Abstract
Purpose
Pain, fatigue, dyspnea, and distress are commonly reported cancer-related symptoms, but few studies have examined the effects of multiple concurrent symptoms in longer-term cancer survivors. We examined the impact of varying degrees of symptom burden on health-related quality of life (HRQOL) and performance status in surgically treated non-small cell lung cancer (NSCLC) survivors.
Methods
A sample of 183 NSCLC survivors 1–6 years post-surgical treatment completed questionnaires assessing five specific symptoms (pain, fatigue, dyspnea, depression, and anxiety), HRQOL, and performance status. The number of concurrent clinically significant symptoms was calculated as an indicator of symptom burden.
Results
Most survivors (79.8 %) had some degree of symptom burden, with 30.6 % reporting one clinically significant symptom, 27.9 % reporting two symptoms, and 21.3 % reporting three or more symptoms. Physical HRQOL significantly decreased as the degree of symptom burden increased, but mental HRQOL was only significantly decreased in those with three or more symptoms. Receiver-operating characteristic (ROC) curves showed that having multiple concurrent symptoms (two or more) was most likely associated with limitations in functioning (area under a ROC curve=0.75, sensitivity=0.81, specificity=0.54).
Conclusions
Two or more clinically significant symptoms are identified as the “tipping point” for showing adverse effects on HRQOL and functioning. This highlights the need for incorporating multiple-symptom assessment into routine clinical practice. Comprehensive symptom management remains an important target of intervention for improved post-treatment HRQOL and functioning among lung cancer survivors.
Keywords: Lung cancer survivor, Symptom burden, Quality of life, Pain, Depression, Anxiety
Introduction
With improvements in the detection and treatment of cancer, more individuals are diagnosed in early stages and surviving long after treatment ends [1–4]. Yet, for this growing population, substantial consequences of the disease and treatments remain [2, 4–11]. Studies of post-treatment sequelae have focused mainly on single symptoms, but symptoms rarely present in isolation [3–9, 12–24], and emerging research suggests that multiple symptoms may stem from the same etiology (e.g., inflammatory cytokines) [22, 25, 26]. Under-treated symptoms often interfere with cancer survivors’ work [9], activities of daily living [2, 9, 15, 16, 18], and interpersonal relationships [9, 27]. Greater understanding is needed of how combinations of cancer-related symptoms impact the health and well-being of survivors [3, 28].
Patients with lung cancer commonly experience more symptoms than individuals diagnosed with other cancers [4, 6]. The number and type of symptoms experienced by lung cancer patients during and shortly after treatment have been significantly related to impaired functioning, psychological distress, and overall health-related quality of life (HRQOL) [29, 30]. To date, research examining the effects of multiple symptoms among individuals with lung cancer has primarily focused on the active treatment phase or in those with advanced disease [5, 19]. Cooley et al. [14] were among the first to examine the experience of symptoms in lung cancer patients over the course of the disease. In their longitudinal study of 117 patients, symptom distress was moderately high at the time of diagnosis, decreased 3 months after diagnosis, and then increased at 6 months. Less is known about the post-treatment sequelae in longer-term lung cancer survivors or the impact that these symptoms have on functioning at this post-treatment stage.
Patient-reported symptom burden is an important area of assessment and includes the presence, frequency, and severity of multiple symptoms that go untreated or unrelieved [5]. It is defined as the combined impact of disease- and treatment-related symptoms on the ability of individuals to function as they did before onset of their disease or therapy [5]. Pain, fatigue, dyspnea, depression, and anxiety are symptoms commonly reported by lung cancer patients [12]. Studies of various cancer patient and survivor groups have found relationships among these symptoms. Pain is a frequent symptom in cancer survivors [3, 31, 32] and is highly correlated with depression and anxiety [3, 9, 27]. Fatigue is one of the most commonly reported symptoms and one of the most distressing [14, 33]. Cancer-related fatigue and depression are separate constructs but are highly correlated in research studies and can be difficult to distinguish in clinical practice [3]. Although dyspnea is common in many cancer populations, it is most common in patients with lung cancer [33]. Patients with dyspnea often become anxious, which can then exacerbate the sensation of breathlessness, creating a regenerative feedback loop [27, 33].
Performance status and HRQOL are major clinical outcomes that are negatively correlated with cancer- and treatment-related symptoms [15, 16], and higher symptom burden has been associated with poor performance status, worse HRQOL, and even an increased risk of death [19] in newly diagnosed lung cancer. Most interventions have been designed to target a single symptom, and they are not designed to address the complexity of multiple symptoms [3, 9, 20, 22]. Multiple-symptom models offer insights into the experiences of cancer survivors by examining how symptoms interact with each other and the additive effect they may have on patient outcomes (i.e., HRQOL and daily functioning) [24].
