Abstract
OBJECTIVE
The aim of this study was to determine the childhood clinical predictors of early adult outcomes in pediatric-onset obsessive-compulsive disorder (OCD) and to assess whether dimensional subtypes of OCD and the presence of comorbid tic symptoms influence long-term outcomes.
METHODS
We conducted a longitudinal cohort study in which 45 of 62 eligible children with OCD were reassessed an average of 9 years later, in early adulthood. Main outcome measures included expert-rated, obsessive-compulsive (OC) symptom severity and time to remission of OC symptoms. Baseline clinical characteristics were evaluated in terms of their influence on OCD severity in adulthood and time to remission of OC symptoms.
RESULTS
Forty-four percent of subjects were determined to have subclinical OC symptoms at the follow-up evaluation. The absence of a comorbid tic disorder and the presence of prominent hoarding symptoms were associated with the persistence of OCD symptoms. Female gender, earlier age at childhood assessment, later age of OCD onset, more-severe childhood OCD symptoms, and comorbid oppositional defiant disorder also were associated with persistence of OCD symptoms into adulthood.
CONCLUSIONS
These results confirm that a significant proportion of treated children with OCD experience remission by adulthood. The presence of comorbid tics heralds a positive outcome, whereas primary hoarding symptoms are associated with persistent OCD.
Keywords: obsessive-compulsive disorder, longitudinal study, compulsive hoarding, tic disorder
The prevalence of obsessive-compulsive disorder (OCD) in epidemiological studies of pediatric populations ranges from 1% to 3%, a rate similar to that observed in adult populations. 1–3 The majority of OCD cases in adulthood arise after the teenage years, which suggests that many of the pediatric-onset cases of OCD may remit by adulthood. A meta-analysis of previous follow-up studies of pediatric-onset OCD suggested that as many as 40% to 59% of cases remit.4 However, many of the studies included in the previous meta-analysis were conducted before the widespread use of effective, evidence-based treatments for OCD, such as selective serotonin reuptake inhibitors (SSRIs) and cognitive-behavioral therapy, and before the introduction of validated clinical rating scales for OCD. In addition, follow-up intervals in most of the studies ranged from 1 to 5 years; therefore, the studies had limited ability to assess adult outcomes. To our knowledge, no previous studies have assessed outcomes after a sufficient period to determine early adult outcomes in OCD.
Pediatric OCD has been hypothesized to be a distinct developmental subtype of OCD.5 In contrast to adult-onset OCD, pediatric OCD is associated with a distinct prepubertal age of onset, male predominance, strong family history of OCD, and high rate of comorbid tic disorders and attention-deficit/hyperactivity disorder (ADHD).5 In previous prospective longitudinal studies of pediatric-onset OCD, poorer long-term outcomes were associated with earlier age of OCD onset, increased duration of illness before treatment, inpatient hospitalization because of OCD, comorbid tic disorder, and poor initial response to pharmacologic treatment.4,6
A recent meta-analysis of 21 factor-analysis studies involving >5000 subjects demonstrated a fairly robust 4-factor structure for OCD that was remarkably consistent across the lifespan.7 The 4 OCD factors were (1) symmetry factor, which contained symmetry obsessions and ordering, repeating, and counting compulsions; (2) forbidden thoughts factor, which contained aggression and sexual and religious obsessions; (3) cleaning factor, which contained contamination obsessions and cleaning compulsions; and (4) hoarding factor, which contained hoarding obsessions and compulsions. These OCD symptom dimensions have been associated with distinct patterns of comorbid psychiatric conditions, different patterns of heritability, and specific genetic polymorphisms, as well as distinct symptom-associated patterns of neural activity, as measured with functional MRI.8–11 Symptom dimensions have been associated with different responses to pharmacologic and non-pharmacologic treatments.12,13
The goals of this study were to determine childhood clinical predictors of early adult outcomes in pediatric-onset OCD and to determine whether specific obsessive-compulsive (OC) symptom dimensions are associated with outcomes of pediatric-onset OCD assessed in early adulthood. We specifically hypothesized that OCD symptom dimensions and comorbid tic disorders would be associated with persistence of OCD symptoms into early adulthood.
METHODS
Subjects
All participants were recruited through the Yale Tourette syndrome (TS)/OCD clinic. Eligible participants were required (1) to have a childhood diagnosis of OCD, (2) to participate in MRI or neuropsychological testing studies before 16 years of age (these were prerequisites for having a detailed clinical research evaluation at that time), and (3) to be >16 years of age at the time of the potential follow-up interview. Adulthood follow-up interviews took place, on average, 9 years after initial childhood evaluations. Exclusionary criteria in those earlier studies included a history of seizure, head trauma with loss of consciousness, ongoing or past substance abuse, or an IQ of <80. Parent written informed consent and subject assent were obtained at the childhood baseline assessment and subject informed consent at the follow-up evaluation. Compensation for participation was provided at both time points under the guidelines of the human investigations committee.
