Abstract
Objectives
Blood culture contamination is a common and preventable problem in the emergency department (ED). In a previous single-center study, changing the process of ED blood culture collection from the traditional “clean,” nonsterile procedure to a fully sterile procedure with standardized use of sterile gloves, large volume chlorhexidine skin antisepsis, and fenestrated sterile drapes resulted in a substantial reduction in contamination. The objective of the current study was to evaluate the effectiveness of this sterile blood culture collection process for reducing blood culture contamination in two community hospital EDs.
Methods
The authors implemented the sterile blood culture collection process in the ED of two hospitals, including Hospital A, which historically had a contamination rate of approximately 5%, and Hospital B, with a 2.5% historical contamination rate. Using an interrupted times series design and segmented regression analysis to adjust for secular trends and autocorrelation, the monthly percentages of cultures contaminated at each hospital during an intervention period (sterile technique) were compared to a 10-month baseline period immediately preceding implementation (clean technique). At Hospital A, the full sterile blood culture collection process was used throughout the 16-month intervention period. At Hospital B, user feedback indicated poor compliance due to difficulty implementing the fenestrated drape component; therefore, the process was simplified to the modified sterile collection process, in which the fenestrated drape component was dropped and sterile gloves and large volume skin antisepsis were emphasized. Hence, at Hospital B, two intervention periods were compared to the baseline period—the 8-month intervention period 1 (full sterile process) and the subsequent 8-month intervention period 2 (modified sterile process).
Results
Hospital A: During the baseline period 165 / 3417 (4.83%) cultures were contaminated, while 142 / 5238 (2.71%) were contaminated during the intervention period (p < 0.01). In the segmented regression model, the full sterile blood culture collection process was associated with an immediate 2.68% (95% confidence interval [CI]: 1.43% to 3.52%) absolute reduction in contamination and sustained reductions during the entire intervention period. Hospital B: During the baseline, 63/2509 (2.51%) cultures were contaminated. In intervention period 1 with the full sterile process, 51/1865 (2.73%) cultures were contaminated (p=0.65), with segmented regression results showing no changes compared to baseline. After simplification of the process to address poor compliance, the modified sterile process during intervention period 2 was associated with a significant reduction in contamination, with 17/1860 (0.91%) cultures contaminated (p<0.01 compared to baseline). The segmented regression model demonstrated the modified sterile process was associated with an immediate 1.53% (95% CI: 1.00% to 1.88%) absolute reduction in contamination with significant sustained reductions.
Conclusions
Changing the method of blood culture collection from the commonly-used non-sterile technique to a sterile process resulted in significant reductions in blood culture contamination at two community hospital EDs, including one with low baseline contamination. Monitoring the implementation process at both sites was important to identify and overcome operational challenges. At one study site, simplification of the process by removing the fenestrated drape component was a key for successful implementation.
Introduction
Blood cultures are commonly performed in the emergency department (ED) due to the important diagnostic and prognostic information they can provide clinicians.1 Unfortunately, false positive blood culture results due to specimen contamination with skin bacteria are also common in the ED.2–7 These contaminated blood cultures have significant negative consequences for individual patients and the overall healthcare system, because clinicians respond to these false positive tests by initiating diagnostic studies and treatments that would otherwise not have been pursued.8–14 Examples of patient morbidity caused by contaminated blood cultures include increased hospital length of stay, unnecessary ED visits, and unneeded tests and procedures, such as echocardiograms and central line insertions.9 The Clinical and Laboratory Standards Institute recommends a national benchmark for each healthcare institution to maintain a contamination rate < 3%.15
Blood cultures are frequently collected in the ED, because patients with acute infections often undergo initial, pre-antibiotic testing in this setting.4,6,9 The proportion of contaminated blood cultures is frequently higher in the ED compared to other healthcare settings, likely due to variability in techniques used for specimen collection, relatively high turnover of staff in the ED, and the need to rapidly collect cultures prior to an initial dose of antibiotics.2,4,6,9 Therefore, interventions that specifically address the increased vulnerability of the ED for blood culture contamination can have a profound impact on reducing contamination overall within the healthcare system.
