Abstract
Purpose
Prone positioning for ARDS has been performed for decades without definitive evidence of clinical benefit. A recent multicenter trial demonstrated for the first time significantly reduced mortality with prone positioning. This meta-analysis was performed to integrate these findings with existing literature and test whether differences in tidal volume explain conflicting results among randomized trials.
Methods
Studies were identified using MEDLINE, EMBASE, Cochrane Register of Controlled Trials, LILACS, and citation review. Included were randomized trials evaluating the effect on mortality of prone versus supine positioning during conventional ventilation for ARDS. The primary outcome was risk ratio of death at 60 days meta-analyzed using random effects models. Analysis stratified by high (>8 ml/kg predicted body weight) or low (≤8 ml/kg PBW) mean baseline tidal volume was planned a priori.
Results
Seven trials were identified including 2,119 patients, of whom 1,088 received prone positioning. Overall, prone positioning was not significantly associated with the risk ratio of death (RR 0.83; 95 % CI 0.68–1.02; p = 0.073; I2 = 64 %). When stratified by high or low tidal volume, prone positioning was associated with a significant decrease in RR of death only among studies with low baseline tidal volume (RR 0.66; 95 % CI 0.50–0.86; p = 0.002; I2 = 25 %). Stratification by tidal volume explained over half the between-study heterogeneity observed in the unstratified analysis.
Conclusions
Prone positioning is associated with significantly reduced mortality from ARDS in the low tidal volume era. Substantial heterogeneity across studies can be explained by differences in tidal volume.
Keywords: Acute respiratory distress syndrome, Acute lung injury, Prone position, Patient positioning, Meta-analysis, Randomized controlled trial
Introduction
Acute respiratory distress syndrome (ARDS) accounts for an estimated 75,000 deaths and 3.6 million hospital days annually in the USA [1]. Mechanical ventilation with low tidal volumes [2, 3] and early neuromuscular blockade [4] have been shown definitively to reduce mortality from ARDS. Despite these advancements, mortality from ARDS remains high, ranging from 27 to 45 % for mild to severe disease [5, 6].
Prone positioning has been used for four decades in patients with ARDS [7]. Its physiological benefits are well described: improved ventilation-perfusion matching [8], recruitment of dependent lung regions [9, 10], optimized chest wall mechanics [11], and enhanced drainage of tracheobronchial secretions [12]. Yet, these physiological benefits have failed to translate into reduced mortality in multiple clinical trials of prone positioning for ARDS [13–18].
By contrast, a recent multicenter randomized trial by Guérin et al. [19] found that prone positioning reduced the risk of death by half. While previous meta-analyses and post hoc analyses have suggested benefit in patients with the highest overall illness severity [13, 16, 20] and with severe ARDS [13, 21, 22], no such benefit was seen even in the largest prior randomized trials enrolling 300–800 patients each [13, 14, 18].
Therefore, we performed the following meta-analysis of randomized trials of prone positioning in ARDS to identify sources of heterogeneity between studies that would account for their divergent findings. We anticipated that the gradual adoption of low tidal volume ventilation over the last decade would largely explain the conflicting results of these trials [23–25]. We hypothesized that prone positioning reduces 60-day mortality from ARDS only when injurious high tidal volumes are avoided.
Methods
Identification of trials
Studies were identified using computerized literature searches of MEDLINE, EMBASE, LILACS, and Cochrane Central Register of Controlled Trials. The following Medical Subject Headings (MeSH) and keywords were combined to retrieve relevant articles: randomized controlled trial OR controlled clinical trial OR random* OR trial OR groups AND prone position (MeSH) OR supine position (MeSH) OR patient positioning (MeSH) OR prone OR proning OR supine AND respiratory distress syndrome, adult (MeSH) OR acute lung injury OR ARDS OR respiratory distress syndrome OR respiratory failure.
Selection of studies
Using the PICOS framework [26], two independent reviewers (SBM, SS) applied criteria to evaluate studies for inclusion. Disagreement in study selection was addressed through evaluation by a third reviewer (JRB) and final determination achieved by team consensus. Eligible studies involved adult patients meeting the Berlin definition of ARDS [5, 27], including those previously classified as having acute lung injury (PaO2:FiO2 201–300 mmHg) under the 1994 American-European Consensus Conference definition [28]. Studies were required to assess the intervention prone positioning compared to supine positioning during conventional mechanical ventilation. Co-interventions in addition to prone positioning were permitted. Studies that did not report mortality were excluded. Only randomized controlled trials were eligible.
Data extraction
Two authors (JRB, SBM) independently extracted data using a standardized data collection form. Discrepancies in collected data were addressed through team consensus. One included trial, Taccone et al. [18], stratified at randomization according to moderate or severe ARDS (PaO2:FiO2 100–200 or <100 mmHg). It was decided before data extraction to treat this trial as two separate studies in the meta-analysis because prior post hoc analyses have suggested the benefit of prone positioning may vary with ARDS severity [13, 21, 22].
Evaluation of primary outcome
The primary outcome was risk ratio (RR) of death at 60 days among patients assigned to prone compared to supine positioning. When not directly reported, 60-day mortality was determined by digitally enhancing the Kaplan-Meier plot to measure directly the relative distance from zero of the product limit estimator at 60 days [29]. In one study [15], neither 60-day mortality nor a Kaplan–Meier plot was available; the corresponding author provided the required information.
Quality assessment
Study quality was evaluated by considering concealment of allocation, completeness of follow-up, blinded analysis, crossover between study arms, post hoc exclusions, and early trial discontinuation. The Jadad score for randomized trials [30] was not considered because blinding of patients or caregivers to the study intervention was not possible.