Of an eligible sample of 62 subjects evaluated at the baseline interview, 46 (74%) elected to participate. Reasons for nonparticipation included subject refusal to participate in a follow-up interview (n = 2) and inability to locate subjects (n = 14). At the follow-up interview, it was found that 1 participant had since been diagnosed as having Asperger disorder. He and his mother were reinterviewed by a trained psychiatrist, and it was determined that he likely had Asperger disorder rather than OCD in childhood; therefore, he was excluded from subsequent analyses because he was not representative of the population we were trying to study. Demographic data, as assessed at the childhood baseline interview, did not differ significantly between participants and nonparticipants, except for age at baseline evaluation (Table 1).
TABLE 1.
Demographic Comparison of Participants and Nonparticipants
| Participants | Nonparticipants | |
|---|---|---|
| N | 45 | 16 |
| Age at evaluation, mean ± SD, ya | 12.1 ± 2.0 | 9.9 ± 1.6 |
| Male, n, % | 34 (76) | 12 (75) |
| TS, n, % | 18 (40) | 11 (69) |
| ADHD, n, % | 18 (40) | 6 (38) |
| Children’s Y-BOCS score, mean ± SD | 11.6 ± 8.1 | 14.2 ± 9.8 |
| Yale Global Tic Severity Scale score, mean ± SD | 8.0 ± 10.8 | 14.2 ± 10.8 |
Participants differed significantly from nonparticipants only in having a significantly older age at baseline evaluation. This result is not surprising, given that the main reason for nonparticipation was our inability to locate subjects for follow-up assessment.
P < .001.
Interview Procedure at Childhood Baseline Assessment
Childhood baseline assessments occurred before 16 years of age and included current and worst-ever measures using the Children’s Yale-Brown Obsessive Compulsive Scale (Y-BOCS) and Yale Global Tic Severity Scale.14,15 The Schedule for Tourette and Other Behavioral Syndromes was used to survey other comorbid psychiatric illnesses. 16 Neuropsychiatric diagnoses were established through a best-estimate consensus procedure performed by 2 child psychiatrists after a review of all available materials.17 Available information typically included findings of a clinical interview performed by an attending physician in the TS/OCD clinic and a structured clinical interview performed by a trained research assistant. Participants were then classified, on the basis of these diagnoses, with respect to their diagnosis status (yes or no) for TS, chronic tic disorder (CTD), ADHD, major depressive disorder (depression), comorbid anxiety disorder besides OCD (anxiety), and oppositional defiant disorder (ODD). If a child received a diagnosis of social phobia, separation anxiety disorder, or generalized anxiety disorder, then he or she was considered to have a comorbid anxiety disorder. Participants and their families were also asked, at that time, about the age of onset of the OC symptoms.
Interview Procedure at Adulthood Follow-up Evaluation
Follow-up evaluations in early adulthood included current and worst-ever ratings on the Y-BOCS and Yale Global Tic Severity Scale.14,18 Screening for comorbid psychiatric conditions was conducted with the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Axis I Disorders, and a standardized medication history was obtained.19 In addition, an interview with a parent or a close cohabitating adult was performed when possible, to verify symptom severity. We asked all subjects who had experienced significant improvement the age at which their OC symptoms remitted, which was defined in the interviews as the “the age your OC symptoms improved substantially.”
Data Analyses
All statistical analyses were performed with SAS 9.1 (SAS Institute, Cary, NC). Our primary outcome variable for analysis was the time from the initial childhood assessment to remission of OC symptoms. A participant was considered to have experienced remission if the Y-BOCS score at the follow-up evaluation was <8. A Y-BOCS score of <8 was chosen as our criterion for remission because a score of 8 differentiates subclinical (Y-BOCS scores of 0 –7) from clinical (Y-BOCS scores of ≥8) OCD symptoms.17 The age of OCD remission was determined through patient self-report at the follow-up evaluation. Time to remission was calculated by subtracting the age of the baseline childhood assessment from the age of remission.
The Kaplan-Meier method (implemented with SAS Proc Lifetest) was used to plot the overall survival function of our data and to test whether our a priori childhood clinical variables influenced the overall survival curve. Subject data were stratified according to comorbid CTD and primary OCD symptom dimension (symmetry, cleaning, forbidden thoughts, or hoarding). The Wilcoxon test was used to compare survival curves, because this method places greater weight on the time points that have more observations. A strict Bonferroni correction was used to set our threshold for statistical significance at P < .025.
We then used the Cox proportional-hazard model of survival analysis (Proc Phreg command in SAS) to construct the best-fitting exploratory model for our data. In our model, we examined the following variables of interest: age of onset, OCD severity at baseline childhood assessment, age at baseline assessment, depression, anxiety, TS, CTD, ADHD, conduct disorder, ODD, gender, and worst-ever OCD severity. Our best-fitting model was built by using the following steps: (1) we identified all variables that were significantly associated with time to OCD remission in univariate analyses; (2) all significant univariate terms were entered into the same model, individual terms were identified on the basis of the least significant P value, and then terms were removed if they did not improve the fit of the model significantly, according to the log-likelihood test; and (3) all nonsignificant univariate predictors of time to remission were added back into the model, and they were kept if they improved the fit of the model, according to the log-likelihood test (with a threshold of P < .05). We then added the dimensional measures of OCD to the best-fitting model with traditional clinical data, to determine whether this approach improved the fit of the model. We measured dimensional OC symptoms as follows: (1) factor scores for OCD dimensions were computed in a previously published study,20 (2) OC symptoms in each dimension were rated as absent, present, or severe, and (3) participants were categorized as primarily having hoarding symptoms or not. These dimensional approaches were included in our final best-fitting model only if they improved the goodness of fit, according to the log-likelihood test.
