Abstract
Rationale: We attenuated virulent Bordetella pertussis by genetically eliminating or detoxifying three major toxins. This strain, named BPZE1, is being developed as a possible live nasal vaccine for the prevention of whooping cough. It is immunogenic and safe when given intranasally in adult volunteers.
Objectives: Before testing in human infants, we wished to examine the potential effect of BPZE1 on a common pediatric infection (respiratory syncytial virus [RSV]) in a preclinical model.
Methods: BPZE1 was administered before or after RSV administration in adult or neonatal mice. Pathogen replication, inflammation, immune cell recruitment, and cytokine responses were measured.
Measurements and Main Results: BPZE1 alone did not cause overt disease, but induced efflux of neutrophils into the airway lumen and production of IL-10 and IL-17 by mucosal CD4+ T cells. Given intranasally before RSV infection, BPZE1 markedly attenuated RSV, preventing weight loss, reducing viral load, and attenuating lung cell recruitment. Given neonatally, BPZE1 also protected against RSV-induced weight loss even through to adulthood. Furthermore, it markedly increased IL-17 production by CD4+ T cells and natural killer cells and recruited regulatory cells and neutrophils after virus challenge. Administration of anti–IL-17 antibodies ablated the protective effect of BPZE1 on RSV disease.
Conclusions: Rather than enhancing RSV disease, BPZE1 protected against viral infection, modified viral responses, and enhanced natural mucosal resistance. Prevention of RSV infection by BPZE1 seems in part to be caused by induction of IL-17.
Clinical trial registered with www.clinicaltrials.gov (NCT 01188512).
Keywords: bronchiolitis, vaccine, whooping cough, mucosal immunity, innate immunity
At a Glance Commentary
Scientific Knowledge on the Subject
Viral infections are known to promote bacterial pneumonia, but bacterial exposure may also modulate responses to viruses. Mechanisms that underlie these interactions are diverse and often poorly defined.
What This Study Adds to the Field
In mice, a novel genetically engineered live bacterial vaccine (designed to protect against whooping cough) did not exacerbate the inflammatory response to infection with respiratory syncytial virus; it instead provided considerable protection. This nonspecific effect was long-lasting, and depended on the cytokine IL-17.
Respiratory tract infections (RTIs) are the most frequent cause of childhood morbidity and mortality worldwide. Despite many advances in the prevention and treatment of RTIs, they currently cause more than 4 million deaths per year. Most of the deaths occur in young children, mainly because no satisfactory vaccine is available, or existing effective vaccines do not reach those children who require protection.
Bordetella pertussis and respiratory syncytial virus (RSV) are both important causes of RTI in young children throughout the world. RSV is the major cause of viral bronchiolitis in infants (1), and triggers wheezing disease in later childhood (2). Despite more than 50 years of research, a safe and effective vaccine remains elusive and treatment remains supportive. Most children are infected by 1 year of age, and virtually all by the third RSV season. Although infection induces serum antibodies, they are insufficient to protect reliably against reinfection, which occurs approximately every 3–5 years throughout life. The occurrence and severity of infection remains highly unpredictable. The reasons for resistance or susceptibility remain poorly understood. B. pertussis is a common cause of bacterial RTI, sometimes causing severe and even life-threatening whooping cough in infants. Vaccines have been available for several decades, but none is sufficiently effective and safe in young infants, probably because of suboptimal T-cell function in the newborn (3). However, natural B. pertussis RTI does protect against reinfection, even in children as young as 1 month of age (4). This observation prompted us to develop a live attenuated B. pertussis mutant to be delivered by the nasal route to mimic natural infection without causing disease.
In this live vaccine strain, named BPZE1, the tracheal cytotoxin and dermonecrotic toxin were genetically removed and pertussis toxin (PT) was genetically detoxified by two independent mutations (5). A single nasal administration of BPZE1 protects mice against infection with wild-type B. pertussis (6) and is safe and immunogenic when given intranasally to healthy adult volunteers. In addition, nasal administration of BPZE1 protects mice from the effects of influenza A infection (7). The mechanism underlying this effect is unknown, but it is intriguing that reduced lung inflammation, cytokine release, and tissue damage is seen, whereas viral load is unaffected.
In this study we investigated the effects of BPZE1 on the course of RSV infection in mice. We found that prior BPZE1 infection changes the response to subsequent RSV challenge, and that the effects are surprisingly long-lasting. The innate imprinting caused by BPZE1 was associated with up-regulation of IL-17A accompanied by induction of regulatory cells (Foxp3+ or IL-10+ CD4). The effects of BPZE1 on RSV infection could be largely prevented by depletion of IL-17. Our studies are the first to show that nasal colonization with benign live-vaccine bacteria can induce substantial and durable protection against an unrelated common viral pathogen by the production of IL-17.
Some of the results of these studies have been previously reported in the form of abstracts (8, 9).
Methods
Mice and Infections
All procedures were performed in accordance with UK Home Office guidelines. Six- to 8-week-old female BALB/c mice were anesthetized and intranasally infected with 106 attenuated BPZE1 (6) or virulent B. pertussis (BpSM) (5) and/or 5 × 105 pfu RSV or phosphate-buffered saline control. Mice of 2–5 days of age were infected intranasally with 106 BPZE1 and 8- to 10-week adult mice were challenged with 5 × 105 pfu RSV. For depletions, mice were injected with 100-μg anti–IL-17 antibody (clone 50104, R&D, Abingdon, UK) or 100-μg isotype control (IgG2a) intraperitoneally 1 day before and every other day after RSV challenge.
Bacterial and Viral Procedures
BPZE1 is a streptomycin-resistant B. pertussis Tohama I derivative with a deleted dermonecrotic-encoding gene, producing inactivated PT and background levels of tracheal cytotoxin (6). BPZE1 and virulent BpSM stocks were generated by culturing the bacteria for 72 hours at 37°C in Stainer-Scholte medium, as described (10); viable counts were determined by plating on supplemented Bordet-Gengou agar (Difco, Detroit, MI) incubated at 37°C for 48 hours. To monitor colonization, lungs were homogenized and plated onto Bordet-Gengou agar plates for 120 hours. Plaque-purified human RSV (strain A2) was grown in Hep2 cells in serum-free Dulbecco’s modified Eagle medium media (11). Virus load was measured by plaque assay or quantitative polymerase chain reaction for RSV L-gene (12).
