Abstract
Limited prospective data are available on the importance of estimated glomerular filtration rate (GFR) and proteinuria in the prediction of all-cause mortality (ACM) in community-based elderly populations. We examined the relationship between GFR or proteinuria and ACM in 949 randomly selected community-dwelling elderly subjects (aged ≥65 years) over a 5-year period. A spot urine sample was used to measure proteinuria by the dipstick test, and GFR was estimated using the chronic kidney disease-epidemiology collaboration (CKD-EPI) equation. Information about mortality and causes of death was collected by direct enquiry with the subjects and from the national mortality data. Compared to subjects without proteinuria, those with proteinuria of grade ≥1+ had a 1.725-fold (1.134–2.625) higher risk of ACM. Compared to subjects with GFR ≥90 ml/min/1.73 m2, those with GFR<45 ml/min/1.73 m2 had a 2.357 -fold (1.170–4.750) higher risk for ACM. Among the 403 subjects included in the analysis of renal progression, the annual rate of GFR change during follow-up period was −0.52±2.35 ml/min/1.73 m2/year. The renal progression rate was 7.315-fold (1.841–29.071) higher in subjects with GFR<60 ml/min/1.73 m2 than in those with GFR ≥60 ml/min/1.73 m2. Among a community-dwelling elderly Korean population, decreased GFR of <45 ml/min/1.73 m2 and proteinuria were independent risk factors for ACM.
Introduction
The prevalence of chronic kidney disease (CKD) has been increasing, causing concern both worldwide and in Korea [1]. Age is one of the most important risk factors related to the prevalence of CKD. The prevalence of a glomerular filtration rate (GFR) of <60 ml/min/1.73 m2 is 0.9% in subjects aged <60 years but increases to 51.2% among those aged ≥80 years in the USA [2]. The prevalence of albuminuria has been reported as 32.7% among a population of subjects aged ≥80 years [2]. However, studies evaluating the association of age with incident CKD among community-dwelling elderly populations in a prospective cohort are limited [2]. Studies on GFR changes with aging have shown that GFR declines steadily at a rate of 0.96 ml/min/1.73 m2 annually after 30–40 years of age and decreased more rapidly after the age of 60 years [3], [4]. Therefore, it is debatable whether mildly decreased GFR without definite evidences of renal damage, such as proteinuria or azotemia-related complications, should be considered as a “disease” in the elderly population [5]. In older populations, the prevalence of some of the CKD-related complications in subjects with GFR of 45–59 ml/min/1.73 m2 was not higher than that in subjects with GFR ≥60 ml/min/1.73 m2 [6]. A recent meta-analysis by the CKD prognosis consortium showed that the risks for mortality and renal progression were high in older populations with mildly decreased GFR [7]. However, there were several points to be cleared, which were reported in community-based prospective cohort studies. GFR has not been found to be a risk factor for all-cause mortality (ACM) [8], and the criterion of decreased renal function, in terms of GFR<60 ml/min/1.73 m2, has been found to be less suitable for the prediction of cardiovascular mortality (CVM) [9] and ACM [10]. Furthermore, albuminuria, rather than GFR, has been found to be a risk factor for incident cardiovascular disease [11]. Another issue is that Korean data included in the consortium were obtained from participants who underwent health examination in 2 selected centers and did not represent the community population [12]. Therefore, we sought to prospectively analyze the decline in the GFR, incidence of renal progression, and the effect of GFR and proteinuria on ACM in a randomly selected, community-based, elderly population residing in a Korean city, over a 5-year follow-up period.
