Abstract
Importance
Regional left ventricular (LV) wall thinning is believed to represent chronic transmural myocardial infarction and scar tissue. However, recent case reports using delayed-enhancement cardiovascular magnetic resonance (CMR) imaging raise the possibility that thinning may occur with little or no scarring.
Objective
To evaluate patients with regional myocardial wall thinning and to determine scar burden and potential for functional improvement.
Design, Setting, and Patients
Investigator-initiated, prospective, 3-center study conducted from August 2000 through January 2008 in 3 parts to determine (1) in patients with known coronary artery disease (CAD) undergoing CMR viability assessment, the prevalence of regional wall thinning (end-diastolic wall thickness ≤5.5 mm), (2) in patients with thinning, the presence and extent of scar burden, and (3) in patients with thinning undergoing coronary revascularization, any changes in myocardial morphology and contractility.
Main Outcomes and Measures
Scar burden in thinned regions assessed using delayed-enhancement CMR and changes in myocardial morphology and function assessed using cine-CMR after revascularization.
Results
Of 1055 consecutive patients with CAD screened, 201 (19% [95% CI, 17% to 21%]) had regional wall thinning. Wall thinning spanned a mean of 34% (95% CI, 32% to 37% [SD, 15%]) of LV surface area. Within these regions, the extent of scarring was 72% (95% CI, 69% to 76% [SD, 25%]); however, 18% (95% CI, 13% to 24%) of thinned regions had limited scar burden (≤50% of total extent). Among patients with thinning undergoing revascularization and follow-up cine-CMR (n=42), scar extent within the thinned region was inversely related to regional (r=−0.72, P<.001) and global (r=−0.53, P<.001) contractile improvement. End-diastolic wall thickness in thinned regions with limited scar burden increased from 4.4 mm (95% CI, 4.1 to 4.7) to 7.5 mm (95% CI, 6.9 to 8.1) after revascularization (P<.001), resulting in resolution of wall thinning. On multivariable analysis, scar extent had the strongest association with contractile improvement (slope coefficient, −0.03 [95% CI, −0.04 to −0.02]; P<.001) and reversal of thinning (slope coefficient, −0.05 [95% CI, −0.06 to −0.04]; P<.001).
Conclusions and Relevance
Among patients with CAD referred for CMR and found to have regional wall thinning, limited scar burden was present in 18% and was associated with improved contractility and resolution of wall thinning after revascularization. These findings, which are not consistent with common assumptions, warrant further investigation.
REGIONAL MYOCARDIAL WALL thinning is believed to represent chronic myocardial infarction. A recent task force report on the definition of myocardial infarction by the European Society of Cardiology, American College of Cardiology, American Heart Association, and the World Heart Federation concludes that “[i]maging evidence of a region … that is thinned and fails to contract, in the absence of a non-ischaemic cause” is diagnostic for chronic myocardial infarction.1 Accordingly, many experts state that viability testing is unnecessary for regions with wall thinning, because this is synonymous with scar tissue.2-6
However, recent case reports incorporating the use of delayed-enhancement cardiovascular magnetic resonance (CMR) imaging raise the possibility that this viewpoint is incorrect.7,8 These single-patient reports indicate that myocardial regions with severe wall thinning do not necessarily consist entirely of scar tissue but instead may have minimal or no scarring. Thus, some areas of myocardial thinning may represent viable myocardium and have the potential for recovery of function.
In the current 3-center study, we systematically evaluated several aspects of myocardial thinning. First, in consecutive patients with known coronary artery disease (CAD) undergoing delayed-enhancement CMR viability assessment, we examined the prevalence of regional myocardial thinning. Second, we investigated how often limited scarring can occur in the setting of myocardial thinning. Third, in a subset of patients undergoing coronary revascularization specifically of the thinned region, we assessed whether limited scarring in the thinned region would be associated with functional improvement after revascularization.
METHODS
Patients and Protocol
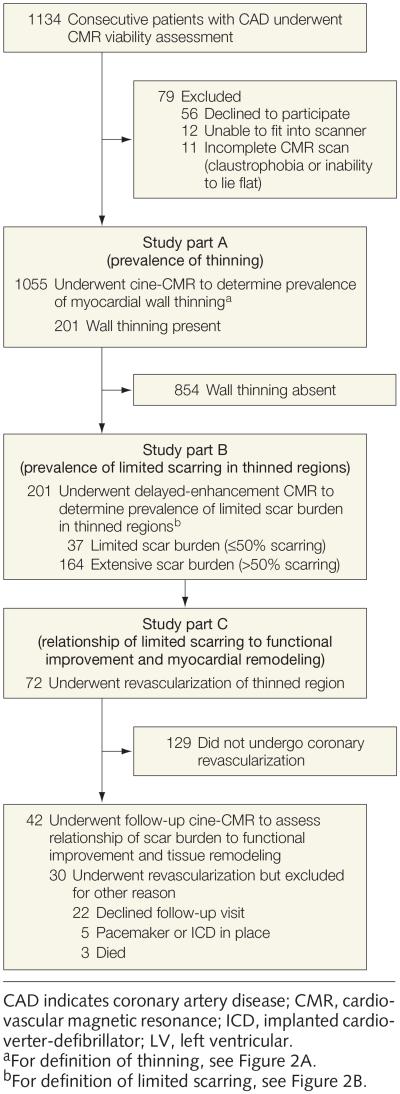
This prospective study, conducted from August 2000 through January 2008, consisted of 3 parts (FIGURE 1). Patients were enrolled at 3 sites: Duke Cardiovascular Magnetic Resonance Center, Durham, North Carolina; The Heart Group PLLC, Nashville, Tennessee; and Northwestern University, Chicago, Illinois. All patients provided written informed consent for this protocol, which was approved by the institutional review board at each site.