Self et al2 developed a new process for collecting blood culture specimens using a fully sterile procedure with standardized use of sterile gloves, skin antisepsis with 3 ml of a chorhexidine-based skin wash, creation of a sterile field with a fenestrated drape, and a procedural checklist. Implementation of this sterile blood culture collection process in the tertiary-care, academic hospital ED where it was developed resulted in immediate and sustained reduction in blood culture contamination compared to the historically high levels of contamination previously seen in that ED.2
Following the success of that single-center study, important questions about generalizability were raised, including: (1) Can the sterile blood culture collection process reduce contamination in other EDs not under direct supervision of the quality improvement team that developed it? (2) Does the sterile collection process need to be simplified for successful use in community hospital EDs? And (3) can the sterile collection process reduce contamination to very low levels (< 1%) in EDs that have maintained contamination < 3% using usual care, nonsterile collection techniques? In this study, we sought to answers these questions by implementing the sterile blood culture collection process in two community hospital EDs and measuring its effectiveness at reducing contamination.
Methods
Study Design
This study evaluated the effectiveness of a quality improvement intervention at two study sites using an interrupted time series design.16–17 The sterile blood culture collection process was implemented in the EDs of Hospital A and Hospital B, where traditional, non-sterile techniques were used to collect cultures prior to this study. The impact of this process change was evaluated by comparing the monthly percentages of contaminated blood cultures before and after implementation of the sterile blood culture collection process using segmented regression analysis.18–19 The governing Institutional Review Boards of the participating hospitals approved this study with waiver of informed consent. Research funding was provided by the Society for Academic Emergency Medicine, Emergency Medicine Patient Safety Foundation, and the National Center for Advancing Translational Sciences. CareFusion, Inc. provided Chloraprep® devices for use during the study. These funding sources had no role in the conduct of the study or interpretation of its results.
Study Setting and Population
The study was conducted in the EDs of two community hospitals in the United States (Hospital A and Hospital B). ED and hospital leadership at both sites volunteered for participation in this study with a goal of reducing blood culture contamination in their EDs. The study team included quality improvement leaders from the hospitals’ management company and a collaborating academic medical center, as well as local improvement teams at both hospitals.
Hospital A is a suburban community hospital with approximately 350 in-patient beds, 34,000 annual ED visits and an average of 350 blood cultures collected monthly in the ED. Prior to this study, ED nurses and laboratory phlebotomy staff collected blood cultures using non-sterile techniques, including non-sterile gloves, skin anti-sepsis with a 0.67 ml solution of 2% chlorhexidine gluconate/70% isopropyl alcohol, and no preparation of distinct sterile fields. Historically, approximately 5% of blood cultures collected in this ED were contaminated with skin bacteria. Repeated efforts to reduce contamination by educating nurses and phlebotomists about skin antisepsis failed to result in sustained improvement. The local quality improvement team for this study included the ED nursing director, ED nurse educator, laboratory manager, and ED Medical Staff Director. The study team’s goal was to use the sterile collection process to decrease the rate of contamination below the 3% benchmark.
Hospital B is located in a small city and has approximately 220 in-patient beds, 27,000 annual ED visits and 250 blood cultures collected monthly in the ED. Before this study, ED nurses and laboratory phlebotomists collected cultures in the ED. Typically, nurses collected blood for the first set of blood cultures at the time of peripheral intravenous catheter placement and phlebotomists collected blood for subsequent cultures later during the patient’s ED stay. Both nurses and phlebotomists used similar collection techniques, including non-sterile gloves, skin antisepsis with a 1.5 ml solution of 2% chlorhexidine gluconate/70% isopropyl alcohol, and no sterile draping to create procedural fields. Historically, the contamination rate was approximately 2.5%, below the 3% benchmark. The local study team included the ED nursing manager and laboratory manager. The study team sought to reduce contamination below 1% with implementation of the sterile blood culture collection process.
