Abstract
RNAi is a promising potential therapeutic approach for many diseases. A major barrier to its clinical translation is the lack of efficient delivery systems for siRNA. Among nonviral vectors, nonionic surfactant vesicles (niosomes) have shown a great deal of promise in terms of their efficacy and toxicity profiles. Nonionic surfactants have been shown to be a superior alternative to phospholipids in several studies. There is a large selection of surfactants with various properties that have been incorporated into niosomes. Therefore, there is great potential for innovation in terms of nisome composition. This article summarizes recent advancements in niosome technology for the delivery of siRNA.
Keywords: gene delivery, niosome, nonionic surfactant vesicle, siRNA delivery
Delivery of RNAi therapeutics
Gene silencing-based therapy has the potential to transform modern medicine [1,2]. Synthetic siRNAs or miRNA mimics can be incorporated into RNA-induced silencing complexes to knockdown target genes [3]. A related therapeutic strategy to inhibit miRNA function is by introduction of miR inhibitors, sometimes known as anti-miRs or antagomirs. It is worth noting that RNAi can be induced by gene transfer in the form of shRNAs, by either viral or nonviral vectors, which are then transcribed and processed into active miRNA duplexes by innate endonucleases drosha and dicer. However, this strategy requires nuclear delivery of a large DNA molecule, such as a plasmid, which faces increased challenges in delivery [2,4]. By contrast, synthetic RNAi agents are much smaller in size (duplexes of oligomers of approximately 21 bases in length or in the case of anti-miRs, short single-stranded oligomers) and can incorporate chemical modifications into the backbone and termini for improved stability [5]. Despite these advantages, oligonucleotide agents are high-molecular-weight polyanions that cannot readily diffuse across cellular membranes, which presents a significant barrier to delivery [6,7]. Moreover, these molecules need to resist degradation during systemic circulation, extravasate and get across the cellular membrane to reach the cytoplasmic site of therapeutic action. The most promising strategies for RNAi delivery seem to be a combination of oligonucleotide backbone chemical modifications and formulation into nanoparticles [8].
Nanoparticles for nucleic acid delivery are typically synthesized by a self-assembly process driven by electrostatic interactions between a cationic polymer or lipid and an anionic nucleic acid, such as siRNA. A net positive charge of the assembled particles can mediate cellular uptake via electrostatic adhesion to cellular surfaces, which carry a slight negative charge. This basis of charge interaction is very helpful in facilitating siRNA delivery in vitro [3–4]. However, in vivo, highly charged particles are rapidly cleared from the circulation due to strong interactions with plasma components and the reticuloendothelial system [9]. As a result, there is generally little correlation between optimal compositions for high delivery efficiency of a delivery vehicle in vitro and in vivo [10]. To optimize in vivo delivery, it is important to make stable nanoparticles that can survive circulation, but not so stable that they are rendered inactive following cellular internalization. Achieving this balance requires rational design of nanoparticle composition [11]. A well-known system for siRNA delivery is based on stable nucleic acid lipid particles, for example, with a composition of cholesterol, dipalmitoylphosphatidylcholine, 3-N-[(ωmethoxy poly(ethylene glycol)2000)carbamoyl]-1,2-dimyrestyloxypropylamine and the cationic component 1,2-dilinoleyloxy-3-N,N-dimethylaminopropane [12]. Stable nucleic acid lipid particles are currently in Phase I and II clinical trials for the delivery of siRNAs; ALN-VSP02 and TKM-PLK1, respectively. Lipid nanoparticles for siRNA delivery have been the subject of several recent review articles [13–15]. Since delivery is key to the successful clinical translation of siRNA therapeutics, the need for additional effort in this area of research is evident.
Nonionic surfactant vesicles for nucleic acid delivery
Similar to the zwitterionic phospholipids that form liposomes, nonionic surfactants with a cylindrical geometry are capable of forming bilayer vesicles, termed ‘niosomes’. Like liposomes, these vesicles have been used for drug [16–31], gene [32–35] and siRNA [36,37] delivery. Niosomes and liposomes differ in several respects. First, surfactants are generally lower in cost and are potentially more stable than phospholipids, which are subject to oxidation and degradation by phospholipases. Indeed, niosomes may have longer shelf lives and be more stable than liposomes in vivo. The selection of synthetic surfactants may be further advantageous to lipids derived from natural sources, which can differ from batch to batch in terms of purity. A large selection of surfactants that display a wide range of properties desirable for specific drug delivery applications are readily available.
Composition of niosomes
Niosomes are typically composed of nonionic surfactants and cholesterol [38]. The ability to form bilayer vesicles from surfactants depends on their hydrophile–lipophile balance (HLB) value and critical packing parameter, as well as other factors. A critical packing parameter in the range of 0.5–1 indicates that a surfactant is likely to form spherical vesicles [39,40]. The optimal HLB value for high-loading efficiency niosomes is approximately 8.6. Surfactants with either higher or lower HLB values form vesicles with lower stability and lower volumes of entrapment [41–44]. The addition of cholesterol may be used to further increase the stability of niosomes.