The purpose of the present study was to examine symptom burden in a sample of long-term survivors of early-stage non-small cell lung cancer (NSCLC), with the goal of identifying the “tipping point” at which the number and severity of symptoms adversely affects functioning and quality of life. The specific aims of this study were to (1) assess the prevalence and severity of symptom burden 1–6 years post-treatment, (2) identify demographic and medical correlates of moderate–high symptom burden, and (3) determine the degree of symptom burden that corresponds most with functional impairment. We hypothesized that individuals with multiple concurrent symptoms would report poorer HRQOL and performance status. Such data are important in understanding the needs of post-treatment cancer survivors, who are at risk for experiencing chronic symptoms that may impair quality of life. The overall aim of this analysis is to contribute to the design of multiple-symptom-focused interventions to reduce symptom burden for a growing cancer survivor population. Such data may enable health care providers for survivors to take a comprehensive and integrated view of symptom presentation by their patients and promote steps to address multi-symptom burden.
Methods
Participants
Patients were identified from queries of institutional clinical and research databases at Memorial Sloan-Kettering Cancer Center (MSKCC). Eligibility criteria included diagnosis of primary stage IA or IB NSCLC, surgical resection with curative intent, 1–6 years post-surgical treatment, no evidence of disease at time of recruitment, oncologist-granted permission for study participation, no severe psychiatric or cognitive impairment judged to interfere with participation, and ability to give informed consent.
Procedures
All potentially eligible participants (n =503) were identified from the database and mailed a consent form and letter inviting study participation. They were called 2 weeks later to confirm eligibility and interested participants provided verbal informed consent. All participants were offered print educational resources summarizing the availability of onsite psychosocial and rehabilitation services for cancer survivors. The study was approved by the Institutional Review Board. Previous papers detailing prevalence of fatigue [34], dyspnea [35], HRQOL [36], and health behaviors [37] have been published from this cohort. This manuscript builds on these previous papers by examining the burden of multiple symptoms in lung cancer survivors’ HRQOL and daily functioning.
Measures
Demographic variables
Participants reported their age, gender, race/ethnicity, education, marital/partnership status, employment status, and income.
Medical variables
The thoracic surgical database identified pathological disease stage, time since surgical resection, type of surgical resection, and use of video-assisted thoracoscopic surgery (VATS) (yes/no). Participants also reported on the presence of 21 comorbid medical conditions [38].
Health-related quality of life
The SF-36v2 [39] is a 36-item self-reported HRQOL scale, and it has been used in previous studies of lung cancer survivors [10]. Further rationale for the use of this measure in this population can be found in the work of Ostroff et al. [36]. The measure consists of two component factors: physical health (PCS) and mental health (MCS). Component scores demonstrated good internal consistency (PCS: α =0.93, MCS: α =0.95).
Performance status
The Self-Reported Karnofsky Performance Scale (SR-KPS) is a measure of current performance ability [15]. Participants indicate which statement best describes their current level of functioning. The modified response scale ranges from 1 (able to carry out normal activity with no physical complaints) to 7 (severely disabled with hospital admission required) [15].
Symptom burden
The following measures were chosen because of their empirically validated clinical cutoff points:
• Pain
The Brief Pain Inventory (BPI) [31] is a self-report measure of pain severity and interference, and it has been used in other studies of cancer survivors [7]. Pain severity scores were determined by averaging the mean of participants’ responses to items 1 (“pain at its worst in the last week”), 2 (“pain at its least in the last week”), and 3 (“pain on average in the last week”). Pain severity items were rated on an 11-point scale from 0 (“no pain”) to 10 (“pain as bad as you can imagine”). The BPI has been well-validated [31] and showed good internal consistency (α =0.95). Based on previous research, clinically significant pain was defined as a score of 4 or more [32].
• Fatigue
The Brief Fatigue Inventory (BFI) is a nine-item measure of clinically relevant fatigue [40]. The BFI evaluates fatigue “right now” and “during the past week” (worst and usual), as well as the severity and interference of fatigue with daily functioning during the past week. Fatigue is rated on a scale of 0 (“no fatigue”) to 10 (“severe fatigue”). A score of 4 or higher has been used previously as an indicator of clinically significant fatigue [40], and it was the cutoff score we used in this study. The BFI has been used with cancer patient populations [40] and evidenced good internal consistency in the current sample (α =0.94).
• Dyspnea
The Baseline Dyspnea Index (BDI) [41] is a self-report measure of current dyspnea severity along three dimensions: overall functional impairment, task magnitude, and effort magnitude. Dyspnea is graded on a five-point (0 to 4) scale from “very severe” to “no impairment.” Item responses are summed with lower scores indicating greater dyspnea. The BDI has been validated in patients undergoing lung surgery [42] and showed good internal consistency in the current sample (α =0.91). Based on previous research, clinically significant dyspnea was defined as a score of 9 or less [35].
• Anxiety and depression
The Hospital Anxiety and Depression Scale (HADS) [43] is a self-report measure of mood disturbance designed for use in medical populations. It consists of a seven-item anxiety scale (α =0.92) and a seven-item scale of depressive symptoms (α =0.87). Each item contains four response options (rated 0 to 3), and a summed score of 8 or above on either scale is indicative of clinically significant symptomatology [43].