Figure 1.
Study Flow
Part A: Prevalence of Thinning
The purpose of part A was to evaluate the prevalence of myocardial thinning. Consecutive patients with CAD who were scheduled for CMR viability assessment were screened and asked to participate unless they (1) were younger than 18 years, (2) had experienced an acute myocardial infarction (MI) within last 2 weeks, (3) had nonischemic myocardial disease (eg, hypertrophic cardiomyopathy, myocarditis), or (4) were unable to provide consent. A total of 1134 patients were approached, of whom 56 (5%) declined to participate, 12 (1%) were unable to fit into the scanner, and 11 (1%) did not complete CMR assessment for various reasons (eg, claustrophobia, inability to lie flat). One thousand fifty-five patients completed CMR and were included in the analysis. Similar to prior publications, myocardial thinning was defined as an end-diastolic wall thickness (EDWT) of 5.5 mm or less on cine-CMR.4
Part B: Scar Burden in Thinned Regions
The purpose of part B was to evaluate the prevalence of limited scar burden (defined as ≤50% of total extent) in thinned myocardium. This pre-specified cutoff was chosen based on prior delayed-enhancement CMR studies demonstrating that regions with scar burden greater than 50% have low likelihood of functional recovery after revascularization.9,10 However, data were also evaluated using scar extent as a continuous variable.
Part C: Relationship of Scar Burden to Functional Improvement and Tissue Remodeling
The objective of part C was to determine the relationship of scar burden to functional improvement and myocardial tissue remodeling in the thinned region following coronary revascularization. The decision to undergo revascularization was guided by all relevant clinical and imaging data and made at the discretion of the treating physician and the patient.
Cardiovascular Magnetic Resonance
Image Acquisition
Images were acquired on 1.5T scanners (Siemens Sonata/Avanto) as described previously.9 Briefly, cine-CMR images were acquired using a steady-state free-precession sequence. Short-axis views were obtained every 1 cm to cover the entire left ventricle (6 mm thickness, 4 mm gap). Gadolinium contrast (gadopentetate dimeglumine, gadoteridol, or gadoversetamide) was administered intravenously (0.15 mmoL/kg), and delayed-enhancment CMR images were acquired 10 to 15 minutes later using a segmented inversion-recovery sequence in the same views used for cine-CMR.
Image Analysis
Cardiovascular magnetic resonance images were evaluated masked to patient identity and clinical information. For patients who underwent revascularization and returned for follow-up cine-CMR, the follow-up scan was analyzed by an observer blinded to all delayed-enhancement data and different from the individual who analyzed the baseline images. Quantitative measurements were made by manual planimetry.
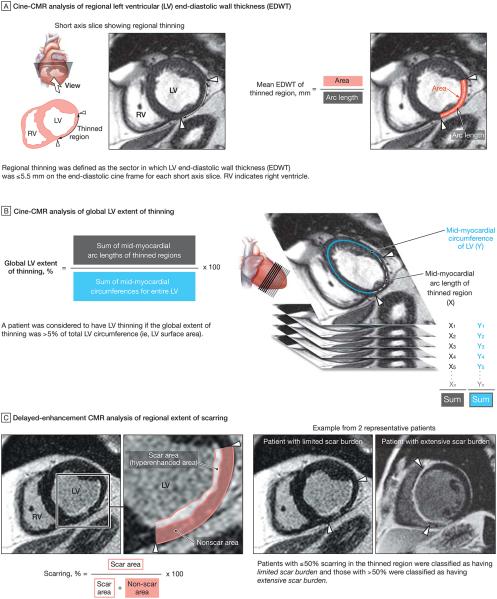
Determination of Wall Thinning
Regional thinning was defined quantitatively as the sector in which EDWT was 5.5 mm or less on the end-diastolic cine frame for each short-axis slice (FIGURE 2A). The global left ventricular (LV) extent of thinning was calculated by summing the mid-myocardial circumference of the thinned region and dividing by the mid-myocardial circumference of the entire left ventricle (summation of all short-axis cine slices), multiplied by 100 (Figure 2B). On a per-patient basis, thinning was deemed present if the global extent of thinning was more than 5% of total LV circumference (ie, LV surface area).
Figure 2.
Cardiovascular Magnetic Resonance (CMR) Analyses
Scar Extent in Thinned Regions
Cine images were used to determine the representative view that went through the central core of the thinned region. Quantitative analysis was then performed on the corresponding delayed-enhancement CMR slice after 4-fold interpolation. The extent of scarring (hyperenhanced region) in the thinned region was expressed as a percentage of the total area of thinned myocardium (Figure 2C). Patients with 50% or less scarring in the thinned region were classified as having limited scar burden and those with more than 50% scarring as having extensive scar burden.
LV Volumes, Ejection Fraction, and Mass
Endocardial borders on the stack of short-axis cine images in end diastole and end systole, and the epicardial borders in end diastole, were traced using planimetry. Left ventricular ejection fraction (LVEF) was calculated by subtracting end-systolic volume from end-diastolic volume and dividing by end-diastolic volume. Left ventricular mass was calculated by subtracting endocardial from epicardial volumes and multiplying by myocardial density (1.05 g/cm3).11
Regional Functional Improvement and Myocardial Tissue Remodeling
For patients who returned for follow-up CMR after revascularization, analyses of end-diastolic and end-systolic wall thickness were performed in the same manner as for baseline measurements. The thinned region identified on the prerevascularization images was registered to the same region on the postrevascularization images using conventional landmarks (right ventricular insertion sites and papillary muscles).