Nonionic surfactants
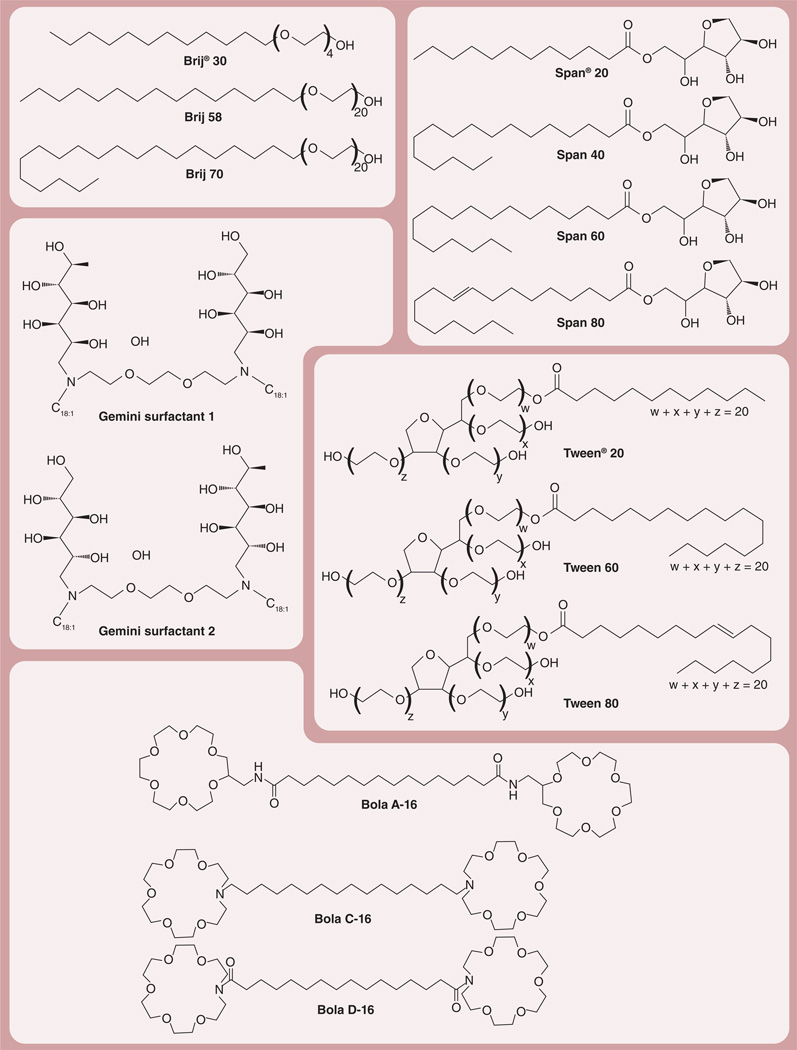
Common nonionic surfactants that can be used to prepare niosomes are classified by chemical structure as described below (Figure 1) [38,39,45,46].
-
▪
Alkyl ethers and alkyl glyceryl ethers such as polyoxyethylene 4 lauryl ether (Brij® 30), polyoxyethylene cetyl ethers (Brij 58) and polyoxyethylene stearyl ethers (Brij 72 and Brij 76; Sigma Aldrich, MO, USA);
-
▪
Sorbitan fatty acid esters such as Span® 20, Span 40, Span 60 and Span 80 (Sigma Aldrich);
-
▪
Polyoxyethylene fatty acid esters (polysorbate) such as Tween® 20, Tween 60 and Tween 80 (Sigma Aldrich);
- ▪
- ▪
Figure 1. Nonionic surfactant commonly used to prepare niosomes (facing page).
Structures of alkyl ethers and alkyl glyceryl ethers (Brij®; Sigma Aldrich, MO, USA), sorbitan fatty acid esters (Span®; Sigma Aldrich), polyoxyethylene fatty acid esters (Tween®; Sigma Aldrich), Gemini surfactants 1 and 2, and Bola surfactants.
Cholesterol
Cholesterol may be combined with nonionic surfactants for the preparation of niosomes [51,52]. The gel-to-liquid phase transition of niosomes can be impeded by adding cholesterol, resulting in niosomes that are more rigid and less likely to lose the drug [39]. In a recent study, a 1:1 ratio of cholesterol and nonionic surfactants was shown to be optimal for producing physically stable niosomes [38], possibly owing to interactions between the surfactant and cholesterol [53].
Charge inducer components
Niosomes are often stabilized by the inclusion of a charged lipid. Examples of commonly used anionic lipids include dicetyl phosphate, dihexadecyl phosphate and lipoamine acid. For nucleic acid delivery, a cationic surfactant may be used so that the niosomes form electrostatic complexes with the negatively charged oligonucleotides [39,46]. A slight net surface charge of the nisome–oligonucleotide complexes leads to increased colloidal stability. However, excessive net charge can lead to rapid removal from systemic circulation by the reticuloendothelial system. Thus, a careful balancing of charge is essential to form an effective delivery vehicle.
Applications of niosomes
Niosomes constitute a versatile delivery platform that can be used in various pharmaceutical applications in addition to oligonucleotide delivery, as described in Table 1.
Table 1.
Pharmaceutical applications of niosomes.
| Fields of application | Type of encapsulated drugs/agents | Ref. |
|---|---|---|
| Drug delivery | NSAIDs, anticancer, antibacterial, antifungal, antiviral, steroids, antiglaucoma, antidiabetics, local anesthetics, muscle relaxants, diagnostic agents, contraceptives, hormones, vitamins | [16–31] |
| Immunization | DNA vaccine, vaccine adjuvants | [54–61] |
| Protein/peptide drugs | Insulin, vasopressin | [38,53–64] |
| Nucleic acid delivery | Plasmid DNA, oligonucleotide siRNA |
[32–35] [36,37] |
NSAID: Nonsteroidal anti-inflammatory drug.