Data analysis
Descriptive statistics were calculated for demographic and medical variables. Symptom measure scores were dichotomized (yes/no) based on cutoffs (see “Measures” section) for pain, fatigue, dyspnea, anxiety, and depression. The total number of symptoms (i.e., number of symptoms coded “yes”) classified the degree of symptom burden. Chi-squared tests, Kruskal–Wallis H tests, and Mann–Whitney U tests were chosen based on their ability to analyze ranked data and were used to examine demographic, medical, and HRQOL correlates of the degree of symptom burden. Receiver-operating characteristic (ROC) curves were used to determine the degree of symptom burden that corresponds most with functional impairment. A ROC curve is a plot of a test's true-positive rate (sensitivity) over the false-positive rate (1-specificity) measured at a cutoff value. The area under a ROC curve (AUC) is an estimate of the ability to accurately discriminate a patient identified as a “case” from a patient who does not meet case criteria [44]. The AUC can range from 0.5 (suggesting that the test offers no discrimination) to 1.0 (suggesting that the test offers perfect discrimination).
Results
Participants
Of the 503 NSCLC patients identified in the database, 222 did not meet the eligibility criteria. The most common reasons for exclusion were current malignancy (n =71), more than 6 years since resection (n =46), deceased (n =26), diagnosis of stage II–IV disease (n =25), and non-English speaking (n =17). Of the eligible patients, 19 were unable to be reached and 78 declined participation. The most common reasons for refusal included 23 lack of interest, 20 wished to avoid discussing cancer, and 10 felt too ill to participate. A total of 183 patients provided informed consent and participated in the study (65 % participation rate). There were no significant differences between participating and non-participating patients with regard to age, gender, time since surgical resection, or pathological disease stage (ps>.05). Data collection took place from September 2005 through July 2007.
Demographic and medical characteristics
Participants had a mean age of 69.0 (SD=9.9) years, were mainly female (63.9 %), non-Hispanic White (93.4 %), married/partnered (61.2 %), and had a college degree (47.0 %). They had a mean of 2.9 (SD=1.2) years since treatment and 2.4 (SD=1.6) comorbid diseases. VATS procedure was used in 15 % of the participants. See Tables 1 and 2 for additional data on demographic and medical characteristics.
Table 1.
Demographic characteristics of sample (N = 183)
| Characteristic | Number | Percent |
|---|---|---|
| Age (years) (M = 69.0, SD = 9.9) | ||
| <65 | 52 | 28.4 |
| 65–74 | 73 | 39.9 |
| 75+ | 58 | 31.7 |
| Gender | ||
| Female | 117 | 63.9 |
| Relationship | ||
| Married/partnered | 112 | 61.2 |
| Race/ethnicity | ||
| Non-Hispanic White | 171 | 93.4 |
| Non-Hispanic Black | 5 | 2.7 |
| Non-Hispanic other | 1 | 0.6 |
| Hispanic | 6 | 3.3 |
| Education | ||
| College degree | 86 | 47.0 |
| Employment status | ||
| Employed | 49 | 26.8 |
| Income | ||
| >$50,000 | 92 | |
| Missing (n) | (33) | 61.3 |
Table 2.
Medical characteristics of sample (N = 183)
| Characteristic | Number | Percent |
|---|---|---|
| Pathological disease stage | ||
| IA | 125 | 68.3 |
| IB | 58 | 31.7 |
| Time since surgical resection (years) (M = 2.9, SD = 1.2) | ||
| 1–<2 | 23 | 12.6 |
| 2–<3 | 54 | 29.5 |
| 3–<4 | 48 | 26.2 |
| 4–<5 | 41 | 22.4 |
| 5–<6 | 17 | 9.3 |
| Type of surgical resection | ||
| Wedge | 26 | 14.3 |
| Segmentectomy | 17 | 9.3 |
| Lobectomy | 133 | 73.1 |
| Biliobectomy | 4 | 2.2 |
| Pneumonectomy | 2 | 1.1 |
| Missing (n) | (1) | |
| Use of video-assisted thoracic surgery (VATS) | ||
| Yes | 28 | 15.5 |
| Missing (n) | (3) | |
| Number of self-reported comorbid medical conditions (M = 2.3, SD = 1.5) | ||
| 0 | 20 | 10.9 |
| 1 | 40 | 21.9 |
| 2 | 42 | 23.0 |
| 3 | 30 | 16.4 |
| 4 | 32 | 17.5 |
| ≥5 | 19 | 10.4 |
Prevalence and severity of symptom burden
The frequency of clinically significant symptoms reported among the sample was as follows: 57.9 % pain, 13.7 % fatigue, 57.9 % dyspnea, 21.9 % anxiety, and 8.2 % depression. Based on the number of symptoms present, participants were categorized into one of four degrees of symptom burden: 37 (20.2 %) had none, 56 (30.6 %) had one, 51 (27.9 %) had two, and 39 (21.3 %) had three or more concurrent symptoms. Patients with only one symptom most frequently reported dyspnea (44.6 %) or pain (41.1 %). Those with two symptoms most frequently reported dyspnea and pain (82.4 %). Among those with three or more symptoms, dyspnea, pain, and fatigue were the most common symptom cluster (41.0 %).
