Abstract
Objective
Sleep disturbance, reduced quality-of-life (QOL), and other components of “sickness behavior” in patients with chronic rhinosinusitis (CRS) are poorly understood. These complex changes in central behavior are due to the effects of immune mediators acting in the brain. We hypothesized that immune mediators that have been associated with CRS are also associated with sickness behavior, somnifacient complaints, and CRS disease specific QOL.
Study Design
Pilot
Methods
Twenty patients with CRS were prospectively enrolled and completed the Pittsburgh Sleep Quality Index (PSQI), disease-specific QOL, and olfactory instruments. Ethmoid mucosa was obtained and RT-PCR was performed for the cytokines IL-4,-13, and transforming growth factor-beta (TGF-β). Average change in crossover threshold was calculated and differences in gene expression were correlated with sleep quality, CRS specific QOL and disease severity.
Results
Patients with CRS reported overall poor sleep quality and poor CRS specific QOL with significant correlations between them. Increased expression of TGF-β (r=−0.443; p=0.050) and IL-4 (r=−0.548; p=0.012) correlated with sleep dysfunction while IL-13 expression was linearly associated with worse sleep quality (PSQI scores r=−0.417; p=0.075). IL-4 and TGF-β expression was not associated with CRS disease severity or QOL, while significantly higher levels of IL-13 expression correlated with worse CRS disease severity and QOL.
Conclusion
Patients with CRS exhibited behavioral changes commonly referred to as sickness behavior which include poor sleep quality and reduced QOL. The up-regulation of IL-4 and TGF-β may contribute to inflammatory brain-mediated effects on sleep quality, while IL-13 may be a pleiotropic signaling molecule influencing sleep, QOL, and CRS disease severity.
Level of Evidence
NA / 2b
MeSH Key Words: Sinusitis, sleep, quality of life, rhinology, inflammation mediators
INTRODUCTION
Chronic rhinosinusitis (CRS) is associated with a substantial reduction in quality-of-life (QOL).1 Poor QOL may be considered an equivalent to “sickness behavior,” which is associated with reduction in motivation, depression, fatigue, pain, malaise, in addition to significant detrimental effects on sleep behavior.2 The field of clinical psychoneuroimmunology grew out of the observation that inflammatory cytokines produced in the periphery are the same as those that act within the brain to promote these central behaviors.
Little is known about the etiology and pathophysiology of sickness behavior in patients with CRS. Increased somnolence is an important component of sickness behavior, and evidence demonstrates that cytokines and other inflammatory mediators may play an integral role in this process. Patients with CRS have a high prevalence of poor sleep quality which, in turn, is significantly correlated with decreased QOL.3 Sleep is regulated in part through humoral signaling via cytokines and their downstream products.4 The cytokines interleukin (IL)-1β and tumor necrosis factor (TNF)-α are involved in the physiological regulation of rapid eye movement (REM) sleep and non-rapid eye movement (NREM) sleep in both health and disease.5 IL-1β and TNF-α induce sleep when administered centrally or systemically5–9, and substances that activate or up-regulate these cytokines (e.g., bacterial cell walls, murayml dipeptide and influenza virus) increase sleep. In contrast, antagonists (e.g., antibodies, soluble receptors) of IL-1β and TNF-α can act to decrease sleep. For instance, the cytokines IL-4, IL-13, and transforming growth factor (TGF)-β are thought to inhibit these pro-somnogenic cytokines.10–12 There is an associated up-regulation of IL-4, TGF-β and IL-13 in CRS 13,14 and these mediators have been shown to decrease sleep following intracerebral injections.15–17 Elevated levels of IL-4 correlated with increased latency to REMS, decreased time in REMS, and decreased latency to sleep onset.18 Taken as a whole, several key cytokines including IL-4, IL-13 and TGF-β are anti-somnogenic and may associate with sickness behavior in patients with CRS (Table 1). We therefore sought to quantify their expression in patients with CRS, and hypothesized that cytokine expression would correlate with sickness behavior as measured by sleep quality and disease-specific QOL.
Table 1.
Mediators implicated in non-rapid eye movement sleep, rapid eye movement sleep, and chronic rhinosinusitis.
| Somnogenic Substances/Immune Mediators | Effects NREMS | Effects REMS | CRS | References |
|---|---|---|---|---|
| Interleukin-1 | ↑ ↔ ↓ | + | Krueger5, Lennard,46 Mullol47 | |
| Interleukin-1 Receptor Antagonist | ↓ | Opp 48 | ||
| Interleukin-4 | ↓ | + | Krueger5, Kushikata16, | |
| Interleukin-6 | ↑ ↔ ↓ | + | Bauer49, Vgontzas50, Hogan51, Ghaffar52 | |
| Interleukin-10 | ↓ | + | Krueger5, Kushikata15, Opp53, Toth54, Jyonouchi 55 | |
| Interleukin-13 | ↓ | + | Kubota17, al Ghamdi,14 Reh56 | |
| Inferferon alpha | ↑ ↔ ↓ | ↓ | Bohnet57 | |
| Soluble tumor necrosis factor receptor | ↓ | Kubota58 | ||
| Tissue Growth Factor Beta | ↓ | + | Kubota,17 Kuo13 | |
| Histamine | ↓ | + | Tashiro,59 | |
| TLR | ↑ | ↓ | + | Wisor,60 Sartorius,61 Lane62 |
| Prostaglandin | ↑ ↔ ↓ | ↑ | Sri Kantha,63 Hayaishi,64 Urade,65 Krueger66 |
NREMS, non-rapid eye movement sleep; REMS, rapid eye movement sleep; CRS, chronic rhinosinusitis; TLR, toll-like receptor. “↑”,”↔”, and “↓” indicate mean influence on NREMS or REMS.
METHODS
Human Subjects
Adult study subjects with CRS were enrolled between February, 2011 and November, 2012 into an ongoing, prospective, translational research cohort. Study subjects underwent standard clinical examinations consisting of physical evaluations, patient history, computed tomography (CT) imaging of the paranasal sinuses, as well as bilateral rigid sinonasal endoscopy.
Inclusion criteria consisted of: 1) a current diagnosis of CRS as defined by the 2007 Adult Sinusitis Guidelines19, 2) previous treatment with a minimum of oral, broad spectrum or culture directed antibiotics (≥2 weeks duration) and either topical nasal corticosteroid sprays (≥3 week duration) or at least a 5-day trial of systemic steroid therapy, 3) voluntary election of endoscopic sinus surgery (ESS) as the next treatment option. Patients were also required to complete all necessary enrollment study questionnaires and the adult consent and authorization process in English. The Institutional Review Board at OHSU monitored and approved all investigational and repository protocols. Each patient was assured that study participation was completely voluntary and participation in no way affected the standard of care.
