ABSTRACT
Keratin 18 (K18 or KRT18) undergoes caspase-mediated cleavage during apoptosis, the significance of which is poorly understood. Here, we mutated the two caspase-cleavage sites (D238E and D397E) in K18 (K18-DE), followed by transgenic overexpression of the resulting mutant. We found that K18-DE mice develop extensive Fas-mediated liver damage compared to wild-type mice overexpressing K18 (K18-WT). Fas-stimulation of K18-WT mice or isolated hepatocytes caused K18 degradation. By contrast, K18-DE livers or hepatocytes maintained intact keratins following Fas-stimulation, but showed hypo-phosphorylation at a major stress-kinase-related keratin 8 (K8) phosphorylation site. Although K18-WT and K18-DE hepatocytes showed similar Fas-mediated caspase activation, K18-DE hepatocytes were more ‘leaky’ after a mild hypoosmotic challenge and were more susceptible to necrosis after Fas-stimulation or severe hypoosmotic stress. K8 hypophosphorylation was not due to the inhibition of kinase binding to the keratin but was due to mutation-induced inaccessibility to the kinase that phosphorylates K8. A stress-modulated keratin phospho-mutant expressed in hepatocytes phenocopied the hepatocyte susceptibility to necrosis but was found to undergo keratin filament reorganization during apoptosis. Therefore, the caspase cleavage of keratins might promote keratin filament reorganization during apoptosis. Interference with keratin caspase cleavage shunts hepatocytes towards necrosis and increases liver injury through the inhibition of keratin phosphorylation. These findings might extend to other intermediate filament proteins that undergo proteolysis during apoptosis.
KEY WORDS: Apoptosis, Intermediate filament, Necrosis
INTRODUCTION
<@?tlb 10.95pt?>Microfilaments, microtubules and intermediate filament (IF) proteins make up the three major cytoskeletal protein families (Ku et al., 1999). IFs include a large group of nuclear proteins and cell-specific cytoplasmic proteins (Fuchs and Weber, 1994; Kim<@?tlb 11pt?> and Coulombe, 2007; Herrmann et al., 2007; Butin-Israeli et al., 2012), with the epithelial-cell-specific keratins (K) making up the largest subgroup of IFs (Coulombe and Omary, 2002). Keratins are classed as type I (K9–K28; K31–K40) and II (K1–K8; K71–K86; Schweizer et al., 2006), and all epithelial cells express at least one type I and one type II keratin that form obligate non-covalent heteropolymers (Coulombe and Omary, 2002). For example, adult hepatocytes express the K8/K18 pair, which protects hepatocytes from a variety of mechanical and non-mechanical stresses (Ku et al., 1995; Marceau et al., 2001; Omary et al., 2009). The importance of K8/K18 in protecting hepatocytes from apoptosis was clearly demonstrated in several keratin-related genetic animal models. For example, hepatocytes or mice with a mutation or deletion of K8 or K18 are significantly more susceptible than their wild-type counterparts to apoptosis mediated by Fas or concanavalin A (Gilbert et al., 2001; Ku et al., 2003: Omary et al., 2009). In addition, the expression of mutant human K18-R90C in mice leads to dramatic hepatocyte fragility upon liver perfusion, keratin filament disruption and an increased susceptibility to drug-induced liver injury (Ku et al., 1995; Ku et al., 1996; Toivola et al., 1998). The importance of K8 and K18 in modulating the susceptibility of the cell to apoptosis is highly dependent on the apoptosis pathway. For example, K8-null hepatocytes that are cultured ex vivo are more susceptible to Fas-mediated apoptosis but not to apoptosis mediated by a combination of tumor necrosis factor (TNF) and cycloheximide as compared with hepatocytes isolated from wild-type (WT) mice (Gilbert et al., 2001). Similarly, mice that overexpress human (h) K18-R90C are more susceptible to apoptosis mediated by Fas but not TNF as compared with mice that overexpress WT hK18 (Ku et al., 2003).
K18 and other type I keratins or IFs, including desmin, vimentin and lamins, undergo caspase-mediated cleavage during apoptosis (Ku et al., 1997; Caulín et al., 1997; Rao et al., 1996; Byun et al., 2001; Ku and Omary, 2001; Chen et al., 2003; Schutte et al., 2004). Caspase cleavage of hK18 occurs initially at the tail-domain Asp397 (D394ALD↓) then at the rod-domain Asp238 (V235EVD↓) (Fig. 1A), leading to keratin reorganization and epithelial-cell apoptosis (Ku et al., 1997; Caulín et al., 1997; Leers et al., 1999; Ku and Omary, 2001; Schutte et al., 2004). Caspase-cleaved keratins accumulate in spheroid inclusions in apoptotic cells (MacFarlane et al., 2000). Only one prior study has examined the role of the caspase-mediated cleavage of IFs during apoptosis. That study showed that the cardiomyocytes of mice overexpressing TNFα could be protected from apoptosis by the expression of a version of desmin that was mutated at the caspase-cleavage site (Panagopoulou et al., 2008). However, the effect of re-introducing wild-type desmin versus a form that was resistant to caspase cleavage was not compared, so one cannot be certain whether the observed protection was due to the mutation of the desmin caspase-digestion site. Moreover, the overall physiological significance of IF cleavage during apoptosis, and its relevance to filament organization and disease-related mutations, is unknown.
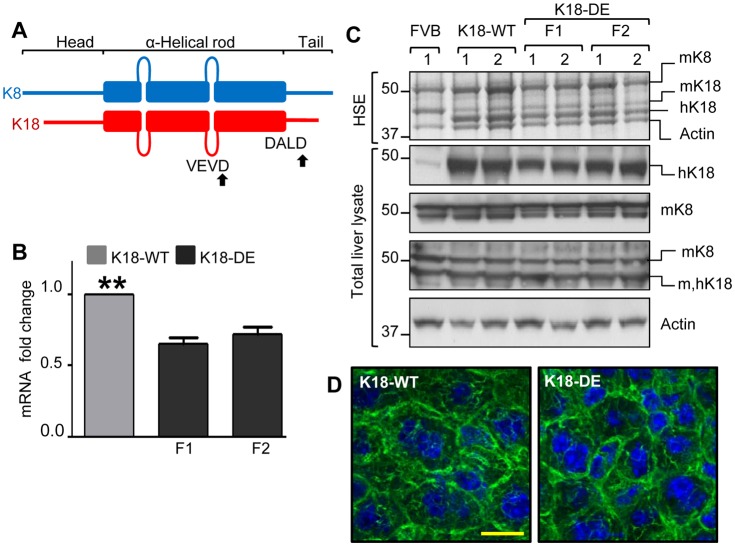
Fig. 1.
Characterization of K18 D238/397E (K18-DE) transgenic mice. (A) A schematic representation of the protein domain organization of K8 and K18. The ‘head’, ‘rod’ and ‘tail’ domains are common features of all IF proteins (Fuchs and Weber 1994; Kim and Coulombe, 2007). The K18 sites of caspase-mediated aspartate cleavage [which are conserved in both human (h) and mouse (m) sequences] are indicated by arrows. (B) hK18-WT and K18-DE mRNA levels in mouse liver. Two transgenic mouse founder lines (F1 and F2) that express K18-DE were generated and were compared with K18-WT mice for their relative liver mRNA levels. Data show the mean±s.e.m. (three independent livers). **P<0.01. (C) The expression of K18-WT and K18-DE protein in mouse liver. Liver tissue from FVB non-transgenic mice was used as a control. High-salt extraction (HSE) was used to generate an enriched keratin-containing fraction (Ku et al., 2004) that was analyzed by SDS-PAGE followed by Coomassie staining. Because of the maintenance of a 1∶1 ratio of K8 and K18 proteins, the decrease in mouse K18 is compensated for by the overexpressed human (h)K18 (which gives a similar profile to the heterozygous expression of a mutant protein in the context of a human disease). In parallel, total liver homogenates were analyzed by blotting using antibodies against the indicated proteins. The actin blot is included as a loading control. Each lane represents a separate liver. (D) The keratin filament organization of K18-WT and K18-DE in mouse liver. Liver sections were immunostained with antibody against hK18 (green) and counterstained with DAPI to show the nuclei (blue). Scale bar: 20 µm.
Here, we tested the physiological significance of caspase-mediated K18 digestion during hepatocyte cell death, in vivo and ex vivo, using transgenic mice that overexpress K18-WT or K18 that is mutated at Asp238 and Asp397 (to render the keratin resistant to caspase digestion). We show that inhibition of K18 caspase digestion, by K18 mutation, promotes hepatocyte ‘leakiness’ and necrosis, and interferes with the reorganization of keratin filaments, in a context-dependent manner, in association with K8 hypophosphorylation.
