Abstract
Cardiac myosin binding protein-C (cardiac MyBP-C, cardiac C protein) belongs to a family of proteins implicated in both regulatory and structural functions of striated muscle. For the cardiac isoform, regulatory phosphorylation in vivo by cAMP-dependent protein kinase (PKA) upon adrenergic stimulation is linked to modulation of cardiac contraction. The sequence of human cardiac MyBP-C now reveals regulatory motifs specific for this isoform. Site-directed mutagenesis identifies a LAGGGRRIS loop in the N-terminal region of cardiac MyBP-C as the key substrate site for phosphorylation by both PKA and a calmodulin-dependent protein kinase associated with the native protein. Phosphorylation of two further sites by PKA is induced by phosphorylation of this isoform-specific site. This phosphorylation switch can be mimicked by aspartic acid instead of phosphoserine. Cardiac MyBP-C is therefore specifically equipped with sensors for adrenergic regulation of cardiac contraction, possibly implicating cardiac MyBP-C in cardiac disease. The gene coding for cardiac MyBP-C has been assigned to the chromosomal location 11p11.2 in humans, and is therefore in a region of physical linkage to subsets of familial hypertrophic cardiomyopathy (FHC). This makes cardiac MyBP-C a candidate gene for chromosome 11-associated FHC.
Full text
PDF



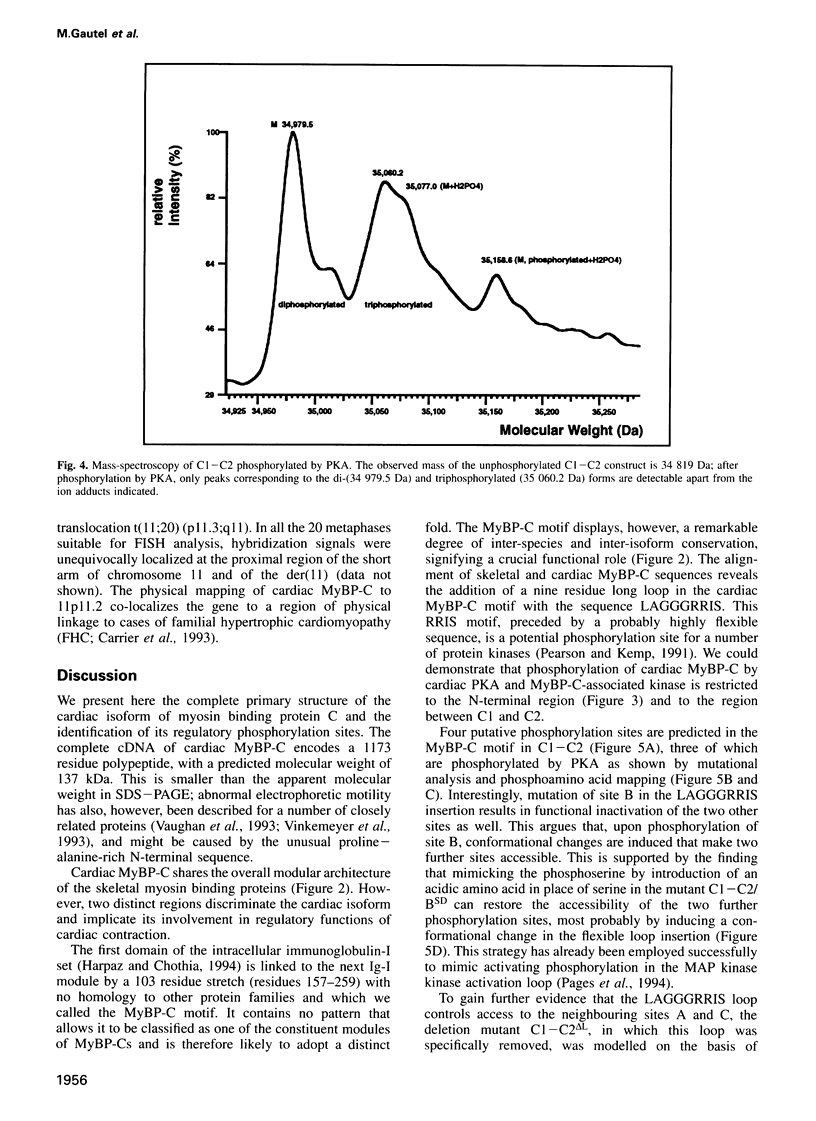
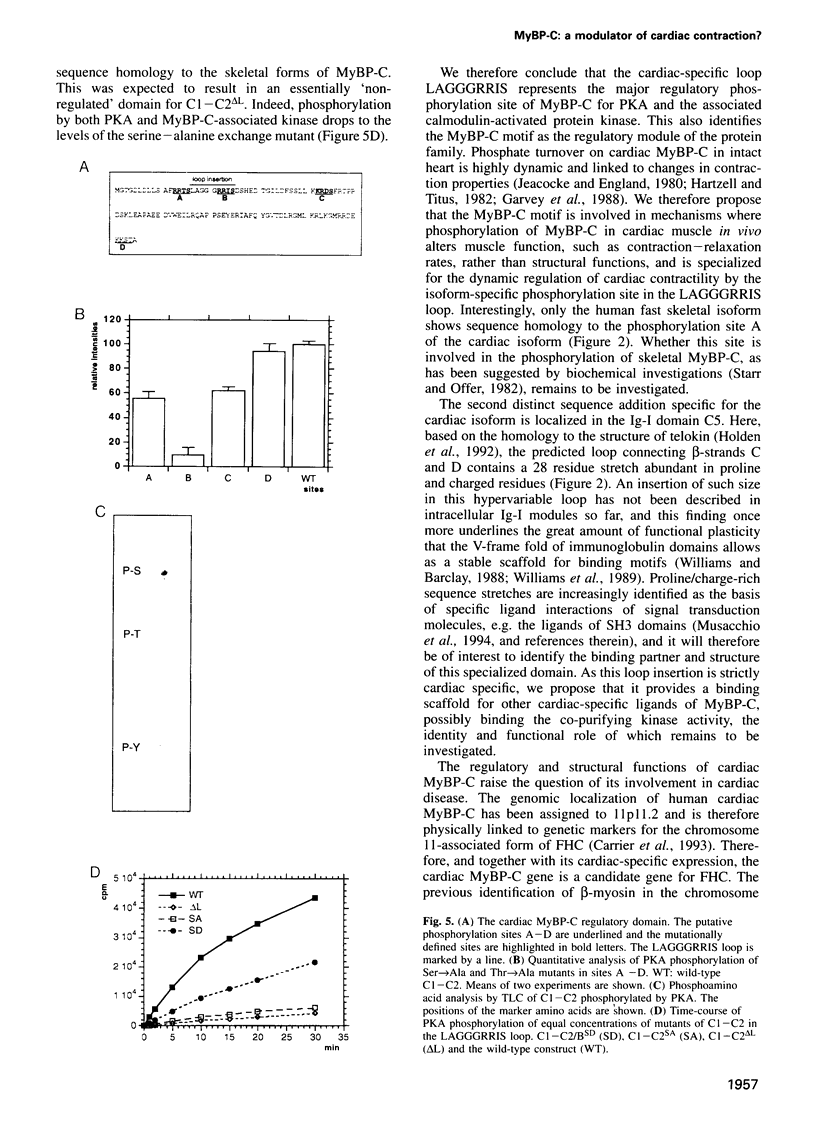
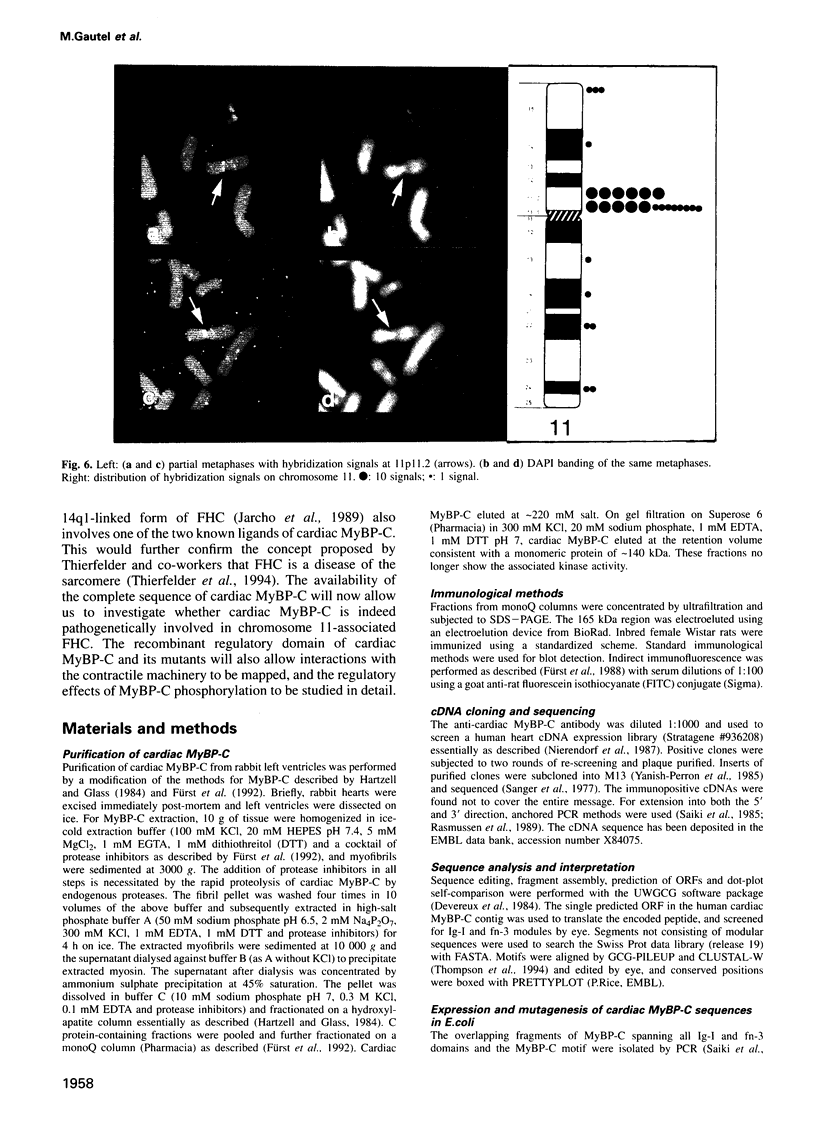


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benian G. M., Kiff J. E., Neckelmann N., Moerman D. G., Waterston R. H. Sequence of an unusually large protein implicated in regulation of myosin activity in C. elegans. Nature. 1989 Nov 2;342(6245):45–50. doi: 10.1038/342045a0. [DOI] [PubMed] [Google Scholar]
- Bennett P., Craig R., Starr R., Offer G. The ultrastructural location of C-protein, X-protein and H-protein in rabbit muscle. J Muscle Res Cell Motil. 1986 Dec;7(6):550–567. doi: 10.1007/BF01753571. [DOI] [PubMed] [Google Scholar]
- Blumenthal D. K., Charbonneau H., Edelman A. M., Hinds T. R., Rosenberg G. B., Storm D. R., Vincenzi F. F., Beavo J. A., Krebs E. G. Synthetic peptides based on the calmodulin-binding domain of myosin light chain kinase inhibit activation of other calmodulin-dependent enzymes. Biochem Biophys Res Commun. 1988 Oct 31;156(2):860–865. doi: 10.1016/s0006-291x(88)80923-9. [DOI] [PubMed] [Google Scholar]