Each study subject who met inclusion criteria was asked to report demographic, social, and medical history data including: age, gender, household income, current tobacco use, alcohol consumption, nasal polyposis, depression, asthma, allergies (both history and/or confirmed allergic testing via skin prick or modified radioallergosorbent testing), history of prior sinus surgery, acetylsalicylic acid (ASA) intolerance, and cystic fibrosis. All subjects diagnosed with obstructive sleep apnea (OSA) by testing or history were excluded from final analyses. Patients with a diagnosis of CRS with nasal polyposis were excluded as nasal polyps are an independent predictor of worse sleep quality due to nasal obstruction as measured by the PSQI.20
Mucosa Tissue Collection
Those patients meeting inclusion criteria underwent ESS, per the current standard of care. The precise time of tissue collection was recorded for each subject. Prior to ESS, patients were instructed to complete one week of oral prednisone in a tapering fashion as well as oral antibiotics. Mucosal tissue specimens (~4.0mm2) were taken from the anterior ethmoid cells during ESS. Following removal from the sinonasal cavity, mucosa was separated from underlying bony fragments, placed into labeled centrifuge tubes (Bioexpress Corp. Kaysville, UT) containing 1.0 ml. of RNAlater (Applied Biosystems/Life Technologies Corp., Carlsbad, CA) and then transferred and stored using cryovials (Nalge Nunc International/Thermo Fisher Scientific, Rochester, NY) in a −80°F freezer for extraction and processing.
Real time quantitative polymerase chain reaction (RT-PCR)
Ribonucleic acid (RNA) for reverse transcription polymerase chain reaction (RT-PCR) procedures was extracted from RNAlater stabilized mucosal specimens using the RNeasy Mini Kit according to the manufacturer’s instructions (Qiagen Incorporated, Valencia, CA). After the RNA was isolated, RNA purification was performed using the RNeas-Free DNase Set (Qiagen Incorporated, Valencia, CA). A total of 5μg. RNA was used to synthesize a complementary deoxyribonucleic acid (cDNA) probe using the RT2 first strand kit (SABiosciences, Frederick, MD) for hybridization to a custom made human gene array (Applied Biosystems Inc, Carlsbad, CA) membrane including: IL-4, IL-13, TGF-β and glyceraldehyde-3-phosphate dehydrogenase (GAPDH; selected as reference gene). Semi-quantitative real-time RT-PCR was performed using the ABI StepOnePlus system (Applied Biosystems Inc., Carlsbad, CA) protocol. The threshold cycle (Ct) was defined as the fractional cycle number at which reporter fluorescence is generated by cleavage of the cDNA probe at a fixed threshold above baseline. Delta Ct (ΔCt) was calculated for each sample as the difference between Ct values of each target gene and GAPDH gene, while higher average ΔCt values represent less gene expression.
Sleep Quality Assessment – the Pittsburgh Sleep Quality Index
All patients completed the Pittsburgh Sleep Quality Index (PSQI) at initial study enrollment. The PSQI is an 18-item, self-reported measure of sleep quality and duration during the four week time period preceding survey completion. The PSQI contains seven subdomain scores including: subjective sleep quality, sleep latency, sleep duration, sleep efficiency, sleep disturbance, sleep medication usage, and daytime dysfunction. Subdomain component scores (score range: 0 – 3) are assessed using a publicly available scoring algorithm and added to obtain a total score (score range: 0 – 21). Higher total and subdomain PSQI scores suggest worse sleep quality.21
Disease-Specific Quality of Life Measures
Study participants completed two CRS-specific QOL instruments: the 30-item Rhinosinusitis Disability Index (RSDI) and the 22-item Sinonasal Outcome Test (SNOT-22). The RSDI (total score range: 0–120) is a validated, disease-specific survey instrument consisting of three subscales developed to measure the impact of CRS on a patient’s physical (score range: 0–44), functional (score range: 0–36), and emotional (score range: 0–40) subdomains. Higher scores indicate a greater impact of chronic sinonasal disease.22 The SNOT-22 is a validated outcome measure for chronic sinonasal conditions (score range: 0–120). Lower total scores on the SNOT-22 suggest better symptom severity and overall QOL.23 The enrolling physicians were blinded to all survey responses for the study duration.
Clinical Measures of Disease Severity
Computed tomography (CT) images were staged in accordance with the Lund-Mackay bilateral scoring system (score range: 0–24) where higher scores represent greater severity of disease.24 Endoscopic examinations were staged using the Lund-Kennedy endoscopy staging system (score range: 0–20) where higher scores represent worse disease severity. This semi-quantitative staging system quantifies bilateral severity of nasal polyposis, discharge, edema, scarring, and crusting.25 All visualizations were scored by the enrolling physician at OHSU at the time of enrollment. Olfactory function was measured at the initial enrollment period using The Brief Smell Identification Test (B-SIT; Sensonics, Inc, Haddon Heights, NJ). The B-SIT is a validated 12-item, non-invasive test of olfactory function that employs 12 microencapsulated odorant strips in a “scratch-’n-sniff” format (score range: 0–12) with higher scores indicating a better sense of smell.26
Statistical Analysis
All patient-level data was collected, scored, and transferred into a commercially available statistical software database package (SPSS ver.21, IBM Corp., Armonk, NY) for comparative analysis. Descriptive statistics were compiled for patient history, sleep quality, disease-specific QOL, and all clinical measure of disease severity and included means, standard deviations (SD), frequencies, and ranges. Graphical analysis was utilized to assess normality of linearity of each patient factor and those immune mediators of interest. Spearman’s correlation coefficients (rs) were used to evaluate bivariate correlations between all ordinal and continuous PSQI scores, clinical measures of disease severity, disease-specific QOL scores, and individual measures of gene expression. P-values were reported for each two-tailed, bivariate test where appropriate.
RESULTS
General Characteristics
Twenty patients without nasal polyposis met inclusion criteria, completed all eligibility requirements and were subsequently enrolled in the study. The breakdown by patient characteristics is provided in Table 2. The average age of all study participants was 46.0(14.3) years (range: 24–64 years) with a majority of female subjects (n=13; 65%). The study participants were primarily Caucasian (n=18; 90%), 30% had depression (n=6), 40% had a history of asthma (n=8), 30% were diagnosed with atopy via skin prick or mRAST (n=6), 75% reported alcohol use (n=15) and 30% had a prior history of sinus surgery (n=6). The majority of patients reporting average alcohol use (14/15; 93%) described consumption below established limits recognized as moderate drinking (14 drinks / week for men; 7 drinks / week for women).27
Table 2.
