Abstract
Psoralen-conjugated triple-helix-forming oligonucleotides have been used to generate site-specific mutations within mammalian cells. To investigate factors influencing the efficiency of oligonucleotide-mediated gene targeting, the processing of third-strand-directed psoralen adducts was compared in normal and repair-deficient human cells. An unusually high mutation frequency and an altered mutation pattern were seen in xeroderma pigmentosum variant (XPV) cells compared with normal, xeroderma pigmentosum group A (XPA), and Fanconi anemia cells. In XPV, targeted mutations were produced in the supF reporter gene carried in a simian virus 40 vector at a frequency of 30%, 3-fold above that in normal or Fanconi anemia cells and 6-fold above that in XPA. The mutations generated by targeted psoralen crosslinks and monoadducts in the XPV cells formed a pattern distinct from that in the other three cell lines, with mutations occurring not just at the damaged site but also at adjacent base pairs. Hence, the XPV cells may have an abnormality in trans-lesion bypass synthesis during repair and/or replication, implicating a DNA polymerase or an accessory factor as a basis of the defect in XPV. These results may help to elucidate the repair deficiency in XPV, and they raise the possibility that genetic manipulation via triplex-targeted mutagenesis may be enhanced by modulation of the XPV-associated activity in normal cells.
Full text
PDF



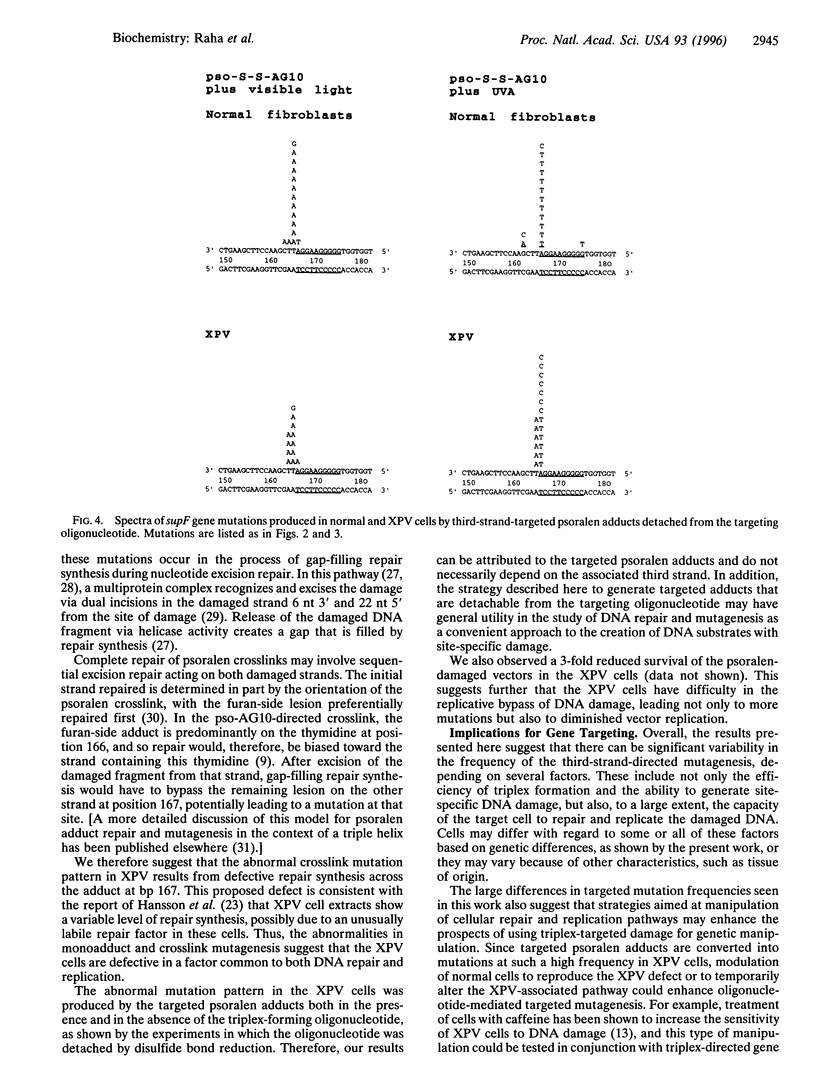

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beal P. A., Dervan P. B. Second structural motif for recognition of DNA by oligonucleotide-directed triple-helix formation. Science. 1991 Mar 15;251(4999):1360–1363. doi: 10.1126/science.2003222. [DOI] [PubMed] [Google Scholar]
- Boyer J. C., Kaufmann W. K., Brylawski B. P., Cordeiro-Stone M. Defective postreplication repair in xeroderma pigmentosum variant fibroblasts. Cancer Res. 1990 May 1;50(9):2593–2598. [PubMed] [Google Scholar]
- Cooney M., Czernuszewicz G., Postel E. H., Flint S. J., Hogan M. E. Site-specific oligonucleotide binding represses transcription of the human c-myc gene in vitro. Science. 1988 Jul 22;241(4864):456–459. doi: 10.1126/science.3293213. [DOI] [PubMed] [Google Scholar]
- Diatloff-Zito C., Papadopoulo D., Averbeck D., Moustacchi E. Abnormal response to DNA crosslinking agents of Fanconi anemia fibroblasts can be corrected by transfection with normal human DNA. Proc Natl Acad Sci U S A. 1986 Sep;83(18):7034–7038. doi: 10.1073/pnas.83.18.7034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasparro F. P., Havre P. A., Olack G. A., Gunther E. J., Glazer P. M. Site-specific targeting of psoralen photoadducts with a triple helix-forming oligonucleotide: characterization of psoralen monoadduct and crosslink formation. Nucleic Acids Res. 1994 Jul 25;22(14):2845–2852. doi: 10.1093/nar/22.14.2845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths T. D., Ling S. Y. Effect of UV light on DNA chain growth and replicon initiation in xeroderma pigmentosum variant cells. Mutagenesis. 1991 Jul;6(4):247–251. doi: 10.1093/mutage/6.4.247. [DOI] [PubMed] [Google Scholar]