RESULTS
Generation and characterization of K18 D238/396E mice
To investigate the significance of keratin caspase cleavage during apoptosis, transgenic mice that overexpress human hK18 D238/397E (K18-DE) (Fig. 1A) were generated and characterized. Total RNA was isolated from two founder lines (F1 and F2) of K18-DE mice and was compared with that isolated from K18-WT mice. Livers from the two K18-DE founders expressed similar levels of hK18 mRNA, but expressed less hK18 mRNA than K18-WT livers (Fig. 1B). Consistent with the mRNA levels, both K18-DE founders expressed less human K18-DE protein in their livers than the K18-WT mice did, but the overall keratin levels were similar in WT and K18-DE mice (Fig. 1C). Importantly, keratin filament organization of K18-WT and K18-DE was similar (Fig. 1D), thereby indicating that the K18-D238/397E double mutation did not interfere with normal keratin filament organization in hepatocytes under basal conditions. The K18-DE mice were healthy, with normal epithelial tissue histology and had similar weights to their K18-WT counterparts (data not shown).
K18-DE mice are more susceptible to Fas-induced apoptotic liver injury in association with K8 hypophosphorylation
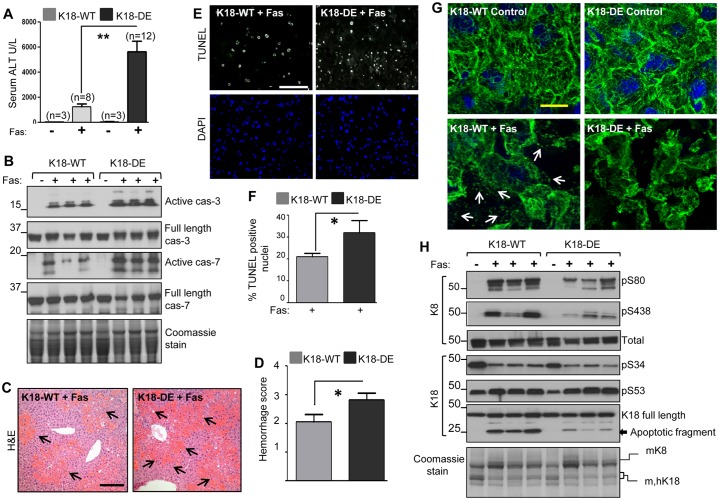
We investigated the significance of keratin cleavage by caspases during apoptosis. K18-WT and K18-DE mice were challenged with the monoclonal anti-Fas antibody Jo2 to induce apoptosis, followed by biochemical and histological analysis. Both K18-WT and K18-DE control mice had normal serum alanine aminotransferase (ALT) levels, whereas, after Fas activation, K18-DE mice had significantly higher ALT compared to K18-WT mice (Fig. 2A). Livers from the K18-DE mice that were given Jo2 showed increased caspase 3 and 7 activation (Fig. 2B), hemorrhage (Fig. 2C,D) and more TUNEL-positive nuclei (Fig. 2E,F) as compared to K18-WT livers. The Jo2-induced apoptosis resulted in the reorganization of keratin filaments into dots in K18-WT livers (Fig. 2G, arrows), whereas the K18-DE livers had a relatively intact keratin network (Fig. 2G). Because keratin filament reorganization during hepatocyte apoptosis occurs in association with keratin hyperphosphorylation (Ku and Omary, 2001; Schutte et al., 2004), we assessed keratin phosphorylation during Jo2-induced liver injury. Contrary to what might have been predicted, K18-DE livers had decreased K8 phosphorylation at serine residues 80 and 438 (pS80/pS438) as compared to K18-WT livers; however, K18 phosphorylation (pS34/pS53) was similar in both K18-WT and K18-DE mice (Fig. 2H). As expected, despite the increased caspase activation and liver damage in K18-DE mice (Fig. 2B–F), the K18-DE protein could not be cleaved, and only a limited amount of the K18 apoptotic fragment was detected (Fig. 2H) because of the presence of residual WT murine K18 in the K18-DE mice. These results indicate that caspase-cleavage-resistant K18-DE mice are more susceptible to Fas-induced apoptotic liver injury in association with K8 hypophosphorylation than are K18-WT mice. The increased in vivo susceptibility of K18-DE mice to Fas-mediated injury, as compared with K18-WT mice, was not observed when exposing mice to TNFα (supplementary material Fig. S1). This result is in line with prior findings showing that K8-null hepatocytes or mice that overexpress K18-R90C are more susceptible to apoptosis mediated by Fas, but not TNF, compared to their WT counterparts (Gilbert et al., 2001; Ku et al., 2003).
Fig. 2.
K18-DE mice are more susceptible to Fas-mediated liver injury. (A) Serological analysis of the serum ALT levels in K18-WT and mutant mice with or without Jo2 (Fas) treatment (n, number of animals). Mice were killed 4 h after the administration of Jo2, followed by serum analysis (for Jo2-treated animals, nearly one-half were from the F1 line and the other half were from the F2 line). Data show the mean±s.e.m. **P<0.01. (B) Fas-induced caspase activation in the liver. Total-liver homogenates were prepared from the livers of mice with or without Fas stimulation, followed by immunoblot analysis of equal fractions using antibodies against full-length and active caspases 3 or 7. The Coomassie stain of the lysates is included to show equal protein loading. Each lane represents an individual mouse liver. (C) Fas-induced liver damage was assessed by hematoxylin and eosin (H&E) staining. The arrows indicate areas of hemorrhage. Scale bar: 100 µm. (D) A quantification of Fas-induced hemorrhage in K18-WT and K18-DE mice. H&E sections from K18-WT (15 mice) and K18-DE (22 mice) were scored using multiple images. Data show the mean±s.e.m. *P<0.05. (E) Fas-induced hepatocyte apoptosis was assessed by TUNEL and DAPI staining. Scale bar: 40 µm. (F) The percentage of TUNEL-positive nuclei of K18-WT and K18-DE mice after Jo2 treatment. Three to five fields from three animals per condition were used for quantification. Data show the mean±s.e.m. *P<0.05. (G) Fas-induced keratin filament reorganization in K18-WT and K18-DE mouse livers. Liver sections were stained with hK18-specific antibody (green) and DAPI (blue). The arrows indicate the disassembled keratin dot-like fragments that are seen in the K18-WT but not K18-DE livers. Scale bar: 20 µm. (H) Immunoblot analysis of keratin phosphorylation upon Fas-induced liver injury in K18-WT and K1-DE mice. Livers were processed to obtain high-salt extracts, followed by immunoblotting with antibodies specific to phospho-K8 (pS80, pS438), phospho-hK18 (pS34, pS53) and total K8 and K18. Also included is the Coomassie stain of the extracts showing similar loading. Each lane represents samples from an individual mouse liver and the experiment was repeated at least three times.
K18-DE hepatocytes undergo cell death by necrosis
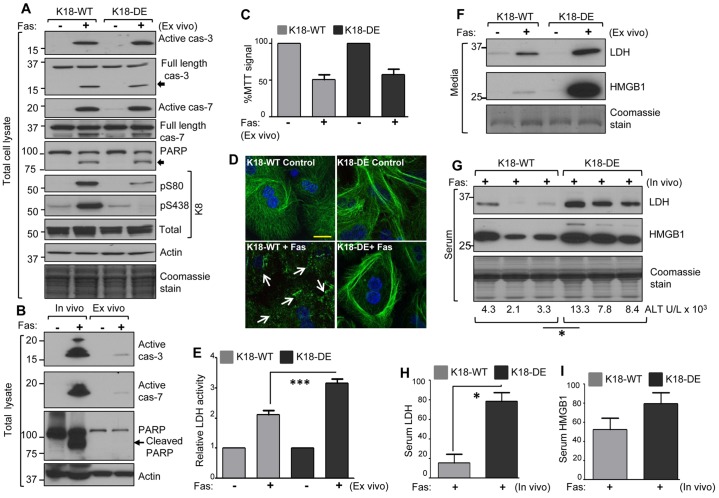
To explore the mechanism by which K18-DE mice are more susceptible to Fas-mediated apoptotic liver injury, primary hepatocytes from K18-WT and K18-DE mice were challenged with Jo2, followed by analysis of the hepatocyte damage. In contrast to the in vivo situation, ex vivo K18-WT and K18-DE hepatocytes showed similar caspase 3 and 7 activation and poly (ADP-ribose) polymerase (PARP) cleavage (Fig. 3A). However, similar to the in vivo findings, K18-DE hepatocytes showed decreased K8 S80/S438 phosphorylation following the induction of apoptosis as compared with K18-WT hepatocytes (Fig. 3A). Of note, the ex vivo culture of hepatocytes rendered them relatively resistant to Fas-mediated apoptosis, compared with in vivo Fas stimulation (as shown by the differences in caspase activation and PARP cleavage between the in vivo and ex vivo cells, Fig. 3B). Consistent with the similar caspase activation in the K18-WT and K18-DE hepatocytes ex vivo, treatment with Jo2 resulted in similar mitochondrial damage in K18-WT and K18-DE primary hepatocytes as determined by the MTT assay (Fig. 3C). Notably, K18-WT hepatocytes displayed marked reorganization of keratin filament into dots (Fig. 3D, arrows), whereas K18-DE hepatocytes had a relatively intact keratin network (Fig. 3D).