In posthoc analysis, we compared baseline characteristics of subjects classified as remitters versus nonremitters in adulthood follow-up evaluations. All continuous variables were examined by using Student’s t test, and categorical variables were examined by using Fisher’s exact test.
RESULTS
Interview Times
Childhood baseline interviews took place at an average age of 12.1 ± 2.0 years (range: 8.0 –15.8 years). The average age for early adulthood follow-up evaluations was 21.1 ± 3.1 years (range: 16.0 –27.0 years). The average period between the initial and follow-up interviews was 9.0 ± 2.9 years.
Clinical Course of OC Symptoms
The average age of onset of OC symptoms was 8.0 ± 2.5 years (range: 4 –13 years). The average Y-BOCS score at the follow-up evaluation was 10.2 ± 8.6, compared with 25.6 ± 7.1 at the worst-ever assessment (age: 11.3 ± 2.4 years). Twenty subjects (44%) had minimal OC symptoms at the follow-up evaluation and were classified as having experienced remission (Children’s Y-BOCS scores of <8) (Table 2). Fourteen subjects (31%) had mild OC symptoms (Y-BOCS scores of 8 –15) at the follow-up evaluation, whereas 6 subjects (13%) had moderate OC symptoms (Y-BOCS scores of 16 –23) and 5 subjects (11%) had severe OC symptoms (Y-BOCS scores of 24–40).
TABLE 2.
Characteristics of Subjects With Remitted Versus Nonremitted OCD
| Remitted (N = 20) | Nonremitted (N = 24) | |
|---|---|---|
| Demographic characteristics | ||
| Age, mean ± SD, y | 12.1 ± 2.1 | 12.1 ± 2.1 |
| Male, n (%)a | 18 (90) | 16 (67) |
| Age of onset, mean ± SD, y | 7.6 ± 2.5 | 8.4 ± 2.6 |
| Symptoms | ||
| Duration of symptoms, mean ± SD, y | 4.5 ± 2.1 | 3.7 ± 3.5 |
| OCD severity score, mean ± SD | ||
| Childhood assessmentb | 8.4 ± 4.7 | 14.3 ± 9.4 |
| Worst-ever childhood evaluationa | 23.7 ± 8.4 | 27.8 ± 6.0 |
| Comorbid conditions in childhood, n (%) | ||
| TS | 10 (50) | 7 (29) |
| CTDb | 16 (80) | 10 (42) |
| ADHD | 10 (50) | 8 (33) |
| Depression | 6 (30) | 7 (29) |
| ODD | 6 (30) | 7 (29) |
| Comorbid conditions in adulthood, n (%) | ||
| Depression | 4 (20) | 9 (38) |
| Bipolar disorder | 0 (0) | 3 (13) |
| Psychotic disorder | 0 (0) | 1 (4) |
| Alcohol abuse/dependence | 5 (25) | 5 (21) |
| Substance abuse/dependence | 7 (35) | 7 (29) |
| Body dysmorphic disorder | 0 (0) | 2 (8) |
| Primary OCD symptom dimension | ||
| Cleaning | 7 (35) | 5 (21) |
| Forbidden thoughts | 6 (30) | 5 (21) |
| Symmetry | 6 (30) | 5 (21) |
| Hoardingb | 1 (5) | 9 (38) |
| Treatment, n (%) | ||
| Childhood baseline assessment | ||
| Cognitive behavioral therapy | 10 (50) | 11 (46) |
| SSRIs | 19 (95) | 22 (92) |
| Antipsychotic agents | 11 (55) | 12 (50) |
| α2-Adrenergic receptor agonists | 7 (35) | 6 (25) |
| Adulthood follow-up assessment | ||
| SSRIs | 11 (55) | 16 (67) |
| Antipsychotic agents | 3 (15) | 4 (17) |
| α2-Adrenergic receptor agonists | 1 (5) | 1 (4) |
Subjects with pediatric-onset OCD whose OC symptoms remitted in adulthood were more likely to have primary hoarding symptoms and less likely to have comorbid CTDs, compared with nonremitters.
P < .1.
P < .05.
Comorbid Conditions and Medication Use at Follow-up Evaluation
At the follow-up evaluation, 13 subjects (29%) had a history of major depressive disorder, and 2 (4%) met criteria for current major depression. One subject was diagnosed as having a psychotic disorder and another as having bipolar disorder during the follow-up interval. No subjects had bipolar disorder or psychotic symptoms at the childhood assessment.
