Abstract
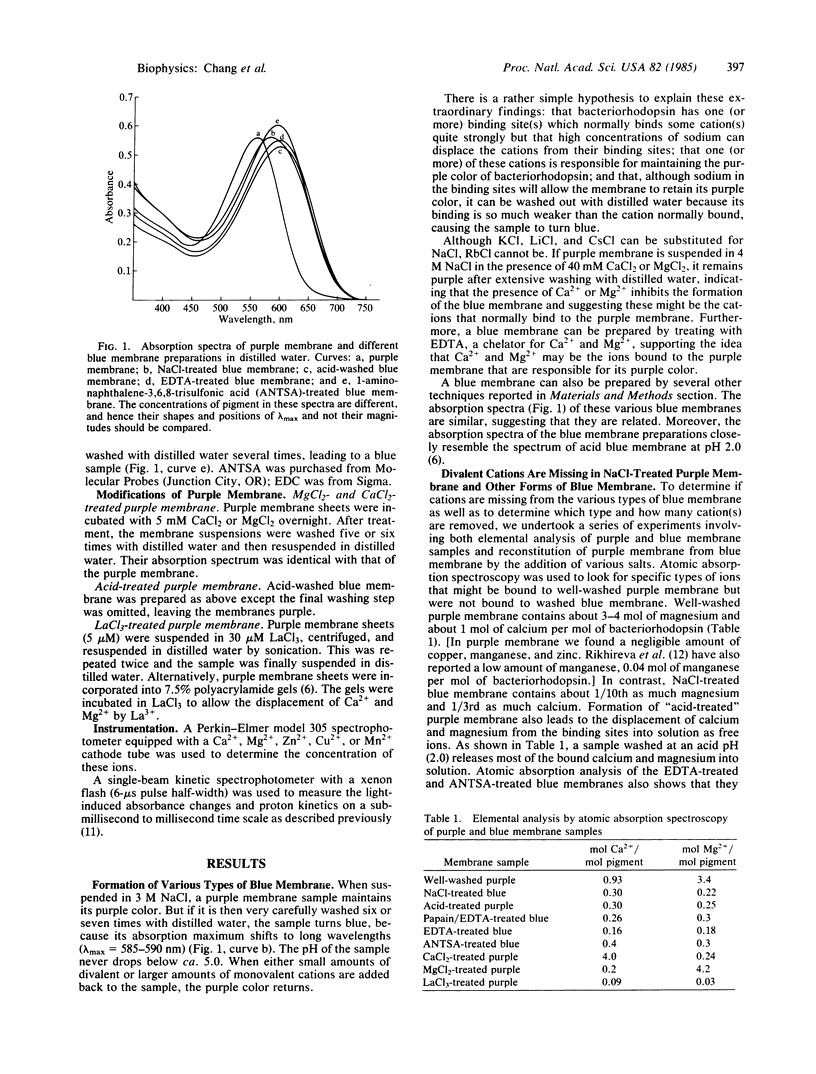
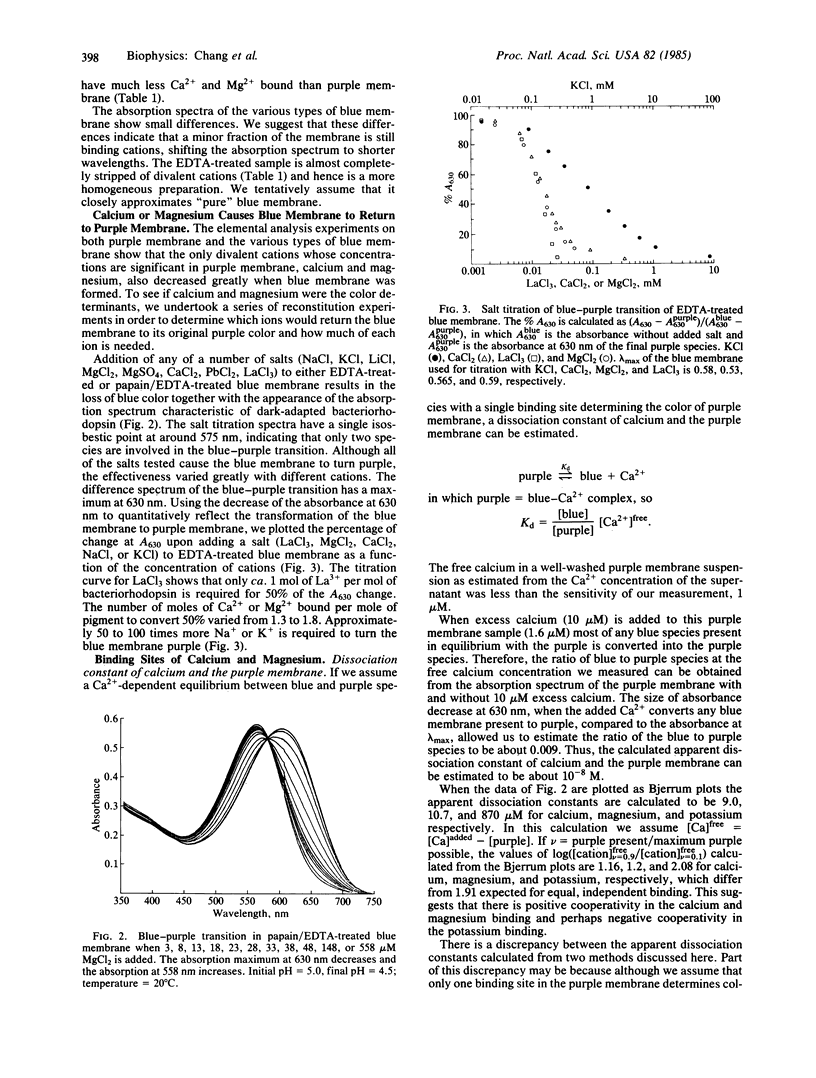
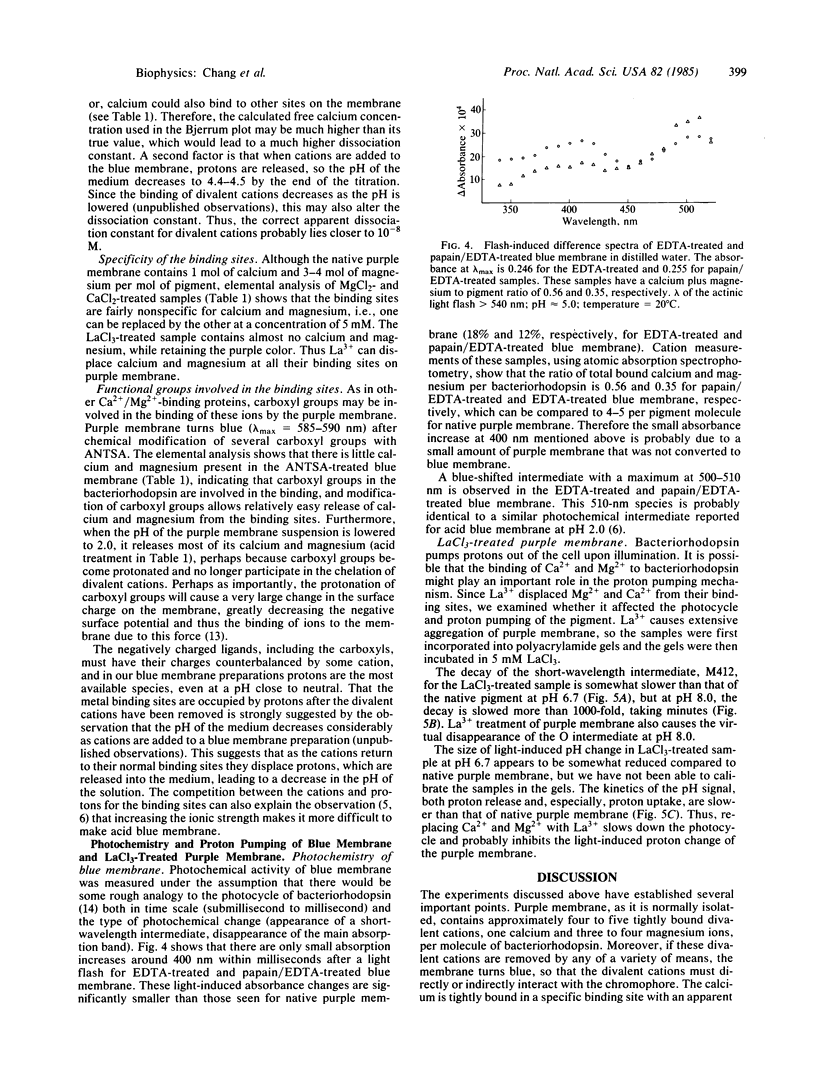
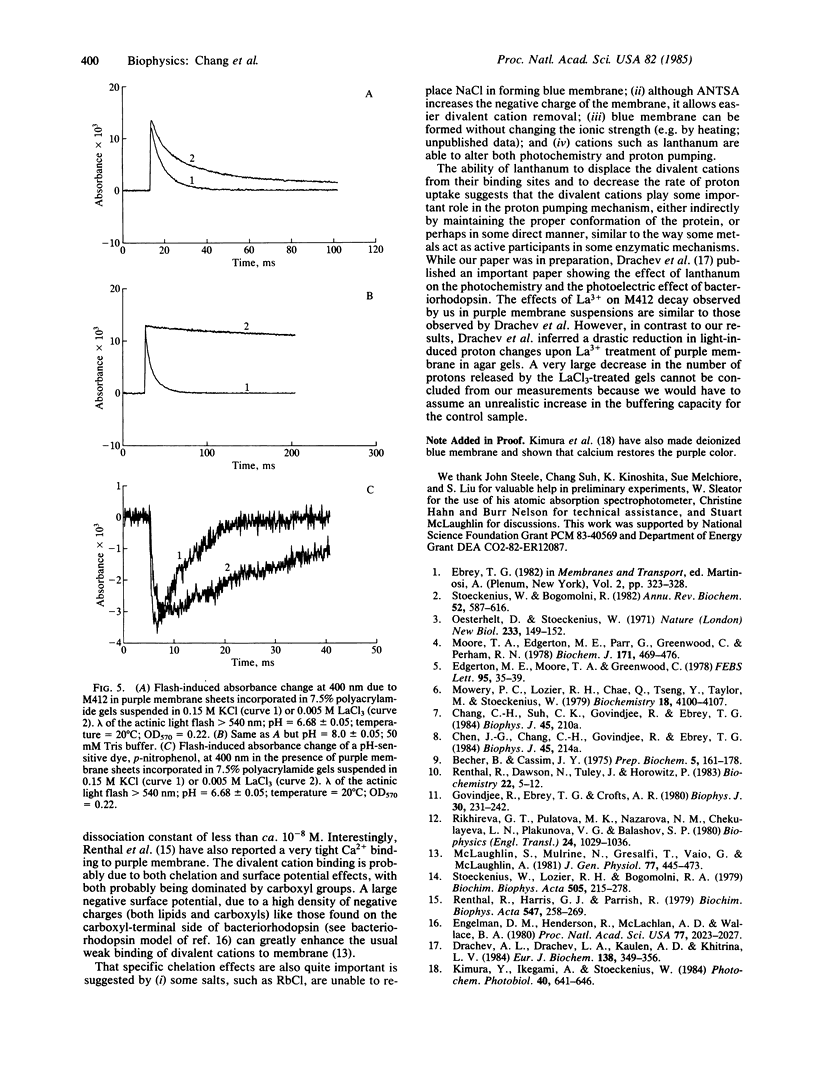
We have found that extensively washed purple membrane has about 1 calcium and 3-4 magnesium ions bound per bacteriorhodopsin molecule. When these divalent cations are removed by any of a variety of means, the pigment changes its color from purple to blue (λmax ≈ 600 nm). This blue pigment, which can be formed at near neutral pH, is probably very similar to blue species formed when the pH of a purple membrane sample is lowered to ≈2. When any of a wide variety of cations are added to a blue membrane preparation, the characteristic purple color of bacteriorhodopsin returns. Divalent and trivalent cations are much more efficient than monovalent cations in restoring the purple color and are effective at a ratio approaching one cation per pigment molecule. Besides shifting the absorption spectrum, removal of the divalent cations drastically alters the photochemical cycle of bacteriorhodopsin, including abolishing the unprotonated Schiff base (M-type) intermediate. Finally, lanthanum not only displaces the divalent cations normally bound to the purple membrane but also greatly reduces both the rate of decay of the M412 intermediate and proton uptake.