- Boyle W. J., van der Geer P., Hunter T. Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods Enzymol. 1991;201:110–149. doi: 10.1016/0076-6879(91)01013-r. [DOI] [PubMed] [Google Scholar]
- Carrier L., Hengstenberg C., Beckmann J. S., Guicheney P., Dufour C., Bercovici J., Dausse E., Berebbi-Bertrand I., Wisnewsky C., Pulvenis D. Mapping of a novel gene for familial hypertrophic cardiomyopathy to chromosome 11. Nat Genet. 1993 Jul;4(3):311–313. doi: 10.1038/ng0793-311. [DOI] [PubMed] [Google Scholar]
- Dennis J. E., Shimizu T., Reinach F. C., Fischman D. A. Localization of C-protein isoforms in chicken skeletal muscle: ultrastructural detection using monoclonal antibodies. J Cell Biol. 1984 Apr;98(4):1514–1522. doi: 10.1083/jcb.98.4.1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Einheber S., Fischman D. A. Isolation and characterization of a cDNA clone encoding avian skeletal muscle C-protein: an intracellular member of the immunoglobulin superfamily. Proc Natl Acad Sci U S A. 1990 Mar;87(6):2157–2161. doi: 10.1073/pnas.87.6.2157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fürst D. O., Osborn M., Nave R., Weber K. The organization of titin filaments in the half-sarcomere revealed by monoclonal antibodies in immunoelectron microscopy: a map of ten nonrepetitive epitopes starting at the Z line extends close to the M line. J Cell Biol. 1988 May;106(5):1563–1572. doi: 10.1083/jcb.106.5.1563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fürst D. O., Vinkemeier U., Weber K. Mammalian skeletal muscle C-protein: purification from bovine muscle, binding to titin and the characterization of a full-length human cDNA. J Cell Sci. 1992 Aug;102(Pt 4):769–778. doi: 10.1242/jcs.102.4.769. [DOI] [PubMed] [Google Scholar]
- Garvey J. L., Kranias E. G., Solaro R. J. Phosphorylation of C-protein, troponin I and phospholamban in isolated rabbit hearts. Biochem J. 1988 Feb 1;249(3):709–714. doi: 10.1042/bj2490709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harpaz Y., Chothia C. Many of the immunoglobulin superfamily domains in cell adhesion molecules and surface receptors belong to a new structural set which is close to that containing variable domains. J Mol Biol. 1994 May 13;238(4):528–539. doi: 10.1006/jmbi.1994.1312. [DOI] [PubMed] [Google Scholar]
