Abstract
Mitochondrial diseases are an unusually genetically and phenotypically heterogeneous group of disorders, which are extremely challenging to treat. Currently, apart from supportive therapy, there are no effective treatments for the vast majority of mitochondrial diseases. Huge scientific effort, however, is being put into understanding the mechanisms underlying mitochondrial disease pathology and developing potential treatments. To date, a variety of treatments have been evaluated by randomized clinical trials, but unfortunately, none of these has delivered breakthrough results. Increased understanding of mitochondrial pathways and the development of many animal models, some of which are accurate phenocopies of human diseases, are facilitating the discovery and evaluation of novel prospective treatments. Targeting reactive oxygen species has been a treatment of interest for many years; however, only in recent years has it been possible to direct antioxidant delivery specifically into the mitochondria. Increasing mitochondrial biogenesis, whether by pharmacological approaches, dietary manipulation or exercise therapy, is also currently an active area of research. Modulating mitochondrial dynamics and mitophagy and the mitochondrial membrane lipid milieu have also emerged as possible treatment strategies. Recent technological advances in gene therapy, including allotopic and transkingdom gene expression and mitochondrially targeted transcription activator-like nucleases, have led to promising results in cell and animal models of mitochondrial diseases, but most of these techniques are still far from clinical application.
Linked Articles
This article is part of a themed issue on Mitochondrial Pharmacology: Energy, Injury & Beyond. To view the other articles in this issue visit http://dx.doi.org/10.1111/bph.2014.171.issue-8
Keywords: mitochondrial diseases, models for mitochondrial disorders, treatments for mitochondrial disorders, clinical trials for mitochondrial disorders, nutritional and cofactor support in mitochondrial disorders, mitochondrial biogenesis, gene therapy for mitochondrial diseases
Introduction
Recently, the term mitochondrial disorders has become very popular, finding its place in reference to many metabolic diseases, Alzheimer's disease, Parkinson's disease, type 2 diabetes and aging (Patti and Corvera, 2010; Coskun et al., 2012; Bratic and Larsson, 2013). In this review, the term mitochondrial diseases denotes a group of inborn errors affecting the oxidative phosphorylation (OXPHOS) system of the mitochondrion. Mitochondrial diseases have a prevalence of approximately 1 in 5000 (Thorburn, 2004). They are a common and unusually heterogeneous group of metabolic disorders, which may present in patients of any age and virtually with any symptoms (Munnich et al., 1992). The most important function of OXPHOS is to produce ATP, and therefore, it is not surprising that any defects in this system will be most evident in tissues with high energy demands, for example, the skeletal muscle, brain, liver or heart.
A mitochondrial disorder is part of the differential diagnosis in individuals presenting with myopathy, cardiomyopathy, peripheral neuropathies, stroke-like episodes, seizures, external ophthalmoplegia, sensorineural hearing loss (SNHL), exercise intolerance, diabetes, liver failure, Fanconi syndrome or gastrointestinal symptoms such as nausea, vomiting, pseudo-obstruction, severe dysmotility and faltering growth. These symptoms can occur in isolation or be part of a multisystemic disease presentation (Rahman and Hanna, 2009; Vafai and Mootha, 2012). Currently, there are no effective treatments for the vast majority of mitochondrial disorders and there is an urgent need to develop and evaluate novel therapies. This article discusses the challenges to developing treatment strategies for these complex disorders; models for assessing efficacy of candidate therapies; current therapeutic approaches and the results of previous clinical trials; and the most promising treatments on the horizon, including pharmacological and other strategies for stimulating mitochondrial biogenesis, antioxidant approaches and gene therapy for mitochondrial DNA (mtDNA) and nuclear DNA-encoded mitochondrial disorders.
Mitochondrial biology and disease
The mitochondrial respiratory chain (RC) and OXPHOS system
The OXPHOS system is composed of five multimeric complexes (complexes I–IV comprise the RC and complex V is the ATP synthase), which are embedded in the mitochondrial inner membrane. The complexes are composed of multiple subunits, encoded by mtDNA or nuclear DNA. Overall, the RC is composed of approximately 90 proteins. Thirteen of these proteins are encoded by mtDNA, a circular double-stranded DNA molecule, ∼16.6 kb long. Additionally, the mtDNA also encodes 2 rRNAs and 22 tRNAs, which are required for the intramitochondrial synthesis of these 13 proteins. The remaining proteins are translated in the cytosol and transported through the TIM/TOM complex in their precursor form. Once in the correct mitochondrial compartment, the precursor is assembled to form the final protein (Wiedemann et al., 2004).
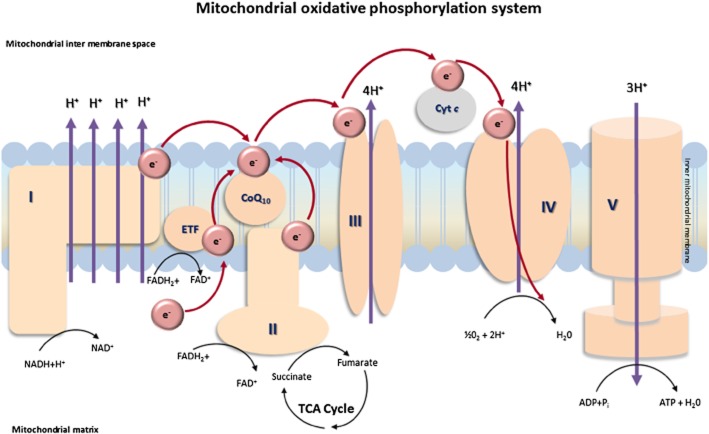
The purpose of the RC is to generate a proton-motive force (complexes I, III and IV are proton-pumping enzymes) across the mitochondrial inner membrane, which is utilized to power ATP synthase, allowing it to produce ATP from ADP and inorganic phosphate – the final step of OXPHOS. Electrons in the form of NADH and FADH2, which are derived from the Krebs cycle and β-oxidation, provide the free energy required to pump protons across the membrane. Complex I (NADH : ubiquinone oxidoreductase) oxidizes NADH to NAD+ and H+ and passes two electrons to ubiquinone (coenzyme Q10, CoQ10) reducing it to ubiquinol. Simultaneously, complex II (succinate : ubiquinone oxidoreductase), the enzyme that links the Krebs cycle to OXPHOS, oxidizes FADH to FAD+ and H+ and reduces ubiquinone, while also oxidizing succinate to fumarate, both of which are intermediates of the Krebs cycle. From ubiquinol, electrons are transported to complex III (ubiquinol : cytochrome c oxidoreductase), which facilitates the transfer of electrons to cytochrome c, which in turn reduces complex IV (cytochrome c oxidase, COX). Finally, electrons are donated to molecular oxygen and water is formed (Wallace et al., 2010) (Figure 1).
Figure 1.
Mitochondrial OXPHOS system. I, NADH : ubiquinone oxidoreductase; II, succinate : ubiquinone oxidoreductase; III, ubiquinol : cytochrome c oxidoreductase; IV, COX; V, ATP synthase.
Mitochondrial genetics and disease
Mitochondrial disorders may be caused by mutations in the mtDNA or in the nuclear DNA. Disorders originating from mtDNA mutations follow a uniparental mode of inheritance, and can only be transmitted from the mother to the child. Disorders caused by mtDNA mutations are complicated by the phenomenon of heteroplasmy (coexistence of mutant and wild-type mtDNA), which arises because of the high copy number of mtDNA. For each mutation, a specific critical threshold of mutation load exists, above which RC function is impaired and disease ensues (Taylor and Turnbull, 2005). Generally speaking, lower mutation load is usually associated with less severe symptoms presenting in adulthood (Rahman and Hanna, 2009).
Mitochondrial disorders due to nuclear DNA mutations follow Mendelian inheritance (DiMauro, 1999). The majority of subunits (∼77), which comprise the mitochondrial RC, are encoded by nuclear DNA. Many additional proteins required for correct functioning of the RC are also encoded by nuclear DNA, which explains why the majority of mitochondrial disorders are caused by mutations in nuclear DNA. Overall, 1500 genes have been suggested as potential causes of mitochondrial disease (Calvo et al., 2006), and so far, more than 130 of these have been linked to various mitochondrial disorders (Vafai and Mootha, 2012; Rahman, 2013). These genes are involved in a range of functions including assembly of RC enzyme complexes, maintenance and expression of the mtDNA, cofactor metabolism and biosynthesis and metabolism of mitochondrial membrane lipids. Wide-scale adoption of next generation sequencing approaches has led to a rapid escalation in the number of known disease genes in the last few years, and it is likely that the number of genes linked to mitochondrial disease will soon exceed 200. This huge genetic heterogeneity contributes to the phenotypic heterogeneity of mitochondrial diseases, and presents enormous challenges in devising effective therapies for these disorders.
Challenges in treatment of mitochondrial diseases
Difficulties in designing and implementing clinical trials
Development of successful treatments for mitochondrial diseases has proved to be extremely difficult. For other inborn errors of metabolism, therapeutic options include reduction of metabolic load (by dietary manipulation), removal of toxic metabolites, enzyme replacement, enzyme enhancement and organ (liver or bone marrow) transplantation. However, the situation for mitochondrial disorders is rather more complex, for a number of reasons. The main challenges are caused by the extreme genetic and phenotypic heterogeneity of mitochondrial disorders, as discussed above, making it very difficult to collect sufficiently large groups of patients to conduct adequately powered, statistically valid, randomized, double-blinded, placebo-controlled clinical trials. Ideally, clinical trials should be conducted on a group of patients with the same genetic defect, with the same presentation and biochemical findings, at a similar stage of disease progression and, in the case of mutations in mtDNA, a similar mutation load. When dealing with mitochondrial patients collecting a large group of patients that would meet these criteria is virtually impossible in a single centre. Clinical trials that could satisfy these criteria could only exist in the context of multicentre international collaborations (Suomalainen, 2011). A final challenge in implementing clinical trials for mitochondrial disease is a lack of clinically relevant, universally agreed, validated outcome measures.
Other challenges
Despite enormous progress in understanding mitochondrial biology and mitochondrial diseases in recent years, delivering therapeutic molecules to mitochondria is still challenging. This is mainly due to the relative inaccessibility of the mitochondrion, so that mitochondrially targeted drugs need to pass across several membranes in order to reach the matrix (Heller et al., 2012). Some techniques that allow synthetic molecules to be mitochondrially targeted include utilizing the mitochondrial targeting signal peptide, which is a short peptide chain that in vivo is attached to proteins that are translated within the cytosol but destined for the mitochondria; conjugating lipophilic cations to small molecules to allow them to accumulate within mitochondria by utilizing the mitochondrial membrane potential; and dequalinium-derived vesicles (so-called DQAsomes) with similar properties to liposomes, which bind DNA and theoretically may be used to deliver small DNA molecules into mitochondria (Yamada et al., 2007).
Recent developments in the therapy of mitochondrial disorders
Completed clinical trials
Owing to the inherent difficulties in clinical trial design, few randomized double-blinded placebo-controlled trials for mitochondrial disease have been completed. These were the subject of an excellent recent review by Douglas Kerr (Kerr, 2013) and were also critically evaluated in a recently updated Cochrane systematic review (Pfeffer et al., 2012). Only 12 studies of 1335 abstracts screened fulfilled Cochrane inclusion criteria for well-conducted unbiased clinical trials (Pfeffer et al., 2012; summarized in Table 1). The treatments that have been investigated under such conditions include dichloroacetate (DCA) in five studies and creatine in four studies, either alone or as a cocktail with CoQ10 and lipoic acid. Single studies examined the efficacy of CoQ10, dimethylglycine and whey-based cysteine supplementation. These clinical trials were mostly conducted in small numbers of patients (9 of the 12 studies included fewer than 17 subjects) and in most cases, the patients had different genetic defects. Dramatic responses were not observed in any of these 12 clinical trials, and no effect was seen in four studies (Table 1). DCA is a pharmacological inhibitor of pyruvate dehydrogenase kinase (PDK), an enzyme that inhibits pyruvate dehydrogenase (PDH). Inhibiting PDK results in reduced lactate levels by maintaining PDH in its active state (Stacpoole et al., 1997). Differing results were observed in the five clinical trials of DCA (Table 1) but, importantly, the randomized, controlled clinical trial of DCA in patients with mitochondrial encephalomyopathy (MELAS) had to be terminated because of peripheral nerve toxicity (Kaufmann et al., 2006). CoQ10 supplementation increased serum CoQ10 levels and decreased serum lactate after 1 min of cycle ergometry but other outcome measures were not significantly changed (Glover et al., 2010). Combined supplementation with creatine, CoQ10 and lipoic acid showed a significant reduction in plasma lactate levels, and slowed the progression of loss of ankle dorsiflexion strength (Rodriguez et al., 2007). Dimethylglycine and whey-based cysteine supplementation showed no significant improvement (Liet et al., 2003; Mancuso et al., 2010). Much has been learned from these completed clinical trials regarding optimization of methodological design and the importance of selecting clinically relevant primary end points. It is encouraging that there was a trend towards larger numbers of participants in the studies conducted in more recent years (Table 1). This is likely to reflect a combination of factors: improved trial design, increased numbers of patients receiving a genetic diagnosis of mitochondrial disease and increased accessibility of clinical trials to patients.
Table 1.
Randomized, double-blinded, placebo-controlled clinical trials in mitochondrial disease
| Treatment | Disease | No. of participants | Type of trial | Outcome | References |
|---|---|---|---|---|---|
| CoQ10 | MELAS, PEO, complex I deficiency, NARP, LHON | 30 | Randomized, placebo-controlled, double-blind crossover | Serum CoQ10 increased, lactate levels decreased after 1 min of cycle ergometry, but no significant change in other endpoints | Glover et al., 2010 |
| Creatine | MELAS and MM | 7 | Randomized, placebo-controlled, double-blind crossover | Increased handgrip strength, NIDT and post-exercise lactate | Tarnopolsky et al., 1997 |
| CPEO and MM | 16 | Randomized, placebo-controlled, double-blind crossover | No effect | Klopstock et al., 2000 | |
| CPEO and KSS | 15 | Randomised, placebo-controlled crossover | No effect | Kornblum et al., 2005 | |
| DCA | MM, CPEO, KSS, Leigh syndrome, MELAS | 11 | Randomised, placebo-controlled, double-blind crossover | Decreased blood lactate, pyruvate and alanine at rest and post-exercise, some improvements in brain MRS | De Stefano et al., 1995 |
| CPEO, MERRF, MM | 8 | Randomized, placebo-controlled, double-blind crossover | Decreased resting and exercise lactate and pyruvate | Vissing et al., 2001 | |
| Mitochondrial RC disorders | 9 | Randomized, placebo-controlled, double-blind crossover | Decreased blood lactate levels during exercise | Duncan et al., 2004 | |
| MELAS m.3243A>G | 30 | Randomized, placebo-controlled crossover | No effect. Study terminated due to side effects (peripheral neuropathy) | Kaufmann et al., 2006 | |
| Congenital lactic acidosis | 43 | Randomized, double-blinded, placebo-controlled parallel group | Reduced blood lactate levels post high carbohydrate meal | Stacpoole et al., 2006 | |
| Dimethylglycine | SLSJ-COX | 5 | Randomized, placebo-controlled crossover | No effect | Liet et al., 2003 |
| Whey-based cysteine | PEO | 13 | Randomized, placebo-controlled, double-blind crossover | Glutathione levels increased. Advanced oxidation protein products and ferric-reducing antioxidant power increased | Mancuso et al., 2010 |
| Combination therapy (creatine, α-lipoic acid and CoQ10) | CPEO, KSS, MELAS, MNGIE, MM | 16 | Randomized, placebo-controlled, double-blind crossover | Decreased plasma lactate, slower disease progression (measured by peak angle dorsiflexion strength). | Rodriguez et al., 2007 |
CPEO, chronicprogressive external ophthalmoplegia; MERRF, myoclonic epilepsy, ragged red fibres; NIDT, non-ischaemic, isometric, dorsiflexion torque; SLSJ-COX, Saguenay-Lac-Saint-Jean COX deficiency.
