Abstract
Adrenal myelolipomas are rare benign tumors of the adrenal cortex composed of adipose and hematopoietic cells. They have been postulated to arise from repeated stimulation by stress, inflammation and ACTH oversecretion. Myelolipomas are usually detected incidentally on imaging and do not require any active intervention besides regular follow-up by imaging. However, myelolipomas may insidiously grow to large sizes and cause mass effects and hemorrhage. Timely diagnosis and surgical resection are curative and lifesaving.
Key words: Myelolipoma, Adrenal myelolipoma, Benign tumor, Resection
Introduction
Adrenal myelolipomas are benign tumors which are usually asymptomatic and diagnosed incidentally on imaging or during autopsy. They were first described by Gierke in 1905 and were termed as myelolipomas by Oberling in 1929 [1, 2]. They are uncommon benign tumors of the adrenal cortex constituting approximately for 3–5% of all primary adrenal tumors and are increasingly being diagnosed due to improved imaging techniques [3, 4]. Adrenal myelolipomas are most frequently detected after the fifth decade and are seen almost equally in both genders. They are commonly found as unilateral masses, but occasional bilateral masses have been documented [5]. A right-sided predominance in unilateral tumors has also been reported [6]. They are usually asymptomatic but myelolipomas may attain large sizes and present as abdominal masses [7, 8]. Patients may also present with symptoms of hematuria and hypertension [9]. Their size may also cause mass effect by compressing other intra-abdominal organs [10]. Malignant transformation has not been documented in these tumors. Diagnosis of giant adrenal myelolipomas in a timely manner may prevent complications due to hemorrhage and can be lifesaving.
Case Description
A 52-year-old man with a past medical history significant for atrial fibrillation, diabetes type 2 and hypertension for 30 years was admitted with early satiety for the past 3 months and progressive dyspnea on exertion for 3 weeks. He also complained of gradual unintentional weight loss of approximately 100 pounds over the last 10 years. He had been a non-smoker and a moderate alcohol user for the last 30 years. On initial presentation to the hospital, he denied shortness of breath at rest or chest discomfort and was in no acute distress.
His blood pressure was 204/93 mm Hg on the left arm and 194/88 mm Hg on the right arm with an irregular heart rate of 54 beats/min and oxygen saturation of 96% on room air. The lungs were clear to auscultation, jugular venous distension was noted at 13 cm above the sternal angle and 3+ bilateral leg edema with scrotal edema. Cardiac auscultation was pertinent for 3/6 systolic ejection murmur over the left sternal border. His abdomen felt firm and lacking in tympanicity, but it was non-tender in all four quadrants. Bowel sounds were audible but faint. Laboratory values were notable for a brain natriuretic peptide of 980 ng/l, creatinine of 1.2 mg/dl, BUN of 20 mg/dl, HCO3 of 31 mEq/l, INR of 1.23, Hb of 12.5 g/dl, Na of 140 mEq/l, K of 3.7 mEq/l and negative cardiac troponins. He was monitored in the Intensive Care Unit for decompensated heart failure with uncontrolled hypertension.
To promote diuresis and reduce afterload, he was administered intravenous furosemide 80 mg, oral metolazone 2.5 mg, oral hydralazine 75 mg every 6 h, oral clonidine 0.3 mg, lisinopril 40 mg, isosorbide mononitrate 120 mg and losartan 100 mg. Overnight, he had an episode of moderate self-limited epistaxis with minimal improvement in blood pressure control. He remained in slow atrial fibrillation with a lowest recorded heart rate of 38 beats/min. However, he denied dizziness at any time despite a negative fluid balance of 1.8 liters. Renal function worsened with an increase of creatinine to 1.6 mg/dl.
An echocardiogram showed a left ventricular ejection fraction of 30% with four-chamber dilatation, moderately reduced right ventricular systolic function, severe mitral regurgitation and moderate pulmonary hypertension. He underwent right heart catheterization showing a right atrial pressure of 12 mm Hg, right ventricular pressure (systolic/diastolic) of 60/12 mm Hg, pulmonary artery pressure of 60/23 mm Hg and pulmonary capillary wedge pressure of 20 mm Hg and a cardiac index of 2.3 liters/min. A left heart catheterization revealed clean coronaries, and alcohol was thought to be the likely etiology of non-ischemic cardiomyopathy in this patient. Urine catecholamine and vanillylmandelic acid were measured and a magnetic resonance angiography of the renal arteries was performed to evaluate for secondary causes of hypertension. This showed non-obstructed renal arteries bilaterally.