Study Protocol
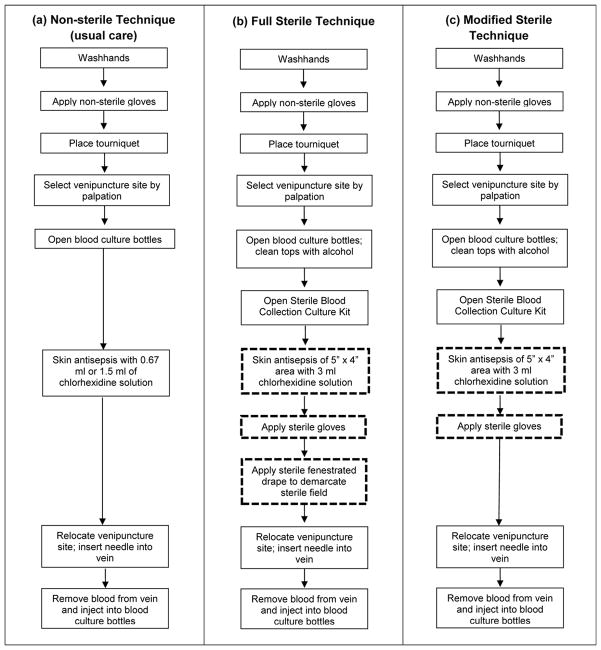
We evaluated the impact of changing the blood culture collection technique from the usual care, non-sterile methods historically used at each study site to standardized sterile blood culture collection techniques (Figure 1). Palpation of a venipuncture site with non-sterile hands after skin antisepsis has been identified as a likely cause of contamination using usual care techniques.2 The sterile collection process allows palpation within a sterile field to identify veins for venipuncture with minimal risk of introducing skin contaminants.2 This sterile technique has been previously described2 and includes application of sterile gloves and creation of a large sterile field with skin antisepsis using a 3 ml solution of 2% chlorhexidine gluconate/70% isopropyl alcohol (Chloraprep®; CareFusion, San Diego, CA) and a fenestrated sterile drape. Sterile blood culture collection kits (packaged by LSL Industries, Inc.; Chicago, IL) were stocked at the participating sites throughout the study period. These kits contained materials needed to perform blood culture collection using the sterile technique, including the skin antisepsis device, fenestrated drape, butterfly needle and checklist outlining the procedure.
Figure 1.
Description of blood culture specimen collection techniques, including (a) non-sterile technique used during the baseline periods; (b) full sterile technique; and (c) modified sterile technique. Key steps that are different among the techniques are highlighted with bold, dashed outlines.
Study team members visited both participating sites during the implementation process. During these site visits, local leaders were trained on the sterile blood culture collection process. These local leaders then trained the nurses and phlebotomists who collected blood cultures in the ED. Training methods included videos demonstrating the technique, hands-on workshops, and interactive question-and-answer sessions. Methods to regularly stock the sterile blood culture kits, monitor blood culture contamination, and elicit feedback about the program from its front-line users were established at each site.
Consistent with quality improvement methodology,20–21 ED and laboratory personnel at the participating sites were aware of the ongoing study throughout the intervention periods and the intervention was designed to be adaptable based on real-time feedback from these frontline workers. Contamination rates were consistently shared by the local quality improvement teams with their nurses and phlebotomists. When blood culture contamination increased, the study team investigated potential causes and considered changes to the intervention to improve its performance in the local setting. The intervention was adapted one time during the study at Hospital B. Approximately 8 months after implementation of the sterile blood culture collection process at Hospital B, there had been no significant change in contamination compared to pre-implementation levels. Feedback from nurses and phlebotomists revealed that compliance with the sterile technique was low because use of the fenestrated drape was thought to be too cumbersome in their setting. Therefore, the study team simplified the collection process at this site by eliminating the fenestrated drape component and focusing on consistently using sterile gloves and performing meticulous skin antisepsis with 3 ml chlorhexidine devices. The simplified process, called the Modified Sterile Collection Process, was implemented at Hospital B only (Figure 1C). Implementation involved hands-on training with each nurse and phlebotomists on performing sterile collection without a drape.
Study Periods
The study spanned from January 2011 through March 2013 at both sites. At Hospital A, local leaders calculated contamination percentages monthly and the study period was divided into 27 months, with the monthly percentage of cultures contaminated serving as the primary outcome for analysis. During the ten months prior to implementation (January–October 2011), usual care, non-sterile techniques were used to collect cultures; this was defined as the baseline period for the study (Figure 2). During November 2011, the study team completed its on-site visit and local providers were trained on the sterile technique; November 2011 was considered a transition phase and not included in either the pre-implementation baseline period or post-implementation intervention period. The full sterile blood culture collection process was completely implemented in December 2011 and was used to collect blood cultures for the 16-month intervention period, December 2011 through March 2013.
Figure 2.
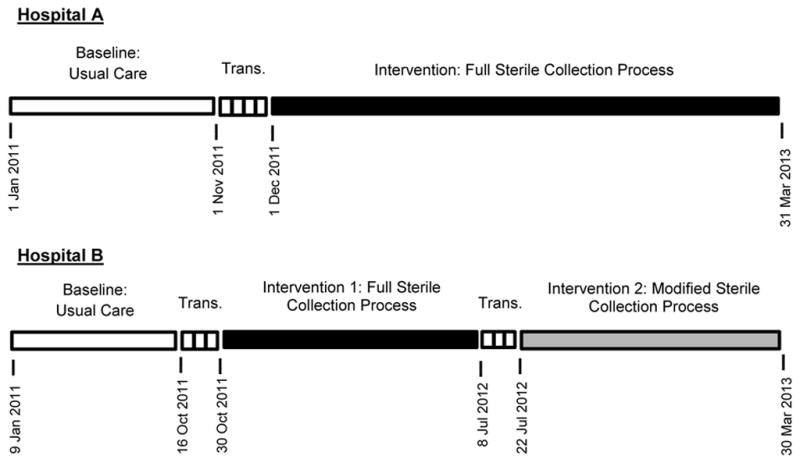
Definition of study periods at both study sites, Hospital A and Hospital B. Trans: transition phase.