Outcome measures and natural history studies
When investigating potential therapeutic strategies, the correct choice of outcome measure(s) is very important. For mitochondrial disorders, some of the most commonly measured outcomes include biochemical assessment, such as plasma lactate and pyruvate, and muscle RC enzyme assays; histopathological and histochemical assessment; and neuroimaging including brain MRI and magnetic resonance spectroscopy (MRS) of brain or muscle. However, lactate is not universally elevated in patients with mitochondrial disease and is subject to acute variations so may not be a reliable biomarker in clinical trials. Serial muscle biopsies are invasive; therefore, muscle histology and RC enzyme activities are not practical biomarkers. Neuroimaging also requires general anaesthesia in young children and may not be a sufficiently sensitive method to detect relatively small changes during the period of a clinical trial. Better biomarkers are clearly needed. Fibroblast growth factor-21 (FGF21), a serum cytokine involved in lipid metabolism, has recently been suggested as a potential biomarker for mitochondrial myopathies. In Deletor mice, FGF21 levels correlated with increased COX-negative fibres (Tyynismaa et al., 2010), and subsequently, FGF21 was found to be elevated in patients with mitochondrial disorders with skeletal muscle involvement (Suomalainen et al., 2011). A composite score can be useful in clinical trials, and wide adoption of the same score would facilitate comparison of clinical trials. Examples of composite scores include the Global Assessment of Treatment Efficacy (GATE) score, which was utilized in two clinical trials of DCA (Kaufmann et al., 2006; Stacpoole et al., 2006) and the Newcastle paediatric mitochondrial disease scale (NPMDS), which was used in a study of the novel antioxidant EPI-743 (Phoenix et al., 2006; Martinelli et al., 2012).
Another important recent development is the publication of natural history data from large national cohorts with single gene disorders, such as a nationwide prospective study of nearly 100 Japanese patients with MELAS (Yatsuga et al., 2012) and a multinational retrospective study of >50 patients with SURF1 deficiency (Wedatilake et al., 2013). These historical data will be vital comparative data for future clinical trials.
Development of models for mitochondrial disease
Animal and cell models play a crucial role in understanding and developing treatments for many diseases, and especially so for diseases that are as heterogeneous as mitochondrial disorders.
Cell models
Initial studies involved cell-based models of mitochondrial disease. Yeast mitochondrial disease models have been used in high throughput screening of drug libraries to identify novel candidate therapies (Schwimmer et al., 2006). Patient-derived cell lines, especially lymphocytes and fibroblasts, which can be obtained through relatively non-invasive procedures, are useful models to study mitochondrial function, diseases and potential therapies (Robinson et al., 1990). Although there are many benefits in using these cell lines, some considerations need to be taken into account. For example, one of the main drawbacks in using fibroblasts is that some RC deficiencies are tissue specific, and the defect might not be detectable in these cells. Additionally, both fibroblasts and lymphocytes (healthy and diseased) rely on glycolysis as their main source of energy; therefore, RC assays may not show significant differences in enzyme activities in cells obtained from patients compared with controls. In cell culture, substituting galactose for glucose may be used to help combat this issue, forcing cells to rely on OXPHOS as their main source of ATP, thus allowing any enzyme deficiencies to be more apparent (Robinson, 1996). Myoblasts are also a useful model to study mitochondrial diseases since they more closely resemble the affected tissue, but obtaining muscle tissue does require invasive biopsy and (in children) general anaesthesia. An alternative is to use myogenic transdifferentiation of fibroblasts into myoblasts by MyoD transfection (Bulst et al., 2012). Generation of induced pluripotent stem cells from patient fibroblasts and reprogramming into neuronal cells will allow the development of more relevant models for neurological mitochondrial diseases (Cherry et al., 2013). Further difficulties arise when the cell line is derived from a patient with a mtDNA mutation. As discussed above (section ‘Mitochondrial genetics and disease’) mtDNA mutations are often heteroplasmic with at least two subpopulations being present, one wild type and the other mutated. In patient cell lines, mtDNA populations often drift towards the wild type, although the converse (increasing mutant load) may occur in some instances. Cell lines of higher passage numbers are thus likely to contain less mutant mtDNA (van de Corput et al., 1997). Generation of transmitochondrial cybrids with 100% mutant load can overcome this difficulty; these are created by fusing enucleated cells containing mutant mtDNA with a ‘ρ zero’ nuclear donor cell line, which has been depleted of mtDNA (King and Attardi, 1989) and then selecting for cybrids with 100% mutated mtDNA.
Animal models
One of the earliest animal models for mitochondrial diseases was a Drosophila melanogaster with a technical knockout (TKO) of the gene encoding the mitochondrial ribosomal protein S12. Detailed phenotypic characterization revealed its similarity to mitochondrial sensorineural deafness (Toivonen et al., 2001). Other Drosophila models have been reported more recently (Fernandez-Ayala et al., 2010; Debattisti and Scorrano, 2013). Another animal model for mitochondrial disorders is the nematode Cenorrhabditis elegans but only limited phenotypes could be studied in this simple organism, such as biochemical function and effects on survival, motility and reproduction. One study examined riboflavin supplementation in a C. elegans model of complex I deficiency (mutation in the nuo-1 orthologue of the human NDUFV1 gene), and showed improved assembly and activity of complexes I and IV, increased ATP production, decreased reactive oxygen species (ROS) production and improved general metabolic function (Grad and Lemire, 2006). The last few years have witnessed the development of a plethora of mouse models for mitochondrial disorders (Table 2), generated by various strategies including constitutive and conditional gene KO and mutagenesis with ENU (N-ethyl-N-nitrosourea). Both mtDNA and nuclear genes have been targeted. Examples of mouse models with mtDNA mutations include the Mito-mouse harbouring a heteroplasmic single mtDNA deletion at high mutation load (Inoue et al., 2000), and a mouse model of Leber's hereditary optic neuropathy (LHON) containing the ND6 P25L human mtDNA mutation (Lin et al., 2012). The LHON mouse presents similar features to the human phenotype (Lin et al., 2012) making it a useful tool to investigate potential pharmacological and gene therapy treatment approaches for this disease.
Table 2.
Examples of mouse models for mitochondrial diseases
| Gene | Human phenotype | Mouse model | Mouse phenotype | References |
|---|---|---|---|---|
| ANT1 | ADPEO (cardiomyopathy in only recessive case reported) | Ant1 (−/−) | Ragged-red fibres, mitochondrial proliferation, cardiomyopathy, very high serum lactate levels. | Graham et al., 1997 |
| POLG | Alpers disease ARPEO ADPEO | Polg (−/−) Polg (+/−) | Embryonically lethal. Severe mtDNA depletion. Slight reduction in mtDNA, normal development. | Hance et al., 2005 |
| C10orf2 (PEO1) | ADPEO MDDS (hepatocerebral) | Twinkle ‘Deletor’ mouse | Accumulate multiple mtDNA deletions with progressive COX deficiency and late-onset myopathy. | Tyynismaa et al., 2005 |
| RRM2B | ADPEO MDDS (encephalomyopathic) | Rrm2b (−/−) | Normal at birth, at 6 weeks show growth retardation and die prematurely | Kimura et al., 2003 |
| TK2 | MDDS (myopathic) | Tk2 (−/−) Tk2 (H126N) | Normal at birth, at 7 days show growth retardation, severe hypothermia, severe mtDNA depletion in muscle, heart, liver and spleen. Death at 30 days. Growth retardation, tremor, ataxic gait and severe weakness on day 10. MtDNA depletion. | Akman et al., 2008; Zhou et al., 2008 |
| MPV17 | MDDS (hepatocerebral) | Mpv17 (−/−) | Adult mice show nephrotic syndrome and chronic renal failure. | Weiher et al., 1990 |
| TFAM | None reported to date | Tfam (−/−) | Embryonically lethal. Severe mtDNA depletion and no detectable OXPHOS. | Larsson et al., 1998 |
| ND6 | Leigh syndrome | Nd6 (P25L) | Optic atrophy, reduced complex I and increased oxidative stress. | Lin et al., 2012 |
| NDUFS4 | Leigh syndrome | Ndufs4 (−/−) | Encephalomyopathy, ataxia at 5 weeks, death ∼7 weeks. Slow growth, lethargy, loss of motor skills, blindness and high serum lactate. | Kruse et al., 2008 |
| NDUFS6 | Fatal infantile lactic acidosis | Ndufs6 (−/−) | Cardiomyopathy at 4 months (males) and 8 months (females) and death. | Ke et al., 2012 |
| SDHD | Paraganglioma | Sdhd (−/−) Sdhd (+/−) | Homozygous KO lethal. Heterozygous KO has a decreased Complex II activity. | Piruat et al., 2004 |
| BCS1L | GRACILE syndrome (cholestasis with iron overload, intrauterine growth restriction, amino aciduria, lactic acidosis and early death), complex III deficiency | Bcs1l (S78G) | Failure to thrive, liver steatosis, fibrosis and cirrhosis, tubulopathy, complex III deficiency, premature death. | Leveen et al., 2011 |
| SURF1 | COX-deficient Leigh syndrome | Surf1 (−/−) | High rates of embryonic lethality. Reduced birth weight, reduced complex IV activity in muscle. | Agostino et al., 2003 |
| SCO2 | Cardio-encephalomyopathy | Sco2 (−/−) Sco2 (E140K) | Homozygous KO lethal. Complex IV deficiency, no cardiomyopathy and normal life span. | Yang et al.,; 20102010 |
| COX10 | Encephalomyopathy with renal tubulopathy, Leigh syndrome | Cox10 (−/−) | Slowly progressing myopathy at 3 months, severe complex IV deficiency. | Diaz et al., 2005 |
| PDSS2 | Encephalomyopathy and nephrotic syndrome, CoQ10 deficiency | Kd/kd (spontaneous mutation) | Progressive renal failure. | Lyon and Hulse, 1971 |
| COQ9 | Fatal multisystem disease with CoQ10 deficiency | Coq9 (R239X/R239X) | Fatal encephalomyopathy. | Garcia-Corzo et al., 2013 |
| HSP40 | None reported to date | Hsp40 (−/−) | Dilated cardiomyopathy, RC deficiency and decreased mtDNA levels. Death before 10 weeks. | Hayashi et al., 2006 |
ADPEO, autosomal dominant progressive external ophthalmoplegia; ARPEO, autosomal recessive progressive external ophthalmoplegia.
For nuclear genes, mouse models are now available for RC subunits (e.g. the NDUFS4 and NDUFS6 subunits of complex I, SDHD subunit of complex II and RISP subunit of complex III) and assembly factors (e.g. BCS1L for complex III and COX10, SCO2 and SURF1 for complex IV), and genes involved in maintenance and expression of the mtDNA (Table 2). Mice with defects in mtDNA maintenance genes include KOs of POLG, PEO1 (also known as C10orf2, encoding the Twinkle helicase), TK2 and RRM2B (Table 2). Some of these mouse models display embryonic lethality, others relatively little phenotype, but several have clinical phenotypes similar to the respective human diseases, enabling preclinical trials to be performed. The results of trials performed in these mouse models are discussed in the relevant sections below.
Establishment of national mitochondrial disease consortia
A promising recent advance has been the creation of national consortia aimed at recruiting large cohorts of patients affected by mitochondrial disease. These include the UK MRC Mitochondrial Disease Patient Cohort Study (Nesbitt et al., 2013); the Nationwide Italian Collaborative Network of Mitochondrial Diseases (Mancuso et al., 2013); and the North American Mitochondrial Disease Consortium (DiMauro, 2013). The existence of these consortia should greatly facilitate recruitment into future clinical trials for mitochondrial disease.
Current treatment strategies for mitochondrial diseases
Identification of treatable disorders
One of the most important tasks in the management of mitochondrial disease is the identification of those few disorders that are exquisitely responsive to specific therapies. These include defects of CoQ10 biosynthesis, which may present with infantile-onset encephalomyopathy, nephrotic syndrome, SNHL, ataxia, seizures or isolated myopathy (Rahman et al., 2012). Early initiation of CoQ10 supplementation is related to clinical outcome (Montini et al., 2008), but it should be noted that not all patients respond clinically to CoQ10 supplementation (Rahman et al., 2001; Duncan et al., 2009).
Leigh syndrome is almost invariably a devastating progressive neurodegenerative disorder, but rare treatable causes include biotinidase deficiency (which responds to doses of biotin of 5–10 mg·day−1), and also the biotin thiamine responsive basal ganglia disease (BTBGD), in which much higher doses of biotin are needed, typically at least 5 mg·kg−1·day−1 (Alfadhel et al., 2013). Patients with BTBGD may also present with an acute Wernicke-like encephalopathy, and need thiamine as well as biotin for optimal clinical response, which is not surprising since the gene mutated in this disorder, SLC19A3, encodes a thiamine transporter (Fassone et al., 2013).
Recently, we have also observed (secondary) RC enzyme deficiencies in patients with riboflavin transporter disorders in the Brown Vialetto Van Laere spectrum (Foley et al., 2013). These patients show clinical improvement following high-dose riboflavin (vitamin B2) supplementation. Riboflavin has also been reported to be beneficial for patients with mutations in ACAD9, a gene encoding a flavin-dependent enzyme, which was originally thought to be involved in fat oxidation but which now appears to play a more significant role in complex I assembly (Scholte et al., 1995; Gerards et al., 2011). Although anecdotal reports of benefit from riboflavin have been reported for patients with other causes of complex I deficiency (Ogle et al., 1997), and also some patients with complex II deficiency (Bugiani et al., 2006), so far, no clinical trials have been performed to evaluate this formally. Ongoing studies assessing efficacy of riboflavin therapy in two mouse models of nuclear-encoded complex I deficiency may pave the way for clinical trials (Rahman and Thorburn, 2013).
Supportive therapy
Despite the many and increasingly sophisticated efforts towards finding a suitable cure for mitochondrial disease, currently, most patients are primarily offered symptomatic treatment. Nevertheless, symptom management is important for patients with mitochondrial diseases, improving their quality of life and in some instances prolonging survival (Rahman and Hanna, 2009).
General supportive measures
Examples of symptom management approaches include anticonvulsant drugs for mitochondrial seizure disorders (although sodium valproate should be avoided in patients with POLG mutations because of the risk of liver failure; Rahman, 2012); eyelid surgery for ptosis; hearing aids and cochlear implants for patients with SNHL; pacemakers and implantable defibrillators for cardiac conduction defects; medical treatment of cardiomyopathies; pancreatic enzymes and insulin for pancreatic failure (particularly seen in patients with Kearns–Sayre syndrome (KSS) and in patients with maternally inherited diabetes and deafness); thyroxine, growth hormone and cortisol replacement in patients with hormonal deficiencies; blood transfusions for patients with Pearson syndrome and other sideroblastic anaemias; electrolyte replacement for patients with significant renal tubular losses (as in many children and young people with KSS); and nutritional support (gastrostomy or even parenteral feeding) for patients with prominent gastrointestinal symptoms including dysphagia, vomiting, diarrhoea and failure to gain or maintain weight (Rahman, 2013).