Materials and Methods
Design of KLoSHA and Study Population
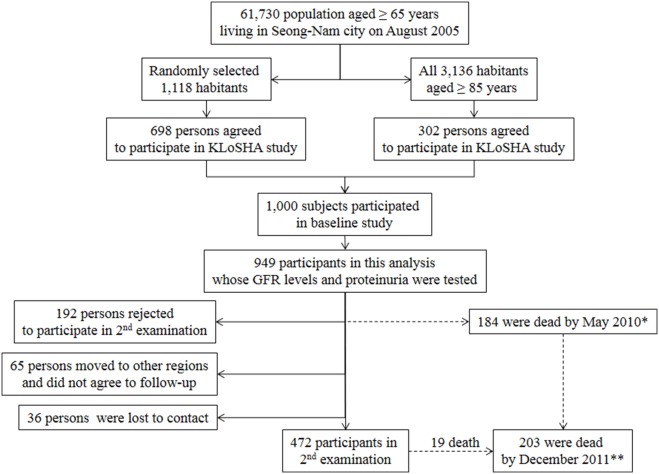
This study was conducted as a part of the Korean Longitudinal Study on Health and Aging (KLoSHA), which included a randomly selected, community-based, elderly population. The detailed design of the KLoSHA has been described in our previous report [13]. This study protocol was reviewed and approved by the institutional review board of the Seoul National University Bundang Hospital (B-0508/023-003). The study was conducted in accordance with the Declaration of Helsinki. The baseline study was conducted from September 2005 to September 2006; the follow-up study, from May 2010 to March 2012. After obtaining written informed consent from all participants, the assessments were performed at SNUBH. Among 1,000 subjects originally included in the KLoSHA, 949 subjects with baseline serum creatinine and proteinuria, as determined by the dipstick test (Figure 1) were enrolled in the current study.
Figure 1. Participants in KLoSHA study.
*Mortality was detected by direct contact and the national database. **Mortality was identified by the national database.
Demographic and Clinical Characteristics
Blood pressure values considered for the analysis were the means of 3 measurements. All medications taken by the participants were gathered and identified. Diabetes mellitus (DM) was defined by the use of anti-diabetic medicines, a serum fasting glucose level ≥126 mg/dl, or hemoglobin A1c (HbA1c) level ≥6.5%. Hypertension was defined as systolic blood pressure (SBP) ≥140 mmHg, diastolic blood pressure (DBP) ≥90 mmHg, or the use of anti-hypertensive medication. Proteinuria was defined as protein ≥1+, as determined by a dipstick urine test, while hematuria was defined as a red blood cell (RBC) count ≥5 per high-power field, as examined by a light microscopic examination of a urine sample. Serum creatinine level was measured by the alkaline picrate Jaffe kinetic method using an automatic analyzer (Toshiba 200FR, Tokyo, Japan). Serum creatinine levels were calibrated to an assay traceable on an isotope dilution mass spectrometry (IDMS) device (Roche diagnostics). GFR was calculated using the CKD-epidemiology collaboration (CKD-EPI) equation [14]. The participants were categorized into 4 groups on the basis of the GFR. Proteinuria was determined by the urine dipstick test, using an automated urine analyzer (CLINITEK ATLAS, Siemens Healthcare Diagnostics, Deerfield, IL, USA). Anemia was defined by hemoglobin levels <12 g/dL and <13 g/dL in women and men, respectively.
Outcomes
We obtained information about the survival status of the subjects participating in the baseline study via contact through telephone, cellular phone, and/or mail. This information was then confirmed by comparison against the mortality data maintained by the National Statistical Office (NSO) of Korea for the period between September 2005 and December 2011, using each individual’s unique identification number. The estimated GFR decline rate for the participants who were examined a second time was determined using covariate analysis (ANCOVA). We defined renal progression as an annual decrease rate in GFR of ≥2.5 ml/min/1.73 m2/year during the follow-up period and GFR<45 ml/min/1.73 m2 at the second examination [15].
Statistical Analyses
All analyses were performed using SPSS (SPSS version 20.0, Chicago, IL). We compared the cumulative incidences of ACM among the participants by using the log-rank test. Cox’s hazard proportional analysis was used to estimate the hazard ratios (HRs) for mortality. HRs for ACM was adjusted for age, gender, habit of smoking, CRP, cholesterol, triglyceride, albumin, platelet, and hemoglobin. We selected these adjusting factors which showed significant associations with mortality in the univariate analysis. Formal tests for multiplicative interactions were conducted by comparing −2 log likelihood in regression models, including the full population, with and without interaction terms (for example, risk factor 1 * risk factor 2). We compared the incidence of renal progression in the different groups using logistic regression analysis. The number of subjects with renal progression was small, and we grouped the subjects according to the GFR level of 60 ml/min/1.73 m2. P-values of 0.05 were considered statistically significant.
