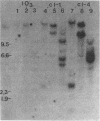
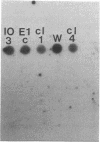
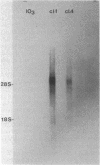
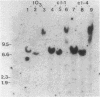
Abstract
Friend murine leukemia virus (Fr-MuLV) is a replication-competent murine retrovirus that induces acute nonlymphocytic leukemias in NFS/n mice. Fr-MuLV disease is divided into two stages based on the ability of the leukemia cells to grow in culture and transplant into syngeneic mice. Hematopoietic cells taken from the early stage of disease after Fr-MuLV infection grow as immortal myeloid cell lines in the presence of WEHI-3 cell-conditioned medium (CM) or interleukin 3. These growth factor-dependent cell lines do not grow in culture in the absence of CM and do not form tumors in syngeneic animals. If these Fr-MuLV-infected cells are superinfected with Abelson murine leukemia virus (Ab-MuLV), they lose their dependence on WEHI-3 CM and proliferate in culture in the absence of exogenous growth factors. Concomitant with the loss of growth factor dependence in culture, the Ab-MuLV-infected cell lines become tumorigenic in syngeneic mice. This secondary level of transformation is Ab-MuLV specific. Fr-MuLV-immortalized myeloid cell lines superinfected with Harvey murine sarcoma virus (Ha-MuSV) or amphotropic virus remain dependent on WEHI-3 CM for growth in vitro and are not tumorigenic in vivo. Neither Ab-MuLV- nor Ha-MuSV-infected normal mouse myeloid cell cultures produce growth factor-independent or tumorigenic cell lines. We conclude that at least two genetic events are needed to convert a murine myeloid precursor into a tumorigenic cell line. The first event occurs in Fr-MuLV-infected mice, generating cells that are growth factor dependent but immortal in vitro. The second event, which can be accomplished by Ab-MuLV infection, converts these immortal myeloid precursors into growth factor-independent and tumorigenic cells.
Full text
PDF

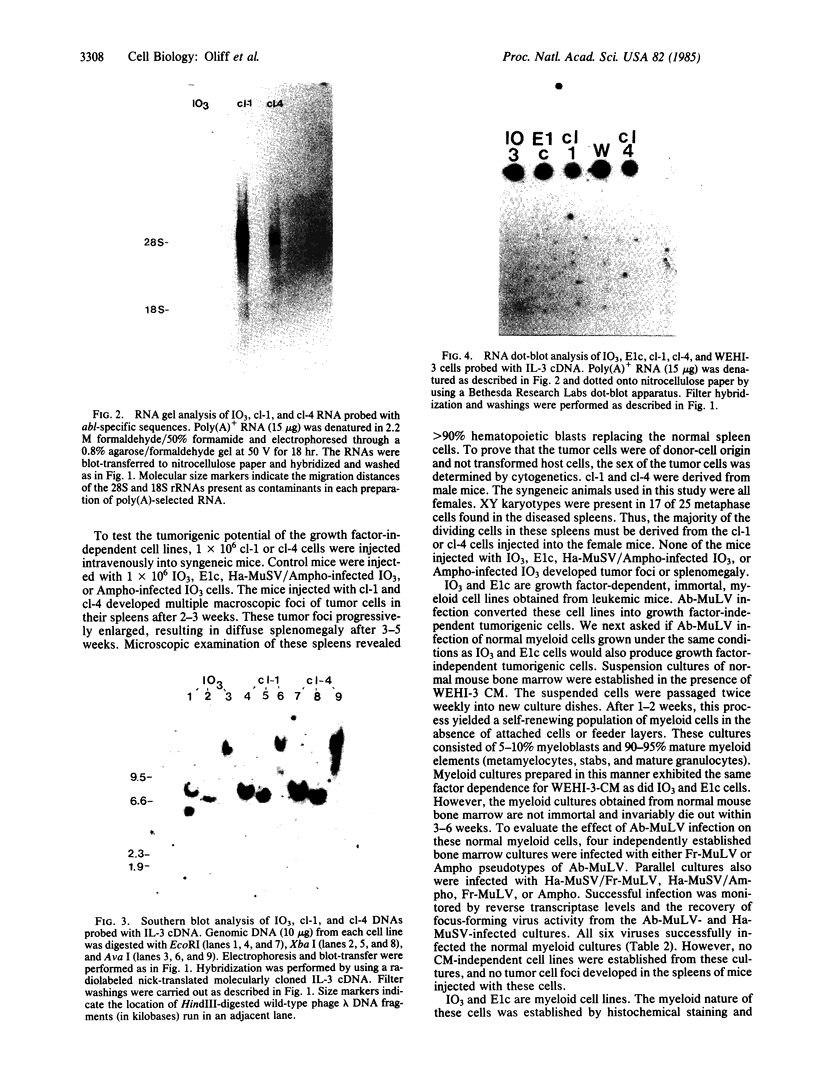


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abelson H. T., Rabstein L. S. Lymphosarcoma: virus-induced thymic-independent disease in mice. Cancer Res. 1970 Aug;30(8):2213–2222. [PubMed] [Google Scholar]
- Boss M., Greaves M., Teich N. Abelson virus-transformed haematopoietic cell lines with pre-B-cell characteristics. Nature. 1979 Apr 5;278(5704):551–553. doi: 10.1038/278551a0. [DOI] [PubMed] [Google Scholar]
- Chattopadhyay S. K., Oliff A. I., Linemeyer D. L., Lander M. R., Lowy D. R. Genomes of murine leukemia viruses isolated from wild mice. J Virol. 1981 Sep;39(3):777–791. doi: 10.1128/jvi.39.3.777-791.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dexter T. M., Garland J., Scott D., Scolnick E., Metcalf D. Growth of factor-dependent hemopoietic precursor cell lines. J Exp Med. 1980 Oct 1;152(4):1036–1047. doi: 10.1084/jem.152.4.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eliyahu D., Raz A., Gruss P., Givol D., Oren M. Participation of p53 cellular tumour antigen in transformation of normal embryonic cells. Nature. 1984 Dec 13;312(5995):646–649. doi: 10.1038/312646a0. [DOI] [PubMed] [Google Scholar]
