Abstract
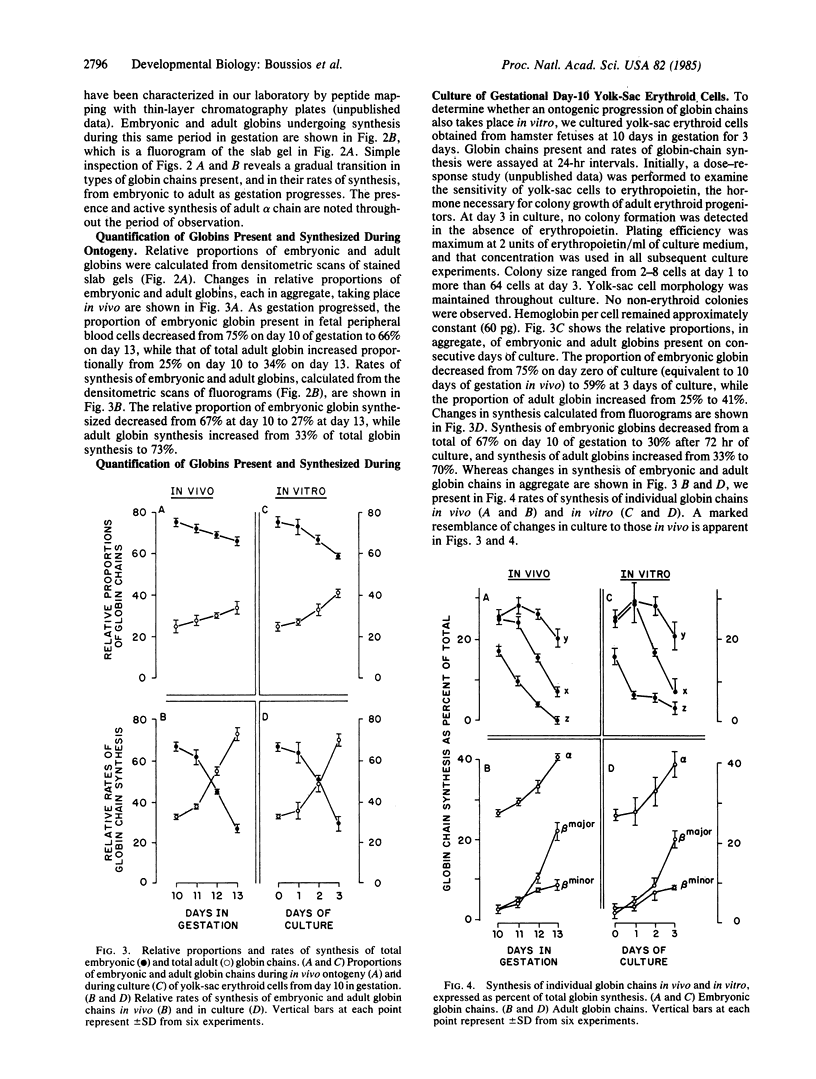
During mammalian hemoglobin ontogeny, synthesis of the earliest globin chains (embryonic) is ultimately replaced by synthesis of globin chains (adult) characteristic of the fully formed organism. Elements of control of initiation, progression, and completion of globin-chain ontogeny are poorly understood. In search of a cell culture system in which ontogeny might be studied under closely controlled experimental conditions, we chose erythroid cells of the hamster embryo. First, the ontogeny of globin chains was defined in these yolk-sac-derived erythroid cells from day 10 through day 13 in gestation. Amounts of individual embryonic and adult globin chains were quantified, as were their rates of synthesis. Next, analogous studies were performed on yolk-sac erythroid cells from day 10 in gestation (prior to the appearance of fetal liver) grown in culture for 3 days, corresponding to days 10-13 in vivo. The ontogenic program in culture was virtually identical to that in vivo. Approximately 70% of active globin synthesis was embryonic at day 10 in gestation (day 0 of culture), declining to 30% by day 13 in gestation (day 3 of culture). Whereas only trace synthesis of the adult non-alpha chains (beta major and beta minor) were initially observed, their combined active synthesis achieved a level of approximately 30% 3 days later both in vivo and in culture. Cell hemoglobin content and cell morphology were similar in both systems. We conclude that an ontogenic program for globin-chain synthesis exists in these primitive erythroid cells, overriding possible influences of cell environment. Further, we suggest that these cells in culture provide a means of examining cell mechanisms associated with globin-gene ontogeny under controlled experimental conditions.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alter B. P., Goff S. C. A murine model for the switch from fetal to adult hemoglobin during ontogeny. Blood. 1980 Dec;56(6):1100–1105. [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Boussios T., Bertles J. F., Clegg J. B. Simultaneous expression of globin genes for embryonic and adult hemoglobins during mammalian ontogeny. Science. 1982 Dec 17;218(4578):1225–1227. doi: 10.1126/science.6183746. [DOI] [PubMed] [Google Scholar]
- Boyer S. H., Crosby E. F., Noyes A. N. Hemoglobin switching in non-anemic sheep. I. Mediation by plasma from anemic animals. Johns Hopkins Med J. 1968 Aug;123(2):85–91. [PubMed] [Google Scholar]
- Brotherton T. W., Chui D. H., Gauldie J., Patterson M. Hemoglobin ontogeny during normal mouse fetal development. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2853–2857. doi: 10.1073/pnas.76.6.2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fantoni A., Bank A., Marks P. A. Globin composition and synthesis of hemoglobins in developing fetal mice erythroid cells. Science. 1967 Sep 15;157(3794):1327–1329. doi: 10.1126/science.157.3794.1327. [DOI] [PubMed] [Google Scholar]
- Fantoni A., De la Chapelle A., Marks P. A. Synthesis of embryonic hemoglobins during erythroid cell development in fetal mice. J Biol Chem. 1969 Feb 25;244(4):675–681. [PubMed] [Google Scholar]
- Iscove N. N., Sieber F., Winterhalter K. H. Erythroid colony formation in cultures of mouse and human bone marrow: analysis of the requirement for erythropoietin by gel filtration and affinity chromatography on agarose-concanavalin A. J Cell Physiol. 1974 Apr;83(2):309–320. doi: 10.1002/jcp.1040830218. [DOI] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- McLeod D. L., Shreeve M. M., Axelrad A. A. Improved plasma culture system for production of erythrocytic colonies in vitro: quantitative assay method for CFU-E. Blood. 1974 Oct;44(4):517–534. [PubMed] [Google Scholar]
- Miller R. G., Phillips R. A. Separation of cells by velocity sedimentation. J Cell Physiol. 1969 Jun;73(3):191–201. doi: 10.1002/jcp.1040730305. [DOI] [PubMed] [Google Scholar]
- Whitney J. B., 3rd Differential control of the synthesis of two hemoglobin beta chains in normal mice. Cell. 1977 Dec;12(4):863–871. doi: 10.1016/0092-8674(77)90150-7. [DOI] [PubMed] [Google Scholar]
- Wong P. M., Clarke B. J., Carr D. H., Chui D. H. Adult hemoglobins are synthesized in erythroid colonies in vitro derived from murine circulating hemopoietic progenitor cells during embryonic development. Proc Natl Acad Sci U S A. 1982 May;79(9):2952–2956. doi: 10.1073/pnas.79.9.2952. [DOI] [PMC free article] [PubMed] [Google Scholar]