At Hospital B, local leaders calculated contamination percentages weekly. In order to more closely match the monthly time intervals for the data at Hospital A, the study period at Hospital B was divided into 4-week blocks with the percentage of cultures contaminated during these 4-week blocks serving as the primary outcome. During the baseline period, which included ten 4-week blocks from January through mid-October 2011, blood cultures were collected with usual care, non-sterile techniques (Figure 2). Implementation of the full sterile blood culture collection process occurred during a 2-week transition phase in October 2011. The following nine 4-week blocks from November 2011 through early July 2012 were considered Intervention Period 1. Because of reported low compliance with the original intervention, the modified sterile collection process was introduced and implemented during a second 2-week transition phase in July 2012. Then, the modified process was used during Intervention Period 2, which spanned from late July 2012 through March 2013.
Outcome Measures
The primary outcome was the percentage of blood cultures contaminated in each participating ED. A blood culture was classified as contaminated if one or more of the following organisms grew in only one culture in a series of blood cultures collected from the same patient during a single ED visit: Aerococcus species, α-hemolytic Streptococcus, Bacillus species except anthracis, coagulase negative Staphylococcus species except lugdunensis, Corynebacterium species, Micrococcus species, and Propionibacterium species.22
Data Analysis
We evaluated the effectiveness of the sterile blood culture interventions to reduce blood culture contamination separately at each study site using segmented linear regression analysis.18–19 This is a statistical technique to evaluate the immediate and sustained impact of a change in a process while accounting for secular trends over time. At Hospital A, we compared the percentage of blood culture contaminated after implementation of the full sterile collection process (intervention period) to the baseline period. At Hospital B, we compared both Intervention Period 1 (full sterile collection process) and Intervention Period 2 (modified sterile collection process) to the baseline period. Transition phases were not included in statistical comparisons.
In initial before-after analyses, percentages of cultures contaminated during each intervention period were compared to the baseline period at the same hospital using the Chi-squared test. Next, we built three segmented linear regression models to compare each intervention period to the baseline period: (1) Hospital A intervention period compared to Hospital A baseline; (2) Hospital B intervention period 1 compared to Hospital B baseline; and (3) Hospital B intervention period 2 compared to Hospital B baseline. The dependent variable for the models was the percentage of cultures contaminated during monthly intervals at Hospital A and 4-week intervals at Hospital B. The dependent variable underwent natural log transformation to stabilize variances of these terms. Independent variables included study period (baseline, intervention 1, intervention 2) and secular trends during the baseline period and intervention period being evaluated. Terms for secular trends were included to evaluate the impact of the intervention independent of time trends in the data. Since error terms of consecutive observations could be correlated, we accounted for first order autocorrelation using autoregressive integrated moving average (ARIMA) models.19 Confidence intervals for independent variables in the models were calculated using robust standard errors.
To quantify the impact of the interventions, we compared contamination percentages observed during the intervention periods to percentages expected during the same time period assuming no intervention had been implemented.18–19 The baseline period trend line was projected into the intervention periods to make these comparisons. We specifically quantified the absolute and relative impact of the interventions at the first and last time point during each intervention period.
For power calculations, we used historical blood culture data at each site, the chi-square test for before-after comparisons, and a familywise type I error of 0.05. We assumed 350 monthly cultures would be collected at Hospital A and 5% of these cultures would be contaminated during the baseline period. Our goal was to detect a change in contamination to less than 3% during the intervention period. Using these estimates, the baseline period and intervention period each needed to be at least 6 months long for 90% power to detect the desired detectable difference. A single comparison (intervention period to baseline period) was made at Hospital A, and a two-sided p-value < 0.05 was considered significant. At Hospital B, we assumed 250 cultures would be collected during each 4-week block and 2.5% of cultures would be contaminated during the baseline period. Our goal at Hospital B was to detect a change in contamination to less than 1%. At least seven 4-week blocks were needed during the baseline period and intervention period for 90% power to detect this change. At Hospital B, two comparisons were made (intervention period 1 to baseline, and intervention period 2 to baseline); in order to maintain a familywise type 1 error probability of 0.05, we corrected the significance level for each pairwise comparison with a Bonferroni adjustment, such that a p-value < 0.025 was considered significant for each pair-wise comparison.