Intraobserver and Interobserver Agreement
Images in a random sample of 10% of the population in study part B (21 patients) were reanalyzed 6 months later to determine intraobserver and interobserver agreement for measurements of EDWT, systolic wall thickening, and transmural extent of scarring. Intraobserver and interobserver agreement values were similar, with a bias of less than 0.2 mm for EDWT and systolic wall thickening and less than 1% for extent of scarring within the thinned region, with an SD of the difference of less than 0.3 mm for EDWT and systolic wall thickening and less than 4.7% for extent of scarring.
Coronary Angiography
Angiograms were reviewed in conjunction with the cine-CMR images to identify the coronary artery supplying the thinned region. The maximum severity of stenosis was analyzed using standard quantitative coronary angiography methods.12 The extent of collateral flow was determined using the Rentrop classification scheme (0, no collateral flow; 1, collateral flow leading to side branches; 2, collateral flow leading to side branches and a portion of the epicardial segment; 3, collateral flow leading to side branches and the entire epicardial segment).13
Electrocardiography
The Minnesota Code, a standardized classification system for electrocardiograms, was used to determine the presence of prior Q-wave myocardial infarction (codes 1-1-1 to 1-2-7) based on Q-wave location, amplitude, and duration.14,15 Infarct size as determined by electrocardiogram was estimated using the 54-criteria, 32-point Selvester QRS scoring system.16
Statistical Analysis
Continuous data are presented as mean (SD). Two-sample t tests were used to compare continuous data between 2 groups. Comparisons between discrete data were made using χ2 tests; the Fisher exact test was used when cell count was less than 5. We used linear regression analyses to examine the relationships between scar burden and functional and morphological parameters at baseline and changes in these parameters with revascularization. Paired t tests were used to compare regional systolic wall thickening and EDWT before and after revascularization in subgroups with limited scarring and with extensive scarring. Multivariable logistic or linear regression analyses (as appropriate) were performed to identify clinical and imaging characteristics associated with limited scarring, functional improvement, and tissue remodeling. Variables with P<.05 on bivariable analysis were considered candidate variables in the multivariable models unless there were fewer than 3 total candidate variables, in which case the 3 most significant variables were included.
All statistical tests were 2-tailed; P<.05 was considered statistically significant. SAS version 9.2 (SAS Institute Inc) was used to perform the statistical analyses.
RESULTS
Part A: Prevalence of Thinning
Among the 1055 patients screened and who provided consent, 201 (19% [95% CI, 17% to 21%]) had myocardial wall thinning. The mean circumferential extent of thinning was substantial, encompassing 34% (95% CI, 32% to 37% [SD, 15%]) of total LV surface area. Baseline characteristics are shown in the TABLE. Patients with thinning were predominantly men (79%), had CAD risk factors (mean, 2.3 [SD, 1.0]) and significant LV dysfunction (LVEF, 32.6% [95% CI, 31.0% to 34.2%]; SD, 11.5%), and most had history of MI (71%) and electrocardiographic Q waves (67%).
Table.
Baseline Characteristics of Patients With Wall Thinning
| Characteristic | No. (%) | P Value | ||
|---|---|---|---|---|
| All (n = 201) |
Scar Burden | |||
| Extensive (>50%) (n = 164) |
Limited (≤50%) (n = 37)a |
|||
| Age, mean (SD), y | 64.3 (11.9) | 63.7 (11.9) | 67.2 (11.5) | .11 |
| Men | 158 (79) | 130 (79) | 28 (76) | .63 |
| Cardiac risk factors Hypercholesterolemia |
163 (81) | 136 (83) | 27 (73) | .16 |
| Hypertension | 128 (64) | 102 (62) | 26 (70) | .36 |
| Cigarette smoking | 62 (31) | 47 (29) | 15 (41) | .16 |
| Family history of premature CAD | 62 (31) | 48 (29) | 14 (38) | .31 |
| Diabetes | 57 (28) | 47 (29) | 10 (27) | .84 |
| Risk factors present, mean (SD) | 2.3 (1.0) | 2.3 (1.0) | 2.5 (1.0) | .37 |
| Angina | 105 (52) | 85 (52) | 20 (54) | .71 |
| NYHA functional status (classification score), mean (SD) | 2.2 (1.1) | 2.2 (0.9) | 2.3 (0.9) | .79 |
| I | 53 (26) | 44 (27) | 9 (24) | |
| II | 58 (29) | 46 (28) | 12 (32) | |
| III | 80 (40) | 67 (41) | 13 (35) | |
| IV | 10 (5) | 7 (4) | 3 (8) | |
| Cardiac history Prior myocardial infarction |
142 (71) | 119 (73) | 23 (62) | .21 |
| Prior revascularization | 98 (51) | 82 (52) | 16 (46) | .53 |
| Cardiac medications β-Blockers |
144 (72) | 120 (73) | 24 (65) | .31 |
| ACE inhibitors and/or angiotensin receptor blockers | 133 (66) | 110 (67) | 23 (62) | .57 |
| HMG reductase inhibitors | 125 (62) | 105 (64) | 20 (54) | .26 |
| Aspirin | 152 (76) | 126 (77) | 26 (70) | .40 |
| Thienopyridines | 22 (11) | 20 (12) | 2 (5) | .23 |
| Electrocardiographyb Any Q waves |
131 (67) | 108 (68) | 23 (62) | .47 |
| Q waves corresponding to thinned region | 112 (57) | 94 (59) | 18 (49) | .23 |
| Selvester score, mean (SD)c | 6.6 (3.7) | 6.9 (3.6) | 5.2 (3.9) | .009 |
| Coronary angiography, mean (SD)d No. of vessels with CAD (stenosis ≥70%) |
2.03 (0.90) | 2.02 (0.93) | 2.08 (0.80) | .71 |
| Maximum stenosis in artery to thinned region | 84 (30) | 81 (320 | 95 (17) | <.001 |
| Rentrop collateral score to thinned region 0 |
123 (62) | 108 (66) | 15 (41) | .009 |
| 1 | 15 (8) | 11 (7) | 4 (11) | |
| 2 | 25 (13) | 17 (10) | 8 (22) | |
| 3 | 37 (19) | 27 (17) | 10 (27) | |
| Cardiovascular magnetic resonance, mean (SD) Global LV ejection fraction, % |
32.6 (11.5) | 32.1 (11.0) | 34.7 (13.4) | .22 |
| LV surface area thinned, % | 34.2 (15.3) | 34.9 (14.9) | 31.1 (17.0) | .17 |
| Thinned region, mm End-diastolic wall thickness |
4.2 (0.8) | 4.1 (0.9) | 4.5 (0.7) | .02 |
| End-systolic wall thickness | 4.1 (0.9) | 4.1 (0.9) | 4.2 (0.7) | .36 |
| Systolic wall thickening | −0.1 (0.4) | −0.0 (0.3) | −0.2 (0.7) | .06 |
Abbreviations: ACE, angiotensin-converting enzyme; CAD, coronary artery disease; HMG, 3-hydroxy-3-methyl-glutaryl; LV, left ventricular; New York Heart Assocation.