Bronchoalveolar Lavage and Cell Isolation
Bronchoalveolar lavage (BAL)-accessible cells were obtained by repeated instillation of 1 ml of phosphate-buffered saline containing 12 mM lidocaine by the trachea in killed animals. Supernatants were stored at −80°C for cytokine analysis and BAL cells were centrifuged onto microscope slides using a cytospin (Thermo Scientific, Loughborough, UK) before staining with hematoxylin and eosin (Reagena, Gamidor, UK) and analyzed by light microscopy (300 cells per slide). For total lung cell preparations, lungs were homogenized and digested with collagenase D (50 μg/ml, Sigma-Aldrich, Gillingham, UK) for 30 minutes and red blood cells were lysed. Count Bright counting beads (Invitrogen, Paisley, UK) were used in combination with the live/dead marker 7AAD to determine cell numbers.
Flow Cytometry
Cells were incubated with LIVE/DEAD Fixable cell stain (Invitrogen), then stained for 20 minutes with CD3-Pacific Blue, CD4-PerCP-Cy5.5, CD8-PE or fluorescein isothiocyanate, Nkp46–fluorescein isothiocyanate, DX5-PE-Cy7, and CD19-APC (all from BD Bioscience, Oxford, UK). Cells were gated for live cells, singlets, and lymphocytes before analysis. For intracellular cytokine production cells were stimulated with 100 ng/ml phorbol myristate acetate and 1 μg/ml ionomycin for 3 hours, adding Golgi stop (BD) after the first hour. Cells were fixed (BD Cytofix/Cytoperm) and permeabilized (BD permwash), then incubated with IFN-γ-PerCp-Cy5.5 or IFN-γ-PE, IL-10-APC, IL-17-PE, and IL-4-APC antibodies before analysis on an LSR II (BD, UK), collecting 100,000 live events. Data were analyzed using FlowJo (v7.6.5, Treestar Ashland, OR).
Antigen-specific ELISA
RSV antigen– and B. pertussis filamentous hemagglutinin (FHA)-specific immunoglobulins were measured as described (13, 14).
Cytokine ELISA
mIL-4, IL-10, IL-17, and IFN-γ were measured in BAL fluids using R&D Duoset kits. Limits of detection were as follows: IL-4, 7.5 pg; IL-10, 15 pg; IL-17, 7.5 pg; and IFN-γ, 15 pg.
Statistical Analysis
GraphPad Prism software (La Jolla, CA) was used to analyze data, generally shown as mean ± SEM of five animals per group. Mann-Whitney test and two-way analysis of variance were used to compare data.
Results
BPZE1 Colonization of Lung Induces a Transient Influx of Neutrophils and Primes T Cells toward IL-10 and IL-17 Production
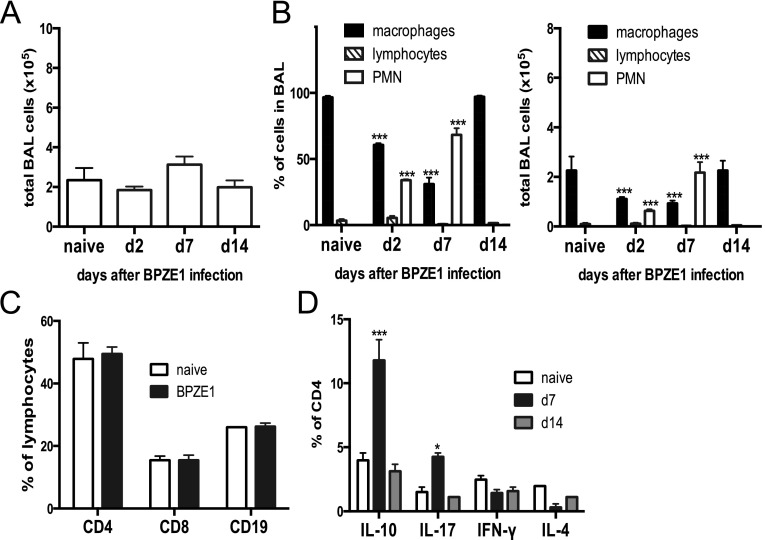
Intranasal administration of BPZE1 did not cause weight loss (data not depicted) or significant cell influx to the airways (Figure 1A). It did, however, cause an alteration in BAL cell composition, inducing a transient neutrophilia that peaked on Day 7 (P < 0.001 compared with naive) (Figure 1B). By Day 14, the BAL cell composition returned to normal and macrophages again predominated (Figure 1B). Lung lymphocyte subsets (analyzed by flow cytometry) showed no change in T- and B-cell proportions after BPZE1 infection (Figure 1C; total lung number shown in Figure E3 in the online supplement). However, there was a transient induction of IL-10– and IL-17–producing CD4+ T cells 7 days after BPZE1 infection (Figure 1D; total lung number shown in Figure E3).
Figure 1.
Lung cellular response to BPZE1 infection. Mice were treated intranasally with phosphate-buffered saline (naive) or BPZE1 and analyzed 2, 7, and 14 days after infection. (A) Total number of cells recruited to the airways shows no significant change (P > 0.05). (B) BPZE1 infection induces a transient polymorphonuclear (PMN) cell efflux into the airway (open bars) P < 0.001 as compared with naive (two-way analysis of variance) with corresponding decline in the proportion of macrophages (black bars), whereas the proportion of lymphocytes (hatched bars) does not change significantly over time; this is mirrored in cell numbers. (C) Lymphocyte populations in the lung are similar 14 days after phosphate-buffered saline (naive, open bars) or BPZE1 (black bars) inoculation. (D) Intracellular cytokine staining of lung cells from BPZE1-infected mice stimulated with phorbol myristate acetate/ionomycin shows a transient boost of IL-10 and IL-17 production by CD4+ T cells on Day 7 (black bars Day 7; gray bars Day 14) (*P < 0.05, ***P < 0.001 as compared with naive; two-way analysis of variance; similar data in two experiments, six animals per group). BAL = bronchoalveolar lavage.
