Abstract
Integrin αvβ3 is most likely the foremost modulator of angiogenesis among all known integrins. Recombinant disintegrin DisBa-01, originally obtained from snake venom glands, binds to αvβ3, thereby significantly inhibiting adhesion and generating in vivo anti-metastatic ability. However, its function in mediator production is not clear. Here, we observed that the mediators VEGF-A, IL-8, and TGF-β are not produced by human umbilical vein endothelial cells (HUVEC cell line) or monocyte/macrophage cells (SC cell line) when cells adhered to vitronectin. However, when exposed to DisBa-01, HUVECs produced higher levels of TGF-β, and SC cells produced higher levels of VEGF-A. Nonetheless, HUVECs also showed an enhancement of apoptosis after losing adherence when exposed to disintegrin, which is a characteristic of anoikis. We propose that disintegrin DisBa-01 could be used to modulate integrin αvβ3 functions.
Keywords: DisBa, HUVEC, IL-8, TGF, VEGF, cancer, endothelial cell, integrin αvβ3, macrophage
Introduction
Integrins are dimeric transmembrane proteins that act in stable adhesion or mobility and can trigger intracellular signaling, thus modifying morphology, migration, and survival.1 Most cells in the human body require integrin adhesion to avoid anchorage-dependent apoptosis, a process known as anoikis.2 Among the 24 known integrins, αvβ3 (CD51/CD61) is most likely the most significant modulator of angiogenesis.3 Integrin αvβ3 may bind to a variety of compounds by interacting with the motif arginyl-glycyl-aspartic acid (RGD), but it usually binds strongly to vitronectin.1 It is expressed mainly in areas of significant inflammation, such as in cancer or wound healing, and it is overexpressed by tumor cells but is nearly absent in normal tissues of adults. Cells derived from bone marrow, such as monocytes/macrophages, also express this integrin, although not in high levels.4 Because of its characteristics, αvβ3 integrin is an interesting target for modulating pathology-associated angiogenesis.
Recombinant disintegrin DisBa-01 is a monomeric protein derived from a metalloproteinase from the venom glands of the snake Rhinocerophis alternatus; it binds mainly to the β3 subunit of αvβ3 and αIIbβ3 integrins. Its structure and production have previously been reported.5 This disintegrin has been established as possessing the important ability to inhibit cell adhesion to vitronectin, but it does not significantly affect adhesion of tumor lineage cells that present αvβ3 integrin to fibronectin.5,6 It also shows in vivo anti-metastatic abilities,5 reduces in vitro messenger ribonucleic acid (mRNA) for vascular endothelial growth factor (VEGF) receptors type 1 and 2,7 and has an anti-thrombotic role in vitro.6 Such functions could make DisBa-01 an interesting molecule in treating cancer. Mediator production triggered by DisBa-01 is not well studied, and it is widely known that integrins and mediator receptors tend to interact upon activation, sometimes creating a positive feedback, which is found in many human cancers.8
Therefore, in this work, we analyzed whether disintegrin DisBa-01 modifies mediator production and apoptosis in cells bearing αvβ3 integrin and whether this production differs from that when cells are exposed to vitronectin. The cells studied herein are immortalized and human: human umbilical vein endothelial cell line (HUVEC) and monocyte/macrophage cell line (SC) are utilized because endothelial cells and macrophages are widely present in the tumor microenvironment and may contribute dramatically to tumor growth or rejection.
Results
Influence of DisBa-01 on cell viability
Cell viability (Fig. 1) was observed after 24 h of exposure to DisBa-01 at 5 or 10 µM or controls. It was observed that, compared with untreated cells, HUVECs presented at least 71.76 ± 12.77% viable cells and SC cells presented at least 81.91 ± 8.56% viable cells, with no differences in all comparisons (P > 0.05). This viability allowed us to continue with further experiments.