Keywords: calcium, magnesium, lanthanum, proton pumping
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Becher B. M., Cassim J. Y. Improved isolation procedures for the purple membrane of Halobacterium halobium. Prep Biochem. 1975;5(2):161–178. doi: 10.1080/00327487508061568. [DOI] [PubMed] [Google Scholar]
- Drachev A. L., Drachev L. A., Kaulen A. D., Khitrina L. V. The action of lanthanum ions and formaldehyde on the proton-pumping function of bacteriorhodopsin. Eur J Biochem. 1984 Jan 16;138(2):349–356. doi: 10.1111/j.1432-1033.1984.tb07922.x. [DOI] [PubMed] [Google Scholar]
- Edgerton M. E., Moore T. A., Greenwood C. Salt reversal of the acid-induced changes in purple membrane from Halobacterium halobium. FEBS Lett. 1978 Nov 1;95(1):35–39. doi: 10.1016/0014-5793(78)80046-5. [DOI] [PubMed] [Google Scholar]
- Engelman D. M., Henderson R., McLachlan A. D., Wallace B. A. Path of the polypeptide in bacteriorhodopsin. Proc Natl Acad Sci U S A. 1980 Apr;77(4):2023–2027. doi: 10.1073/pnas.77.4.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Govindjee R., Ebrey T. G., Crofts A. R. The quantum efficiency of proton pumping by the purple membrane of Halobacterium halobium. Biophys J. 1980 May;30(2):231–242. doi: 10.1016/S0006-3495(80)85091-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura Y., Ikegami A., Stoeckenius W. Salt and pH-dependent changes of the purple membrane absorption spectrum. Photochem Photobiol. 1984 Nov;40(5):641–646. doi: 10.1111/j.1751-1097.1984.tb05353.x. [DOI] [PubMed] [Google Scholar]
- McLaughlin S., Mulrine N., Gresalfi T., Vaio G., McLaughlin A. Adsorption of divalent cations to bilayer membranes containing phosphatidylserine. J Gen Physiol. 1981 Apr;77(4):445–473. doi: 10.1085/jgp.77.4.445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore T. A., Edgerton M. E., Parr G., Greenwood C., Perham R. N. Studies of an acid-induced species of purple membrane from Halobacterium halobium. Biochem J. 1978 May 1;171(2):469–476. doi: 10.1042/bj1710469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mowery P. C., Lozier R. H., Chae Q., Tseng Y. W., Taylor M., Stoeckenius W. Effect of acid pH on the absorption spectra and photoreactions of bacteriorhodopsin. Biochemistry. 1979 Sep 18;18(19):4100–4107. doi: 10.1021/bi00586a007. [DOI] [PubMed] [Google Scholar]
- Oesterhelt D., Stoeckenius W. Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat New Biol. 1971 Sep 29;233(39):149–152. doi: 10.1038/newbio233149a0. [DOI] [PubMed] [Google Scholar]
- Renthal R., Dawson N., Tuley J., Horowitz P. Constraints on the flexibility of bacteriorhodopsin's carboxyl-terminal tail at the purple membrane surface. Biochemistry. 1983 Jan 4;22(1):5–12. doi: 10.1021/bi00270a601. [DOI] [PubMed] [Google Scholar]
- Renthal R., Harris G. J., Parrish R. Reaction of the purple membrane with a carbodiimide. Biochim Biophys Acta. 1979 Aug 14;547(2):258–269. doi: 10.1016/0005-2728(79)90009-4. [DOI] [PubMed] [Google Scholar]
- Stoeckenius W., Bogomolni R. A. Bacteriorhodopsin and related pigments of halobacteria. Annu Rev Biochem. 1982;51:587–616. doi: 10.1146/annurev.bi.51.070182.003103. [DOI] [PubMed] [Google Scholar]
- Stoeckenius W., Lozier R. H., Bogomolni R. A. Bacteriorhodopsin and the purple membrane of halobacteria. Biochim Biophys Acta. 1979 Mar 14;505(3-4):215–278. doi: 10.1016/0304-4173(79)90006-5. [DOI] [PubMed] [Google Scholar]