Organ and stem cell transplantation
In situations where the disease targets a single organ such as the heart or liver, organ transplantation can be considered, after careful exclusion of significant neurological involvement (DiMauro and Mancuso, 2007; Rahman, 2013). Allogeneic haematopoietic stem cell transplantation (AHSCT) is currently the only effective way to replace thymidine phosphorylase activity in patients with mitochondrial neurogastrointestinal encephalopathy (MNGIE), and has quickly become accepted as the ‘standard of care’, although it has not yet been evaluated in a clinical trial setting. So far, AHSCT for MNGIE has been associated with ∼50% mortality from disease progression or transplant-related complications (Halter et al., 2011). An internationally agreed transplant protocol aims to reduce toxicity of pre-transplant conditioning (Halter et al., 2011), and a formal clinical trial is planned (Kerr, 2013).
Cerebral folate deficiency
Other supportive therapies are aimed at replacing metabolites that appear to be low in specific subgroups of patients with mitochondrial disease. For example, low CSF levels of the major transport folate 5-methyltetrahydrofolate (5-MTHF) have been documented in several patients with KSS over the last 30 years (Allen et al., 1983). The underlying pathogenic mechanisms are debated, but are likely to involve impairment of active transport of 5-MTHF into the CSF, either because of ATP insufficiency or through ROS-mediated damage to the choroid plexus cells responsible for folate transport (Hyland et al., 2010; Spector and Johanson, 2010; Serrano et al., 2012). Cerebral folate deficiency (CFD) in KSS is associated with cerebral white matter changes, seizures and learning and behavioural difficulties. Treatment with folinic acid led to clinical and radiological improvement in an affected patient (Pineda et al., 2006). Further studies are needed to determine which mitochondrial disease patients are at risk of CFD, understand the mechanisms underpinning CFD, and optimize therapeutic protocols for folinic acid replacement and monitoring in these patients.
Amino acid supplementation
The amino acids L-arginine, citrulline and taurine have been proposed as potential therapeutic agents in the syndrome of MELAS. Strokes in this condition are thought to result from vascular endothelial dysfunction. The observation of low citrulline levels in some affected patients led to the hypothesis that disturbed nitric oxide homeostasis contributes to the pathogenesis of MELAS (Naini et al., 2005) It was therefore suggested that supplementation with arginine, which is required for nitric oxide synthesis, might stabilize vascular function in these patients. A series of open-label studies performed by a Japanese group demonstrated reduced frequency and severity of stroke-like episodes in MELAS patients with the common m.3243A>G mutation (Koga et al., 2005; 2006; 2010). More recently, a group at Baylor College has provided preliminary evidence that citrulline may be even more effective than arginine in MELAS (El-Hattab et al., 2012). Finally, an open-label study of two Japanese subjects showed amelioration of epilepsy and prevention of strokes following high-dose oral taurine administration over a 9 year period (Rikimaru et al., 2012). The authors suggested that providing exogenous taurine reduces the aminoacylation defect associated with the m.3243A>G mutation. Formal double-blinded randomized clinical trials are needed to confirm all of these findings, but are difficult to design and implement because of the episodic and highly unpredictable occurrence of strokes in MELAS.
Therapies still in development
Despite the disappointing results obtained from the clinical trials performed so far in mitochondrial disease, there are many ongoing studies (in vitro and in vivo) that provide hope that some pharmacological approaches may be beneficial to subgroups of patients.
Strategies for increasing mitochondrial biogenesis
An approach that has attracted considerable attention is stimulation of mitochondrial biogenesis, with the intention of increasing mitochondrial function simply by having a greater mitochondrial mass. However controversy exists regarding whether increasing biogenesis of damaged as well as normal mitochondria will ultimately be beneficial or harmful. Mitochondrial biogenesis is under complex regulatory control, requiring coordinated transcription of multiple proteins encoded in two cellular compartments. This allows mitochondrial function to be tailored in vivo according to nutrient and oxygen availability, hormonal signals, differing metabolic demands and rate of cell proliferation. The transcriptional co-activator PPAR-γ co-activator 1-α (PGC-1α) coordinates mitochondrial biogenesis via a cascade of nuclear-encoded hormone receptors, transcription factors and transcriptional co-activators, including PPARs, oestrogen-related receptors, thyroid hormone receptors, nuclear respiratory factors NRF1 and 2 and the transcription factors CREB and YY1 (see Andreux et al., 2013; Dominy and Puigserver, 2013) (Figure 2). In recent years, this signalling cascade has become an attractive therapeutic target to manipulate mitochondrial function, and several methods to increase mitochondrial biogenesis have been explored, including pharmacological strategies, dietary manipulation and exercise therapy.
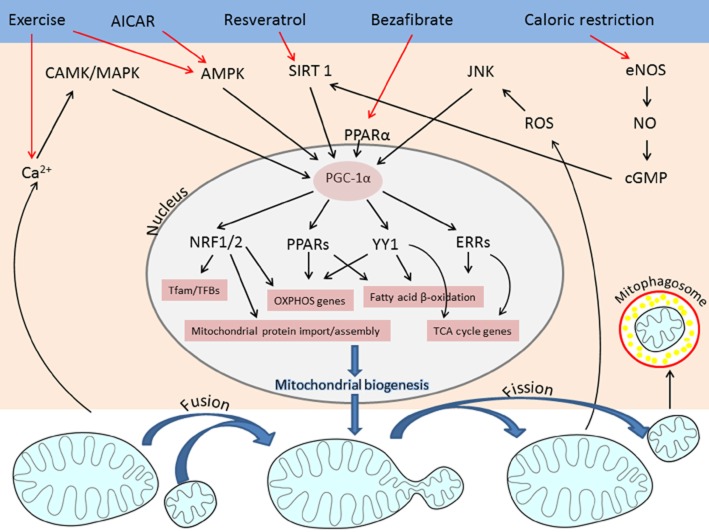
Figure 2.
Schematic representation of pathways regulating mitochondrial biogenesis. External factors (exercise, calorie restriction, stress or small molecules such as bezafibrate, resveratrol or AICAR) up-regulate the expression of PGC-1α, which in turn activates NRF1/2, PPAR, YY1 and ERR transcription factors. These are required for up-regulation of key mitochondrial genes including those that encode OXPHOS subunits, Krebs cycle enzymes, fatty acid β oxidation and proteins involved in mitochondrial protein import and assembly.
Pharmacological approaches
Bezafibrate
PGC-1α overexpression was shown to ameliorate mitochondrial disease in two murine models of COX deficiency: a muscle-specific Cox10 KO (Wenz et al., 2008) and a constitutive Surf1 KO (Viscomi et al., 2011). Following these proof of principle experiments, bezafibrate, a synthetic ligand of PPARα, was used to treat the same mouse models (Wenz et al., 2008; Viscomi et al., 2011) and also a third mouse model, the Deletor mouse containing a dominant mutation in the Twinkle helicase (Yatsuga and Suomalainen, 2012). The results of bezafibrate treatment differed in these three mouse models. COX activity improved in the muscle-specific Cox10 KO mice but not in the Surf1 KO or Deletor mice (Wenz et al., 2008; Viscomi et al., 2011; Yatsuga and Suomalainen, 2012). Similarly, there was evidence of increased mitochondrial biogenesis in the Cox10 KO mice but not in the Deletor mice (Wenz et al., 2008; Yatsuga and Suomalainen, 2012). A problem with the mouse models is that mice develop abnormal lipid metabolism and hepatomegaly in response to bezafibrate (Viscomi et al., 2011; Yatsuga and Suomalainen, 2012). However, this appears to be a rodent-specific effect since bezafibrate has been used safely to treat hyperlipidaemia in hundreds of thousands of people (Prescribing and Primary Care Services and Health and Social Care Information Centre, 2012). Taken together, the results of these studies are promising and suggest that bezafibrate may be beneficial in some mitochondrial disorders. An open-label clinical trial of bezafibrate in the fatty acid oxidation disorder carnitine palmitoyl transferase 2 (CPT2) deficiency was associated with increased physical activity and reduced muscle pain (Bonnefont et al., 2010). In view of these encouraging preliminary results, it seems likely that clinical trials in patients with primary mitochondrial diseases will follow in the near future.
Resveratrol
Other potential targets to increase mitochondrial biogenesis are the sirtuins, which are NAD+-dependent protein deacetylases whose substrates include PGC-1α and the mitochondrial transcription factor TFAM. Resveratrol activates the sirtuin SIRT1 and has been shown to improve mitochondrial fatty acid oxidation in fibroblasts with defects in CPT2 and very long-chain acylCoA dehydrogenase (Bastin et al., 2011). In endothelial cell culture studies resveratrol increased mitochondrial mass and mtDNA content as well the expression of PGC-1α, NRF-1 and TFAM (Csiszar et al., 2009). Recently, a phase I trial of a novel SIRT1 activator, SRT2104, appeared to show improved mitochondrial function in healthy elderly human volunteers (Libri et al., 2012). However, efficacy of this novel molecule has not yet been investigated in mitochondrial disease states. Ongoing trials include an open-label study of resveratrol in Friedreich's ataxia at the Murdoch Children's Research Institute in Melbourne, Australia (Delatycki, 2012). Friedreich's ataxia is an autosomal recessive degenerative disorder caused in most cases by a GAA triplet expansion in the FRDA gene encoding frataxin. Frataxin is involved in regulating mitochondrial iron transport, and FRDA mutations result in secondary deficiencies of mitochondrial iron-sulphur cluster containing enzymes (Rötig et al., 1997).
AICAR
AMP activated PK (AMPK) is a cellular energy sensor (Hardie, 2007) that is another attractive target for modifying mitochondrial bioenergetics in mitochondrial disease. AMPK is activated at high AMP/ATP ratios (i.e. relative energy deficiency states) and acts to phosphorylate several enzymes involved in stimulating catabolism (e.g. by increasing glucose transport and fatty acid oxidation) and inhibiting anabolism (e.g. by reducing glycogen synthesis and lipogenesis). Chronic AMPK activation has also been implicated in transcriptional up-regulation of mitochondrial biogenesis, again via the PGC-1α signalling cascade (Jager et al., 2007) since AMPK increases NAD+ levels that increase SIRT1 activity (Beher et al., 2009; Canto et al., 2009), which up-regulates PGC-1α as discussed above. The AMPK activator 5-aminoimidazole-4-carboxamide ribonucleoside (AICAR) increased mitochondrial biogenesis and ATP levels and decreased ROS in human complex I deficient fibroblasts (Golubitzky et al., 2011) and has also shown promise in mouse models of COX deficiency (Viscomi et al., 2011).
Dietary approaches
The ketogenic diet (KD) has also received a lot of attention in recent years as a possible treatment for mitochondrial diseases. The KD is a high-fat, low-carbohydrate diet that appears to have a number of effects. Fatty acid utilization by mitochondrial β-oxidation is stimulated by the KD, leading to formation of ketone bodies, which provide an alternative energy source for the brain, heart and skeletal muscle. Ketone bodies are metabolized to acetyl-CoA, which feeds into the Krebs cycle and thence to the RC and OXPHOS system to ultimately generate ATP, at least partially bypassing complex I. Increased ketone bodies have also been associated with increased expression of OXPHOS genes, possibly via a similar response to starvation. Starvation causes stress to the cell, which results in activation of many transcription factors and cofactors (including SIRT1, AMPK,PGC-1α) that ultimately increase mitochondrial biogenesis (Nunnari and Suomalainen, 2012). The KD has been investigated in both mitochondrial disease cellular and mouse models. Initial studies reported promising preliminary results: KD reduced mutation load of a heteroplasmic mtDNA deletion in a cybrid model (Santra et al., 2004), increased the expression levels of uncoupling proteins in mice (Sullivan et al., 2004), up-regulated genes involved in mitochondrial biogenesis (Bough et al., 2006) and increased mitochondrial glutathione levels (Jarrett et al., 2008) in rats. A preclinical trial in the Deletor mouse revealed slowing of mitochondrial myopathy progression in mice treated with the KD (Ahola-Erkkila et al., 2010). In humans, there are anecdotal reports of (often transient) benefit of the KD in mitochondrial disease (Malojcic et al., 2004; Laugel et al., 2007), particularly in patients with epilepsy (Kang et al., 2007). The KD has yet to be tested in a randomized, double-blinded clinical trial.
Exercise therapy
Exercise therapy was initially explored as a method for ‘heteroplasmy shifting’, that is, to reduce the relative proportion of mutant to wild-type mtDNA in patients with heteroplasmic mtDNA deletions and point mutations (Taivassalo et al., 1999). However, open-label clinical studies of aerobic exercise in patients with heteroplasmic mtDNA mutations resulted in a mild increase or no change in mutant mtDNA, yet were associated with clinical benefit in terms of increased exercise tolerance and improved quality of life (Taivassalo et al., 2001; 2006). A study of resistance training showed increased muscle strength, again without any significant change in mtDNA mutation load (Murphy et al., 2008). A long-term randomized crossover clinical trial is in progress (Haller, 2009). The mechanisms underlying efficacy of exercise in mitochondrial disease are not completely understood. The prevailing hypothesis is that exercise stimulates mitochondrial biogenesis but evidence to support this is currently lacking in mitochondrial disease patients. In one study, there was a baseline increased expression of PGC-1α in patients with mitochondrial myopathies but no further increase was observed following a period of endurance training (Adhihetty et al., 2007). This study did demonstrate increased expression of the antioxidant enzyme Mn-superoxide dismutase following training, as well as increases in a mitochondrial import protein and in cytochrome c, suggesting that increased antioxidant defence and electron transport may contribute to the adaptive responses following exercise in mitochondrial disease (Adhihetty et al., 2007).
It is important to bear in mind that all of the above strategies to increase mitochondrial biogenesis are interrelated as they all converge on PGC-1α and its downstream signalling cascade. Thus, a combination of pharmacological interventions, diet and exercise may be needed to combat mitochondrial disease. However, evaluating such a complex multifactorial intervention in a clinical trial would clearly be extremely challenging.
Antioxidant approaches
The mitochondrion is the main site of ROS production within the cell. ROS are physiologically important in cell signalling, and in healthy individuals, this is a tightly regulated process. In patients with defects in the RC, however, inefficient transfer of electrons between the four RC complexes results in accumulation of electrons. These can react with O2 to form superoxide anions (O2•−) (Wallace et al., 2010). Depending on the precise genetic defect, a defective RC will often lead to increased ROS production, which will further damage the RC complexes. For example, in cultured human astrocytes, complex IV deficiency led to decreased complex II+III activity (Hargreaves et al., 2007), which would further exacerbate ROS production. It has been suggested that increased ROS levels are one of the most important factors underlying the development of a phenotype in patients with mitochondrial diseases. The importance of this is highlighted by the observation of severe phenotypes in some patients with relatively mild RC defects, which would not be expected to result in a significant decrease in ATP synthesis. Accordingly, inhibition of the RC increased superoxide formation prior to impairment of cellular energy metabolism in an astrocyte model (Jacobson et al., 2005).