Figure 1. Cell viability measured by MTT salt after exposure to DisBa-01. Cell viability was observed after HUVECs or SC cells in culture were exposed to adhesion buffer (C), phorbol 12-myristate 13-acetate (PMA), or to DisBa-01 (DB). The results are expressed as the mean and standard error of at least four independent experiments, in which C is considered to represent maximum possible viability. Data were analyzed using the Tukey-Kramer test; P > 0.05 in all comparisons.
Apoptosis assays of cells that sustained adhesion or not
Apoptosis was assessed by observing externalized phosphatidylserine on cell membranes and DNA breakage after 1.5 h of exposure to DisBa-01, followed by cellular adhesion on vitronectin for 24 h (Fig. 2). Neither HUVECs nor SC cells presented higher levels of externalized phosphatidylserine if the cells were adherent (P > 0.05 on all comparisons). However, HUVECs exposed to DisBa-01 that lost adhesion to vitronectin presented more DNA breakage compared with those exposed to buffer that could sustain adhesion (P < 0.05). This result was not observed for SC cells (P > 0.05).

Figure 2. Apoptosis in cells that did or did not maintain adhesion after exposure to DisBa-01. HUVECs and SC cells were exposed to DisBa-01 at 10 µM (DB) or adhesion buffer alone (C) and left to adhere to vitronectin-coated plates. Apoptosis was observed by (A) detection of externalized phosphatidylserine or (B) targeting DNA breaks. The results are expressed as the mean and standard deviation of three independent experiments. Data were analyzed using the Dunn test; *P < 0.05. All other comparisons are not statistically significant.
Cytokine production after cellular adhesion to vitronectin
In this study, we quantified cytokine production after cells were left to adhere in a vitronectin-coated culture plate for 24 h (Fig. 3). It was observed that HUVECs exposed to vitronectin or control produced similar amounts of IL-8, TGF-β, or VEGF-A (P > 0.05 in all comparisons). Further, SC cells did not produce significant amounts of mediators in the same conditions (P > 0.05 in all comparisons).
Figure 3. Mediator production after adhesion to vitronectin. Production of VEGF-A, IL-8, and TGF-β1 was observed in the supernatants of HUVECs or SC cells after exposure to vitronectin (Vit) or bovine serum albumin (C) for 24 h. The results are expressed as the mean and standard error of at least three independent experiments. Data were analyzed using the Tukey-Kramer test; P > 0.05 in all comparisons.
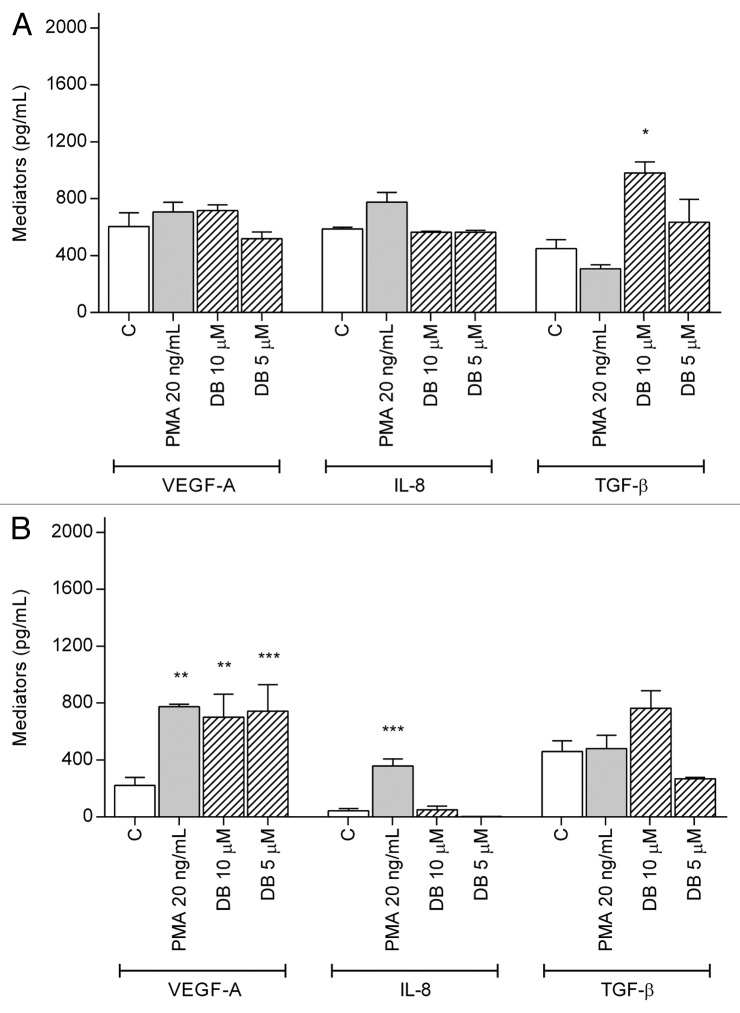
Cytokine production when cells were stimulated with DisBa-01