Niosomes as nucleic acid carriers
Like cationic liposomes, cationic niosomes can be used for nucleic acid delivery. Typical components of cationic niosomes include a nonionic surfactant, cholesterol and a cationic lipid [33,35]. Niosomes have been used successfully for topical gene delivery [46] and for synthesis of DNA vaccines [54,55]. In one study, plasmid delivery into rat skin mediated by niosomes resulted in high levels of β-galactosidase and luciferase reporter gene expression [56]. Manosroi et al. demonstrated the use of elastic cationic niosomes composed of Tween 61 (Sigma Aldrich), cholesterol and DDAB as a topical delivery system for the tyrosinase gene as a treatment for vitiligo [57]. To improve the stability and cellular delivery of oligodeoxynucleotides (ODNs), Huang et al. prepared PEGylated cationic niosomes by modifying cationic niosomes with a PEG-conjugated lipid. Complexes of PEGylated cationic niosomes and ODNs showed a neutral ζ-potential with a particle size of approximately 300 nm. The PEG modification significantly decreased the serum binding and particle aggregation in the presence of serum, provided greater resistance to serum nuclease and enhanced the efficiency of ODN delivery [32]. Vyas et al. developed a niosome-based delivery system for DNA vaccines. DNA encoding hepatitis B surface antigen was encapsulated in niosomes composed of Span 85 (Sigma Aldrich) and cholesterol. The results showed that immunization using topical niosomes can elicit a comparable serum antibody titer and cytokine levels compared with those following intramuscular administration of recombinant hepatitis B surface antigen and topical administration of DNA vaccines in liposomes [54]. Gene transfer efficiency mediated by cationic niosomes is influenced by their composition, including the types of surfactants and cationic lipids used [32–35,58]. Paecharoenchai et al. investigated the effect of the structure of cationic lipids on pDNA transfection in HeLa cells mediated by cationic niosomes. The cationic niosomes were composed of Span 20, cholesterol and spermine derivative-based cationic lipids with varying acyl chain lengths (carbon [C]14, C16 and C18). The results showed that the transfection efficiency of the Span 20–niosomes was the highest for the spermine–C14 formulation following the order of: spermine–C14 > spermine–C16 > spermine–C18. In addition, Span 20–niosomes showed low cytotoxicity and hemolytic activity [35].
Niosomes as siRNA carriers
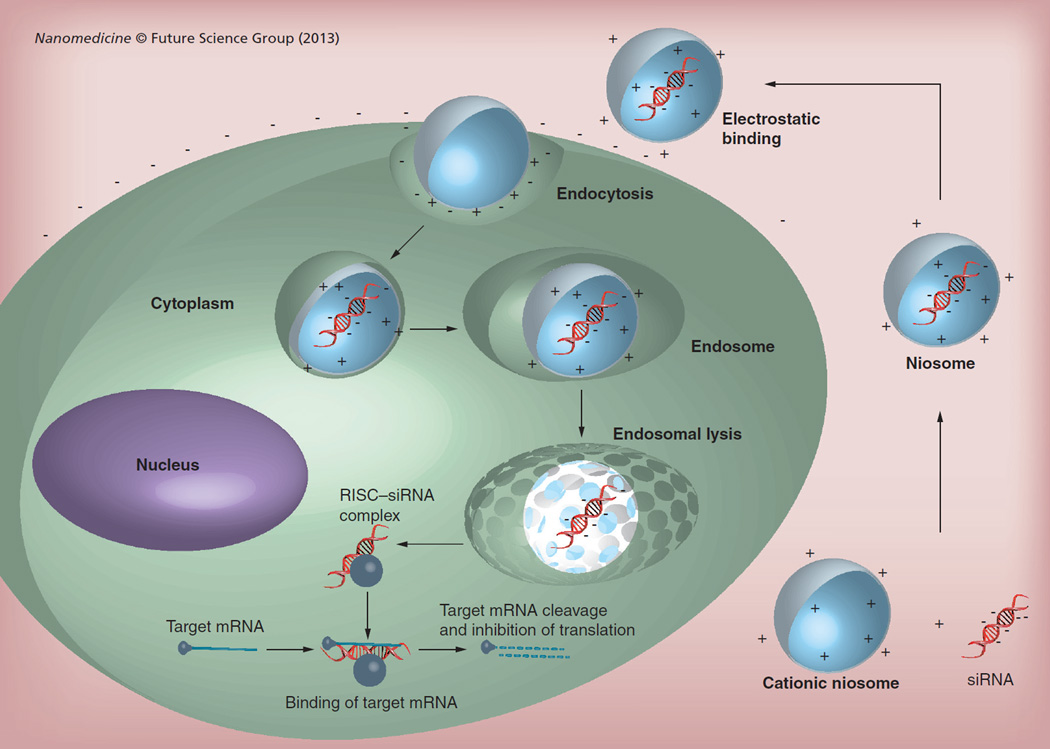
In addition to gene delivery, niosomes have been shown to facilitate siRNA delivery [36,37]. A general scheme for noisome-mediated siRNA delivery is shown in Figure 2. For example, a Span 80-based cationic niosome formulation, also known as SPANosome, was developed specifically for siRNA delivery. It contained 1,2-dioleoyl-3-trimethylammonium-propane as a cationic lipid and d-α-tocopheryl PEG-1000 succinate as a PEGylating lipid [36] at 1 or 5 n/n%. The particle size of empty SPANosomes was 25–40 nm, which decreased with increasing amounts of d-α-tocopheryl PEG-1000 succinate in the formulation. Optimization of the SPANosome–siRNA formulation was carried out by altering the siRNA/surfactant ratio. A decrease in this ratio resulted in smaller particle sizes and increased ζ-potential. The SPANosome–siRNA formulation showed colloidal stability for at least 3 weeks.