Power analyses were performed with PS software.23 Other statistical analyses were conducted with Stata/IC 12.0 (College Station, TX).
Results
Hospital A
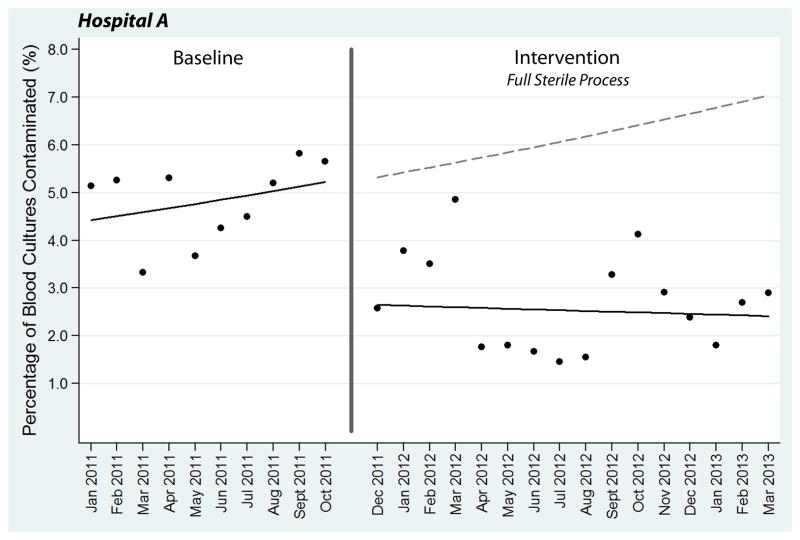
Using usual care collection techniques during the baseline period at Hospital A, 165 (4.83%) of 3,417 total blood cultures were contaminated. Comparatively, only 142 (2.71%) of 5,238 cultures were contaminated during the intervention period after implementation of the full sterile blood culture collection process (p < 0.01). In the segmented regression model, implementation of the sterile collection process was associated with an immediate absolute 2.68% (95% confidence interval [CI]: 1.43% to 3.52%) reduction and relative 50.25% (26.88% to 66.14%) reduction in contamination (Figure 3). At the end of the study period in March 2013, the sterile collection process was associated with an absolute 4.62% (95% CI: 1.87% to 5.91%) and relative 65.70% (95% CI: 26.61% to 83.96%) reduction in contamination compared to projected estimates from the baseline period (Figure 3). These reductions are represented in Figure 3 by the difference between the solid regression line in the intervention period and dotted projected line extending from the baseline period.
Figure 3.
Segmented regression analysis of blood culture contamination at Hospital A. Each data point represents the percentage of blood cultures contaminated during a single month. The transition phase in November 2011 is denoted by a vertical line. Solid lines represent best-fit linear regression lines, while the dotted line is the baseline regression line projected into the intervention period.
Hospital B
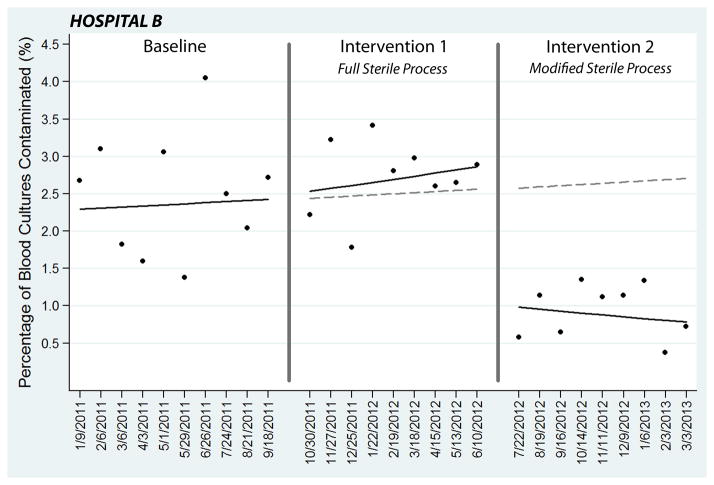
At Hospital B, 63 (2.51%) of 2,509 blood cultures collected during the baseline period with usual care techniques were contaminated. During intervention period 1 after implementation of the full sterile collection process, 51 (2.73%) of 1,865 cultures were contaminated (p=0.65). The segmented regression model comparing intervention period 1 to baseline demonstrated no significant change in contamination, with an absolute 0.30% (95% CI: −0.83% to 2.16%) change in contamination at the end of intervention period 1 period compared to projected estimates from the baseline period (Figure 4).