DisBa-01 was added to each cell line in a sterile 96-well culture plate, and cytokine production was measured after 24 h (Fig. 4). HUVECs produced higher levels of TGF-β (P < 0.05), and SC cells produced enhanced levels of VEGF-A (P < 0.01). Neither cell lineage produced higher levels of IL-8 when exposed to DisBa-01 (P > 0.05 in all comparisons). PMA generated no production of mediators for HUVECs, but it triggered production of VEGF-A and IL-8 for monocyte/macrophage SC cells (P < 0.01 and P < 0.001, respectively).
Figure 4. Production of mediators when cells were stimulated with DisBa-01. Production of VEGF-A, IL-8, and TGF-β was observed in supernatants after (A) HUVECs or (B) SC cells were exposed to adhesion buffer (C), phorbol 12-myristate 13-acetate (PMA), or DisBa-01 (DB). The results are expressed as the mean and standard error of at least three independent experiments. Data were analyzed using the Tukey-Kramer test of samples compared with C; *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
Disintegrin DisBa-01 is not a cytotoxic compound for adhered cells when used in a variety of doses, and its main function is to inhibit the adhesion to vitronectin.5 Thus, we tried to observe anoikis in two relevant cell lines treated with DisBa-01. HUVECs presented a higher percentage of cells with DNA breakage after the exposure to disintegrin caused a loss of adhesion. HUVECs express significant levels of αvβ3 integrin,9 and it is reasonable to hypothesize that anoikis could have been triggered in this scenario. However, treated cells that maintained adhesion showed similar levels of DNA breakage compared with treated cells that lost adhesion. For SC cells, other receptors may sustain adhesion to vitronectin, or anoikis could occur through other mechanisms of cell death, as reported by others.2 Although tumor cells eventually acquire some level of anoikis resistance,10 which is fundamental for metastasis formation,11 the ability to activate anoikis could be useful for cancer therapy.
Angiogenesis is an essential part of wound healing and a normal menstrual cycle but also supports tumor growth and is associated with inflammation, ischemia, and retinopathies. Mediators produced either by stressed cells or by nearby cells help initiate inflammatory processes and tissue modification as necessary for expanding vasculature.12,13 Inflammatory and endothelial cells, as well as tumor cells, may modulate angiogenesis by producing a variety of mediators.
DisBa-01 triggers the production of mediators involved in angiogenesis in a different way from that when integrin αvβ3 associates with vitronectin. In our study, vitronectin did not lead to mediator production. The overexpression of IL-8 mRNA after cellular adhesion to vitronectin has been reported,14 although the presence of mRNA may not indicate an increase in protein production per se, and VEGF-A release may not occur either.15 To the best of our knowledge, there is no information about TGF-β production after adhesion to vitronectin except our own results.