Demographic and medical correlates of symptom burden
Variables that were significantly associated (p <.05) with higher symptom burden (two or more concurrent symptoms) were not married/partnered (χ2=5.19), lower education (χ2=3.75), retired or unemployed (χ2=5.83), lower income (χ2=7.93), multiple comorbidities (χ2=7.58), and not having the VATS procedure (χ2=6.46). Analysis on race/ethnicity was not conducted due to a lack of heterogeneity of participants. Age, gender, disease stage, type of resection, and time since surgical resection were not significantly associated with higher symptom burden (ps>.05).
HRQOL and symptom burden
A Kruskal–Wallis H test showed a significant overall group difference in physical health HRQOL by symptom burden (H(3)=72.29, p <0.01), with Mann–Whitney U tests revealing differences (ps<0.01) between each level of symptom burden (no symptoms: M =55.61, SD=4.65; one symptom: M =50.76, SD=8.28; two symptoms: M =46.36, SD=7.64; three or more symptoms: M =33.65, SD=12.14). A Kruskal–Wallis H test also showed a significant overall group difference in mental health HRQOL (H(3)=32.93, p <0.01), with Mann–Whitney U tests showing significant differences (ps<0.01) only between the highest level of symptom burden (three or more symptoms) and all other degrees of symptom burden (no symptoms: M =56.39, SD=7.82; one symptom: M =55.45, SD=7.19; two symptoms: M =54.36, SD=7.74; three or more symptoms: M =45.85, SD=10.37).
Symptom burden and performance status
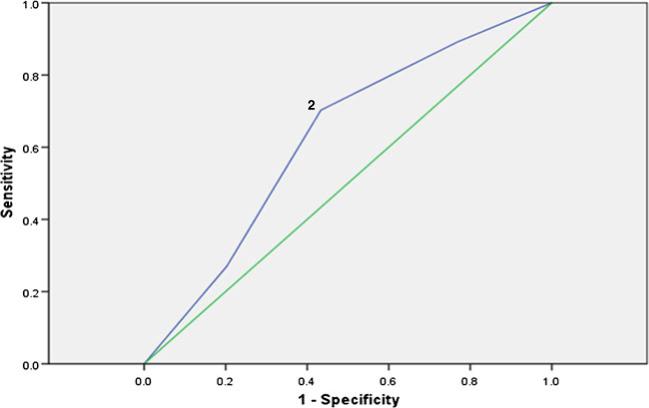
ROC curve analyses were used to examine the level of symptom burden that corresponded to limitations in functioning. Based on clinical experience and previous research [45], we used a SR-KPS cutoff score of 2 (“Able to carry on normal activity, minor symptoms of disease or side effects of treatment”) as an initial marker of impaired functioning (n =67, 36.6 %). Results showed symptom burden effectively discriminated between the patients with normal functioning and those who reported some level of impairment (AUC=0.63, SE=0.05, 95 % CI=0.53–0.72, non-parametric p <0.05). Visual inspection of the ROC curve (Fig. 1) and the sensitivity and specificity values suggested that experiencing two or more concurrent symptoms corresponded most to impairment in functioning (sensitivity=0.70, specificity=0.57).
Fig. 1.
ROC curve using a SR-KPS cutpoint of 1. ROC results (plotting sensitivity as a function of 1-specificity). Optimal cutoff values in the ROC curve for degree of symptom burden and functional limitation (using Self-Reported Karnofsky Performance Scale [SR-KPS]) in (n =183) NSCLC survivors. The area under the curve (AUC) was 0.63. The diagonal line corresponds to the ROC curve produced by random prediction
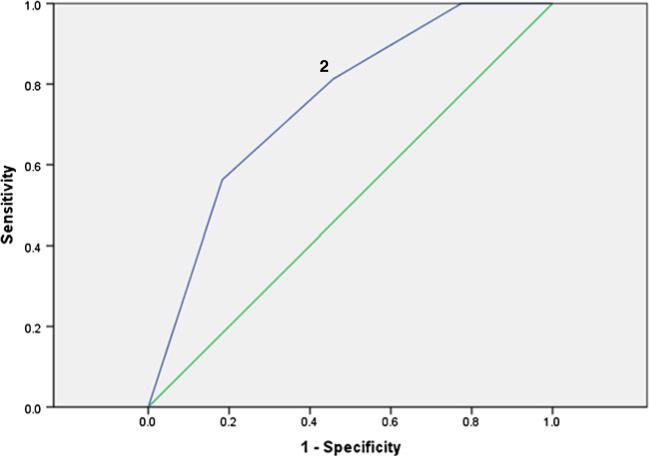
We then used a SR-KPS cutoff score of 3 (“Normal activity with effort, some symptoms of disease or side effects of treatment”), which indicates greater impairment in functioning (n =30, 16.4 %). Results showed improved accuracy and discrimination between patients with normal functioning and those with impaired functioning (AUC=0.75, SE=0.06, 95 % CI=0.64–0.87, non-parametric p <0.01). Visual inspection of the ROC curve (Fig. 2) and the sensitivity and specificity values again suggested that having two or more concurrent symptoms corresponded most to impairment in functioning (sensitivity=0.81, specificity=0.46).
