Abstract
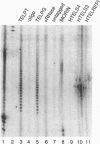
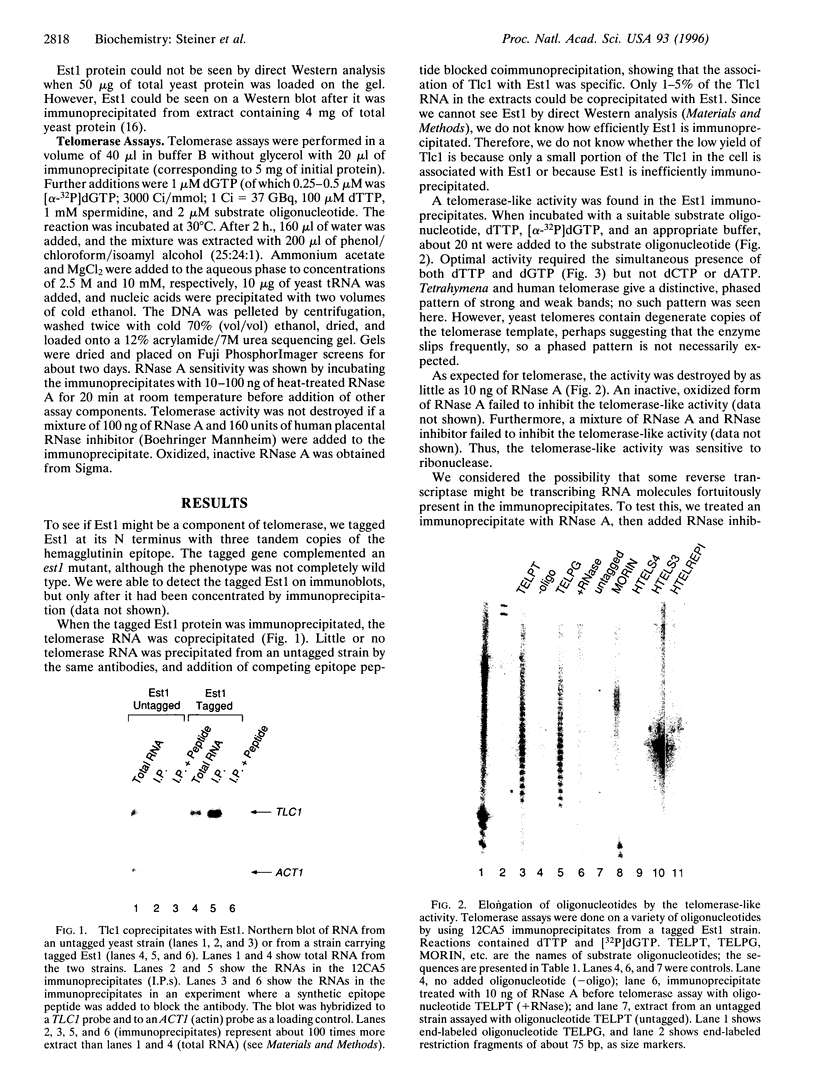
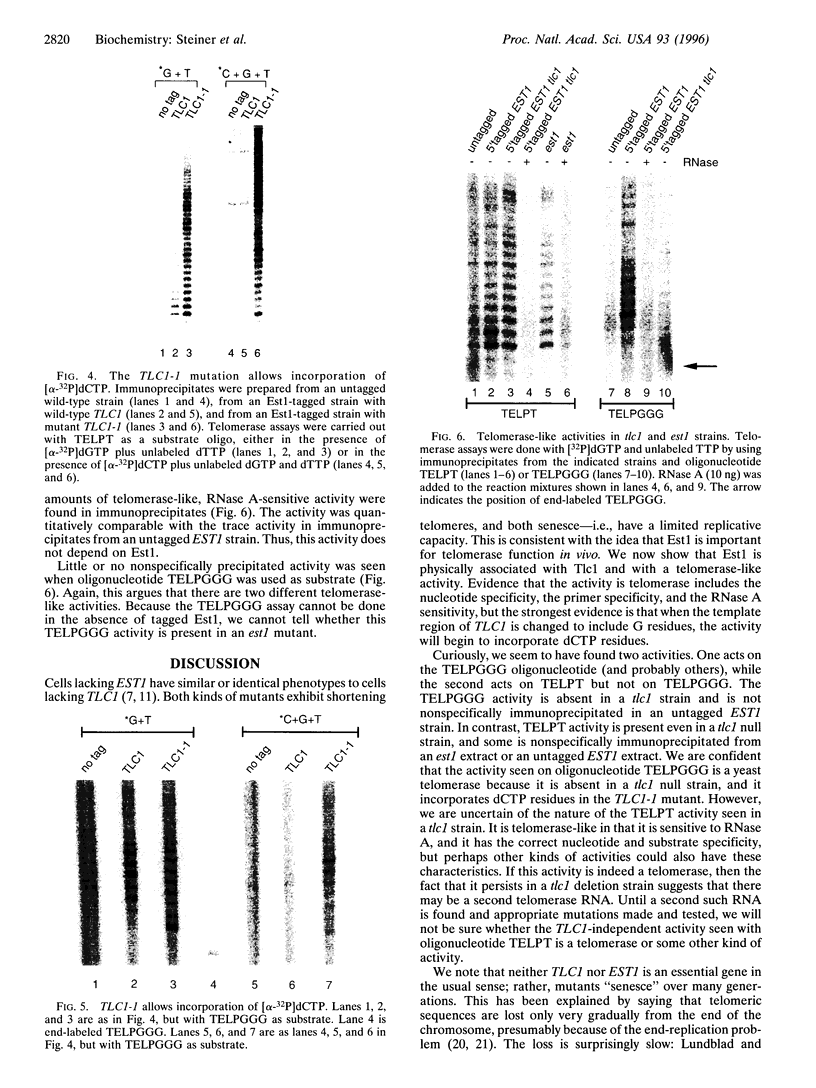
The est1 mutant was previously identified because it is defective in telomere maintenance and displays a senescent phenotype. To see if Est1 might be a component of yeast telomerase, we examined immunoprecipitated Est1. The yeast telomerase RNA Tlc1 specifically coprecipitated with Est1. Furthermore, the Est1 immunoprecipitates contained a telomerase-like activity. As expected for yeast telomerase, the activity elongated telomeric primers, it required dGTP and dTTP but not dATP or dCTP, and it was sensitive to RNase A. Further evidence suggesting that the activity was telomerase was obtained from experiments using a TLC1-1 mutant strain, which has a mutant telomerase template containing dG residues. The activity immunoprecipitated from TLC1-1 mutant strains incorporated 32P-labeled dCTP, while activity from TLC1 strains did not. Use of different telomeric primer substrates revealed two distinguishable telomerase-like activities: one was dependent on TLC1, and one was not. The TLC1-independent activity may be due to a second yeast telomerase RNA, or it may be some other kind of activity.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Autexier C., Greider C. W. Functional reconstitution of wild-type and mutant Tetrahymena telomerase. Genes Dev. 1994 Mar 1;8(5):563–575. doi: 10.1101/gad.8.5.563. [DOI] [PubMed] [Google Scholar]
- Biessmann H., Mason J. M. Genetics and molecular biology of telomeres. Adv Genet. 1992;30:185–249. doi: 10.1016/s0065-2660(08)60321-1. [DOI] [PubMed] [Google Scholar]
- Blackburn E. H. Telomerases. Annu Rev Biochem. 1992;61:113–129. doi: 10.1146/annurev.bi.61.070192.000553. [DOI] [PubMed] [Google Scholar]
- Blackburn E. H. Telomeres: no end in sight. Cell. 1994 Jun 3;77(5):621–623. doi: 10.1016/0092-8674(94)90046-9. [DOI] [PubMed] [Google Scholar]