Statistical analysis
The primary outcome of 60-day mortality was chosen a priori because it represents the maximum follow-up interval reported in all studies selected for analysis. Potential sources of heterogeneity considered a priori were use of low tidal volume ventilation, duration of respiratory failure prior to prone positioning, and dose (in hours/day) of prone positioning [25]. Before data extraction, an analysis plan was developed to account for anticipated sources of heterogeneity, consisting of stratification by high (>8 ml/kg predicted body weight, PBW) versus low (≤8 ml/kg PBW) mean baseline tidal volume.
Initial meta-analysis was carried out using random effects models to calculate risk ratios with 95 % confidence intervals for each included study and a combined risk ratio for all studies. This method was selected for its proven ability to account for within- and between-study variability [31]. Heterogeneity was quantified with the Q-statistic and I2 index. Cumulative meta-analysis was also performed to evaluate the potential effects of gradual widespread adoption of lung-protective ventilation and other advances in critical care over time.
To evaluate whether tidal volume was a significant source of inter-study heterogeneity, additional random effects models were developed after stratifying by mean baseline tidal volume. Unrestricted maximum likelihood mixed effects regression was performed to evaluate for a dose-response relationship between mortality (log risk ratio) and mean baseline tidal volume (ml/kg PBW). Random effects models also were used to evaluate the effect of high (≥12 h/day) versus low (<12 h/day) proning dose.
To evaluate for publication bias, a funnel scatter plot of sample and effect size was constructed. Publication bias was also assessed via Begg-Mazumdar rank correlation and Egger’s regression to account for selection bias impact on the overall combined significance of the results. The meta-analysis was conducted using Comprehensive Meta-Analysis v2 (Biostat, Eaglewood, NJ, USA) with conscious effort to follow PRISMA guidelines [32].
Results
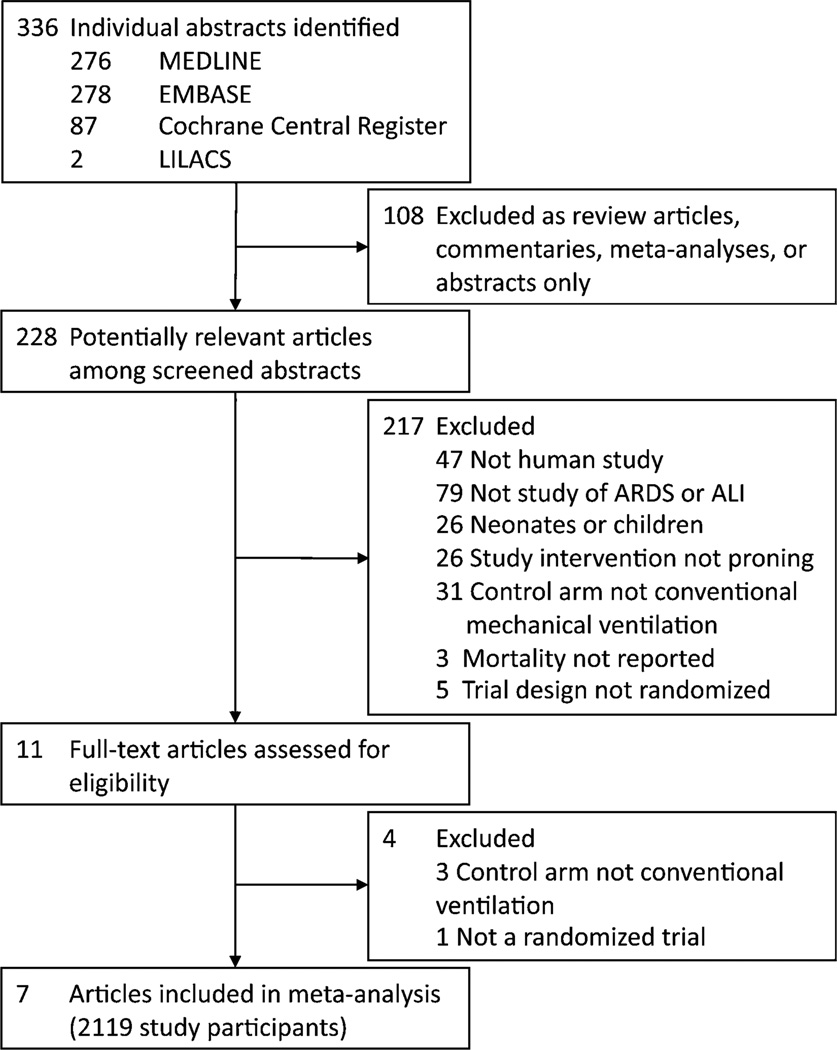
The initial search yielded 643 citations comprising 336 unique abstracts (Fig. 1). After excluding 108 abstracts that did not represent peer-reviewed primary research and 217 that did not meet the inclusion criteria, 11 articles were identified for full-text review to assess for eligibility. Four of these 11 studies were excluded after review for non-conventional ventilation in the control arm or non-randomized design. Seven randomized trials encompassing 2,119 patients were included for meta-analysis. A total of 1,088 patients were mechanically ventilated in the prone position and 1,031 in the supine position. One included study, Taccone et al. [18], stratified patients at randomization according to moderate or severe ARDS (PaO2:FiO2 100–200 or <100 mmHg) and was treated as two separate trials throughout the analysis.
Fig. 1.