Fig. 2.
ROC curve using a SR-KPS cutpoint of 2. ROC results (plotting sensitivity as a function of 1-specificity). Optimal cutoff values in the ROC curve for degree of symptom burden and functional limitation (using Self-Reported Karnofsky Performance Scale [SR-KPS]) in (n =183) NSCLC survivors. The area under the curve (AUC) was 0.75. The diagonal line corresponds to the ROC curve produced by random prediction
Discussion
To our knowledge, this study is the first to examine and report the tipping point at which symptom burden (symptom intensity and number) specifically affect functioning and quality of life in a sample of NSCLC survivors 1–6 years post-surgical resection. This paper adds to the literature by examining these relationships in a longer-term survivor population where, years out from surgical resection, symptoms are likely more chronic. We found that survivors reporting two or more clinically significant symptoms experienced impaired functioning and quality of life. Further, we found that nearly half of participants experienced multiple concurrent symptoms, most frequently dyspnea and pain. This is consistent with Cheville et al. who found high prevalence of symptom clusters, even years after treatment, despite no evidence of active cancer [6].
Our research adds to the existing literature showing that the decline in HRQOL associated with increased symptoms [4, 29, 30] may reflect a physical, more than an emotional, aspect of HRQOL in the lives of lung cancer survivors. For example, previous studies [3, 9, 27] have found pain to be highly correlated with depression and anxiety in cancer patient samples but may not apply to longer-term survivors, who may have learned to cope with or adapt to pain. Such an explanation is consistent with our finding that symptom burden was more strongly associated with physical HRQOL, but mental HRQOL was only associated when symptom burden was at the highest level. In addition, symptom burden was most associated with functional impairment when experienced as two or more concurrent symptoms [16]. This highlights the importance of assessing not only presence and severity but also the functional impact of multiple concurrent symptoms in lung cancer survivors.
Our results indicate that a subgroup of lung cancer survivors, namely, those who are single, not working, have lower education, lower income, and multiple comorbidities, may be at greater risk of higher symptom burden and thereby, has more limitations in daily functioning. The VATS approach was also associated with lower symptom burden and warrants further research. Targeting thoracotomy patients as an at-risk population for greater intervention may be indicated to address long-term symptoms. A comprehensive symptom assessment and management program may be particularly beneficial for these long-term post-treatment individuals, where symptoms are less likely to fluctuate and improve over time.
Study strengths and limitations
Specific strengths of this study include a good participation rate (65 %) and no indication of sample bias. In addition, well-validated symptom measures were used with empirically established cutoff scores representing clinically significant symptoms. Such brief standardized measures can be readily used in clinical practice to evaluate the presence and severity of concurrent symptoms and the extent of functional impairment.
There are limitations with regard to the generalizability of findings, given that participants were all survivors of stage I NSCLC came from a single cancer institution and were mostly non-Hispanic White and well-educated. Only 2.7 % of the participants were non-Hispanic Black, a group in which lung cancer rates are the highest and escalating [1]. Future studies should examine symptom burden and performance status among a more heterogeneous sample of NSCLC survivors. Although this study assessed common symptoms of NSCLC, it did not include other symptoms, such as sleep problems and cognitive impairment [10, 19, 33]. Studies using more detailed assessments of performance status are needed to determine the extent of impairment in daily functioning. In addition, although time since resection was not found to be significantly related to symptom burden, further examination of the role of comorbidities should be explored. Comparison against a non-cancer control cohort with similar comorbidities would help establish to what extent symptoms are related to cancer diagnosis and treatment. Finally, the cross-sectional research design prohibits the examination of a causal relationship between symptom burden and HRQOL or performance variables. Longitudinal studies can offer additional insights by monitoring symptom burden over time. Future studies using longitudinal design to assess symptom burden, HRQOL, and follow-up can perhaps identify multi-symptom burden earlier, enabling earlier intervention, which may improve HRQOL and functioning over subsequent years.
Conclusions and clinical implications
Results of this study suggest multiple symptoms are often experienced concurrently in early-stage, long-term lung cancer survivors. This implies that a broad array of symptoms should be assessed rather than focusing only on the most prominent ones that patients self-report. The number of symptoms, rather than any particular type, appears to impact HRQOL and functioning. The high prevalence and impact of moderate to high symptom burden represent a target for improved clinical care. Long-term survivors with multiple concurrent symptoms may be particularly vulnerable and should be targeted for more intensive symptom management interventions. Survivors with less education, those not working, and those with multiple medical comorbidities may be most at risk for experiencing higher symptom burden in survivorship.