- Cohn M., Blackburn E. H. Telomerase in yeast. Science. 1995 Jul 21;269(5222):396–400. doi: 10.1126/science.7618104. [DOI] [PubMed] [Google Scholar]
- Collins K., Kobayashi R., Greider C. W. Purification of Tetrahymena telomerase and cloning of genes encoding the two protein components of the enzyme. Cell. 1995 Jun 2;81(5):677–686. doi: 10.1016/0092-8674(95)90529-4. [DOI] [PubMed] [Google Scholar]
- Greider C. W., Blackburn E. H. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature. 1989 Jan 26;337(6205):331–337. doi: 10.1038/337331a0. [DOI] [PubMed] [Google Scholar]
- Greider C. W. Chromosome first aid. Cell. 1991 Nov 15;67(4):645–647. doi: 10.1016/0092-8674(91)90058-7. [DOI] [PubMed] [Google Scholar]
- Kramer K. M., Haber J. E. New telomeres in yeast are initiated with a highly selected subset of TG1-3 repeats. Genes Dev. 1993 Dec;7(12A):2345–2356. doi: 10.1101/gad.7.12a.2345. [DOI] [PubMed] [Google Scholar]
- Lin J. J., Zakian V. A. An in vitro assay for Saccharomyces telomerase requires EST1. Cell. 1995 Jun 30;81(7):1127–1135. doi: 10.1016/s0092-8674(05)80017-0. [DOI] [PubMed] [Google Scholar]
- Lue N. F., Wang J. C. ATP-dependent processivity of a telomerase activity from Saccharomyces cerevisiae. J Biol Chem. 1995 Sep 15;270(37):21453–21456. doi: 10.1074/jbc.270.37.21453. [DOI] [PubMed] [Google Scholar]
- Lundblad V., Blackburn E. H. An alternative pathway for yeast telomere maintenance rescues est1- senescence. Cell. 1993 Apr 23;73(2):347–360. doi: 10.1016/0092-8674(93)90234-h. [DOI] [PubMed] [Google Scholar]
- Lundblad V., Szostak J. W. A mutant with a defect in telomere elongation leads to senescence in yeast. Cell. 1989 May 19;57(4):633–643. doi: 10.1016/0092-8674(89)90132-3. [DOI] [PubMed] [Google Scholar]
- Morin G. B. Recognition of a chromosome truncation site associated with alpha-thalassaemia by human telomerase. Nature. 1991 Oct 3;353(6343):454–456. doi: 10.1038/353454a0. [DOI] [PubMed] [Google Scholar]