Fig. 3.
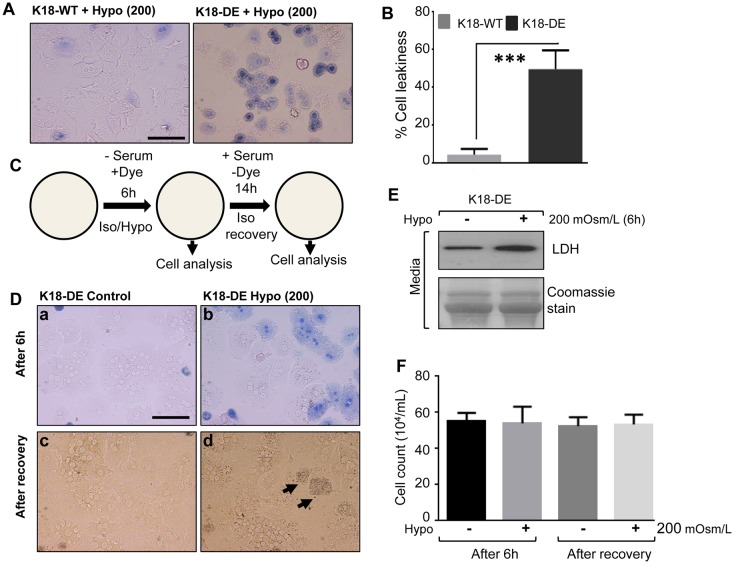
K18-DE hepatocytes are more susceptible to necrosis after Fas stimulation. (A) Fas-induced caspase activation and keratin phosphorylation in mouse primary hepatocytes. Hepatocytes were isolated from K18-WT and K18-DE mice. After overnight equilibration, cultured hepatocytes were treated with Jo2 for 6 h, followed by preparation of a total-cell lysate and immunoblotting with antibodies against caspases 3 and 7, PARP, K8 and phospho-K8. The arrows indicate the cleaved PARP and caspase products. An actin blot and a Coomassie stain of the analyzed gels are included as loading controls. (B) Western blot analysis to compare caspase activation and PARP cleavage in vivo and ex vivo. Total-cell lysates were obtained from the livers of K18-WT mice (in vivo; injected with Jo2, 0.1 µg/g, 4 h before killing) or from isolated hepatocytes (ex vivo; cultured with Jo2, 0.5 µg/ml for 6 h). Equal fractions of the lysates were compared for caspase activation and PARP cleavage. An actin blot is included as a loading control. Note that ex vivo caspase activation and PARP cleavage, under the conditions used, are markedly lower as compared to the in vivo situation. (C) Fas-induced mitochondrial damage in hepatocytes that were isolated from K18-WT and K18-DE mice was assessed using an MTT assay. Data show the mean±s.e.m. (D) Fas-induced reorganization of keratin filaments. Primary hepatocytes similar to those used in A were stained with anti-hK18 antibody (green) and counterstained with DAPI (blue). The arrows highlight the keratin dot-like fragments in K18-WT hepatocytes (lower-left panel) that are not seen in K18-DE hepatocytes (lower-right panel). Scale bar: 20 µm. (E) The relative LDH activity in culture medium obtained from K18-WT and K18-DE primary hepatocytes (with or without Fas). Data show the mean±s.e.m. ***P<0.001. (F) Immunoblot analysis of the necrosis markers LDH and HMGB1. Culture media from the cells used in A were concentrated to an equal volume, and were then analyzed by SDS-PAGE and Coomassie staining (which shows the secreted albumin band) or by blotting using antibodies against LDH and HMGB1. The results are representative of three independent experiments. (G) Fas-induced LDH and HMGB1 release in vivo. Sera were collected from K18-WT and K18-DE mice that were pre-treated with Jo2 for 4 h, and were analyzed by blotting as for F. The Coomassie stain is included as a loading control, and the serum ALT from each animal is indicated. *P<0.05. (H,I) Densitometric quantification of serum LDH and HMGB1 (from the samples shown in G). Data show the mean±s.e.m. *P<0.05
The observed difference in the susceptibility of K18-DE hepatocytes to Fas-induced apoptosis in vivo versus ex vivo led us to hypothesize that ex vivo K18-DE hepatocytes might undergo cell death through non-apoptotic processes such as necrosis. To test this idea, we analyzed hepatocyte culture medium for the release of the necrosis markers lactate dehydrogenase (LDH) and high mobility group box 1 protein (HMGB1) (Štros, 2010). The culture medium of K18-DE hepatocytes had higher LDH activity (Fig. 3E), and more LDH and HMGB1 protein (Fig. 3F), as compared with K18-WT hepatocytes. However, there was no significant difference in the number of dead cells (based on Trypan Blue uptake) between K18-WT and K18-DE primary hepatocytes (supplementary material Fig. S2). Similarly, the release of LDH and HMGB1 into the serum following Fas-mediated apoptotic liver injury was significantly higher in K18-DE as compared to K18-WT mice (Fig. 3G–I). These results suggest that the inhibition of caspase digestion of K18 promotes hepatocyte leakiness and necrosis during Fas-induced apoptotic liver injury.
The mutation of K18 caspase digestion sites promotes hypoosmotic-stress-induced hepatocyte leakiness and cell death independently of caspase activation
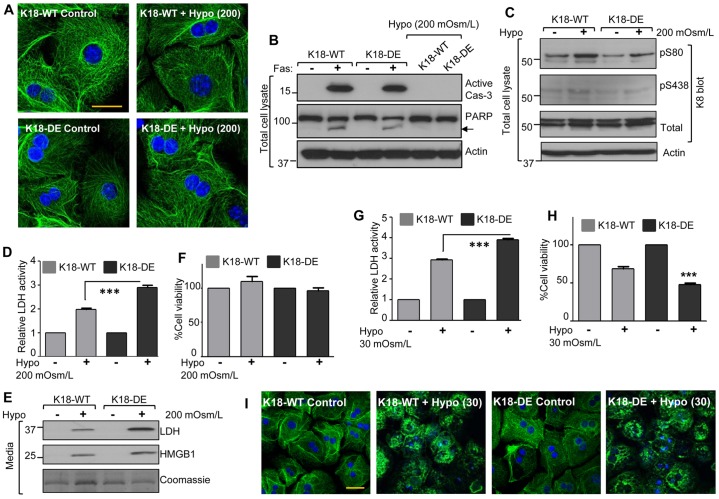
Given the established function of K8/K18 in protecting hepatocytes from mechanical and nonmechanical forms of injury (Ku et al., 1995; Loranger et al., 1997; Ku and Omary, 2006), we compared the responses of K18-WT and K18-DE primary hepatocytes that were challenged with mild hypoosmotic stress (200 mOsm/l). Hypoosmotic stress can be considered to be a form of mechanical stress because it induces cell swelling. Keratin immunostaining showed no evident filament reorganization in K18-WT or K18-DE hepatocytes (Fig. 4A); and there was no detectable caspase activation or PARP cleavage, in contrast to the hepatocyte response to Fas stimulation (Fig. 4B). However, similar to the response to Jo2 treatment, K18-DE hepatocytes showed hypophosphorylation of K8 S80 (and to a lesser extent S438) (Fig. 4C), thereby indicating that the reduced K8 phosphorylation in response to stress in K18-DE livers is independent of caspase activation and keratin filament reorganization.
Fig. 4.
