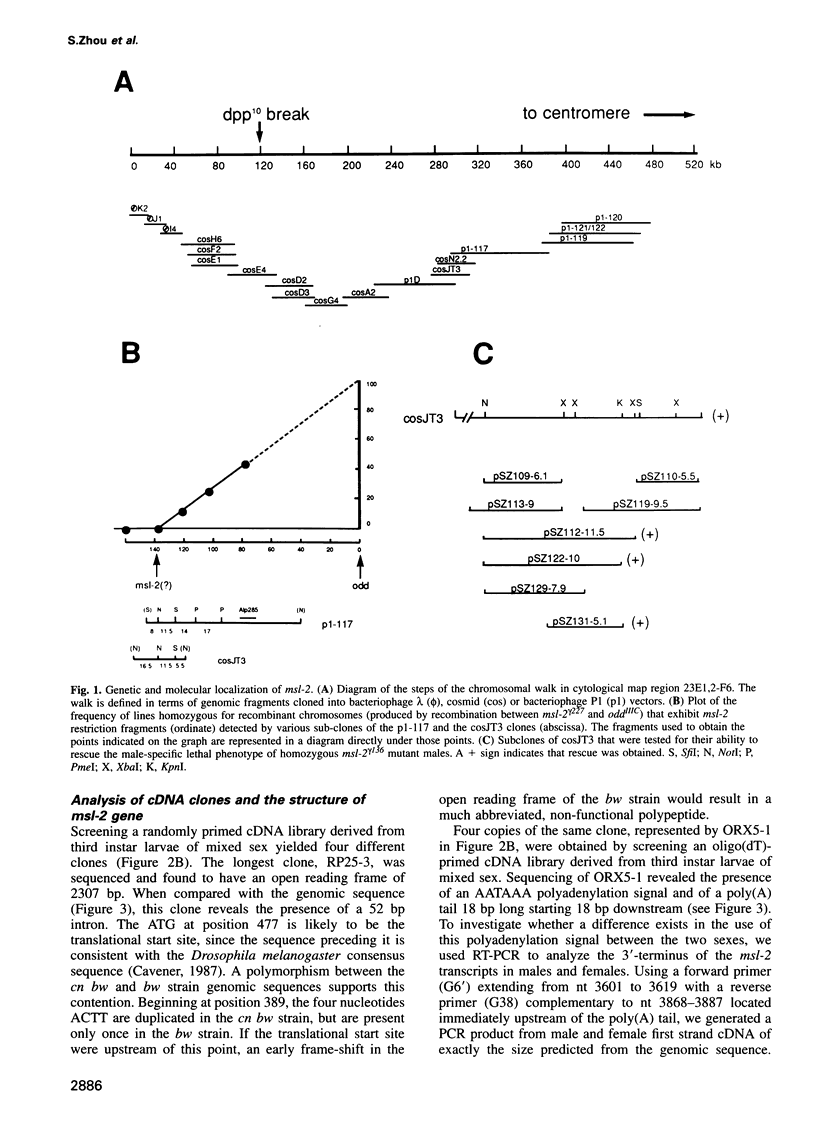
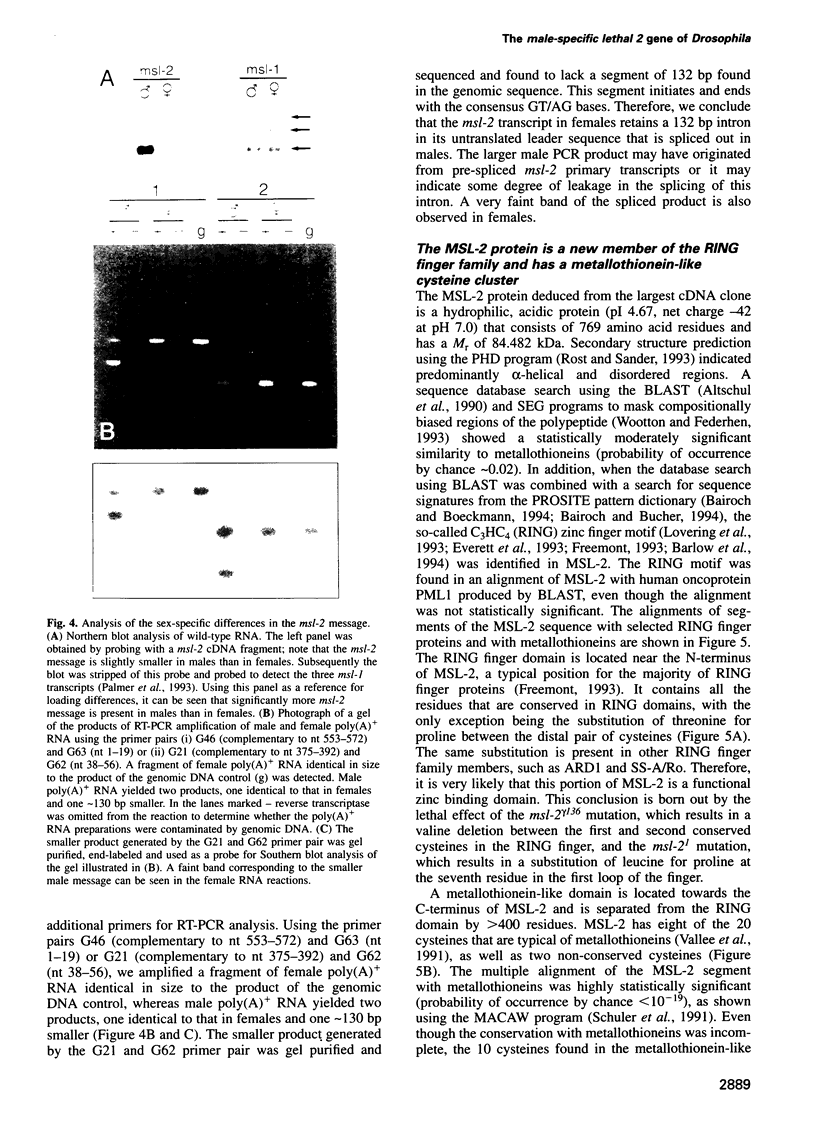
Abstract
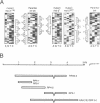
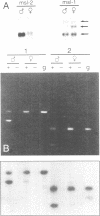
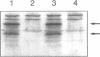
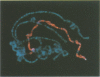
In Drosophila the equalization of X-linked gene products between males and females, i.e. dosage compensation, is the result of a 2-fold hypertranscription of most of these genes in males. At least four regulatory genes are required for this process. Three of these genes, maleless (mle), male-specific lethal 1 (msl-1) and male-specific lethal 3 (msl-3), have been cloned and their products have been shown to interact and to bind to numerous sites on the X chromosome of males, but not of females. Although binding to the X chromosome is negatively correlated with the function of the master regulatory gene Sex lethal (Sxl), the mechanisms that restrict this binding to males and to the X chromosome are not yet understood. We have cloned the last of the known autosomal genes involved in dosage compensation, male-specific lethal 2 (msl-2), and characterized its product. The encoded protein (MSL-2) consists of 769 amino acid residues and has a RING finger (C3HC4 zinc finger) and a metallothionein-like domain with eight conserved and two non-conserved cysteines. In addition, it contains a positively and a negatively charged amino acid residue cluster and a coiled coil domain that may be involved in protein-protein interactions. Males produce a msl-2 transcript that is shorter than in females, due to differential splicing of an intron of 132 bases in the untranslated leader. Using an antiserum against MSL-2 we have shown that the protein is expressed at a detectable level only in males, where it is physically associated with the X chromosome. Our observations suggest that MSL-2 may be the target of the master regulatory gene Sxl and provide the basic elements of a working hypothesis on the function of MSL-2 in mediating the 2-fold increase in transcription that is characteristic of dosage compensation.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altschul S. F., Boguski M. S., Gish W., Wootton J. C. Issues in searching molecular sequence databases. Nat Genet. 1994 Feb;6(2):119–129. doi: 10.1038/ng0294-119. [DOI] [PubMed] [Google Scholar]
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Bairoch A., Boeckmann B. The SWISS-PROT protein sequence data bank: current status. Nucleic Acids Res. 1994 Sep;22(17):3578–3580. [PMC free article] [PubMed] [Google Scholar]
- Bairoch A., Bucher P. PROSITE: recent developments. Nucleic Acids Res. 1994 Sep;22(17):3583–3589. [PMC free article] [PubMed] [Google Scholar]
- Baker B. S., Gorman M., Marín I. Dosage compensation in Drosophila. Annu Rev Genet. 1994;28:491–521. doi: 10.1146/annurev.ge.28.120194.002423. [DOI] [PubMed] [Google Scholar]