At the baseline childhood evaluation, 41 subjects (89%) were taking SSRIs. At the follow-up evaluation, 27 (60%) patients continued to take SSRIs, whereas 14 patients (31%) had stopped using them. Only 4 participants (9%) had never used SSRIs. Two subjects (4%) were using α2- adrenergic receptor agonists at the adulthood follow-up evaluation, compared with 9 (20%) at the childhood baseline evaluation. Seven subjects (15%) were taking antipsychotic medications at the adulthood follow-up evaluation, compared with 22 subjects (48%) at the childhood evaluation. Subjects with remitted versus nonremitted OCD did not differ significantly in medication use at the baseline childhood evaluation or adulthood follow-up evaluation.
Kaplan-Meier Model for a Priori Predictors of Time to OCD Remission
Kaplan-Meier survival plots demonstrated reduced time to remission of OC symptoms for subjects with comorbid tic disorders ( ; P = .02) (Fig 1). Primary OCD symptom dimension also was associated with the likelihood of remission at trend levels ( ; P = .09) (Fig 2). This result seemed to be attributable primarily to a significantly decreased likelihood of remission for individuals with primary hoarding symptoms, compared with other children with OCD ( ; P = .02).
FIGURE 1.
Survival curves comparing patients with OCD with and without comorbid CTD (red curve indicates patients with comorbid CTD; black curve, patients without CTD; circles, censored observations). Proportions of patients with OCD who experienced remission are indicated on the y-axis, and time since childhood baseline assessment is displayed on the x-axis. Patients with OCD with comorbid CTD experienced decreased time to remission of OC symptoms ( ; P = .02).
FIGURE 2.
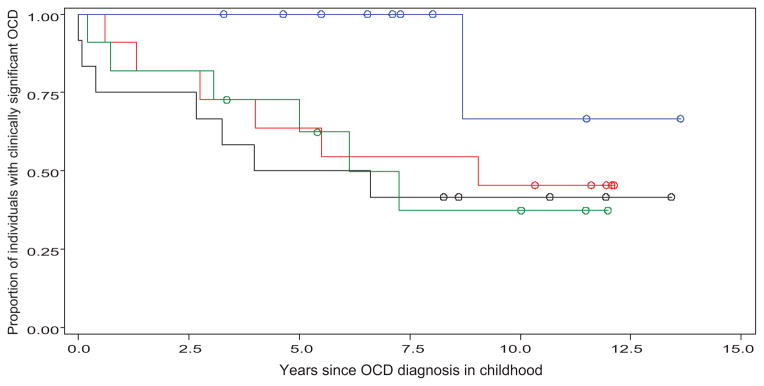
Survival curves comparing patients with OCD according to primary symptom dimension (red curve indicates symmetry; green curve, cleaning; black curve, forbidden thoughts; blue curve, hoarding; circles, censored observations). Proportions of patients with OCD who experienced remission are displayed on the y-axis, and time since childhood baseline assessment is displayed on the x-axis. OCD symptom dimensions were associated with differences in the likelihood of remission at trend levels ( ; P = .09). Primary hoarding symptoms were associated with a significantly decreased likelihood of remission ( ; P = .02), compared with nonhoarding OCD symptoms.
Cox Proportional-Hazard Model for Time to OCD Remission
Our best-fitting model, according to the likelihood ratio test, is depicted in Table 3. Female gender, absence of comorbid tic symptoms, more-severe initial OC symptoms, younger age at childhood assessment, older age of onset, comorbid ODD, and primary hoarding symptoms were associated with increased time to remission of OCD symptoms. Using patient factor scores for OCD symptom dimensions and classifying OCD severity in each dimension did not improve the overall fit of our model, compared with using the clinical variables alone (our best-fitting model without the primary hoarding symptom variable).
TABLE 3.
Cox Proportional-Hazard Model of Time to Remission of Pediatric-Onset OC Symptoms
| Parameter | Hazard Ratio (95% Confidence Interval) | P |
|---|---|---|
| Gender | 0.06 (0.02–0.15) | .002 |
| Childhood Children’s Y-BOCS score | 0.89 (0.85–0.94) | .018 |
| Age at baseline assessment | 2.00 (1.64–2.43) | <.001 |
| CTD | 4.50 (2.15–9.42) | .042 |
| ODD | 0.17 (0.08–0.35) | .014 |
| Age of OCD onset | 0.77 (0.70–0.84) | .004 |
| Primary hoarding symptoms | 0.09 (0.03–0.25) | .023 |
A lower hazard ratio indicates an increased time to remission of OC symptoms. Female gender, more-severe initial OC symptoms, younger age at initial assessment, later age of onset, comorbid ODD, absence of comorbid tic symptoms, and primary hoarding symptoms were associated with increased time to remission. Overall model fit statistics were as follows: likelihood ratio: ; P < .0001 (N = 45).
Comparison of Children With OCD Remission in Adulthood Versus No Remission
Table 2 compares the characteristics of children whose OC symptoms remitted during adulthood with the characteristics of children whose symptoms did not. Decreased OCD severity at childhood assessment, presence of a comorbid tic disorder, and absence of hoarding symptoms were associated with remission in adulthood. Male gender and decreased worst-ever childhood OCD severity were associated with OCD remission at trend levels.