Results
Baseline Characteristics
The mean age of the participants was 75.8±9.0 years. DM was present in 24.2% of the subjects; hypertension, in 73.8%. The GFR was ≥90 ml/min/1.73 m2 in 15.2% of the subjects; 60–89 ml/min/1.73 m2, in 60.5%; 45–59 ml/min/1.73 m2, in 16.5%; and <45 ml/min/1.73 m2, in 7.8%. Proteinuria of grade ≥1+was noted in 8.2% of the population. The proportion of subjects with GFR<60 ml/min/1.73 m2 was higher in the oldest old group, aged ≥75 years, than in the group of subjects aged 65–74 years (41.2% vs. 11.2%) (Table 1).
Table 1. Basal characteristics of elderly population at baseline study according to age.
| All | Age<75 years | Age≥75 years | p-value | |
| Number | 949 | 534 | 415 | |
| Age (years) | 75.8±9.0 | 68.8±2.9 | 84.8±5.6 | <0.001 |
| Gender (Male, %) | 45.4 | 45.5 | 45.3 | 0.950 |
| BMI (kg/m2) | 24.0±3.3 | 24.6±3.2 | 22.9±3.2 | <0.001 |
| DBP (mmHg) | 82.7±10.6 | 83.5±10.3 | 81.5±10.9 | 0.003 |
| SBP (mmHg) | 132.3±17.9 | 132.3±16.8 | 132.3±19.2 | 0.973 |
| Smoking (%) | 0.202 | |||
| Never | 58.9 | 59.9 | 57.6 | |
| Ex-smoker | 29.6 | 27.5 | 32.3 | |
| Current smoker | 11.5 | 12.5 | 10.1 | |
| Diabetes mellitus (%) | 24.2 | 25.7 | 22.4 | 0.328 |
| Hypertension (%) | 73.8 | 72.6 | 75.4 | 0.279 |
| Hemoglobin (g/dL) | 13.7±1.5 | 14.0±1.4 | 13.3±1.5 | <0.001 |
| Anemia (%) | 8.6 | 4.3 | 14.3 | <0.001 |
| Creatinine (mg/dL) | 0.93±0.37 | 0.89±0.35 | 0.98±0.40 | 0.001 |
| GFR (ml/min/1.73 m2) | 72.2±17.0 | 78.1±14.5 | 64.6±17.0 | <0.001 |
| ≥90 | 15.2 | 24.2 | 3.6 | <0.001 |
| 60–89 | 60.5 | 64.6 | 55.2 | |
| 45–59 | 16.5 | 8.2 | 27.2 | |
| <45 | 7.8 | 3.0 | 14.0 | |
| Proteinuria by dipstick (%) | 0.012 | |||
| none | 83.6 | 86.7 | 79.5 | |
| trace | 8.2 | 6.6 | 10.4 | |
| 1+ or more | 8.2 | 6.7 | 10.1 | |
| Hematuria (%) | 10.5 | 9.7 | 11.6 | 0.363 |
| Medication (%) | ||||
| ACEI or ARB | 14.4 | 12.7 | 16.6 | 0.091 |
| Anti-platelet agent | 21.1 | 20.6 | 21.7 | 0.686 |
| Statin | 8.1 | 9.4 | 6.5 | 0.110 |
| No of AntiHTN (%) | 0.196 | |||
| 0 | 53.5 | 55.4 | 51.1 | |
| 1 | 28.9 | 28.5 | 29.4 | |
| 2 | 10.7 | 9.4 | 12.5 | |
| ≥3 | 6.9 | 7.7 | 7.0 |
GFR: Calculated by CKD-EPI equation, BMI: body mass index, Proteinuria: measured by dipstick test, Anemia: defined in female with hemoglobin less than 12 g/dL and, in male, less than 13 g/dL. Hematuria: RBC≥5/HPF, Anti-platelet agent: aspirin 100 mg, triflusal, sarpogrelate, or clopidogrel, No of AntiHTN: number of antihypertensive medication.