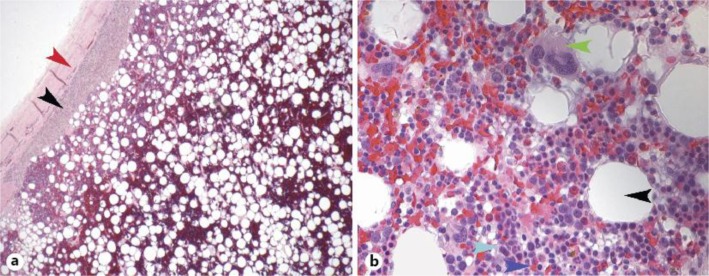
A minimally enhancing 22 × 16 × 23 cm mass was visualized in the left upper and mid-abdomen displacing the spleen and stomach posterosuperiorly, the pancreas anteriorly and the left kidney inferiorly (fig. 1b). The left adrenal gland was not visible. A contrast-enhanced CT scan of the abdomen to further characterize the mass showed no infiltration of the surrounding vasculature or organs, and the left adrenal gland was visualized although it could not be confirmed if the mass was arising from the adrenals (fig. 1a). The suggested radiological differential diagnoses were: liposarcoma, teratoma and myelolipoma. A needle biopsy of this mass showed adipose tissue with foci of extramedullary hematopoiesis consistent with myelolipoma without any evidence of malignancy, confirming this mass to be a benign myelolipoma (fig. 2a, b).
Fig. 1.
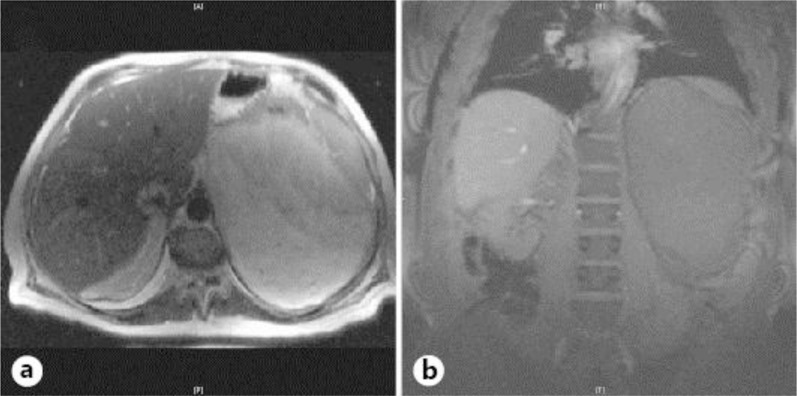
a CT scan of the abdominal mass seen in transverse view. b MRI scan of the abdominal mass seen in coronal view.
Fig. 2.
a Low-power magnification of the biopsy showing capsule (red arrowhead), adrenal tissue (black arrowhead) and an admixture of adipose and hematopoietic cells. b High-power magnification of the biopsy showing adipocytes (black arrowhead), erythroid precursors (cyan arrowhead), lymphoid precursors (blue arrowhead) and megakaryocytes (green arrowhead). Bony spicules and sinusoids are conspicuously absent from the specimen confirming it is a myelolipoma.
Urinary epinephrine, metanephrine and vanillylmandelic acid were within normal limits. A renal perfusion scan showed a small left kidney with reduced perfusion contributing 22% to the overall tubular function without obstructive uropathy. The lower extremity edema was partly ascribed to be due to inferior vena cava compression, and diuretics were decreased due to worsening renal function. Given the bradycardia and severe congestive heart failure, an electrophysiologist was consulted for evaluation and need for pacemaker implantation. Bradycardia was ascribed to be partly due to the Cushing reflex secondary to elevated blood pressure, and as the patient had no symptoms secondary to a low heart rate, pacemaker implantation was not advised. Despite maximum doses of clonidine, isosorbide, hydralazine, losartan and lisinopril, at best a moderate blood pressure control was achieved preoperatively.