Figure 4.
Segmented regression analysis of blood culture contamination at Hospital B. Each data point represents the percentage of blood cultures contaminated during a 4-week block. Two-week transition phases between the periods are denoted with vertical lines. Solid lines represent best-fit linear regression lines. The dotted lines are the baseline period regression line projected into intervention period 1 and intervention period 2.
In July 2012, the fenestrated draping component of the intervention was removed, and the modified sterile blood culture collection process was introduced with renewed emphasis on sterile glove use and meticulous skin antisepsis. With this modified process, 17 of 1,860 (0.91%) cultures collected during intervention period 2 were contaminated (p < 0.01, comparing intervention period 2 to baseline). In the segmented regression model comparing intervention period 2 and the baseline period, implementation of the modified sterile collection process was associated with a significant reduction in contamination, including a 1.53% (95% CI: 1.00% to 1.88%) absolute reduction at the first point in intervention period 2, corresponding to a 59.48% (95% CI: 38.90% to 73.12%) relative reduction (Figure 4). At the last time point of intervention period 2, the intervention was associated with a 1.87% (95% CI: 1.01 to 2.29) absolute and 69.00% (95% CI: 37.41 to 84.64) relative reduction compared to projected estimates from the baseline.
Discussion
By converting the process of blood culture collection from a clean, non-sterile procedure to a sterile technique, we significantly reduced blood culture contamination in two community hospital EDs.
Hospital A historically had contamination percentages of approximately 5%, which was higher than the national median and 3% benchmark.15,22 The goal at Hospital A was to use the sterile blood culture collection intervention to consistently maintain contamination < 3% by the end of the study. Immediately after implementation of the full sterile blood culture collection process, contamination percentages fell, but remained in the 3% to 5% range for four months (Figure 3). At this point, leaders at Hospital A refocused on reducing contamination by retraining all personnel who collected blood cultures in the ED on the sterile collection process and ensuring the sterile blood culture kits were readily available in the ED. Study leaders discussed options for changing the intervention at this point, but decided to continue with the full sterile blood culture collection process with increased emphasis on compliance through efforts of local quality improvement leaders. These efforts were very effective. Beginning with the fifth month after implementation (April 2012), contamination percentages were < 3% during 10 of the next 12 months.
Hospital B historically maintained contamination at approximately 2.5% in the ED. Despite this level being lower than the national 3% benchmark, ED and hospital leaders believed they could reduce contamination even lower, and set an ambitious goal of 1% contamination. Implementation of the full sterile blood culture process did not have a significant effect on contamination (Figure 4). We explored why this process change did not lead to reduced contamination through a series of interviews with the nurses and phlebotomists who collected cultures. They expressed reluctance to use a fenestrated sterile drape for each blood draw, viewing this as an unnecessary and time-consuming step when contamination rates had been “acceptable” using the old, faster methods. The local clinicians and study team suspected compliance was poor with all components of the sterile collection process due to resistance to the fenestrated drape component. Therefore, approximately 8 months after initial implementation of the full sterile blood culture collection process (July 2012), the study team and local leaders modified the intervention by removing the fenestrated drape and reemphasized use of sterile gloves and meticulous, large-volume skin antisepsis. This modified sterile blood culture technique led to an immediate and sustained reduction in contamination with monthly rates < 1.5% and near the 1% goal for nine consecutive months (Figure 4).
Results of this study combined with previous work suggest implementation of sterile blood culture collection techniques can reduce ED blood culture contamination in many settings, including large academic hospitals,2 pediatric hospitals,4 and community hospitals with both high (Hospital A in this study) and low (Hospital B in this study) baseline contamination.
Success of sterile blood culture collection at Hospital B only after simplification of the process based on feedback from frontline users suggests that continuous monitoring and communication with the local implementation team are important the success of this process change. Furthermore, this suggests the fenestrated drape may not be an essential component of the process. After the success at Hospital B using the modified sterile process without the fenestrated drape, we discussed the potential of similar modifications at Hospital A. However, leaders at Hospital A preferred to continue with the full sterile process, believing that the drape helped focus nurses and phlebotomists on maintaining sterility and was important at their site, given their history of very high contamination rates and initial success with the full sterile process. We believe both the full sterile technique and modified technique are viable strategies. Future work will focus on quantifying the impact of specific components of the sterile process, including the fenestrated drape. For EDs seeking to reduce blood culture contamination, a pragmatic approach may be to develop a local sterile blood culture collection protocol that maintains the central concept described here—creation of sterile fields during blood culture collection to enable nurses to palpate veins with minimal risk of introducing contamination—while modifying specific elements of the process based on local experience to facilitate enthusiasm for change and adherence to the new process.