Prior RSV Does Not Affect Response to BPZE1 Infection
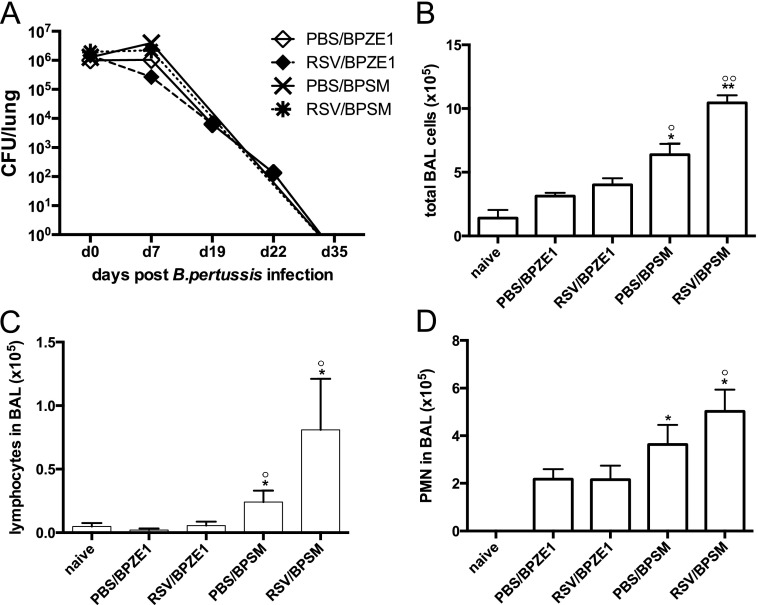
To examine whether prior RSV infection affected BPZE1 or BpSM responses, mice were infected with BPZE1 or virulent BpSM 2 weeks after RSV infection. RSV was undetectable in all groups by the time of bacterial infection and remained undetectable throughout bacterial challenge (not depicted). Both B. pertussis strains colonized the lung to a similar extent, regardless of prior RSV infection, and could be detected in homogenized lungs up to Day 22 postinfection (102 cfu per lung) (Figure 2A). All tested groups cleared BPZE1 or BpSM infection after 35 days.
Figure 2.
Interactions between respiratory syncytial virus (RSV), BPZE1, and virulent Bordetella pertussis (BpSM) infections. (A) Mice were infected with RSV 14 days before BPZE1 (RSV/BPZE1, solid diamonds) and virulent B. pertussis (RSV/BpSM, black stars) or inoculated with phosphate-buffered saline (PBS) before BPZE1 (PBS/BPZE1, open diamonds) or BpSM (PBS/BpSM, black crosses), and the CFU were determined in the lungs by blood agar culture at the indicated time points. (B–D) Mice were infected with RSV or treated with PBS 14 days before BPZE1 or BpSM infection and analyzed on Day 7. (B) Total numbers of bronchoalveolar lavage (BAL) cell influx after bacterial infection. (C) Numbers of lymphocytes in BAL. (D) Numbers of polymorphonuclear (PMN) in BAL. *P < 0.05, **P < 0.01 compared with naive; °P < 0.05, °°P < 0.01 compared with BPZE1/RSV, Mann-Whitney test; similar data in two experiments, six animals per group.
Infection with virulent BpSM led to an inflammatory response in the lungs, which was increased by previous RSV infection (Figure 2B). This was accompanied by efflux of lymphocytes and neutrophils into the BAL (Figures 2C and 2D). In contrast, previous RSV infection did not induce or exaggerate the immune response toward BPZE1 (Figures 2B–2D).
BPZE1 Prevents Weight Loss, Reduces Viral Load, and Alters Inflammation after RSV Infection
Infection with RSV A2 induces a substantial dose-dependent weight loss in mice, peaking at Day 6 of infection. Priming with BPZE1 14 days before challenge with RSV completely abolished the weight loss induced by RSV (Figure 3A). All groups recovered to 110% of their original weight by Day 18. Viral load usually peaks on Day 4, and prior BPZE1 inoculation reduced RSV RNA copy numbers after challenge twofold to threefold (Figure 3B) by Days 4 and 5. The kinetics of RSV- or FHA-specific antibodies were unaffected and no cross-reactivity was detected by coinfection (see Figure E1).
Figure 3.
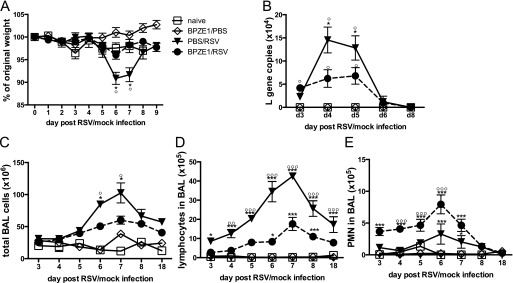
BPZE1 infection prior to respiratory syncytial virus (RSV) attenuates viral infection. Mice were treated with phosphate-buffered saline (PBS) (naive, open squares), infected with BPZE1 and then inoculated with PBS (BPZE1/PBS, open diamonds), treated with PBS and then infected with RSV (PBS/RSV, solid triangles), or infected with BPZE1 before RSV challenge (BPZE1/RSV, solid circles, dotted lines). RSV challenge 14 days after BPZE1 or PBS inoculation = Day 0 on the graphs. (A) Weight patterns after RSV or PBS challenge, showing that BPZE1 protects against RSV-induced weight loss. (B) Number of L gene copies 3, 4, and 5 days after RSV or PBS inoculation shows that BPZE1 reduces subsequent RSV replication (*P < 0.05 as compared with BPZE1/RSV, Mann-Whitney). (C) Prior BPZE1 inoculation reduces the total bronchoalveolar lavage (BAL) cell response to RSV infection and (D) lymphocyte numbers in BAL and (E) enhances the polymorphonuclear (PMN) response to RSV infection. *P < 0.05, ***P < 0.001 as compared with naive (two-way analysis of variance); °P < 0.05, °°°P < 0.001 as compared with BPZE1/RSV (two-way analysis of variance); the data shown are representative of three experiments, six animals per group.
BPZE1 inoculation before RSV challenge significantly reduced cell numbers recruited to the airway after RSV infection (Figure 3C). The recruited cells were mainly lymphocytes 5–8 days after RSV infection, the numbers of which were significantly reduced by prior BPZE1 infection (Figure 3D). The percentage of macrophages in the BAL of BPZE1/RSV- infected animals was significantly increased throughout the time course, when compared with RSV-infected mice (see Figure E2).
BPZE1 priming before RSV infection led to a transient increase of polymorphonuclear cells 3–7 days after RSV challenge (Figure 3E). Importantly, we did not observe eosinophil recruitment in any group at any given time point (data not shown).
