Abstract
Patient: Male, 9
Final Diagnosis: Duchenne muscular dystrophy
Symptoms: Hyporeflexia • hypotonia • weaknes of lower limbs
Medication: —
Clinical Procedure: —
Specialty: Neurology
Objective:
Congenital defects/diseases
Background:
Duchenne muscular dystrophy (DMD) is a fatal, genetic, progressive, degenerating muscle disorder. Current treatment options are palliative. Newer options of cellular therapy promise to alter the disease process. Preclinical studies have successfully tested myogenic, neurogenic potential and dystrophin expression of bone marrow mononuclear cells.
Case Report:
We treated a 9-year-old boy suffering from DMD with serial autologous bone marrow mononuclear cell transplantations followed by multidisciplinary rehabilitation. Brooke-Vignos score was 10 and he was wheelchair-bound. Over 36 months, gradual progressive improvement was noticed in muscle strength, ambulation with assistive devices, fine motor movements, Brooke-Vignos score, and functional independence measure score. Nine months after the transplantation, electromyography findings showed development of new normal motor unit potentials of the vastus medialis muscle.
Conclusions:
Magnetic resonance imaging scan of musculoskeletal systems showed no increase in fatty infiltration. This case report provides early investigative findings or the restorative effects of cellular therapy in DMD.
MeSH Keywords: Stem Cell Transplantation, Autologous Bone Marrow Mono Nuclear Cells, Electromyography, Muscular Dystrophy, Duchenne – congenital
Background
Duchenne muscular dystrophy (DMD) is a progressive degeneration of the striated muscles of the body, and has a fatal prognosis. The disease is caused by mutation, deletion, or duplication of the dystrophin gene [1,2], leading to synthesis of functionally impotent dystrophin. Dystrophin is a protein essential to maintaining the integrity of the exoskeleton of the muscle cells [3]. Dystrophin is also a structural component of neurons, glial cells, and Schwann cells. The role of dystrophin in the development and function of these cells is not as well defined as that in muscles, but it is presumed to influence synaptic activity [4,5].
The disease manifests as progressive weakness of the muscles, leading to loss of function. Children typically exhibit loss of ambulation in the second decade of life, followed by premature death [6]. Treatment of muscular dystrophy consists of medical, rehabilitation, and surgical management aiming to preserve the function of the individual and prolonging their independence [7,8]. These treatments contribute very little towards altering the pathology of the disease. Cellular transplantation has exhibited dystrophin expression in the muscles of dystrophic mice and various cell types have shown myogenic potential and neurogenic potential [9–11]. Cellular transplantation in humans is safe and feasible and has shown positive clinical effects in people with muscular dystrophy [12,13].
We present a case of DMD monitored over 36 months treated with serial adult autologous Bone marrow mononuclear cell (BMMNC) transplantations followed by long-term multidisciplinary rehabilitation.
Case Report
A 9-year-old boy had a history of difficulty in getting up from the floor, climbing stairs, and frequent falls since the age of 4 years. The increasing weakness in the lower limbs led to toe walking and loss of ambulation by the age of 8 years. The weakness then progressed to the upper extremities and performing overhead activities was difficult. Diagnosis of DMD was confirmed based on the clinical features, elevated serum creatinine phosphokinase (CPK) levels (5460 IU/L), and genetic analysis with multiplex polymerase chain reaction for dystrophin gene showing deletion of exons 51 and 52. Despite regular medical management and physiotherapy, the muscle strength continued to deteriorate.
On examination, there was evident calf and deltoid muscle pseudo-hypertrophy. He presented with hypotonia and hyporeflexia. Posture was lordotic. Muscular strength was measured by manual muscle testing, using a scale devised by our experienced physiotherapists based on the modified Medical Research Council’s manual muscle testing scale (mMRC MMT). Since mMRC-MMT does not sub-classify grades 1 and 2 according to partial Range of Motion (ROM), in our scale (mMRC MMT – I) grades 1 and 2 were subdivided. This allowed us to measure the subtle changes in the strength as observed in patients with DMD (Appendix 1). Proximal muscle strength was less than in the distal muscles (Table 1). Ambulation was wheelchair-dependent. Functionally, he was dependent for all of his daily activities and his functional independence measure (FIM) score was 85. Brooke-Vignos scale score was 10. In leisure activities, he could swim for up to 60 minutes without fatigue.