K18-DE hepatocytes are more susceptible than K18-WT cells to necrosis and cell leakiness after exposure to hypoosmotic stress, and this is independent of caspase activation. (A) Mild hypoosmotic stress does not alter keratin filament organization. Isolated primary hepatocytes from K18-WT and K18-DE mice were exposed to mild hypoosmotic conditions (200 mOsm/l for 6 h) and were then stained with anti-K18 antibody (green) and DAPI (blue) to assess filament reorganization. Scale bar: 20 µm. (B,C) The effect of mild hypoosmotic stress on caspase activation and keratin phosphorylation. Lysates from the cells in panel A and from cells treated with Jo2 (0.5 µg/ml for 6 h) were analyzed by blotting using antibodies against PARP, active caspase-3, K8 and phospho-K8. Note that there is no PARP cleavage (indicated by the arrow) or caspase activation during injury induced by hypoosmotic stress. The relative phosphorylation ratio of K8 S80, estimated by densitometry scanning after correction for total K8, in K18-WT and K18-DE hepatocytes after hypoosmotic stress is 2.25∶1, respectively (lane two versus lane four; representative of three independent experiments). The actin blot shows equal protein loading. (D,E) Mild hypoosmotic stress induced LDH and HMGB1 release. The culture media from cells used in A were collected and concentrated, and then analyzed for LDH activity or by blotting with antibodies against LDH and HMGB1. Data show the mean±s.e.m. ***P<0.001. (F) The effect of mild hypoosmotic stress on cell death. The percentage of viable cells after primary hepatocytes from K18-WT and K18-DE mice were challenged with hypoosmolar (200 mOsm/l) conditions is shown. Data show the mean±s.e.m. (G) Severe hypoosmotic stress induced LDH release. Hepatocytes from K18-WT and K18-DE mice were exposed to severe hypoosmotic conditions (30 mOsm/l for 6 h) and the activity of the LDH that was released into the medium was measured. Data show the mean±s.e.m. (H) Severe hypoosmotic stress induced cell death, as shown by calculation of the percentage cell viability. Data show the mean±s.e.m. ***P<0.001. (I) Severe hypoosmotic stress induced keratin filament reorganization. Hepatocytes, as in A, were exposed to hypoosmotic conditions (30 mOsm/l for 6 h), followed by staining using antibodies against K18 (green) and DAPI (blue). Scale bar: 50 µm.
We then analyzed the release of LDH and HMGB1 to cell culture medium during mild hypoosmotic stress conditions. In response to hypoosmotic stress, the medium from K18-DE hepatocytes had higher LDH activity compared with that from K18-WT hepatocytes, and the K18-DE cells released more LDH and HMGB1 protein into the culture medium (Fig. 4D,E). This release likely reflects an increase in cell leakiness (i.e., a decrease in plasma membrane integrity) because there was no significant change in cell count (Fig. 4F). However, in comparison to K18-WT cells, exposure of K18-DE cells to severe hypoosmotic conditions (30 mOsm/l) resulted in higher LDH activity in the medium (Fig. 4G) and more prominent cell death (Fig. 4H) that was independent of caspase activation (supplementary material Fig. S3A). Both K18-WT and K18-DE hepatocytes underwent similar keratin filament reorganization in response to severe hypoosmotic stress (Fig. 4I). Therefore, K18-DE hepatocytes can undergo context-dependent keratin filament reorganization similar to K18-WT cells, independent of caspase-mediated keratin filament collapse.
K18-DE hepatocyte leakiness in response to mild hypoosmotic conditions is reversible
We next examined the effect of the K18 mutation (K18-DE) on hepatocyte leakiness by Trypan Blue uptake, and asked whether cell leakiness is reversible. Hepatocytes were isolated from K18-WT and K18-DE mice and were allowed to attach overnight, then challenged with mild hypoosmotic stress (200 mOsm/l) in the presence of Trypan Blue in serum-free medium. After 6 h, K18-DE hepatocytes showed more dye uptake compared to K18-WT cells (Fig. 5A,B), which is consistent with loss of membrane integrity and release of LDH under the same conditions (Fig. 4D,E). In order to test whether hepatocyte leakiness promotes cell death or is potentially reversible, we performed the experiment outlined in Fig. 5C. K18-DE hepatocytes were exposed to hypoosmotic stress for 6 h, followed by the culture of duplicate samples in isotonic medium for a 14-h chase period. We then assessed the effects of the chase period on the cellular retention of Trypan Blue and on cell viability. As anticipated, K18-DE hepatocytes that were challenged with mild hypoosmotic stress showed Trypan Blue uptake (Fig. 5D, panels a and b), accompanied by an increased release of LDH (Fig. 5E). Notably, after the 14-h chase, the leaky cells recovered and only a few cells retained the blue dye (Fig. 5D, panels c and d), without any evidence of cell death (Fig. 5F). Taken together, these results show that the mutation of the two K18 caspase digestion sites promotes reversible hepatocyte leakiness under mild hypoosmotic conditions, or cell death under severe hypoosmotic conditions.
Fig. 5.
K18-DE hepatocytes are leakier than K18-WT but leakiness can be rescued under mild hypoosmotic conditions. Hepatocytes were isolated from K18-WT and K18-DE mice. After overnight attachment, the cells were challenged with mild hypoosmotic stress (200 mOsm/l) under serum-free conditions for 6 h in the presence of Trypan Blue (1%). (A) Brightfield images showing the dye uptake of K18-WT and K18-DE hepatocytes. Scale bar: 50 µm. (B) Quantification of cell leakiness as assessed by the percentage of stained cells. ***P<0.001. (C) A schematic showing the design of the recovery experiment. Iso, isotonic; Hypo, hypotonic. (D) K18-DE hepatocytes recover after mild hypoosmotic stress. The hepatocytes were isolated from K18-DE mice. After overnight attachment, the cells were challenged with isotonic (300 mOsm/l) or mild hypoosmotic stress (200 mOsm/l) under serum-free conditions for 6 h in the presence of Trypan Blue (1%). The upper-left and -right brightfield images (a and b) show the dye uptake after 6 h. After 6 h, the medium was replaced with regular culture medium to allow hepatocyte recovery. The lower-left and -right brightfield images (c and d) show the recovery. Arrows indicate the cells that retained the blue dye. Scale bar: 50 µm. (E) Mild hypoosmotic conditions induced LDH release. Culture media from the cells used for the experiment outlined in D were collected after 6 h (D, panels a and b) and concentrated, and then were analyzed for LDH by immunoblotting. Coomassie stain is used as a loading control. (F) Quantification of the cell death induced by a 6-h exposure to mild hypoosmotic stress.
K18 D238E mutation renders K8-S80 less susceptible to phosphorylation by reducing the access of kinase
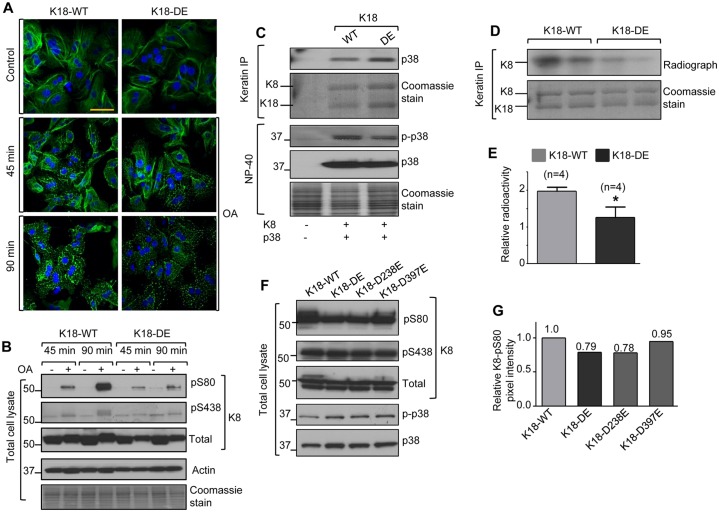
The fact that the K18-DE double mutation led to the reduced hyperphosphorylation of K8 after Fas stimulation or hypoosmotic stress raised the possibility that it has a trans-inhibitory effect on K8 phosphorylation, potentially through a conformational alteration that directly or indirectly modulates the access of a phosphatase or kinase to K8. In order to distinguish between these two possibilities, we first examined a potential phosphatase role by taking advantage of the known effect of phosphatase inhibition by okadaic acid, which results in dramatic keratin filament disassembly and K8/K18 hyperphosphorylation due to the inhibition of protein phosphatase-1 and -2A (Ku et al., 2004; Gehringer, 2004). This approach also allowed us to address whether the inability of K18-DE-containing filaments to reorganize during apoptosis is related to the limited K8 hyperphosphorylation response. Treatment of K18-WT and K18-DE hepatocytes with okadaic acid resulted in similar patterns of filament reorganization (Fig. 6A). However, K18-DE hepatocytes still showed marked K8 hypophosphorylation (particularly at S80; Fig. 6B, compare lanes 4 and 8) relative to K18-WT hepatocytes, whereas K18 phosphorylation at S34 was similar between the two hepatocyte lines, and thus was unaffected by the K18-DE mutation (supplementary material Fig. S3B). Therefore, the effects of K18 mutation are unlikely to be due to known keratin phosphatases per se. This also provides further evidence that the ability of K18-DE mutation to interfere with K8/K18 filament reorganization is context-dependent and occurs preferentially in the setting of apoptosis.
Fig. 6.