As a result of increased ROS levels, GSH levels will also decrease. GSH is the main antioxidant present within mitochondria and functions to metabolize hydrogen peroxide and prevent peroxidation of phospholipids such as cardiolipin (Mari et al., 2009). Decreased mitochondrial GSH levels will result in even higher ROS levels and more damage to the RC, resulting in yet more ROS production forming a vicious cycle. Complex IV activity is directly related to the levels of mitochondrial GSH (Heales et al., 1996). Increased ROS levels and GSH depletion will result in peroxidation of cardiolipin. Cardolipin is an unsaturated phospholipid, which is important for the formation of supercomplexes as well as the retention of cytochrome c within the mitochondrial inner membrane. Supercomplex formation is necessary for the RC enzymes to function effectively (Schagger and Pfeiffer, 2000), while retention of cytochrome c within the inner membrane is crucial in the prevention of cellular apoptosis (Orrenius and Zhivotovsky, 2005). In skeletal muscle samples with complex I and/or complex IV deficiency, cytosolic cytochrome c levels were found to be higher than in controls (Oppenheim et al., 2009). We have demonstrated low GSH in skeletal muscle (Hargreaves et al., 2005) and plasma (Salmi et al., 2012) samples from patients with RC deficiencies.
Understanding the importance of ROS in the context of mitochondrial disease opens many doors to potential treatments. N-acetylcysteine, a drug currently approved and commonly used in treatment of paracetamol overdose could be beneficial. N-acetylcysteine promotes GSH synthesis by increasing the availability of cysteine, which is rate-limiting for GSH biosynthesis (Ferreira et al., 2011). Addition of GSH to skeletal muscle homogenate, even in the presence of oxidizing peroxynitrate, prevented cardiolipin oxidation (Pope et al., 2008). A similar finding was observed with the use of Trolox (vitamin E analogue) (Heales et al., 1994; Pope et al., 2008). Melatonin has also been shown to inhibit cardiolipin oxidization and reduce cytochrome c release (Petrosillo et al., 2009).
Various antioxidants have been used to treat mitochondrial disease. CoQ10 is the most commonly prescribed antioxidant in mitochondrial disease, although a randomized placebo-controlled double-blind crossover clinical trial did not reveal dramatic efficacy of this agent (Glover et al., 2010) as discussed above. A phase III clinical trial in children with RC deficiencies is ongoing (Stacpoole et al., 2012). Several modifications of the CoQ10 molecule have also been investigated. Idebenone has a shorter isoprenyl chain length and is said to have better blood–brain penetrance than CoQ10. Several clinical trials have evaluated idebenone in Friedreich's ataxia and there is a suggestion that it may slow neurological progression in this disorder (see Kerr, 2013). A study in LHON did not show a significant change in the primary outcome measure (best eye recovery) but there was a suggestion of improvement in a subgroup (Klopstock et al., 2011). A phase IIa (dose-finding) randomized placebo-controlled double-blind trial of idebenone in MELAS is ongoing. MitoQ is CoQ10 conjugated to the lipophilic cation triphenylphosphonium, so that it acts as a mitochondria-targeted antioxidant (Kelso et al., 2001). MitoQ has been used in mouse models of disorders associated with secondary mitochondrial dysfunction including Parkinson's and Alzheimer's diseases but has not been used in primary mitochondrial diseases.
EPI-743 is a structurally modified variant of CoQ10 with bis-methyl groups replacing the bis-methoxy groups on the quinone ring, and a chain length of three isoprenyl units rather than ten. This synthetic molecule was found to be the most potent antioxidant of several hundred structurally modified CoQ10 molecules screened in a cell model system, with 1000-fold increased antioxidant properties compared with native CoQ10 (Enns et al., 2012). EPI-743 was subsequently evaluated in a series of open-label trials in an end-of-life setting in several centres in the United States (Enns et al., 2012) and in Rome, and was reported to slow disease progression compared with historical natural history data (Martinelli et al., 2012). However, the extremely unpredictable natural history of Leigh syndrome creates great challenges in interpreting data from such open-label studies. A formal randomized double-blind crossover clinical trial of EPI-743 in children with Leigh syndrome is in progress (Klein, 2012).
Targeting mitochondrial dynamics and mitophagy
The increasing recognition of mitochondria as highly dynamic organelles has led to a new focus for targeting novel therapeutic strategies (Andreux et al., 2013). Mitochondrial mass and morphology results from a delicate balance of several interrelated processes, fission/fusion and biogenesis/mitophagy (Figure 2), and modifying each of these processes may have a role in the treatment of different mitochondrial disorders (reviewed by Andreux et al., 2013; Stetler et al., 2013). Genetically defined disorders of mitochondrial dynamics include defects of mitochondrial fusion caused by mutations in MFN2 or OPA1, presenting as Charcot-Marie-Tooth type 2A and autosomal dominant optic atrophy, respectively (Alexander et al., 2000; Delettre et al., 2000; Zuchner et al., 2004), and impaired mitochondrial fission caused by mutations in DRP1 and MFF (Waterham et al., 2007; Shamseldin et al., 2012). The recent discovery of specific inhibitors of mitochondrial fusion (M-hydrazone) and fission (MDIVI-1 and P110) may provide therapeutic potential for these disorders (Cassidy-Stone et al., 2008; Wang et al., 2012; Andreux et al., 2013; Qi et al., 2013) but preclinical studies are first needed to investigate efficacy and possible adverse effects. These novel agents may also be useful to modify mitophagy in other mitochondrial disorders not primarily caused by fission/fusion defects (Andreux et al., 2013).
Mitochondrial membrane lipids as therapeutic targets
Mitochondrial membrane fluidity and plasticity is intimately related to its lipid content. The first disorder linked to abnormal mitochondrial lipid metabolism was Barth syndrome of dilated cardiomyopathy associated with cyclical neutropenia, growth retardation and 3-methylglutaconic aciduria (Clarke et al., 2013). Barth syndrome is caused by abnormal metabolism of cardiolipin (Schlame and Ren, 2006), the most specific phospholipid component of the mitochondrial membrane. Recently four other disorders have been linked to abnormalities of mitochondrial phospholipid biosynthesis or remodelling (Lamari et al., 2013): rhabdomyolysis associated with mutations in LPIN1 (Zeharia et al., 2008; Michot et al., 2012), Sengers syndrome (hypertrophic cardiomyopathy and cataracts) caused by AGK mutations (Mayr et al., 2012), MEGDEL (3-methylglutaconic aciduria, SNHL, encephalomyopathy and Leigh-like syndrome) associated with SERAC1 mutations (Wortmann et al., 2012), and congenital muscular dystrophy caused by mutations in CHKB, encoding the first step of phosphatidylcholine biosynthesis (Mitsuhashi et al., 2011). Targeting mitochondrial lipid metabolism has thus become an attractive therapeutic strategy, not just for these specific disorders, but also for other mitochondrial diseases since altering the mitochondrial membrane lipid composition by dietary manipulation has been shown to affect ATP synthesis, ROS production and the membrane potential (see Monteiro et al., 2013).
Other pharmacological approaches
Nucleoside replacement
Mitochondrial DNA depletion syndrome (MDDS) may be caused by nuclear-encoded defects of the mtDNA replication machinery or of nucleoside salvage. Nucleoside imbalance is thought to underlie disease pathogenesis in the latter group, and in theory replacing the deficient deoxyribonucleosides might correct these disorders. Accordingly, addition of dAMP and dGMP prevented mtDNA depletion in deoxyguanosine kinase-deficient patient fibroblasts (Taanman et al., 2003) and myotubes (Bulst et al., 2009). A potential concern regarding the use of this form of therapy in vivo is that nucleoside pools are tightly regulated and theoretically replacing one or two nucleosides may result in iatrogenically induced nucleoside imbalance and thereby exacerbate the mtDNA replication defect in affected patients. Recently, it has been proposed that simultaneous administration of deoxyribonucleosides and inhibitors of their catabolism may avoid unwarranted side effects (Camara et al., 2013). Further studies are in progress.
α-Lipoic acid
α-Lipoic acid is a cofactor of three mitochondrial enzymes (PDH, α-ketoglutarate dehydrogenase and branched chain ketoacid decarboxylase) and was first tried as a treatment for PDH deficiency nearly 25 years ago (Byrd et al., 1989). Recently, it has been shown that a subgroup of patients with abnormal mitochondrial energy metabolism have defects in lipoic acid synthesis (Mayr et al., 2011). It is possible that treatment with lipoic acid may benefit this group of patients, although no formal studies have been reported yet. As discussed above, a trial of lipoic acid in combination with creatine and CoQ10 in a heterogeneous group of mitochondrial disease patients showed only mild benefit (Table 2; Rodriguez et al., 2007).
Enzyme replacement therapy
Unlike lysosomal storage disorders, for which recombinant enzyme replacement therapy (ERT) has been approved or is in clinical trial for more than 10 different disorders (Desnick and Schuchman, 2012), no ERTs are currently available for mitochondrial disorders. However, enzyme replacement has been attempted for MNGIE using purified thymidine phosphorylase enzyme encapsulated in the patient's own red blood cells (Moran et al., 2008), so far without long-term clinical success. Very recently, a preclinical toxicity evaluation of this system has been performed in mice and Beagle dogs, and was complicated by infusion-related immune responses in both species (Levene et al., 2013). Finally, one study described the construction of active thymidine phosphorylase encapsulating nanoreactors as novel enzyme delivery vehicles (De Vocht et al., 2010) but clinical utility of this method has not yet been reported in either cell or animal models of MNGIE.
Miscellaneous approaches
Other specific therapies that have been proposed include bypass of complex I using succinate, which is metabolized via complex II (Oguro et al., 2004) or triacylglycerol supplementation (metabolized via fatty acid oxidation, resulting in FADH2 production, which feeds into complex II). Triacylglycerol infusion in four patients with complex I deficiency resulted in increased exercise tolerance (Roef et al., 2002b) but no effect on plasma lactate levels (Roef et al., 2002a). Metronidazole has been used to reduce sulphide production by intestinal anaerobes in ethylmalonic encephalopathy, a disorder of sulphur detoxification (Viscomi et al., 2010). N-acetylcysteine has also been used in the same disorder, in order to replenish GSH as a means of buffering sulphide (Viscomi et al., 2010).
Gene therapy
Although this review is primarily focussed on pharmacological strategies, it is important to mention some recent potentially exciting developments in gene therapy approaches for mitochondrial diseases. Gene therapy for mtDNA defects is particularly challenging since therapeutic DNA molecules need to pass across the two mitochondrial membranes in addition to the plasma membrane. Furthermore, mtDNA gene therapy strategies also need to take into account the high copy number of mtDNA, with thousands of copies per cell.
Allotopic gene expression
Recoding genes that are normally encoded by mtDNA so that they can be inserted into and expressed from the nucleus is known as allotopic gene expression. This technique was used successfully to transfer the recoded mitochondrial MTATP6 gene and thereby rescue the ATP synthesis defect in cybrids containing the m.8993T>G mutation, which is associated with maternally inherited Leigh syndrome and neuropathy, ataxia and retinitis pigmentosa (NARP) (Manfredi et al., 2002). Furthermore, allotopic expression of the MTND4 gene prevented blindness in a rat model of LHON (Manfredi et al., 2002; Ellouze et al., 2008). A human clinical trial of allotopic gene therapy in LHON is in progress but has not yet been reported (Lam et al., 2010).
Transkingdom gene therapy
Several studies have used transkingdom gene therapy, where a gene from one species is used to correct a disorder in another species, to target mitochondrial diseases. Transkingdom gene therapy was first suggested as a possible treatment for complex I deficiencies in 2006 (Yagi et al., 2006) and subsequently adeno-associated virus (AAV) tagging of the yeast alternative NADH dehydrogenase NDI1 was used to treat an animal model of LHON (Marella et al., 2010). Similarly, the yeast alternative oxidase bypasses complexes III and IV and was shown to rescue COX deficiency in human cultured cells (Dassa et al., 2009) and a mouse model (El-Khoury et al., 2013). Transgenic expression of D. melanogaster deoxyribonucleoside kinase (Dm-dNK) was able to rescue myopathic MDDS in mice with thymidine kinase deficiency (Krishnan et al., 2013).
Other gene therapy approaches for mtDNA defects
DNA delivery into the mitochondrion has been attempted using liposome-based nanocarriers such as Mito-Porter (Yasuzaki et al., 2010) or a DQAsome transfection system (Lyrawati et al., 2011), delivery of cytosolic tRNAs into mitochondria as a method to aid mitochondrial translation (Mahata et al., 2006) and using restriction enzymes to specifically degrade mutant mtDNA (Tanaka et al., 2002). Heteroplasmy shifting has also been achieved using antisense oligonucleotides in cybrids containing a heteroplasmic mtDNA deletion (Comte et al., 2013). Very recently, a new approach used transcription activator-like effector nucleases engineered to localize to mitochondria, to eliminate mutant mtDNA from cybrids containing the m.14459G>A mutation associated with LHON plus dystonia (Bacman et al., 2013). The efficacy of these approaches is strongly debated, and preclinical studies will be required to ensure the safety of these novel agents in whole organisms.
Gene therapy for nuclear defects
Gene therapy for nuclear-encoded mitochondrial disorders is technically less challenging and AAV and lentiviral-mediated gene therapy has been performed in murine models of ethylmalonic encephalopathy and MNGIE, respectively, with encouraging results (Torres-Torronteras et al., 2011; Di Meo et al., 2012). Similar approaches would be applicable for other nuclear-encoded mitochondrial diseases.
Pronuclear and spindle cell transfer
Germ line gene therapy has been proposed for mtDNA mutations using the techniques of pronuclear transfer and maternal spindle cell transfer, as a potential method for preventing transmission of mutated mtDNA from the mother to the embryo. In the case of pronuclear transfer, donor and recipient zygotes are enucleated and the recipient's nucleus is then fused into the enucleated donor zygote. This has been demonstrated to be technically possible in research using abnormally fertilized human embryos (Craven et al., 2010). Maternal spindle cell transfer (MSCT) involves transfer of nuclear material between donor and recipient unfertilized metaphase II oocytes, and several Rhesus monkeys have been born from oocytes manipulated in this way (Tachibana et al., 2009). More recently, MSCT has been shown to be an effective method to replace mtDNA in human oocytes (Tachibana et al., 2013). There has been considerable discussion regarding the ethics surrounding these procedures (Bredenoord and Braude, 2010; Nuffield Council on Bioethics UK, 2012), but recently, after an extensive public consultation process (Pitts-Tucker, 2012), the UK government has granted permission for further human research to be conducted using these techniques (House of Commons Hansard Debates 25 June, 2013).
Although all of these studies give promise for future therapies, they are still very far from being offered to patients.
Conclusions
The complexity of the mitochondrial organelle and the disorders associated with its dysfunction creates unusual challenges for developing effective treatments. There are particular difficulties in clinical trial design for this group of heterogeneous disorders with unpredictable clinical courses. The few well-designed clinical trials that have been conducted to date have failed to identify any clearly effective treatments for mitochondrial disease. However, recent developments including the establishment of national and international consortia aimed at collecting large well-characterized cohorts of patients affected by mitochondrial disease, the availability of mouse models of numerous mitochondrial disorders, the identification of novel drug targets including components of the complex signalling cascades controlling mitochondrial biogenesis, and the realization of novel gene therapy approaches, all herald promise for developing and testing new mitochondrial therapeutic agents. Given the extreme clinical, biochemical and genetic heterogeneity of mitochondrial disease, it is extremely unlikely that a ‘one size fits all’ universal panacea exists. Rather, it is more likely that different interventions will be effective for different subgroups of mitochondrial diseases. Ultimately, it is hoped that it will be possible to deliver a ‘personalized medicine’ approach to patients affected by mitochondrial disease, with the goal of long-term survival with good quality of life.