Limitations
In this quality improvement study, we evaluated the effect of a multifaceted improvement bundle. Using this design, we were not able to separately evaluate the impact of individual components of the bundle, such as the use of sterile gloves or specific skin antisepsis devices. Success of the modified sterile process at Hospital B suggests the fenestrated drape component of the process may be unnecessary and potentially interfere with implementation in some settings. Further study is needed to understand the utility of including the fenestrated drape in the process. Administrative leaders and the ED staff at both participating hospitals volunteered for this study and were highly motivated to improve their contamination rates. This internal motivation was likely a key component to reducing contamination and the results of this study may not be generalizable to other settings without supportive leadership or enthusiasm for change. We gauged adherence with the new process for blood culture collection via frequent communication with the nurses and phlebotomists who collected the cultures. Direct observation of blood culture collection by the study team would have provided more precise measurements of adherence, but was not feasible in this study due to the nature of ED blood culture collection occurring 24 hours per day, seven days a week. Similar to previous blood culture studies,2,14,22 we used a “series definition” for contamination with a culture classified as contaminated if only one culture within a series collected from the same patient grew the same skin contaminant organism. This definition may overestimate or underestimate the true contamination rate.
Conclusions
Implementing sterile techniques for blood culture collection resulted in significant reductions in specimen contamination at two community hospital EDs. Changing the procedure for blood culture collection from commonly-used non-sterile methods to a sterile process is feasible and can be effective at reducing contamination in busy community hospital EDs, even in departments with baseline contamination levels less than the 3% benchmark. A simplified sterile collection process that does not include fenestrated draping is an alternative to the full sterile technique and was successful at reducing contamination at one site that had difficulty implementing the full sterile process. Real-time evaluation of the implementation process, feedback from end-users, and adaptation of the intervention to overcome local barriers of implementation appear to be important components to successfully reducing blood culture contamination using a standardized sterile collection process.
Acknowledgments
Funding:
Supported by the 2011 Society for Academic Emergency Medicine / Emergency Medicine Patient Safety Foundation Patient Safety Research Fellowship Grant (WHS).
Supported by a research grant from the National Center for Advancing Translational Sciences [grant number KL2 TR000446 to WHS].
Research supplies provided by CareFusion, Inc.
Footnotes
Potential Conflicts of Interest:
WHS and TRT report submitting a US patent for a blood culture collection device and process. CareFusion, the makers of Chloraprep®, provided materials for use in this study.
References
- 1.Weinstein MP, Towns ML, Quartey SM, Mirrett S, Reimer LG, Parmigiani G. The clinical significance of positive blood cultures in the 1990s: A prospective comprehensive evaluation of the microbiology, epidemiology and outcome of bacteremia and fungemia in adults. Clin Infect Dis. 1997;24:584–602. doi: 10.1093/clind/24.4.584. [DOI] [PubMed] [Google Scholar]
- 2.Self WH, Speroff T, Grijalva CG, McNaughton CD, Ashburn J, Liu D, et al. Reducing blood culture contamination in the emergency department: An interrupted time series quality improvement study. Acad Emerg Med. 2013;20:89–97. doi: 10.1111/acem.12057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Archibald LK, Pallangyo K, Kazembe P, Reller LB. Blood culture contamination in Tanzania, Malawi, and the United States: a microbiological tale of three cities. J Clin Microbiol. 2006;44:4425–4429. doi: 10.1128/JCM.01215-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hall RT, Domenico HJ, Self WH, Hain PD. Reducing the blood culture contamination rate in a pediatric emergency department and subsequent cost savings. Pediatrics. 2013;131:e292–297. doi: 10.1542/peds.2012-1030. [DOI] [PubMed] [Google Scholar]