K18-DE hepatocytes undergo keratin filament reorganization independently of keratin hypophosphorylation. (A,B) Okadaic acid induced keratin filament reorganization and phosphorylation. Hepatocytes from K18-WT and K18-DE mice were cultured with or without okadaic acid (OA, 1 µM) for the indicated times, followed by staining using an antibody against K18 (green) and DAPI (nuclei). Scale bar: 50 µm. Total-cell lysates were then prepared, followed by blotting using antibodies against the indicated proteins. The relative phosphorylation ratio of K8 S80, estimated by densitometry scanning after correction for total K8, in K18-WT and K18-DE hepatocytes after okadaic acid treatment is 2.4∶1, respectively (B, lane four versus lane eight). The results are representative of two independent experiments. (C) p38 kinase and keratin association. BHK cells were co-transfected with K8 and p38 kinase, together with K18-WT or K18-DE. After 24 h, keratins were immunoprecipitated, followed by blotting of the precipitates and input lysates with antibodies against the indicated proteins. (D,E) In vitro keratin phosphorylation. Keratins were immunoprecipitated from K18-WT and K18-DE mouse livers, followed by in vitro phosphorylation of the precipitates using p38 kinase in the presence of [γ-32P]ATP. The Coomassie stain of the keratin immunoprecipitate shows the input protein. The autoradiograph shows the in vitro K8 phosphorylation, which was quantified and is shown in E. n, the number of livers used for individual immunoprecipitates. *P<0.05. (F,G) The effect of individual mutation of the K18 caspase-cleavage sites on K8 phosphorylation. CHO cells were co-transfected with K8 and p38 together with one of four K18 constructs (K18-WT, K18-DE, K18-D238E or K18-D397E). After 24 h, total-cell lysates were prepared and blotted with antibodies against K8, K8-pS80, p38 and phospho-p38. The relative K8-pS80 pixel intensity to that of K8 for each transfection was measured and normalized to K18-WT (set to a value of 1.0). Similar results were reproduced using the BHK-transfection system.
Previous reports have shown that K8/K18 associates with p38 and Jun kinases, which in turn phosphorylate K8-S80 in the context of apoptosis and cell stress (Ku et al., 2002; He et al., 2002). We tested the effect of the K18-DE double mutation on the association of p38 kinase with K8/K18 using a baby hamster kidney (BHK) cell transfection system. Similar levels of p38 were associated with both K18-WT and K18-DE (Fig. 6C), thereby indicating that the K18-DE double mutation does not interfere with keratin binding to p38. We then tested whether K18-DE renders its binding partner, K8, a less favorable substrate for phosphorylation by p38 kinase. As shown in Fig. 6D,E, K18-DE mutation resulted in a significant decrease in K8 phosphorylation when K8/K18 immunoprecipitates (containing either WT or mutant K18) were used in an in vitro phosphorylation assay with recombinant p38 kinase. Analysis of the individual or double K18 mutants (D238E, D397E and D238E/D397E) showed that it was the K18 D238E mutation that preferentially rendered K8 S80 less susceptible to phosphorylation (Fig. 6F,G) in a trans fashion, as a result of kinase inaccessibility.
The K8-S74A mutant biases hepatocytes towards necrosis
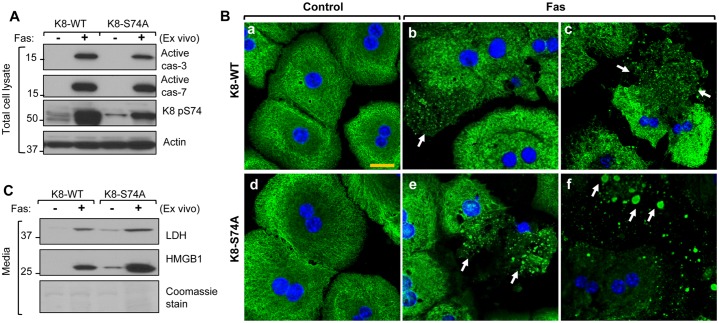
The observed susceptibility of K18-DE cells to necrosis, as compared with K18-WT hepatocytes, might be due to resistance of the mutant protein to caspase-mediated filament reorganization or the reduced K8 phosphorylation or both. To distinguish between these possibilities, primary hepatocytes from mice that overexpress hK8-WT or hK8-S74A [equivalent to K8-S80 in mouse (Ku and Omary, 2006)] were challenged with Fas. Both K8-WT and K8-S74A hepatocytes showed similar caspase activation and, as expected, K8-S74A hepatocytes showed reduced S74A phosphorylation (Fig. 7A, the residual phospho-antibody reactivity in K8-S74A hepatocytes is derived from endogenous WT K8). Unlike K18-DE hepatocytes, K8-S74A hepatocytes undergo caspase-mediated filament reorganization in a similar manner to K8-WT (Fig. 7B), thereby indicating that human K8 S74 (and, by extrapolation, mouse K8 S80) phosphorylation is not directly involved with caspase-mediated filament collapse. However, K8-S74A hepatocytes are more susceptible to necrosis as compared with their WT counterparts, as determined by LDH and HMGB1 release and keratin immunostaining after 6 h of Fas treatment (Fig. 7B,C). This indicates that the inhibition of K18 caspase cleavage interferes with keratin filament reorganization independently of K8 S74 phosphorylation but predisposes the cell to necrosis in association with the inability of K8 to mount a hyperphosphorylation response.
Fig. 7.
K8-S74A hepatocytes undergo keratin filament reorganization but are more susceptible to necrosis during Fas-induced apoptosis. (A) Hepatocytes were isolated from human (h) K8-WT and hK8-S74A livers, cultured overnight and then treated with Fas for 6 h. Total-cell lysates were then blotted using antibodies specific to active caspases 3 and 7 and phospho-K8. An actin blot is included as a loading control. (B) Fas-induced reorganization of keratin filaments. Primary hepatocytes similar to those used in A were stained with anti-keratin antibody (green) and counterstained using DAPI (blue). hK8-S74A hepatocytes undergo caspase-mediated filament collapse similar to K8-WT hepatocytes (arrows). In addition, hK8-S74A hepatocytes are more susceptible to necrotic cell death as shown by the more disrupted keratin filaments (compare panels c and f). Scale bar: 20 µm. (C) Culture media from the cells used in A were concentrated to an equal volume followed by analysis by SDS-PAGE and Coomassie staining (which shows the secreted albumin) or by blotting using antibodies against LDH and HMGB1.
DISCUSSION
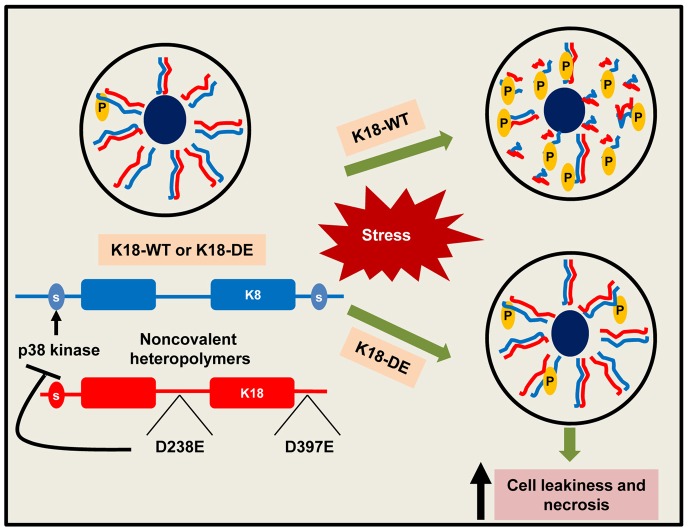
Our findings provide a novel potential mechanism for the pathogenesis of some IF diseases. Some disease-related IF mutations that render the IF protein resistant to caspase cleavage reside within the linker L1–2 caspase box (supplementary material Fig. S4). In the context of such mutations, our findings indicate that cell injury promotes necrosis or promotes cell leakiness in situations, such as mild hypoosmotic stress, that do not cause cell death per se (Figs 4,5). Although the mouse model that we generated has two K18 mutations (within the caspase box and the K18 tail domain), we attribute the observed effect primarily to the caspase-box linker-region mutation because it was the linker mutation (K18-D238E) or the double mutation (K18-DE) but not the tail-domain mutation (K18-D397E) that inhibited K8 S80 phosphorylation (Fig. 6F,G). These findings might extend to IF proteins that harbor the linker-region caspase box (with an XEXD or XDXD motif; where X is a non-charged amino acid) (Ku and Omary, 2001; Marceau et al., 2007), with the exception of type II keratins, which are not caspase substrates (Ku et al., 1997). The necrosis phenotype that we observed occurred either in the context of what otherwise is considered primarily apoptotic cell death (upon Fas stimulation) or caspase-independent cell death (upon severe hypoosmotic stimulation). This provides a novel potential mode of cell injury in IF-related diseases; injury due to mutations that interfere with caspase cleavage (supplementary material Fig. S4), possibly by increasing the rigidity of the cytoskeleton and causing it to be unable to undergo stress-induced phosphorylation (or potentially other post-translational changes) and reorganization. By contrast, other rod-domain mutations such as K18-R90C [or its equivalent K14-R125C that causes severe epidermolysis bullosa simplex (Coulombe et al., 2009)] directly disrupt keratin filaments in association with increased keratin phosphorylation at alternative sites such as K18 S53 (Ku et al., 1995; Ku et al., 2003).