Literature search strategy and study selection
Study quality
All studies were multicenter randomized controlled trials. In all cases, concealed allocation was achieved through either a centralized process or sealed envelopes. Loss to follow-up occurred rarely but was accounted for in detail. Post hoc exclusions occurred only for withdrawal of consent or mistaken inclusion in enrollment. Three trials were terminated early for slow enrollment [13, 16, 17], leading to a risk of insufficient statistical power to detect differences between groups.
Patient and study characteristics
Table 1 summarizes key features of the included studies. The primary endpoint for five of the seven trials was mortality at a specified time of follow-up [13, 14, 17–19]. One trial’s primary endpoint was ICU mortality [16] and the other duration of mechanical ventilation [15]. Between 40 and 802 subjects were enrolled in each trial. Degree of hypoxemia required for study eligibility became increasingly severe over time. Targeted daily dose of prone positioning ranged from 6 to >20 h/day, with higher doses in more recent trials. All trials used lower levels of PEEP than currently recommended [33].
Table 1.
Key design features of included studies
| Gattinoni 2001 [13] |
Guérin 2004 [14] |
Voggenreiter 2005 [15] |
Mancebo 2006 [16] |
Fernandez 2008 [17] |
Taccone 2009 [18] |
Guérin 2013 [19] |
|
|---|---|---|---|---|---|---|---|
| Study characteristics | |||||||
| No. subjects enrolled | 304 | 802 | 40 | 142 | 42 | 344 | 474 |
| No. of sites | 30 | 21 | 2 | 13 | 17 | 25 | 27 |
| Inclusion | ≤300 | ≤300 | ≤300 | ≤200 | ≤200 | Stratified: | <150 |
| PaO2:FiO2 range, mmHg | 100–200; <100 | ||||||
| Inclusion cause of ARDS | Any | Any | Multiple trauma | Any | Any | Any | Any |
| Prior proning experience | Not reported | Yes | Not reported | Yes | Not reported | Yes | Yes |
| Geographic region | Italy, Switzerland | France | Germany | Mexico, Spain | Spain | Italy, Spain | France, Spain |
| Enrollment period | 1996–1999 | 1998–2002 | 1999–2001 | 1998–2002 | 2003–2007 | 2004–2008 | 2008–2011 |
| Study interventions | |||||||
| Proning dose recommended, h/day | ≥6 | ≥8 | 8–23 | 20 | Up to 20 | ≥20 | ≥16 |
| Proning dose achieved, h/day | 7.0 ± 1.8 | 8.0 [7.7, 9.8] | 11 ± 5 | 17 | Not reported | 18 ± 4 | 17 ± 3 |
| Proning treatment course end criteria | P:F > 200 on PEEP ≥ 5 or P:F > 300 on PEEP ≥ 10 | Study-specific “improvement criteria” | P:F > 300 for 48 h | FiO2 ≤ 0.45 & SaO2 ≥ 93 % at PEEP ≤ 8 | P:F > 250 at PEEP ≤ 8 for 12 h | Resolution of acute respiratory failure | P:F ≥ 150 at PEEP ≤ 10 & FiO2 < 0.6 for 4 h |
| Proning treatment course, no. days | 10 | 4 [2.0, 6.0] | Not reported | 10.1 ± 10.3 | Not reported | 8.4 ± 6.3 | 4 ± 4 |
| Days ventilated before proning | Not reported | Not reported | Not reported | <2 | <2 | <3 | <1.5 |
| PEEP titration strategy | Usual care | Usual care | Recommended PEEP ≥ 12 for FiO2 > 0.5 | Recommended PEEP 10–15 | ARDSnet ARMA | Study-specific protocol similar to ALVEOLI low PEEP | ALVEOLI low PEEP |
| Study quality | |||||||
| Concealed allocation | Centralized | Sealed envelopes | Centralized | Sealed envelopes | Centralized | Centralized | Centralized |
| Post hoc exclusions | None | 5 withdrew consent, 4 inclusion mistake | None | None | None | 2 inclusion mistake | 7 inclusion mistake, 1 proxy issues |
| Cross-over from supine to prone, no. (%) | 12 (7.9) | 81 (21.4) | None | 5 (8.3) | 2 (10.5) | 20 (11.5) | 17 (7.4) |
| Follow-up for main outcome | Complete | 2 lost to follow-up | Complete | 1 lost to follow-up, 5 missing data | 2 lost to follow-up | Complete | Complete |
| Early termination | Yes: slow enrollment | No | No | Yes: slow enrollment | Yes: slow enrollment | No | No |
| Blinded analysis | Not reported | Not reported | Not reported | Not reported | Not reported | Yes | Yes |
Numerical data are presented as mean ± standard deviation, median [interquartile range], or number (%) as appropriate
Patient characteristics and individual study outcomes are summarized in Table 2. The earliest studies are characterized by the highest tidal volumes and highest mortality. Four studies [13–16] began enrollment before publication of the landmark NHLBI ARDS Network trial that demonstrated reduced mortality with low tidal volumes [2]. Four studies [15, 17–19] targeted tidal volumes in the current recommended range of 6–8 ml/kg [34, 35]. Adverse events, including unintended extubation, were rare.
Table 2.