Based on 50% or less scarring in the thinned region.
Available in 195 patients.
Infarct size as determined by electrocardiogram was estimated using the 32-point Selvester QRS scoring system, in which each point is designed to represent approximately 3% of LV mass.
Available in 199 patients.
Part B: Scar Burden in Thinned Regions
Of the 201 patients with wall thinning, 198 had scarring within the thinned region. The mean extent of scarring was 72% (95% CI, 69% to 76% [SD, 25%]); however, 18% (95% CI, 13% to 24%) of patients had scarring involving less than 50% of the thinned region (FIGURE 3A). Although there was an association between wall thickness and scar extent, the relationship was poor, because the extent of scarring was highly variable for each level of wall thickness (r=−0.22, P=.001) (Figure 3B).
Figure 3.
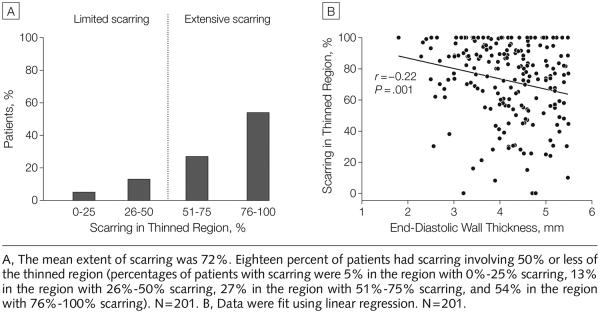
Correlation Between End-Diastolic Wall Thickness and Scar Burden Within the Thinned Region
Also shown in the Table are the characteristics of patients with 50% or less scarring in the thinned region (limited scarring), compared with those with more than 50% (extensive scarring). Characteristics were similar between groups, including LVEF, the extent of LV surface area that was thinned, and the fraction with Q waves. The only characteristics that were different were lower Selvester score (mean, 5.2 [95% CI, 3.0 to 6.5] for those with ≤50% scarring vs 6.9 [95% CI, 6.4 to 7.5] for those with >50% scarring, P=.009), greater severity of stenosis in the coronary vessel supplying the thinned region (95% [95% CI, 89% to 100%] vs 81% [95% CI, 76% to 86%], P<.001), higher Rentrop collateral score (P=.009), and slightly higher EDWT (4.5 [95% CI, 4.2 to 4.7] mm vs 4.1 [95% CI, 4.0 to 4.2] mm, P=.02) in patients with limited scarring.
Multivariable logistic regression analysis (eTable 1) demonstrated that significant associations with limited scar burden were Selvester score (odds ratio [OR], 0.87 [95% CI, 0.78 to 0.97]; P=.01), degree of coronary stenosis supplying the thinned region (OR, 1.03 [95% CI, 1.00 to 1.06]; P=.03), and EDWT (OR, 1.86 [95% CI, 1.12 to 3.12]; P=.02).
Part C: Relationship of Scar Burden to Functional Improvement and Tissue Remodeling
Seventy-two patients underwent revascularization, which included the coronary artery supplying the thinned region. Of these, 42 (35 surgical, 7 percutaneous) returned for follow-up cine-CMR after revascularization (mean, 116 [SD, 73] days). No patient experienced a cardiac event between the initial and follow-up CMR. Of the 30 patients in whom follow-up CMR was unavailable, 3 died; 5 underwent placement of a pacemaker or implantable cardioverter-defibrillator device; and 22 declined to return. Patients with follow-up CMR were similar to those without follow-up for all baseline characteristics except frequency of diabetes (19% vs 43%, respectively; P=.03).