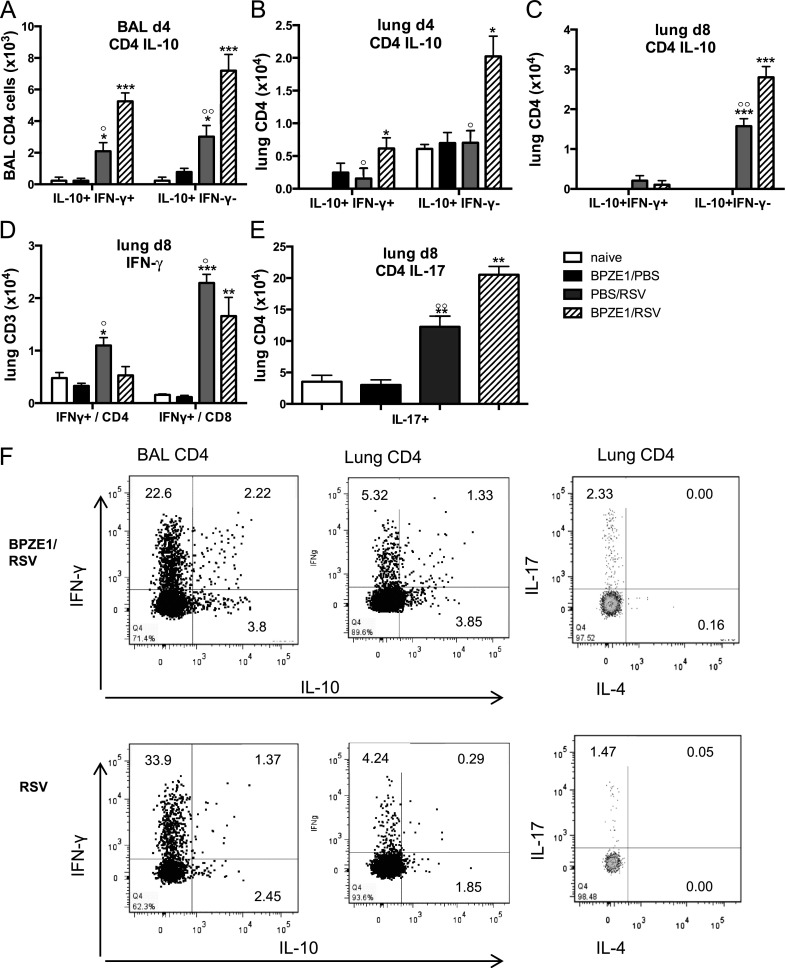
We used flow cytometry to analyze intracellular production of IFN-γ, IL-4, IL-17, and IL-10 at Days 4 (peak of viral load) and 8 (peak of immunopathology) after RSV challenge (Figure 4). Prior BPZE1 infection did not affect single IFN-γ production by CD4+ or CD8+ T cells on Day 4 (data not shown), but enhanced numbers of IL-10–producing CD4+ T cells in BAL (Figure 4A) and lung (Figure 4B), including cells coproducing IFN-γ. IL-10 production in CD4+ lung cells was enhanced by prior BPZE1 infection, although RSV infection alone also induced IL-10 expression (Figures 4A and 4C). In contrast, by Day 8 IFN-γ production by CD4+ and by CD8+ T cells in lung was significantly reduced by prior BPZE1 infection (Figure 4D). Interestingly, CD4+ T cell IL-17 production was induced by RSV infection, and this was enhanced by prior BPZE1 infection (Figure 4E). Levels of cytokines in the BAL fluid showed enhancement of IL-17 and reductions in regulated on activation normal T-cell expressed and secreted (RANTES) and tumor necrosis factor-α in mice previously infected with BPZE1 (see Figure E4). Examples of flow cytometric results from representative mice are shown in Figure 4F.
Figure 4.
BPZE1 prior to respiratory syncytial virus (RSV) infection boosts IL-10 and IL-17 production by CD4+ cells. Mice were treated with phosphate-buffered saline (PBS) (naive, open bars), infected with BPZE1, and inoculated with PBS (BPZE1, black bars), treated with PBS and infected with RSV (RSV, gray bars), or infected with BPZE1 before RSV challenge (BPZE1/RSV, hatched bars). Bronchoalveolar lavage (BAL) and lung lymphocytes were analyzed by flow cytometry for intracellular cytokine production after phorbol myristate acetate/ionomycin stimulation on Days 4 and 8 after RSV infection. IL-10 single and IL-10/IFN-γ double-producing CD4+ T cells in (A) BAL on Day 4, (B) lung on Day 4, (C) lung on Day 8. (D) IFN-γ production by CD4+ and CD8+ T cells in lung on Day 8. (E) IL-17–producing CD4+ T cells in lung on Day 8. (F) Example of fluorescence-activated cell sorter plots for IFN-γ, IL-10, IL-17, and IL-4 intracellular staining in CD4+ lung cells of RSV- and BPZE1/RSV-treated mice. *P < 0.05, **P < 0.01, ***P < 0.001 as compared with naive; °P < 0.05, °°P < 0.01, as compared with BPZE1/RSV; the data shown are representative of three experiments, six animals per group.
BPZE1 Priming of Neonatal Mice Protects against RSV Challenge in Adulthood
Mice primed with BPZE1 on Days 2–5 of life and challenged with RSV in adulthood were protected from RSV-induced weight loss (Figure 5A). We also observed an increase in the number of neutrophils in the BAL of these mice (data not shown). Intracellular expression of IL-17A was also increased in CD3+CD4+ T cells and natural killer (NK) cells (CD3−DX5+) (Figures 5B and 5C; see Figure E5), whereas IL-10 expression was unaffected (data not shown). Macrophages and neutrophils were not found to produce substantial amounts of IL-17 in this setting (see Figures E6–E8).
Figure 5.
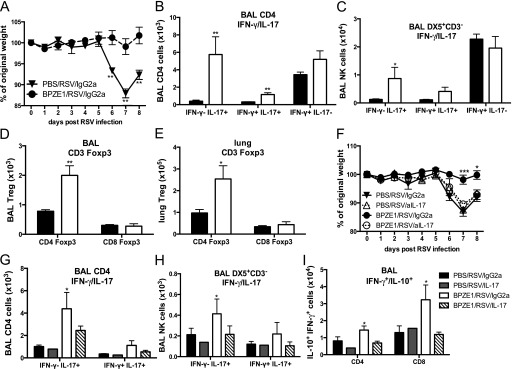
Priming of neonatal mice with BPZE1 induces IL-17 production and protects against adult respiratory syncytial virus (RSV) challenge. Neonatal mice (2–5 d old) were primed with phosphate-buffered saline (PBS/RSV, solid triangles, black bars) or BPZE1 (BPZE1/RSV, solid circles, white bars) and challenged with RSV in adulthood (age 8 wk). (A) Weight loss after RSV challenge. (B) CD4+ T cells producing IFN- γ, IL-17, or both. (C) Natural killer (NK) cells (DX5+CD3−) producing IFN-γ, IL-17, or both. Foxp3+ regulatory T cells (Tregs) in airways bronchoalveolar lavage (BAL) (D) and lung (E). *P < 0.05, **P < 0.01, ***P < 0.001 as compared with PBS/RSV; the data shown are representative of three experiments, four to six animals per group. (F–H) Depletion of IL-17 in vivo. Neonate mice were primed with BPZE1 or PBS and challenged 8 weeks later with RSV. On Days −1, 1, 3, 5, and 7, mice received an anti–IL-17 or control IgG2a antibody intraperitoneally (PBS/RSV/IgG2a = RSV+ control antibody, solid triangles, black bars; PBS/RSV/aIL-17 = RSV+ anti-IL-17 antibody, open triangles, gray bars; BPZE1/RSV/IgG2a = BPZE1+ RSV+ control antibody, solid circles, white bars; BPZE1/RSV/aIL-17 = BPZE1+ RSV+ anti–IL-17 antibody, open circles, hatched bars). (F) Weight loss after adult RSV challenge. IL-17 single or IL-17 IFN-γ double-producing (G) CD4+ T cells and (H) NK cells. (I) IFN-γ/IL-10 double-producing T cells in BAL. *P < 0.05, **P < 0.01, ***P < 0.001 compared with PBS/RSV/IgG2a; the data shown are representative of three experiments, five to six animals per group.