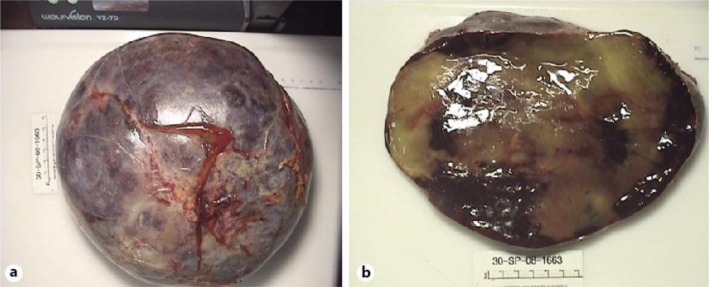
The patient was taken for surgical removal of the mass. During surgery, the mass was found to be soft, highly vascular but well encapsulated and attached only to the left adrenal gland via a small portion that was resected along with the mass (fig. 3a, b). There was minimal blood loss during the surgery, and the patient had an uneventful recovery in the postoperative period. A renal perfusion scan done 8 days after surgical removal showed 36% contribution to the overall tubular function. Blood pressure control improved after surgery and the patient was gradually titrated off clonidine, hydralazine and lisinopril. Bradycardia resolved with few episodes of atrial fibrillation with rapid ventricular response that were controlled with β-blockers and digoxin. The antihypertensive medications that the patient was discharged on were carvedilol 40 mg, valsartan 320 mg and isosorbide mononitrate 240 mg.
Fig. 3.
a Resected specimen showing a well-encapsulated tumor. b Resected specimen cut surface revealing adipose tissue with interspersed areas of hemorrhage.
Discussion
Myelolipomas are composed of fat and hematopoietic tissue and are usually biochemically inactive. They are thought to arise from the reticuloendothelial cells of the adrenal gland in response to repeated stimulation by stress, inflammation and ACTH [11, 12, 13]. Both the adipose and the hematopoietic cells have been noted to have the same pattern of X-chromosome inactivation and have been postulated to arise by clonal proliferation from a common mesenchymal stem cell [14]. Moreover, the chromosomal translocation (3;21)(q25;p11) has been detected in myelolipomas in patients with hematological malignancies, which has led to the speculation of bone marrow origin of these tumors [15]. Extra-adrenal myelolipomas have been documented in the mediastinum, lung, thoracic spine, liver mesentery, spleen, kidney and pelvis [16, 17, 18, 19, 20, 21]. They have been noted to be more common in women [22]. Extra-adrenal myelolipomas may mimic extramedullary hematopoietic tumors especially since bone spicules have been reported [23]. Extramedullary hematopoietic tumors generally have a higher content of erythroid cells and less fat tissue compared to extra-adrenal myelolipomas which have a relatively higher number of lymphoid cells [24].
Adrenal myelolipomas are best imaged by CT due to the high negative attenuation value. However, calcification is seen in approximately 27% of the cases and the presence of a higher proportion of hematopoietic tissues may mimic an adrenal adenoma [4, 25, 26]. Ultrasonography is not a very useful diagnostic modality, especially if the tumors are smaller in size (<2 cm). An MRI has a similar diagnostic utility as a CT scan in adrenal myelolipomas with a high fat content [27]. The gold standard for diagnosis is tissue biopsy which typically shows hematopoietic cells admixed with mature adipose cells [6].
Adrenal myelolipomas can be associated with hyperaldosteronism, congenital adrenal hyperplasia and Cushing's syndrome [28]. They are known to be biochemically inactive but a case of adrenal myelolipoma secreting cortisol has been reported [8]. Adrenal myelolipomas <5 cm are conservatively managed with sequential imaging, but larger masses are preferably resected due to the risk of hemorrhage [29]. Laparoscopic resection has been successfully used to reduce hospital stay and improve perioperative outcomes, but it is not preferable for tumors >10 cm or in tumors with infiltration or extensive adhesions [30, 31]. Our patient was fortunate not to have any hemorrhagic complications. His clinical presentation was compatible with symptoms related to mass effect of the giant myelolipoma, which resolved after surgical resection.
Conclusion
Adrenal myelolipomas are benign tumors with favorable outcomes when diagnosed in a timely manner. Their insidious clinical course becomes apparent only when mass effects are manifested in the form of an abdominal mass, flank pain, hypertension and pedal edema. It is important to differentiate them from other malignancies, such as liposarcoma, and to ensure that the myelolipoma is not biochemically active. Adequate follow-up of small masses and surgical resection of large masses are the standard of care which provide for lesser morbidity and mortality.
