Abstract
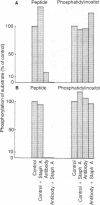
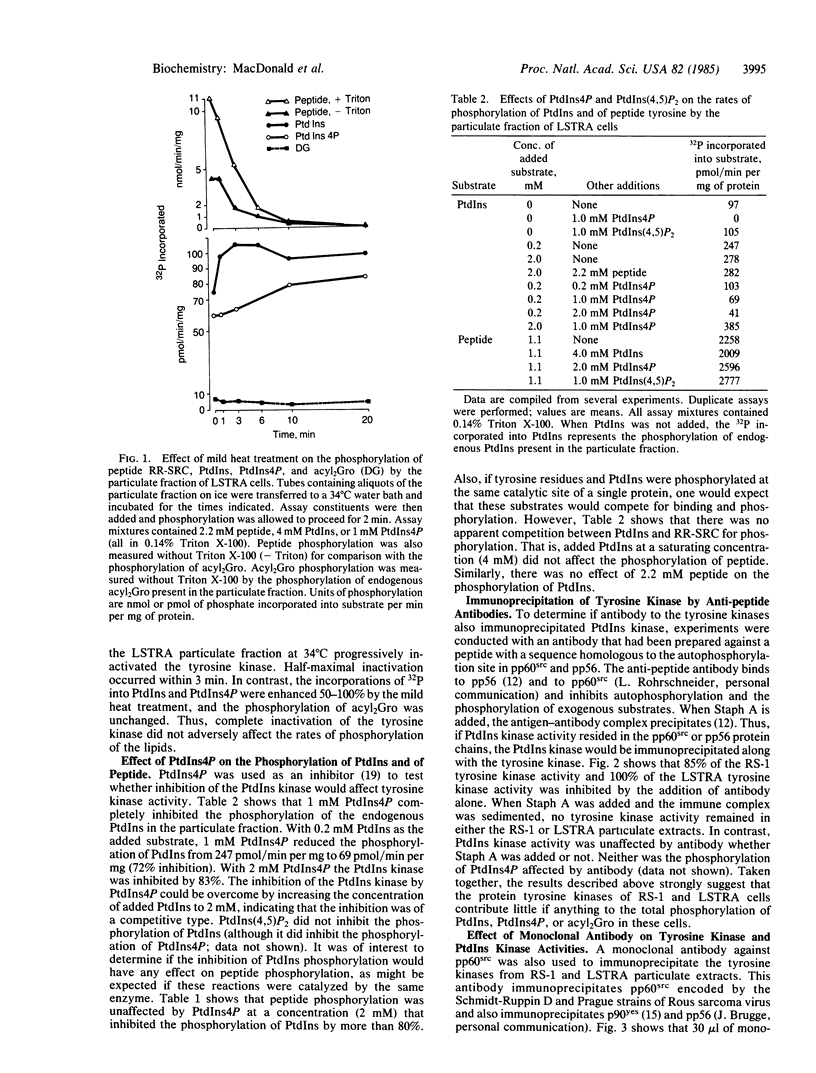
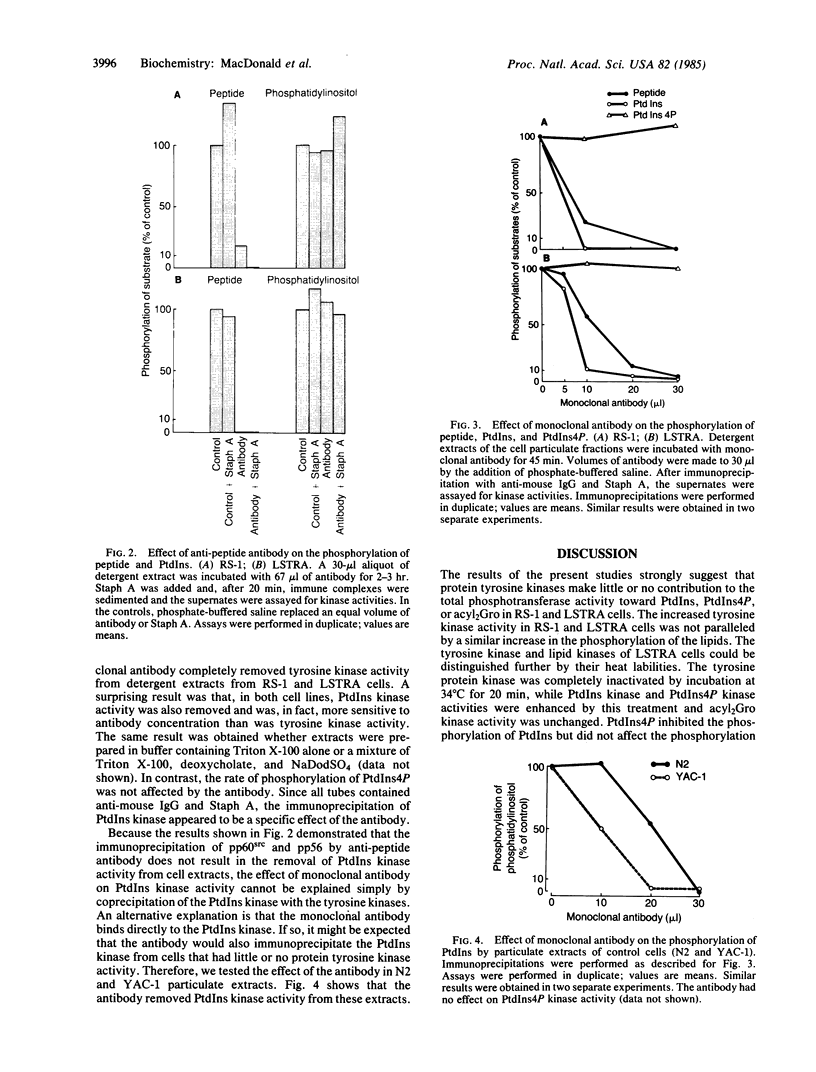
Two transformed rodent cell lines (RS-1 and LSTRA) were studied in vitro to determine if their major protein tyrosine kinases catalyzed the phosphorylation of phosphatidylinositol (PtdIns), phosphatidylinositol 4-phosphate (PtdIns4P), or diacylglycerol. RS-1 cells, transformed by Rous sarcoma virus, contain high levels of pp60src; LSTRA cells, transformed by Moloney murine leukemia virus, contain a tyrosine kinase (pp56) that is the product of an unknown cellular gene. Rates of phosphorylation of peptide tyrosine were elevated more than 20-fold in RS-1 and LSTRA particulate fractions compared to fractions from suitable control cells (N2 and YAC-1), but there was not a proportional increase in rates of phosphorylation of PtdIns, PtdIns4P, or diacylglycerol. Heat (34 degrees C) completely inactivated the LSTRA tyrosine kinase, while it enhanced the phosphorylation of PtdIns and PtdIns4P and had no effect on the phosphorylation of diacylglycerol. PtdIns4P inhibited the phosphorylation of PtdIns but had no effect on tyrosine kinase activity. An antibody, raised against a peptide with a sequence homologous to the autophosphorylation site of pp60src, immunoprecipitated tyrosine kinase activity from RS-1 and LSTRA extracts but had no effect on PtdIns kinase or PtdIns4P kinase activity. These results provide evidence that the phosphorylations of tyrosine and PtdIns are catalyzed by different proteins. An additional observation was that a monoclonal antibody that binds to pp60src and pp56 removed PtdIns kinase as well as tyrosine kinase activity from RS-1 and LSTRA particulate extracts. This antibody also removed PtdIns kinase from N2 and YAC-1 extracts, in which tyrosine kinase activity was low or undetectable. Thus, the anti-pp60src monoclonal antibody may recognize the PtdIns kinase in addition to pp60src and pp56.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berridge M. J., Irvine R. F. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature. 1984 Nov 22;312(5992):315–321. doi: 10.1038/312315a0. [DOI] [PubMed] [Google Scholar]