During RSV infection, Foxp3+ CD4+ regulatory T cell (Tregs) numbers were elevated in both the BAL and lungs of adult mice primed with BPZE1 as neonates, compared with nonprimed mice (Figures 5D and 5E).
Protective Effects of BPZE1 Are Inhibited by Blocking IL-17
To test the relevance of IL-17 to these effects, mice primed with BPZE1 as neonates were injected with an anti–IL-17 antibody 1 day before adult RSV challenge and on alternate days thereafter. Strikingly, blocking of IL-17 reestablished the weight loss prevented by neonatal BPZE1 priming (Figure 5F) but did not otherwise further enhance inflammation in animals infected with RSV. Furthermore, depletion of IL-17 prevented the accumulation of IL-17 single and IL-17/IFN-γ double producers in CD4+ T cells and NK cells (Figures 5G and 5H) found in protected neonates. Also, the recruitment of CD4+ and CD8+ T cells and IFN-γ/IL-10 double producers was impaired (Figure 5I).
Discussion
In this study, we evaluated the effect of BPZE1 in the modulation of virus-induced airway inflammation in mice. We found that RSV infection did not interfere with B. pertussis colonization of the lung but significantly attenuated RSV disease when BPZE1 infection preceded RSV challenge. Virus infections (e.g., RSV and influenza) may alter the pulmonary immune defense against bacterial infections (15, 16), but we found that prior RSV infection did not aggravate inflammatory responses to BPZE1. However, responses to virulent B. pertussis were enhanced.
BPZE1 was designed to protect against whooping cough in children less than 6 months of age. We therefore analyzed the effects of immunizing neonatal mice with BPZE1 on the outcome of RSV challenge in adulthood; even after this long interval, BPZE1 continued to protect against RSV disease. Part of this effect could be a result of innate sensing of bacteria (with BPZE1 as source of TLR ligands) leading to mucosal imprinting, analogous to the effects of CpG in neonatal mice (17). However, recent studies with influenza A show the need of live BPZE1 because heat-killed bacteria did not provide protection (7) thus indicating a combination of effects.
In humans, it has been reported that episodes of virulent Bordetella infection may be associated with less severe RSV bronchiolitis (18), supporting the possibility that live attenuated B. pertussis vaccines might have beneficial effects on viral infections. However, both RSV and B. pertussis infections in children are associated with a higher risk of developing allergic disorders or asthma (19–21). By contrast, BPZE1 has been shown to prevent allergic ovalbumin-induced airway inflammation (22) and contact dermatitis (23) in mice. Hence, enhanced maturation of the immune system and induction of Th1/Th17 responses (rather than the Th2 priming, thought to be responsible for enhanced RSV disease after formalin-inactivated vaccine administration [24]) might be an additional benefit of administration of BPZE1 in infancy.
Neonatal BPZE1 infection enhanced the induction of IL-17A and recruitment of neutrophils to the airways during adult RSV infection. This, as in our adult challenge studies, was not associated with enhanced pathology. These findings are similar to those from studies of TLR ligand administration, shown to influence RSV disease by neutrophil recruitment without inducing pathology (25). The enhanced virus clearance observed in mice primed with BPZE1 may possibly be caused by enhancement of neutrophil recruitment because neutrophils may have antiviral actions by NET formation or oxidative killing (26); alternatively, enhanced Th17 responses could increase antiviral effects by activation of NK cells (27) or by direct antiviral effects of Th17-polarized CD4 T-cell effector cells (28). Although we observed significantly reduced viral load after BPZE1 priming, this does not fully account for the beneficial effects of BPZE1. In our experience, the levels of RSV replication seen in BPZE1-vaccinated mice would normally be associated with greater weight loss and lung inflammation than that seen in the present studies. In addition, depletion of IL-17 did not increase viral persistence (see Figure E5). Furthermore, studies of the protective effects of BPZE1 on influenza (7) show no reduction of viral load but increased survival highlighting the immunomodulatory rather than antiviral aspects of BPZE1.
Neutrophils may promote Th1 responses that are protective against other murine infections (29, 30). Human studies also indicate an important role for neutrophils, linking neutrophils with impaired TLR4 signaling to severe RSV bronchiolitis (31). In the present studies we found that BPZE1 inoculation resulted in a very moderate neutrophil recruitment (<12% of BAL cells), and BPZE1 colonization caused no lung inflammation or neutrophil excess by Day 7 after infection, suggesting that BPZE1-mediated recruitment of neutrophils does not contribute to airway inflammation. The number of lung neutrophils seems to be tightly regulated, to resolve infection without excessive inflammation (32).
BPZE1 infection before RSV challenge decreased the levels of IFN-γ and induced stable production of IL-10 in the BAL fluid. IFN-γ produced by NK and CD8+ cells is a normal component of protective antiviral responses, but excessive IFN-γ production can be detrimental in RSV disease. IL-10 has immunosuppressive effects, and we and others have shown that IL-10 is crucial in a mouse model of RSV infection and its depletion leads to increased disease (33–35). Thus, an increase in IL-10 is expected to reduce immunopathology and interestingly B. pertussis FHA protein has been shown to induce IL-10–producing Tr1 cells (36). However, IL-10+ T cells were not significantly enhanced after neonatal priming with BPZE1, arguing against an exclusive role for IL-10 in this situation. We have previously shown that Tregs modulate RSV disease (37), so increased Treg numbers could in part be responsible for the effects that we observed in the present studies. However, we only observed increased Treg numbers after neonatal priming and not after adult priming, although BPZE1 provides protection in both scenarios. We therefore infer that both Foxp3-positive Tregs and IL-10–producing CD4 T cells can contribute to BPZE1 protection, separately providing synergistic roles in supporting the beneficial effect to BPZE1 that depends, in either case, on IL-17 production.