Table 1.
Changes in the muscle strength over 36 months as measured by mMRC-MMT (I) scale.
| Date | mMRC-MMT (I) grade at assessment (Before 1st transplantation) | mMRC-MMT (I) grade at 9 months (Before 2nd transplantation) | mMRC-MMT (I) grade at 24 months (Before 3rd transplantation) | mMRC-MMT (I) grade at 33 months (Before 4th transplantation) | mMRC-MMT (I) grade at 36 months | |
|---|---|---|---|---|---|---|
|
| ||||||
| Muscle groups | ||||||
| Hip | Flexors | 3− | 3 | 1+ | 2 | 2 |
| Extensors | 1 | 2+ | 1+ | 1++ | 1++ | |
| Abductors | 2+ | 2++ | 2+ | 2+ | 2+ | |
| Adductors | 2 | 2++ | 2++ | 2++ | 2++ | |
|
| ||||||
| Knee | Flexors | 3+ | 3+ | 3+ | 3 | 3 |
| Extensors | 3− | 3 | 3 | 3 | 3 | |
|
| ||||||
| Ankle and foot | Tibialis anterior | 3+ | 3+ | 3+ | 3++ | 3++ |
| Tibialis posterior | 4 | 4 | 4 | 3++ | 3++ | |
| Peronius longus | 2+ | 2+ | 2+ | 3++ | 3++ | |
| Peroneous brevis | 2+ | 2+ | 2+ | 3++ | 3++ | |
|
| ||||||
| Trunk | Abdominals upper | 2 | 2++ | 2++ | 2++ | 2++ |
|
| ||||||
| Neck | Flexors | 3+ | 3+ | 3+ | 3++ | 3++ |
| Extensors | 3+ | 3+ | 3+ | 2+ | 2+ | |
|
| ||||||
| Shoulder | Flexors | 3+ | 3+ | 3+ | 3 | 3++ |
| Extensors | 3+ | 3+ | 3+ | 3 | 2+ | |
|
| ||||||
| Arm | Biceps | 3+ | 3+ | 2++ | 2+ | 2+ |
| Brachialis | 3+ | 3+ | 2+ | 2+ | 2+ | |
| Triceps | 3+ | 3+ | 2++ | 2+ | 2+ | |
| Brachioradialis | 3+ | 3+ | 2++ | 2+ | 2+ | |
|
| ||||||
| Forearm, wrist and hand | Supinators | 3+ | 3+ | 3++ | 3++ | 3++ |
| Pronators | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Extensors | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Flexors | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Flexor Pollicis Longus | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Flexor Policis Brevis | 3 | 3 | 3++ | 3++ | 3++ | |
| Extensor Pollicis Longus | 3++ | 3++ | 3++ | 3++ | 3++ | |
| Extensor Policis Brevis | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Adductor Policis | 3++ | 3++ | 3++ | 3++ | 3++ | |
| Abductor Pollicis Longus | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Opponens Pollicis | 3+ | 3+ | 3++ | 3++ | 3++ | |
| Palmar Interossei | 4 | 4 | 4 | 4 | 4 | |
On investigations, serum CPK levels were elevated (5144 IU/l). His musculoskeletal magnetic resonance imaging (MRI – MSK) showed extensive fatty infiltration in the pelvic girdle muscles, moderate fatty infiltration in thigh and leg muscles, and mild infiltration in foot, arm, forearm, and hand muscles. Electromyography (EMG) showed generalized intrinsic muscle disease as suggested by presence of myopathic action potentials and absence of normal motor unit potentials.
Pre-intervention assessment and intervention
Selection of this patient for the treatment was based on the World Medical Association Revised Declaration of Helsinki. Ethical approval was obtained from the Institutional Committee for Stem Cell Research and Therapy (IC-SCRT), NeuroGen, Brain and Spine Institute, Mumbai, India. Parents of the patient provided informed consent for the procedure and subsequent reporting.