- Eva A., Robbins K. C., Andersen P. R., Srinivasan A., Tronick S. R., Reddy E. P., Ellmore N. W., Galen A. T., Lautenberger J. A., Papas T. S. Cellular genes analogous to retroviral onc genes are transcribed in human tumour cells. Nature. 1982 Jan 14;295(5845):116–119. doi: 10.1038/295116a0. [DOI] [PubMed] [Google Scholar]
- FRIEND C., HADDAD J. R. Tumor formation with transplants of spleen or liver from mice with virus-induced leukemia. J Natl Cancer Inst. 1960 Dec;25:1279–1285. [PubMed] [Google Scholar]
- Greenberger J. S., Davisson P. B., Gans P. J., Moloney W. C. In vitro induction of continuous acute promyelocytic leukemia cell lines by Friend or Abelson murine leukemia virus. Blood. 1979 May;53(5):987–1001. [PubMed] [Google Scholar]
- Hankins W. D., Scolnick E. M. Harvey and Kirsten sarcoma viruses promote the growth and differentiation of erythroid precursor cells in vitro. Cell. 1981 Oct;26(1 Pt 1):91–97. doi: 10.1016/0092-8674(81)90036-2. [DOI] [PubMed] [Google Scholar]
- Land H., Parada L. F., Weinberg R. A. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature. 1983 Aug 18;304(5927):596–602. doi: 10.1038/304596a0. [DOI] [PubMed] [Google Scholar]
- MacDonald M. E., Mak T. W., Bernstein A. Erythroleukemia induction by replication-competent type C viruses cloned from the anemia- and polycythemia-inducing isolates of Friend leukemia virus. J Exp Med. 1980 Jun 1;151(6):1493–1503. doi: 10.1084/jem.151.6.1493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neel B. G., Hayward W. S., Robinson H. L., Fang J., Astrin S. M. Avian leukosis virus-induced tumors have common proviral integration sites and synthesize discrete new RNAs: oncogenesis by promoter insertion. Cell. 1981 Feb;23(2):323–334. doi: 10.1016/0092-8674(81)90128-8. [DOI] [PubMed] [Google Scholar]
- Oliff A. I., Hager G. L., Chang E. H., Scolnick E. M., Chan H. W., Lowy D. R. Transfection of molecularly cloned Friend murine leukemia virus DNA yields a highly leukemogenic helper-independent type C virus. J Virol. 1980 Jan;33(1):475–486. doi: 10.1128/jvi.33.1.475-486.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliff A., Collins L., Mirenda C. Molecular cloning of Friend mink cell focus-inducing virus: identification of mink cell focus-inducing virus-like messages in normal and transformed cells. J Virol. 1983 Nov;48(2):542–546. doi: 10.1128/jvi.48.2.542-546.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliff A., Oliff I., Schmidt B., Famulari N. Isolation of immortal cell lines from the first stage of murine leukemia virus-induced leukemia. Proc Natl Acad Sci U S A. 1984 Sep;81(17):5464–5467. doi: 10.1073/pnas.81.17.5464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliff A., Ruscetti S., Douglass E. C., Scolnick E. Isolation of transplantable erythroleukemia cells from mice infected with helper-independent Friend murine leukemia virus. Blood. 1981 Aug;58(2):244–254. [PubMed] [Google Scholar]
- Payne G. S., Courtneidge S. A., Crittenden L. B., Fadly A. M., Bishop J. M., Varmus H. E. Analysis of avian leukosis virus DNA and RNA in bursal tumours: viral gene expression is not required for maintenance of the tumor state. Cell. 1981 Feb;23(2):311–322. doi: 10.1016/0092-8674(81)90127-6. [DOI] [PubMed] [Google Scholar]