Patient characteristics and outcomes by study
| Gattinoni 2001 [13] |
Guérin 2004 [14] |
Voggenreiter 2005 [15] |
Mancebo 2006 [16] |
Fernandez 2008 [17] |
Taccone 2009 (mod. ARDS) [18] |
Taccone 2009 (sev. ARDS) [18] |
Guérin 2013 [19] | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prone | Supine | Prone | Supine | Prone | Supine | Prone | Supine | Prone | Supine | Prone | Supine | Prone | Supine | Prone | Supine | |
| Patient characteristics at baseline | ||||||||||||||||
| No. subjects | 152 | 152 | 413 | 378 | 21 | 19 | 76 | 60 | 21 | 19 | 94 | 98 | 74 | 76 | 237 | 229 |
| SAPS II score | 40 ± 14 | 40 ± 16 | 45 ± 15 | 46 ± 16 | Not reported | 43 ± 16 | 38 ± 14 | 37 ± 11 | 39 ± 13 | 39 ± 14c | 43.0 ± 14.6c | 45 ± 15 | 47 ± 17 | |||
| Tidal volume, cc/kg PBW | 10.3 ± 2.7 | 10.3 ± 2.9 | 8.1 ± 2.0a | 8.1 ± 1.9a | Target 6–8b | 8.3 ± 1.7 | 8.6 ± 1.6 | 7.4 ± 1.1 | 7.1 ± 1.1 | 8.2 ± 1.7c | 7.7 ± 1.6c | 6.1 ± 0.6 | 6.1 ± 0.6 | |||
| Set PEEP, cm H2O | 9.7 ± 2.9 | 9.6 ± 3.2 | 7.9 ± 3.4 | 7.5 ± 3.2 | 12 ± 4 | 11 ± 3 | 12.4 ± 1.9 | 12.3 ± 2.4 | 12.1 ± 3.1 | 14.2 ± 4.4 | 9 ± 2c | 11 ± 3c | 10 ± 3 | 10 ± 4 | ||
| PaO2:FiO2 | 125 ± 49 | 130 ± 48 | 150 ± 59 | 155 ± 59 | 215 ± 63 | 228 ± 75 | 132 ± 74 | 161 ± 94 | 153 ± 59 | 157 ± 83 | 141 ± 27c | 77 ± 16c | 100 ± 30 | 100 ± 20 | ||
| Outcomes | ||||||||||||||||
| Mortality at 60d, % | 57.2 | 51.6 | 41.1 | 40.3 | 4.8 | 15.8 | 41.9d | 53.4d | 38.1 | 52.6 | 31.7 | 37.2 | 44.6 | 54.8 | 20.1 | 37.7 |
| Ventilator time among survivors (d) | Not reported | 16.9 ± 11.4 | 17.6 ± 13.7 | 30 ± 17 | 33 ± 23 | Not reported | 12.0 ± 10.6 | 7.6 ± 7.6 | 23 [10–28] | 19 [7–28] | 27 [12–28] | 18 [11–28] | 17 ± 16 | 19 ± 21 | ||
| Unplanned extubation, no. (%) | 1 (0.5) | 0 (0) | 44 (10.7) | 47 (12.4) | 1 (4.8) | 1 (5.3) | 6 (7.9) | 1 (1.7) | 1 (4.8) | 1 (5.3) | 12 (12.8) | 6 (6.1) | 6 (8.1) | 2 (2.6) | 31 (13.3) | 25 (10.9) |
Numerical data are presented as mean ± standard deviation, median [interquartile range], or number (%) as appropriate. Patient characteristics are baseline values prior to study intervention
Tidal volume was reported for patients in the volume-control mode per measured body weight and not predicted body weight. Approximately 12 % of subjects received another mode of ventilation as chosen by the clinical team
Baseline tidal volume was not reported
SAPS II score, tidal volume, set PEEP, and PaO2:FiO2 were not reported separately for prone and supine groups
Kaplan-Meier product limit estimator was presented only for intensive care unit survival, the primary outcome of this study. Post-discharge survival was not reported in this study
Prone positioning and mortality
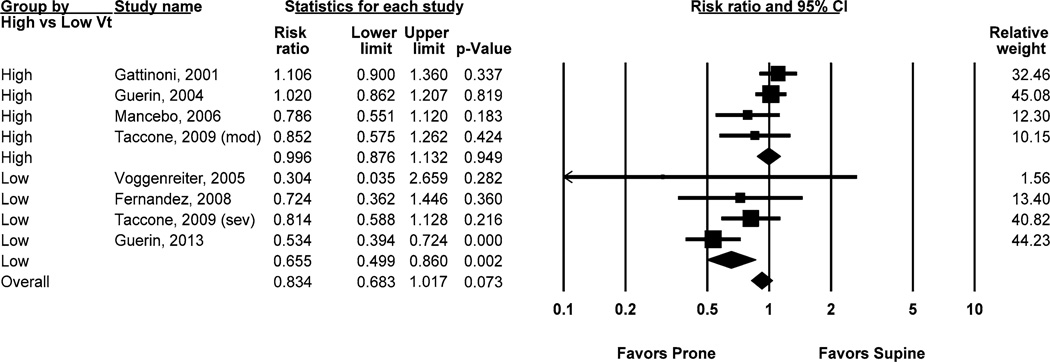
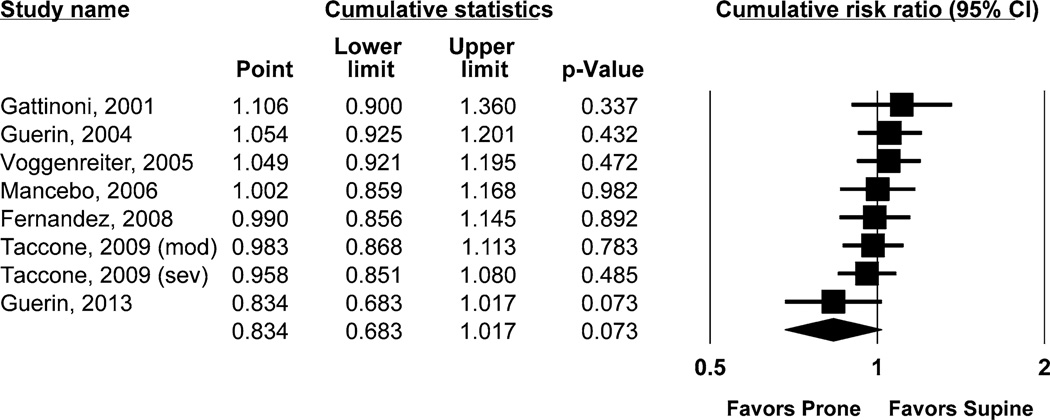
Among individual studies, only the most recent trial by Guérin et al. [19] demonstrated significantly lower mortality with prone positioning. Collectively, risk ratio of death at 60 days was 0.83 (95 % CI 0.68–1.02; p = 0.073) (Fig. 2). Significant heterogeneity was found across studies (Q-statistic = 19.6; I2 = 64 %; p = 0.006). Cumulative meta-analysis with successive addition of increasingly recent studies demonstrated a clear time trend toward reduced mortality with prone positioning (Fig. 3).