Serological, biochemical, and hematological blood tests, chest X-ray, electrocardiogram, and 2-D echocardiography a week before autologous BMMNC transplantation established preoperative fitness. Granulocyte colony-stimulating factor (GCSF) 300 mcg was administered subcutaneously 48 hours and 24 hours prior to the MNCs transplantation. GCSF was given to enhance mobilization of BMMNCs [14]. A range of medical and allied health professionals conducted a detailed assessment. Muscles with mMRC MMT (I) score less than 3 and of functional importance were selected for intramuscular transplantation of BMMNC. Motor points (the point where the innervating nerve enters the muscle belly) were identified and marked by an experienced physiotherapist.
On the day of transplantation, 100 ml of bone marrow was aspirated from the anterior superior iliac spine under local anesthesia using a bone marrow aspiration needle and was collected in heparinized tubes. Density gradient method was used to separate the BMMNC fraction. Fluorescence-activated cell sorting (FACS) analysis showed 98% viability of the cells and a CD34+ count of 1.45%. Half of the cell fraction was injected intrathecally at the level between L4 and L5. The remaining cells were then diluted in the patient’s own cerebrospinal fluid (CSF) due to the properties of CSF that harbor cell growth [15]. Cells were then injected intramuscularly, bilaterally in the specific motor points of biceps, triceps, hamstrings, quadriceps, glutei, back extensors, and abdominals. Methyl prednisolone (1 gm) in 500 ml of Ringer lactate solution was administered intravenously to reduce the immediate inflammation. Intrathecally and intramuscularly, a combined total of 33×106 cells were injected.
BMMNC transplantation was combined with vigorous rehabilitation during the subsequent days before discharge. This was undertaken by a physiotherapist, an occupational therapist, a speech therapist, and a psychologist. At 1 week the patient was discharged and a detailed home program was prescribed to be followed after discharge under professional guidance.
Positive response as explained in the results provided the rationale for subsequent transplantations. The patient underwent serial cellular transplantations at 9, 21, and 31 months after the first transplantation. The transplantation procedure was replicated, including the muscles chosen for intramuscular injections. A detailed follow-up assessment, MRI-MSK scan, and EMG-NCV study were repeated prior to subsequent transplantations. MRI-MSK and EMG-NCV were conducted on the same unit with the same parameters as before.
Results
Following the transplantations, there was a gradual increase in the strength of various muscle groups. Most of the muscle groups maintained strength (Table 1). The Brooke and Vignos scale scores improved over a period of 3 years from 10 to 8 (Table 2). FIM scores increased from 85 to 92 at the end of 36 months (Table 2). MRI-MSK showed no increase in the fatty infiltration until the end of the follow-up period. EMGNCV study showed improvement in vastus medialis muscles 9 months after first transplantation, which was maintained until after 3 years.
Table 2.
Changes in various outcome measures over the period of 36 months.
| Outcome measures | At assessment months (before 1st transplantation) | At 9 months (before 2nd transplantation) | At 24 months (before 3rd transplantation) | At 33 months (before 4th transplantation) | At 36 months |
|---|---|---|---|---|---|
| Functional Independence Measure score | 85 | 92 | 92 | 92 | 92 |
| Brooke-Vignos scale score | 10 | 10 | 9 | 8 | 8 |
After the first transplantation, there was a significant improvement in the ankle range of motion and exercise tolerance. The strength of abdominals and hip flexors, extensors, and adductors had increased.
One month after the second transplantation, he could stand holding a walker. Three months after the second transplantation, he started walking wearing calipers with the help of a walker. He was independent in maintaining perineal hygiene. He could sit up from supine position faster and with greater ease.
After the third transplantation, he experienced ease in overhead activities and the compensatory behavior had reduced. Walking endurance had increased with push knee splints and walker to 90 minutes. It was possible to take a few steps without a walker. His dynamic standing balance was better. Swimming endurance had improved to 90 minutes. Frequency of falls had also reduced.
After the fourth transplantation, the above-mentioned changes were still maintained. His exercise tolerance had increased further. There was further improvement in dynamic balance while standing and walking. Speed of walking had increased. Handwriting was neat and legible. Frequency of falls had reduced significantly, with no episode of falling noted in 8 months. There was a visible change in his hand writing (Figure 1).
Figure 1.

Changes in the handwriting over the period of 24 months. (A) Hand writing sample at assessment. (B) Hand writing sample at 24 months.