- Hartzell H. C., Glass D. B. Phosphorylation of purified cardiac muscle C-protein by purified cAMP-dependent and endogenous Ca2+-calmodulin-dependent protein kinases. J Biol Chem. 1984 Dec 25;259(24):15587–15596. [PubMed] [Google Scholar]
- Hartzell H. C., Titus L. Effects of cholinergic and adrenergic agonists on phosphorylation of a 165,000-dalton myofibrillar protein in intact cardiac muscle. J Biol Chem. 1982 Feb 25;257(4):2111–2120. [PubMed] [Google Scholar]
- Hofmann P. A., Hartzell H. C., Moss R. L. Alterations in Ca2+ sensitive tension due to partial extraction of C-protein from rat skinned cardiac myocytes and rabbit skeletal muscle fibers. J Gen Physiol. 1991 Jun;97(6):1141–1163. doi: 10.1085/jgp.97.6.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holden H. M., Ito M., Hartshorne D. J., Rayment I. X-ray structure determination of telokin, the C-terminal domain of myosin light chain kinase, at 2.8 A resolution. J Mol Biol. 1992 Oct 5;227(3):840–851. doi: 10.1016/0022-2836(92)90226-a. [DOI] [PubMed] [Google Scholar]
- Jarcho J. A., McKenna W., Pare J. A., Solomon S. D., Holcombe R. F., Dickie S., Levi T., Donis-Keller H., Seidman J. G., Seidman C. E. Mapping a gene for familial hypertrophic cardiomyopathy to chromosome 14q1. N Engl J Med. 1989 Nov 16;321(20):1372–1378. doi: 10.1056/NEJM198911163212005. [DOI] [PubMed] [Google Scholar]
- Jeacocke S. A., England P. J. Phosphorylation of a myofibrillar protein of Mr 150 000 in perfused rat heart, and the tentative indentification of this as C-protein. FEBS Lett. 1980 Dec 15;122(1):129–132. doi: 10.1016/0014-5793(80)80418-2. [DOI] [PubMed] [Google Scholar]
- Labeit S., Gautel M., Lakey A., Trinick J. Towards a molecular understanding of titin. EMBO J. 1992 May;11(5):1711–1716. doi: 10.1002/j.1460-2075.1992.tb05222.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Le Grice S. F., Grüninger-Leitch F. Rapid purification of homodimer and heterodimer HIV-1 reverse transcriptase by metal chelate affinity chromatography. Eur J Biochem. 1990 Jan 26;187(2):307–314. doi: 10.1111/j.1432-1033.1990.tb15306.x. [DOI] [PubMed] [Google Scholar]
- Mierendorf R. C., Percy C., Young R. A. Gene isolation by screening lambda gt11 libraries with antibodies. Methods Enzymol. 1987;152:458–469. doi: 10.1016/0076-6879(87)52054-7. [DOI] [PubMed] [Google Scholar]
- Moos C., Feng I. N. Effect of C-protein on actomyosin ATPase. Biochim Biophys Acta. 1980 Oct 1;632(2):141–149. doi: 10.1016/0304-4165(80)90071-9. [DOI] [PubMed] [Google Scholar]