Demographic breakdown of patient characteristics.
| Characteristics: | Mean (SD) | Range | n(%) |
|---|---|---|---|
| Age (yrs) | 46.0 (14.3) | [24 – 64] | |
| Males | 7 (35.0) | ||
| Females | 13 (65.0) | ||
| Depression | 6 (30.0) | ||
| Asthma | 8 (40.0) | ||
| Allergy by Testing | 6 (30.0) | ||
| Alcohol use | 15 (75.0) | ||
| Previous Sinus | |||
| Surgery | 6 (30.0) | ||
| Endoscopy score | 4.6 (1.9) | [2 – 10] | |
| CT score | 10.6 (4.4) | [2 – 19] | |
| B-SIT olfactory score | 10.7 (1.9) | [4 – 12] |
SD=standard deviation; yrs=years; CT=computed tomography, B-SIT=Brief Smell Identification Test
Correlations between Sleep Quality and Quality of Life
Patients reported overall poor sleep quality with a mean total PSQI score of 11.3(4.5) with a range 3–20. Patients also reported an overall poor disease-specific QOL with a mean total RSDI survey score of 44.1(19.8), range: 5–81, and SNOT-22 of 53.4(14.9), range: 24–76. Correlation between PSQI total and sub-domain scores, disease-specific QOL scores, and clinical outcome measures are reported in Table 3. Sub-domain scores of the PSQI including sleep duration, disturbance, and dysfunction were associated with disease-specific QOL while the physical subdomain of the RSDI did not correlate to either the total or subdomain scores of the PSQI. Significant correlations were found between PSQI total scores and both RSDI and SNOT-22 total scores.
Table 3.
Correlation coefficients (rs) between the PSQI scores, disease-specific QOL scores, and clinical measures
| Outcomes: | Sleep Quality | Sleep Latency | Sleep Duration | Sleep Efficiency | Sleep Disturbance | Sleep Medication | Daytime Dysfunction | PSQI Total |
|---|---|---|---|---|---|---|---|---|
| RSDI Physical | 0.271 | −0.060 | 0.197 | 0.162 | 0.406 | 0.092 | 0.439 | 0.355 |
| RSDI Functional | 0.098 | 0.002 | 0.305 | 0.209 | 0.646** | 0.072 | 0.728* | 0.483** |
| RSDI Emotional | 0.263 | 0.200 | 0.286 | 0.218 | 0.701** | 0.109 | 0.776* | 0.533** |
| RSDI Total | 0.194 | −0.015 | 0.274 | 0.203 | 0.638** | 0.076 | 0.654** | 0.460** |
| SNOT-22 | 0.329 | 0.115 | 0.510** | 0.340 | 0.652** | −0.215 | 0.563** | 0.596** |
| Endoscopy score | −0.418 | −0.430 | 0.128 | −0.043 | 0.191 | −0.233 | 0.445 | −0.028 |
| CT score | −0.224 | −0.378 | −0.188 | −0.201 | 0.090 | 0.010 | 0.220 | −0.137 |
| B-SIT Olfactory score | −0.174 | 0.050 | 0.273 | 0.332 | −0.217 | 0.084 | 0.037 | 0.045 |
rs=Spearman’s correlation coefficient; PSQI=Pittsburgh Sleep Quality Index; QOL=quality-of-life; RSDI=Rhinosinusitis Disability Index; SNOT-22=Sinonasal Outcome Test-22; CT=computed tomography; B-SIT=Brief Smell Identification Test.
indicates p-value (two-tailed) significant less than 0.001;
indicates p-value less than 0.05.
Correlations between Sleep Quality, Quality of Life and Disease Severity
No significant associations were found between PSQI total and subdomain scores, Lund-Kennedy endoscopy scores, Lund-Mackay CT scores, or B-SIT olfaction function scores. Consistent with prior studies, disease severity as measured by endoscopy, CT or B-SIT olfaction scores did not significantly correlate to disease specific QOL (Table 3).
Correlations between Cytokine Gene Expression and Sleep Quality
Increased gene expression of TGF-β (mean ΔCt = 3.916 (0.396); range: 3.019 – 4.724) significantly correlated with poor PSQI sleep duration scores (r= −0.443; p=0.050), while elevated levels of IL-4 gene expression (mean ΔCt = 12.025 (1.188); range: 10.364 – 14.224) were significantly correlated with worse sleep disturbance (r= −0.548; p=0.012; Table 4) scores. Increased gene expression of IL-13 (mean ΔCt = 13.421 (1.687); range: 7.438 – 16.111) in patients with CRS was found to have a possible linear association with worse PSQI total sleep scores, however statistical significance was not reached (rs= −0.417; p=0.075). Increased levels of both IL-4 (rs = −0.308; p=0.187) and TGF-β (rs = −0.293; p=0.209) gene expression were found to have weak, non-significant correlations with PSQI total sleep scores.
Table 4.
Coefficient correlations (rs) between mean PSQI sub-domains with cytokine gene expression (ΔCt)
| Inflammatory mediators: | Sleep Quality | Sleep Latency | Sleep Duration | Sleep Efficiency | Sleep Disturbance | Sleep Medication | Daytime Dysfunction | PSQI Total |
|---|---|---|---|---|---|---|---|---|
| IL-4 | −0.023 | −0.293 | −0.376 | −0.057 | −0.548** | −0.371 | −0.250 | −0.308 |
| IL-13 | −0.332 | −0.127 | −0.296 | −0.128 | −0.356 | 0.038 | −0.308 | −0.407 |
| TGF-β | −0.368 | −0.088 | −0.443** | −0.146 | 0.000 | −0.126 | 0.056 | −0.293 |
rs=Spearman’s correlation coefficient; PSQI=Pittsburgh Sleep Quality Index; IL=interleukin; TGF-β= transforming growth factor-beta.
indicates p-value less than 0.05.
Cytokine Gene Expression, QOL and Clinical Measures of Disease Severity
Correlation between gene expression, CRS-specific QOL scores, and clinical measures of CRS disease severity scores are reported in Table 5. Gene expression did not correlate with RSDI total and sub-domain scores. IL-4 and TGF-β gene expression did not correlate with any clinical measure of disease severity, however moderate correlations were found between IL-13 gene expression and CRS disease severity as measured by the SNOT-22 and CT scores.
Table 5.