All-cause Mortality
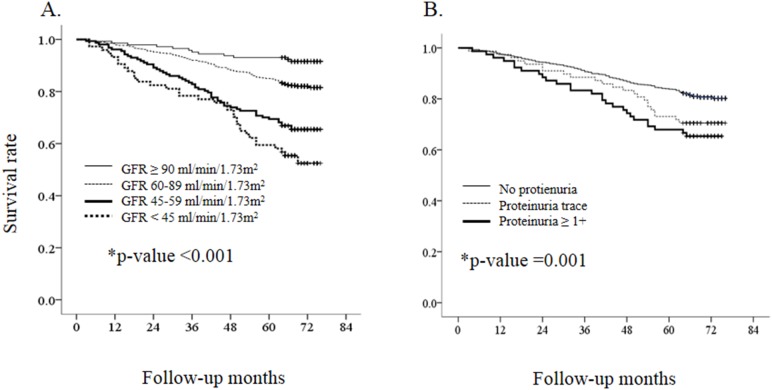
The follow-up duration for mortality was 63.4±16.4 months after the baseline visit. Overall, there were 203 cases of ACM and 4.05 deaths/100 person-years (Table 2). The estimated 5-year survival rates were 91.5% in GFR group 1, 89.7% in GFR group 2, 66.9% in GFR group 3, and 52.5% in GFR group 4 (p<0.001; Figure 2). The estimated 5-year survival rates were 92.4% in subjects with no proteinuria, 70.5% in subjects with trace proteinuria, and 65.4% in subjects with proteinuria ≥1+ (p = 0.001; Figure 2). In the estimation of mortality, no interactions were noted between the GFR group and proteinuria group, age group, or gender and between proteinuria group and age group or gender. Subjects with proteinuria of grade ≥1+ had a 1.725-fold higher risk for ACM than those without proteinuria (p = 0.011), and subjects with GFR<45 ml/min/1.73 m2 had a 2.357-fold higher risk for ACM than those with GFR ≥90 ml/min/1.73 m2 (p = 0.016; Table 3).
Table 2. The event rate of all-cause mortality according to basal characteristics.
| N | ACM rate | p * | ||
| /100 PY | (95% CI) | |||
| All | 949 | 4.05 | 3.98–4.11 | |
| Gender | 0.005 | |||
| Male | 431 | 4.92 | 4.80–5.06 | |
| Female | 518 | 3.34 | 3.27–3.41 | |
| Age (years) | <0.001 | |||
| <75 | 534 | 1.27 | 1.25–1.28 | |
| ≥75 | 415 | 8.48 | 8.20–8.77 | |
| DM | 0.825 | |||
| no | 719 | 4.10 | 4.02–4.18 | |
| yes | 230 | 3.90 | 3.78–4.03 | |
| Hypertension | 0.576 | |||
| no | 248 | 3.81 | 3.69–3.94 | |
| yes | 700 | 4.14 | 4.06–4.22 | |
| DBP (mmHg) | 0.246 | |||
| <80 | 196 | 5.03 | 4.82–5.27 | |
| 80–89 | 492 | 3.67 | 3.62–3.78 | |
| ≥90 | 259 | 4.03 | 3.92–4.15 | |
| SBP (mmHg) | 0.707 | |||
| <130 | 397 | 4.30 | 4.18–4.42 | |
| 130–149 | 350 | 3.73 | 3.64–3.83 | |
| ≥150 | 200 | 4.15 | 4.02–4.30 | |
| BP (mmHg) | 0.895 | |||
| <130/80 | 165 | 4.23 | 4.04–4.44 | |
| ≥130/80 | 782 | 4.02 | 3.95–4.09 | |
| GFR (ml/min/1.73 m2) | <0.001 | |||
| ≥90 | 144 | 1.47 | 1.43–1.51 | |
| 60–89 | 574 | 3.32 | 3.26–3.38 | |
| 45–59 | 157 | 7.10 | 6.74–7.50 | |
| <45 | 74 | 10.23 | 9.36–11.29 | |
| Proteinuria | 0.001 | |||
| none | 793 | 3.61 | 3.55–3.67 | |
| trace | 78 | 5.79 | 5.44–6.20 | |
| ≥1+ | 78 | 7.14 | 6.63–7.75 |
*ACM: all-cause mortality, p-value: calculated by Pearson’s Chi-square test, 100 PY: 100 person-years.
Figure 2. The survival rate according to GFR and proteinuria.
A. GFR group for all-caused mortality. B. Proteinuria group for all-caused mortality *p-value by Log-rank test.
Table 3. The significant effect on mortality of proteinuria and GFR.
| HR for ACM* | |
| Proteinuria group | |
| None | 1.000 (ref.) |
| trace | 1.235 (0.777–1.963) |
| ≥1+ | 1.725 (1.134–2.625) |
| GFR group | |
| ≥90 | 1.000 (ref.) |
| 60–89 | 1.370 (0.746–2.517) |
| 45–59 | 1.654 (0.841–3.254) |
| <45 | 2.357 (1.170–4.750) |
*Cox’s hazard proportional model adjusted with age, gender, habit of smoking, CRP, cholesterol, triglyceride, albumin, platelet, and hemoglobin.