DISCUSSION
Findings from this follow-up study of pediatric-onset OCD indicate that a significant proportion of treated children experience remission by early adulthood. The proportion of remitters in our study ranged from 44% (Y-BOCS scores of <8) to 58% (Y-BOCS scores of <10), depending on the criteria used for remission. These results are consistent with a meta-analysis of previous studies of shorter duration, which estimated rates of remission of 40% to 59%.4 In the present study, we were able to describe the life course of pediatric-onset OCD, in which children experience onset at ~8 years of age and experience their worst-ever symptoms just before initial treatment, at 11 years of age. These results are in striking contrast to long-term outcome studies with adults, in which rates of remission of OC symptoms according to such strict criteria are much lower.21
Children with OCD and comorbid CTD had a briefer time to remission of OCD symptoms, compared with subjects without comorbid tics. Sixty-two percent of subjects with pediatric-onset OCD and comorbid CTD experienced remission by adulthood, compared with 22% of those who did not have comorbid CTD (Table 2). Tics among patients with OCD were associated previously with comparable responses to cognitive behavioral therapy, poor short-term responses to sertraline treatment, 22 and improved responses to antipsychotic augmentation.12 Comorbid tics in children may indicate a developmental trajectory of OCD symptoms that mirrors that of tics in children with both disorders. Tic symptoms typically reach their worst-ever severity between 10 and 12 years of age and then improve during the course of adolescence.24,25 The developmental changes that are responsible for improving tics in children during adolescence also may help ameliorate comorbid OCD symptoms in these children.
Participants with primary hoarding symptoms seemed to have poorer long-term outcomes, compared with other children with OCD, with rates of remission of 10%, compared with 54% for children with other primary OC symptoms. Experts have suggested that the hoarding OC symptom dimension should be used as a specifier in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Indeed, a strong case can be made that compulsive hoarding in the absence of other OCD symptoms should be listed as a diagnostic entity separate from OCD.26,27 Genome-wide association studies have found an area on chromosome 14 that suggests linkage with compulsive hoarding but not other OC symptoms.28 Functional neuroimaging studies demonstrated distinct patterns of glucose metabolism and brain activation during symptom provocation paradigms when hoarding and nonhoarding patients with OCD were compared.29 Anatomic imaging studies demonstrated gray matter differences when compulsive hoarders were compared with other patients with OCD.30 Hoarding symptoms also have been associated with poor treatment responses. Short-term trials demonstrated poorer responses to cognitive-behavioral therapy and pharmacotherapy for patients with OCD with primary hoarding symptoms. 12,31–35 In children with OCD, hoarding symptoms are associated with poor insight, magical thinking, ordering and arranging compulsions, and higher levels of anxiety, aggression, somatic complaints, and overall externalizing and internalizing symptoms.36 These data all suggest that compulsive hoarders may have distinct psychopathologic features and diagnoses, compared with other individuals with OCD.
In multivariate, exploratory, survival analyses, we found an association of increased baseline OCD severity with longer time to remission. This association is not surprising, given that we used Y-BOCS scores of <8 as our criterion for remission. Individuals with more-severe OCD would need a greater degree of improvement to achieve this metric. In addition, we found that a later age of onset and younger age at evaluation were associated with increased time to remission. This finding is in contrast to previous long-term outcome studies associating early age of onset and longer illness duration with poorer outcomes in univariate analyses.4,6 The longer follow-up period, the larger proportion of children with comorbid tic disorders, and the use of multivariate analysis in our study may explain this difference. The association of decreased duration of treatment before assessment with greater persistence of OC symptoms suggests that greater acuity of onset of OC symptoms in children may portend poor outcomes. Another possible explanation is that unknown family and child factors could produce both greater treatment-seeking and poorer long-term prognoses. In our best-fitting model, comorbid ODD was associated with poor outcomes. Previous studies demonstrated that comorbid ODD is a predictor of poor responses to pharmacologic and behavioral therapies among children with OCD.37,38
Our study has several important limitations. We achieved a 74% participation rate for all eligible participants in the follow-up portion of our study. Nonparticipants might have differed systematically from those who did participate. The only discernible difference between participants and nonparticipants at the follow-up evaluation, however, was the significantly older age at baseline assessment of participants, compared with nonparticipants. This finding was not surprising, because our inability to locate eligible participants was largely responsible for individuals not participating. Participant recall bias or measurement error in the recall of age at remission also is a possibility. Prospective studies with assessments at regular intervals are needed to confirm our findings. In addition, multiple clinical and dimensional variables were tested in the multivariate Cox survival models, which thus have a higher probability of type I error. Therefore, our findings in these models should be regarded as generating rather than confirming hypotheses. Participants were treated clinically with both pharmacologic and behavioral therapies throughout the follow-up period; therefore, our outcomes and predictor variables are indicative of the clinical course and not necessarily the natural history of pediatric-onset OCD.
