Accumulating evidence supports the critical role of B-cell receptor (BCR) activation in chronic lymphocytic leukaemia (CLL) pathogenesis (Herishanu, et al 2011). Recent preclinical and clinical studies indicate BCR signal inhibitors, specifically the Bruton tyrosine kinase (BTK) inhibitor Ibrutinib and phosphatidylinositide 3-kinase (PI3K) δ inhibitor CAL101, have impressive preclinical and clinical efficacy in CLL (Byrd, et al 2013, Herman, et al 2010). Akt is downstream of the signalling cascade following BCR ligation. Preclinical studies performed using co-culture of CLL B-cells with marrow stromal cells highlight the importance of PI3K/Akt signalling in the activation of both cell types during their interactions that support survival of CLL B-cells (Ding, et al 2010, Edelmann, et al 2008). MK2206 is a highly selective oral allosteric Akt inhibitor found to have minimal side effects in a phase 1 trial of patients with refractory solid tumours (Yap, et al 2011). The present study determined the maximum tolerated dose (MTD) of MK2206 as a single-agent and its ability to target Akt in tumour cells. Here we explore the in vitro efficacy of MK2206 on CLL B-cell survival, its potential synergy with bendamustine, and its impact on BCR ligation events including cytokine release both in vitro and in vivo. Materials and methods are detailed in Appendix S1.
We initially tested the effect of the Akt inhibitor, MK2206, on CLL B-cell survival. Dose and time- dependent apoptosis was induced by Akt inhibition with MK2206 (Figure 1A, n = 37, appendix S1). Unlike CLL cells, normal peripheral blood mononuclear cells did not undergo apoptosis after exposure to MK2206. Basal phosphorylation of AktS473 was detected at variable levels amongst tested CLL samples (n = 7) but was completely abolished by MK2206 treatment at the 1 μM dose (Figure 1B). MK2206 also resulted in cleavage of the poly (ADP-ribose) polymerase (PARP) protein and significantly decreased the activation of Akt target p70S6K. However, expression of the p85 subunit of PI3K, which is upstream of Akt, was not affected. Next we tested whether established prognostic factors would be associated with differential CLL apoptosis levels induced by MK2206. No significant differences in MK2206-induced cell death were found based on the presence or absence of IGHV mutation, del (17p13), positivity of ZAP70 and CD38 (Figure 1C-F). These results indicated that MK2206 is able to induce apoptosis in CLL B-cells independent of adverse prognostic factors.
Figure 1.
Effect of MK2206 treatment on CLL apoptosis. (A) The mean and standard error of apoptosis levels of peripheral blood mononuclear cells (PBMC) isolated from 37 CLL patients and 5 normal subjects. (B) Western blot analysis showed treatment with MK2206 decreased levels of pAktS473, p70S6KT389 and induced PARP cleavage but had no effect on total Akt, the p85 subunit of PI3K, and total p70S6K. Results from a representative patient are shown. MK2006-induced apoptosis levels did not differ based on IGHV mutation status (C, p > 0.05 for tested doses), the presence or absence of del (17p13) (D, p > 0.05 for tested doses), or ZAP70 and CD38 expression (E and F, p > 0.05 for tested doses). To determine whether MK2206 in combination treatment with bendamustine at 50% inhibitory concentration (IC50) doses of both drugs yielded greater cell death than expected from summation alone, cytotoxicity data were analysed using the CalcuSyn software program (Biosoft, Cambridge, UK), using the method of Chou and Talalay (Chou and Talalay 1984). A combination index (CI) of 0.8 – 1.2 was considered to indicate an additive effect, CI >1.2 an antagonistic effect, and CI <0.8 a synergistic effect. An additive (n = 5) or synergistic (n=6) was seen in a majority (11/12; 92%) of a patients (G and H). The mean and standard error of apoptosis levels of PBMC isolated from 12 CLL patients are shown in (H). BEN, bendamustine.