Both BPZE1 priming in adults before RSV challenge and neonatal priming with BPZE1 followed by adult challenge led to the induction of IL-17–producing cells. We found that although IL-17 in adult primed mice was mainly derived from Th17 (CD4+ IL-17A+) cells, IL-17 in neonatally primed mice was produced by both CD3+CD4+ T cells and by DX5+CD3− NK cells. Macrophages and neutrophils only produced minimal amounts in both models. In vitro, BPZE1 has been shown to prime human Th1/Th17 cell responses (38) and suppressor T cells. PT has been shown to induce IL-17–producing cells in vivo (39). BPZE1 produces PT in an enzymatically inactive form, suggesting that enzymatically inactive PT may still be able to induce IL-17 in vivo (39). Furthermore, human dendritic cells (DCs) infected with BPZE1 induce Th17 cells, whereas DCs treated with BpSM LPS promote a Th2-biased response (40). However, human DCs seem to respond differently to mouse cells, in that virulent B. pertussis does not induce Th2 cytokines but a solid Th1/Th17 response (41) and Th17 and Th1 immune responses both contribute to the immunity conferred by natural pertussis infection in nonhuman primates (42).
Depletion of IL-17A abolished the ability of BPZE1 to protect against RSV-induced pathology, suggesting that IL-17–producing lymphocytes are crucial in preventing viral pathology. The RSV NS1 protein is known to inhibit interferon, but also to block Th17 induction and reduce neutrophil recruitment and protective CD103+CD8+ T cells (43). Furthermore, Th17 responses have been shown to be beneficial in other types of viral airway inflammation (28) and in the defense against pneumonia, where IL-17 is crucial for antibody-independent protection against Pseudomonas aeruginosa (44).
It is possible that IL-17 has immunopathologic effects in RSV infection (45), but a recent human study linked IL-17 to recovery of children hospitalized with RSV (46) and the role of IL-17 in RSV disease seems to be highly dependent on context (47, 48). In our studies, the effects of IL-17 may be mediated by promoting the neutrophil responses. However, experiments involving human monocyte-derived DCs suggest direct effects on signal transduction in antigen-presenting cells, leading to differential priming of T cells (unpublished observations).
We have therefore shown that BPZE1 does not enhance RSV disease, but rather exerts a beneficial immunomodulatory effect on subsequent RSV infection. After priming with BPZE1 in the neonatal period, this beneficial effect lasts through to adulthood and the effects are dependent on the induction of IL-17. Because BPZE1 can be genetically manipulated to express additional antigens (49, 50), it may in the future be used as a live attenuated vaccine vector to deliver antigens from additional pathogens (including RSV), thus providing polyvalent protection against multiple respiratory infections by both specific and nonspecific immunologic effects.
Acknowledgments
Acknowledgment
The authors thank Dr. Lydia Durant and Dr. Jonathan Dodd for critical reading of the manuscript and Martijn J. Schuijs and Christa Benjamin for excellent technical help.
Footnotes
Supported by a grant from the European Commission under the grant agreement #201502 (ChildINNOVAC) and the Wellcome Trust (087805/Z/08/Z).
Author Contributions: C.S. and D.S. performed and analyzed all experiments. C.S., W.O., and P.J.O. designed and planned experiments. X.R., D.R., and C.L. designed and constructed the attenuated vaccine. C.S. and P.J.O. wrote the manuscript.
Originally Published in Press as DOI: 10.1164/rccm.201307-1227OC on November 21, 2013
This article has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Smyth RL, Openshaw PJ. Bronchiolitis. Lancet. 2006;368:312–322. doi: 10.1016/S0140-6736(06)69077-6. [DOI] [PubMed] [Google Scholar]
- 2.Blanken MO, Rovers MM, Molenaar JM, Winkler-Seinstra PL, Meijer A, Kimpen JLL, Bont L Dutch RSV Neonatal Network. Respiratory syncytial virus and recurrent wheeze in healthy preterm infants. N Engl J Med. 2013;368:1791–1799. doi: 10.1056/NEJMoa1211917. [DOI] [PubMed] [Google Scholar]
- 3.Mascart F, Hainaut M, Peltier A, Verscheure V, Levy J, Locht C. Modulation of the infant immune responses by the first pertussis vaccine administrations. Vaccine. 2007;25:391–398. doi: 10.1016/j.vaccine.2006.06.046. [DOI] [PubMed] [Google Scholar]
- 4.Mascart F, Verscheure V, Malfroot A, Hainaut M, Piérard D, Temerman S, Peltier A, Debrie A-S, Levy J, Del Giudice G, et al. Bordetella pertussis infection in 2-month-old infants promotes type 1 T cell responses. J Immunol. 2003;170:1504–1509. doi: 10.4049/jimmunol.170.3.1504. [DOI] [PubMed] [Google Scholar]
- 5.Antoine R, Alonso S, Raze D, Coutte L, Lesjean S, Willery E, Locht C, Jacob-Dubuisson F. New virulence-activated and virulence-repressed genes identified by systematic gene inactivation and generation of transcriptional fusions in Bordetella pertussis. J Bacteriol. 2000;182:5902–5905. doi: 10.1128/jb.182.20.5902-5905.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mielcarek N, Debrie A-S, Raze D, Bertout J, Rouanet C, Younes AB, Creusy C, Engle J, Goldman WE, Locht C. Live attenuated B. pertussis as a single-dose nasal vaccine against whooping cough. PLoS Pathog. 2006;2:e65. doi: 10.1371/journal.ppat.0020065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li R, Lim A, Phoon MC, Narasaraju T, Ng JKW, Poh WP, Sim MK, Chow VT, Locht C, Alonso S. Attenuated Bordetella pertussis protects against highly pathogenic influenza A viruses by dampening the cytokine storm. J Virol. 2010;84:7105–7113. doi: 10.1128/JVI.02542-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schnoeller C, Roux X, Sawant D, Raze D, Locht C, Olszewska W, Openshaw PJ.Live attenuated Bordetella pertussis vaccine candidate modulates viral lung inflammation. Presented at the British Society of Immunology Meeting, Liverpool, UK, December 6–12., 2010 [Google Scholar]