Despite these limitations, this study provides data on early adult outcomes in a large cohort of children with OCD. It confirms the results of previous long-term outcome studies of pediatric-onset OCD in suggesting that long-term outcomes seem better than those for adults with OCD. We found that comorbid tic symptoms were associated with improved long-term outcomes in pediatric-onset OCD. Although comorbid tic symptoms have been associated with poor short-term responses to pharmacologic treatment in pediatric-onset OCD, tics may suggest a clinical course of OCD that is more likely to remit and that follows a developmental trajectory similar to that of tics. This study expands on previous results by suggesting that specific subtypes of OCD (particularly hoarding) may have worse outcomes than others, which emphasizes the importance of developing better behavioral and pharmacologic therapies for treating children with compulsive hoarding symptoms.
WHAT’S KNOWN ON THIS SUBJECT
Approximately one half of children with OCD experience remission of OCD symptoms with appropriate treatment.
WHAT THIS STUDY ADDS
Forty-four percent of children with OCD were not experiencing significant OCD symptoms at the follow-up evaluation in early adulthood. The presence of prominent hoarding symptoms and the absence of comorbid tic symptoms were associated with persistence of OCD symptoms.
Acknowledgments
We acknowledge National Institute of Mental Health support of the Yale Child Study Center Research Training Program (Drs Bloch and Leckman), grant K05MH076273 (Dr Leckman), grant K02MH074677 (Dr Peterson), and grant R01MH068318 (Dr Peterson), the National Institutes of Health Loan Repayment Program (Dr Bloch), the support of the Tourette’s Syndrome Association (Drs Bloch and Leckman), an American Psychiatric Institute for Research and Education/Eli Lilly psychiatric research fellowship (Dr Bloch), an American Academy of Child and Adolescent Psychiatry/Eli Lilly pilot research award (Dr Bloch), and the American Psychiatric Association/National Institute of Mental Health Program for Minority Research Training in Psychiatry (Dr Landeros-Weisenberger).
ABBREVIATIONS
- OCD
obsessive-compulsive disorder
- TS
Tourette syndrome
- ADHD
attention-deficit/hyperactivity disorder
- Y-BOCS
Yale-Brown Obsessive Compulsive Scale
- ODD
oppositional defiant disorder
- SSRI
selective serotonin reuptake inhibitor
- CTD
chronic tic disorder
- OC
obsessive-compulsive
Footnotes
This work was presented in part at the American Academy of Child and Adolescent Psychiatry meeting; October 23–28, 2007; Boston, MA; American College of Neuropsychopharmacology meeting; December 3–7, 2006; Hollywood, FL; American College of Neuropsychopharmacology meeting; December 9 –13, 2007; Boca Raton, FL; American College of Neuropsychopharmacology meeting; December 7–11, 2008; Waikoloa, HI; and Obsessive Compulsive Foundation meeting; August 8 –10, 2008; Boston, MA.
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
References
- 1.Douglass HM, Moffitt TE, Dar R, McGee R, Silva P. Obsessive-compulsive disorder in a birth cohort of 18-year-olds: prevalence and predictors. J Am Acad Child Adolesc Psychiatry. 1995;34(11):1424–1431. doi: 10.1097/00004583-199511000-00008. [DOI] [PubMed] [Google Scholar]
- 2.Flament MF, Whitaker A, Rapoport JL, et al. Obsessive compulsive disorder in adolescence: an epidemiological study. J Am Acad Child Adolesc Psychiatry. 1988;27(6):764–771. doi: 10.1097/00004583-198811000-00018. [DOI] [PubMed] [Google Scholar]
- 3.Narrow WE, Rae DS, Robins LN, Regier DA. Revised prevalence estimates of mental disorders in the United States: using a clinical significance criterion to reconcile 2 surveys’ estimates. Arch Gen Psychiatry. 2002;59(2):115–123. doi: 10.1001/archpsyc.59.2.115. [DOI] [PubMed] [Google Scholar]
- 4.Stewart SE, Geller DA, Jenike M, Pauls D, Shaw D, Mullin B, et al. Long-term outcome of pediatric obsessive-compulsive disorder: a meta-analysis and qualitative review of the literature. Acta Psychiatr Scand. 2004;110(1):4–13. doi: 10.1111/j.1600-0447.2004.00302.x. [DOI] [PubMed] [Google Scholar]
- 5.Leckman JF, Grice DE, Barr LC, de Vries AL, Martin C, Cohen DJ, et al. Tic-related vs. non-tic-related obsessive compulsive disorder. Anxiety. 1994;1(5):208–215. [PubMed] [Google Scholar]