Figure 2. Niosome–siRNA delivery pathway.
siRNA forms an electrostatic complex with the cationic niosome. Niosomes are then internalized by the cell and siRNA is released into the cytoplasm to associate with RISC. RISC degrades the sense strand of siRNA and recruits the target mRNA, which is subsequently degraded, thus inhibiting translation.
Gene silencing activity mediated by SPANosome
Gene silencing activity was evaluated in MDA-MB-231-green fluorescent protein (GFP) cells, which were stably transfected with GFP, using SPANosomes carrying the siRNA siGFP. GFP silencing was shown to be dose dependent and reached 66% with the optimized formulation, which was greater than the silencing activity of Lipofectamine® 2000 (Life Technologies, CA, USA). In another experiment, the aromatase gene silencing mediated by SPANosome–siRNA was investigated in SK-Br-3 cells with a siRNA targeting aromatase, siArom. The results based on aromatase assay showed approximately a 77% knockdown by SPANosome–siRNA and the efficiency was greater than that of lipofectamine–siRNA. No significant cytotoxicity in MDA-MB-231 cells was observed with the SPANosome–siRNA formulations at concentrations below 20 µg/ml (~100 nM) siRNA [36]. SPANosomes have yet to be tested in vivo for siRNA delivery so their safety remains to be established [56–58].
Cellular uptake mechanism of SPANosome–siRNA
SPANosome–siRNA was shown to be internalized by tumor cells primarily through the caveolae-mediated pathway, which does not lead to lysosomal delivery and, thus, is less degradative. By contrast, the pathway used by lipofectamine–siRNA was primarily clathrin-mediated endocytosis [37]. Intracellular trafficking of SPANosome–siRNA was studied using molecular beacons as probes of cytoplasmic delivery [37]. The results showed that SPANosome–siRNA had a longer intracellular half-life and greater delivery of molecular beacons into the cytoplasm relative to cationic liposomes–siRNA. Since Span 80 is known to form nonbilayer cubic phases, it may promote the destabilization of the endosomal membrane and subsequently enhance cytosolic delivery of the molecular beacon. Additionally, Huang et al. reported that Spans enhanced transfection mediated by cationic liposomes. This effect might be due to the abilities of Span to destabilize an endosomal membrane and also to promote phase transition from the lamellar phase to inverted hexagonal phase, resulting in cytoplasmic release of DNA [59]. Therefore, nonionic surfactants, such as Span 80, can be considered as ‘helper lipids’ to cationic lipids with greater efficiency than conventional helper lipids such as 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine and cholesterol, which are less active in the presence of serum. Given the wide selection of nonionic surfactants commercially available, there is ample space for innovation and optimization of niosome formulations for siRNA delivery.
Some recent publications on niosomes as gene/siRNA carriers are listed in Table 2.
Table 2.
Niosome-based gene/siRNA delivery systems.
| Niosome compositions | DNA/siRNA | Model | Results | Ref. |
|---|---|---|---|---|
| Tween® 61 (Sigma Aldrich, MO, USA):Chol:DDAB (1:1:0.5 M ratio) | Tyrosinase plasmid (pMEL34) | Rat skin | Higher tyrosinase activity than the free plasmid | [57] |
| Tween 61:Chol:DDAB (1:1:0.5 M ratio) | Luciferase plasmid | Rat skin | Enhanced transdermal absorption of luciferase plasmid | [58] |
| Span® 20 (Sigma Aldrich):Chol:spermine–C14 Span 20:Chol:spermine–C16 Span 20:Chol:spermine–C18 (2.5:2.5:1 M ratio) |
pEGFP-C2 | HeLa cell line | High transfection efficiency with low cytotoxicity and low hemolytic effect; transfection efficiency is affected by cationic lipid structure | [35] |
| GDL:Chol:POE-10 | β-galactosidase reporter | Rat skin | Intense staining of follicular and epidermal cells | [56] |
| Span 85 (Sigma Aldrich):Chol (7:3 M ratio) | HBsAg | BALB/c mice | High serum antibody titer and endogenous cytokines | [54] |
| Span 40 (Sigma Aldrich):DC-Chol (1:1 M ratio) with PEG2000-DSPE (5 mol%) | Oligodeoxynucleotides | COS-7 cell line | PEGylated cationic niosomes showed a higher efficiency of oligodeoxynucleotide cellular uptake and decreased the binding of serum protein | [32] |
| DOTAP:Span 80:TPGS (50:49:1/50:45:5 M ratio) SPANosomes | siGFP, siArom | MDA-MB-231 cells with/without stably transfected GFP, aromatase-expressing cell line SK-Br-3 | Transfection efficiency greater than cationic liposome-based reagent, 66% GFP gene silencing, 77% aromatase gene silencing | [36] |
| DOTAP:Span 80:TPGS (50:49:1 M ratio) SPANosomes | FAM-siRNA, Cy3-MB, Silencer® renilla luciferase siRNA (Invitrogen, CA, USA) | SK Hep-1 cells with stable luciferase expression, (SK Hep-1 Luc) and flow cytometry | Divergent cellular pharmacokinetic profiles of the niosomes and liposomes were associated with different cellular entry pathways | [37] |
Chol: Cholesterol; DC-Chol: 1-cholesteryl 3-N-(dimethylaminoethyl) carbamate; DDAB: Dimethyl dioctadecyl ammonium bromide; DOTAP: 1,2-dioleoyl-3-trimethylammonium-propane; FAM: Carboxyfluorescein; GDL: Glyceryl dilaurate; GFP: Green fluorescent protein; HBsAg: Hepatitis B surface antigen; PEG2000-DSPE: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-(PEG)-2000; POE-10: Polyoxyethylene-10 stearyl ether; siArom: siRNA targeting aromatase; siGFP: Silencer® enhanced GFP siRNA; Spermine–C14: N1,N1-dimyristeroyloxyethyl-spermine; Spermine-C16: N1,N1-palmitoyloxyethyl-spermine; Spermine–C18: N1,N1-steroyloxyethyl-spermine; TPGS: d-α-tocopheryl PEG-1000 succinate.