To assess whether MK2206 could enhance the drug sensitivity of CLL cells to the established agent (bendamustine) in relapsed disease (Fischer, et al 2011), we next explored the impact of MK2206 in combination with bendamustine on CLL apoptosis. An additive or synergistic affect was observed when MK2206 was added to bendamustine in 11 of the 12 CLL samples tested (figure 1G and 1H).
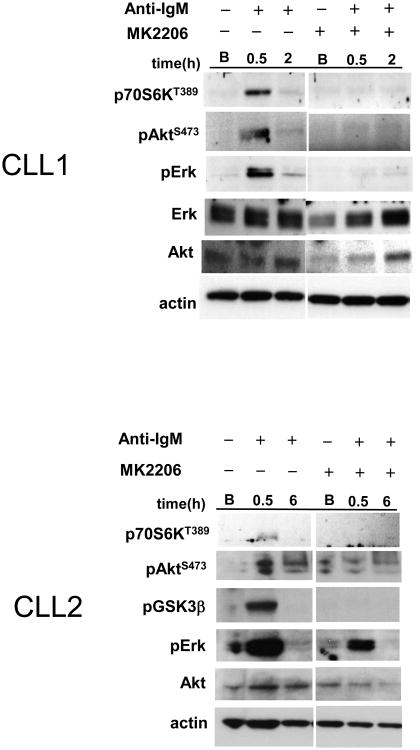
Given the prognostic significance of BCR-induced cytokines (Sivina, et al 2011), we subsequently tested if Akt inhibition with MK2206 could abrogate BCR-mediated cytokine secretion and signal activation. BCR ligation with anti-IgM significantly increased production of selective immune cytokines (CCL3, CCL4, CCL2, IL-2Rα, IL-1Rα and IL-8) among the 30 cytokines tested (Figure 2A-B, n = 7). CCL3 and CCL4 upregulation are consistent with the previous report (Burger, et al 2009). The increase of CCL2, IL-2Rα, IL-1Rα and IL-8 following BCR ligation of CLL has not been previously reported. Pre-treatment with MK2206 abrogated the increase of CCL3, CCL4, CCL2 and IL-2Rα (Figure 2A), but not IL-1Rα and IL-8 (Figure 2B). Variable degrees of pAktS473, pGSK3βS9, p44/42Erk T202/Y204 and p70S6K T389 activation were also observed at 30 min after BCR stimulation (Figure 2C). MK2206 pretreatment reduced or abolished the activation of pAktS473, pGSK3βS9 and p70S6KT389 induced by anti-IgM and reduced the activation of p44/42Erk T202/Y204 to a variable degree. Collectively, these in vitro results indicate that MK2206 selectively abolishes BCR induced CCL3, CCL4, CCL2 and IL-2Rα production, probably via abrogating Akt and Erk signals. Other BCR-induced cytokines, such as IL-8 and IL-1Rα however were not affected by MK2206, suggesting these cytokines are induced by BCR-initiated signals other than those mediated by Akt or Erk phosphorylation.
Figure 2.
MK2206 selectively abrogates immune cytokine secretion induced by BCR ligation on CLL cells. (A, B) The secretion of CCL3, CCL4, CCL2, IL-2Rα, IL-1Rα and IL-8 increased with time following BCR ligation. MK2206 treatment significantly abrogates the increase of CCL3, CCL4, CCL2 and IL-2Rα (**, p < 0.01; *, p < 0.05) (A), but does not affect the increased production of IL-1Rα and IL-8 (B). base: baseline. (C) MK2206 reduces the AKT and the ERK activation mediated by BCR ligation in a variable degree on tested CLL cells (n=5, 2 representative casesshown). Akt downstream target GSK3β and p70S6K activation also were reduced. B: baseline. (D) Top panels: MK2206 monotherapy administered one time to CLL patients was associated with increases of absolute lymphocyte count (ALC) at one week post-therapy in CLL patients (n=8). Bottom panel: a single administered dose of MK2206 to CLL patients resulted in decreased CCL3/CCL4/CCL2 plasma levels in the majority of tested patients at one week post-therapy, but these levels were increased following administration of one oral dose of MK2206 therapy in 2 patients (CLL2, CLL6). D0 ALC: baseline absolute lymphocyte count. D8 ALC: ALC at day 8 after one dose of MK2206 administered at day 1. The percentage change of ALC or cytokine was calculated as: Percent ALC increase = (D8 ALC − D0 ALC)/D0 ALC; Percent cytokine reduction = (D0 cytokine − D8 cytokine)/D0 cytokine.