- 6.Leonard HL, Swedo SE, Lenane MC, et al. A 2- to 7-year follow-up study of 54 obsessive-compulsive children and adolescents. Arch Gen Psychiatry. 1993;50(6):429–439. doi: 10.1001/archpsyc.1993.01820180023003. [DOI] [PubMed] [Google Scholar]
- 7.Bloch MH, Landeros-Weisenberger A, Rosario MC, Pittenger C, Leckman JF. Meta-analysis of the symptom structure of obsessive-compulsive disorder. Am J Psychiatry. 2008;165(12):1532–1542. doi: 10.1176/appi.ajp.2008.08020320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mataix-Cols D, Wooderson S, Lawrence N, Brammer MJ, Speckens A, Phillips ML. Distinct neural correlates of washing, checking, and hoarding symptom dimensions in obsessive-compulsive disorder. Arch Gen Psychiatry. 2004;61(6):564–576. doi: 10.1001/archpsyc.61.6.564. [DOI] [PubMed] [Google Scholar]
- 9.Hasler G, Kazuba D, Murphy DL. Factor analysis of obsessive-compulsive disorder YBOCS-SC symptoms and association with 5-HTTLPR SERT polymorphism. Am J Med Genet B Neuropsychiatr Genet. 2006;141B(4):403– 408. doi: 10.1002/ajmg.b.30309. [DOI] [PubMed] [Google Scholar]
- 10.Hasler G, LaSalle-Ricci VH, Ronquillo JG, et al. Obsessive-compulsive disorder symptom dimensions show specific relationships to psychiatric comorbidity. Psychiatry Res. 2005;135(2):121–132. doi: 10.1016/j.psychres.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 11.Mataix-Cols D, Rosario-Campos MC, Leckman JF. A multidimensional model of obsessive-compulsive disorder. Am J Psychiatry. 2005;162(2):228–238. doi: 10.1176/appi.ajp.162.2.228. [DOI] [PubMed] [Google Scholar]
- 12.Mataix-Cols D, Rauch SL, Manzo PA, Jenike MA, Baer L. Use of factor-analyzed symptom dimensions to predict outcome with serotonin reuptake inhibitors and placebo in the treatment of obsessive-compulsive disorder. Am J Psychiatry. 1999;156(9):1409–1416. doi: 10.1176/ajp.156.9.1409. [DOI] [PubMed] [Google Scholar]
- 13.Mataix-Cols D, Marks IM, Greist JH, Kobak KA, Baer L. Obsessive-compulsive symptom dimensions as predictors of compliance with and response to behaviour therapy: results from a controlled trial. Psychother Psychosom. 2002;71(5):255–262. doi: 10.1159/000064812. [DOI] [PubMed] [Google Scholar]
- 14.Leckman JF, Riddle MA, Hardin MT, et al. The Yale Global Tic Severity Scale: initial testing of a clinician-rated scale of tic severity. J Am Acad Child Adolesc Psychiatry. 1989;28(4):566–573. doi: 10.1097/00004583-198907000-00015. [DOI] [PubMed] [Google Scholar]
- 15.Scahill L, Riddle MA, McSwiggin-Hardin M, et al. Children’s Yale-Brown Obsessive Compulsive Scale: reliability and validity. J Am Acad Child Adolesc Psychiatry. 1997;36(6):844–852. doi: 10.1097/00004583-199706000-00023. [DOI] [PubMed] [Google Scholar]
- 16.Pauls DL, Alsobrook JP, 2nd, Goodman W, Rasmussen S, Leckman JF. A family study of obsessive-compulsive disorder. Am J Psychiatry. 1995;152(1):76–84. doi: 10.1176/ajp.152.1.76. [DOI] [PubMed] [Google Scholar]
- 17.Leckman JF, Sholomskas D, Thompson WD, Belanger A, Weissman MM. Best estimate of lifetime psychiatric diagnosis: a methodological study. Arch Gen Psychiatry. 1982;39(8):879–883. doi: 10.1001/archpsyc.1982.04290080001001. [DOI] [PubMed] [Google Scholar]
- 18.Goodman WK, Price LH, Rasmussen SA, et al. The Yale-Brown Obsessive Compulsive Scale, part I: development, use, and reliability. Arch Gen Psychiatry. 1989;46(11):1006–1011. doi: 10.1001/archpsyc.1989.01810110048007. [DOI] [PubMed] [Google Scholar]
- 19.First M, Spitzer R, Gibbon M, Williams J. Structured Clinical Interview for DSM-IV Axis I Disorders-Patient Edition. New York, NY: New York Psychiatric Institute; 1995. [Google Scholar]
- 20.Leckman JF, Grice DE, Boardman J, Zhang H, Vitale A, Bondi C, et al. Symptoms of obsessive-compulsive disorder. Am J Psychiatry. 1997;154(7):911–917. doi: 10.1176/ajp.154.7.911. [DOI] [PubMed] [Google Scholar]
- 21.Skoog G, Skoog I. A 40-year follow-up of patients with obsessive-compulsive disorder. Arch Gen Psychiatry. 1999;56(2):121–127. doi: 10.1001/archpsyc.56.2.121. [DOI] [PubMed] [Google Scholar]
- 22.March JS, Franklin ME, Leonard H, et al. Tics moderate treatment outcome with sertraline but not cognitive-behavior therapy in pediatric obsessive-compulsive disorder. Biol Psychiatry. 2007;61(3):344–347. doi: 10.1016/j.biopsych.2006.09.035. [DOI] [PubMed] [Google Scholar]