Conclusion
siRNA and other oligonucleotide-based therapeutics represent great opportunities for drug development. Developing efficient delivery systems is the key to their successful clinical translation. Niosomes have shown superior activities over well-known lipid-based delivery systems. Careful selection of surfactant and lipid components determines the encapsulation, pharmacokinetic and release properties of niosomes. Like liposomes, niosomes may have applications in many pharmaceutical fields including conventional drug delivery, protein/peptide delivery, vaccine delivery and oligonucleotide delivery. Current data appear to suggest that the success of niosomes for siRNA delivery may be due to a combination of caveolae-mediated cellular entry and the membrane bilayer destabilization effect characteristic of surfactant molecules.
Future perspective
Niosome technology for the delivery of ODNs and siRNA is still in its early stages and there is much room for improvement and innovation. A large variety of surfactants and lipid combinations that could benefit the delivery system remain untested. Concerns relating to particle size and long-term colloidal stability will need to be addressed by careful adjustment of surface charge parameters and perhaps postproduction considerations such as lyophilization. Determination of the in vivo efficacy of the formulation will be necessary moving forward to determine if off-target toxicity is a limiting factor for niosomes. Thus far, niosomes have only been tested in vitro or topically; demonstration of efficacy via paternal administration would further expand its application clinically. The application of targeting agents such as antibodies may also be of benefit to niosome formulations should off-target toxicity present an issue. Taken together, niosomes represent an exciting opportunity for the treatment of cancer and other diseases that do not respond well to traditional methods of treatment.
Executive summary.
Delivery of RNAi therapeutics
-
▪
The potential of RNAi therapeutics has been largely limited by inefficient methods of delivery.
-
▪
Nonviral vectors, which take advantage of electrostatic interactions with RNAi therapeutics, form stable complexes that promote delivery to the intracellular target.
Nonionic surfactant vesicles for nucleic acid delivery
-
▪
Niosomes possess a variety of chemical properties that make them advantageous relative to the classically used phospholipids.
-
▪
Niosomes are composed of nonionic surfactants, cholesterol and charge-inducer components.
Applications of niosomes
-
▪
Niosomes have shown success in the delivery of several classes of drug, including nucleic acid-based drugs.
-
▪
Among niosomes, SPANosomes, based on the surfactant Span® 80 (Sigma Aldrich, MO, USA), have experienced success owing to utilization of the caveolae-mediated pathway for cellular entry.
Conclusion
-
▪
The development of carrier systems is essential to the implementation of RNAi therapeutics.
-
▪
Niosomes demonstrate increased efficacy over conventional lipid-based delivery systems.
Future perspective
-
▪
Further optimization and characterization of niosome formulations will potentiate its activity and open doors for new treatment opportunities for patients.
Acknowledgments
The authors wish to thank the Commission of Higher Education (Thailand) and the Thailand Research Funds through the Golden Jubilee PhD Program (Grant No. PHD/0092/2551) for financial support.
Footnotes
Financial & competing interests disclosure
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as:
▪ of interest
- 1.Reischl D, Zimmer A. Drug delivery of siRNA therapeutics: potentials and limits of nanosystems. Nanomedicine. 2009;5:8–20. doi: 10.1016/j.nano.2008.06.001. [DOI] [PubMed] [Google Scholar]
- 2. Wang J, Lu Z, Wientjes GM, Au JL. Delivery of siRNA therapeutics: barriers and carriers. AAPS J. 2010;12(4):492–503. doi: 10.1208/s12248-010-9210-4. ▪ Provides a general overview of the barriers that nanoparticles need to overcome in order to be effective nanocarriers.
- 3. Schroeder A, Levins CG, Cortez C, Langer R, Anderson DG. Lipid-based nanotherapeutics for siRNA delivery. J. Intern. Med. 2010;267(1):9–21. doi: 10.1111/j.1365-2796.2009.02189.x. ▪ Cites examples of several commonly used lipids in siRNA delivery and details examples of their use in clinical applications.