These preclinical studies support trials of MK2206 in combination with bendamustine for CLL patients. To that end, we initiated a phase 1/2 trial of MK2206 in combination with bendamustine and rituximab for patients with relapsed CLL (N1087) in 2011. N1087 is currently open for accrual with 10 patients enrolled to date. To evaluate the single agent effect of MK2206, this trial involves a 1-week run-in of MK2206 monotherapy prior to initiating bendamustine and rituximab (Appendix S1). Similar to the finding of lymphocyte mobilization from other BCR targeting therapies, all patients evaluated (n=8) had increases of their absolute lymphocyte count (ALC) after a single dose of MK2206 monotherapy (Figure 2D, mean increase of 8 patients: 56%). The increase in ALC after the first dose of MK2206 was modest in three of these patients (6.3%, 6.7% and 11.0% increase respectively), and more dramatic in the remaining 5 patients (mean increase of 84.3%; Figure 2D).
To explore the clinical relevance of our in vitro results showing that MK2206 abrogates BCR-mediated CCL2/3/4 secretion, we investigated the effect of a single-dose MK2206 on the plasma levels of these cytokines in trial patients after one week. The majority of the tested 9 patients had a decrease in CCL3 (mean ± standard error of the mean [n=7]: 25% ± 6% reduction), CCL4 ([n=7], 22% ± 3% reduction) and CCL2 level ([n=6], 20% ± 7% reduction) after a single-dose MK2206 (Figure 2D). In contrast, two patients were found to have small increases of these three cytokines after the single-dose of MK2206. Notably these two patients also experienced the smallest increase in ALC. Although our sample size is too small to draw a definitive conclusion, these findings corroborate our in vitro results and suggest that the reduction of the BCR-stimulated CCL2/3/4 secretion is associated with lymphocyte mobilization.
Collectively, our in vitro and in vivo findings indicate that MK2206 induces apoptosis of CLL B-cells and is able to block the signal activation and cytokine up-regulation induced by BCR ligation. Single agent MK2206 is capable of mobilizing leukaemic B-cells and decreases CCL3, CCL4 and CCL2 production in vivo. We believe that this data strongly encourages further clinical testing of MK2206 either alone or in combination with traditional chemotherapeutic drugs in CLL.
Supplementary Material
Acknowledgments
This work was supported by funding from NCI grant K23CA160345 (WD), NCI grant CA95241 (NEK) and Cephalon (TDS). Dr. Shanafelt is a Clinical Scholar of the Leukemia and Lymphoma Society.
Footnotes
Authorship Contributions: WD designed research and performed the experiments, analysed and interpreted the results, and wrote the paper. TDS and NEK designed research, interpreted the results, and wrote the paper. CEL, CS and TRS performed the experiments and interpreted the results. CE designed research, provided scientific input, review, and discussion. SK provided reagent and provided review and input. JFL, TGC, DAB, MC recruited patients, provided input and discussion.
Disclosure of Conflicts of Interest: TDS has received research support from Celgene, Hospira, Cephalon, Genentech, Glaxo-Smith-Kline and Polyphenon E International. SK has consulting relations with Merck, Celgene, Millennium, Novartis, Sanofi and Onyx (all uncompensated). CE was on the advisory board of Merck over one year ago. NEK has received research support from Celgene.