Our findings also provide the first direct evidence that the caspase cleavage of K18 (which might translate to other IFs) is necessary for keratin filament reorganization during apoptosis ex vivo and in vivo. However, the K18-DE mutations that block K18 digestion by caspases do not interfere with filament reorganization in other contexts such as hypoosmotic stress or okadaic-acid-induced hyperphosphorylation. Interestingly, K18-DE mutation reduced the ability of K8 to undergo stress-induced hyperphosphorylation in a unique trans manner (i.e. by affecting the partner keratin), yet had no detectable effect on K18 phosphorylation at S34. This is likely to reflect a mutation-induced conformational alteration of the keratin heterodimer that renders K8 S80 a poor substrate for its kinase(s) (Fig. 8). As such, p38 kinase was still able to bind to K8/K18-DE just as well as to K8/K18-WT, but it could not phosphorylate its target efficiently. Of note, the phospho-mutant of the human equivalent of mouse K8 S80 (K8-S74A) had no inhibitory effect on caspase-mediated filament reorganization (Fig. 7B). This indicates that the observed resistance of K18-DE to caspase-mediated filament reorganization is not due to K8 hypophosphorylation at the conserved human/mouse S74/S80 sites per se (although we cannot exclude the importance of other keratin phosphorylation sites). With regard to the difference in caspase activation in vivo versus ex vivo in the context of K18-WT and K18-DE hepatocytes or livers (whereby the K18-DE double mutation in vivo, but not ex vivo, is associated with increased caspase activation versus K18-WT), we envision a sequence of in vivo events that are precipitated by increased necrosis. We suggest the establishment of a feedback loop in which the increased necrosis promotes further apoptosis in the context of the whole organ. The potential contribution (if any) of circulating HMGB1, LDH or other released proteins to this process is not known.
Fig. 8.
A model of the consequences of K18-DE mutation during simple epithelial-cell injury. For WT-K18, stress conditions that are associated with apoptotic or non-apoptotic injury result in keratin filament disassembly (depending on the nature of the stress) and keratin hyperphosphorylation at murine K8 S80 (or at S74 of human K8), which serves as a stress sensor (Ku et al., 2002; He et al., 2002; Snider and Omary, 2014). By contrast, the K18-DE mutation renders the keratin filaments unable to reorganize in association with keratin hypophosphorylation. The K18-DE mutation likely leads to a conformational change that limits the access of p38 kinase (and possibly other kinases) to K8 S80 (and possibly to other keratin phosphorylation sites). Consequently, this drives K18-DE hepatocytes towards cell leakiness and necrosis, depending on the extent and nature of the insult.
Our results differ from those reported for desmin, whereby the parallel caspase-box mutation D263E attenuates cardiomyocyte apoptosis (Panagopoulou et al., 2008). However, the two experimental systems are very different, in that desmin-D263 is introduced as a transgene into mice that overexpress TNF-α in a desmin-null background, but no comparison was made with transgenic mice that overexpress desmin-WT. Taken together, our findings provide several new functional roles for K18 cleavage at its caspase box, including a context-dependent role in promoting filament reorganization during apoptosis, facilitation of stress-induced keratin phosphorylation and protection of hepatocytes from necrosis. These findings have broad potential implications for other intermediate-filament proteins that undergo caspase-mediated cleavage and that harbor potential disease-causing mutations that interfere with caspase digestion (supplementary material Fig. S4).
MATERIALS AND METHODS
Generation of K18-DE transgenic mice
The hK18 D238/D397E (K18-DE) mutant was generated in a 10-kb hK18 genomic DNA using a TransformerTM mutagenesis kit (Clontech). The mutated hK18 genomic DNA was injected into fertilized pronuclei from FVB/n mice. Tail genomic DNA from progeny mice was screened by PCR amplification using the hK18-specific primer set: 5′-CAGAAGGCCAGCTTGGAGAAC-3′ and 5′-ATCTCCTGATCCCAGCACGTG-3′. The hK18-WT, hK8-WT and hK18-S74A mice in FVB/n background have been used extensively by us and others (Ku et al., 1995; Ku et al., 1996; Toivola et al., 1998; Ku and Omary, 2001; Ku and Omary, 2006). All animal experiments were performed according to approved guidelines.
Isolation of RNA and qPCR analysis
Total RNA was isolated from mouse liver using an RNeasy kit (Qiagen) according to the manufacturers' instructions. RNA was reverse transcribed into cDNA using the TaqMan reverse-transcription kit (Applied Biosystems). For qPCR analysis, the cDNA was amplified with Brilliant SYBR green master mix using the hK18 gene-specific primers 5′-CCACCGGGATAGCCGGGGGTC-3′ and 5′-GTAATGGCTCCAGTCTCTGACCTGGG-3′ (MyiQ real-time PCR detection system; Bio-Rad Laboratories). Gapdh was used as the internal control. The relative mRNA of K18-DE to Gapdh was then normalized to that of K18-WT (which was set at a value of 1.0) to calculate the mRNA fold change.
Jo2 administration, liver tissue and blood analysis
For the in vivo experiments, mice (age and sex matched) were fasted overnight followed by intra-peritoneal injection of Jo2 antibody (0.10 µg/g mouse weight, BD Pharmingen). After 4 h, mice were killed by CO2 inhalation and the livers were isolated and divided into pieces that were either stored in liquid nitrogen (for biochemical analysis), embedded in optimum cutting temperature compound and stored at −80°C (for immune staining), or fixed in 10% formalin (for hematoxylin and eosin staining and histological analysis). Blood samples were collected from the mice by intra-cardiac puncture just after CO2 inhalation and were stored overnight at 4°C before analysis. Serum ALT levels were determined using a Vetscan-vs2 instrument (Abaxis), employing the comprehensive diagnostic profile.
Isolation and treatment of primary mouse hepatocytes
Primary hepatocytes from K18-WT and K18-DE mice were isolated as described previously (Snider et al., 2011), with the modifications that are outlined below. Mice were anesthetized with 50 mg/kg Nembutal (Ovation Pharmaceuticals). The liver was perfused with 3–5 ml of perfusion medium (Hanks balanced salt solution containing 0.5 mM EGTA, 5.5 mM glucose and 1% penicillin-streptomycin, pH 7.7–7.8, 37°C) through the portal vein with a flow rate of 3 ml/min, followed by perfusion with 15–20 ml of digestion medium [Williams Medium E supplemented with 1% penicillin-streptomycin and 150 units/ml of collagenase-II (Worthington), pH 7.7–7.8, 37°C] at the same flow rate. The perfused liver was placed in a sterile Petri dish containing ice-cold wash medium (William's medium E supplemented with 1% penicillin-streptomycin) and was subjected to mechanical breakdown. The cell suspension was filtered through a 70-µm cell strainer and was pelleted by centrifugation (50 g for 2 min at 4°C). The cell pellet was resuspended in 6 ml of ice-cold wash medium and was separated on an ice-cold Percoll gradient [Sigma, 15% in phosphate-buffered saline (PBS) pH 7.5, 50 g for 10 min] to remove dead cells. The cell pellet was washed again with ice-cold wash medium and was resuspended in culture medium [William's medium E supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin, 37°C] and plated at a density of 0.25×106 cells/ml on collagen-I-coated six-well plates (BD BioCoat) for biochemical experiments, four-well chamber slides for immunostaining or 96-well plates for MTT assays. After 1 h of cell attachment (37°C, under 5% CO2), the cell culture medium was replaced with fresh medium and was allowed to equilibrate for an additional 12 h (37°C, under 5% CO2). The cultured hepatocytes were then treated with Jo2 (0.5 µg/ml, 6 h), okadaic acid (1 µM) or hypoosmotic medium (200 or 30 mOsm/l for 6 h) in the absence (for experiments to analyze cell culture medium for HMGB1 and LDH release) or presence of FBS.
Preparation of liver and hepatocyte lysates and biochemical analysis
High-salt extraction (HSE) was performed as described previously (Ku et al., 2004). Total-liver or primary-hepatocyte lysates were prepared by homogenizing the liver tissue or cells using 2× Tris-Glycine SDS sample buffer. Sera were also mixed with 2× SDS-containing sample buffer before analysis. Proteins were separated using SDS-PAGE and then were stained with Coomassie Blue or were transferred to polyvinylidene fluoride membranes followed by blotting with antibodies against active or total caspases 3 and 7, PARP, p38, phospho-p38 (Cell Signaling), LDH (LifeSpan BioSciences), HMGB1 (Abcam), actin (Neomarkers), K8-pS80, K8-pS438, total K8, K18-pS34, K18-pS53 or total K18 (Ku et al., 2004).