- Moos C., Mason C. M., Besterman J. M., Feng I. N., Dubin J. H. The binding of skeletal muscle C-protein to F-actin, and its relation to the interaction of actin with myosin subfragment-1. J Mol Biol. 1978 Oct 5;124(4):571–586. doi: 10.1016/0022-2836(78)90172-9. [DOI] [PubMed] [Google Scholar]
- Moos C., Offer G., Starr R., Bennett P. Interaction of C-protein with myosin, myosin rod and light meromyosin. J Mol Biol. 1975 Sep 5;97(1):1–9. doi: 10.1016/s0022-2836(75)80017-9. [DOI] [PubMed] [Google Scholar]
- Musacchio A., Saraste M., Wilmanns M. High-resolution crystal structures of tyrosine kinase SH3 domains complexed with proline-rich peptides. Nat Struct Biol. 1994 Aug;1(8):546–551. doi: 10.1038/nsb0894-546. [DOI] [PubMed] [Google Scholar]
- Obinata T., Reinach F. C., Bader D. M., Masaki T., Kitani S., Fischman D. A. Immunochemical analysis of C-protein isoform transitions during the development of chicken skeletal muscle. Dev Biol. 1984 Jan;101(1):116–124. doi: 10.1016/0012-1606(84)90122-2. [DOI] [PubMed] [Google Scholar]
- Offer G., Moos C., Starr R. A new protein of the thick filaments of vertebrate skeletal myofibrils. Extractions, purification and characterization. J Mol Biol. 1973 Mar 15;74(4):653–676. doi: 10.1016/0022-2836(73)90055-7. [DOI] [PubMed] [Google Scholar]
- Okagaki T., Weber F. E., Fischman D. A., Vaughan K. T., Mikawa T., Reinach F. C. The major myosin-binding domain of skeletal muscle MyBP-C (C protein) resides in the COOH-terminal, immunoglobulin C2 motif. J Cell Biol. 1993 Nov;123(3):619–626. doi: 10.1083/jcb.123.3.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagès G., Brunet A., L'Allemain G., Pouysségur J. Constitutive mutant and putative regulatory serine phosphorylation site of mammalian MAP kinase kinase (MEK1). EMBO J. 1994 Jul 1;13(13):3003–3010. doi: 10.1002/j.1460-2075.1994.tb06599.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson R. B., Kemp B. E. Protein kinase phosphorylation site sequences and consensus specificity motifs: tabulations. Methods Enzymol. 1991;200:62–81. doi: 10.1016/0076-6879(91)00127-i. [DOI] [PubMed] [Google Scholar]
- Politou A. S., Gautel M., Joseph C., Pastore A. Immunoglobulin-type domains of titin are stabilized by amino-terminal extension. FEBS Lett. 1994 Sep 19;352(1):27–31. doi: 10.1016/0014-5793(94)00911-2. [DOI] [PubMed] [Google Scholar]