Acknowledgments
M. K. is supported by a PhD studentship from Vitaflo International. S. R. is supported by Great Ormond Street Hospital Children's Charity and research in her group is funded by the Muscular Dystrophy Campaign, Sparks and The Wellcome Trust.
Glossary
- AAV
adeno-associated virus
- AICAR
5-aminoimidazole-4-carboxamide ribonucleotide
- CFD
cerebral folate deficiency
- COX
cytochrome c oxidase
- CPT2
carnitine palmitoyl transferase 2
- DCA
dichloroacetate
- ERT
enzyme replacement therapy
- FGF21
fibroblast growth factor-21
- KD
ketogenic diet
- KO
knockout
- KSS
Kearns–Sayre syndrome
- LHON
Leber's hereditary optic neuropathy
- MDDS
mitochondrial DNA depletion syndrome
- MELAS
mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes
- MNGIE
mitochondrial neurogastrointestinal encephalopathy
- MRS
magnetic resonance spectroscopy
- MSCT
maternal spindle cell transfer
- mtDNA
mitochondrial DNA
- NARP
neuropathy, ataxia and retinitis pigmentosa
- OXPHOS
oxidative phosphorylation
- PDH
pyruvate dehydrogenase
- PDK
pyruvate dehydrogenase kinase
- PGC-1α
PPAR γ co-activator 1-α
- RC
respiratory chain
- ROS
reactive oxygen species
Conflict of interest
The authors declare that M. K.'s PhD studentship is funded by Vitaflo International.
References
- Adhihetty PJ, Taivassalo T, Haller RG, Walkinshaw DR, Hood DA. The effect of training on the expression of mitochondrial biogenesis-and apoptosis-related proteins in skeletal muscle of patients with mtDNA defects. Am J Physiol Endocrinol Metab. 2007;293:E672–E680. doi: 10.1152/ajpendo.00043.2007. [DOI] [PubMed] [Google Scholar]
- Agostino A, Invernizzi F, Tiveron C, Fagiolari G, Prelle A, Lamantea E, et al. Constitutive knockout of Surf1 is associated with high embryonic lethality, mitochondrial disease and cytochrome c oxidase deficiency in mice. Hum Mol Genet. 2003;12:399–413. doi: 10.1093/hmg/ddg038. [DOI] [PubMed] [Google Scholar]
- Ahola-Erkkila S, Carroll CJ, Peltola-Mjosund K, Tulkki V, Mattila I, Seppanen-Laakso T, et al. Ketogenic diet slows down mitochondrial myopathy progression in mice. Hum Mol Genet. 2010;19:1974–1984. doi: 10.1093/hmg/ddq076. [DOI] [PubMed] [Google Scholar]
- Akman HO, Dorado B, Lopez LC, Garcia-Cazorla A, Vila MR, Tanabe LM, et al. Thymidine kinase 2 (H126N) knockin mice show the essential role of balanced deoxynucleotide pools for mitochondrial DNA maintenance. Hum Mol Genet. 2008;17:2433–2440. doi: 10.1093/hmg/ddn143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander C, Votruba M, Pesch UE, Thiselton DL, Mayer S, Moore A, et al. OPA1, encoding a dynamin-related GTPase, is mutated in autosomal dominant optic atrophy linked to chromosome 3q28. Nat Genet. 2000;26:211–215. doi: 10.1038/79944. [DOI] [PubMed] [Google Scholar]
- Alfadhel M, Almuntashri M, Jadah RH, Bashiri FA, Al Rifai MT, Al Shalaan H, et al. Biotin-responsive basal ganglia disease should be renamed biotin-thiamine-responsive basal ganglia disease: a retrospective review of the clinical, radiological and molecular findings of 18 new cases. Orphanet J Rare Dis. 2013;8:83. doi: 10.1186/1750-1172-8-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen RJ, DiMauro S, Coulter DL, Papadimitriou A, Rothenberg SP. Kearns–Sayre syndrome with reduced plasma and cerebrospinal fluid folate. Ann Neurol. 1983;13:679–682. doi: 10.1002/ana.410130620. [DOI] [PubMed] [Google Scholar]
- Andreux PA, Houtkooper RH, Auwerx J. Pharmacological approaches to restore mitochondrial function. Nat Rev Drug Discov. 2013;12:465–483. doi: 10.1038/nrd4023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bacman SR, Williams SL, Pinto M, Peralta S, Moraes CT. Specific elimination of mutant mitochondrial genomes in patient-derived cells by mitoTALENs. Nat Med. 2013;19:1111–1113. doi: 10.1038/nm.3261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bastin J, Lopes-Costa A, Djouadi F. Exposure to resveratrol triggers pharmacological correction of fatty acid utilization in human fatty acid oxidation-deficient fibroblasts. Hum Mol Genet. 2011;20:2048–2057. doi: 10.1093/hmg/ddr089. [DOI] [PubMed] [Google Scholar]
- Beher D, Wu J, Cumine S, Kim KW, Lu SC, Atangan L, et al. Resveratrol is not a direct activator of SIRT1 enzyme activity. Chem Biol Drug Des. 2009;74:619–624. doi: 10.1111/j.1747-0285.2009.00901.x. [DOI] [PubMed] [Google Scholar]
- Bonnefont JP, Bastin J, Laforet P, Aubey F, Mogenet A, Romano S, et al. Long-term follow-up of bezafibrate treatment in patients with the myopathic form of carnitine palmitoyltransferase 2 deficiency. Clin Pharmacol Ther. 2010;88:101–108. doi: 10.1038/clpt.2010.55. [DOI] [PubMed] [Google Scholar]
- Bough KJ, Wetherington J, Hassel B, Pare JF, Gawryluk JW, Greene JG, et al. Mitochondrial biogenesis in the anticonvulsant mechanism of the ketogenic diet. Ann Neurol. 2006;60:223–235. doi: 10.1002/ana.20899. [DOI] [PubMed] [Google Scholar]
- Bratic A, Larsson NG. The role of mitochondria in aging. J Clin Invest. 2013;123:951–957. doi: 10.1172/JCI64125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bredenoord AL, Braude P. Ethics of mitochondrial gene replacement: from bench to bedside. BMJ. 2010;341:c6021. doi: 10.1136/bmj.c6021. [DOI] [PubMed] [Google Scholar]
- Bugiani M, Lamantea E, Invernizzi F, Moroni I, Bizzi A, Zeviani M, et al. Effects of riboflavin in children with complex II deficiency. Brain Dev. 2006;28:576–581. doi: 10.1016/j.braindev.2006.04.001. [DOI] [PubMed] [Google Scholar]
- Bulst S, Abicht A, Holinski-Feder E, Muller-Ziermann S, Koehler U, Thirion C, et al. In vitro supplementation with dAMP/dGMP leads to partial restoration of mtDNA levels in mitochondrial depletion syndromes. Hum Mol Genet. 2009;18:1590–1599. doi: 10.1093/hmg/ddp074. [DOI] [PubMed] [Google Scholar]
- Bulst S, Holinski-Feder E, Payne B, Abicht A, Krause S, Lochmuller H, et al. In vitro supplementation with deoxynucleoside monophosphates rescues mitochondrial DNA depletion. Mol Genet Metab. 2012;107:95–103. doi: 10.1016/j.ymgme.2012.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrd DJ, Krohn HP, Winkler L, Steinborn C, Hadam M, Brodehl J, et al. Neonatal pyruvate dehydrogenase deficiency with lipoate responsive lactic acidaemia and hyperammonaemia. Eur J Pediatr. 1989;148:543–547. doi: 10.1007/BF00441554. [DOI] [PubMed] [Google Scholar]
- Calvo S, Jain M, Xie X, Sheth SA, Chang B, Goldberger OA, et al. Systematic identification of human mitochondrial disease genes through integrative genomics. Nat Genet. 2006;38:576–582. doi: 10.1038/ng1776. [DOI] [PubMed] [Google Scholar]
- Camara Y, Gonzalez-Vioque E, Scarpelli M, Torres-Torronteras J, Marti R. Feeding the deoxyribonucleoside salvage pathway to rescue mitochondrial DNA. Drug Discov Today. 2013;18:950–957. doi: 10.1016/j.drudis.2013.06.009. [DOI] [PubMed] [Google Scholar]
- Canto C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009;458:1056–1060. doi: 10.1038/nature07813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassidy-Stone A, Chipuk JE, Ingerman E, Song C, Yoo C, Kuwana T, et al. Chemical inhibition of the mitochondrial division dynamin reveals its role in Bax/Bak-dependent mitochondrial outer membrane permeabilization. Dev Cell. 2008;14:193–204. doi: 10.1016/j.devcel.2007.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherry AB, Gagne KE, McLoughlin EM, Baccei A, Gorman B, Hartung O, et al. Induced pluripotent stem cells with a mitochondrial DNA deletion. Stem Cells. 2013;31:1287–1297. doi: 10.1002/stem.1354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke SL, Bowron A, Gonzalez IL, Groves SJ, Newbury-Ecob R, Clayton N, et al. Barth syndrome. Orphanet J Rare Dis. 2013;8:23. doi: 10.1186/1750-1172-8-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Comte C, Tonin Y, Heckel-Mager AM, Boucheham A, Smirnov A, Aure K, et al. Mitochondrial targeting of recombinant RNAs modulates the level of a heteroplasmic mutation in human mitochondrial DNA associated with Kearns Sayre syndrome. Nucleic Acids Res. 2013;41:418–433. doi: 10.1093/nar/gks965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van de Corput MP, van den Ouweland JM, Dirks RW, Hart LM, Bruining GJ, Maassen JA, et al. Detection of mitochondrial DNA deletions in human skin fibroblasts of patients with Pearson's syndrome by two-color fluorescence in situ hybridization. J Histochem Cytochem. 1997;45:55–61. doi: 10.1177/002215549704500108. [DOI] [PubMed] [Google Scholar]
- Coskun P, Wyrembak J, Schriner SE, Chen HW, Marciniack C, Laferla F, et al. A mitochondrial etiology of Alzheimer and Parkinson disease. Biochim Biophys Acta. 2012;1820:553–564. doi: 10.1016/j.bbagen.2011.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craven L, Tuppen HA, Greggains GD, Harbottle SJ, Murphy JL, Cree LM, et al. Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease. Nature. 2010;465:82–85. doi: 10.1038/nature08958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Csiszar A, Labinskyy N, Pinto JT, Ballabh P, Zhang H, Losonczy G, et al. Resveratrol induces mitochondrial biogenesis in endothelial cells. Am J Physiol Heart Circ Physiol. 2009;297:H13–H20. doi: 10.1152/ajpheart.00368.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dassa EP, Dufour E, Gonçalves S, Paupe V, Hakkaart GA, Jacobs HT, et al. Expression of the alternative oxidase complements cytochrome c oxidase deficiency in human cells. EMBO Mol Med. 2009;1:30–36. doi: 10.1002/emmm.200900001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Stefano N, Matthews PM, Ford B, Genge A, Karpati G, Arnold DL. Short-term dichloroacetate treatment improves indices of cerebral metabolism in patients with mitochondrial disorders. Neurology. 1995;45:1193–1198. doi: 10.1212/wnl.45.6.1193. [DOI] [PubMed] [Google Scholar]
- De Vocht C, Ranquin A, Van GJ, Vanhaecke T, Rogiers V, Van GP, et al. Polymeric nanoreactors for enzyme replacement therapy of MNGIE. J Control Release. 2010;148:e19–e20. doi: 10.1016/j.jconrel.2010.07.011. [DOI] [PubMed] [Google Scholar]
- Debattisti V, Scorrano L. D. melanogaster, mitochondria and neurodegeneration: small model organism, big discoveries. Mol Cell Neurosci. 2013;55:77–86. doi: 10.1016/j.mcn.2012.08.007. [DOI] [PubMed] [Google Scholar]
- Delatycki MB. 2012. An open label clinical pilot study of resveratrol as treatment for friedreich ataxia. NCT01339884. Available at: http://www.clinicaltrials.gov/ct2/show/NCT01339884?term=NCT01339884&rank=1 (accessed 20 August 2013)
- Delettre C, Lenaers G, Griffoin JM, Gigarel N, Lorenzo C, Belenguer P, et al. Nuclear gene OPA1, encoding a mitochondrial dynamin-related protein, is mutated in dominant optic atrophy. Nat Genet. 2000;26:207–210. doi: 10.1038/79936. [DOI] [PubMed] [Google Scholar]
- Desnick RJ, Schuchman EH. Enzyme replacement therapy for lysosomal diseases: lessons from 20 years of experience and remaining challenges. Annu Rev Genomics Hum Genet. 2012;13:307–335. doi: 10.1146/annurev-genom-090711-163739. [DOI] [PubMed] [Google Scholar]
- Di Meo I, Auricchio A, Lamperti C, Burlina A, Viscomi C, Zeviani M. Effective AAV-mediated gene therapy in a mouse model of ethylmalonic encephalopathy. EMBO Mol Med. 2012;4:1008–1014. doi: 10.1002/emmm.201201433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz F, Thomas CK, Garcia S, Hernandez D, Moraes CT. Mice lacking COX10 in skeletal muscle recapitulate the phenotype of progressive mitochondrial myopathies associated with cytochrome c oxidase deficiency. Hum Mol Genet. 2005;14:2737–2748. doi: 10.1093/hmg/ddi307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiMauro S. Mitochondrial encephalomyopathies: back to Mendelian genetics. Ann Neurol. 1999;45:693–694. doi: 10.1002/1531-8249(199906)45:6<693::aid-ana2>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- DiMauro S. Mitochondrial encephalomyopathies – fifty years on: the Robert Wartenberg Lecture. Neurology. 2013;81:281–291. doi: 10.1212/WNL.0b013e31829bfe89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiMauro S, Mancuso M. Mitochondrial diseases: therapeutic approaches. Biosci Rep. 2007;27:125–137. doi: 10.1007/s10540-007-9041-4. [DOI] [PubMed] [Google Scholar]
- Dominy JE, Puigserver P. Mitochondrial biogenesis through activation of nuclear signaling proteins. Cold Spring Harb Perspect Biol. 2013;5:1–16. doi: 10.1101/cshperspect.a015008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan AJ, Bitner-Glindzicz M, Meunier B, Costello H, Hargreaves IP, Lopez LC, et al. A nonsense mutation in COQ9 causes autosomal-recessive neonatal-onset primary coenzyme Q10 deficiency: a potentially treatable form of mitochondrial disease. Am J Hum Genet. 2009;84:558–566. doi: 10.1016/j.ajhg.2009.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan GE, Perkins LA, Theriaque DW, Neiberger RE, Stacpoole PW. Dichloroacetate therapy attenuates the blood lactate response to submaximal exercise in patients with defects in mitochondrial energy metabolism. J Clin Endocrinol Metab. 2004;89:1733–1738. doi: 10.1210/jc.2003-031684. [DOI] [PubMed] [Google Scholar]