- Barlow P. N., Luisi B., Milner A., Elliott M., Everett R. Structure of the C3HC4 domain by 1H-nuclear magnetic resonance spectroscopy. A new structural class of zinc-finger. J Mol Biol. 1994 Mar 25;237(2):201–211. doi: 10.1006/jmbi.1994.1222. [DOI] [PubMed] [Google Scholar]
- Bell L. R., Horabin J. I., Schedl P., Cline T. W. Positive autoregulation of sex-lethal by alternative splicing maintains the female determined state in Drosophila. Cell. 1991 Apr 19;65(2):229–239. doi: 10.1016/0092-8674(91)90157-t. [DOI] [PubMed] [Google Scholar]
- Bell L. R., Maine E. M., Schedl P., Cline T. W. Sex-lethal, a Drosophila sex determination switch gene, exhibits sex-specific RNA splicing and sequence similarity to RNA binding proteins. Cell. 1988 Dec 23;55(6):1037–1046. doi: 10.1016/0092-8674(88)90248-6. [DOI] [PubMed] [Google Scholar]
- Belote J. M., Lucchesi J. C. Control of X chromosome transcription by the maleless gene in Drosophila. Nature. 1980 Jun 19;285(5766):573–575. doi: 10.1038/285573a0. [DOI] [PubMed] [Google Scholar]
- Belote J. M., Lucchesi J. C. Male-specific lethal mutations of Drosophila melanogaster. Genetics. 1980 Sep;96(1):165–186. doi: 10.1093/genetics/96.1.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boggs R. T., Gregor P., Idriss S., Belote J. M., McKeown M. Regulation of sexual differentiation in D. melanogaster via alternative splicing of RNA from the transformer gene. Cell. 1987 Aug 28;50(5):739–747. doi: 10.1016/0092-8674(87)90332-1. [DOI] [PubMed] [Google Scholar]
- Bone J. R., Lavender J., Richman R., Palmer M. J., Turner B. M., Kuroda M. I. Acetylated histone H4 on the male X chromosome is associated with dosage compensation in Drosophila. Genes Dev. 1994 Jan;8(1):96–104. doi: 10.1101/gad.8.1.96. [DOI] [PubMed] [Google Scholar]
- Breen T. R., Lucchesi J. C. Analysis of the dosage compensation of a specific transcript in Drosophila melanogaster. Genetics. 1986 Mar;112(3):483–491. doi: 10.1093/genetics/112.3.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brendel V., Bucher P., Nourbakhsh I. R., Blaisdell B. E., Karlin S. Methods and algorithms for statistical analysis of protein sequences. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2002–2006. doi: 10.1073/pnas.89.6.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgess W. H., Jemiolo D. K., Kretsinger R. H. Interaction of calcium and calmodulin in the presence of sodium dodecyl sulfate. Biochim Biophys Acta. 1980 Jun 26;623(2):257–270. doi: 10.1016/0005-2795(80)90254-8. [DOI] [PubMed] [Google Scholar]