- 23.Bloch MH, Landeros-Weisenberger A, Kelmendi B, Coric V, Bracken MB, Leckman JF. A systematic review: antipsychotic augmentation with treatment refractory obsessive-compulsive disorder. Mol Psychiatry. 2006;11(7):622– 632. doi: 10.1038/sj.mp.4001823. [DOI] [PubMed] [Google Scholar]
- 24.Bloch MH, Peterson BS, Scahill L, et al. Adulthood outcome of tic and obsessive-compulsive symptom severity in children with Tourette syndrome. Arch Pediatr Adolesc Med. 2006;160(1):65– 69. doi: 10.1001/archpedi.160.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leckman JF, Zhang H, Vitale A, et al. Course of tic severity in Tourette syndrome: the first two decades. Pediatrics. 1998;102(1):14–19. doi: 10.1542/peds.102.1.14. [DOI] [PubMed] [Google Scholar]
- 26.Saxena S. Is compulsive hoarding a genetically and neurobiologically discrete syndrome? Implications for diagnostic classification. Am J Psychiatry. 2007;164(3):380–384. doi: 10.1176/ajp.2007.164.3.380. [DOI] [PubMed] [Google Scholar]
- 27.Leckman JF, Bloch MH. A developmental and evolutionary perspective on obsessive-compulsive disorder: whence and whither compulsive hoarding? Am J Psychiatry. 2008;165(10):1229–1233. doi: 10.1176/appi.ajp.2008.08060891. [DOI] [PubMed] [Google Scholar]
- 28.Samuels J, Shugart YY, Grados MA, et al. Significant linkage to compulsive hoarding on chromosome 14 in families with obsessive-compulsive disorder: results from the OCD Collaborative Genetics Study. Am J Psychiatry. 2007;164(3):493– 499. doi: 10.1176/ajp.2007.164.3.493. [DOI] [PubMed] [Google Scholar]
- 29.Saxena S, Brody AL, Maidment KM, et al. Cerebral glucose metabolism in obsessive-compulsive hoarding. Am J Psychiatry. 2004;161(6):1038–1048. doi: 10.1176/appi.ajp.161.6.1038. [DOI] [PubMed] [Google Scholar]
- 30.Gilbert AR, Mataix-Cols D, Almeida JR, Lawrence N, Nutche J, Diwadkar V, et al. Brain structure and symptom dimension relationships in obsessive-compulsive disorder: a voxel-based morphometry study. J Affect Disord. 2008;109(1–2):117–126. doi: 10.1016/j.jad.2007.12.223. [DOI] [PubMed] [Google Scholar]
- 31.Abramowitz JS. Effectiveness of psychological and pharmacological treatments for obsessive-compulsive disorder: a quantitative review. J Consult Clin Psychol. 1997;65(1):44–52. doi: 10.1037//0022-006x.65.1.44. [DOI] [PubMed] [Google Scholar]
- 32.Rufer M, Fricke S, Moritz S, Kloss M, Hand I. Symptom dimensions in obsessive-compulsive disorder: prediction of cognitive-behavior therapy outcome. Acta Psychiatr Scand. 2006;113(5):440–446. doi: 10.1111/j.1600-0447.2005.00682.x. [DOI] [PubMed] [Google Scholar]
- 33.Stein DJ, Andersen EW, Overo KF. Response of symptom dimensions in obsessive-compulsive disorder to treatment with citalopram or placebo. Rev Bras Psiquiatr. 2007;29(4):303–307. doi: 10.1590/s1516-44462007000400003. [DOI] [PubMed] [Google Scholar]
- 34.Saxena S, Brody AL, Schwartz JM, Baxter LR. Neuroimaging and frontal-subcortical circuitry in obsessive-compulsive disorder. Br J Psychiatry Suppl. 1998;35:26–37. [PubMed] [Google Scholar]
- 35.Masi G, Millepiedi S, Mucci M, Bertini N, Milantoni L, Arcangeli F. A naturalistic study of referred children and adolescents with obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2005;44(7):673– 681. doi: 10.1097/01.chi.0000161648.82775.ee. [DOI] [PubMed] [Google Scholar]
- 36.Storch EA, Lack CW, Merlo LJ, et al. Clinical features of children and adolescents with obsessive-compulsive disorder and hoarding symptoms. Compr Psychiatry. 2007;48(4):313–318. doi: 10.1016/j.comppsych.2007.03.001. [DOI] [PubMed] [Google Scholar]
- 37.Storch EA, Merlo LJ, Larson MJ, et al. Impact of comorbidity on cognitive-behavioral therapy response in pediatric obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2008;47(5):583–592. doi: 10.1097/CHI.0b013e31816774b1. [DOI] [PubMed] [Google Scholar]
- 38.Geller DA, Biederman J, Stewart SE, et al. Impact of comorbidity on treatment response to paroxetine in pediatric obsessive-compulsive disorder: is the use of exclusion criteria empirically supported in randomized clinical trials? J Child Adolesc Psychopharmacol. 2003;13(suppl 1):S19–S29. doi: 10.1089/104454603322126313. [DOI] [PubMed] [Google Scholar]