These findings highlight the importance of incorporating multi-focal symptom assessment into routine clinical practice. Effective interventions need to be developed for survivors to prevent and treat symptom clusters. The findings of the current study also underscore the critical need for ongoing survivorship care, as recommended by the Institute of Medicine [28]. An interdisciplinary, collaborative approach is needed to detect, monitor, and treat not only complex and multiple physical symptoms but also the psychosocial needs of survivors. Symptom management remains an important target of intervention to promote optimal functioning and HRQOL among lung cancer survivors.
Acknowledgments
This project was supported in part by the Byrne Foundation (study design and data collection), grant T32CA009461 from the National Cancer Institute (data analysis, interpretation, and presentation), and P30 CA08748 (Behavioral Research Methods Core). We thank Melissa Ozim and Syncia Sabain for their assistance with data collection and management, and the study participants for their valued contribution.
Footnotes
Conflict of interest Funding sponsors were not involved in the conduct of the study or development of the submission. Manuscript authors had full control of all primary data and would be happy to comply with allowing the journal to review study data if requested. Preliminary findings were presented as a poster at the 33rd Annual Society for Behavioral Medicine Conference held in New Orleans, LA, USA from 11–14 April 2012.
Contributor Information
Amy E. Lowery, School of Medicine, University of Pittsburgh, 5115 Centre Ave, Suite 140, Pittsburgh, PA, USA Department of Psychiatry & Behavioral Sciences, Memorial Sloan-Kettering Cancer Center, New York, NY, USA.
Paul Krebs, Department of Population Health, NYU Langone Medical Center, New York, NY, USA; VA New York Harbor Healthcare System, New York, NY, USA.
Elliot J. Coups, Rutgers Cancer Institute of New Jersey, New Brunswick, NJ, USA Department of Medicine, Rutgers Robert Wood Johnson Medical School, New Brunswick, NJ, USA; Department of Health Education and Behavioral Science, Rutgers School of Public Health, Piscataway, NJ, USA; Rutgers, The State University of New Jersey, New Brunswick, NJ, USA.
Marc B. Feinstein, Department of Medicine, Memorial Sloan-Kettering Cancer Center, New York, NY, USA
Jack E. Burkhalter, Department of Psychiatry & Behavioral Sciences, Memorial Sloan-Kettering Cancer Center, New York, NY, USA
Bernard J. Park, Department of Thoracic Surgery, Hackensack University Medical Center, Hackensack, NJ, USA
Jamie S. Ostroff, Department of Psychiatry & Behavioral Sciences, Memorial Sloan-Kettering Cancer Center, New York, NY, USA
References
- 1.American Cancer Society . Cancer facts & figures 2012. American Cancer Society; Atlanta: 2012. [Google Scholar]
- 2.Hewitt M, Rowland JH, Yancik R. Cancer survivors in the United States: age, health, and disability. J Gerontol A: Biol Med Sci. 2003;58:82–91. doi: 10.1093/gerona/58.1.m82. [DOI] [PubMed] [Google Scholar]
- 3.National Institute of Health NIH State-of-the-Science Statement on symptom management in cancer: pain, depression, and fatigue. NIH Consens State Sci Statements. 2002;19(4):1–29. [PubMed] [Google Scholar]
- 4.Shi Q, Smith TG, Michonski JD, Stein KD, Kaw C, Cleeland CS. Symptom burden in cancer survivors 1 year after diagnosis: a report from the American Cancer Society's Studies of Cancer Survivors. Cancer. 2011;117:2779–2790. doi: 10.1002/cncr.26146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burkett VS, Cleeland CS. Symptom burden in cancer survivorship. J Cancer Survivorship Res Pract. 2007;1:167–175. doi: 10.1007/s11764-007-0017-y. [DOI] [PubMed] [Google Scholar]
- 6.Cheville AL, Novotny PJ, Sloan JA, et al. Fatigue, dyspnea, and cough comprise a persistent symptom cluster up to five years after diagnosis with lung cancer. J Pain Symptom Manag. 2011;42:202–212. doi: 10.1016/j.jpainsymman.2010.10.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eversley R, Estrin D, Dibble S, Wardlaw L, Pedrosa M, Favila-Penney W. Post-treatment symptoms among ethnic minority breast cancer survivors. Oncol Nurs Forum. 2005;32:250–256. doi: 10.1188/05.ONF.250-256. [DOI] [PubMed] [Google Scholar]