Disclosure Statement
The authors declare that there was no funding for this study.
References
- 1.Gierke E. Über Knochenmarksgewebe in der Nebenniere. Beitr Path Anat. 1905;7:311–324. [Google Scholar]
- 2.Oberling C. Les formations myelolipomateuses. Bull Assoc Fr Etud Cancer. 1929;18:234–246. [Google Scholar]
- 3.Porcaro AB, Novella G, Ficarra V, Cavalleri S, Antoniolli SZ, Curti P. Incidentally discovered adrenal myelolipoma. Report on 3 operated patients and update of the literature. Arch Ital Urol Androl. 2002;74:146–151. [PubMed] [Google Scholar]
- 4.Lam KY, Lo CY. Adrenal lipomatous tumours: a 30 year clinicopathological experience at a single institution. J Clin Pathol. 2001;54:707–712. doi: 10.1136/jcp.54.9.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Amendolara M, Barbarino C, Bucca D, Guarnieri F, Novello GB, Romano FM, et al. Giant and bilateral adrenal myelolipoma. Case report (in Italian) G Chir. 2008;29:85–88. [PubMed] [Google Scholar]
- 6.Kenney PJ, Wagner BJ, Rao P, Heffess CS. Myelolipoma: CT and pathologic features. Radiology. 1998;208:87–95. doi: 10.1148/radiology.208.1.9646797. [DOI] [PubMed] [Google Scholar]
- 7.Bovo G, Picozzi SC, Viganò P, Giuberti A, Casu M, Manganini V, Mazza L, Strada GR. Giant adrenal myelolipoma: report of a case and review of the literature. Minerva Urol Nefrol. 2007;59:455–458. [PubMed] [Google Scholar]
- 8.Brogna A, Scalisi G, Ferrara R, Bucceri AM. Giant secreting adrenal myelolipoma in a man: a case report. J Med Case Rep. 2011;5:298. doi: 10.1186/1752-1947-5-298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cristofaro MG, Lazzaro F, Fava MG, Aversa C, Musella M. Giant adrenal myelolipoma: a case report and review of the literature (in Italian) Ann Ital Chir. 2004;75:677–681. [PubMed] [Google Scholar]
- 10.Olobatuyi FA, Maclennan GT. Myelolipoma. J Urol. 2006;176:1188. doi: 10.1016/j.juro.2006.06.095. [DOI] [PubMed] [Google Scholar]
- 11.Han M, Burnett AL, Fishman EK, Marshall FF. The natural history and treatment of adrenal myelolipoma. J Urol. 1997;157:1213–1216. [PubMed] [Google Scholar]
- 12.Meyer A, Behrend M. Presentation and therapy of myelolipoma. Int J Urol. 2005;12:239–243. doi: 10.1111/j.1442-2042.2005.01034.x. [DOI] [PubMed] [Google Scholar]
- 13.Romano G, Cocchiara G, Calderone F, Buscemi G, Gioè FP, Alongi G, Buscemi G, Romano M. Laparoscopic treatment of adrenal myelolipoma: case report and review of the literature (in Italian) G Chir. 2006;27:228–231. [PubMed] [Google Scholar]
- 14.Bishop E, Eble JN, Cheng L, Wang M, Chase DR, Orazi A, O'Malley DP. Adrenal myelolipomas show nonrandom X-chromosome inactivation in hematopoietic elements and fat: support for a clonal origin of myelolipomas. Am J Surg Pathol. 2006;30:838–843. doi: 10.1097/01.pas.0000202044.05333.17. [DOI] [PubMed] [Google Scholar]
- 15.Chang KC, Chen PI, Huang ZH, Lin YM, Kuo PL. Adrenal myelolipoma with translocation (3;21)(q25;p11) Cancer Genet Cytogenet. 2002;134:77–80. doi: 10.1016/s0165-4608(01)00592-1. [DOI] [PubMed] [Google Scholar]
- 16.Pascual Garcia X, Bujons Tur A, Rodriguez Faba O, Gomez Ruiz JJ, Palou Redorta J, Villavicencio Mavrich H. Extraadrenal perirenal myelolipoma: report of a case and review of the literature (in Spanish) Actas Urol Esp. 2007;31:932–934. doi: 10.1016/s0210-4806(07)73751-8. [DOI] [PubMed] [Google Scholar]