Integrin αvβ3 generates different outcomes when challenged by DisBa-01 rather than vitronectin, resulting in the significant production of TGF-β for HUVECs and VEGF-A for SC cells. The literature also shows that DisBa-01 seems to be capable of triggering VEGF-A production by human fibroblasts even in lower concentrations, although it suppresses the expression of mRNA for VEGF receptors 1 and 2.7
Cells treated with DisBa-01 usually demonstrate a significant loss of adhesion,5 which could be exploited as an anti-metastatic adjuvant treatment in cancer. Here, we demonstrated that tumor-associated cells could also be influenced by DisBa-01 to produce mediators, if they present integrin αvβ3 on cellular surface, which is normally the case for bone marrow-derived cells, such as monocyte/macrophages. In addition, integrin αvβ3 can be overexpressed at sites of significant inflammation.4 The mediators VEGF-A and TGF-β act in a contradictory fashion: TGF-β usually inhibits the proliferation of nearby cells,16 whereas VEGF-A stimulates cell proliferation17 and enhances the expression of integrin αvβ3.8
TGF-β is a member of a family of secreted growth factors produced as an inactive complex17 by several cell types, including macrophages and some tumor cells.18 TGF-β participates in embryogenesis and development, in adults it helps maintaining homeostasis and affects cell proliferation, apopstosis, immune regulation, and matrix synthesis. Its unbalance is associated with pathologies as cancer and autoimmune diseases.19 Signaling generated by TGF-β is also an indispensable pathway for angiogenesis.13
On the other hand, VEGF is a mitogen for endothelial cells with a potent angiogenic activity and an ability to stimulate migration through extracellular matrix and it can act in very low concentrations, below 1 nm/L. It can be synthetized by endothelial cells, macrophages, tumor cells, among others. VEGF can stimulate tumor growth directly and by generating angiogenesis.18
While TGF-β attracts monocytes and accentuates angiogenesis and clearance roles for those cells, VEGF-A enhances the proliferation of endothelial cells, which, along with fibroblasts, synthesize and deposit extracellular matrix. As a positive feedback, TGF-β upregulates VEGF-A expression.16,20 It is interesting to notice that integrin αvβ3 interacts with the VEGF receptors using their intracellular domains, amplifying signals from both of them, and stimulating angiogenesis. This interaction can be hyperstimulated by VEGF-A and can be abrogated indirectly by TGF-β.21
Because of unique features of this cytokines, DisBa-01 could enhance angiogenesis locally. However, TGF-β presents contradictory roles in this scenario: for one side it stimulates apoptosis and inhibits proliferation and for another side it leads to epithelial–mesenchymal transition and stimulates angiogenesis. However, this disintegrin has the ability to greatly inhibit cellular adhesion of cells bearing αvβ3 integrin,5 being an interesting anti-metastatic compound for future clinical use. Particularly in the case of cancer, angiogenesis is a possible collateral effect in theoretical chemotherapy treatment using DisBa-01, and the risk and benefits should be evaluated for every patient.
Based on our observations of apoptosis in HUVECs after adhesion loss and elevated production of VEGF-A by SC cells and TGF-β by HUVECs, which was not induced by cellular adhesion to vitronectin, DisBa-01 could be used to modulate αvβ3 integrin functions, especially in pathological conditions.
Conclusions
Monocyte/macrophage lineage SC cells, when exposed to DisBa-01, produced higher levels of VEGF-A. In addition, endothelial cell lineage HUVECs produced higher levels of TGF-β. Nonetheless, HUVECs also showed an enhancement of apoptosis after losing adherence upon exposure to disintegrin, which is a characteristic of anoikis. These results clearly indicate that disintegrin DisBa-01 affects cells that express surface αvβ3 integrin in a different way than when this integrin interacts with vitronectin. Because this integrin is highly expressed on inflamed tissues, but not on normal microenvironments, this modulation could be interesting for future clinical use.
Materials and Methods
Cells were exposed for 24 h to adhesion buffer or to the following compounds diluted in adhesion buffer: 5 or 10 µM DisBa-01, or 1 to 40 ng/mL phorbol 12-myristate 13-acetate (PMA, Sigma-Aldrich). All experiments were performed at least four distinct times and in triplicate.