Functional Improvement
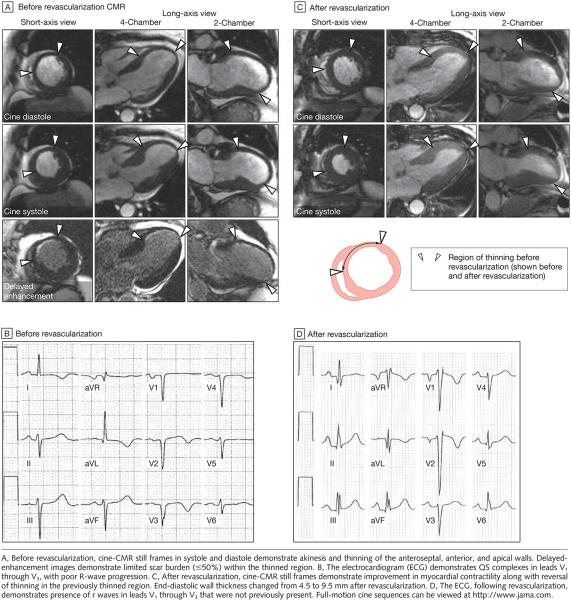
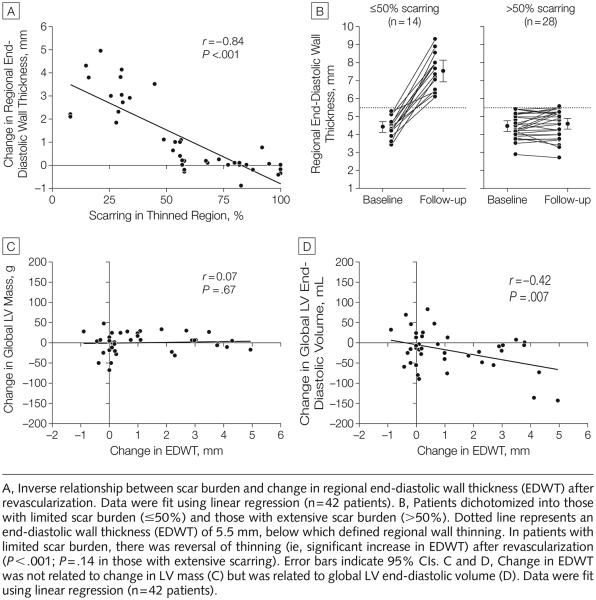
Within the thinned region, there was an inverse relationship between the extent of scarring and contractile improvement after revascularization (r = −0.72, P<.001) (FIGURE 4A). After dichotomizing patients into those with limited scar burden (≤50%) and those with extensive scar burden (>50%), only the group with limited scar burden demonstrated contractile improvement in the thinned region, with a mean increase of 2.3 mm (SD, 1.1 mm; range, 1.0-4.2 mm) (P<.001) in absolute systolic wall thickening (Figure 4B). Also observed was an inverse relationship between the extent of scarring in the thinned region and improvement in global LVEF after revascularization (r=−0.53, P<.001) (Figure 4C). FIGURE 6 demonstrates typical images in a patient with limited scarring in the thinned region who experienced significant improvement in both regional and global function after revascularization.
Figure 4.
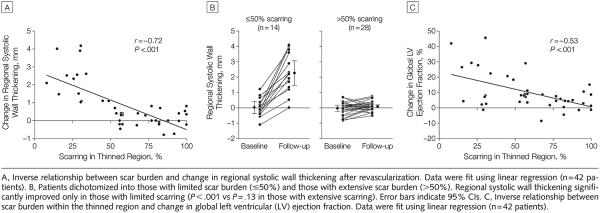
Relationship of Scarring to Functional Improvement
Figure 6.
Cardiovascular Magnetic Resonance (CMR) Imaging and Electrocardiographic Changes in an Example Patient with Wall Thinning and Limited Scar Burden
Myocardial Remodeling
As shown in FIGURE 5A, there was an inverse relationship between the extent of scarring in the thinned region and increase in EDWT after revascularization (r=−0.84, P<.001). Before revascularization, wall thinning was similar in patients with limited and extensive scarring (4.4 mm [95% CI, 4.1 to 4.7] vs 4.5 mm [95% CI, 4.2 to 4.7], P=.80). After revascularization, the group with limited scarring uniformly demonstrated an increase in EDWT, with a mean change of 3.1 mm (SD, 1.0 mm; range, 1.1-4.9 mm) (P<.001), whereas patients with extensive scarring demonstrated no change in EDWT (Figure 5B).
Figure 5.
Relationship of Scarring to Myocardial Remodeling
To ascertain if the increase in EDWT was attributable to active myocardial growth or simply a passive change in LV geometry, we related change in EDWT with change in global LV mass and change in LV end-diastolic volume. Figure 5C demonstrates that after revascularization there was no relationship between change in EDWT and change in LV mass (r=0.07, P=.67). Conversely, an increase in EDWT after revascularization was associated with a reduction in LV end-diastolic volume (r=−0.42, P=.007) (Figure 5D).
The patient example in Figure 6 demonstrates that limited scarring in the thinned region was associated with disappearance of wall thinning after revascularization. There was also disappearance of Q waves after revascularization. Twenty-two patients had Q waves corresponding to the thinned region before revascularization; among these, the subgroup with reversal of wall thinning after revascularization was more likely to have disappearance of Q waves compared with the subgroup without reversal of wall thinning (4/5 [80%] vs 0/17 [0%]; P<.001).
Bivariable and Multivariable Analysis
eTable 2 reports the results of bivariable and multivariable regression analysis regarding associations with functional and remodeling changes after revascularization. On multivariable analysis, scar extent had the strongest association with regional contractile improvement (slope coefficient, −0.03 [95% CI, −0.04 to −0.02]; P<.001), global contractile improvement (slope coefficient, −0.21 [95% CI, −0.32 to −0.11]; P<.001), and reversal of thinning (slope coefficient, −0.05 [95% CI, −0.06 to −0.04]; P<.001). The adjusted-model R2 values representing the proportion of variance explained for the linear regression models of contractile improvement and remodeling are reported in eTable 2.