ACM: all-cause mortality, ref.: reference group, HR: Hazard ratio, CI: confidence interval, NC: cannot be calculated.
GFR Decline and Renal Progression
The follow-up study was conducted 59.4±6.9 months after the baseline visit. Among the 472 subjects who underwent the follow-up examination, 403 were included in the analysis of renal progression. The annual rate of GFR change during the follow-up period was −0.52±2.35 ml/min/1.73 m2/year. Compared to participants without DM or lower GFR, those with DM or lower GFR showed a more rapid decline in GFR during the follow-up period (Table 4). The proportion of subjects with renal progression was 11/403 (2.7%). There was no interaction between GFR group and age or DM for renal progression. Negative interactions were found between gender and GFR group (p-interactions = 0.047). Multiple logistic regression analysis revealed that the renal progression rate was 7.315-fold (1.841–29.071) higher in subjects with GFR<60 ml/min/1.73 m2 than in those with GFR ≥60 ml/min/1.73 m2.
Table 4. GFR decline according to baseline characteristics in participants followed up 2nd examination.
| N | GFR decline * | p-value | N | GFR decline * | p-value | ||
| Mean (95% CI) | Mean (95% CI) | ||||||
| All | 403 | 0.52 (0.31–0.73) | SBP (mmHg) | 0.510 | |||
| Gender | 0.237 | <130 | 164 | 0.42 (0.07–0.76) | |||
| Male | 206 | 0.64 (0.35–0.93) | 130–149 | 158 | 0.49 (0.16–0.82)) | ||
| Female | 197 | 0.15 (0.09–0.69) | ≥150 | 81 | 0.77 (0.29–1.26) | ||
| Age (years) | 0.677 | BP (mmHg) | 0.068 | ||||
| <75 | 308 | 0.47 (0.15–0.78) | <130/80 | 67 | 0.08 (0.44 ** –0.59) | ||
| ≥75 | 75 | 0.68 (0.11 ** –1.47) | ≥130/80 | 336 | 0.61 (0.38–0.83) | ||
| DM | <0.001 | GFR (ml/min/1.73 m2) | 0.012 | ||||
| no | 304 | 0.29 (0.05–0.53) | ≥90 | 84 | 0.29 (0.40 **–0.96) | ||
| yes | 99 | 1.21 (0.79–1.63) | 60–89 | 260 | 0.38 (0.11–0.64) | ||
| HTN | 0.084 | 45–59 | 41 | 1.22 (0.33–2.10) | |||
| no | 112 | 0.22 (0.18–0.62) | <45 | 18 | 2.88 (1.41–4.35) | ||
| yes | 291 | 0.63 (0.39–0.88) | Proteinuria | 0.892 | |||
| DBP (mmHg) | 0.589 | none | 357 | 0.53 (0.31–0.75) | |||
| <80 | 75 | 0.28 (0.21–0.78) | trace | 24 | 0.56 (0.30 ** –1.42) | ||
| 80–89 | 216 | 0.57 (0.28–0.86) | ≥1+ | 22 | 0.31 (0.59 **–1.21) | ||
| ≥90 | 112 | 0.57 (0.15–0.99) |
*GFR decline*: GFR decline rate (ml/min/1.73 m2/year) estimated by covariate analysis adjusted for age, gender, DM, HTN, GFR, and proteinuria by dipstick test at baseline study.
**The number with bold character indicates GFR increase rate.
Discussion
In this community-based elderly cohort, the grade of GFR was found to be an independent risk factor for ACM. However, subjects with a GFR of 45–59 ml/min/1.73 m2 did not have higher risk for ACM than those with GFR ≥90 ml/min/1.73 m2. Proteinuria was an independent risk factor for ACM. The annual rate of GFR decline was 0.52 ml/min/1.73 m2. GFR<60 ml/min/1.73 m2 was an important risk factor for renal progression.