- 4.Spagnou S, Miller AD, Keller M. Lipidic carriers of siRNA: differences in the formulation, cellular uptake, and delivery with plasmid DNA. Biochemistry. 2004;43:13348–13356. doi: 10.1021/bi048950a. [DOI] [PubMed] [Google Scholar]
- 5.Deleavey GF, Damha MJ. Designing chemically modified oligonucleotides for targeted gene silencing. Chem. Biol. 2012;19(8):937–954. doi: 10.1016/j.chembiol.2012.07.011. [DOI] [PubMed] [Google Scholar]
- 6.Vicentini FT, Borgheti-Cardoso LN, Depieri LV, et al. Delivery systems and local administration routes for therapeutic siRNA. Pharm. Res. 2013;30(4):915–931. doi: 10.1007/s11095-013-0971-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rhinn H, Largeau C, Bigey P, et al. How to make siRNA lipoplexes efficient? Add a DNA cargo. Biochim. Biophys. Acta. 2009;1790:219–230. doi: 10.1016/j.bbagen.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 8.Lin PJ, Tam YY, Hafez I, et al. Influence of cationic lipid composition on uptake and intracellular processing of lipid nanoparticle formulations of siRNA. Nanomedicine. 2013;9(2):233–246. doi: 10.1016/j.nano.2012.05.019. [DOI] [PubMed] [Google Scholar]
- 9.Ho EA, Osooly M, Strutt D, et al. Characterization of long-circulating cationic nanoparticle formulations consisting of a two-stage PEGylation step for the delivery of siRNA in a breast cancer tumor model. J. Pharm. Sci. 2013;102(1):227–236. doi: 10.1002/jps.23351. [DOI] [PubMed] [Google Scholar]
- 10.Whitehead KA, Matthews J, Chang PH, et al. In vitro–in vivo translation of lipid nanoparticles for hepatocellular siRNA delivery. Am. Chem. Soc. Nano. 2012;6(8):6922–6929. doi: 10.1021/nn301922x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Semple SC, Akinc A, Chen J, et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 2010;28:172–176. doi: 10.1038/nbt.1602. [DOI] [PubMed] [Google Scholar]
- 12.Heyes J, Palmer L, Bremner K, MacLachlan I. Cationic lipid saturation influences intracellular delivery of encapsulated nucleic acids. J. Control. Release. 2005;107(2):276–287. doi: 10.1016/j.jconrel.2005.06.014. [DOI] [PubMed] [Google Scholar]
- 13.Gavrilov K, Saltzman WM. Therapeutic siRNA: principles, challenges, and strategies. Yale J. Biol. Med. 2012;85(2):187–200. [PMC free article] [PubMed] [Google Scholar]
- 14.David S, Pitard B, Beno JP, Passirani C. Non-viral nanosystems for systemic siRNA delivery. Pharmacol. Res. 2010;62:100–114. doi: 10.1016/j.phrs.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 15.Allen TM, Cullis PR. Liposomal drug delivery systems: from concept to clinical applications. Adv. Drug Deliv. Rev. 2013;65(1):36–48. doi: 10.1016/j.addr.2012.09.037. [DOI] [PubMed] [Google Scholar]
- 16.Fang JY, Hong CT, Chiu WT, Wang YY. Effect of liposomes and niosomes on skin permeation of enoxacin. Int. J. Pharm. 2001;219:61–72. doi: 10.1016/s0378-5173(01)00627-5. [DOI] [PubMed] [Google Scholar]
- 17.Shahiwala A, Misra A. Studies in topical application of niosomally entrapped nimesulide. J. Pharm. Pharm. Sci. 2002;5:220–225. [PubMed] [Google Scholar]
- 18.Pando D, Caddeo C, Manconi M, Fadda AM, Pazos C. Nanodesign of olein vesicles for the topical delivery of the antioxidant resveratrol. J. Pharm. Pharmacol. 2013;65(8):1158–1167. doi: 10.1111/jphp.12093. [DOI] [PubMed] [Google Scholar]
- 19.Tavano L, Vivacqua M, Carito V, Muzzalupo R, Caroleo MC, Nicoletta F. Doxorubicin loaded magneto-niosomes for targeted drug delivery. Colloids Surf. B Biointerfaces. 2013;102:803–807. doi: 10.1016/j.colsurfb.2012.09.019. [DOI] [PubMed] [Google Scholar]
- 20.Bayindir ZS, Yuksel N. Characterization of niosomes prepared with various nonionic surfactants for paclitaxel oral delivery. J. Pharm. Sci. 2010;99:2049–2060. doi: 10.1002/jps.21944. [DOI] [PubMed] [Google Scholar]
- 21.Abdelbary G, El-Gendy N. Niosome-encapsulated gentamicin for ophthalmic controlled delivery. AAPS Pharm. Sci. Tech. 2008;9:740–747. doi: 10.1208/s12249-008-9105-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jadon PS, Gajbhiye V, Jadon RS, Gajbhiye KR, Ganesh N. Enhanced oral bioavailability of griseofulvin via niosomes. AAPS Pharm. Sci. Tech. 2009;10(4):1186–1192. doi: 10.1208/s12249-009-9325-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ruckmani K, Sankar V, Sivakumar M. Tissue distribution, pharmacokinetics and stability studies of zidovudine delivered by niosomes and proniosomes. J. Biomed. Nanotechnol. 2010;6:43–51. doi: 10.1166/jbn.2010.1101. [DOI] [PubMed] [Google Scholar]
- 24.Marianeccia C, Paolino D, Celia C, Fresta M, Carafa M, Alhaique F. Non-ionic surfactant vesicles in pulmonary glucocorticoid delivery: characterization and interaction with human lung fibroblasts. J. Control. Release. 2010;147:127–135. doi: 10.1016/j.jconrel.2010.06.022. [DOI] [PubMed] [Google Scholar]