Correlation coefficients (rs) between gene expression, disease-specific QOL, and clinical measures of disease severity in patients with CRS.
| Inflammatory mediators: | RSDI Physical | RSDI Functional | RSDI Emotional | RSDI Total | SNOT-22 | Endoscopy Score | CT Score | B-SIT Olfactory Score |
|---|---|---|---|---|---|---|---|---|
| IL-4 | 0.125 | −0.063 | −0.332 | −0.096 | −0.014 | −0.206 | −0.089 | −0.138 |
| IL-13 | −0.318 | −0.252 | −0.291 | −0.285 | −0.467** | −0.250 | −0.461** | 0.118 |
| TGF-β | 0.177 | 0.293 | 0.173 | 0.248 | 0.096 | −0.136 | −0.438 | −0.059 |
rs=Spearman’s correlation coefficient; QOL=quality-of-life; CRS=chronic rhinosinusitis; RSDI=Rhinosinusitis Disability Index; SNOT-22=Sinonasal Outcome Test-22; CT=computed tomography; B-SIT=Brief Smell Identification Test; IL=interleukin; TGF-β=transforming growth factor-beta.
indicates p-value less than 0.05.
Cytokine Gene Expression and Circadian Rhythm
Correlation coefficients comparing IL-4, IL-13 and TGF-β gene expression to time of day tissue collection demonstrated no significant circadian variation (rs ≤ 0.223; p ≥ 0.346) (Figure 1–3).
Figure 1.
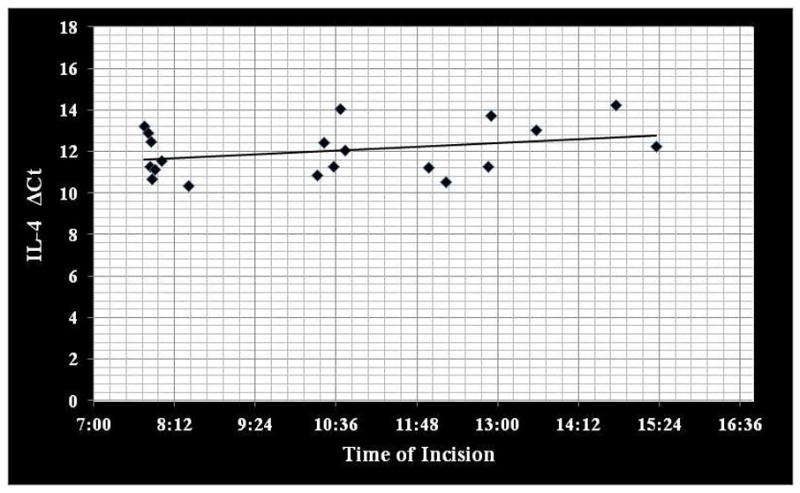
Correlation between IL-4 gene expression (ΔCt) to time of day mucosal tissue was collected. IL, interleukin (rs = 0.197; p=0.405).
Figure 3.
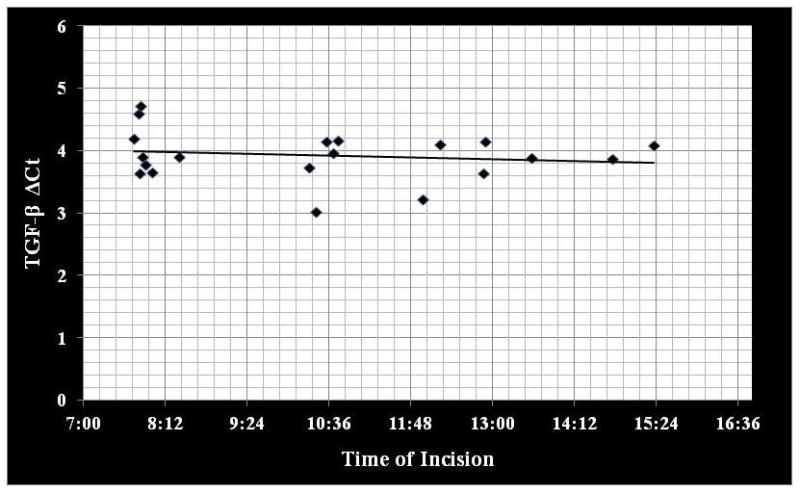
Correlation between TGF-β gene expression (ΔCt) to time of day mucosal tissue was collected. TGF-β=transforming growth factor-beta (rs = −0.164; p=0.490).
DISCUSSION
The rationale behind this pilot study was to determine if associations exist between sleep inhibiting immune mediators and sleep quality in patients with CRS, while simultaneously evaluating whether gene expression variation is merely a consequence of the pathogenesis of the disease itself. We observed the following: 1) patients with CRS exhibit sickness behavior as measured by an overall poor sleep quality and reduced disease specific QOL, 2) CRS disease severity does not play a role in either sleep quality or QOL, 3) IL-4 and TGF-β gene expression associated with some aspects of CRS sleep dysfunction but were not pervasively significant across all measures of sleep quality 4) increased IL-13 expression correlated with worse CRS-specific QOL, greater disease severity and trended towards associating with sleep dysfunction. Taken together, these findings suggest that anti-somnogenic cytokine expression in patients with CRS may play a role in CRS sickness behavior.
Intuitively sleep quality predicts reduced QOL and these measures are often associated with greater disease severity due to an activated inflammatory response.28–31 In a similar manner, we found that a progressive decline in sleep quality significantly correlates with an overall worse CRS-specific QOL. Interestingly, we found that sleep quality in patients with CRS did not associate with CRS-specific QOL or disease severity. Likewise, neither IL-4 nor TGF-β expression was associated with any of the CRS-specific QOL or disease severity instruments. In contrast, IL-13 was associated with worse CRS-specific QOL and greater disease severity. These findings suggest that poor sleep quality in patients with CRS does not associate with disease severity but does associate with CRS related QOL assessments.
The pathophysiology of poor sleep in patients with CRS is most likely multifactorial and humoral signaling through inflammatory mediators may be one of the factors. Humoral sleep regulation in the central nervous system (CNS) is thought to be controlled via sleep regulatory substances (cytokines and growth factors) that alter input-output signaling among interconnected neuronal groups found in sleep centers and in the cortex of the CNS.4 There is considerable evidence linking pro-inflammatory cytokines such as TNF-α and IL1-β to sleep regulation32 while anti-somnogenic cytokines such as IL-4, IL-10, 1L-13 and TGF-β are thought to inhibit sleep by blocking sleep regulatory substances such as IL-1β and TNF-α.15–17 IL-13 inhibits IL-1β,10 through the induction and release of the IL-1 type II decoy receptor.33,34 In a similar fashion, IL-13 and TGF-β suppress both IL-1β and TNF-α production,12,35 and suppress pro-inflammatory cytokine production through the up regulation of IL-1 receptor antagonists.11
We found that the increased expression of IL-4, IL-13 and TGF-β tended to be linearly associated with poor sleep quality. Increased expression of IL-4 was significantly associated with greater sleep disturbance, while increased TGF-β expression significantly correlated with worse sleep duration. Expression of IL-13 appears to be a pleiotropic factor in CRS sickness behavior, exhibiting multiple associations with worse CRS-specific disease severity scores as well as worse disease specific QOL.