Studies analyzing the relationships between GFR or proteinuria and outcomes in the elderly have shown variable results depending on the characteristics of the enrolled population. Recent meta-analyses have shown that the HRs of low GFR (<60 ml/min/1.73 m2) with ACM [7], [16], [17], CVM[16], and end stage renal disease (ESRD) [7] were similar meanings irrespective of patient age. The studies included all of community populations, high-risk populations for CKD, and CKD populations and showed differences compared to community-based studies. The relationship between serum creatinine level and mortality was most evident in populations with traditional cardiovascular risk factors or renal insufficiency, and not in community-based cohorts [18]. Among patients (age, >55 years) who had documented vascular disease or diabetes with symptoms of end organ damage, the HRs for ACM according to GFR levels increased in proportion to the severity of the traditional risk factors for cardiovascular disease [19]. A study on patients visiting the outpatient clinics showed a less evident relationship between mild-to-moderately decreased GFR and outcomes. O’Hara et al. reported that, compared to elderly patients with GFR ≥60 ml/min/1.73 m2, those with a GFR of 50–59 ml/min/1.73 m2 did not have an increased risk for death [20]. A prospective cohort study of community-dwelling persons aged ≥65 years showed that GFR was not a risk factor for ACM and CVM [8]. Therefore, a randomly sampled community-based cohort with representativeness is more suitable for determining the effect of changes of GFR with aging on outcomes.
In this respect, the KLoSHA protocol offered several advantages in the evaluation of the relationship between renal parameters and outcomes. The majority of these data were sampled by random selection from non-institutionalized habitants. A prospective study provides higher quality of evidences than those provided by a cross-sectional study. We were able to contact almost all the participants for confirmation of mortality and verified this information against the data maintained by the National Statistical Office of Korea. All Koreans have a unique identifier, which is the primary key to obtain individualized data. Finally, serum creatinine was measured in a single institution throughout the study period and represented as IDMS-traceable value, which was suitable for application of the CKD-EPI equation.
The definition of GFR for the assessment of increased risk of mortality in elderly population can also vary with the nature of the included population. Several prospective community-based cohort studies showed that a GFR value of 60 ml/min/1.73 m2 was not a suitable cut-off for the prediction of ACM [8], [9], [20] or CVM [9], [10] among the elderly population. Other studies, including those on community populations, showed no relationship between CVM and GFR in populations of subjects aged ≥65 years [8], [18], [21] or ≥70 years [17]. The other issue is the effect of albuminuria among participants with decreased GFR on mortality. In elderly patients aged ≥60 years, GFR<60 ml/min/1.73 m2 estimated by serum creatinine was found to be a risk factor in participants with albuminuria, but not for those without albuminuria [22]. Albuminuria has been reported as a risk factor for ACM and CVM in elderly populations with high risk(s) [11], [16] and in community-based elderly cohorts [11], [23], [24], [25]. For example, in the PREVEND study, albuminuria, but not GFR, was found to be associated with CVM and ACM [11].
A few prospective studies have evaluated the association of age with the decline in estimated GFR with aging [2]. A longitudinal study on an elderly cohort (60–93 years) in Sweden revealed that the GFR estimated by the CKD-EPI equation declined at an annual rate of 1.042 ml/min/1.73 m2 in men and 0.970 ml/min/1.73 m2 in women [26]; these rates are higher than those noted in this study. Imai et al. reported that the annual decline in GFR using the MDRD equation in an elderly population (60–79 years) followed for 10 years [27] was 0.32–0.39 ml/min/1.73 m2 per year, which is similar to that obtained in our study. Ethnic differences in the rate of decline in GFR should be accounted for in a well-designed study. We used the criteria for renal progression used in the KDIGO controversy conferences [15]. Our study revealed that a GFR<60 ml/min/1.73 m2 was the most important risk factor of renal progression, as observed in other studies [8], [28].
There are several limitations in the interpretation of this study’s results. The CKD-EPI equation has not been fully validated in elderly Koreans, although our colleagues have reported that the ethnic coefficients of the CKD-EPI equations were close to “1,” on the basis of a study on 131 CKD patients and healthy volunteers [29]. We followed the participants over 5 years but 29.3% of initial population was not followed up at the second examination. The significant percent of dropout rate is an important limitation of this study. Especially, renal progression was evaluated in only subjects who completed follow up examination. Several criteria for renal outcomes have been proposed thus far, and we used the criteria defined at the KDIGO controversies conference [15]. In addition, we evaluated a relatively small numbers of subjects with severely decreased renal function and proteinuria. The results should be carefully interpreted in subjects with eGFR<45 ml/min/1.73 m2 or proteinuria. We evaluate the renal outcome on the basis of the difference of serum creatinine at baseline and at the end of the follow up study, and we did not confirm the sustained decline in eGFR during follow up period. Finally, this study is based on a city population, and the generalization of the results for a nationwide population or for all Asians is debatable.