Immunofluorescence staining and confocal imaging
The immunostaining of liver sections and cells was carried out as described previously (Ku et al., 2004). Briefly, liver sections (5-µm thick) were fixed with acetone (for 10 min at −20°C), and primary cultured hepatocytes were fixed with methanol (for 10 min at −20°C) and air dried (for 1 h). Nonspecific binding was blocked by incubation in blocking buffer [PBS with 2.5% wt/vol bovine serum albumin (BSA) and 2% goat serum] for 10 min. Fixed and air-dried liver sections or cells were incubated with antibody specific to hK18 (Ku et al., 2004) in blocking buffer for 1 h at 22°C, followed by three 5 min washes in PBS and incubation with Alexa-Fluor-conjugated antibody (30 min). The slides were washed three times (5 min/wash) in PBS, air dried and mounted in ProLong Gold containing DAPI (Invitrogen). Sections were imaged using a laser-scanning confocal microscope (FluoView 500; Olympus) with a ×60 oil immersion (1.4 NA) objective.
Determination of the percentage of apoptotic cells and the extent of hemorrhage
The percentage of apoptotic cells in liver sections was determined using a terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) kit (Roche) as described previously (Weerasinghe et al., 2011). The extent to which hemorrhage replaced normal architecture was scored qualitatively in a blinded fashion as follows: 0, no hemorrhage; 1, <20% hemorrhage; 2, 21–40% hemorrhage; 3, 41–60% hemorrhage; 4, 61–80% hemorrhage and 5, >80% hemorrhage. The scores were then used for statistical analysis as reported previously (Weerasinghe et al., 2011).
Analysis of the medium of cultured cells to assess cell leakiness
After individual treatments, culture plates were centrifuged (at 275 g for 5 min) and then the culture medium was collected without disturbing the cells. The collected medium was again centrifuged (at 275 g for 5 min) and the supernatant was used for analysis. The released LDH activity was measured using a colorimetric LDH cytotoxicity detection kit (Clontech). Briefly, 100 µl of culture medium was incubated with 100 µl of reaction mixture (freshly prepared by mixing the recommended proportions of catalyst and dye solution) in a 96-well format (at 37°C for 5–10 min). The absorbance (at 490 nm) was measured using a BioTek Synergy 2 microplate reader and was analyzed using Gen5TM data collection and analysis software. The relative LDH activity was calculated by dividing the absorbance of each treated replicate by the average absorbance of the untreated control (set at a value of 1.0). Each sample was analyzed in four to eight replicates. For biochemical analysis, the culture medium was concentrated using Centricon YM-10 filter devices (Millipore) (typically, 2 ml was concentrated to 0.1 ml) and then mixed with SDS-PAGE sample buffer for subsequent immunoblotting.
MTT assay
The assay was performed in a 96-well format using the Vybrant MTT cell-proliferation-assay kit (Molecular Probes) as recommended by the supplier. Briefly, hepatocyte culture medium (after specific treatments) was replaced with 100 µl of fresh medium containing 10 µl of MTT stock solution and this was incubated for 30 min at 37°C. 100 µl of SDS-HCl solution was then added and incubated for 1–2 h (until the color developed). Absorbance at 570 nm was measured using a BioTek Synergy 2 microplate reader and was analyzed using Gen5TM data collection and analysis software. The percent MTT signal was calculated by dividing the absorbance of each treated replicate by the average absorbance of the untreated control (set to 100%). Each sample was analyzed in six to eight replicates.
DNA constructs and cell transfection
The keratin 8 wild type (K8-WT), K18-WT, K18-D238E/D397E, K18-D238E and K18-D397E constructs were described previously (Ku and Omary, 2001). The pMT3 p38 construct was obtained from Addgene. BHK-21 (baby hamster kidney) or CHO (chinese hamster ovary) cells were transfected using Lipofectamine 2000 (Invitrogen) as recommended by the supplier. Neither cell line expresses keratins but both express vimentin as their major cytoplasmic IF protein.
Keratin immunoprecipitation and in vitro phosphorylation
Cells were lysed in 1% NP-40/PBS-EDTA buffer or liver tissues were homogenized in 1% Empigen/PBS-EDTA lysis buffer and were subsequently used for immunoprecipitation with anti-human K18 antibody (L2A1) that was conjugated to protein-G Dynabeads (Invitrogen). In vitro kinase assays were performed as reported previously (Ku et al., 2004). Briefly, 25 µl of immune-complex-associated magnetic beads (total bead volume) was mixed with 25 µl of 1× kinase buffer (25 mM Tris-HCl pH 7.5, 5 mM β-glycerophosphate, 10 mM MgCl2, 0.1 mM Na3VO4 and 1 mM MnCl2) and heated to 90°C for 2 min to remove any endogenous-associated kinase activity. After cooling to room temperature, the supernatant was replaced with 25 µl of 1× kinase buffer containing 10 µCi of [γ-32P]ATP (PerkinElmer), 20 µM ATP (Sigma) and 1 unit of p38 MAP kinase (R&D Systems), and was incubated at 37°C for 15 min. The kinase reaction was quenched by the addition of 25 µl of 2×SDS-PAGE sample buffer followed by heating to 90°C for 4 min. Samples then underwent gel analysis and autoradiography.
Statistical analysis
All statistical analyses were performed using a one-way analysis of variance (ANOVA) or an unpaired Student's t-test in GraphPad Prism 5 statistical software. The pixel intensities of the scanned immunoblots were determined using Adobe Photoshop CS2 version 9.0. Errors bars show standard error of the mean (s.e.m.).
Supplementary Material
Acknowledgments
We thank David Moons (University of Michigan, Ann Arbor, MI) and Daniel Portney (University of Michigan, Ann Arbor, MI) for assistance with some of the experiments.
Footnotes
Competing interests
The authors declare no competing interests.
Author contributions
S.V.W.W., N.-O.K. and M.B.O. designed the experiments and wrote the manuscript. S.V.W.W. and N.-O.K. performed most of the experiments with assistance from P.J.A. and R.K. All authors read and edited the manuscript.
Funding
This work was supported by the National Institutes of Health [grant number DK47918]; and the Department of Veterans Affairs (to M.B.O.); the National Institutes of Health [grant number DK34933 to the University of Michigan]; the Korean Research World Class University project [grant number R31-10086]; and the Korea Health Industry Development Institute [grant number A111769 to N.-O.K]. Deposited in PMC for release after 12 months.