Discussion
DMD is characterized by progressive muscle weakness. At the cellular level this can be explained as muscle necrosis that exceeds the regenerative capacity of the muscles. To endure the severities of every muscle contraction, muscle fibers have specialized cytoskeletal protein complexes, dystrophin. These complexes make it possible for the myocytes to endure the mechanical stresses of the muscle contraction. In DMD, abnormalities in the dystrophin gene lead to non-expression of the protein, dystrophin. This leads to sarcolemmal disruption and cell necrosis [1,3]. Repair and regeneration of these cells is engineered by local stem cells and infiltrating inflammatory cells. These local stem cells are termed “satellite” cells. There is, however, only a limited pool of satellite cells in the muscles; therefore, in DMD it cannot meet the increasing demands of accelerated cell necrosis [16]. In the presence of increased necrotic tissue, the extracellular environment also becomes conducive to necrosis.
The current management of DMD is restricted to maintaining the functional independence of the patient for as long as possible using various therapeutic strategies. The medical management aims at reducing the early inflammatory process and slowing the muscle necrosis [7,8]. These management strategies only delay the imminent outcome of the disease. DMD still leads to early loss of ambulation and functional dependence. New advances in the management of DMD using exon skipping, gene therapy, and cellular therapy show some promise of being able to alter the disease process, which may lead to further improvement in disease progression and survival of these patients. Exon skipping can be described as the process whereby a DNA analogue corrects the transcription of dystrophin, skipping the genetic abnormality that leads to incomplete but potentially better functioning protein sequence [17]. Gene therapy aims at introducing the absent dystrophin gene using various vectors. Several practical difficulties have prevented gene therapy from being a clinically feasible and viable option at present [18].
Mononuclear cell transplantation has been successfully tested for its myogenic and neurogenic properties and ability to express dystrophin in regenerated muscles in animals [9–11]. Stem cells isolated from various donor sites (e.g., bone marrow, umbilical cord, adipose tissue, and menstrual blood) [19] have been used for the treatment of muscular dystrophy. However, the evidence is still inconclusive about which of these are most effective for skeletal muscle regeneration. The evidence about the dosage and frequency of stem cell transplantation also remains ambiguous. The important conclusion is that there are no adverse effects of the autologous bone marrow mononuclear cell transplantation in muscular dystrophy [12,13]. Although primarily a muscle disease, involvement of neural structures has been observed. Dystrophin has been thought to play a role in the neuronal synapses and myelination of the nerves by anchoring the Schwann cells [20,21]. Nervous system impairment is manifested as impaired cognition in some children with DMD [22]. Therefore, half of the cell fraction was transplanted intrathecally to address the neural component in DMD.
In our patient we used BMMNC transplantation followed by rigorous rehabilitation. It has been observed that physical activity facilitates the effectiveness of the stem cell transplantation in muscular dystrophy [23]. Stem cell transplantation is mainly found to have its beneficial effects through various paracrine mechanisms of angiogenesis, stimulation of various growth factors, and stimulation satellite stem cells [24]. Our patient showed improved muscle recruitment and improved function 3 years after the stem cell transplantation. This suggests some alteration in the disease process, as the natural course of the disease shows reduction of muscular strength by 0.3 MMT units/ year [25]. Lue et al found a 3.9% reduction in muscle strength every year [26]. In our clinical experience, measuring the strength of the muscles using mMRC-MMT was not sensitive to measure this subtle decline and we therefore further subdivided the scale using the same principles (Appendix 1). This scale was standardized for all the patients and was able to measure the subtle changes in muscle strength. The impressive objective finding was MRI-msk with no increase in the fatty infiltration (Figure 2A, 2B). Analysis of serial MRI scans of children with DMD suggests a 5% increase of fatty infiltration every year [27]. No significant increase in the fatty infiltration in 36 months suggests altered disease progression. EMG studies also suggested improvement in the recruitment of the vastus medialis muscle, which is a key muscle in patellar stability and knee stability while walking [28]. This was functionally reflected as ability to stand and walk independently, maintained over 3 years.
Figure 2.