In conclusion, we found that in a community-dwelling elderly Korean population, a decreased GFR<45 ml/min/1.73 m2 and proteinuria were independent risk factors for ACM. With aging, the annual rate of decrease in GFR was 0.52 ml/min/1.73 m2/year and the baseline GFR was the most important risk factor for renal progression.
Funding Statement
This work was supported by an independent Research Grant (IRG) from Pfizer Global Pharmaceuticals (grant number 06-05-039) and a Grant for developing Seongnam Health Promotion Program for the Elderly from Seongnam City Government in Korea (grant number 800-20050211. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Kim S, Lim CS, Han DC, Kim GS, Chin HJ, et al. (2009) The prevalence of chronic kidney disease (CKD) and the associated factors to CKD in urban Korea: a population-based cross-sectional epidemiologic study. J Korean Med Sci 24: S11–S21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Bowling CB, Muntner P (2012) Epidemiology of chronic kidney disease among older adults: a focus on the oldest old. J Gerontol A Biol Sci Med Sci 67: 1379–1386. [DOI] [PubMed] [Google Scholar]
- 3. Davies DF, Shock NW (1950) Age changes in glomerular filtration rate, effective renal plasma flow, and tubular excretory capacity in adult males. J Clin Invest 29: 496–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lindeman RD, Tobin J, Shock NW (1985) Longitudinal studies on the rate of decline in renal function with age. J Am Geriatr Soc 33: 278–285. [DOI] [PubMed] [Google Scholar]
- 5. Glassock RJ, Winearls C (2009) Ageing and the glomerular filtration rate: truths and consequences. Trans Am Clin Climatol Assoc 120: 419–428. [PMC free article] [PubMed] [Google Scholar]
- 6. Bowling CB, Inker LA, Gutiérrez OM, Allman RM, Warnock DG, et al. (2011) Age-specific associations of reduced estimated glomerular filtration rate with concurrent chronic kidney disease complications. Clin J Am Soc Nephrol 6: 2822–2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hallan SI, Matsushita K, Sang Y, Mahmoodi BK, Black C, et al. (2012) Chronic Kidney Disease Prognosis Consortium: Age and association of kidney measures with mortality and end-stage renal disease. JAMA 308: 2349–2360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Dalrymple LS, Katz R, Kestenbaum B, Shlipak MG, Sarnak MJ, et al. (2011) Chronic kidney disease and the risk of end-stage renal disease versus death. J Gen Intern Med 26: 379–385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Stengel B, Metzger M, Froissart M, Rainfray M, Berr C, et al. (2011) Epidemiology and prognostic significance of chronic kidney disease in the elderly–the Three-City prospective cohort study. Nephrol Dial Transplant 26: 3286–3295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nerpin E, Ingelsson E, Risérus U, Sundström J, Larsson A, et al. (2011) The combined contribution of albuminuria and glomerular filtration rate to the prediction of cardiovascular mortality in elderly men. Nephrol Dial Transplant 26: 2820–2827. [DOI] [PubMed] [Google Scholar]
- 11. Smink PA, Lambers Heerspink HJ, Gansevoort RT, de Jong PE, Hillege HL, et al. (2012) Albuminuria, estimated GFR, traditional risk factors, and incident cardiovascular disease: the PREVEND (Prevention of Renal and Vascular Endstage Disease) study. Am J Kidney Dis 60: 804–811. [DOI] [PubMed] [Google Scholar]
- 12. Kimm H, Yun JE, Jo J, Jee SH (2009) Low serum bilirubin level as an independent predictor of stroke incidence: a prospective study in Korean men and women. Stroke 40: 3422–3427. [DOI] [PubMed] [Google Scholar]
- 13. Chin HJ, Song YR, Lee JJ, Lee SB, Kim KW, et al. (2008) Moderately decreased renal function negatively affects the health-related quality of life among the elderly Korean population: a population-based study. Nephrol Dial Transplant 23: 2810–2817. [DOI] [PubMed] [Google Scholar]