COMMENT
The pathophysiology of regional wall thinning following transmural MI has undergone considerable investigation. Animal models have demonstrated that slippage of myocyte bundles plays a fundamental role in infarct expansion, remodeling, and ultimately wall thinning.17,18 Resorption during infarct healing, with removal of necrotic debris and resolution of tissue edema, may also play a role.19 With either mechanism, only complete rather than partial infarction of the myocardium at risk is postulated to result in substantial wall thinning.20-23 These experimental studies form the physiological basis for the view that regional wall thinning is indicative of transmural MI and absent residual viability.17,20-24 This concept has been perpetuated, despite a paucity of experimental data concerning the effects of chronic ischemia without infarction (ie, hibernating myocardium) on diastolic wall thickness.
Studies in patients have reinforced the viewpoint that regional wall thinning is indicative of transmural MI and have focused on the presence of focal thinning observable on conventional cardiac imaging as a straightforward, accurate marker of nonviable scar tissue.3-6 Baer et al4 and Schmidt et al6 (using cine-MRI) and Cwajg et al3 and La Canna et al5 (using echocardiography) concluded that a simple measurement of EDWT (<5-6 mm) indicates irreversible myocardial damage and thus obviates the need for viability testing. These investigations were small (43, 40, 45, and 28 patients, respectively), and only a fraction of patients or segments in each cohort had wall thinning. Nonetheless, these reports have become the basis for proposed clinical algorithms in which an early finding of wall thinning (<5-6 mm) precludes further viability testing or coronary revascularization, because thinned regions are assumed to represent permanent scar tissue without residual viability.2
To our knowledge, the current study is the first to systematically investigate a cohort of patients with regional wall thinning. Of 201 patients identified by CMR as having wall thinning, most had significant LV dysfunction (LVEF, 32.6% [SD, 11.5%]), multivessel CAD, and thinning of a substantial portion of the left ventricle (34.2% [SD, 15.3%] of LV surface area). Among this cohort, 18% (95% CI, 13% to 24%) of thinned regions had limited or no scarring observed using delayed-enhancement CMR. Because the lack of scarring was associated with significant contractile improvement and reverse remodeling with resolution of wall thinning following revascularization, we believe the data indicate that myocardial thinning is potentially reversible and therefore should not be considered a permanent state.
Our results suggest that common clinical characteristics will not be useful in predicting whether thinned regions have limited scar tissue. There were no differences in age, sex, cardiac risk factors, angina or heart failure symptoms, or presence of Q waves between patients with extensive or limited scar burden. Although patients with extensive scarring were less likely to have collateral flow observable on coronary angiography, it is notable that 41% of those with limited scarring had no detectable angiographic collateral flow. The findings suggest that these clinical characteristics should not be used to assess viability in a region of thinning.
As a group, patients with limited scarring had a very high degree of stenosis in the coronary artery perfusing the thinned region (mean stenosis, 95%). This suggests that lesser grades of stenosis may be sufficient to result in resting dysfunction without infarction but are insufficient to result in the unique condition of resting dysfunction and thinning without infarction. Although speculative, this suggests that thinning of viable regions requires a significant reduction in resting blood flow and represents an extreme form of hibernating myocardium with a delicate balance between resting ischemia and infarction. In this situation, one might expect reduced activity on myocardial perfusion imaging and an exhausted coronary flow reserve with an inability to respond to inotropic stimulation, despite limited or no scar tissue within the thinned region.
Independent of functional changes, it has been suggested that preservation of LV geometry may be an important end point after revascularization, because ongoing LV remodeling is a major determinant of poor prognosis.25-27 Indeed, a few studies have reported that “reverse” remodeling may occur following coronary revascularization, particularly in patients with substantial myocardial viability.26,27 The results of the current study extend previous observations and document a greater range in the progression and regression of LV remodeling as well as a different type of remodeling process. Whereas prior reports focused on global changes in the LV cavity—measures of volumes, sphericity, or both—we have shown that the myocardial wall may thin and revert back to full thickness as long as limited scarring is present. These results indicate that the end stage of remodeling is better determined by tissue composition (ie, scarring) rather than any set level of morphological changes to the LV cavity or LV wall.
A possible mechanism for the disappearance of wall thinning is suggested by the documentation that reverse remodeling following revascularization was not accompanied by any change in total LV myocardial mass (Figure 5C). We hypothesize that slippage of myocyte bundles,17 the primary mechanism of myocardial infarct expansion, may also occur in the setting of chronic ischemia without infarction and that this process is reversible following coronary revascularization. This hypothesis requires further investigation.
Our study has a number of limitations. First, CMR was performed in patients in whom revascularization was being considered rather than in a randomized fashion. Hence, the results may be subject to selection bias, and the prevalence of wall thinning associated with limited scarring reported in our study population may be different from that found more generally in patients with stable chronic CAD or in those in whom revascularization is not indicated.
Second, follow-up CMR was performed in a limited number of patients and in none who did not undergo revascularization. Thus, this is another potential source of selection bias, and there remains some uncertainty regarding the conditions and the degree to which thinned regions can recover function. For instance, although we have previously shown that medical therapy can favorably influence LVEF and cavity dimensions in patients with chronic heart failure,28 it remains unknown if optimal medical therapy can result in disappearance of wall thinning.
Third, outcome data on our patient population are not available. Accordingly, we are unable to comment on whether the disappearance of wall thinning in association with recovery of contractility leads to an improvement in prognosis.