- Casnellie J. E., Gentry L. E., Rohrschneider L. R., Krebs E. G. Identification of the tyrosine protein kinase from LSTRA cells by use of site-specific antibodies. Proc Natl Acad Sci U S A. 1984 Nov;81(21):6676–6680. doi: 10.1073/pnas.81.21.6676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casnellie J. E., Harrison M. L., Hellstrom K. E., Krebs E. G. A lymphoma cell line expressing elevated levels of tyrosine protein kinase activity. J Biol Chem. 1983 Sep 10;258(17):10738–10742. [PubMed] [Google Scholar]
- Casnellie J. E., Harrison M. L., Hellstrom K. E., Krebs E. G. A lymphoma protein with an in vitro site of tyrosine phosphorylation homologous to that in pp60src. J Biol Chem. 1982 Dec 10;257(23):13877–13879. [PubMed] [Google Scholar]
- Casnellie J. E., Harrison M. L., Pike L. J., Hellström K. E., Krebs E. G. Phosphorylation of synthetic peptides by a tyrosine protein kinase from the particulate fraction of a lymphoma cell line. Proc Natl Acad Sci U S A. 1982 Jan;79(2):282–286. doi: 10.1073/pnas.79.2.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cikes M., Friberg S., Jr, Klein G. Progressive loss of H-2 antigens with concomitant increase of cell-surface antigen(s) determined by Moloney leukemia virus in cultured murine lymphomas. J Natl Cancer Inst. 1973 Feb;50(2):347–362. doi: 10.1093/jnci/50.2.347. [DOI] [PubMed] [Google Scholar]
- GLYNN J. P., BLANCO A. R., GOLDIN A. STUDIES ON INDUCED RESISTANCE AGAINST ISOTRANSPLANTS OF VIRUS-INDUCED LEUKEMIA. Cancer Res. 1964 Apr;24:502–508. [PubMed] [Google Scholar]
- Gallis B., Bornstein P., Brautigan D. L. Tyrosylprotein kinase and phosphatase activities in membrane vesicles from normal and Rous sarcoma virus-transformed rat cells. Proc Natl Acad Sci U S A. 1981 Nov;78(11):6689–6693. doi: 10.1073/pnas.78.11.6689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heldin C. H., Westermark B. Growth factors: mechanism of action and relation to oncogenes. Cell. 1984 May;37(1):9–20. doi: 10.1016/0092-8674(84)90296-4. [DOI] [PubMed] [Google Scholar]
- Hui D. Y., Harmony J. A. Phosphatidylinositol turnover in mitogen-activated lymphocytes. Suppression by low-density lipoproteins. Biochem J. 1980 Oct 15;192(1):91–98. doi: 10.1042/bj1920091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipsich L. A., Lewis A. J., Brugge J. S. Isolation of monoclonal antibodies that recognize the transforming proteins of avian sarcoma viruses. J Virol. 1983 Nov;48(2):352–360. doi: 10.1128/jvi.48.2.352-360.