DisBa-01 expression and purification
Recombinant disintegrin DisBa-01 was expressed and purified at São Carlos Federal University (UFSCAR) under the supervision of Prof HS Selistre-de-Araújo. This process has been previously reported by Ramos et al.5 The sequence data of DisBa-01 have been submitted to the GenBank databases under accession number AY259516. The presence of endotoxins was evaluated using an E-toxate kit (Sigma-Aldrich, #E8779) for each batch of disintegrin, according to the manufacturer’s instructions for the qualitative assay. Samples were negative for endotoxin presence. Disintegrin was quantified using the method proposed by Lowry et al.22 and modified by Fryer et al.23
Cell lineages
The cell lines used in this study are immortalized Human Umbilical Vascular Endothelial Cell (HUVEC) (American Type Culture Collection [ATCC, #CRL-2873]), which constitutively expresses integrin αvβ3,9 and SC (ATCC, #CRL-9855), an immortalized human monocyte/macrophage cell line.24 All cell lineages were maintained on appropriate cell media, as suggested by ATCC, at 37 °C and 5% CO2. During experimentation, the cells were maintained with 50 U/mL penicillin, 0.1 µg/mL streptomycin, and 0.25 µg/mL amphotericin B. Cells were harvested during passage number of at least 10 and no more than 20.
Coated culture plates
Sterile culture plates with 96 wells were coated with 80 μl of vitronectin (0.5 μg/mL, Sigma-Aldrich, #V8379), as suggested by the manufacturer. Then, plates were blocked with 200 μL of a bovine serum albumin solution (1% bovine serum albumin [BSA] diluted in an adhesion buffer, which consisted of 5 mM KCl, 150 mM NaCl, 20 mM HEPES, 5 mM MgCl2, and 1 mM MnCl2 at pH 7.4) for 1 h at room temperature. A control plate was incubated with 2% BSA in phosphate-buffered saline for 18 h at 4 °C and then blocked. Before their use, the coated plates were washed with 200 µL of adhesion buffer. Cells were able to adhere to mentioned substrates as observed by counting adhered cells in different concentrations using crystal violet (data not shown).
Cytotoxicity assay
The cell viability assay used in this study measures the ability of viable cells to cleave the tetrazolium ring present in MTT (3-[4,5-dimethylthiazol]-2,5-diphenyl-tetrazolium; Sigma-Aldrich, #M5655). Cells in culture medium (5 × 104 cells/mL for HUVEC and 5 × 105 cells/mL for SC) were exposed to compounds diluted in adhesion buffer 1:1 (v/v). The compounds tested were DisBa-01 at 5 and 10 µM, or phorbol 12-myristate 13-acetate (PMA, Sigma-Aldrich, #P8139) at concentrations ranging from 1 to 40 ng/mL. The plates were incubated for 24 h at 37 °C and 5% CO2. After this period, the supernatant was removed, and the cells were exposed to MTT solution at a final concentration of 1 mg/mL and incubated at 37 °C for 3 (HUVECs) or 4 h (SC cells). After incubation, the culture medium was removed, and the formazan crystals were dispersed in 100 µL of isopropanol. The optical density of the resulting solution was determined using a spectrophotometer at 540 nm and 620 nm. Cell viability was calculated as a percentage of total cells, assuming the negative control to be 100% viable.
Anoikis
Cells (1 × 105 cells/mL) were suspended in adhesion buffer with or without disintegrin DisBa-01 at 10 µM and incubated for 30 min at 37 °C. Then, cell suspensions were delicately homogenized and transferred to a coated plate and incubated for 24 h at 37 °C and 5% CO2. Non-adhered cells were transferred to a test tube, and adhered cells were washed, trypsinized, and transferred to a different test tube. Cells were washed and reactions were performed using Annexin V-FITC Apoptosis Detection Kit (eBioscience, # BMS500FI) or Apo-Direct Apoptosis Detection Kit (eBioscience, #88-6611-88), according to the manufacturer's instructions.