The mechanisms through which local inflammatory cytokines signal the central nervous system in patients with CRS to induce sickness behavior is unknown. However, good evidence suggests that immune mediators may signal the brain via transport across the blood brain barrier (BBB), through the circumventricular organs, or act locally, as opposed to systemically, through the stimulation or alteration of afferent neuronal transmission.36–38
Cytokines released locally in response to local infectious and inflammatory stimuli, act primarily in an autocrine or paracrine (as opposed to endocrine) fashion to further potentiate the inflammatory response. Cytokines are proteins whose molecular characteristics such as size (e.g., 150 amino acids for IL-1β) and hydrophilic properties inhibit their movement through the BBB. Therefore, their ability to act directly on the brain to induce behavior has been debated. For these reasons cytokines are thought to act at brain sites that lack a BBB, known as circumventricular organs due to their close association with the brain ventricles. There are some problems with this hypothesis as sickness behavior can precede and subside before inflammatory cytokines are detected in the blood,39 and cytokine levels have not been detected in many circumstances, thereby implying a non-blood source However, it has been shown that pro-inflammatory cytokines are extremely potent, requiring only femtomole amounts to illicit behavior manifestations, making it plausible that assays in use are not sensitive enough to measure circulating levels. In this circumstance gene expression profiles could represent the cumulative action of many hard to detect events. In addition, the measured inflammatory response in the periphery would be the cumulative sum of all cytokine production diminishing the specificity. Taken as a whole these results are consistent with the idea that elevated cytokines in the blood do not always correlate with the physiology of behavior after immune stimulation, which implies a redundant signaling method is most likely in place.
Locally released cytokines, such as IL-1β, communicate with the brain, in part, through afferent nerve transmission, and thereby influence sickness behavior.38,40,41 The expression and action of these immune mediators are regulated and modified by various molecular intermediates including IL-4, IL-13 and TGF-β. These cytokines, released locally, can inhibit IL-1β through NFkB, IL-R antagonist,11 inducing decoy receptors42 and decreasing cell surface expression of IL-1 receptors.43,44 Because these cytokines balance downstream expression, it is possible we are seeing increased expression due to an over stimulation of IL-1β; increasing doses of IL-1β have divergent effects on sleep in animal studies as lower doses of IL-1β increase sleep, whereas high-doses inhibit sleep and induce fever.7 Further investigation elucidating the role IL-1β plays in CRS sickness behavior should be carried out. Our understanding of these neural pathways from the immune system to the brain is in its early stages.
Ultimately, CNS input via neuronal or humoral signaling can dramatically influence neuronal output which may ultimately explain sickness behavior seen in patients with CRS and gives insight into the brain’s bi-directional relationship with the immune system. The fact that cytokines act within the brain to induce sickness behavior may begin to help explain the pathophysiology of CRS, thereby, leading to improved treatment of those “sickness behaviors” commonly associated with CRS ultimately improving patients QOL.
The strengths of this pilot investigation include prospective enrollment and translational design, as well as use of validated, disease-specific survey instruments. There are caveats to consider in the interpretation of these results. When measuring gene expression multiple assumptions are made including; 1) more abundant transcripts are more important, 2) changes in expression indicate a biologic difference, 3) and most importantly gene expression is a “proxy” measure for transcription/translational events. We acknowledge that the medical treatment of CRS in the days, weeks and months prior to obtaining mucosal samples, has the potential to impact mucosal inflammation and gene expression such that glucocorticoids have been shown to have complex effects on cellular signaling in nasal polyp tissue.45 To our knowledge there have been no well-designed studies systematically examining gene expression following perioperative oral steroids in patients without nasal polyposis, leaving a large gap in the current knowledge. This treatment paradigm represents our current standard of practice and was not modified for this study for strict research purposes. Taken as a whole, the results presented herein, should be reviewed with this in mind. Future studies evaluating gene expression following inflammatory modifying medication would further advance our understanding. This pilot study, by definition, reports on a small cohort in an attempt to generate hypotheses for future study and help delineate a research direction. Given the limited small sample size, correcting for multiple, pair-wise comparisons was not implemented. Readers should therefore consider the potential for spurious results due to a higher likelihood of committing a type I error. In addition, cross-sectional investigations, such as this, are innately limited in generalizability and cannot infer a causal association.
CONCLUSION
Chronic rhinosinusitis is an organ-specific inflammatory disease associated with dramatic systemic maladaptive behaviors such as sleep dysfunction and poor QOL. Although the underlying etiology and pathophysiology remains largely unknown, based on the literature, and our preliminary findings, it is logical for one to assume that immune mediators are playing a role in sleep dysfunction, QOL, and to disease severity in patients with CRS. Future studies examining systemic and local cytokine profiles such as those presented in Table 1 and their association to “sickness” will be instrumental in delineating the complex neuro-immune interactions we observe as physicians.
Figure 2.
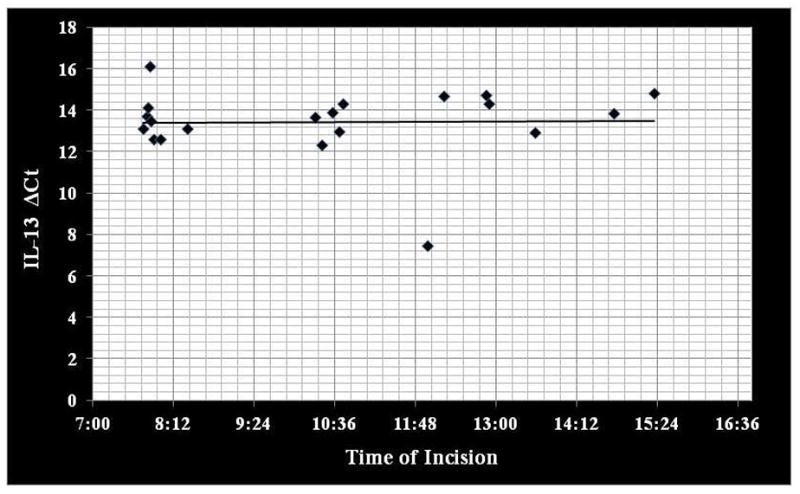
Correlation between IL-13 gene expression (ΔCt) to time of day mucosal tissue was collected. IL, interleukin (rs = 0.223; p=0.346).