- Politou A. S., Gautel M., Pfuhl M., Labeit S., Pastore A. Immunoglobulin-type domains of titin: same fold, different stability? Biochemistry. 1994 Apr 19;33(15):4730–4737. doi: 10.1021/bi00181a604. [DOI] [PubMed] [Google Scholar]
- Rasmussen U. B., Basset P., Daniel J. Y. Direct amplification of cDNA inserts from lambda libraries using the cloning-adapter as primer for PCR. Nucleic Acids Res. 1989 Apr 25;17(8):3308–3308. doi: 10.1093/nar/17.8.3308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi E., Zarrilli R., Zuffardi O. Regional assignment of the gene coding for a human Graves' disease autoantigen to 10q21.3-q22.1. Hum Genet. 1993 Feb;90(6):653–654. doi: 10.1007/BF00202485. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Scharf S., Faloona F., Mullis K. B., Horn G. T., Erlich H. A., Arnheim N. Enzymatic amplification of beta-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science. 1985 Dec 20;230(4732):1350–1354. doi: 10.1126/science.2999980. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlender K. K., Bean L. J. Phosphorylation of chicken cardiac C-protein by calcium/calmodulin-dependent protein kinase II. J Biol Chem. 1991 Feb 15;266(5):2811–2817. [PubMed] [Google Scholar]
- Schultheiss T., Lin Z. X., Lu M. H., Murray J., Fischman D. A., Weber K., Masaki T., Imamura M., Holtzer H. Differential distribution of subsets of myofibrillar proteins in cardiac nonstriated and striated myofibrils. J Cell Biol. 1990 Apr;110(4):1159–1172. doi: 10.1083/jcb.110.4.1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schweizer D. Counterstain-enhanced chromosome banding. Hum Genet. 1981;57(1):1–14. [PubMed] [Google Scholar]
- Soteriou A., Gamage M., Trinick J. A survey of interactions made by the giant protein titin. J Cell Sci. 1993 Jan;104(Pt 1):119–123. doi: 10.1242/jcs.104.1.119. [DOI] [PubMed] [Google Scholar]
- Starr R., Offer G. Preparation of C-protein, H-protein, X-protein, and phosphofructokinase. Methods Enzymol. 1982;85(Pt B):130–138. doi: 10.1016/0076-6879(82)85016-7. [DOI] [PubMed] [Google Scholar]
- Starr R., Offer G. The interaction of C-protein with heavy meromyosin and subfragment-2. Biochem J. 1978 Jun 1;171(3):813–816. doi: 10.1042/bj1710813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Studier F. W., Rosenberg A. H., Dunn J. J., Dubendorff J. W. Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol. 1990;185:60–89. doi: 10.1016/0076-6879(90)85008-c. [DOI] [PubMed] [Google Scholar]