- Hansson J., Keyse S. M., Lindahl T., Wood R. D. DNA excision repair in cell extracts from human cell lines exhibiting hypersensitivity to DNA-damaging agents. Cancer Res. 1991 Jul 1;51(13):3384–3390. [PubMed] [Google Scholar]
- Havre P. A., Glazer P. M. Targeted mutagenesis of simian virus 40 DNA mediated by a triple helix-forming oligonucleotide. J Virol. 1993 Dec;67(12):7324–7331. doi: 10.1128/jvi.67.12.7324-7331.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Havre P. A., Gunther E. J., Gasparro F. P., Glazer P. M. Targeted mutagenesis of DNA using triple helix-forming oligonucleotides linked to psoralen. Proc Natl Acad Sci U S A. 1993 Aug 15;90(16):7879–7883. doi: 10.1073/pnas.90.16.7879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J. C., Svoboda D. L., Reardon J. T., Sancar A. Human nucleotide excision nuclease removes thymine dimers from DNA by incising the 22nd phosphodiester bond 5' and the 6th phosphodiester bond 3' to the photodimer. Proc Natl Acad Sci U S A. 1992 Apr 15;89(8):3664–3668. doi: 10.1073/pnas.89.8.3664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hélène C. The anti-gene strategy: control of gene expression by triplex-forming-oligonucleotides. Anticancer Drug Des. 1991 Dec;6(6):569–584. [PubMed] [Google Scholar]
- Jones C. J., Wood R. D. Preferential binding of the xeroderma pigmentosum group A complementing protein to damaged DNA. Biochemistry. 1993 Nov 16;32(45):12096–12104. doi: 10.1021/bi00096a021. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A. Misalignment-mediated DNA synthesis errors. Biochemistry. 1990 Sep 4;29(35):8003–8011. doi: 10.1021/bi00487a001. [DOI] [PubMed] [Google Scholar]
- Laquerbe A., Moustacchi E., Fuscoe J. C., Papadopoulo D. The molecular mechanism underlying formation of deletions in Fanconi anemia cells may involve a site-specific recombination. Proc Natl Acad Sci U S A. 1995 Jan 31;92(3):831–835. doi: 10.1073/pnas.92.3.831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmann A. R., Kirk-Bell S., Arlett C. F., Paterson M. C., Lohman P. H., de Weerd-Kastelein E. A., Bootsma D. Xeroderma pigmentosum cells with normal levels of excision repair have a defect in DNA synthesis after UV-irradiation. Proc Natl Acad Sci U S A. 1975 Jan;72(1):219–223. doi: 10.1073/pnas.72.1.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letai A. G., Palladino M. A., Fromm E., Rizzo V., Fresco J. R. Specificity in formation of triple-stranded nucleic acid helical complexes: studies with agarose-linked polyribonucleotide affinity columns. Biochemistry. 1988 Dec 27;27(26):9108–9112. doi: 10.1021/bi00426a007. [DOI] [PubMed] [Google Scholar]
- Misra R. R., Vos J. M. Defective replication of psoralen adducts detected at the gene-specific level in xeroderma pigmentosum variant cells. Mol Cell Biol. 1993 Feb;13(2):1002–1012. doi: 10.1128/mcb.13.2.1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moser H. E., Dervan P. B. Sequence-specific cleavage of double helical DNA by triple helix formation. Science. 1987 Oct 30;238(4827):645–650. doi: 10.1126/science.3118463. [DOI] [PubMed] [Google Scholar]