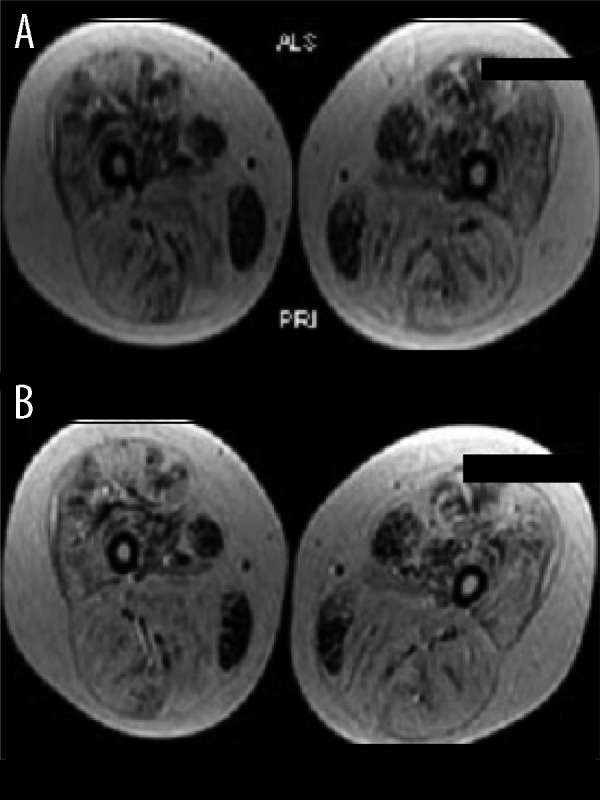
(A) Musculoskeletal MRI assessment before cellular transplantation. (B) Musculoskeletal MRI assessment 24 months after cellular transplantation.
Conclusions
Although cellular therapy is still in its primitive stages, this case report indicates it may have the potential to alter the disease pathology in DMD. Further robust analysis and trials with sophisticated methodology are needed to determine the optimum dosage, source, and frequency of transplantation. This case report provides early imaging evidence for effects of cellular therapy in DMD.
Appendix 1.
Comparison of the grades of the scales mMRC-MMT and mMRC-MMT (I).
| mMRC-MMT grade | Description | mMRC-MMT (I) grade | Description |
|---|---|---|---|
| 0 | No Movement | 0 | No movement |
| 1 | A flicker of movement is seen or felt in the muscle | 1 | A flicker of movement is seen or felt in the muscle |
| 2 | Muscle moves the joint when gravity is eliminated | 1+ | Muscle moves the joint through up to 1/3rd of the ROM when gravity is eliminated |
| 1++ | Muscle moves the joint more than 1/3rd less than 2/3rd of the ROM when gravity is eliminated | ||
| 2− | Muscle moves the joint more than 2/3rd but less than the full ROM | ||
| 2 | Muscle moves the joint through complete ROM when gravity is eliminated | ||
| 3− | Muscle moves the joint against gravity, but not through full mechanical range of motion | 2+ | Muscle moves the joint up to 1/3rd ROM against gravity |
| 2++ | Muscle moves the joint >1/3rd, <2/3rd of ROM against gravity | ||
| 3− | Muscle moves the joint more than 2/3rd but less than complete ROM | ||
| 3 | Muscle cannot hold the joint against resistance but moved the joint fully against gravity | 3 | Muscle moves the joint through complete ROM against gravity |
| 3+ | Muscle moves the joint fully against gravity and is capable of transient resistance, but collapses abruptly | 3+ | Muscle moves the joint against gravity and moderate resistance up to 1/3rd of ROM |
| 3++ | Muscle moves the joint against gravity and moderate resistance from 1/3rd to 2/3rd of ROM | ||
| 4− | Same as grade 4, but muscle holds the joint only against minimal resistance | 4− | Muscle moves the joint more than 2/3rd but less than complete ROM against gravity and moderate resistance |
| 4 | Muscle holds the joint against a combination of gravity and moderate resistance | 4 | Muscle moves the joint against gravity and moderate resistance though complete ROM |
| 4+ | Same as grade 4 but muscle holds the joints against moderate to maximal resistance | 4+ | Muscle moves the joint against gravity and moderate to maximal resistance up to 1/3rd of ROM |
| 5− | Barely detectable weakness | 4++ | Muscle moves the joint against gravity and moderate to maximal resistance from 1/3rd to 2/3rd of ROM (Barely detectable weakness) |
| 5 | Normal strength | 5 | Muscle moves the joint against gravity and moderate to maximal resistance though complete ROM (Normal Strength) |
References:
- 1.Peddareddygari LR, Pillai BH, Nochlin D, et al. Phenotype-genotype analysis of dystrophinopathy caused by duplication mutation in Dystrophin gene in an African patient. Afr Health Sci. 2011;11(4):607–9. [PMC free article] [PubMed] [Google Scholar]