Supplementary material available online at http://jcs.biologists.org/lookup/suppl/doi:10.1242/jcs.138479/-/DC1
References
- Brockmann K., Meins M., Taubert A., Trappe R., Grond M., Hanefeld F. (2003). A novel GFAP mutation and disseminated white matter lesions: adult Alexander disease? Eur. Neurol. 50, 100–105 10.1159/000072507 [DOI] [PubMed] [Google Scholar]
- Butin-Israeli V., Adam S. A., Goldman A. E., Goldman R. D. (2012). Nuclear lamin functions and disease. Trends Genet. 28, 464–471 10.1016/j.tig.2012.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byun Y., Chen F., Chang R., Trivedi M., Green K. J., Cryns V. L. (2001). Caspase cleavage of vimentin disrupts intermediate filaments and promotes apoptosis. Cell Death Differ. 8, 443–450 10.1038/sj.cdd.4400840 [DOI] [PubMed] [Google Scholar]
- Caulín C., Salvesen G. S., Oshima R. G. (1997). Caspase cleavage of keratin 18 and reorganization of intermediate filaments during epithelial cell apoptosis. J. Cell Biol. 138, 1379–1394 10.1083/jcb.138.6.1379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen F., Chang R., Trivedi M., Capetanaki Y., Cryns V. L. (2003). Caspase proteolysis of desmin produces a dominant-negative inhibitor of intermediate filaments and promotes apoptosis. J. Biol. Chem. 278, 6848–6853 10.1074/jbc.M212021200 [DOI] [PubMed] [Google Scholar]
- Coulombe P. A., Omary M. B. (2002). ‘Hard’ and ‘soft’ principles defining the structure, function and regulation of keratin intermediate filaments. Curr. Opin. Cell Biol. 14, 110–122 10.1016/S0955--0674(01)00301--5 [DOI] [PubMed] [Google Scholar]
- Coulombe P. A., Kerns M. L., Fuchs E. (2009). Epidermolysis bullosa simplex: a paradigm for disorders of tissue fragility. J. Clin. Invest. 119, 1784–1793 10.1172/JCI38177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs E., Weber K. (1994). Intermediate filaments: structure, dynamics, function, and disease. Annu. Rev. Biochem. 63, 345–382 10.1146/annurev.bi.63.070194.002021 [DOI] [PubMed] [Google Scholar]
- Gehringer M. M. (2004). Microcystin-LR and okadaic acid-induced cellular effects: a dualistic response. FEBS Lett. 557, 1–8 10.1016/S0014--5793(03)01447--9 [DOI] [PubMed] [Google Scholar]
- Gilbert S., Loranger A., Daigle N., Marceau N. (2001). Simple epithelium keratins 8 and 18 provide resistance to Fas-mediated apoptosis. The protection occurs through a receptor-targeting modulation. J. Cell Biol. 154, 763–774 10.1083/jcb.200102130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- He T., Stepulak A., Holmström T. H., Omary M. B., Eriksson J. E. (2002). The intermediate filament protein keratin 8 is a novel cytoplasmic substrate for c-Jun N-terminal kinase. J. Biol. Chem. 277, 10767–10774 10.1074/jbc.M111436200 [DOI] [PubMed] [Google Scholar]
- Herrmann H., Bär H., Kreplak L., Strelkov S. V., Aebi U. (2007). Intermediate filaments: from cell architecture to nanomechanics. Nat. Rev. Mol. Cell Biol. 8, 562–573 10.1038/nrm2197 [DOI] [PubMed] [Google Scholar]
- Kim S., Coulombe P. A. (2007). Intermediate filament scaffolds fulfill mechanical, organizational, and signaling functions in the cytoplasm. Genes Dev. 21, 1581–1597 10.1101/gad.1552107 [DOI] [PubMed] [Google Scholar]
- Ku N. -O., Zhou X., Toivola D. M., Omary M. B. (1999). The cytoskeleton of digestive epithelia in health and disease. Am. J. Physiol. 227, G1108–37 [DOI] [PubMed] [Google Scholar]
- Ku N-O., Omary M. B. (2001). Effect of mutation and phosphorylation of type I keratins on their caspase-mediated degradation. J. Biol. Chem. 276, 26792–26798 10.1074/jbc.M103315200 [DOI] [PubMed] [Google Scholar]
- Ku N-O., Omary M. B. (2006). A disease- and phosphorylation-related nonmechanical function for keratin 8. J. Cell Biol. 174, 115–125 10.1083/jcb.200602146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ku N-O., Michie S., Oshima R. G., Omary M. B. (1995). Chronic hepatitis, hepatocyte fragility, and increased soluble phosphoglycokeratins in transgenic mice expressing a keratin 18 conserved arginine mutant. J. Cell Biol. 131, 1303–1314 10.1083/jcb.131.5.1303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ku N-O., Michie S. A., Soetikno R. M., Resurreccion E. Z., Broome R. L., Oshima R. G., Omary M. B. (1996). Susceptibility to hepatotoxicity in transgenic mice that express a dominant-negative human keratin 18 mutant. J. Clin. Invest. 98, 1034–1046 10.1172/JCI118864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ku N-O., Liao J., Omary M. B. (1997). Apoptosis generates stable fragments of human type I keratins. J. Biol. Chem. 272, 33197–33203 10.1074/jbc.272.52.33197 [DOI] [PubMed] [Google Scholar]
- Ku N-O., Azhar S., Omary M. B. (2002). Keratin 8 phosphorylation by p38 kinase regulates cellular keratin filament reorganization: modulation by a keratin 1-like disease causing mutation. J. Biol. Chem. 277, 10775–10782 10.1074/jbc.M107623200 [DOI] [PubMed] [Google Scholar]
- Ku N-O., Soetikno R. M., Omary M. B. (2003). Keratin mutation in transgenic mice predisposes to Fas but not TNF-induced apoptosis and massive liver injury. Hepatology 37, 1006–1014 10.1053/jhep.2003.50181 [DOI] [PubMed] [Google Scholar]
- Ku N-O., Toivola D. M., Zhou Q., Tao G. Z., Zhong B., Omary M. B. (2004). Studying simple epithelial keratins in cells and tissues. Methods Cell Biol. 78, 489–517 10.1016/S0091--679X(04)78017--6 [DOI] [PubMed] [Google Scholar]
- Lanktree M., Cao H., Rabkin S. W., Hanna A., Hegele R. A. (2007). Novel LMNA mutations seen in patients with familial partial lipodystrophy subtype 2 (FPLD2; MIM 151660). Clin. Genet. 71, 183–186 10.1111/j.1399--0004.2007.00740.x [DOI] [PubMed] [Google Scholar]
- Leers M. P., Kölgen W., Björklund V., Bergman T., Tribbick G., Persson B., Björklund P., Ramaekers F. C., Björklund B., Nap M. et al. (1999). Immunocytochemical detection and mapping of a cytokeratin 18 neo-epitope exposed during early apoptosis. J. Pathol. 187, 567–572 [DOI] [PubMed] [Google Scholar]
- Loranger A., Duclos S., Grenier A., Price J., Wilson-Heiner M., Baribault H., Marceau N. (1997). Simple epithelium keratins are required for maintenance of hepatocyte integrity. Am. J. Pathol. 151, 1673–1683 [PMC free article] [PubMed] [Google Scholar]
- MacFarlane M., Merrison W., Dinsdale D., Cohen G. M. (2000). Active caspases and cleaved cytokeratins are sequestered into cytoplasmic inclusions in TRAIL-induced apoptosis. J. Cell Biol. 148, 1239–1254 10.1083/jcb.148.6.1239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marceau N., Loranger A., Gilbert S., Daigle N., Champetier S. (2001). Keratin-mediated resistance to stress and apoptosis in simple epithelial cells in relation to health and disease. Biochem. Cell Biol. 79, 543–555 10.1139/o01--138 [DOI] [PubMed] [Google Scholar]
- Marceau N., Schutte B., Gilbert S., Loranger A., Henfling M. E., Broers J. L., Mathew J., Ramaekers F. C. (2007). Dual roles of intermediate filaments in apoptosis. Exp. Cell Res. 313, 2265–2281 10.1016/j.yexcr.2007.03.038 [DOI] [PubMed] [Google Scholar]
- Omary M. B., Ku N-O., Strnad P., Hanada S. (2009). Toward unraveling the complexity of simple epithelial keratins in human disease. J. Clin. Invest. 119, 1794–1805 10.1172/JCI37762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panagopoulou P., Davos C. H., Milner D. J., Varela E., Cameron J., Mann D. L., Capetanaki Y. (2008). Desmin mediates TNF-α-induced aggregate formation and intercalated disk reorganization in heart failure. J. Cell Biol. 181, 761–775 10.1083/jcb.200710049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao L., Perez D., White E. (1996). Lamin proteolysis facilitates nuclear events during apoptosis. J. Cell Biol. 135, 1441–1455 10.1083/jcb.135.6.1441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schutte B., Henfling M., Kölgen W., Bouman M., Meex S., Leers M. P., Nap M., Björklund V., Björklund P., Björklund B. et al. (2004). Keratin 8/18 breakdown and reorganization during apoptosis. Exp. Cell Res. 297, 11–26 10.1016/j.yexcr.2004.02.019 [DOI] [PubMed] [Google Scholar]
- Schweizer J., Bowden P. E., Coulombe P. A., Langbein L., Lane E. B., Magin T. M., Maltais L., Omary M. B., Parry D. A., Rogers M. A. et al. (2006). New consensus nomenclature for mammalian keratins. J. Cell Biol. 174, 169–174 10.1083/jcb.200603161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snider N. T., Omary M. B. (2014). Post-translational modifications of intermediate filament proteins: mechanisms and functions. Nat. Rev. Mol. Cell Biol. 15, 163–77 10.1038/nrm3753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snider N. T., Weerasinghe S. V., Singla A., Leonard J. M., Hanada S., Andrews P. C., Lok A. S., Omary M. B. (2011). Energy determinants GAPDH and NDPK act as genetic modifiers for hepatocyte inclusion formation. J. Cell Biol. 195, 217–229 10.1083/jcb.201102142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Štros M. (2010). HMGB proteins: interactions with DNA and chromatin. Biochim. Biophys. Acta 1799, 101–113 10.1016/j.bbagrm.2009.09.008 [DOI] [PubMed] [Google Scholar]
- Toivola D. M., Omary M. B., Ku N-O., Peltola O., Baribault H., Eriksson J. E. (1998). Protein phosphatase inhibition in normal and keratin 8/18 assembly-incompetent mouse strains supports a functional role of keratin intermediate filaments in preserving hepatocyte integrity. Hepatology 28, 116–128 10.1002/hep.510280117 [DOI] [PubMed] [Google Scholar]
- Weerasinghe S. V., Moons D. S., Altshuler P. J., Shah Y. M., Omary M. B. (2011). Fibrinogen-γ proteolysis and solubility dynamics during apoptotic mouse liver injury: heparin prevents and treats liver damage. Hepatology 53, 1323–1332 10.1002/hep.24203 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.