Fig. 2.
Effect of prone positioning on mortality, overall and stratified by mean baseline tidal volume. High tidal volume was defined as >8 ml/kg predicted body weight and low tidal volume as ≤8 ml/kg predicted body weight
Fig. 3.
Cumulative meta-analysis forest plot over time, demonstrating a trend favoring prone positioning with inclusion of each successive study
Effect of proning stratified by tidal volume
To test the a priori hypothesis that prone positioning reduces mortality only when injurious high tidal volumes are avoided, stratified analysis was performed by high (>8 ml/kg PBW) versus low (≤8 ml/kg PBW) mean baseline tidal volume (Fig. 2). After stratification, prone positioning was associated with a significant decrease in risk ratio of death for studies that used low tidal volumes (RR = 0.66; 95 % CI 0.50–0.86; p = 0.002) but not high tidal volumes (RR 1.00; 95 % CI 0.88–1.13; p = 0.949). The difference in risk ratio of death between strata was highly significant (p < 0.001). Stratification by tidal volume also substantially reduced heterogeneity. I2 decreased from 64 % in the combined model to 11 and 25 % in the stratified high and low tidal volume models, respectively. The Q-statistic similarly decreased from 19.6 in the combined model to 3.4 and 4.0 in the stratified high and low tidal volume models, respectively. Within-stratum heterogeneity was not significant in either group. Meta-regression demonstrated a dose–response relationship between mean baseline tidal volume (in ml/kg PBW) and log-transformed risk ratio of death at 60 days with proning. A decrease in mean baseline tidal volume of 1 ml/kg PBW was associated with a decrease in risk ratio of death by 16.7 % (95 % CI 6.1–28.3; p = 0.001). Adjusted analysis accounting for potential confounders and other predictors to yield a more reliable meta-regression effect estimate could not be performed because of the small number of included studies.
In post hoc analysis, data were re-analyzed incorporating Taccone et al. [18] as a single study to determine whether the a priori decision to treat this reference as two studies affected the results. Treating Taccone et al. as a single study had no substantial impact on the risk ratio of death with prone positioning (RR = 0.67; 95 % CI 0.49–0.92; p = 0.014 for studies with low mean baseline tidal volume). Moreover, stratifying the analysis instead by whether the protocol specified a low tidal volume strategy produced the same groupings achieved with stratification by mean baseline tidal volume.
Other potential sources of heterogeneity
Stratified analysis by high (≥12 h/day) or low (<12 h/ day) proning dose demonstrated a significant reduction in mortality with high doses (RR = 0.71; 95 % CI 0.56–0.90; p = 0.004) but not low doses (RR = 1.05; 95 % CI 0.92–1.19; p = 0.472). However, substantial within-stratum heterogeneity remained among studies of high proning dose, with a Q-statistic of 5.4 and I2 of 44 %. Duration of respiratory failure prior to proning was not pursued because it was only reported in the four most recent studies [16–19] (Table 1).
Mean SAPS score was strikingly similar across all studies (Table 2) such that analysis by overall illness severity was not pursued as a source of heterogeneity. Similarly, while maximum allowed PaO2:FiO2 for patient inclusion decreased with successive studies (Table 1), mean PaO2:FiO2 at enrollment ranged between 100 and 161 mmHg for six of the seven included trials [13, 14, 16–19] (Table 2). The lone trial with a higher PaO2:FiO2 also was the smallest study included in the analysis [15]. Therefore, analysis by mean baseline PaO2:FiO2 was not pursued.
Assessment for publication bias
Funnel plot analysis, Egger’s regression, and Begg-Mazumdar rank correlation did not suggest evidence of publication bias (Kendall’s tau = −0.286, p = 0.322; Egger’s regression intercept −1.94; p = 0.129).
Discussion
This meta-analysis demonstrates that prone positioning significantly reduces mortality from ARDS when used with low tidal volume ventilation. No such significant benefit was seen among studies employing higher tidal volumes or when all studies were considered irrespective of tidal volume. Stratification by low or high mean tidal volume at baseline accounted for more than half the heterogeneity observed in the unstratified analysis. Two unique aspects of this study may account for these findings differing from prior meta-analyses.
First, prior meta-analyses focused on disease-related factors, namely degree of hypoxemia [21, 22] and overall illness severity [20], to account for heterogeneity and to suggest reasons for failure of demonstrable mortality benefit in clinical trials. Yet, mean SAPS score and PaO2:FiO2 were remarkably similar across studies, even as inclusion criteria for later studies targeted more severely ill patients. In fact, only one trial reported a mean baseline PaO2:FiO2 above 161 mmHg [15]. Additionally, only one trial performed stratified randomization by degree of hypoxemia [18]. All other trials included, without stratification, patients who would be classified with both moderate and severe ARDS by the Berlin definition [5], and three trials [13–15] included patients with mild ARDS in addition to moderate and severe forms. Similarly, only one trial performed stratified randomization by illness severity [17]. No other trials accounted a priori for effect modification by illness severity in the study design. Differences in hypoxemia and illness severity are patient-specific factors that would best be evaluated with an individual patient-level meta-analysis.