- 2.Basak J, Dasgupta UB, Banerjee TK, et al. Analysis of dystrophin gene deletions by multiplex PCR in eastern India. Neurology India. 2006;54(3):310–11. doi: 10.4103/0028-3886.27164. [DOI] [PubMed] [Google Scholar]
- 3.Fink RH, Stephenson DG, Williams DA. Physiological properties of skinned fibers from normal and dystrophic (Duchenne) human muscle activated by Ca2+ and Sr2+ J Physiol. 1990;420:337–53. doi: 10.1113/jphysiol.1990.sp017916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Waite A, Tinsley CL, Locke M, Blake DJ. The neurobiology of the dystrophin-associated glycoprotein complex. Ann Intern Med. 2009;41(5):344–59. doi: 10.1080/07853890802668522. [DOI] [PubMed] [Google Scholar]
- 5.Waite A, Brown SC, Blake DJ. The dystrophin-glycoprotein complex in brain development and disease. Trends Neurosci. 2012;35(8):487–96. doi: 10.1016/j.tins.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 6.Yiu EM, Kornberg AJ. Duchenne muscular dystrophy. Neurology India. 2008;56(3):236–47. doi: 10.4103/0028-3886.43441. [DOI] [PubMed] [Google Scholar]
- 7.Bushby K, Finkel R, Birnkrant DJ, et al. DMD Care Considerations Working Group. Diagnosis and management of Duchenne muscular dystrophy, part 1: diagnosis, and pharmacological and psychosocial management. Lancet Neurol. 2010;9(1):77–93. doi: 10.1016/S1474-4422(09)70271-6. [DOI] [PubMed] [Google Scholar]
- 8.Bushby K, Finkel R, Birnkrant DJ, et al. DMD Care Considerations Working Group. Diagnosis and management of Duchenne muscular dystrophy, part 2: implementation of multidisciplinary care. Lancet Neurol. 2010;9(2):177–89. doi: 10.1016/S1474-4422(09)70272-8. [DOI] [PubMed] [Google Scholar]
- 9.Torrente Y, Tremblay JP, Pisati F, et al. Intra arterial injection of muscle-derived CD34 (+) Sca-1(+) stem cells restores dystrophin in mdx mice. J Cell Biol. 2001;152(2):335–48. doi: 10.1083/jcb.152.2.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ferrari G, Mavilio F. Myogenic stem cells from the bone marrow: a therapeutic alternative for muscular dystrophy? Neuromuscul Disord. 2002;12(Suppl.1):S7–10. doi: 10.1016/s0960-8966(02)00102-5. [DOI] [PubMed] [Google Scholar]
- 11.Song S, Song S, Zhang H, et al. Comparison of neuron-like cells derived from bone marrow stem cells to those differentiated from adult brain neural stem cells. Stem Cells Dev. 2007;16(5):747–56. doi: 10.1089/scd.2007.0027. [DOI] [PubMed] [Google Scholar]
- 12.Sharma A, Gokulchandran N, Chopra G, et al. Administration of autologous bone marrow-derived mononuclear cells in children with incurable neurological disorders and injury is safe and improves their quality of life. Cell Transplant. 2012;21(Suppl.1):S79–90. doi: 10.3727/096368912X633798. [DOI] [PubMed] [Google Scholar]
- 13.Sharma A, Paranjape A, Sane H, et al. Cellular Transplantation Alters the Disease Progression in Becker’s Muscular Dystrophy. Case Rep Transplant. 2013;2013:909328. doi: 10.1155/2013/909328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haas R, Murea S. The role of granulocyte colony-stimulating factor in mobilization and transplantation of peripheral blood progenitor and stem cells. Cytokines Mol Ther. 1995;1(4):249–70. Erratum in: Cytokines Mol Ther, 1996; 2(1): 136. [PubMed] [Google Scholar]