- 17.Huang W, Zhao Y, Yin X, Qi Q. Primary myelolipoma of the lung: a case of report and review of literature. Pol J Pathol. 2012;63:204–206. doi: 10.5114/pjp.2012.31507. [DOI] [PubMed] [Google Scholar]
- 18.Vaziri M, Sadeghipour A, Pazooki A, Shoolami LZ. Primary mediastinal myelolipoma. Ann Thorac Surg. 2008;85:1805–1806. doi: 10.1016/j.athoracsur.2007.11.023. [DOI] [PubMed] [Google Scholar]
- 19.Radhi J. Hepatic myelolipoma. J Gastrointestin Liver Dis. 2010;19:106–107. [PubMed] [Google Scholar]
- 20.Cina SJ, Gordon BM, Curry NS. Ectopic adrenal myelolipoma presenting as a splenic mass. Arch Pathol Lab Med. 1995;119:561–563. [PubMed] [Google Scholar]
- 21.Ghaouti M, Znati K, Jahid A, Zouaidia F, Bernoussi Z, Mahassini N. Renal myelolipoma: a rare extra-adrenal tumor in a rare site: a case report and review of the literature. J Med Case Rep. 2013;7:92. doi: 10.1186/1752-1947-7-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.George SA, Manipadam MT, Thomas R. Primary myelolipoma presenting as a nasal cavity polyp: a case report and review of the literature. J Med Case Rep. 2012;6:127. doi: 10.1186/1752-1947-6-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sagan D, Zdunek M, Korobowicz E. Primary myelolipoma of the chest wall. Ann Thorac Surg. 2009;88:e39–e41. doi: 10.1016/j.athoracsur.2009.06.085. [DOI] [PubMed] [Google Scholar]
- 24.Fowler MR, Williams RB, Alba JM, Byrd CR. Extra-adrenal myelolipomas compared with extramedullary hematopoietic tumors: a case of presacral myelolipoma. Am J Surg Pathol. 1982;6:363–374. doi: 10.1097/00000478-198206000-00009. [DOI] [PubMed] [Google Scholar]
- 25.Cintron E, Quintero EC, Perez MR, Ramirez-de Arellano GA, Manich BV. Computed tomography, sonographic, and radiographic findings in adrenal myelolipoma. Urology. 1984;23:608–610. doi: 10.1016/0090-4295(84)90085-2. [DOI] [PubMed] [Google Scholar]
- 26.Musante F, Derchi LE, Zappasodi F, Bazzocchi M, Riviezzo GC, Banderali A, Cicio GR. Myelolipoma of the adrenal gland: sonographic and CT features. AJR Am J Roentgenol. 1988;151:961–964. doi: 10.2214/ajr.151.5.961. [DOI] [PubMed] [Google Scholar]
- 27.Musante F, Derchi LE, Bazzocchi M, Avataneo T, Gandini G, Pozzi Mucelli RS. MR imaging of adrenal myelolipomas. J Comput Assist Tomogr. 1991;15:111–114. doi: 10.1097/00004728-199101000-00017. [DOI] [PubMed] [Google Scholar]
- 28.Jung SI, Kim SO, Kang TW, Kwon DD, Park K, Ryu SB. Bilateral adrenal myelolipoma associated with hyperaldosteronism: report of a case and review of the literature. Urology. 2007;70:1223.e11–1223.e13. doi: 10.1016/j.urology.2007.09.046. [DOI] [PubMed] [Google Scholar]
- 29.Russell C, Goodacre BW, vanSonnenberg E, Orihuela E. Spontaneous rupture of adrenal myelolipoma: spiral CT appearance. Abdom Imaging. 2000;25:431–434. doi: 10.1007/s002610000061. [DOI] [PubMed] [Google Scholar]
- 30.Novitsky YW, Czerniach DR, Kercher KW, Perugini RA, Kelly JJ, Litwin DE. Feasibility of laparoscopic adrenalectomy for large adrenal masses. Surg Laparosc Endosc Percutan Tech. 2003;13:106–110. doi: 10.1097/00129689-200304000-00009. [DOI] [PubMed] [Google Scholar]
- 31.Tsuru N, Suzuki K, Ushiyama T, Ozono S. Laparoscopic adrenalectomy for large adrenal tumors. J Endourol. 2005;19:537–540. doi: 10.1089/end.2005.19.537. [DOI] [PubMed] [Google Scholar]