- 9.Schnoeller C, Roux X, Sawant D, Raze D, Locht C, Olszewska W, Openshaw PJ.Modulation of viral lung inflammation by live attenuated Bordetella pertussis vaccine. Presented at the World Immune Regulation Meeting IV, Davos, Switzerland, March 29–April 1,2010 [Google Scholar]
- 10.Menozzi FD, Gantiez C, Locht C. Identification and purification of transferrin- and lactoferrin-binding proteins of Bordetella pertussis and Bordetella bronchiseptica. Infect Immun. 1991;59:3982–3988. doi: 10.1128/iai.59.11.3982-3988.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee DCP, Harker JAE, Tregoning JS, Atabani SF, Johansson C, Schwarze J, Openshaw PJM. CD25+ natural regulatory T cells are critical in limiting innate and adaptive immunity and resolving disease following respiratory syncytial virus infection. J Virol. 2010;84:8790–8798. doi: 10.1128/JVI.00796-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Olszewska W, Ispas G, Schnoeller C, Sawant D, Van de Casteele T, Nauwelaers D, Van Kerckhove B, Roymans D, De Meulder M, Rouan MC, et al. Antiviral and lung protective activity of a novel respiratory syncytial virus fusion inhibitor in a mouse model. Eur Respir J. 2011;38:401–408. doi: 10.1183/09031936.00005610. [DOI] [PubMed] [Google Scholar]
- 13.Mielcarek N, Debrie A-S, Mahieux S, Locht C. Dose response of attenuated Bordetella pertussis BPZE1-induced protection in mice. Clin Vaccine Immunol. 2010;17:317–324. doi: 10.1128/CVI.00322-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Harker JA, Godlee A, Wahlsten JL, Lee DCP, Thorne LG, Sawant D, Tregoning JS, Caspi RR, Bukreyev A, Collins PL, et al. Interleukin 18 coexpression during respiratory syncytial virus infection results in enhanced disease mediated by natural killer cells. J Virol. 2010;84:4073–4082. doi: 10.1128/JVI.02014-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Loving CL, Brockmeier SL, Vincent AL, Palmer MV, Sacco RE, Nicholson TL. Influenza virus coinfection with Bordetella bronchiseptica enhances bacterial colonization and host responses exacerbating pulmonary lesions. Microb Pathog. 2010;49:237–245. doi: 10.1016/j.micpath.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 16.Didierlaurent A, Goulding J, Patel S, Snelgrove R, Low L, Bebien M, Lawrence T, van Rijt LS, Lambrecht BN, Sirard J-C, et al. Sustained desensitization to bacterial Toll-like receptor ligands after resolution of respiratory influenza infection. J Exp Med. 2008;205:323–329. doi: 10.1084/jem.20070891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yamaguchi Y, Harker JA, Wang B, Openshaw PJ, Tregoning JS, Culley FJ. Preexposure to CpG protects against the delayed effects of neonatal respiratory syncytial virus infection. J Virol. 2012;86:10456–10461. doi: 10.1128/JVI.01082-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Abu Raya B, Bamberger E, Kassis I, Kugelman A, Srugo I, Miron D. Bordetella pertussis infection attenuates clinical course of acute bronchiolitis. Pediatr Infect Dis J. 2013;32:619–621. doi: 10.1097/INF.0b013e3182877973. [DOI] [PubMed] [Google Scholar]
- 19.Dong W, Selgrade MK, Gilmour MI. Systemic administration of Bordetella pertussis enhances pulmonary sensitization to house dust mite in juvenile rats. Toxicol Sci. 2003;72:113–121. doi: 10.1093/toxsci/kfg015. [DOI] [PubMed] [Google Scholar]
- 20.Ennis DP, Cassidy JP, Mahon BP. Prior Bordetella pertussis infection modulates allergen priming and the severity of airway pathology in a murine model of allergic asthma. Clin Exp Allergy. 2004;34:1488–1497. doi: 10.1111/j.1365-2222.2004.02042.x. [DOI] [PubMed] [Google Scholar]
- 21.Sigurs N, Aljassim F, Kjellman B, Robinson PD, Sigurbergsson F, Bjarnason R, Gustafsson PM. Asthma and allergy patterns over 18 years after severe RSV bronchiolitis in the first year of life. Thorax. 2010;65:1045–1052. doi: 10.1136/thx.2009.121582. [DOI] [PubMed] [Google Scholar]
- 22.Kavanagh H, Noone C, Cahill E, English K, Locht C, Mahon BP. Attenuated Bordetella pertussis vaccine strain BPZE1 modulates allergen-induced immunity and prevents allergic pulmonary pathology in a murine model. Clin Exp Allergy. 2010;40:933–941. doi: 10.1111/j.1365-2222.2010.03459.x. [DOI] [PubMed] [Google Scholar]
- 23.Li R, Cheng C, Chong SZ, Lim ARF, Goh YF, Locht C, Kemeny DM, Angeli V, Wong WSF, Alonso S. Attenuated Bordetella pertussis BPZE1 protects against allergic airway inflammation and contact dermatitis in mouse models. Allergy. 2012;67:1250–1258. doi: 10.1111/j.1398-9995.2012.02884.x. [DOI] [PubMed] [Google Scholar]
- 24.Openshaw PJ, Culley FJ, Olszewska W. Immunopathogenesis of vaccine-enhanced RSV disease. Vaccine. 2001;20:S27–S31. doi: 10.1016/s0264-410x(01)00301-2. [DOI] [PubMed] [Google Scholar]
- 25.Cyr SL, Angers I, Guillot L, Stoica-Popescu I, Lussier M, Qureshi S, Burt DS, Ward BJ. TLR4 and MyD88 control protection and pulmonary granulocytic recruitment in a murine intranasal RSV immunization and challenge model. Vaccine. 2009;27:421–430. doi: 10.1016/j.vaccine.2008.10.073. [DOI] [PubMed] [Google Scholar]
- 26.Lin AM, Rubin CJ, Khandpur R, Wang JY, Riblett M, Yalavarthi S, Villanueva EC, Shah P, Kaplan MJ, Bruce AT. Mast cells and neutrophils release IL-17 through extracellular trap formation in psoriasis. J Immunol. 2011;187:490–500. doi: 10.4049/jimmunol.1100123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kumar V, Sharma A. Neutrophils: Cinderella of innate immune system. Int Immunopharmacol. 2010;10:1325–1334. doi: 10.1016/j.intimp.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 28.McKinstry KK, Strutt TM, Buck A, Curtis JD, Dibble JP, Huston G, Tighe M, Hamada H, Sell S, Dutton RW, et al. IL-10 deficiency unleashes an influenza-specific Th17 response and enhances survival against high-dose challenge. J Immunol. 2009;182:7353–7363. doi: 10.4049/jimmunol.0900657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen L, Watanabe T, Watanabe H, Sendo F. Neutrophil depletion exacerbates experimental Chagas’ disease in BALB/c, but protects C57BL/6 mice through modulating the Th1/Th2 dichotomy in different directions. Eur J Immunol. 2001;31:265–275. doi: 10.1002/1521-4141(200101)31:1<265::AID-IMMU265>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 30.Tateda K, Moore TA, Newstead MW, Tsai WC, Zeng X, Deng JC, Chen G, Reddy R, Yamaguchi K, Standiford TJ. Chemokine-dependent neutrophil recruitment in a murine model of Legionella pneumonia: potential role of neutrophils as immunoregulatory cells. Infect Immun. 2001;69:2017–2024. doi: 10.1128/IAI.69.4.2017-2024.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Halfhide CP, Brearey SP, Flanagan BF, Hunt JA, Howarth D, Cummerson J, Edwards S, Hart CA, Smyth RL. Neutrophil TLR4 expression is reduced in the airways of infants with severe bronchiolitis. Thorax. 2009;64:798–805. doi: 10.1136/thx.2008.107821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nembrini C, Marsland BJ, Kopf M.IL-17-producing T cells in lung immunity and inflammation J Allergy Clin Immunol 2009123986–994.quiz 995–996 [DOI] [PubMed] [Google Scholar]