- Olovnikov A. M. A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J Theor Biol. 1973 Sep 14;41(1):181–190. doi: 10.1016/0022-5193(73)90198-7. [DOI] [PubMed] [Google Scholar]
- Schneider B. L., Seufert W., Steiner B., Yang Q. H., Futcher A. B. Use of polymerase chain reaction epitope tagging for protein tagging in Saccharomyces cerevisiae. Yeast. 1995 Oct;11(13):1265–1274. doi: 10.1002/yea.320111306. [DOI] [PubMed] [Google Scholar]
- Schneider B. L., Steiner B., Seufert W., Futcher A. B. pMPY-ZAP: a reusable polymerase chain reaction-directed gene disruption cassette for Saccharomyces cerevisiae. Yeast. 1996 Feb;12(2):129–134. doi: 10.1002/(sici)1097-0061(199602)12:2<129::aid-yea891>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- Shampay J., Szostak J. W., Blackburn E. H. DNA sequences of telomeres maintained in yeast. Nature. 1984 Jul 12;310(5973):154–157. doi: 10.1038/310154a0. [DOI] [PubMed] [Google Scholar]
- Singer M. S., Gottschling D. E. TLC1: template RNA component of Saccharomyces cerevisiae telomerase. Science. 1994 Oct 21;266(5184):404–409. doi: 10.1126/science.7545955. [DOI] [PubMed] [Google Scholar]
- Thomas B. J., Rothstein R. Elevated recombination rates in transcriptionally active DNA. Cell. 1989 Feb 24;56(4):619–630. doi: 10.1016/0092-8674(89)90584-9. [DOI] [PubMed] [Google Scholar]
- Tyers M., Tokiwa G., Nash R., Futcher B. The Cln3-Cdc28 kinase complex of S. cerevisiae is regulated by proteolysis and phosphorylation. EMBO J. 1992 May;11(5):1773–1784. doi: 10.1002/j.1460-2075.1992.tb05229.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yudkin J. Sugar and disease. Nature. 1972 Sep 22;239(5369):197–199. doi: 10.1038/239197a0. [DOI] [PubMed] [Google Scholar]
- Zakian V. A. Structure and function of telomeres. Annu Rev Genet. 1989;23:579–604. doi: 10.1146/annurev.ge.23.120189.003051. [DOI] [PubMed] [Google Scholar]