- Cavener D. R. Comparison of the consensus sequence flanking translational start sites in Drosophila and vertebrates. Nucleic Acids Res. 1987 Feb 25;15(4):1353–1361. doi: 10.1093/nar/15.4.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cline T. W. The Drosophila sex determination signal: how do flies count to two? Trends Genet. 1993 Nov;9(11):385–390. doi: 10.1016/0168-9525(93)90138-8. [DOI] [PubMed] [Google Scholar]
- Everett R. D., Barlow P., Milner A., Luisi B., Orr A., Hope G., Lyon D. A novel arrangement of zinc-binding residues and secondary structure in the C3HC4 motif of an alpha herpes virus protein family. J Mol Biol. 1993 Dec 20;234(4):1038–1047. doi: 10.1006/jmbi.1993.1657. [DOI] [PubMed] [Google Scholar]
- Freemont P. S. The RING finger. A novel protein sequence motif related to the zinc finger. Ann N Y Acad Sci. 1993 Jun 11;684:174–192. doi: 10.1111/j.1749-6632.1993.tb32280.x. [DOI] [PubMed] [Google Scholar]
- Fukunaga A., Tanaka A., Oishi K. Maleless, a recessive autosomal mutant of Drosophila melanogaster that specifically kills male zygotes. Genetics. 1975 Sep;81(1):135–141. doi: 10.1093/genetics/81.1.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fürst P., Hu S., Hackett R., Hamer D. Copper activates metallothionein gene transcription by altering the conformation of a specific DNA binding protein. Cell. 1988 Nov 18;55(4):705–717. doi: 10.1016/0092-8674(88)90229-2. [DOI] [PubMed] [Google Scholar]
- Gorman M., Baker B. S. How flies make one equal two: dosage compensation in Drosophila. Trends Genet. 1994 Oct;10(10):376–380. doi: 10.1016/0168-9525(94)90135-x. [DOI] [PubMed] [Google Scholar]
- Gorman M., Franke A., Baker B. S. Molecular characterization of the male-specific lethal-3 gene and investigations of the regulation of dosage compensation in Drosophila. Development. 1995 Feb;121(2):463–475. doi: 10.1242/dev.121.2.463. [DOI] [PubMed] [Google Scholar]
- Hilfiker A., Yang Y., Hayes D. H., Beard C. A., Manning J. E., Lucchesi J. C. Dosage compensation in Drosophila: the X-chromosomal binding of MSL-1 and MLE is dependent on Sxl activity. EMBO J. 1994 Aug 1;13(15):3542–3550. doi: 10.1002/j.1460-2075.1994.tb06661.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu S., Fürst P., Hamer D. The DNA and Cu binding functions of ACE1 are interdigitated within a single domain. New Biol. 1990 Jun;2(6):544–555. [PubMed] [Google Scholar]
- Inoue K., Hoshijima K., Sakamoto H., Shimura Y. Binding of the Drosophila sex-lethal gene product to the alternative splice site of transformer primary transcript. Nature. 1990 Mar 29;344(6265):461–463. doi: 10.1038/344461a0. [DOI] [PubMed] [Google Scholar]
- Keyes L. N., Cline T. W., Schedl P. The primary sex determination signal of Drosophila acts at the level of transcription. Cell. 1992 Mar 6;68(5):933–943. doi: 10.1016/0092-8674(92)90036-c. [DOI] [PubMed] [Google Scholar]
- Kuroda M. I., Kernan M. J., Kreber R., Ganetzky B., Baker B. S. The maleless protein associates with the X chromosome to regulate dosage compensation in Drosophila. Cell. 1991 Sep 6;66(5):935–947. doi: 10.1016/0092-8674(91)90439-6. [DOI] [PubMed] [Google Scholar]