- 5.Norberg A, Christopher NC, Ramundo ML, Bower JR, Berman SA. Contamination rates of blood cultures obtained by dedicated phlebotomy vs. intravenous catheter. JAMA. 2003;289:726–729. doi: 10.1001/jama.289.6.726. [DOI] [PubMed] [Google Scholar]
- 6.Qamruddin A, Khanna N, Orr D. Peripheral blood culture contamination in adults and venepuncture technique: prospective cohort study. J Clin Pathol. 2008;61:509–513. doi: 10.1136/jcp.2007.047647. [DOI] [PubMed] [Google Scholar]
- 7.Self WH, Speroff T, McNaughton CD, Wright PW, Miller G, Johnson JG, et al. Blood culture collection through peripheral intravenous catheters increases the risk of specimen contamination among adult emergency department patients. Infect Control Hosp Epidemiol. 2012;33:524–526. doi: 10.1086/665319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bates DW, Goldman L, Lee TH. Contaminant blood cultures and resource utilization: The true consequences of false-positive results. JAMA. 1991;265:365–369. [PubMed] [Google Scholar]
- 9.van der Heijden YF, Miller G, Wright PW, Shepherd BE, Daniels TL, Talbot TR. Clinical impact of blood cultures contaminated with coagulase-negative staphylococci at an academic medical center. Infect Control Hosp Epidemiol. 2011;32:623–625. doi: 10.1086/660096. [DOI] [PubMed] [Google Scholar]
- 10.Little JR, Murray PR, Traynor PS, Spitznagel E. A randomized trial of povidone-iodine compared with iodine tincture for venipuncture site disinfection: Effects on rates of blood culture contamination. Am J Med. 1999;107:119–125. doi: 10.1016/s0002-9343(99)00197-7. [DOI] [PubMed] [Google Scholar]
- 11.Gander RM, Byrd L, DeCrescenzo M, Hirany S, Bowen M, Baughman J. Impact of blood cultures drawn by phlebotomy on contamination rates and health care costs in a hospital emergency department. J Clin Microbiol. 2009;47:1021–1024. doi: 10.1128/JCM.02162-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Souvenir D, Anderson DE, Palpant S, Mroch H, Askin S, Anderson J, et al. Blood cultures positive for coagulase-negative staphylococci: Antisepsis, pseudobacteremia, and therapy of patients. J Clin Microb. 1998;36:1923–1926. doi: 10.1128/jcm.36.7.1923-1926.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zwang O, Albert RK. Analysis of strategies to improve cost effectiveness of blood cultures. J Hosp Med. 2006;1:272–276. doi: 10.1002/jhm.115. [DOI] [PubMed] [Google Scholar]
- 14.Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev. 2006;19:788–802. doi: 10.1128/CMR.00062-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.CLSI. CLSI document M47-A. Wayne PA: Clinical and laboratory standards institute; 2007. Principles and procedures for blood cultures; Approved guideline. [Google Scholar]
- 16.Speroff T, O’Connor GT. Study designs for PDSA quality improvement research. Q Manage Health Care. 2004;13:17–32. doi: 10.1097/00019514-200401000-00002. [DOI] [PubMed] [Google Scholar]
- 17.Eccles M, Grimshaw J, Campbell M, Ramsay C. Research designs for studies evaluating the effectiveness of change and improvement strategies. Qual Saf Health Care. 2003;12:47–52. doi: 10.1136/qhc.12.1.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wagner AK, Soumerai SB, Zhang F, Ross-Degnan D. Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther. 2002;27:299–309. doi: 10.1046/j.1365-2710.2002.00430.x. [DOI] [PubMed] [Google Scholar]
- 19.Box GEP, Jenkins GM, Reinsel GC. Time series analysis: Forecasting and control. 4. Hoboken, NJ: John Wiley & Sons; 2008. [Google Scholar]
- 20.Nelson EC, Batalden PB, Godfrey MM. Quality by design: A clinical microsystems approach. San Francisco, CA: Jossey-Bass; 2007. [Google Scholar]
- 21.Varkey P, Reller MK, Resar RK. Basics of quality improvement in health care. Mayo Clin Proc. 2007;82:735–739. doi: 10.4065/82.6.735. [DOI] [PubMed] [Google Scholar]
- 22.Bekeris LG, Tworek JA, Walsh MK, Valenstein PN. Trends in blood culture contamination: A College of American Pathologists Q-Tracks Study of 356 institutions. Arch Pathol Lab Med. 2005;129:1222–1225. doi: 10.5858/2005-129-1222-TIBCCA. [DOI] [PubMed] [Google Scholar]
- 23.Dupont WD, Plummer WD. Power and sample size calculations: A review and computer program. Controlled Clinical Trials. 1990;11:116–128. doi: 10.1016/0197-2456(90)90005-m. [DOI] [PubMed] [Google Scholar]