- 25.Aggarwal D, Kaur IP. Improved pharmacodynamics of timolol maleate from a mucoadhesive niosomal ophthalmic drug delivery system. Int. J. Pharm. 2005;290:155–159. doi: 10.1016/j.ijpharm.2004.10.026. [DOI] [PubMed] [Google Scholar]
- 26.Tamizharasi S, Dubey A, Rathi V, Rathi JC. Development and characterization of niosomal drug delivery of gliclazide. J. Young Pharm. 2009;1:205–209. [Google Scholar]
- 27.Carafa M, Santucci E, Lucania G. Lidocaine-loaded nonionic surfactant vesicles characterization and in vitro permeation studies. Int. J. Pharm. 2002;231:21–32. doi: 10.1016/s0378-5173(01)00828-6. [DOI] [PubMed] [Google Scholar]
- 28.Keservani RK, Sharma AK, Ramteke S. Novel vesicular approach for topical delivery of baclofen via niosomes. Lat. Am. J. Pharm. 2010;29:1364–1370. [Google Scholar]
- 29.Muller D, Foulon M, Bonnemain B, Vandamme TF. Niosomes as carriers of radiopaque contrast agents for x-ray imaging. J. Microencapsul. 2000;17:227–243. doi: 10.1080/026520400288463. [DOI] [PubMed] [Google Scholar]
- 30.Shataelebi MA, Mostafavi SA, Moghaddas A. Niosome as a drug carrier for topical delivery of N-acetyl glucosamine. Res. Pharm. Sci. 2010;5(2):107–117. [PMC free article] [PubMed] [Google Scholar]
- 31.Desai AR, Raghuveer I, Chitme HR, Chandra R. Development and characterization of niosomal drug delivery of α-tocopherol. Int. J. Chem. Anal. Sci. 2010;1:146–158. [Google Scholar]
- 32.Huang Y, Chen J, Chen X, Gao J, Liang W. PEGylated synthetic surfactant vesicles (niosomes): novel carriers for oligonucleotides. J. Mater. Sci. Mater. Med. 2008;19:607–614. doi: 10.1007/s10856-007-3193-4. [DOI] [PubMed] [Google Scholar]
- 33.Manosroi A, Thathang K, Werner RG, Schubert R, Manosroi J. Stability of luciferase plasmid entrapped in cationic bilayer vesicles. Int. J. Pharm. 2008;356:291–299. doi: 10.1016/j.ijpharm.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 34.He CX, Tabata Y, Gao JQ. Non-viral gene delivery carrier and its three-dimensional transfection system. Int. J. Pharm. 2010;386:232–242. doi: 10.1016/j.ijpharm.2009.11.006. [DOI] [PubMed] [Google Scholar]
- 35.Paecharoenchai O, Niyomtham N, Ngawhirunpat T, Rojanarata T, Yingyongnarongkul B, Opanasopit P. Cationic niosomes composed of spermine-based cationic lipids mediate high gene transfection efficiency. J. Drug Target. 2012;20(9):783–792. doi: 10.3109/1061186X.2012.716846. [DOI] [PubMed] [Google Scholar]
- 36. Zhou C, Mao Y, Sugimoto Y, et al. SPANosomes as delivery vehicles for small interfering RNA (siRNA) Mol. Pharm. 2012;9:201–210. doi: 10.1021/mp200426h. ▪ Demonstrates one of the first uses of a sorbitan monooleate for siRNA delivery.
- 37.Zhou C, Zhang Y, Yu B, Phelps MA, Lee LJ, Lee RJ. Comparative cellular pharmacokinetics and pharmacodynamics of siRNA delivery by SPANosomes and by cationic liposomes. Nanomedicine. 2013;9(4):504–513. doi: 10.1016/j.nano.2012.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mahale NB, Thakkar PD, Mali RG, Walunj DR, Chaudhari SR. Niosomes: novel sustained release nonionic stable vesicular systems–an overview. Adv. Colloid Interface Sci. 2012;(183–184):46–54. doi: 10.1016/j.cis.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 39. Rajera R, Nagpal K, Singh SK, Mishra DN. Niosomes: a controlled and novel drug delivery system. Biol. Pharm. Bull. 2011;34(7):945–953. doi: 10.1248/bpb.34.945. ▪ Provides an excellent overview of the advances in niosome technology over the past decade.
- 40.Šegota S, Težak D. Spontaneous formation of vesicles. Adv. Colloid Interface Sci. 2006;121:51–75. doi: 10.1016/j.cis.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 41.Biswal S, Murthy PN, Sahu J, Sahoo P, Amir F. Vesicles of nonionic surfactants (niosomes) and drug delivery potential. Int. J. Pharm. Sci. Nanotechnol. 2008;1:1–8. [Google Scholar]
- 42.Shahiwala A, Misra A. Studies in topical application of niosomally entrapped nimesulide. J. Pharm. Sci. 2002;5:220–225. [PubMed] [Google Scholar]
- 43.Nasr M, Mansour S, Mortada N, Elshamy AA. Vesicular aceclofenac systems: a comparative study between liposomes and niosomes. J. Microencapsul. 2008;25(7):499–512. doi: 10.1080/02652040802055411. [DOI] [PubMed] [Google Scholar]
- 44.Pham TT, Jaafar-Maalej C, Charcosset C, Fessi H. Liposome and niosome preparation using a membrane contactor for scale-up. Colloids Surf. B Biointerfaces. 2013;94:15–21. doi: 10.1016/j.colsurfb.2011.12.036. [DOI] [PubMed] [Google Scholar]
- 45.Kumar GP, Rajeshwarrao P. Nonionic surfactant vesicular systems for effective drug delivery – an overview. Acta Pharm. Sin. B. 2011;1(4):208–219. [Google Scholar]
- 46.Sankhyan A, Pawar P. Recent trends in niosome as vesicular drug delivery system. J. Appl. Pharm. Sci. 2012;2(6):20–32. [Google Scholar]