Footnotes
Potential Conflicts of Interest: None
Financial Disclosures: Jess C. Mace, MPH, and Timothy L. Smith, MD, MPH are supported by a grant from the National Institute on Deafness and Other Communication Disorders (NIDCD), one of the National Institutes of Health, Bethesda, MD. (R01 DC005805; PI/PD: TL Smith). Public clinical trial registration (http://www.clinicaltrials.gov) ID# NCT01332136. Timothy L. Smith, MD is also a consultant for Intersect ENT (Palo Alto, CA) which is not affiliated with this investigation.
Accepted for oral presentation at the annual meeting of the American Rhinologic Society within the Combined Otolaryngology Spring Meeting (COSM) in Orlando, Florida on April 10–14, 2013.
References
- 1.Rudmik L, Smith TL. Quality of life in patients with chronic rhinosinusitis. Curr Allergy Asthma Rep. 11:247–252. doi: 10.1007/s11882-010-0175-2. [DOI] [PubMed] [Google Scholar]
- 2.Kelley KW, Bluthe RM, Dantzer R, et al. Cytokine-induced sickness behavior. Brain Behav Immun. 2003;17 (Suppl 1):S112–118. doi: 10.1016/s0889-1591(02)00077-6. [DOI] [PubMed] [Google Scholar]
- 3.Alt JA, Smith TL, Mace JC, Soler ZM. Sleep Quality and Disease Severity in Patients with Chronic Rhinosinusitis. Laryngoscope. 2013 doi: 10.1002/lary.24040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Krueger JM, Obal F, Jr, Fang J. Why we sleep: a theoretical view of sleep function. Sleep Med Rev. 1999;3:119–129. doi: 10.1016/s1087-0792(99)90019-9. [DOI] [PubMed] [Google Scholar]
- 5.Krueger JM. The role of cytokines in sleep regulation. Curr Pharm Des. 2008;14:3408–3416. doi: 10.2174/138161208786549281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Opp MR, Krueger JM. Interleukin-1 is involved in responses to sleep deprivation in the rabbit. Brain Res. 1994;639:57–65. doi: 10.1016/0006-8993(94)91764-7. [DOI] [PubMed] [Google Scholar]
- 7.Opp MR, Obal F, Jr, Krueger JM. Interleukin 1 alters rat sleep: temporal and dose-related effects. Am J Physiol. 1991;260:R52–58. doi: 10.1152/ajpregu.1991.260.1.R52. [DOI] [PubMed] [Google Scholar]
- 8.Fang J, Wang Y, Krueger JM. Effects of interleukin-1 beta on sleep are mediated by the type I receptor. Am J Physiol. 1998;274:R655–660. doi: 10.1152/ajpregu.1998.274.3.R655. [DOI] [PubMed] [Google Scholar]
- 9.Susic V, Totic S. “Recovery” function of sleep: effects of purified human interleukin-1 on the sleep and febrile response of cats. Metab Brain Dis. 1989;4:73–80. doi: 10.1007/BF00999497. [DOI] [PubMed] [Google Scholar]
- 10.Deleuran B, Iversen L, Deleuran M, et al. Interleukin 13 suppresses cytokine production and stimulates the production of 15-HETE in PBMC. A comparison between IL-4 and IL-13. Cytokine. 1995;7:319–324. doi: 10.1006/cyto.1995.0040. [DOI] [PubMed] [Google Scholar]
- 11.Muzio M, Sironi M, Polentarutti N, Mantovani A, Colotta F. Induction by transforming growth factor-beta 1 of the interleukin-1 receptor antagonist and of its intracellular form in human polymorphonuclear cells. Eur J Immunol. 1994;24:3194–3198. doi: 10.1002/eji.1830241242. [DOI] [PubMed] [Google Scholar]
- 12.Suzumura A, Sawada M, Yamamoto H, Marunouchi T. Transforming growth factor-beta suppresses activation and proliferation of microglia in vitro. J Immunol. 1993;151:2150–2158. [PubMed] [Google Scholar]
- 13.Kou W, Hu GH, Yao HB, et al. Regulation of transforming growth factor-beta1 activation and expression in the tissue remodeling involved in chronic rhinosinusitis. ORL J Otorhinolaryngol Relat Spec. 2012;74:172–178. doi: 10.1159/000338799. [DOI] [PubMed] [Google Scholar]
- 14.al Ghamdi K, Ghaffar O, Small P, Frenkiel S, Hamid Q. IL-4 and IL-13 expression in chronic sinusitis: relationship with cellular infiltrate and effect of topical corticosteroid treatment. J Otolaryngol. 1997;26:160–166. [PubMed] [Google Scholar]
- 15.Kushikata T, Fang J, Krueger JM. Interleukin-10 inhibits spontaneous sleep in rabbits. J Interferon Cytokine Res. 1999;19:1025–1030. doi: 10.1089/107999099313244. [DOI] [PubMed] [Google Scholar]
- 16.Kushikata T, Fang J, Wang Y, Krueger JM. Interleukin-4 inhibits spontaneous sleep in rabbits. Am J Physiol. 1998;275:R1185–1191. doi: 10.1152/ajpregu.1998.275.4.R1185. [DOI] [PubMed] [Google Scholar]
- 17.Kubota T, Fang J, Kushikata T, Krueger JM. Interleukin-13 and transforming growth factor-beta1 inhibit spontaneous sleep in rabbits. Am J Physiol Regul Integr Comp Physiol. 2000;279:R786–792. doi: 10.1152/ajpregu.2000.279.3.R786. [DOI] [PubMed] [Google Scholar]
- 18.Krouse HJ, Davis JE, Krouse JH. Immune mediators in allergic rhinitis and sleep. Otolaryngol Head Neck Surg. 2002;126:607–613. doi: 10.1067/mhn.2002.125300. [DOI] [PubMed] [Google Scholar]
- 19.Rosenfeld RMAD, Bhattacharyya N, et al. Clinical practice guideline. Otolaryngol Head Neck Surg. 2007;137:S1–31. doi: 10.1016/j.otohns.2007.06.726. [DOI] [PubMed] [Google Scholar]
- 20.Alt JA, Mace J, Smith TL. Nasal Obstruction and Sleep Quality: Weak But Positive Correlation in Chronic Rhinosinusitis. 2013 In Preparation. [Google Scholar]
- 21.Buysse DJ, Reynolds CF, 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 22.Benninger MS, Senior BA. The development of the Rhinosinusitis Disability Index. Arch Otolaryngol Head Neck Surg. 1997;123:1175–1179. doi: 10.1001/archotol.1997.01900110025004. [DOI] [PubMed] [Google Scholar]