- Park C. H., Sancar A. Formation of a ternary complex by human XPA, ERCC1, and ERCC4(XPF) excision repair proteins. Proc Natl Acad Sci U S A. 1994 May 24;91(11):5017–5021. doi: 10.1073/pnas.91.11.5017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parris C. N., Seidman M. M. A signature element distinguishes sibling and independent mutations in a shuttle vector plasmid. Gene. 1992 Aug 1;117(1):1–5. doi: 10.1016/0378-1119(92)90482-5. [DOI] [PubMed] [Google Scholar]
- Praseuth D., Perrouault L., Le Doan T., Chassignol M., Thuong N., Hélène C. Sequence-specific binding and photocrosslinking of alpha and beta oligodeoxynucleotides to the major groove of DNA via triple-helix formation. Proc Natl Acad Sci U S A. 1988 Mar;85(5):1349–1353. doi: 10.1073/pnas.85.5.1349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancar A., Tang M. S. Nucleotide excision repair. Photochem Photobiol. 1993 May;57(5):905–921. doi: 10.1111/j.1751-1097.1993.tb09233.x. [DOI] [PubMed] [Google Scholar]
- Shivji K. K., Kenny M. K., Wood R. D. Proliferating cell nuclear antigen is required for DNA excision repair. Cell. 1992 Apr 17;69(2):367–374. doi: 10.1016/0092-8674(92)90416-a. [DOI] [PubMed] [Google Scholar]
- Strobel S. A., Doucette-Stamm L. A., Riba L., Housman D. E., Dervan P. B. Site-specific cleavage of human chromosome 4 mediated by triple-helix formation. Science. 1991 Dec 13;254(5038):1639–1642. doi: 10.1126/science.1836279. [DOI] [PubMed] [Google Scholar]
- Wang G., Glazer P. M. Altered repair of targeted psoralen photoadducts in the context of an oligonucleotide-mediated triple helix. J Biol Chem. 1995 Sep 22;270(38):22595–22601. doi: 10.1074/jbc.270.38.22595. [DOI] [PubMed] [Google Scholar]
- Wang G., Levy D. D., Seidman M. M., Glazer P. M. Targeted mutagenesis in mammalian cells mediated by intracellular triple helix formation. Mol Cell Biol. 1995 Mar;15(3):1759–1768. doi: 10.1128/mcb.15.3.1759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y. C., Maher V. M., McCormick J. J. Xeroderma pigmentosum variant cells are less likely than normal cells to incorporate dAMP opposite photoproducts during replication of UV-irradiated plasmids. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7810–7814. doi: 10.1073/pnas.88.17.7810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y. C., Maher V. M., Mitchell D. L., McCormick J. J. Evidence from mutation spectra that the UV hypermutability of xeroderma pigmentosum variant cells reflects abnormal, error-prone replication on a template containing photoproducts. Mol Cell Biol. 1993 Jul;13(7):4276–4283. doi: 10.1128/mcb.13.7.4276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waters H. L., Seetharam S., Seidman M. M., Kraemer K. H. Ultraviolet hypermutability of a shuttle vector propagated in xeroderma pigmentosum variant cells. J Invest Dermatol. 1993 Nov;101(5):744–748. doi: 10.1111/1523-1747.ep12371686. [DOI] [PubMed] [Google Scholar]
- Wood R. D., Aboussekhra A., Biggerstaff M., Jones C. J., O'Donovan A., Shivji M. K., Szymkowski D. E. Nucleotide excision repair of DNA by mammalian cell extracts and purified proteins. Cold Spring Harb Symp Quant Biol. 1993;58:625–632. doi: 10.1101/sqb.1993.058.01.069. [DOI] [PubMed] [Google Scholar]
- Wood R. D. Pyrimidine dimers are not the principal pre-mutagenic lesions induced in lambda phage DNA by ultraviolet light. J Mol Biol. 1985 Aug 20;184(4):577–585. doi: 10.1016/0022-2836(85)90304-3. [DOI] [PubMed] [Google Scholar]