- Thierfelder L., Watkins H., MacRae C., Lamas R., McKenna W., Vosberg H. P., Seidman J. G., Seidman C. E. Alpha-tropomyosin and cardiac troponin T mutations cause familial hypertrophic cardiomyopathy: a disease of the sarcomere. Cell. 1994 Jun 3;77(5):701–712. doi: 10.1016/0092-8674(94)90054-x. [DOI] [PubMed] [Google Scholar]
- Thompson J. D., Higgins D. G., Gibson T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994 Nov 11;22(22):4673–4680. doi: 10.1093/nar/22.22.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan K. T., Weber F. E., Einheber S., Fischman D. A. Molecular cloning of chicken myosin-binding protein (MyBP) H (86-kDa protein) reveals extensive homology with MyBP-C (C-protein) with conserved immunoglobulin C2 and fibronectin type III motifs. J Biol Chem. 1993 Feb 15;268(5):3670–3676. [PubMed] [Google Scholar]
- Vaughan K. T., Weber F. E., Fischman D. A. cDNA cloning and sequence comparisons of human and chicken muscle C-protein and 86kD protein. Symp Soc Exp Biol. 1992;46:167–177. [PubMed] [Google Scholar]
- Vinkemeier U., Obermann W., Weber K., Fürst D. O. The globular head domain of titin extends into the center of the sarcomeric M band. cDNA cloning, epitope mapping and immunoelectron microscopy of two titin-associated proteins. J Cell Sci. 1993 Sep;106(Pt 1):319–330. doi: 10.1242/jcs.106.1.319. [DOI] [PubMed] [Google Scholar]
- Weber F. E., Vaughan K. T., Reinach F. C., Fischman D. A. Complete sequence of human fast-type and slow-type muscle myosin-binding-protein C (MyBP-C). Differential expression, conserved domain structure and chromosome assignment. Eur J Biochem. 1993 Sep 1;216(2):661–669. doi: 10.1111/j.1432-1033.1993.tb18186.x. [DOI] [PubMed] [Google Scholar]
- Williams A. F., Barclay A. N. The immunoglobulin superfamily--domains for cell surface recognition. Annu Rev Immunol. 1988;6:381–405. doi: 10.1146/annurev.iy.06.040188.002121. [DOI] [PubMed] [Google Scholar]
- Williams A. F., Davis S. J., He Q., Barclay A. N. Structural diversity in domains of the immunoglobulin superfamily. Cold Spring Harb Symp Quant Biol. 1989;54(Pt 2):637–647. doi: 10.1101/sqb.1989.054.01.075. [DOI] [PubMed] [Google Scholar]
- Yamamoto K., Moos C. The C-proteins of rabbit red, white, and cardiac muscles. J Biol Chem. 1983 Jul 10;258(13):8395–8401. [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]