- 23.Piccirillo JF, Merritt MG, Jr, Richards ML. Psychometric and clinimetric validity of the 20-Item Sino-Nasal Outcome Test (SNOT-20) Otolaryngol Head Neck Surg. 2002;126:41–47. doi: 10.1067/mhn.2002.121022. [DOI] [PubMed] [Google Scholar]
- 24.Lund VJ, Mackay IS. Staging in rhinosinusitis. Rhinology. 1993;31:183–184. [PubMed] [Google Scholar]
- 25.Lund VJ, Kennedy DW. Staging for rhinosinusitis. Otolaryngol Head Neck Surg. 1997;117:35–40. doi: 10.1016/S0194-59989770005-6. [DOI] [PubMed] [Google Scholar]
- 26.Doty R. The Brief Smell Identification Test Administration Manual. Sensonics, Inc; Haddon Heights, NJ: 2001. [Google Scholar]
- 27.CDC. Center for Disease Control and Prevention. Alcohol and Public Health, Fact sheets - Alcohol Use and Health [Google Scholar]
- 28.Omachi TA. Measures of sleep in rheumatologic diseases: Epworth Sleepiness Scale (ESS), Functional Outcome of Sleep Questionnaire (FOSQ), Insomnia Severity Index (ISI), and Pittsburgh Sleep Quality Index (PSQI) Arthritis Care Res (Hoboken) 2012;63 (Suppl 11):S287–296. doi: 10.1002/acr.20544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jankelowitz L, Reid KJ, Wolfe L, Cullina J, Zee PC, Jain M. Cystic fibrosis patients have poor sleep quality despite normal sleep latency and efficiency. Chest. 2005;127:1593–1599. doi: 10.1378/chest.127.5.1593. [DOI] [PubMed] [Google Scholar]
- 30.Batmaz I, Sariyildiz MA, Dilek B, Bez Y, Karakoc M, Cevik R. Sleep quality and associated factors in ankylosing spondylitis: relationship with disease parameters, psychological status and quality of life. Rheumatol Int. 2013;33:1039–1045. doi: 10.1007/s00296-012-2513-2. [DOI] [PubMed] [Google Scholar]
- 31.Wells GA, Li T, Kirwan JR, et al. Assessing quality of sleep in patients with rheumatoid arthritis. J Rheumatol. 2009;36:2077–2086. doi: 10.3899/jrheum.090362. [DOI] [PubMed] [Google Scholar]
- 32.Obal F, Jr, Krueger JM. Biochemical regulation of non-rapid-eye-movement sleep. Front Biosci. 2003;8:d520–550. doi: 10.2741/1033. [DOI] [PubMed] [Google Scholar]
- 33.Colotta F, Re F, Muzio M, et al. Interleukin-13 induces expression and release of interleukin-1 decoy receptor in human polymorphonuclear cells. J Biol Chem. 1994;269:12403–12406. [PubMed] [Google Scholar]
- 34.Re F, Muzio M, De Rossi M, et al. The type II “receptor” as a decoy target for interleukin 1 in polymorphonuclear leukocytes: characterization of induction by dexamethasone and ligand binding properties of the released decoy receptor. J Exp Med. 1994;179:739–743. doi: 10.1084/jem.179.2.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cosentino G, Soprana E, Thienes CP, Siccardi AG, Viale G, Vercelli D. IL-13 down-regulates CD14 expression and TNF-alpha secretion in normal human monocytes. J Immunol. 1995;155:3145–3151. [PubMed] [Google Scholar]
- 36.Quan N, Banks WA. Brain-immune communication pathways. Brain Behav Immun. 2007;21:727–735. doi: 10.1016/j.bbi.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 37.Turnbull AV, Rivier CL. Regulation of the hypothalamic-pituitary-adrenal axis by cytokines: actions and mechanisms of action. Physiol Rev. 1999;79:1–71. doi: 10.1152/physrev.1999.79.1.1. [DOI] [PubMed] [Google Scholar]
- 38.Watkins LR, Maier SF, Goehler LE. Cytokine-to-brain communication: a review & analysis of alternative mechanisms. Life Sci. 1995;57:1011–1026. doi: 10.1016/0024-3205(95)02047-m. [DOI] [PubMed] [Google Scholar]
- 39.Campisi J, Hansen MK, O’Connor KA, et al. Circulating cytokines and endotoxin are not necessary for the activation of the sickness or corticosterone response produced by peripheral E. coli challenge. J Appl Physiol. 2003;95:1873–1882. doi: 10.1152/japplphysiol.00371.2003. [DOI] [PubMed] [Google Scholar]
- 40.Hansen MK, O’Connor KA, Goehler LE, Watkins LR, Maier SF. The contribution of the vagus nerve in interleukin-1beta-induced fever is dependent on dose. Am J Physiol Regul Integr Comp Physiol. 2001;280:R929–934. doi: 10.1152/ajpregu.2001.280.4.R929. [DOI] [PubMed] [Google Scholar]
- 41.Niijima A. The afferent discharges from sensors for interleukin 1 beta in the hepatoportal system in the anesthetized rat. J Auton Nerv Syst. 1996;61:287–291. doi: 10.1016/s0165-1838(96)00098-7. [DOI] [PubMed] [Google Scholar]
- 42.Dubois CM, Ruscetti FW, Keller JR, et al. In vivo interleukin-1 (IL-1) administration indirectly promotes type II IL-1 receptor expression on hematopoietic bone marrow cells: novel mechanism for the hematopoietic effects of IL-1. Blood. 1991;78:2841–2847. [PubMed] [Google Scholar]
- 43.Dubois CM, Ruscetti FW, Palaszynski EW, Falk LA, Oppenheim JJ, Keller JR. Transforming growth factor beta is a potent inhibitor of interleukin 1 (IL-1) receptor expression: proposed mechanism of inhibition of IL-1 action. J Exp Med. 1990;172:737–744. doi: 10.1084/jem.172.3.737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Redini F, Mauviel A, Pronost S, Loyau G, Pujol JP. Transforming growth factor beta exerts opposite effects from interleukin-1 beta on cultured rabbit articular chondrocytes through reduction of interleukin-1 receptor expression. Arthritis Rheum. 1993;36:44–50. doi: 10.1002/art.1780360108. [DOI] [PubMed] [Google Scholar]
- 45.Bolger WE, Joshi AS, Spear S, Nelson M, Govindaraj K. Gene expression analysis in sinonasal polyposis before and after oral corticosteroids: a preliminary investigation. Otolaryngol Head Neck Surg. 2007;137:27–33. doi: 10.1016/j.otohns.2007.01.023. [DOI] [PubMed] [Google Scholar]