- Liu F., Green M. R. Promoter targeting by adenovirus E1a through interaction with different cellular DNA-binding domains. Nature. 1994 Apr 7;368(6471):520–525. doi: 10.1038/368520a0. [DOI] [PubMed] [Google Scholar]
- Lovering R., Hanson I. M., Borden K. L., Martin S., O'Reilly N. J., Evan G. I., Rahman D., Pappin D. J., Trowsdale J., Freemont P. S. Identification and preliminary characterization of a protein motif related to the zinc finger. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2112–2116. doi: 10.1073/pnas.90.6.2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucchesi J. C., Skripsky T. The link between dosage compensation and sex differentiation in Drosophila melanogaster. Chromosoma. 1981;82(2):217–227. doi: 10.1007/BF00286106. [DOI] [PubMed] [Google Scholar]
- Lupas A., Van Dyke M., Stock J. Predicting coiled coils from protein sequences. Science. 1991 May 24;252(5009):1162–1164. doi: 10.1126/science.252.5009.1162. [DOI] [PubMed] [Google Scholar]
- Nagoshi R. N., McKeown M., Burtis K. C., Belote J. M., Baker B. S. The control of alternative splicing at genes regulating sexual differentiation in D. melanogaster. Cell. 1988 Apr 22;53(2):229–236. doi: 10.1016/0092-8674(88)90384-4. [DOI] [PubMed] [Google Scholar]
- Palmer M. J., Mergner V. A., Richman R., Manning J. E., Kuroda M. I., Lucchesi J. C. The male-specific lethal-one (msl-1) gene of Drosophila melanogaster encodes a novel protein that associates with the X chromosome in males. Genetics. 1993 Jun;134(2):545–557. doi: 10.1093/genetics/134.2.545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer M. J., Richman R., Richter L., Kuroda M. I. Sex-specific regulation of the male-specific lethal-1 dosage compensation gene in Drosophila. Genes Dev. 1994 Mar 15;8(6):698–706. doi: 10.1101/gad.8.6.698. [DOI] [PubMed] [Google Scholar]
- Rost B., Sander C. Prediction of protein secondary structure at better than 70% accuracy. J Mol Biol. 1993 Jul 20;232(2):584–599. doi: 10.1006/jmbi.1993.1413. [DOI] [PubMed] [Google Scholar]
- Sakashita E., Sakamoto H. Characterization of RNA binding specificity of the Drosophila sex-lethal protein by in vitro ligand selection. Nucleic Acids Res. 1994 Oct 11;22(20):4082–4086. doi: 10.1093/nar/22.20.4082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuler G. D., Altschul S. F., Lipman D. J. A workbench for multiple alignment construction and analysis. Proteins. 1991;9(3):180–190. doi: 10.1002/prot.340090304. [DOI] [PubMed] [Google Scholar]
- Smoller D. A., Petrov D., Hartl D. L. Characterization of bacteriophage P1 library containing inserts of Drosophila DNA of 75-100 kilobase pairs. Chromosoma. 1991 Sep;100(8):487–494. doi: 10.1007/BF00352199. [DOI] [PubMed] [Google Scholar]