References
- Burger JA, Quiroga MP, Hartmann E, Burkle A, Wierda WG, Keating MJ, Rosenwald A. High-level expression of the T-cell chemokines CCL3 and CCL4 by chronic lymphocytic leukemia B cells in nurselike cell cocultures and after BCR stimulation. Blood. 2009;113:3050–3058. doi: 10.1182/blood-2008-07-170415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrd JC, Furman RR, Coutre SE, Flinn IW, Burger JA, Blum KA, Grant B, Sharman JP, Coleman M, Wierda WG, Jones JA, Zhao W, Heerema NA, Johnson AJ, Sukbuntherng J, Chang BY, Clow F, Hedrick E, Buggy JJ, James DF, O'Brien S. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. The New England journal of medicine. 2013;369:32–42. doi: 10.1056/NEJMoa1215637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Advances in enzyme regulation. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- Ding W, Knox TR, Tschumper RC, Wu W, Schwager SM, Boysen JC, Jelinek DF, Kay NE. Platelet-derived growth factor (PDGF)-PDGF receptor interaction activates bone marrow-derived mesenchymal stromal cells derived from chronic lymphocytic leukemia: implications for an angiogenic switch. Blood. 2010;116:2984–2993. doi: 10.1182/blood-2010-02-269894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelmann J, Klein-Hitpass L, Carpinteiro A, Fuhrer A, Sellmann L, Stilgenbauer S, Duhrsen U, Durig J. Bone marrow fibroblasts induce expression of PI3K/NF-kappaB pathway genes and a pro-angiogenic phenotype in CLL cells. Leukemia research. 2008;32:1565–1572. doi: 10.1016/j.leukres.2008.03.003. [DOI] [PubMed] [Google Scholar]
- Fischer K, Cramer P, Busch R, Stilgenbauer S, Bahlo J, Schweighofer CD, Bottcher S, Staib P, Kiehl M, Eckart MJ, Kranz G, Goede V, Elter T, Buhler A, Winkler D, Kneba M, Dohner H, Eichhorst BF, Hallek M, Wendtner CM. Bendamustine combined with rituximab in patients with relapsed and/or refractory chronic lymphocytic leukemia: a multicenter phase II trial of the German Chronic Lymphocytic Leukemia Study Group. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2011;29:3559–3566. doi: 10.1200/JCO.2010.33.8061. [DOI] [PubMed] [Google Scholar]
- Herishanu Y, Perez-Galan P, Liu D, Biancotto A, Pittaluga S, Vire B, Gibellini F, Njuguna N, Lee E, Stennett L, Raghavachari N, Liu P, McCoy JP, Raffeld M, Stetler-Stevenson M, Yuan C, Sherry R, Arthur DC, Maric I, White T, Marti GE, Munson P, Wilson WH, Wiestner A. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood. 2011;117:563–574. doi: 10.1182/blood-2010-05-284984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman SE, Gordon AL, Wagner AJ, Heerema NA, Zhao W, Flynn JM, Jones J, Andritsos L, Puri KD, Lannutti BJ, Giese NA, Zhang X, Wei L, Byrd JC, Johnson AJ. Phosphatidylinositol 3-kinase-delta inhibitor CAL-101 shows promising preclinical activity in chronic lymphocytic leukemia by antagonizing intrinsic and extrinsic cellular survival signals. Blood. 2010;116:2078–2088. doi: 10.1182/blood-2010-02-271171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivina M, Hartmann E, Kipps TJ, Rassenti L, Krupnik D, Lerner S, LaPushin R, Xiao L, Huang X, Werner L, Neuberg D, Kantarjian H, O'Brien S, Wierda WG, Keating MJ, Rosenwald A, Burger JA. CCL3 (MIP-1alpha) plasma levels and the risk for disease progression in chronic lymphocytic leukemia. Blood. 2011;117:1662–1669. doi: 10.1182/blood-2010-09-307249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yap TA, Yan L, Patnaik A, Fearen I, Olmos D, Papadopoulos K, Baird RD, Delgado L, Taylor A, Lupinacci L, Riisnaes R, Pope LL, Heaton SP, Thomas G, Garrett MD, Sullivan DM, de Bono JS, Tolcher AW. First-in-man clinical trial of the oral pan-AKT inhibitor MK-2206 in patients with advanced solid tumors. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2011;29:4688–4695. doi: 10.1200/JCO.2011.35.5263. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.