Quantification of mediators
Cells (5 × 105 cells/mL) were exposed to vitronectin-coated culture plates and incubated for 24 h at 37 °C and 5% CO2. Non-adherent cells were washed out, the culture medium was replenished, and cells were incubated for 24 h under the same conditions. Supernatants obtained from that experiment and from the culture plates described in the “cytotoxicity assay” section above were analyzed for the mediators VEGF-A, IL-8, and TGF-β using ELISA assay kits (eBioscience, respectively: #BMS277/2, #88-8086, #88-8350), according to instructions. All assays were performed with control standards, as indicated by the manufacturer, and cells were also cultured in the presence of PMA for observing a production if cells were responsive to this compound.
Statistical analysis
Statistical analyses for apoptosis assays were performed using the Kruskal-Wallis one-way analysis of variance (ANOVA), followed by the Dunn test for multiple comparisons. Statistical analyses for mediators were performed using ANOVA, followed by the Tukey-Kramer test. P values of P < 0.05 were considered statistically significant. The program used was GraphPad Prism 5.00 (GraphPad Software).
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Acknowledgments
We thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for funding (#2010/05428-0) and for the scholarships for first author (#2010/01568-2) and second author (#2011/07798-2).
Footnotes
Previously published online: www.landesbioscience.com/journals/celladhesion/article/27698
References
- 1.Konstantopoulos K, Thomas SN. Cancer cells in transit: the vascular interactions of tumor cells. Annu Rev Biomed Eng. 2009;11:177–202. doi: 10.1146/annurev-bioeng-061008-124949. [DOI] [PubMed] [Google Scholar]
- 2.Buchheit CL, Rayavarapu RR, Schafer ZT. The regulation of cancer cell death and metabolism by extracellular matrix attachment. Semin Cell Dev Biol. 2012;23:402–11. doi: 10.1016/j.semcdb.2012.04.007. [DOI] [PubMed] [Google Scholar]
- 3.Danhier F, Le Breton A, Préat V. RGD-based strategies to target alpha(v) beta(3) integrin in cancer therapy and diagnosis. Mol Pharm. 2012;9:2961–73. doi: 10.1021/mp3002733. [DOI] [PubMed] [Google Scholar]
- 4.Desgrosellier JS, Cheresh DA. Integrins in cancer: biological implications and therapeutic opportunities. Nat Rev Cancer. 2010;10:9–22. doi: 10.1038/nrc2748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ramos OHP, Kauskot A, Cominetti MR, Bechyne I, Salla Pontes CL, Chareyre F, Manent J, Vassy R, Giovannini M, Legrand C, et al. A novel α(v)β (3)-blocking disintegrin containing the RGD motive, DisBa-01, inhibits bFGF-induced angiogenesis and melanoma metastasis. Clin Exp Metastasis. 2008;25:53–64. doi: 10.1007/s10585-007-9101-y. [DOI] [PubMed] [Google Scholar]
- 6.Kauskot A, Cominetti MR, Ramos OHP, Bechyne I, Renard JM, Hoylaerts MF, Crepin M, Legrand C, Selistre-de-Araujo HS, Bonnefoy A. Hemostatic effects of recombinant DisBa-01, a disintegrin from Bothrops alternatus. Front Biosci. 2008;13:6604–16. doi: 10.2741/3176. [DOI] [PubMed] [Google Scholar]
- 7.Montenegro CF, Salla-Pontes CL, Ribeiro JU, Machado AZ, Ramos RF, Figueiredo CC, Morandi V, Selistre-de-Araujo HS. Blocking αvβ3 integrin by a recombinant RGD disintegrin impairs VEGF signaling in endothelial cells. Biochimie. 2012;94:1812–20. doi: 10.1016/j.biochi.2012.04.020. [DOI] [PubMed] [Google Scholar]
- 8.Chiodoni C, Colombo MP, Sangaletti S. Matricellular proteins: from homeostasis to inflammation, cancer, and metastasis. Cancer Metastasis Rev. 2010;29:295–307. doi: 10.1007/s10555-010-9221-8. [DOI] [PubMed] [Google Scholar]