- El-Hattab AW, Hsu JW, Emrick LT, Wong LJ, Craigen WJ, Jahoor F, et al. Restoration of impaired nitric oxide production in MELAS syndrome with citrulline and arginine supplementation. Mol Genet Metab. 2012;105:607–614. doi: 10.1016/j.ymgme.2012.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Khoury R, Dufour E, Rak M, Ramanantsoa N, Grandchamp N, Csaba Z, et al. Alternative oxidase expression in the mouse enables bypassing cytochrolme c oxidase blockade and limits mitochondrial ROS overproduction. PLoS Genet. 2013;9:e1003182. doi: 10.1371/journal.pgen.1003182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellouze S, Augustin S, Bouaita A, Bonnet C, Simonutti M, Forster V, et al. Optimized allotopic expression of the human mitochondrial ND4 prevents blindness in a rat model of mitochondrial dysfunction. Am J Hum Genet. 2008;83:373–387. doi: 10.1016/j.ajhg.2008.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enns GM, Kinsman SL, Perlman SL, Spicer KM, Abdenur JE, Cohen BH, et al. Initial experience in the treatment of inherited mitochondrial disease with EPI-743. Mol Genet Metab. 2012;105:91–102. doi: 10.1016/j.ymgme.2011.10.009. [DOI] [PubMed] [Google Scholar]
- Fassone E, Wedatilake Y, DeVile C, Chong WK, Carr LJ, Rahman S. Treatable Leigh-like encephalopathy presenting in adolescence. BMJ Case Rep. 2013 doi: 10.1136/bcr-2013-200838. (in press). doi. 10.1136/bcr-2013-200838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Ayala DJ, Chen S, Kemppainen E, O'Dell KM, Jacobs HT. Gene expression in a Drosophila model of mitochondrial disease. PLoS ONE. 2010;5:e8549. doi: 10.1371/journal.pone.0008549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira LF, Campbell KS, Reid MB. N-acetylcysteine in handgrip exercise: plasma thiols and adverse reactions. Int J Sport Nutr Exerc Metab. 2011;21:146–154. doi: 10.1123/ijsnem.21.2.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foley AR, Menezes MP, Pandraud A, Gonzalez MA, Al-Odaib A, Abrams AJ, et al. Treatable childhood neuronopathy caused by mutations in riboflavin transporter RFVT2. Brain. 2013 doi: 10.1093/brain/awt315. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Corzo L, Luna-Sanchez M, Doerrier C, Garcia JA, Guaras A, Acin-Perez R, et al. Dysfunctional Coq9 protein causes predominant encephalomyopathy associated with CoQ deficiency. Hum Mol Genet. 2013;22:1233–1248. doi: 10.1093/hmg/dds530. [DOI] [PubMed] [Google Scholar]
- Gerards M, van den Bosch BJ, Danhauser K, Serre V, van Weeghel M, Wanders RJ, et al. Riboflavin-responsive oxidative phosphorylation complex I deficiency caused by defective ACAD9: new function for an old gene. Brain. 2011;134:210–219. doi: 10.1093/brain/awq273. [DOI] [PubMed] [Google Scholar]
- Glover EI, Martin J, Maher A, Thornhill RE, Moran GR, Tarnopolsky MA. A randomized trial of coenzyme Q10 in mitochondrial disorders. Muscle Nerve. 2010;42:739–748. doi: 10.1002/mus.21758. [DOI] [PubMed] [Google Scholar]
- Golubitzky A, Dan P, Weissman S, Link G, Wikstrom JD, Saada A. Screening for active small molecules in mitochondrial complex I deficient patient's fibroblasts, reveals AICAR as the most beneficial compound. PLoS ONE. 2011;6:e26883. doi: 10.1371/journal.pone.0026883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grad LI, Lemire BD. Riboflavin enhances the assembly of mitochondrial cytochrome c oxidase in C. elegans NADH-ubiquinone oxidoreductase mutants. Biochim Biophys Acta. 2006;1757:115–122. doi: 10.1016/j.bbabio.2005.11.009. [DOI] [PubMed] [Google Scholar]
- Graham BH, Waymire KG, Cottrell B, Trounce IA, Macgregor GR, Wallace DC. A mouse model for mitochondrial myopathy and cardiomyopathy resulting from a deficiency in the heart/muscle isoform of the adenine nucleotide translocator. Nat Genet. 1997;16:226–234. doi: 10.1038/ng0797-226. [DOI] [PubMed] [Google Scholar]
- Haller RG. 2009. Exercise training and deconditions: implications for therapy in mitochondrial myopathy. NCT00457314. Available at: http://www.clinicaltrials.gov/ct2/show/NCT00457314?term=NCT00457314&rank=1 (accessed 20 August 2013)
- Halter J, Schupbach WM, Casali C, Elhasid R, Fay K, Hammans S, et al. Allogeneic hematopoietic SCT as treatment option for patients with mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): a consensus conference proposal for a standardized approach. Bone Marrow Transplant. 2011;46:330–337. doi: 10.1038/bmt.2010.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hance N, Ekstrand MI, Trifunovic A. Mitochondrial DNA polymerase gamma is essential for mammalian embryogenesis. Hum Mol Genet. 2005;14:1775–1783. doi: 10.1093/hmg/ddi184. [DOI] [PubMed] [Google Scholar]
- Hardie DG. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nat Rev Mol Cell Biol. 2007;8:774–785. doi: 10.1038/nrm2249. [DOI] [PubMed] [Google Scholar]
- Hargreaves IP, Sheena Y, Land JM, Heales SJ. Glutathione deficiency in patients with mitochondrial disease: implications for pathogenesis and treatment. J Inherit Metab Dis. 2005;28:81–88. doi: 10.1007/s10545-005-4160-1. [DOI] [PubMed] [Google Scholar]
- Hargreaves IP, Duncan AJ, Wu L, Agrawal A, Land JM, Heales SJ. Inhibition of mitochondrial complex IV leads to secondary loss complex II-III activity: implications for the pathogenesis and treatment of mitochondrial encephalomyopathies. Mitochondrion. 2007;7:284–287. doi: 10.1016/j.mito.2007.02.001. [DOI] [PubMed] [Google Scholar]
- Hayashi M, Imanaka-Yoshida K, Yoshida T, Wood M, Fearns C, Tatake RJ, et al. A crucial role of mitochondrial Hsp40 in preventing dilated cardiomyopathy. Nat Med. 2006;12:128–132. doi: 10.1038/nm1327. [DOI] [PubMed] [Google Scholar]
- Heales SJ, Bolanos JP, Land JM, Clark JB. Trolox protects mitochondrial complex IV from nitric oxide-mediated damage in astrocytes. Brain Res. 1994;668:243–245. doi: 10.1016/0006-8993(94)90530-4. [DOI] [PubMed] [Google Scholar]
- Heales SJ, Bolanos JP, Brand MP, Clark JB, Land JM. Mitochondrial damage: an important feature in a number of inborn errors of metabolism? J Inherit Metab Dis. 1996;19:140–142. doi: 10.1007/BF01799414. [DOI] [PubMed] [Google Scholar]
- Heller A, Brockhoff G, Goepferich A. Targeting drugs to mitochondria. Eur J Pharm Biopharm. 2012;82:1–18. doi: 10.1016/j.ejpb.2012.05.014. [DOI] [PubMed] [Google Scholar]
- House of Commons Hansard Debates 25 June 2013. 2013. Mitochondrial disease. UK Parliment 565 c60WH. Available at: http://www.publications.parliament.uk/pa/cm201314/cmhansrd/cm130625/halltext/130625h0002.htm#13062568000002 (accessed 20 August 2013)
- Hyland K, Shoffner J, Heales SJ. Cerebral folate deficiency. J Inherit Metab Dis. 2010;33:563–570. doi: 10.1007/s10545-010-9159-6. [DOI] [PubMed] [Google Scholar]
- Inoue K, Nakada K, Ogura A, Isobe K, Goto Y, Nonaka I, et al. Generation of mice with mitochondrial dysfunction by introducing mouse mtDNA carrying a deletion into zygotes. Nat Genet. 2000;26:176–181. doi: 10.1038/82826. [DOI] [PubMed] [Google Scholar]
- Jacobson J, Duchen MR, Hothersall J, Clark JB, Heales SJ. Induction of mitochondrial oxidative stress in astrocytes by nitric oxide precedes disruption of energy metabolism. J Neurochem. 2005;95:388–395. doi: 10.1111/j.1471-4159.2005.03374.x. [DOI] [PubMed] [Google Scholar]
- Jager S, Handschin CS, Pierre J, Spiegelman BM. AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1alpha. Proc Natl Acad Sci U S A. 2007;104:12017–12022. doi: 10.1073/pnas.0705070104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarrett SG, Milder JB, Liang LP, Patel M. The ketogenic diet increases mitochondrial glutathione levels. J Neurochem. 2008;106:1044–1051. doi: 10.1111/j.1471-4159.2008.05460.x. [DOI] [PubMed] [Google Scholar]
- Kang HC, Lee YM, Kim HD, Lee JS, Slama A. Safe and effective use of the ketogenic diet in children with epilepsy and mitochondrial respiratory chain complex defects. Epilepsia. 2007;48:82–88. doi: 10.1111/j.1528-1167.2006.00906.x. [DOI] [PubMed] [Google Scholar]
- Kaufmann P, Engelstad K, Wei Y, Jhung S, Sano MC, Shungu DC, et al. Dichloroacetate causes toxic neuropathy in MELAS: a randomized, controlled clinical trial. Neurology. 2006;66:324–330. doi: 10.1212/01.wnl.0000196641.05913.27. [DOI] [PubMed] [Google Scholar]
- Ke BX, Pepe S, Grubb DR, Komen JC, Laskowski A, Rodda FA, et al. Tissue-specific splicing of an Ndufs6 gene-trap insertion generates a mitochondrial complex I deficiency-specific cardiomyopathy. Proc Natl Acad Sci U S A. 2012;109:6165–6170. doi: 10.1073/pnas.1113987109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelso GF, Porteous CM, Coulter CV, Hughes G, Porteous WK, Ledgerwood EC, et al. Selective targeting of a redox-active ubiquinone to mitochondria within cells: antioxidant and antiapoptotic properties. J Biol Chem. 2001;276:4588–4596. doi: 10.1074/jbc.M009093200. J Biol Chem 282: 14708–14718. [DOI] [PubMed] [Google Scholar]
- Kerr DS. Review of clinical trials for mitochondrial disorders: 1997–2012. Neurother. 2013;10:307–319. doi: 10.1007/s13311-013-0176-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura T, Takeda S, Sagiya Y, Gotoh M, Nakamura Y, Arakawa H. Impaired function of p53R2 in Rrm2b-null mice causes severe renal failure through attenuation of dNTP pools. Nat Genet. 2003;34:440–445. doi: 10.1038/ng1212. [DOI] [PubMed] [Google Scholar]
- King MP, Attardi G. Human cells lacking mtDNA: repopulation with exogenous rhorhomitochondria by complementation. Science. 1989;246:500–503. doi: 10.1126/science.2814477. [DOI] [PubMed] [Google Scholar]
- Klein M. 2012. A phase 2B randomized, placebo controlled, double blind clinical trial of EPI-743 in children with leigh syndrome. NCT01721733. Available at: http://www.clinicaltrials.gov/ct2/show/NCT01721733?term=NCT01721733&rank=1 (accessed 20 August 2013)
- Klopstock T, Querner V, Schmidt F, Gekeler F, Walter M, Hartard M, et al. A placebo-controlled crossover trial of creatine in mitochondrial diseases. Neurology. 2000;55:1748–1751. doi: 10.1212/wnl.55.11.1748. [DOI] [PubMed] [Google Scholar]
- Klopstock T, Yu-Wai-Man P, Dimitriadis K, Rouleau J, Heck S, Bailie M, et al. A randomized placebo-controlled trial of idebenone in Leber's hereditary optic neuropathy. Brain. 2011;134:2677–2686. doi: 10.1093/brain/awr170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koga Y, Akita Y, Nishioka J, Yatsuga S, Povalko N, Tanabe Y, et al. L-arginine improves the symptoms of strokelike episodes in MELAS. Neurology. 2005;64:710–712. doi: 10.1212/01.WNL.0000151976.60624.01. [DOI] [PubMed] [Google Scholar]
- Koga Y, Akita Y, Junko N, Yatsuga S, Povalko N, Fukiyama R, et al. Endothelial dysfunction in MELAS improved by l-arginine supplementation. Neurology. 2006;66:1766–1769. doi: 10.1212/01.wnl.0000220197.36849.1e. [DOI] [PubMed] [Google Scholar]
- Koga Y, Povalko N, Nishioka J, Katayama K, Kakimoto N, Matsuishi T. MELAS and L-arginine therapy: pathophysiology of stroke-like episodes. Ann N Y Acad Sci. 2010;1201:104–110. doi: 10.1111/j.1749-6632.2010.05624.x. [DOI] [PubMed] [Google Scholar]
- Kornblum C, Schroder R, Muller K, Vorgerd M, Eggers J, Bogdanow M, et al. Creatine has no beneficial effect on skeletal muscle energy metabolism in patients with single mitochondrial DNA deletions: a placebo-controlled, double-blind 31P-MRS crossover study. Eur J Neurol. 2005;12:300–309. doi: 10.1111/j.1468-1331.2004.00970.x. [DOI] [PubMed] [Google Scholar]
- Krishnan S, Zhou X, Paredes JA, Kuiper RV, Curbo S, Karlsson A. Transgene expression of Drosophila melanogaster nucleoside kinase reverses mitochondrial thymidine kinase 2 deficiency. J Biol Chem. 2013;288:5072–5079. doi: 10.1074/jbc.M112.437152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruse SE, Watt WC, Marcinek DJ, Kapur RP, Schenkman KA, Palmiter RD. Mice with mitochondrial complex I deficiency develop a fatal encephalomyopathy. Cell Metab. 2008;7:312–320. doi: 10.1016/j.cmet.2008.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam BL, Feuer WJ, Abukhalil F, Porciatti V, Hauswirth WW, Guy J. Leber hereditary optic neuropathy gene therapy clinical trial recruitment: year 1. Arch Ophthalmol. 2010;128:1129–1135. doi: 10.1001/archophthalmol.2010.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamari F, Mochel F, Sedel F, Saudubray JM. Disorders of phospholipids, sphingolipids and fatty acids biosynthesis: toward a new category of inherited metabolic diseases. J Inherit Metab Dis. 2013;36:411–425. doi: 10.1007/s10545-012-9509-7. [DOI] [PubMed] [Google Scholar]
- Larsson NG, Wang J, Wilhelmsson H, Oldfors A, Rustin P, Lewandoski M, et al. Mitochondrial transcription factor A is necessary for mtDNA maintenance and embryogenesis in mice. Nat Genet. 1998;18:231–236. doi: 10.1038/ng0398-231. [DOI] [PubMed] [Google Scholar]
- Laugel V, This-Bernd V, Cormier-Daire V, Speeg-Schatz C, de Saint-Martin A, Fischbach M. Early-onset ophthalmoplegia in Leigh-like syndrome due to NDUFV1 mutations. Pediatr Neurol. 2007;36:54–57. doi: 10.1016/j.pediatrneurol.2006.08.007. [DOI] [PubMed] [Google Scholar]
- Leveen P, Kotarsky H, Morgelin M, Karikoski R, Elmer E, Fellman V. The GRACILE mutation introduced into Bcs1l causes postnatal complex III deficiency: a viable mouse model for mitochondrial hepatopathy. Hepatology. 2011;53:437–447. doi: 10.1002/hep.24031. [DOI] [PubMed] [Google Scholar]