- 47.Wasungu L, Stuart MC, Scarzello M, Engberts JB, Hoekstra D. Lipoplexes formed from sugar-based gemini surfactants undergo a lamellar-to-micellar phase transition at acidic pH. Evidence for a non-inverted membrane-destabilizing hexagonal phase of lipoplexes. Biochim. Biophys. Acta. 2006;1758:1677–1684. doi: 10.1016/j.bbamem.2006.06.019. [DOI] [PubMed] [Google Scholar]
- 48.Muzzalupo R, Tavano L, Trombino S, Cassano R, Picci N, La Mesa C. Niosomes from alpha,omega-trioxyethylene-bis(sodium 2-dodecyloxy-propylenesulfonate): preparation and characterization. Colloids Surf. B Biointerfaces. 2008;64(2):200–207. doi: 10.1016/j.colsurfb.2008.01.026. [DOI] [PubMed] [Google Scholar]
- 49.Muzzalupo R, Nicoletta FP, Trombino S, Cassano R, Iemma F, Picci N. A new crown ether as vesicular carrier for 5-fluoruracil: synthesis, characterization and drug delivery evaluation. Colloids Surf. B Biointerfaces. 2007;58(2):197–202. doi: 10.1016/j.colsurfb.2007.03.010. [DOI] [PubMed] [Google Scholar]
- 50.Yoshimura T, Hasegawa S, Hirashima N, Nakanishi M, Ohwada T. Anchoring and bola cationic amphiphiles for nucleotide delivery. Effects of orientation and extension of hydrophobic regions. Bioorg. Med. Chem. Lett. 2001;11(22):2897–2901. doi: 10.1016/s0960-894x(01)00563-7. [DOI] [PubMed] [Google Scholar]
- 51.Geusens B, Strobbe T, Bracke S, et al. Lipid-mediated gene delivery to the skin. Eur. J. Pharm. Sci. 2011;43:199–211. doi: 10.1016/j.ejps.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 52.Manosroi A, Wongtrakul P, Manosroi J, et al. Characterization of vesicles prepared with various nonionic surfactants mixed with cholesterol. J. Colloids Surf. B Biointerfaces. 2003;30:129–138. [Google Scholar]
- 53.Nasseri B. Effect of cholesterol and temperature on the elastic properties of niosomal membranes. Int. J. Pharm. 2005;300:95–101. doi: 10.1016/j.ijpharm.2005.05.009. [DOI] [PubMed] [Google Scholar]
- 54.Vyas SP, Singh RP, Jain S, et al. Non-ionic surfactant based vesicles (niosomes) for non-invasive topical genetic immunization against hepatitis B. Int. J. Pharm. 2005;296:80–86. doi: 10.1016/j.ijpharm.2005.02.016. [DOI] [PubMed] [Google Scholar]
- 55.Perrie Y, Barralet JE, McNeil S, Vangala A. Surfactant vesicle-mediated delivery of DNA vaccines via the subcutaneous route. Int. J. Pharm. 2004;284:31–41. doi: 10.1016/j.ijpharm.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 56.Raghavachari N, Fahl WE. Targeted gene delivery to skin cells in vivo: a comparative study of liposomes and polymers as delivery vehicles. J. Pharm. Sci. 2002;91(3):615–622. doi: 10.1002/jps.10061. [DOI] [PubMed] [Google Scholar]
- 57.Manosroi J, Khositsuntiwong N, Manosroi W, Gotz F, Werner RG, Manosroi A. Enhancement of transdermal absorption, gene expression and stability of tyrosinase plasmid (pMEL34)-loaded elastic cationic niosomes: potential application in vitiligo treatment. J. Pharm. Sci. 2010;99(8):3533–3541. doi: 10.1002/jps.22104. [DOI] [PubMed] [Google Scholar]
- 58.Huang Y, Rao Y, Chen J, Yang VC, Liang W. Polysorbate cationic synthetic vesicle for gene deliver. J. Biomed. Mater. Res. A. 2011;96:513–519. doi: 10.1002/jbm.a.32999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Huang YZ, Gao JQ, Chen JL, Liang WQ. Cationic liposomes modified with nonionic surfactants as effective non-viral carrier for gene transfer. Colloids Surf. B. Biointerfaces. 2006;49(2):158–164. doi: 10.1016/j.colsurfb.2006.03.014. [DOI] [PubMed] [Google Scholar]
- 60.Pardakhty A, Shakibaie M, Daneshvar H, Khamesipour A, Mohammadi-Khors T, Forootanfar H. Preparation and evaluation of niosomes containing autoclaved Leishmania major: a preliminary study. J. Microencapsul. 2011;29(3):219–224. doi: 10.3109/02652048.2011.642016. [DOI] [PubMed] [Google Scholar]
- 61.Vangala A, Bramwell VW, McNeil S, Christensen D, Agger EM, Perrie Y. Comparison of vesicle based antigen delivery systems for delivery of hepatitis B surface antigen. J. Control. Release. 2013;119(1):102–110. doi: 10.1016/j.jconrel.2007.01.010. [DOI] [PubMed] [Google Scholar]
- 62.Varshosaz J, Pardakhty A, Hajhashemi VI, Najafabadi AR. Development and physical characterization of sorbitan monoester niosomes for insulin oral delivery. Drug Deliv. 2003;10:251–262. doi: 10.1080/drd_10_4_251. [DOI] [PubMed] [Google Scholar]
- 63.Pardakhty A, Varshosaz J, Rouholamini A. In vitro study of polyoxyethylene alkyl ether niosomes for delivery of insulin. Int. J. Pharm. 2007;328:130–141. doi: 10.1016/j.ijpharm.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 64.Manosroi A, Khanrin P, Werner RG, Götz F, Manosroi W, Manosroi J. Entrapment enhancement of peptide drugs in niosomes. J. Microencapsul. 2010;27(3):272–280. doi: 10.3109/02652040903131293. [DOI] [PubMed] [Google Scholar]