- 9.Teklemariam T, Seoane AI, Ramos CJ, Sanchez EE, Lucena SE, Perez JC, Mandal SA, Soto JG. Functional analysis of a recombinant PIII-SVMP, GST-acocostatin; an apoptotic inducer of HUVEC and HeLa, but not SK-Mel-28 cells. Toxicon. 2011;57:646–56. doi: 10.1016/j.toxicon.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Okayama H. Cell cycle control by anchorage signaling. Cell Signal. 2012;24:1599–609. doi: 10.1016/j.cellsig.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 11.Taddei ML, Giannoni E, Fiaschi T, Chiarugi P. Anoikis: an emerging hallmark in health and diseases. J Pathol. 2012;226:380–93. doi: 10.1002/path.3000. [DOI] [PubMed] [Google Scholar]
- 12.Lokmic Z, Musyoka J, Hewitson TD, Darby IA. Hypoxia and hypoxia signaling in tissue repair and fibrosis. Int Rev Cell Mol Biol. 2012;296:139–85. doi: 10.1016/B978-0-12-394307-1.00003-5. [DOI] [PubMed] [Google Scholar]
- 13.van Meeteren LA, Goumans MJ, ten Dijke P. TGF-β receptor signaling pathways in angiogenesis; emerging targets for anti-angiogenesis therapy. Curr Pharm Biotechnol. 2011;12:2108–20. doi: 10.2174/138920111798808338. [DOI] [PubMed] [Google Scholar]
- 14.Antonov AS, Antonova GN, Munn DH, Mivechi N, Lucas R, Catravas JD, Verin AD. αVβ3 integrin regulates macrophage inflammatory responses via PI3 kinase/Akt-dependent NF-κB activation. J Cell Physiol. 2011;226:469–76. doi: 10.1002/jcp.22356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wary KK, Kohler EE, Chatterjee I. Focal adhesion kinase regulation of neovascularization. Microvasc Res. 2012;83:64–70. doi: 10.1016/j.mvr.2011.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Miyazono K, Ehata S, Koinuma D. Tumor-promoting functions of transforming growth factor-β in progression of cancer. Ups J Med Sci. 2012;117:143–52. doi: 10.3109/03009734.2011.638729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Worthington JJ, Fenton TM, Czajkowska BI, Klementowicz JE, Travis MA. Regulation of TGFβ in the immune system: an emerging role for integrins and dendritic cells. Immunobiology. 2012;217:1259–65. doi: 10.1016/j.imbio.2012.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grazul-Bilska AT, Johnson ML, Bilski JJ, Redmer DA, Reynolds LP, Abdullah A, Abdullah KM. Wound healing: the role of growth factors. Drugs Today (Barc) 2003;39:787–800. doi: 10.1358/dot.2003.39.10.799472. [DOI] [PubMed] [Google Scholar]
- 19.Horiguchi M, Ota M, Rifkin DB. Matrix control of transforming growth factor-β function. J Biochem. 2012;152:321–9. doi: 10.1093/jb/mvs089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008;16:585–601. doi: 10.1111/j.1524-475X.2008.00410.x. [DOI] [PubMed] [Google Scholar]
- 21.Nam JO, Son HN, Jun E, Cha K, Lee BH, Park RW, Kim IS. FAS1 domain protein inhibits VEGF165-induced angiogenesis by targeting the interaction between VEGFR-2 and αvβ3 integrin. Mol Cancer Res. 2012;10:1010–20. doi: 10.1158/1541-7786.MCR-11-0600. [DOI] [PubMed] [Google Scholar]
- 22.Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–75. [PubMed] [Google Scholar]
- 23.Fryer HJ, Davis GE, Manthorpe M, Varon S. Lowry protein assay using an automatic microtiter plate spectrophotometer. Anal Biochem. 1986;153:262–6. doi: 10.1016/0003-2697(86)90090-4. [DOI] [PubMed] [Google Scholar]
- 24.Yamamoto A, Ochiai M, Kamachi K, Kataoka M, Toyoizumi H, Arakawa Y, Horiuchi Y. A clinically relevant in vitro pyrogen test using a human cell line that have the similar responsiveness to various pyrogens to that of human peripheral blood cells (hPBC) AATEX. 2007;14:647–53. [Google Scholar]