- 46.Lennard CM, Mann EA, Sun LL, Chang AS, Bolger WE. Interleukin-1 beta, interleukin-5, interleukin-6, interleukin-8, and tumor necrosis factor-alpha in chronic sinusitis: response to systemic corticosteroids. Am J Rhinol. 2000;14:367–373. doi: 10.2500/105065800779954329. [DOI] [PubMed] [Google Scholar]
- 47.Mullol J, Xaubet A, Lopez E, Roca-Ferrer J, Picado C. Comparative study of the effects of different glucocorticosteroids on eosinophil survival primed by cultured epithelial cell supernatants obtained from nasal mucosa and nasal polyps. Thorax. 1995;50:270–274. doi: 10.1136/thx.50.3.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Opp MR, Postlethwaite AE, Seyer JM, Krueger JM. Interleukin 1 receptor antagonist blocks somnogenic and pyrogenic responses to an interleukin 1 fragment. Proc Natl Acad Sci U S A. 1992;89:3726–3730. doi: 10.1073/pnas.89.9.3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bauer J, Hohagen F, Ebert T, et al. Interleukin-6 serum levels in healthy persons correspond to the sleep-wake cycle. Clin Investig. 1994;72:315. doi: 10.1007/BF00180048. [DOI] [PubMed] [Google Scholar]
- 50.Vgontzas AN, Papanicolaou DA, Bixler EO, et al. Circadian interleukin-6 secretion and quantity and depth of sleep. J Clin Endocrinol Metab. 1999;84:2603–2607. doi: 10.1210/jcem.84.8.5894. [DOI] [PubMed] [Google Scholar]
- 51.Hogan D, Morrow JD, Smith EM, Opp MR. Interleukin-6 alters sleep of rats. J Neuroimmunol. 2003;137:59–66. doi: 10.1016/s0165-5728(03)00038-9. [DOI] [PubMed] [Google Scholar]
- 52.Ghaffar O, Lavigne F, Kamil A, Renzi P, Hamid Q. Interleukin-6 expression in chronic sinusitis: colocalization of gene transcripts to eosinophils, macrophages, T lymphocytes, and mast cells. Otolaryngol Head Neck Surg. 1998;118:504–511. doi: 10.1016/s0194-5998(98)70209-8. [DOI] [PubMed] [Google Scholar]
- 53.Opp MR, Smith EM, Hughes TK., Jr Interleukin-10 (cytokine synthesis inhibitory factor) acts in the central nervous system of rats to reduce sleep. J Neuroimmunol. 1995;60:165–168. doi: 10.1016/0165-5728(95)00066-b. [DOI] [PubMed] [Google Scholar]
- 54.Toth LA, Opp MR. Cytokine- and microbially induced sleep responses of interleukin-10 deficient mice. Am J Physiol Regul Integr Comp Physiol. 2001;280:R1806–1814. doi: 10.1152/ajpregu.2001.280.6.R1806. [DOI] [PubMed] [Google Scholar]
- 55.Jyonouchi H, Sun S, Le H, Rimell FL. Evidence of dysregulated cytokine production by sinus lavage and peripheral blood mononuclear cells in patients with treatment-resistant chronic rhinosinusitis. Arch Otolaryngol Head Neck Surg. 2001;127:1488–1494. doi: 10.1001/archotol.127.12.1488. [DOI] [PubMed] [Google Scholar]
- 56.Reh DD, Ramanathan M, Jr, Sultan B, Wang Y, May L, Lane AP. The role of hepatocyte growth factor/c-Met in chronic rhinosinusitis with nasal polyps. Am J Rhinol Allergy. 2010;24:266–270. doi: 10.2500/ajra.2010.24.3485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bohnet SG, Traynor TR, Majde JA, Kacsoh B, Krueger JM. Mice deficient in the interferon type I receptor have reduced REM sleep and altered hypothalamic hypocretin, prolactin and 2′,5′-oligoadenylate synthetase expression. Brain Res. 2004;1027:117–125. doi: 10.1016/j.brainres.2004.08.041. [DOI] [PubMed] [Google Scholar]
- 58.Kubota T, Majde JA, Brown RA, Krueger JM. Tumor necrosis factor receptor fragment attenuates interferon-gamma-induced non-REM sleep in rabbits. J Neuroimmunol. 2001;119:192–198. doi: 10.1016/s0165-5728(01)00382-4. [DOI] [PubMed] [Google Scholar]
- 59.Tashiro M, Mochizuki H, Iwabuchi K, et al. Roles of histamine in regulation of arousal and cognition: functional neuroimaging of histamine H1 receptors in human brain. Life Sci. 2002;72:409–414. doi: 10.1016/s0024-3205(02)02276-2. [DOI] [PubMed] [Google Scholar]
- 60.Wisor JP, Clegern WC, Schmidt MA. Toll-like receptor 4 is a regulator of monocyte and electroencephalographic responses to sleep loss. Sleep. 2011;34:1335–1345. doi: 10.5665/SLEEP.1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sartorius T, Lutz SZ, Hoene M, et al. Toll-like receptors 2 and 4 impair insulin-mediated brain activity by interleukin-6 and osteopontin and alter sleep architecture. FASEB J. 2012;26:1799–1809. doi: 10.1096/fj.11-191023. [DOI] [PubMed] [Google Scholar]
- 62.Lane AP, Truong-Tran QA, Myers A, Bickel C, Schleimer RP. Serum amyloid A, properdin, complement 3, and toll-like receptors are expressed locally in human sinonasal tissue. Am J Rhinol. 2006;20:117–123. [PMC free article] [PubMed] [Google Scholar]
- 63.Sri Kantha S, Matsumura H, Kubo E, et al. Effects of prostaglandin D2, lipoxins and leukotrienes on sleep and brain temperature of rats. Prostaglandins Leukot Essent Fatty Acids. 1994;51:87–93. doi: 10.1016/0952-3278(94)90083-3. [DOI] [PubMed] [Google Scholar]
- 64.Hayaishi O, Urade Y. Prostaglandin D2 in sleep-wake regulation: recent progress and perspectives. Neuroscientist. 2002;8:12–15. doi: 10.1177/107385840200800105. [DOI] [PubMed] [Google Scholar]
- 65.Urade Y, Hayaishi O. Prostaglandin D2 and sleep/wake regulation. Sleep Med Rev. 2011;15:411–418. doi: 10.1016/j.smrv.2011.08.003. [DOI] [PubMed] [Google Scholar]
- 66.Krueger JM, Kapas L, Opp MR, Obal F., Jr Prostaglandins E2 and D2 have little effect on rabbit sleep. Physiol Behav. 1992;51:481–485. doi: 10.1016/0031-9384(92)90168-2. [DOI] [PubMed] [Google Scholar]