Nevertheless, despite these limitations, we believe our study provides new insights into the pathophysiology of thinned myocardium and more broadly the process of reversible ischemic injury. The data show that thinned myocardium may consist of limited scar tissue and can recover function—concepts that are both inconsistent with current views. Moreover, we have observed that limited scar burden identified by delayed-enhancement CMR prior to revascularization is strongly associated with contractile improvement and reversal of wall thinning after revascularization. This is relevant because the viability substudy of the STICH (Surgical Treatment in Ischemic Cardiomyopathy) trial recently reported that conventional viability testing with single-photon emission computed tomography or dobutamine echocardiography cannot help decide who should undergo revascularization or even identify who has improved survival (after adjusting for baseline variables and risk factors).29 In concordance with the STICH substudy, our findings highlight that the pathophysiology of hibernating myocardium is still incompletely understood and that there is much to improve regarding the assessment of viability. The findings provide rationale for future experimental studies on reversible ischemic injury as well as for clinical studies prospectively testing whether CMR guidance for coronary revascularization decisions can improve patient outcome.
Supplementary Material
Acknowledgments
Funding/Support: Ms Parker and Drs Judd and R. J. Kim are supported by National Institutes of Health grant NIH-NHLBI R01-HL64726.
Role of the Sponsor: The National Institutes of Health had no role in the design and conduct of the study; the collection, management, analysis, and interpretation of the data; or the preparation, review, or approval of the manuscript.
Footnotes
Conflict of Interest Disclosures: All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Drs R. J. Kim and Judd reported that they are inventors named on a US patent, owned by Northwestern University, for delayed-enhancement cardiovascular magnetic resonance imaging. Dr Shah reported serving as a consultant to Astellas Pharmaceuticals; receiving grants pending from Siemens Medical Solutions, Astellas Pharmaceuticals, and Methodist Hospital Research Institute; and receiving payment for lectures from AstraZeneca, Lantheus Medical Imaging, and Takeda. No other authors reported disclosures.
Online-Only Material: eTables 1 and 2 and cine sequences are available at http://www.jama.com.
REFERENCES
- 1.Thygesen K, Alpert JS, Jaffe AS, et al. Joint ESC/ACCF/AHA/WHF Task Force for the Universal Definition of Myocardial Infarction. Third universal definition of myocardial infarction. Circulation. 2012;126(16):2020–2035. doi: 10.1161/CIR.0b013e31826e1058. [DOI] [PubMed] [Google Scholar]
- 2.Schinkel AF, Bax JJ, Delgado V, Poldermans D, Rahimtoola SH. Clinical relevance of hibernating myocardium in ischemic left ventricular dysfunction. Am J Med. 2010;123(11):978–986. doi: 10.1016/j.amjmed.2010.03.025. [DOI] [PubMed] [Google Scholar]
- 3.Cwajg JM, Cwajg E, Nagueh SF, et al. Enddiastolic wall thickness as a predictor of recovery of function in myocardial hibernation: relation to rest-redistribution T1-201 tomography and dobutamine stress echocardiography. J Am Coll Cardiol. 2000;35(5):1152–1161. doi: 10.1016/s0735-1097(00)00525-8. [DOI] [PubMed] [Google Scholar]
- 4.Baer FM, Theissen P, Schneider CA, et al. Dobutamine magnetic resonance imaging predicts contractile recovery of chronically dysfunctional myocardium after successful revascularization. J Am Coll Cardiol. 1998;31(5):1040–1048. doi: 10.1016/s0735-1097(98)00032-1. [DOI] [PubMed] [Google Scholar]
- 5.La Canna G, Rahimtoola SH, Visioli O, et al. Sensitivity, specificity, and predictive accuracies of noninvasive tests, singly and in combination, for diagnosis of hibernating myocardium. Eur Heart J. 2000;21(16):1358–1367. doi: 10.1053/euhj.1999.2038. [DOI] [PubMed] [Google Scholar]
- 6.Schmidt M, Voth E, Schneider CA, et al. F-18-FDG uptake is a reliable predictory of functional recovery of akinetic but viable infarct regions as defined by magnetic resonance imaging before and after revascularization. Magn Reson Imaging. 2004;22(2):229–236. doi: 10.1016/j.mri.2003.07.006. [DOI] [PubMed] [Google Scholar]
- 7.Kim RJ, Shah DJ. Fundamental concepts in myocardial viability assessment revisited: when knowing how much is “alive” is not enough. Heart. 2004;90(2):137–140. doi: 10.1136/hrt.2003.023101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.John AS, Dreyfus GD, Pennell DJ. Images in cardiovascular medicine: reversible wall thinning in hibernation predicted by cardiovascular magnetic resonance. Circulation. 2005;111(3):e24–e25. doi: 10.1161/01.CIR.0000153385.62524.85. [DOI] [PubMed] [Google Scholar]
- 9.Kim RJ, Wu E, Rafael A, et al. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. N Engl J Med. 2000;343(20):1445–1453. doi: 10.1056/NEJM200011163432003. [DOI] [PubMed] [Google Scholar]