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macara I. G., Marinetti G. V., Balduzzi P. C. Transforming protein of avian sarcoma virus UR2 is associated with phosphatidylinositol kinase activity: possible role in tumorigenesis. Proc Natl Acad Sci U S A. 1984 May;81(9):2728–2732. doi: 10.1073/pnas.81.9.2728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machicao E., Wieland O. H. Evidence that the insulin receptor-associated protein kinase acts as a phosphatidylinositol kinase. FEBS Lett. 1984 Sep 17;175(1):113–116. doi: 10.1016/0014-5793(84)80581-5. [DOI] [PubMed] [Google Scholar]
- Marche P., Koutouzov S., Meyer P. Metabolism of phosphoinositides in the rat erythrocyte membrane. A reappraisal of the effect of magnesium on the 32P incorporation into polyphosphoinositides. Biochim Biophys Acta. 1982 Mar 12;710(3):332–340. doi: 10.1016/0005-2760(82)90116-3. [DOI] [PubMed] [Google Scholar]
- Michell R. H., Harwood J. L., Coleman R., Hawthorne J. N. Characteristics of rat liver phosphatidylinositol kinase and its presence in the plasma membrane. Biochim Biophys Acta. 1967 Dec 5;144(3):649–658. doi: 10.1016/0005-2760(67)90053-7. [DOI] [PubMed] [Google Scholar]
- Sandmeyer S., Smith R., Kiehn D., Bornstein P. Correlation of collagen synthesis and procollagen messenger RNA levels with transformation in rat embryo fibroblasts. Cancer Res. 1981 Mar;41(3):830–838. [PubMed] [Google Scholar]
- Sefton B. M., Hunter T. Tyrosine protein kinases. Adv Cyclic Nucleotide Protein Phosphorylation Res. 1984;18:195–226. [PubMed] [Google Scholar]
- Shinnick T. M., Lerner R. A., Sutcliffe J. G. Nucleotide sequence of Moloney murine leukaemia virus. Nature. 1981 Oct 15;293(5833):543–548. doi: 10.1038/293543a0. [DOI] [PubMed] [Google Scholar]
- Sugimoto Y., Whitman M., Cantley L. C., Erikson R. L. Evidence that the Rous sarcoma virus transforming gene product phosphorylates phosphatidylinositol and diacylglycerol. Proc Natl Acad Sci U S A. 1984 Apr;81(7):2117–2121. doi: 10.1073/pnas.81.7.2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voronova A. F., Buss J. E., Patschinsky T., Hunter T., Sefton B. M. Characterization of the protein apparently responsible for the elevated tyrosine protein kinase activity in LSTRA cells. Mol Cell Biol. 1984 Dec;4(12):2705–2713. doi: 10.1128/mcb.4.12.2705. [DOI] [PMC free article] [PubMed] [Google Scholar]