- 33.Loebbermann J, Schnoeller C, Thornton H, Durant L, Sweeney NP, Schuijs M, O’Garra A, Johansson C, Openshaw PJ. IL-10 regulates viral lung immunopathology during acute respiratory syncytial virus infection in mice. PLoS ONE. 2012;7:e32371. doi: 10.1371/journal.pone.0032371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sun J, Cardani A, Sharma AK, Laubach VE, Jack RS, Müller W, Braciale TJ. Autocrine regulation of pulmonary inflammation by effector T-cell derived IL-10 during infection with respiratory syncytial virus. PLoS Pathog. 2011;7:e1002173. doi: 10.1371/journal.ppat.1002173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Weiss KA, Christiaansen AF, Fulton RB, Meyerholz DK, Varga SM. Multiple CD4+ T cell subsets produce immunomodulatory IL-10 during respiratory syncytial virus infection. J Immunol. 2011;187:3145–3154. doi: 10.4049/jimmunol.1100764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.McGuirk P, McCann C, Mills KHG. Pathogen-specific T regulatory 1 cells induced in the respiratory tract by a bacterial molecule that stimulates interleukin 10 production by dendritic cells: a novel strategy for evasion of protective T helper type 1 responses by Bordetella pertussis. J Exp Med. 2002;195:221–231. doi: 10.1084/jem.20011288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Loebbermann J, Thornton H, Durant L, Sparwasser T, Webster KE, Sprent J, Culley FJ, Johansson C, Openshaw PJ. Regulatory T cells expressing granzyme B play a critical role in controlling lung inflammation during acute viral infection. Mucosal Immunol. 2012;5:161–172. doi: 10.1038/mi.2011.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fedele G, Bianco M, Debrie A-S, Locht C, Ausiello CM. Attenuated Bordetella pertussis vaccine candidate BPZE1 promotes human dendritic cell CCL21-induced migration and drives a Th1/Th17 response. J Immunol. 2011;186:5388–5396. doi: 10.4049/jimmunol.1003765. [DOI] [PubMed] [Google Scholar]
- 39.Andreasen C, Powell DA, Carbonetti NH. Pertussis toxin stimulates IL-17 production in response to Bordetella pertussis infection in mice. PLoS ONE. 2009;4:e7079. doi: 10.1371/journal.pone.0007079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fedele G, Celestino I, Spensieri F, Frasca L, Nasso M, Watanabe M, Remoli ME, Coccia EM, Altieri F, Ausiello CM. Lipooligosaccharide from Bordetella pertussis induces mature human monocyte-derived dendritic cells and drives a Th2 biased response. Microbes Infect. 2007;9:855–863. doi: 10.1016/j.micinf.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 41.Higgs R, Higgins SC, Ross PJ, Mills KHG. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunol. 2012;5:485–500. doi: 10.1038/mi.2012.54. [DOI] [PubMed] [Google Scholar]
- 42.Warfel JM, Merkel TJ. Bordetella pertussis infection induces a mucosal IL-17 response and long-lived Th17 and Th1 immune memory cells in nonhuman primates. Mucosal Immunol. 2013;6:787–796. doi: 10.1038/mi.2012.117. [DOI] [PubMed] [Google Scholar]
- 43.Munir S, Hillyer P, Le Nouën C, Buchholz UJ, Rabin RL, Collins PL, Bukreyev A. Respiratory syncytial virus interferon antagonist NS1 protein suppresses and skews the human T lymphocyte response. PLoS Pathog. 2011;7:e1001336. doi: 10.1371/journal.ppat.1001336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Priebe GP, Walsh RL, Cederroth TA, Kamei A, Coutinho-Sledge YS, Goldberg JB, Pier GB. IL-17 is a critical component of vaccine-induced protection against lung infection by lipopolysaccharide-heterologous strains of Pseudomonas aeruginosa. J Immunol. 2008;181:4965–4975. doi: 10.4049/jimmunol.181.7.4965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mukherjee S, Lindell DM, Berlin AA, Morris SB, Shanley TP, Hershenson MB, Lukacs NW. IL-17-induced pulmonary pathogenesis during respiratory viral infection and exacerbation of allergic disease. Am J Pathol. 2011;179:248–258. doi: 10.1016/j.ajpath.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Faber TE, Groen H, Welfing M, Jansen KJG, Bont LJ. Specific increase in local IL-17 production during recovery from primary RSV bronchiolitis. J Med Virol. 2012;84:1084–1088. doi: 10.1002/jmv.23291. [DOI] [PubMed] [Google Scholar]
- 47.Openshaw PJ, Chiu C. Protective and dysregulated T cell immunity in RSV infection. Curr Opin Virol. 2013;3:468–474. doi: 10.1016/j.coviro.2013.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Habibi MS, Openshaw PJM. Benefit and harm from immunity to respiratory syncytial virus: implications for treatment. Curr Opin Infect Dis. 2012;25:687–694. doi: 10.1097/QCO.0b013e32835a1d92. [DOI] [PubMed] [Google Scholar]
- 49.Ho SY, Chua SQ, Foo DGW, Locht C, Chow VT, Poh CL, Alonso S. Highly attenuated Bordetella pertussis strain BPZE1 as a potential live vehicle for delivery of heterologous vaccine candidates. Infect Immun. 2008;76:111–119. doi: 10.1128/IAI.00795-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kammoun H, Roux X, Raze D, Debrie A-S, De Filette M, Ysenbaert T, Mielcarek N, Saelens X, Fiers W, Locht C. Immunogenicity of live attenuated B. pertussis BPZE1 producing the universal influenza vaccine candidate M2e. PLoS ONE. 2013;8:e59198. doi: 10.1371/journal.pone.0059198. [DOI] [PMC free article] [PubMed] [Google Scholar]