- 8.Fu OS, Crew KD, Jacobson JS, et al. Ethnicity and persistent symptom burden in breast cancer survivors. J Cancer Survivorship Res Pract. 2009;3:241–250. doi: 10.1007/s11764-009-0100-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harrington CB, Hansen JA, Moskowitz M, Todd BL, Feuerstein M. It's not over when it's over: long-term symptoms in cancer survivors–a systematic review. Int J Psychiatry Med. 2010;40:163–181. doi: 10.2190/PM.40.2.c. [DOI] [PubMed] [Google Scholar]
- 10.Sarna L, Padilla G, Holmes C, Tashkin D, Brecht ML, Evangelista L. Quality of life of long-term survivors of non-small-cell lung cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2002;20:2920–2929. doi: 10.1200/JCO.2002.09.045. [DOI] [PubMed] [Google Scholar]
- 11.Svobodnik A, Yang P, Novotny PJ, et al. Quality of life in 650 lung cancer survivors 6 months to 4 years after diagnosis. Mayo Clin Proc Mayo Clin. 2004;79:1024–1030. doi: 10.4065/79.8.1024. [DOI] [PubMed] [Google Scholar]
- 12.Cheville AL, Novotny PJ, Sloan JA, et al. The value of a symptom cluster of fatigue, dyspnea, and cough in predicting clinical outcomes in lung cancer survivors. J Pain Symptom Manag. 2011;42:213–221. doi: 10.1016/j.jpainsymman.2010.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cleeland CS. Symptom burden: multiple symptoms and their impact as patient-reported outcomes. J Natl Cancer Inst Monogr. 2007;37:16–21. doi: 10.1093/jncimonographs/lgm005. [DOI] [PubMed] [Google Scholar]
- 14.Cooley ME, Short TH, Moriarty HJ. Patterns of symptom distress in adults receiving treatment for lung cancer. J Palliat Care. 2002;18:150–159. [PubMed] [Google Scholar]
- 15.Dodd MJ, Cho MH, Cooper BA, Miaskowski C. The effect of symptom clusters on functional status and quality of life in women with breast cancer. Eur J Oncol Nurs Off J Eur Oncol Nurs Soc. 2010;14:101–110. doi: 10.1016/j.ejon.2009.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dodd MJ, Miaskowski C, Paul SM. Symptom clusters and their effect on the functional status of patients with cancer. Oncol Nurs Forum. 2001;28:465–470. [PubMed] [Google Scholar]
- 17.Esther Kim JE, Dodd MJ, Aouizerat BE, Jahan T, Miaskowski C. A review of the prevalence and impact of multiple symptoms in oncology patients. J Pain Symptom Manag. 2009;37:715–736. doi: 10.1016/j.jpainsymman.2008.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ferreira KA, Kimura M, Teixeira MJ, et al. Impact of cancer-related symptom synergisms on health-related quality of life and performance status. J Pain Symptom Manag. 2008;35:604–616. doi: 10.1016/j.jpainsymman.2007.07.010. [DOI] [PubMed] [Google Scholar]
- 19.Gift AG, Stommel M, Jablonski A, Given W. A cluster of symptoms over time in patients with lung cancer. Nurs Res. 2003;52:393–400. doi: 10.1097/00006199-200311000-00007. [DOI] [PubMed] [Google Scholar]
- 20.Henry DH, Viswanathan HN, Elkin EP, Traina S, Wade S, Cella D. Symptoms and treatment burden associated with cancer treatment: results from a cross-sectional national survey in the U.S. Support Care Cancer Off J Multinatl Assoc Support Care Cancer. 2008;16:791–801. doi: 10.1007/s00520-007-0380-2. [DOI] [PubMed] [Google Scholar]
- 21.Jones JM, Cohen SR, Zimmermann C, Rodin G. Quality of life and symptom burden in cancer patients admitted to an acute palliative care unit. J Palliat Care. 2010;26:94–102. [PubMed] [Google Scholar]
- 22.Kirkova J, Aktas A, Walsh D, Davis MP. Cancer symptom clusters: clinical and research methodology. J Palliat Med. 2011;14:1149–1166. doi: 10.1089/jpm.2010.0507. [DOI] [PubMed] [Google Scholar]
- 23.Miaskowski C, Cooper BA, Paul SM, et al. Subgroups of patients with cancer with different symptom experiences and quality-of-life outcomes: a cluster analysis. Oncol Nurs Forum. 2006;33:E79–E89. doi: 10.1188/06.ONF.E79-E89. [DOI] [PubMed] [Google Scholar]
- 24.Molassiotis A, Wengstrom Y, Kearney N. Symptom cluster patterns during the first year after diagnosis with cancer. J Pain Symptom Manag. 2010;39:847–858. doi: 10.1016/j.jpainsymman.2009.09.012. [DOI] [PubMed] [Google Scholar]
- 25.Rausch SM, Clark MM, Patten C, et al. Relationship between cytokine gene single nucleotide polymorphisms and symptom burden and quality of life in lung cancer survivors. Cancer. 2010;116:4103–4113. doi: 10.1002/cncr.25255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang XS, Shi Q, Williams LA, et al. Inflammatory cytokines are associated with the development of symptom burden in patients with NSCLC undergoing concurrent chemoradiation therapy. Brain Behav Immun. 2010;24:968–974. doi: 10.1016/j.bbi.2010.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Buchanan D, Milroy R, Baker L, Thompson AM, Levack PA. Perceptions of anxiety in lung cancer patients and their support network. Support Care Cancer Off J Multinatl Assoc Support Care Cancer. 2009;18:29–36. doi: 10.1007/s00520-009-0626-2. [DOI] [PubMed] [Google Scholar]