- 15.Miyan JA, Zendah M, Mashayekhi F, Owen-Lynch PJ. Cerebrospinal fluid supports viability and proliferation of cortical cells in vitro, mirroring in vivo development. Cerebrospinal Fluid Res. 2006;3:2. doi: 10.1186/1743-8454-3-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hawke TJ, Garry DJ. Myogenic satellite cells: physiology to molecular biology. J Appl Physiol. 2001;91(2):534–51. doi: 10.1152/jappl.2001.91.2.534. Erratum in: J Appl Physiol, 2001; 91(6): 2414. [DOI] [PubMed] [Google Scholar]
- 17.Arechavala-Gomeza V, Anthony K, Morgan J, Muntoni F. Antisense oligonucleotide-mediated exon skipping for Duchenne muscular dystrophy: progress and challenges. Curr Gene Ther. 2012;12(3):152–60. doi: 10.2174/156652312800840621. [DOI] [PubMed] [Google Scholar]
- 18.Konieczny P, Swiderski K, Chamberlain JS. Gene and cell-mediated therapies for muscular dystrophy. Muscle Nerve. 2013;47(5):649–63. doi: 10.1002/mus.23738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chabort EJ, Myburgh KH, Wiehe JM, et al. Potential myogenic stem cell populations: sources, plasticity, and application for cardiac repair. Stem Cells Dev. 2009;18(6):813–30. doi: 10.1089/scd.2008.0387. [DOI] [PubMed] [Google Scholar]
- 20.Blitzblau R, Storer E, Jacob M. Dystrophin and utrophin isoforms are expressed in glia, but not neurons, of the avian parasympathetic ciliary ganglion. Brain Research. 2008;7(1218):21–34. doi: 10.1016/j.brainres.2008.04.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Luo L, Zhou HY. Co-transplantation of myoblasts and Schwann cells in the therapy of Duchenne muscular dystrophy. Sichuan Da XueXueBao Yi Xue Ban. 2011;42(1):101–5. [PubMed] [Google Scholar]
- 22.Moizard MP, Toutain A, Fournier D, et al. Severe cognitive impairment in DMD: obvious clinical indication for Dp71 isoform point mutation screening. Eur J Hum Genet. 2000;8(7):552–56. doi: 10.1038/sj.ejhg.5200488. [DOI] [PubMed] [Google Scholar]
- Sveen ML, Jeppesen TD, Hauerslev S, et al. Endurance training improves fitness and strength in patients with Becker muscular dystrophy. Brain. 2008;131:2824–31. doi: 10.1093/brain/awn189. [DOI] [PubMed] [Google Scholar]
- 24.Gnecchi M, Zhang Z, Ni A, Dzau VJ. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res. 2008;103(11):1204–19. doi: 10.1161/CIRCRESAHA.108.176826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kilmer DD, Abresch RT, Fowler WM., Jr Serial manual muscle testing in Duchenne muscular dystrophy. Arch Phys Med Rehabil. 1993;74(11):1168–71. [PubMed] [Google Scholar]
- 26.Lue YJ, Jong YJ, Lin YT, Chen SS. [The strength and functional performance of patients with Duchenne muscular dystrophy based on natural history] Gaoxiong Yi Xue Ke Xue Za Zhi. 1992;8(11):597–604. [PubMed] [Google Scholar]
- 27.Kim HK, Merrow AC, Shiraj S, et al. Analysis of fatty infiltration and inflammation of the pelvic and thigh muscles in boys with Duchenne muscular dystrophy (DMD): grading of disease involvement on MR imaging and correlation with clinical assessments. Pediatr Radiol. 2013;43(10):1327–35. doi: 10.1007/s00247-013-2696-z. [DOI] [PubMed] [Google Scholar]
- 28.Sakai N, Luo ZP, Rand JA, An KN. The influence of weakness in the vastus medialis oblique muscle on the patellofemoral joint: an in vitro biomechanical study. Clin Biomech (Bristol, Avon) 2000;15(5):335–39. doi: 10.1016/s0268-0033(99)00089-3. [DOI] [PubMed] [Google Scholar]