- Sosnowski B. A., Belote J. M., McKeown M. Sex-specific alternative splicing of RNA from the transformer gene results from sequence-dependent splice site blockage. Cell. 1989 Aug 11;58(3):449–459. doi: 10.1016/0092-8674(89)90426-1. [DOI] [PubMed] [Google Scholar]
- Sosnowski B. A., Davis D. D., Boggs R. T., Madigan S. J., McKeown M. Multiple portions of a small region of the Drosophila transformer gene are required for efficient in vivo sex-specific regulated RNA splicing and in vitro sex-lethal binding. Dev Biol. 1994 Jan;161(1):302–312. doi: 10.1006/dbio.1994.1030. [DOI] [PubMed] [Google Scholar]
- Stoeckle M. Y., Hanafusa H. Processing of 9E3 mRNA and regulation of its stability in normal and Rous sarcoma virus-transformed cells. Mol Cell Biol. 1989 Nov;9(11):4738–4745. doi: 10.1128/mcb.9.11.4738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tartof K. D., Hobbs C. A. New cloning vectors and techniques for easy and rapid restriction mapping. Gene. 1988 Jul 30;67(2):169–182. doi: 10.1016/0378-1119(88)90394-0. [DOI] [PubMed] [Google Scholar]
- Tatusov R. L., Koonin E. V. A simple tool to search for sequence motifs that are conserved in BLAST outputs. Comput Appl Biosci. 1994 Jul;10(4):457–459. doi: 10.1093/bioinformatics/10.4.457. [DOI] [PubMed] [Google Scholar]
- Thiele D. J. ACE1 regulates expression of the Saccharomyces cerevisiae metallothionein gene. Mol Cell Biol. 1988 Jul;8(7):2745–2752. doi: 10.1128/mcb.8.7.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner B. M., Birley A. J., Lavender J. Histone H4 isoforms acetylated at specific lysine residues define individual chromosomes and chromatin domains in Drosophila polytene nuclei. Cell. 1992 Apr 17;69(2):375–384. doi: 10.1016/0092-8674(92)90417-b. [DOI] [PubMed] [Google Scholar]
- Vallee B. L., Coleman J. E., Auld D. S. Zinc fingers, zinc clusters, and zinc twists in DNA-binding protein domains. Proc Natl Acad Sci U S A. 1991 Feb 1;88(3):999–1003. doi: 10.1073/pnas.88.3.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wootton J. C. Non-globular domains in protein sequences: automated segmentation using complexity measures. Comput Chem. 1994 Sep;18(3):269–285. doi: 10.1016/0097-8485(94)85023-2. [DOI] [PubMed] [Google Scholar]
- Zeng J., Heuchel R., Schaffner W., Kägi J. H. Thionein (apometallothionein) can modulate DNA binding and transcription activation by zinc finger containing factor Sp1. FEBS Lett. 1991 Feb 25;279(2):310–312. doi: 10.1016/0014-5793(91)80175-3. [DOI] [PubMed] [Google Scholar]
- Zeng J., Vallee B. L., Kägi J. H. Zinc transfer from transcription factor IIIA fingers to thionein clusters. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):9984–9988. doi: 10.1073/pnas.88.22.9984. [DOI] [PMC free article] [PubMed] [Google Scholar]