- 14. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, et al. (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Levey AS, de Jong PE, Coresh J, El Nahas M, Astor BC, et al. (2011) The definition, classification, and prognosis of chronic kidney disease: a KDIGO Controversies Conference report. Kidney Int 80: 17–28. [DOI] [PubMed] [Google Scholar]
- 16. Chronic Kidney Disease Prognosis Consortium, Matsushita K, van der Velde M, Astor BC, Woodward M, et al. (2010) Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 375: 2073–2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Tonelli M, Wiebe N, Culleton B, House A, Rabbat C, et al. (2006) Chronic kidney disease and mortality risk: a systematic review. J Am Soc Nephrol 17: 2034–2047. [DOI] [PubMed] [Google Scholar]
- 18. Garg AX, Clark WF, Haynes RB, House AA (2002) Moderate renal insufficiency and the risk of cardiovascular mortality: results from the NHANES I. Kidney Int. 61: 1486–1494. [DOI] [PubMed] [Google Scholar]
- 19. Clase CM, Gao P, Tobe SW, McQueen MJ, Grosshennig A, et al. ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) and TRANSCEND (Telmisartan Randomized Assessment Study in Angiotensin-Converting-Enzyme-Inhibitor Intolerant Subjects with Cardiovascular Disease) (2011) Estimated glomerular filtration rate and albuminuria as predictors of outcomes in patients with high cardiovascular risk: a cohort study. Ann Intern Med 154: 310–318. [DOI] [PubMed] [Google Scholar]
- 20. O’Hare AM, Bertenthal D, Covinsky KE, Landefeld CS, Sen S, et al. (2006) Mortality risk stratification in chronic kidney disease: one size for all ages? J Am Soc Nephrol 17: 846–853. [DOI] [PubMed] [Google Scholar]
- 21. Culleton BF, Larson MG, Wilson PW, Evans JC, Parfrey PS, et al. (1999) Cardiovascular disease and mortality in a community-based cohort with mild renal insufficiency. Kidney Int 56: 2214–2219. [DOI] [PubMed] [Google Scholar]
- 22. van der Velde M, Bakker SJ, de Jong PE, Gansevoort RT (2010) Influence of age and measure of eGFR on the association between renal function and cardiovascular events. Clin J Am Soc Nephrol 5: 2053–2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Muntner P, Bowling CB, Gao L, Rizk D, Judd S, et al. (2011) Age-specific association of reduced estimated glomerular filtration rate and albuminuria with all-cause mortality. Clin J Am Soc Nephrol 6: 2200–2207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Hallan S, Astor B, Romundstad S, Aasarød K, Kvenild K, et al. (2007) Association of kidney function and albuminuria with cardiovascular mortality in older vs younger individuals: The HUNT II Study. Arch Intern Med 167: 2490–2496. [DOI] [PubMed] [Google Scholar]
- 25. Rifkin DE, Katz R, Chonchol M, Fried LF, Cao J, et al. (2010) Albuminuria, impaired kidney function and cardiovascular outcomes or mortality in the elderly. Nephrol Dial Transplant 25: 1560–1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Christensson A, Elmståhl S (2011) Estimation of the age-dependent decline of glomerular filtration rate from formulas based on creatinine and cystatin C in the general elderly population. Nephron Clin Pract 117: c40–c50. [DOI] [PubMed] [Google Scholar]
- 27. Imai E, Horio M, Yamagata K, Iseki K, Hara S, et al. (2008) Slower decline of glomerular filtration rate in the Japanese general population: a longitudinal 10-year follow-up study. Hypertens Res 31: 433–441. [DOI] [PubMed] [Google Scholar]
- 28. Fox CS, Larson MG, Leip EP, Culleton B, Wilson PW, et al. (2004) Predictors of new-onset kidney disease in a community-based population. JAMA 291: 844–850. [DOI] [PubMed] [Google Scholar]
- 29. Lee CS, Cha RH, Lim YH, Kim H, Song KH, et al. (2010) Ethnic coefficients for glomerular filtration rate estimation by the Modification of Diet in Renal Disease study equations in the Korean population. J Korean Med Sci 25: 1616–1625. [DOI] [PMC free article] [PubMed] [Google Scholar]