- Rasheed S., Gardner M. B., Chan E. Amphotropic host range of naturally occuring wild mouse leukemia viruses. J Virol. 1976 Jul;19(1):13–18. doi: 10.1128/jvi.19.1.13-18.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rein A. Interference grouping of murine leukemia viruses: a distinct receptor for the MCF-recombinant viruses in mouse cells. Virology. 1982 Jul 15;120(1):251–257. doi: 10.1016/0042-6822(82)90024-1. [DOI] [PubMed] [Google Scholar]
- Rosenberg N., Baltimore D., Scher C. D. In vitro transformation of lymphoid cells by Abelson murine leukemia virus. Proc Natl Acad Sci U S A. 1975 May;72(5):1932–1936. doi: 10.1073/pnas.72.5.1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruley H. E. Adenovirus early region 1A enables viral and cellular transforming genes to transform primary cells in culture. Nature. 1983 Aug 18;304(5927):602–606. doi: 10.1038/304602a0. [DOI] [PubMed] [Google Scholar]
- Scher C. D., Scolnick E. M., Siegler R. Induction of erythroid leukaemia by Harvey and Kirsten sarcoma viruses. Nature. 1975 Jul 17;256(5514):225–226. doi: 10.1038/256225a0. [DOI] [PubMed] [Google Scholar]
- Scher C. D., Siegler R. Direct transformation of 3T3 cells by Abelson murine leukaemia virus. Nature. 1975 Feb 27;253(5494):729–731. doi: 10.1038/253729a0. [DOI] [PubMed] [Google Scholar]
- Slamon D. J., deKernion J. B., Verma I. M., Cline M. J. Expression of cellular oncogenes in human malignancies. Science. 1984 Apr 20;224(4646):256–262. doi: 10.1126/science.6538699. [DOI] [PubMed] [Google Scholar]
- Sporn M. B., Todaro G. J. Autocrine secretion and malignant transformation of cells. N Engl J Med. 1980 Oct 9;303(15):878–880. doi: 10.1056/NEJM198010093031511. [DOI] [PubMed] [Google Scholar]
- Tambourin P. E., Wendling F., Jasmin C., Smadja-Joffe F. The physiopathology of Friend leukemia. Leuk Res. 1979;3(3):117–129. doi: 10.1016/0145-2126(79)90009-2. [DOI] [PubMed] [Google Scholar]
- Teich N., Boss M., Dexter T. M. Infection of mouse bone marrow cells with Abelson murine leukemia virus and establishment of producer cell lines. Haematol Blood Transfus. 1979;23:487–490. doi: 10.1007/978-3-642-67057-2_63. [DOI] [PubMed] [Google Scholar]
- Troxler D. H., Scolnick E. M. Rapid leukemia induced by cloned friend strain of replicating murine type-C virus. Association with induction of xenotropic-related RNA sequences contained in spleen focus-forming virus. Virology. 1978 Mar;85(1):17–27. doi: 10.1016/0042-6822(78)90408-7. [DOI] [PubMed] [Google Scholar]
- Westin E. H., Wong-Staal F., Gelmann E. P., Dalla-Favera R., Papas T. S., Lautenberger J. A., Eva A., Reddy E. P., Tronick S. R., Aaronson S. A. Expression of cellular homologues of retroviral onc genes in human hematopoietic cells. Proc Natl Acad Sci U S A. 1982 Apr;79(8):2490–2494. doi: 10.1073/pnas.79.8.2490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitlock C. A., Witte O. N. Abelson virus-infected cells can exhibit restricted in vitro growth and low oncogenic potential. J Virol. 1981 Nov;40(2):577–584. doi: 10.1128/jvi.40.2.577-584.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]