By contrast, this study focuses on tidal volume, a modifiable treatment-related factor. Because tidal volume was protocolized inmost trials, it can be assessed reasonably with a study-level meta-analysis. As a result, between-study heterogeneity measured by the Q and I2 statistics is reduced by more than half when stratified by mean baseline tidal volume, indicating tidal volume alone accounted for over half the heterogeneity in the combined meta-analysis. Importantly, post hoc analysis stratified by protocol-specified tidal volume strategy yielded a near-identical effect estimate, alleviating concern about using mean baseline tidal volume to infer management for the length of the study.
Second, this analysis includes the recent study by Guérin et al. [19], the first randomized trial to demonstrate that prone positioning reduces mortality from ARDS. This trial achieved the lowest tidal volumes of all proning studies and had near-universal use of neuromuscular blockade early in the course of ARDS, two proven therapies [2, 4] that may augment the potential benefits of prone positioning. Additionally, Guérin et al. maintained patients in the prone position for an average of 17 ± 3 h/day. This dose is comparable to the most recent trials of prone positioning but far longer than earlier such trials. Proning dose has been suggested previously to correlate with survival [22].
This meta-analysis is limited by the small number of robust studies published on prone positioning for ARDS. Only randomized controlled trials were included to avoid risk of confounding by indication in observational studies from use of prone positioning as rescue for refractory hypoxemia [36–38]. Studies excluded from this analysis but considered in previous meta-analyses involved patients without ARDS [39, 40], neonatal and pediatric patients only [41], high-frequency oscillatory ventilation as the control arm [42], or observational study design without a clearly defined control group [43]. Some of the heterogeneity in prior meta-analyses may have been avoided here by defining more focused inclusion criteria.
It is possible the effect modification attributed here to low tidal volumes is due to other explanatory factors [25]. Stratification by high (≥12 h/day) or low (<12 h/day) proning doses demonstrated a significant reduction in mortality in the high-dose group. However, this approach explained less than one-third of overall heterogeneity between studies (I2 = 44 % in the high-dose proning stratum, compared to I2 = 64 % overall). By comparison, little heterogeneity remained with stratification by mean baseline tidal volume (I2 = 25 % in the low tidal volume stratum), indicating stratification by tidal volume better explained heterogeneity across studies. Still, it is likely that both low tidal volumes and increased proning duration make important contributions to the findings here.
Duration of respiratory failure prior to proning could not be considered here because of underreporting in included trials and may partially account for divergent study findings. While prone positioning may be more effective with increasing ARDS severity [21], similar mean baseline PaO2:FiO2 among six of the seven included trials precluded meaningful trial-level analysis. Furthermore, critical care for other conditions associated with ARDS, most notably sepsis [44], has improved considerably over the same period that low tidal volumes became widely adopted. Regardless, the conclusion regarding the benefit of prone positioning in the modern era would not change. An individual patient-level meta-analysis would provide important insights into the relative contribution of tidal volume, ARDS severity, proning dose, and other potential explanatory factors.
Importantly, the current literature does not capture the potential learning curve of newly introducing prone positioning to an inexperienced intensive care unit. Complications were rare in the experienced centers performing clinical trials of prone positioning [21]. However, complication rates during an introductory period, including dislodgment of life-sustaining tubes and equipment, may well be higher when proning is done in previously inexperienced centers.
Finally, the existing literature fails to consider the role for prone positioning with high-PEEP strategies (Table 1). Most recent trials targeting alveolar recruitment and prevention of atelectrauma have advocated for applying considerably higher PEEP for a given FiO2 requirement [45–50] as part of an open lung-protective approach. A high-PEEP strategy is supported by a previous patient-level meta-analysis that demonstrated reduced mortality from moderate or severe ARDS with this approach [33]. Yet, even the most recent study by Guérin et al. [19] used precisely the low-PEEP strategy from the ARDS Network ALVEOLI trial [45]. Whether the clinical benefit of prone positioning holds true for patients receiving higher PEEP strategies is unknown, although physiological effects may be synergistic [51]. Confirmatory trials must evaluate prone positioning with concurrent open lung-protective ventilation to determine the potential role for broad adoption of prone positioning.
Acknowledgments
The authors would like to thank Gregor Vog-genreiter, MD, for providing additional data for the analysis [15], Paul A. Bain, PhD, for his assistance with the literature search strategy, and Michael A. Stoto, PhD, for his assistance in development of the analysis plan. This work was supported by NIH T32 HL007633, K24 HL093218, and UM1 HL108724.
Footnotes
Conflicts of interest On behalf of all authors, the corresponding author states that there is no conflict of interest.
Contributor Information
Jeremy R. Beitler, Division of Pulmonary and Critical Care Medicine, Brigham and Women’s Hospital, 221 Longwood Avenue, BLI 036, Boston, MA 02115, USA, jbeitler@partners.org, Tel.: +617-732-5619, Fax: +617-732-7337
Shahzad Shaefi, Department of Anesthesia, Critical Care, and Pain Medicine, Beth Israel Deaconess Medical Center, Boston, MA, USA.