- 10.Schvartzman PR, Srichai MB, Grimm RA, et al. Non-stress delayed-enhancement magnetic resonance imaging of the myocardium predicts improvement of function after revascularization for chronic ischemic heart disease with left ventricular dysfunction. Am Heart J. 2003;146(3):535–541. doi: 10.1016/S0002-8703(03)00318-1. [DOI] [PubMed] [Google Scholar]
- 11.Myerson SG, Bellenger NG, Pennell DJ. Assessment of left ventricular mass by cardiovascular magnetic resonance. Hypertension. 2002;39(3):750–755. doi: 10.1161/hy0302.104674. [DOI] [PubMed] [Google Scholar]
- 12.Keane D, Haase J, Slager CJ, et al. Comparative validation of quantitative coronary angiography systems: results and implications from a multicenter study using a standardized approach. Circulation. 1995;91(8):2174–2183. doi: 10.1161/01.cir.91.8.2174. [DOI] [PubMed] [Google Scholar]
- 13.Rentrop KP, Cohen M, Blanke H, Phillips RA. Changes in collateral channel filling immediately after controlled coronary artery occlusion by an angioplasty balloon in human subjects. J Am Coll Cardiol. 1985;5(3):587–592. doi: 10.1016/s0735-1097(85)80380-6. [DOI] [PubMed] [Google Scholar]
- 14.Grimm RH, Jr, Tillinghast S, Daniels K, et al. Unrecognized myocardial infarction: experience in the Multiple Risk Factor Intervention Trial (MRFIT) Circulation. 1987;75(3, pt 2):II6–II8. [PubMed] [Google Scholar]
- 15.Sheifer SE, Gersh BJ, Yanez ND, III, Ades PA, Burke GL, Manolio TA. Prevalence, predisposing factors, and prognosis of clinically unrecognized myocardial infarction in the elderly. J Am Coll Cardiol. 2000;35(1):119–126. doi: 10.1016/s0735-1097(99)00524-0. [DOI] [PubMed] [Google Scholar]
- 16.Selvester RH, Wagner GS, Hindman NB. The Selvester QRS scoring system for estimating myocardial infarct size: the development and application of the system. Arch Intern Med. 1985;145(10):1877–1881. [PubMed] [Google Scholar]
- 17.Weisman HF, Bush DE, Mannisi JA, Weisfeldt ML, Healy B. Cellular mechanisms of myocardial infarct expansion. Circulation. 1988;78(1):186–201. doi: 10.1161/01.cir.78.1.186. [DOI] [PubMed] [Google Scholar]
- 18.Pfeffer MA, Braunwald E. Ventricular remodeling after myocardial infarction: experimental observations and clinical implications. Circulation. 1990;81(4):1161–1172. doi: 10.1161/01.cir.81.4.1161. [DOI] [PubMed] [Google Scholar]
- 19.Fieno DS, Hillenbrand HB, Rehwald WG, et al. Infarct resorption, compensatory hypertrophy, and differing patterns of ventricular remodeling following myocardial infarctions of varying size. J Am Coll Cardiol. 2004;43(11):2124–2131. doi: 10.1016/j.jacc.2004.01.043. [DOI] [PubMed] [Google Scholar]
- 20.Hutchins GM, Bulkley BH. Infarct expansion versus extension: two different complications of acute myocardial infarction. Am J Cardiol. 1978;41(7):1127–1132. doi: 10.1016/0002-9149(78)90869-x. [DOI] [PubMed] [Google Scholar]
- 21.Eaton LW, Bulkley BH. Expansion of acute myocardial infarction: its relationship to infarct morphology in a canine model. Circ Res. 1981;49(1):80–88. doi: 10.1161/01.res.49.1.80. [DOI] [PubMed] [Google Scholar]
- 22.Hochman JS, Bulkley BH. Expansion of acute myocardial infarction: an experimental study. Circulation. 1982;65(7):1446–1450. doi: 10.1161/01.cir.65.7.1446. [DOI] [PubMed] [Google Scholar]
- 23.Pirolo JS, Hutchins GM, Moore GW. Infarct expansion: pathologic analysis of 204 patients with a single myocardial infarct. J Am Coll Cardiol. 1986;7(2):349–354. doi: 10.1016/s0735-1097(86)80504-6. [DOI] [PubMed] [Google Scholar]
- 24.Weisman HF, Healy B. Myocardial infarct expansion, infarct extension, and reinfarction: pathophysiologic concepts. Prog Cardiovasc Dis. 1987;30(2):73–110. doi: 10.1016/0033-0620(87)90004-1. [DOI] [PubMed] [Google Scholar]
- 25.Bolognese L, Neskovic AN, Parodi G, et al. Left ventricular remodeling after primary coronary angioplasty: patterns of left ventricular dilation and long-term prognostic implications. Circulation. 2002;106(18):2351–2357. doi: 10.1161/01.cir.0000036014.90197.fa. [DOI] [PubMed] [Google Scholar]
- 26.Rizzello V, Poldermans D, Boersma E, et al. Opposite patterns of left ventricular remodeling after coronary revascularization in patients with ischemic cardiomyopathy: role of myocardial viability. Circulation. 2004;110(16):2383–2388. doi: 10.1161/01.CIR.0000145115.29952.14. [DOI] [PubMed] [Google Scholar]
- 27.Carluccio E, Biagioli P, Alunni G, et al. Patients with hibernating myocardium show altered left ventricular volumes and shape, which revert after revascularization: evidence that dyssynergy might directly induce cardiac remodeling. J Am Coll Cardiol. 2006;47(5):969–977. doi: 10.1016/j.jacc.2005.09.064. [DOI] [PubMed] [Google Scholar]
- 28.Bello D, Shah DJ, Farah GM, et al. Gadolinium cardiovascular magnetic resonance predicts reversible myocardial dysfunction and remodeling in patients with heart failure undergoing beta-blocker therapy. Circulation. 2003;108(16):1945–1953. doi: 10.1161/01.CIR.0000095029.57483.60. [DOI] [PubMed] [Google Scholar]
- 29.Bonow RO, Maurer G, Lee KL, et al. STICH Trial Investigators Myocardial viability and survival in ischemic left ventricular dysfunction. N Engl J Med. 2011;364(17):1617–1625. doi: 10.1056/NEJMoa1100358. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.