- Levene M, Coleman DG, Kilpatrick HC, Fairbanks LD, Gangadharan B, Gasson C, et al. Preclinical toxicity evaluation of erythrocyte-encapsulated thymidine phosphorylase in BALB/c mice and beagle dogs: an enzyme-replacement therapy for mitochondrial neurogastrointestinal encephalomyopathy. Toxicol Sci. 2013;131:311–324. doi: 10.1093/toxsci/kfs278. [DOI] [PubMed] [Google Scholar]
- Libri V, Brown AP, Gambarota G, Haddad J, Shields GS, Dawes H, et al. A pilot randomized, placebo controlled, double blind phase I trial of the novel SIRT1 activator SRT2104 in elderly volunteers. PLoS ONE. 2012;7:e51395. doi: 10.1371/journal.pone.0051395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liet JM, Pelletier V, Robinson BH, Laryea MD, Wendel U, Morneau S, et al. The effect of short-term dimethylglycine treatment on oxygen consumption in cytochrome oxidase deficiency: a double-blind randomized crossover clinical trial. J Pediatr. 2003;142:62–66. doi: 10.1067/mpd.2003.mpd0333. [DOI] [PubMed] [Google Scholar]
- Lin CS, Sharpley MS, Fan W, Waymire KG, Sadun AA, Carelli V, et al. Mouse mtDNA mutant model of Leber hereditary optic neuropathy. Proc Natl Acad Sci U S A. 2012;109:20065–20070. doi: 10.1073/pnas.1217113109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyon MF, Hulse EV. An inherited kidney disease of mice resembling human nephronophthisis. J Med Genet. 1971;8:41–48. doi: 10.1136/jmg.8.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyrawati D, Trounson A, Cram D. Expression of GFP in the mitochondrial compartment using DQAsome-mediated delivery of an artificial mini-mitochondrial genome. Pharm Res. 2011;28:2848–2862. doi: 10.1007/s11095-011-0544-0. [DOI] [PubMed] [Google Scholar]
- Mahata B, Mukherjee S, Mishra S, Bandyopadhyay A, Adhya S. Functional delivery of a cytosolic tRNA into mutant mitochondria of human cells. Science. 2006;314:471–474. doi: 10.1126/science.1129754. [DOI] [PubMed] [Google Scholar]
- Malojcic B, Brinar V, Poser C, Djakovic V. An adult case of Leigh disease. Clin Neurol Neurosurg. 2004;106:237–240. doi: 10.1016/j.clineuro.2004.02.028. [DOI] [PubMed] [Google Scholar]
- Mancuso M, Orsucci D, Logerfo A, Rocchi A, Petrozzi L, Nesti C, et al. Oxidative stress biomarkers in mitochondrial myopathies, basally and after cysteine donor supplementation. J Neurol. 2010;257:774–781. doi: 10.1007/s00415-009-5409-7. [DOI] [PubMed] [Google Scholar]
- Mancuso M, Orsucci D, Angelini C, Bertini E, Carelli V, Comi GP, et al. Phenotypic heterogeneity of the 8344A>G mtDNA ‘MERRF’ mutation. Neurology. 2013;80:2049–2054. doi: 10.1212/WNL.0b013e318294b44c. [DOI] [PubMed] [Google Scholar]
- Manfredi G, Fu J, Ojaimi J, Sadlock JE, Kwong JQ, Guy J, et al. Rescue of a deficiency in ATP synthesis by transfer of MTATP6, a mitochondrial DNA-encoded gene, to the nucleus. Nat Genet. 2002;30:394–399. doi: 10.1038/ng851. [DOI] [PubMed] [Google Scholar]
- Marella M, Seo BB, Thomas BB, Matsuno-Yagi A, Yagi T. Successful amelioration of mitochondrial optic neuropathy using the yeast NDI1 gene in a rat animal model. PLoS ONE. 2010;5:e11472. doi: 10.1371/journal.pone.0011472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mari M, Morales A, Colell A, Garcia-Ruiz C, Fernandez-Checa JC. Mitochondrial glutathione, a key survival antioxidant. Antioxid Redox Signal. 2009;11:2685–2700. doi: 10.1089/ars.2009.2695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinelli D, Catteruccia M, Piemonte F, Pastore A, Tozzi G, et al. EPI-743 reverses the progression of the pediatric mitochondrial disease – genetically defined Leigh Syndrome. Mol Genet Metab. 2012;107:383–388. doi: 10.1016/j.ymgme.2012.09.007. [DOI] [PubMed] [Google Scholar]
- Mayr JA, Zimmermann FA, Fauth C, Bergheim C, Meierhofer D, Radmayr D, et al. Lipoic acid synthetase deficiency causes neonatal-onset epilepsy, defective mitochondrial energy metabolism, and glycine elevation. Am J Hum Genet. 2011;89:792–797. doi: 10.1016/j.ajhg.2011.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayr JA, Haack TB, Graf E, Zimmermann FA, Wieland T, Haberberger B, et al. Lack of the mitochondrial protein acylglycerol kinase causes Sengers syndrome. Am J Hum Genet. 2012;90:314–320. doi: 10.1016/j.ajhg.2011.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michot C, Hubert L, Romero NB, Gouda A, Mamoune A, Mathew S, et al. Study of LPIN1, LPIN2 and LPIN3 in rhabdomyolysis and exercise-induced myalgia. J Inherit Metab Dis. 2012;35:1119–1128. doi: 10.1007/s10545-012-9461-6. [DOI] [PubMed] [Google Scholar]
- Mitsuhashi S, Ohkuma A, Talim B, Karahashi M, Koumura T, Aoyama C, et al. A congenital muscular dystrophy with mitochondrial structural abnormalities caused by defective de novo phosphatidylcholine biosynthesis. Am J Hum Genet. 2011;88:845–851. doi: 10.1016/j.ajhg.2011.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monteiro JP, Oliveira PJ, Jurado AS. Mitochondrial membrane lipid remodeling in pathophysiology: a new target for diet and therapeutic interventions. Prog Lipid Res. 2013;52:513–528. doi: 10.1016/j.plipres.2013.06.002. [DOI] [PubMed] [Google Scholar]
- Montini G, Malaventura C, Salviati L. Early coenzyme Q10 supplementation in primary coenzyme Q10 deficiency. N Engl J Med. 2008;358:2849–2850. doi: 10.1056/NEJMc0800582. [DOI] [PubMed] [Google Scholar]
- Moran NF, Bain MD, Muqit MM, Bax BE. Carrier erythrocyte entrapped thymidine phosphorylase therapy for MNGIE. Neurology. 2008;71:686–688. doi: 10.1212/01.wnl.0000324602.97205.ab. [DOI] [PubMed] [Google Scholar]
- Munnich A, Rustin P, Rotig A, Chretien D, Bonnefont JP, Nuttin C, et al. Clinical aspects of mitochondrial disorders. J Inherit Metab Dis. 1992;15:448–455. doi: 10.1007/BF01799603. [DOI] [PubMed] [Google Scholar]
- Murphy JL, Blakely EL, Schaefer AM, He L, Wyrick P, Haller RG, et al. Resistance training in patients with single, large-scale deletions of mitochondrial DNA. Brain. 2008;131:2832–2840. doi: 10.1093/brain/awn252. [DOI] [PubMed] [Google Scholar]
- Naini A, Kaufmann P, Shanske S, Engelstad K, De Vivo DC, Schon EA. Hypocitrullinemia in patients with MELAS: an insight into the ‘MELAS paradox’. J Neurol Sci. 2005;229–230:187–193. doi: 10.1016/j.jns.2004.11.026. [DOI] [PubMed] [Google Scholar]
- Nesbitt V, Pitceathly RD, Turnbull DM, Taylor RW, Sweeney MG, Mudanohwo EE, et al. The UK MRC Mitochondrial Disease Patient Cohort Study: clinical phenotypes associated with the m.3243A>G mutation–implications for diagnosis and management. J Neurol Neurosurg Psychiatry. 2013;84:936–938. doi: 10.1136/jnnp-2012-303528. [DOI] [PubMed] [Google Scholar]
- Nuffield Council on Bioethics UK. 2012. Novel techniques for the prevention of mitochondrial DNA disorders: an ethical review. Available at: http://www.nuffieldbioethics.org/sites/default/files/Novel_techniques_for_the_prevention_of_mitochondrial_DNA_disorders_compressed.pdf (accessed 20 August 2013)
- Nunnari J, Suomalainen A. Mitochondria: in sickness and in health. Cell. 2012;148:1145–1159. doi: 10.1016/j.cell.2012.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogle RF, Christodoulou J, Fagan E, Blok RB, Kirby DM, Seller KL, et al. Mitochondrial myopathy with tRNA(Leu(UUR)) mutation and complex I deficiency responsive to riboflavin. J Pediatr. 1997;130:138–145. doi: 10.1016/s0022-3476(97)70323-8. [DOI] [PubMed] [Google Scholar]
- Oguro H, Iijima K, Takahashi K, Nagai A, Bokura H, Yamaguchi S, et al. Successful treatment with succinate in a patient with MELAS. Intern Med. 2004;43:427–431. doi: 10.2169/internalmedicine.43.427. [DOI] [PubMed] [Google Scholar]
- Oppenheim ML, Hargreaves IP, Pope S, Land JM, Heales SJ. Mitochondrial cytochrome c release: a factor to consider in mitochondrial disease? J Inherit Metab Dis. 2009;32:269–273. doi: 10.1007/s10545-009-1061-8. [DOI] [PubMed] [Google Scholar]
- Orrenius S, Zhivotovsky B. Cardiolipin oxidation sets cytochrome c free. Nat Chem Biol. 2005;1:188–189. doi: 10.1038/nchembio0905-188. [DOI] [PubMed] [Google Scholar]
- Patti ME, Corvera S. The role of mitochondria in the pathogenesis of type 2 diabetes. Endocr Rev. 2010;31:364–395. doi: 10.1210/er.2009-0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrosillo G, Moro N, Ruggiero FM, Paradies G. Melatonin inhibits cardiolipin peroxidation in mitochondria and prevents the mitochondrial permeability transition and cytochrome c release. Free Radic Biol Med. 2009;47:969–974. doi: 10.1016/j.freeradbiomed.2009.06.032. [DOI] [PubMed] [Google Scholar]
- Pfeffer G, Majamaa K, Turnbull DM, Thorburn D, Chinnery PF. Treatment for mitochondrial disorders. Cochrane Database Syst Rev. 2012;(4) doi: 10.1002/14651858.CD004426.pub3. CD004426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phoenix C, Schaefer AM, Elson JL, Morava E, Bugiani M, Uziel G, et al. A scale to monitor progression and treatment of mitochondrial disease in children. Neuromuscul Disord. 2006;16:814–820. doi: 10.1016/j.nmd.2006.08.006. [DOI] [PubMed] [Google Scholar]
- Pineda M, Ormazabal A, Lopez-Gallardo E, Nascimento A, Solano A, Herrero MD, et al. Cerebral folate deficiency and leukoencephalopathy caused by a mitochondrial DNA deletion. Ann Neurol. 2006;59:394–398. doi: 10.1002/ana.20746. [DOI] [PubMed] [Google Scholar]
- Piruat JI, Pintado CO, Ortega-Saenz P, Roche M, Lopez-Barneo J. The mitochondrial SDHD gene is required for early embryogenesis, and its partial deficiency results in persistent carotid body glomus cell activation with full responsiveness to hypoxia. Mol Cell Biol. 2004;24:10933–10940. doi: 10.1128/MCB.24.24.10933-10940.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitts-Tucker T. UK fertilisation authority launches consultation on mitochondrial replacement techniques. BMJ. 2012;345:e6259. doi: 10.1136/bmj.e6259. [DOI] [PubMed] [Google Scholar]
- Pope S, Land JM, Heales SJ. Oxidative stress and mitochondrial dysfunction in neurodegeneration; cardiolipin a critical target? Biochim Biophys Acta. 2008;1777:794–799. doi: 10.1016/j.bbabio.2008.03.011. [DOI] [PubMed] [Google Scholar]
- Prescribing and Primary Care Services, Health and Social Care Information Centre. 2012. Prescriptions dispensed in the community: England 2002–12. Available at: https://catalogue.ic.nhs.uk/publications/prescribing/primary/pres-disp-com-eng-2002-12/pres-disp-com-eng-2002-12-rep.pdf (accessed 20 August 2013)
- Qi X, Qvit N, Su YC, Mochly-Rosen D. A novel Drp1 inhibitor diminishes aberrant mitochondrial fission and neurotoxicity. J Cell Sci. 2013;126:789–802. doi: 10.1242/jcs.114439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman S. Mitochondrial disease and epilepsy. Dev Med Child Neurol. 2012;54:397–406. doi: 10.1111/j.1469-8749.2011.04214.x. [DOI] [PubMed] [Google Scholar]
- Rahman S. Gastrointestinal and hepatic manifestations of mitochondrial disorders. J Inherit Metab Dis. 2013;36:659–673. doi: 10.1007/s10545-013-9614-2. [DOI] [PubMed] [Google Scholar]
- Rahman S, Hanna MG. Diagnosis and therapy in neuromuscular disorders: diagnosis and new treatments in mitochondrial diseases. J Neurol Neurosurg Psychiatry. 2009;80:943–953. doi: 10.1136/jnnp.2008.158279. [DOI] [PubMed] [Google Scholar]
- Rahman S, Thorburn DR. 189th ENMC international workshop complex I deficiency: diagnosis and treatment 20–22 April 2012, Naarden, The Netherlands. Neuromuscul Disord. 2013;23:506–515. doi: 10.1016/j.nmd.2013.03.004. [DOI] [PubMed] [Google Scholar]
- Rahman S, Hargreaves I, Clayton P, Heales S. Neonatal presentation of coenzyme Q10 deficiency. J Pediatr. 2001;139:456–458. doi: 10.1067/mpd.2001.117575. [DOI] [PubMed] [Google Scholar]
- Rahman S, Clarke CF, Hirano M. 176th ENMC International Workshop: diagnosis and treatment of coenzyme Q(1)(0) deficiency. Neuromuscul Disord. 2012;22:76–86. doi: 10.1016/j.nmd.2011.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rikimaru M, Ohsawa Y, Wolf AM, Nishimaki K, Ichimiya H, Kamimura N, et al. Taurine ameliorates impaired the mitochondrial function and prevents stroke-like episodes in patients with MELAS. Intern Med. 2012;51:3351–3357. doi: 10.2169/internalmedicine.51.7529. [DOI] [PubMed] [Google Scholar]
- Robinson BH. Use of fibroblast and lymphoblast cultures for detection of respiratory chain defects. Methods Enzymol. 1996;264:454–464. doi: 10.1016/s0076-6879(96)64041-5. [DOI] [PubMed] [Google Scholar]
- Robinson BH, Glerum DM, Chow W, Petrova-Benedict R, Lightowlers R, Capaldi R. The use of skin fibroblast cultures in the detection of respiratory chain defects in patients with lacticacidemia. Pediatr Res. 1990;28:549–555. doi: 10.1203/00006450-199011000-00027. [DOI] [PubMed] [Google Scholar]
- Rodriguez MC, MacDonald JR, Mahoney DJ, Parise G, Beal MF, Tarnopolsky MA. Beneficial effects of creatine, CoQ10, and lipoic acid in mitochondrial disorders. Muscle Nerve. 2007;35:235–242. doi: 10.1002/mus.20688. [DOI] [PubMed] [Google Scholar]
- Roef MJ, de Meer K, Reijngoud DJ, Straver HW, de Barse M, et al. Triacylglycerol infusion does not improve hyperlactemia in resting patients with mitochondrial myopathy due to complex I deficiency. Am J Clin Nutr. 2002a;75:228–236. doi: 10.1093/ajcn/75.2.228. [DOI] [PubMed] [Google Scholar]