Sydney B. Montesi, Pulmonary and Critical Care Unit, Massachusetts General Hospital, Boston, MA, USA
Amy Devlin, Division of Rheumatology, Beth Israel Deaconess Medical Center, Boston, MA, USA.
Stephen H. Loring, Department of Anesthesia, Critical Care, and Pain Medicine, Beth Israel Deaconess Medical Center, Boston, MA, USA
Daniel Talmor, Department of Anesthesia, Critical Care, and Pain Medicine, Beth Israel Deaconess Medical Center, Boston, MA, USA.
Atul Malhotra, Division of Pulmonary and Critical Care Medicine, University of California, San Diego, San Diego, CA, USA.
References
- 1.Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–1693. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- 2.Brower RG, Matthay MA, Morris A, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 3.Malhotra A. Low-tidal-volume ventilation in the acute respiratory distress syndrome. N Engl J Med. 2007;357:1113–1120. doi: 10.1056/NEJMct074213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Papazian L, Forel JM, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363:1107–1116. doi: 10.1056/NEJMoa1005372. [DOI] [PubMed] [Google Scholar]
- 5.ARDS Definition Task Force. Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 6.Schmidt M, Zogheib E, Rozé H, et al. The PRESERVE mortality risk score and analysis of long-term outcomes after extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. Intensive Care Med. 2013;39:1704–1713. doi: 10.1007/s00134-013-3037-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Piehl MA, Brown RS. Use of extreme position changes in acute respiratory failure. Crit Care Med. 1976;4:13–14. doi: 10.1097/00003246-197601000-00003. [DOI] [PubMed] [Google Scholar]
- 8.Pappert D, Rossaint R, Slama K, et al. Influence of positioning on ventilation-perfusion relationships in severe adult respiratory distress syndrome. Chest. 1994;106:1511–1516. doi: 10.1378/chest.106.5.1511. [DOI] [PubMed] [Google Scholar]
- 9.Gattinoni L, Pesenti A, Carlesso E. Body position changes redistribute lung computed-tomographic density in patients with acute respiratory failure: impact and clinical fallout through the following 20 years. Intensive Care Med. 2013;39:1909–1915. doi: 10.1007/s00134-013-3066-x. [DOI] [PubMed] [Google Scholar]
- 10.Chiumello D, Taccone P, Berto V, et al. Long-term outcomes in survivors of acute respiratory distress syndrome ventilated in supine or prone position. Intensive Care Med. 2012;38:221–229. doi: 10.1007/s00134-011-2445-4. [DOI] [PubMed] [Google Scholar]
- 11.Pelosi P, Tubiolo D, Mascheroni D, et al. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am J Respir Crit Care Med. 1998;157:387–393. doi: 10.1164/ajrccm.157.2.97-04023. [DOI] [PubMed] [Google Scholar]
- 12.Mackenzie CF. Anatomy, physiology, and pathology of the prone position and postural drainage. Crit Care Med. 2001;29:1084–1085. doi: 10.1097/00003246-200105000-00046. [DOI] [PubMed] [Google Scholar]
- 13.Gattinoni L, Tognoni G, Pesenti A, et al. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med. 2001;345:568–573. doi: 10.1056/NEJMoa010043. [DOI] [PubMed] [Google Scholar]
- 14.Guérin C, Gaillard S, Lemasson S, et al. Effects of systematic prone positioning in hypoxemic acute respiratory failure: a randomized controlled trial. JAMA. 2004;292:2379–2387. doi: 10.1001/jama.292.19.2379. [DOI] [PubMed] [Google Scholar]
- 15.Voggenreiter G, Aufmkolk M, Stiletto RJ, et al. Prone positioning improves oxygenation in post-traumatic lung injury—a prospective randomized trial. J Trauma. 2005;59:333–341. doi: 10.1097/01.ta.0000179952.95921.49. [DOI] [PubMed] [Google Scholar]
- 16.Mancebo J, Fernández R, Blanch L, et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med. 2006;173:1233–1239. doi: 10.1164/rccm.200503-353OC. [DOI] [PubMed] [Google Scholar]
- 17.Fernández R, Trenchs X, Klamburg J, et al. Prone positioning in acute respiratory distress syndrome: a multicenter randomized clinical trial. Intensive Care Med. 2008;34:1487–1491. doi: 10.1007/s00134-008-1119-3. [DOI] [PubMed] [Google Scholar]
- 18.Taccone P, Pesenti A, Latini R, et al. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2009;302:1977–1984. doi: 10.1001/jama.2009.1614. [DOI] [PubMed] [Google Scholar]
- 19.Guérin C, Reignier J, Richard J-C, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368:2159–2168. doi: 10.1056/NEJMoa1214103. [DOI] [PubMed] [Google Scholar]
- 20.Alsaghir AH, Martin CM. Effect of prone positioning in patients with acute respiratory distress syndrome: a meta-analysis. Crit Care Med. 2008;36:603–609. doi: 10.1097/01.CCM.0000299739.98236.05. [DOI] [PubMed] [Google Scholar]
- 21.Sud S, Friedrich JO, Taccone P, et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med. 2010;36:585–599. doi: 10.1007/s00134-009-1748-1. [DOI] [PubMed] [Google Scholar]
- 22.Abroug F, Ouanes-Besbes L, Dachraoui F, et al. An updated study-level meta-analysis of randomised controlled trials on proning in ARDS and acute lung injury. Crit Care. 2011;15:R6. doi: 10.1186/cc9403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li G, Malinchoc M, Cartin-Ceba R, et al. Eight-year trend of acute respiratory distress syndrome: a population-based study in Olmsted County, Minnesota. Am J Respir Crit Care Med. 2011;183:59–66. doi: 10.1164/rccm.201003-0436OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Malhotra A, Ayas N, Kacmarek R. Prone positioning of patients with acute respiratory failure. N Engl J Med. 2002;346:295–297. [PubMed] [Google Scholar]