- 28.Institute of Medicine . From cancer patient to cancer survivor: lost in transition. The National Academies; Washington DC: 2005. [Google Scholar]
- 29.Portenoy RK, Thaler HT, Kornblith AB, et al. Symptom prevalence, characteristics and distress in a cancer population. Qual Life Res Int J Qual Life Asp Treat Care Rehab. 1994;3:183–189. doi: 10.1007/BF00435383. [DOI] [PubMed] [Google Scholar]
- 30.Sun V, Ferrell B, Juarez G, Wagman LD, Yen Y, Chung V. Symptom concerns and quality of life in hepatobiliary cancers. Oncol Nurs Forum. 2008;35:E45–E52. doi: 10.1188/08.ONF.E45-E52. [DOI] [PubMed] [Google Scholar]
- 31.Cleeland CS, Ryan KM. Pain assessment: global use of the Brief Pain Inventory. Ann Acad Med. 1994;23:129–138. [PubMed] [Google Scholar]
- 32.Serlin RC, Mendoza TR, Nakamura Y, Edwards KR, Cleeland CS. When is cancer pain mild, moderate or severe? Grading pain severity by its interference with function. Pain. 1995;61:277–284. doi: 10.1016/0304-3959(94)00178-H. [DOI] [PubMed] [Google Scholar]
- 33.Temel JS, Pirl WF, Lynch TJ. Comprehensive symptom management in patients with advanced-stage non-small-cell lung cancer. Clin Lung Cancer. 2006;7:241–249. doi: 10.3816/CLC.2006.n.001. [DOI] [PubMed] [Google Scholar]
- 34.Hung R, Krebs P, Coups EJ, et al. Fatigue and functional impairment in early-stage non-small cell lung cancer survivors. J Pain Symptom Manag. 2011;41:426–435. doi: 10.1016/j.jpainsymman.2010.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Feinstein MB, Krebs P, Coups EJ, et al. Current dyspnea among long-term survivors of early-stage non-small cell lung cancer. J Thorac Oncol Off Publ Int Assoc Study Lung Cancer. 2010;5:1221–1226. doi: 10.1097/JTO.0b013e3181df61c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ostroff JS, Krebs P, Coups EJ, et al. Health-related quality of life among early-stage, non-small cell, lung cancer survivors. Lung Cancer. 2011;71:103–108. doi: 10.1016/j.lungcan.2010.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Krebs P, Coups EJ, Feinstein MB, et al. Health behaviors of early-stage non-small cell lung cancer survivors. J Cancer Survivor-ship Res Pract. 2012;6:37–44. doi: 10.1007/s11764-011-0191-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Buist AS, Connett JE, Miller RD, Kanner RE, Owens GR, Voelker HT. Chronic obstructive pulmonary disease early intervention trial (lung health study). Baseline characteristics of randomized participants. Chest. 1993;103:1863–1872. doi: 10.1378/chest.103.6.1863. [DOI] [PubMed] [Google Scholar]
- 39.Ware JJ, Kosinski M, Gandek B. SF-36 health survey: manual and interpretation guide. QualityMetric, Inc.; Lincoln: 2000. [Google Scholar]
- 40.Mendoza TR, Wang XS, Cleeland CS, et al. The rapid assessment of fatigue severity in cancer patients: use of the Brief Fatigue Inventory. Cancer. 1999;85:1186–1196. doi: 10.1002/(sici)1097-0142(19990301)85:5<1186::aid-cncr24>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 41.Mahler DA, Weinberg DH, Wells CK, Feinstein AR. The measurement of dyspnea. Contents, interobserver agreement, and physiologic correlates of two new clinical indexes. Chest. 1984;85:751–758. doi: 10.1378/chest.85.6.751. [DOI] [PubMed] [Google Scholar]
- 42.Eakin EG, Sassi-Dambron DE, Ries AL, Kaplan RM. Reliability and validity of dyspnea measures in patients with obstructive lung disease. Int J Behav Med. 1995;2:118–134. doi: 10.1207/s15327558ijbm0202_3. [DOI] [PubMed] [Google Scholar]
- 43.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 44.Zweig MH, Campbell G. Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem. 1993;39:561–577. [PubMed] [Google Scholar]
- 45.Cella D, Nichol MB, Eton D, Nelson JB, Mulani P. Estimating clinically meaningful changes for the Functional Assessment of Cancer Therapy–Prostate: results from a clinical trial of patients with metastatic hormone-refractory prostate cancer. Value Health J Int Soc Pharmacoecon Outcomes Res. 2009;12:124–129. doi: 10.1111/j.1524-4733.2008.00409.x. [DOI] [PubMed] [Google Scholar]