- Roef MJ, de Meer K, Reijngoud DJ, Straver HW, de Barse M, Kalhan SC, et al. Triacylglycerol infusion improves exercise endurance in patients with mitochondrial myopathy due to complex I deficiency. Am J Clin Nutr. 2002b;75:237–244. doi: 10.1093/ajcn/75.2.237. [DOI] [PubMed] [Google Scholar]
- Rötig A, de Lonlay P, Chretien D, Foury F, Koenig M, Sidi D, et al. Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet. 1997;17:215–217. doi: 10.1038/ng1097-215. [DOI] [PubMed] [Google Scholar]
- Salmi H, Leonard JV, Rahman S, Lapatto R. Plasma thiol status is altered in children with mitochondrial diseases. Scand J Clin Lab Invest. 2012;72:152–157. doi: 10.3109/00365513.2011.646299. [DOI] [PubMed] [Google Scholar]
- Santra S, Gilkerson RW, Davidson M, Schon EA. Ketogenic treatment reduces deleted mitochondrial DNAs in cultured human cells. Ann Neurol. 2004;56:662–669. doi: 10.1002/ana.20240. [DOI] [PubMed] [Google Scholar]
- Schagger H, Pfeiffer K. Supercomplexes in the respiratory chains of yeast and mammalian mitochondria. EMBO J. 2000;19:1777–1783. doi: 10.1093/emboj/19.8.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlame M, Ren M. Barth syndrome, a human disorder of cardiolipin metabolism. FEBS Lett. 2006;580:5450–5455. doi: 10.1016/j.febslet.2006.07.022. [DOI] [PubMed] [Google Scholar]
- Scholte HR, Busch HF, Bakker HD, Bogaard JM, Luyt-Houwen IE, Kuyt LP. Riboflavin-responsive complex I deficiency. Biochim Biophys Acta. 1995;1271:75–83. doi: 10.1016/0925-4439(95)00013-t. [DOI] [PubMed] [Google Scholar]
- Schwimmer C, Rak M, Lefebvre-Legendre L, Duvezin-Caubet S, Plane G, di Rago JP. Yeast models of human mitochondrial diseases: from molecular mechanisms to drug screening. Biotechnol J. 2006;1:270–281. doi: 10.1002/biot.200500053. [DOI] [PubMed] [Google Scholar]
- Serrano M, Perez-Duenas B, Montoya J, Ormazabal A, Artuch R. Genetic causes of cerebral folate deficiency: clinical, biochemical and therapeutic aspects. Drug Discov Today. 2012;17:1299–1306. doi: 10.1016/j.drudis.2012.07.008. [DOI] [PubMed] [Google Scholar]
- Shamseldin HE, Alshammari M, Al-Sheddi T, Salih MA, Alkhalidi H, Kentab A, et al. Genomic analysis of mitochondrial diseases in a consanguineous population reveals novel candidate disease genes. J Med Genet. 2012;49:234–241. doi: 10.1136/jmedgenet-2012-100836. [DOI] [PubMed] [Google Scholar]
- Spector R, Johanson CE. Choroid plexus failure in the Kearns–Sayre syndrome. Cerebrospinal Fluid Res. 2010;7:14. doi: 10.1186/1743-8454-7-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stacpoole PW, Barnes CL, Hurbanis MD, Cannon SL, Kerr DS. Treatment of congenital lactic acidosis with dichloroacetate. Arch Dis Child. 1997;77:535–541. doi: 10.1136/adc.77.6.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stacpoole PW, Kerr DS, Barnes C, Bunch ST, Carney PR, Fennell EM, et al. Controlled clinical trial of dichloroacetate for treatment of congenital lactic acidosis in children. Pediatrics. 2006;117:1519–1531. doi: 10.1542/peds.2005-1226. [DOI] [PubMed] [Google Scholar]
- Stacpoole PW, deGrauw TJ, Feigenbaum AS, Hoppel C, Kerr DS, McCandless SE, et al. Design and implementation of the first randomized controlled trial of coenzyme CoQ(1)(0) in children with primary mitochondrial diseases. Mitochondrion. 2012;12:623–629. doi: 10.1016/j.mito.2012.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stetler AR, Leak RK, Chen J. The dynamics of the mitochondrial organelle as a potential therapeutic target. J Cereb Blood Flow Metab. 2013;33:22–32. doi: 10.1038/jcbfm.2012.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan PG, Rippy NA, Dorenbos K, Concepcion RC, Agarwal AK, Rho JM. The ketogenic diet increases mitochondrial uncoupling protein levels and activity. Ann Neurol. 2004;55:576–580. doi: 10.1002/ana.20062. [DOI] [PubMed] [Google Scholar]
- Suomalainen A. Therapy for mitochondrial disorders: little proof, high research activity, some promise. Semin Fetal Neonatal Med. 2011;16:236–240. doi: 10.1016/j.siny.2011.05.003. [DOI] [PubMed] [Google Scholar]
- Suomalainen A, Elo JM, Pietilainen KH, Hakonen AH, Sevastianova K, Korpela M, et al. FGF-21 as a biomarker for muscle-manifesting mitochondrial respiratory chain deficiencies: a diagnostic study. Lancet Neurol. 2011;10:806–818. doi: 10.1016/S1474-4422(11)70155-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taanman JW, Muddle JR, Muntau AC. Mitochondrial DNA depletion can be prevented by dGMP and dAMP supplementation in a resting culture of deoxyguanosine kinase-deficient fibroblasts. Hum Mol Genet. 2003;12:1839–1845. doi: 10.1093/hmg/ddg192. [DOI] [PubMed] [Google Scholar]
- Tachibana M, Sparman M, Sritanaudomchai H, Ma H, Clepper L, Woodward J, et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature. 2009;461:367–372. doi: 10.1038/nature08368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tachibana M, Amato P, Sparman M, Woodward J, Sanchis DM, Ma H, et al. Towards germline gene therapy of inherited mitochondrial diseases. Nature. 2013;493:627–631. doi: 10.1038/nature11647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taivassalo T, Fu K, Johns T, Arnold D, Karpati G, Shoubridge EA. Gene shifting: a novel therapy for mitochondrial myopathy. Hum Mol Genet. 1999;8:1047–1052. doi: 10.1093/hmg/8.6.1047. [DOI] [PubMed] [Google Scholar]
- Taivassalo T, Shoubridge EA, Chen J, Kennaway NG, DiMauro S, Arnold DL, et al. Aerobic conditioning in patients with mitochondrial myopathies: physiological, biochemical, and genetic effects. Ann Neurol. 2001;50:133–141. doi: 10.1002/ana.1050. [DOI] [PubMed] [Google Scholar]
- Taivassalo T, Gardner JL, Taylor RW, Schaefer AM, Newman J, Barron MJ, et al. Endurance training and detraining in mitochondrial myopathies due to single large-scale mtDNA deletions. Brain. 2006;129:3391–3401. doi: 10.1093/brain/awl282. [DOI] [PubMed] [Google Scholar]
- Tanaka M, Borgeld HJ, Zhang J, Muramatsu S, Gong JS, Yoneda M, et al. Gene therapy for mitochondrial disease by delivering restriction endonuclease SmaI into mitochondria. J Biomed Sci. 2002;9:534–541. doi: 10.1159/000064726. [DOI] [PubMed] [Google Scholar]
- Tarnopolsky MA, Roy BD, MacDonald JR. A randomized, controlled trial of creatine monohydrate in patients with mitochondrial cytopathies. Muscle Nerve. 1997;20:1502–1509. doi: 10.1002/(sici)1097-4598(199712)20:12<1502::aid-mus4>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- Taylor RW, Turnbull DM. Mitochondrial DNA mutations in human disease. Nat Rev Genet. 2005;6:389–402. doi: 10.1038/nrg1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorburn DR. Mitochondrial disorders: prevalence, myths and advances. J Inherit Metab Dis. 2004;27:349–362. doi: 10.1023/B:BOLI.0000031098.41409.55. [DOI] [PubMed] [Google Scholar]
- Toivonen JM, O'Dell KM, Petit N, Irvine SC, Knight GK, Lehtonen M, et al. Technical knockout, a Drosophila model of mitochondrial deafness. Genetics. 2001;159:241–254. doi: 10.1093/genetics/159.1.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Torronteras J, Gomez A, Eixarch H, Palenzuela L, Pizzorno G, et al. Hematopoietic gene therapy restores thymidine phosphorylase activity in a cell culture and a murine model of MNGIE. Gene Ther. 2011;18:795–806. doi: 10.1038/gt.2011.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyynismaa H, Mjosund KP, Wanrooij S, Lappalainen I, Ylikallio E, Jalanko A, et al. Mutant mitochondrial helicase Twinkle causes multiple mtDNA deletions and a late-onset mitochondrial disease in mice. Proc Natl Acad Sci U S A. 2005;102:17687–17692. doi: 10.1073/pnas.0505551102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyynismaa H, Carroll CJ, Raimundo N, Ahola-Erkkila S, Wenz T, Ruhanen H, et al. Mitochondrial myopathy induces a starvation-like response. Hum Mol Genet. 2010;19:3948–3958. doi: 10.1093/hmg/ddq310. [DOI] [PubMed] [Google Scholar]
- Vafai SB, Mootha VK. Mitochondrial disorders as windows into an ancient organelle. Nature. 2012;491:374–383. doi: 10.1038/nature11707. [DOI] [PubMed] [Google Scholar]
- Viscomi C, Burlina AB, Dweikat I, Savoiardo M, Lamperti C, Hildebrandt T, et al. Combined treatment with oral metronidazole and N-acetylcysteine is effective in ethylmalonic encephalopathy. Nat Med. 2010;16:869–871. doi: 10.1038/nm.2188. [DOI] [PubMed] [Google Scholar]
- Viscomi C, Bottani E, Civiletto G, Cerutti R, Moggio M, Fagiolari G, et al. In vivo correction of COX deficiency by activation of the AMPK/PGC-1alpha axis. Cell Metab. 2011;14:80–90. doi: 10.1016/j.cmet.2011.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vissing J, Gansted U, Quistorff B. Exercise intolerance in mitochondrial myopathy is not related to lactic acidosis. Ann Neurol. 2001;49:672–676. [PubMed] [Google Scholar]
- Wallace DC, Fan W, Procaccio V. Mitochondrial energetics and therapeutics. Annu Rev Pathol. 2010;5:297–348. doi: 10.1146/annurev.pathol.4.110807.092314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Wang J, Bonamy GM, Meeusen S, Brusch RG, Turk C, et al. A small molecule promotes mitochondrial fusion in mammalian cells. Angew Chem Int Ed Engl. 2012;51:9302–9305. doi: 10.1002/anie.201204589. [DOI] [PubMed] [Google Scholar]
- Waterham HR, Koster J, van Roermund CW, Mooyer PA, Wanders RJ, Leonard JV. A lethal defect of mitochondrial and peroxisomal fission. N Engl J Med. 2007;356:1736–1741. doi: 10.1056/NEJMoa064436. [DOI] [PubMed] [Google Scholar]
- Wedatilake Y, Brown R, McFarland R, Yaplito-Lee J, Morris AA, Champion M, et al. SURF1 deficiency: a multi-centre natural history study. Orphanet J Rare Dis. 2013;8:96. doi: 10.1186/1750-1172-8-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiher H, Noda T, Gray DA, Sharpe AH, Jaenisch R. Transgenic mouse model of kidney disease: insertional inactivation of ubiquitously expressed gene leads to nephrotic syndrome. Cell. 1990;62:425–434. doi: 10.1016/0092-8674(90)90008-3. [DOI] [PubMed] [Google Scholar]
- Wenz T, Diaz F, Spiegelman BM, Moraes CT. Activation of the PPAR/PGC-1alpha pathway prevents a bioenergetic deficit and effectively improves a mitochondrial myopathy phenotype. Cell Metab. 2008;8:249–256. doi: 10.1016/j.cmet.2008.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Wiedemann N, Frazier AE, Pfanner N. The protein import machinery of mitochondria. J Biol Chem. 2004;279:14473–14476. doi: 10.1074/jbc.R400003200. [DOI] [PubMed] [Google Scholar]
- Wortmann SB, Vaz FM, Gardeitchik T, Vissers LE, Renkema GH, Schuurs-Hoeijmakers JH, et al. Mutations in the phospholipid remodeling gene SERAC1 impair mitochondrial function and intracellular cholesterol trafficking and cause dystonia and deafness. Nat Genet. 2012;44:797–802. doi: 10.1038/ng.2325. [DOI] [PubMed] [Google Scholar]
- Yagi T, Seo BB, Nakamaru-Ogiso E, Marella M, Barber-Singh J, Yamashita T, et al. Possibility of transkingdom gene therapy for complex I diseases. Biochim Biophys Acta. 2006;1757:708–714. doi: 10.1016/j.bbabio.2006.01.011. [DOI] [PubMed] [Google Scholar]
- Yamada Y, Akita H, Kogure K, Kamiya H, Harashima H. Mitochondrial drug delivery and mitochondrial disease therapy – an approach to liposome-based delivery targeted to mitochondria. Mitochondrion. 2007;7:63–71. doi: 10.1016/j.mito.2006.12.003. [DOI] [PubMed] [Google Scholar]
- Yang H, Brosel S, Acin-Perez R, Slavkovich V, Nishino I, Khan R, et al. Analysis of mouse models of cytochrome c oxidase deficiency owing to mutations in Sco2. Hum Mol Genet. 2010;19:170–180. doi: 10.1093/hmg/ddp477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuzaki Y, Yamada Y, Harashima H. Mitochondrial matrix delivery using MITO-Porter, a liposome-based carrier that specifies fusion with mitochondrial membranes. Biochem Biophys Res Commun. 2010;397:181–186. doi: 10.1016/j.bbrc.2010.05.070. [DOI] [PubMed] [Google Scholar]
- Yatsuga S, Suomalainen A. Effect of bezafibrate treatment on late-onset mitochondrial myopathy in mice. Hum Mol Genet. 2012;21:526–535. doi: 10.1093/hmg/ddr482. [DOI] [PubMed] [Google Scholar]
- Yatsuga S, Povalko N, Nishioka J, Katayama K, Kakimoto N, Matsuishi T, et al. MELAS: a nationwide prospective cohort study of 96 patients in Japan. Biochim Biophys Acta. 2012;1820:619–624. doi: 10.1016/j.bbagen.2011.03.015. [DOI] [PubMed] [Google Scholar]
- Zeharia A, Shaag A, Houtkooper RH, Hindi T, de Lonlay P, Erez G, et al. Mutations in LPIN1 cause recurrent acute myoglobinuria in childhood. Am J Hum Genet. 2008;83:489–494. doi: 10.1016/j.ajhg.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X, Solaroli N, Bjerke M, Stewart JB, Rozell B, Johansson M, et al. Progressive loss of mitochondrial DNA in thymidine kinase 2-deficient mice. Hum Mol Genet. 2008;17:2329–2335. doi: 10.1093/hmg/ddn133. [DOI] [PubMed] [Google Scholar]
- Zuchner S, Mersiyanova IV, Muglia M, Bissar-Tadmouri N, Rochelle J, Dadali EL, et al. Mutations in the mitochondrial GTPase mitofusin 2 cause Charcot-Marie-Tooth neuropathy type 2A. Nat Genet. 2004;36:449–451. doi: 10.1038/ng1341. [DOI] [PubMed] [Google Scholar]