- 25.Gattinoni L, Taccone P, Carlesso E, Marini JJ. Prone position in acute respiratory distress syndrome. Rationale, indications, and limits. Am J Respir Crit Care Med. 2013;188:1286–1293. doi: 10.1164/rccm.201308-1532CI. [DOI] [PubMed] [Google Scholar]
- 26.Richardson WS, Wilson MC, Nishikawa J, Hayward RS. The well-built clinical question: a key to evidence-based decisions. ACP J Club. 1995;123:A12–A13. [PubMed] [Google Scholar]
- 27.Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med. 2012;38:1573–1582. doi: 10.1007/s00134-012-2682-1. [DOI] [PubMed] [Google Scholar]
- 28.Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–824. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 29.Earle CC, Pham B, Wells GA. An assessment of methods to combine published survival curves. Med Decis Making. 2000;20:104–111. doi: 10.1177/0272989X0002000113. [DOI] [PubMed] [Google Scholar]
- 30.Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17:1–12. doi: 10.1016/0197-2456(95)00134-4. [DOI] [PubMed] [Google Scholar]
- 31.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 32.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Briel M, Meade M, Mercat A, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA. 2010;303:865–873. doi: 10.1001/jama.2010.218. [DOI] [PubMed] [Google Scholar]
- 34.Putensen C, Theuerkauf N, Zinserling J, et al. Meta-analysis: ventilation strategies and outcomes of the acute respiratory distress syndrome and acute lung injury. Ann Intern Med. 2009;151:566–576. doi: 10.7326/0003-4819-151-8-200910200-00011. [DOI] [PubMed] [Google Scholar]
- 35.Dellinger RP, Levy MM, Rhodes A, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med. 2013;39:165–228. doi: 10.1007/s00134-012-2769-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Raoof S, Goulet K, Esan A, et al. Severe hypoxemic respiratory failure. Chest. 2010;137:1437–1448. doi: 10.1378/chest.09-2416. [DOI] [PubMed] [Google Scholar]
- 37.Kimmoun A, Guerci P, Bridey C, et al. Prone positioning use to hasten veno-venous ECMO weaning in ARDS. Intensive Care Med. 2013;39:1877–1879. doi: 10.1007/s00134-013-3007-8. [DOI] [PubMed] [Google Scholar]
- 38.Boissier F, Katsahian S, Razazi K, et al. Prevalence and prognosis of cor pulmonale during protective ventilation for acute respiratory distress syndrome. Intensive Care Med. 2013;39:1725–1733. doi: 10.1007/s00134-013-2941-9. [DOI] [PubMed] [Google Scholar]
- 39.Beuret P, Carton M-J, Nourdine K, et al. Prone position as prevention of lung injury in comatose patients: a prospective, randomized, controlled study. Intensive Care Med. 2002;28:564–569. doi: 10.1007/s00134-002-1266-x. [DOI] [PubMed] [Google Scholar]
- 40.Watanabe I, Fujihara H, Sato K, et al. Beneficial effect of a prone position for patients with hypoxemia after transthoracic esophagectomy. Crit Care Med. 2002;30:1799–1802. doi: 10.1097/00003246-200208000-00021. [DOI] [PubMed] [Google Scholar]
- 41.Curley MA, Hibberd PL, Fineman LD, et al. Effect of prone positioning on clinical outcomes in children with acute lung injury. JAMA. 2005;294:229–237. doi: 10.1001/jama.294.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Papazian L, Gainnier M, Marin V, et al. Comparison of prone positioning and high-frequency oscillatory ventilation in patients with acute respiratory distress syndrome. Crit Care Med. 2005;33:2162–2171. doi: 10.1097/01.ccm.0000181298.05474.2b. [DOI] [PubMed] [Google Scholar]
- 43.Chan M-C, Hsu J-Y, Liu H–H, et al. Effects of prone position on inflammatory markers in patients with ARDS due to community-acquired pneumonia. J Formos Med Assoc. 2007;106:708–716. doi: 10.1016/S0929-6646(08)60032-7. [DOI] [PubMed] [Google Scholar]
- 44.Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–1377. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 45.Brower RG, Lanken PN, MacIntyre N, et al. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med. 2004;351:327–336. doi: 10.1056/NEJMoa032193. [DOI] [PubMed] [Google Scholar]
- 46.Meade MO, Cook DJ, Guyatt GH, et al. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299:637–645. doi: 10.1001/jama.299.6.637. [DOI] [PubMed] [Google Scholar]
- 47.Mercat A, Richard J-CM, Vielle B, et al. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299:646–655. doi: 10.1001/jama.299.6.646. [DOI] [PubMed] [Google Scholar]
- 48.Talmor D, Sarge T, Malhotra A, et al. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med. 2008;359:2095–2104. doi: 10.1056/NEJMoa0708638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ferguson ND, Cook DJ, Guyatt GH, et al. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med. 2013;368:795–805. doi: 10.1056/NEJMoa1215554. [DOI] [PubMed] [Google Scholar]
- 50.Amato MB, Barbas CS, Medeiros DM, et al. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338:347–354. doi: 10.1056/NEJM199802053380602. [DOI] [PubMed] [Google Scholar]
- 51.Cornejo RA, Díaz JC, Tobar EA, et al. Effects of prone positioning on lung protection in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2013;188:440–448. doi: 10.1164/rccm.201207-1279OC. [DOI] [PubMed] [Google Scholar]