Abstract
Observational studies have revealed that higher serum vitamin E concentrations and increased vitamin E intake and vitamin E supplementation are associated with beneficial effects on glycaemic control in type 2 diabetes mellitus (T2DM). However, whether vitamin E supplementation exerts a definitive effect on glycaemic control remains unclear. This article involves a meta-analysis of randomised controlled trials of vitamin E to better characterise its impact on HbA1c, fasting glucose and fasting insulin. PubMed, EMBASE and the Cochrane Library were electronically searched from the earliest possible date through April 2013 for all relevant studies. Weighted mean difference (WMD) was calculated for net changes using fixed-effects or random-effects models. Standard methods for assessing statistical heterogeneity and publication bias were used. Fourteen randomised controlled trials involving individual data on 714 subjects were collected in this meta-analysis. Increased vitamin E supplementation did not result in significant benefits in glycaemic control as measured by reductions in HbA1c, fasting glucose and fasting insulin. Subgroup analyses revealed a significant reduction in HbA1c (−0.58%, 95% CI −0.83 to −0.34) and fasting insulin (−9.0 pmol/l, 95% CI −15.90 to −2.10) compared with controls in patients with low baseline vitamin E status. Subgroup analyses also demonstrated that the outcomes may have been influenced by the vitamin E dosage, study duration, ethnic group, serum HbA1c concentration, and fasting glucose control status. In conclusion, there is currently insufficient evidence to support a potential beneficial effect of vitamin E supplementation on improvements of HbA1c and fasting glucose and insulin concentrations in subjects with T2DM.
Introduction
Type 2 diabetes mellitus (T2DM) is a global health problem affecting almost 336 million people worldwide or approximately 8.3% of the world population, and the number of affected individuals will dramatically increase in the next 20 years [1]. Poor glycaemic control is responsible for the long-term negative outcomes in T2DM subjects, including microvascular and macrovascular complications, such as cardiovascular events, renal failure, blindness and peripheral neuropathy [2]. Appropriate management of hyperglycaemia is needed to reduce morbidity and the number of complications associated with T2DM.
The association between oxidative stress and T2DM has long been recognised and is based on the observation that hyperglycaemia, hyperinsulinaemia, and insulin resistance can enhance free radical generation and thus contribute to oxidative stress [3]. Oxidative stress can in turn promote haemoglobin glycation [4], [5] and impair insulin signalling and β-cell insulin secretion in T2DM [6], [7]. Thus, it is reasonable to postulate that antioxidants, such as vitamin E, may have benefits effects on glycaemic control in T2DM.
Vitamin E is a powerful lipid-soluble antioxidant that reduces oxidative stress and decreases the oxidative stress-associated damage in T2DM [8]. Anti-hyperglycaemic effects of vitamin E have been hypothesised, tested in laboratory and human studies, and have gained biological plausibility. Observational studies have indicated that vitamin E intake was inversely related to a risk of T2DM [9]. Furthermore, in patients with established T2DM, regular vitamin E supplementation is associated with a significant improvement in glycaemic control [10], which provide justification for trials evaluating vitamin E supplementation and glycaemic control in T2DM subjects.
There are several possible mechanisms underlying the association between vitamin E and glucose metabolism. Vitamin E may prevent the glycosylation of haemoglobin by inhibiting the formation of advanced glycosylation endproducts (AGEs) [11]. Vitamin E can also mitigate the long-term pancreatic β-cell dysfunction caused by oxidative stress in T2DM [12]. Vitamin E supplementation was also found to be associated with other disorders, including Nonalcoholic Steatohepatitis [13] and age-related cataract [14], while there is no evidence to support the use of vitamin E supplements for prevention of cardiovascular diseases [15] and stroke [16].
Vitamin E (tocopherols and tocotrienols) is an essential micronutrient that is acquired primarily through the consumption of fruit, vegetable oils, nuts, and green leafy vegetables [17]. In addition, vitamin E capsules are readily available and often supplied for healthy people, and approximately 12.7% of the adult population in the United States takes vitamin E as dietary supplements [18]. Given that vitamin E intake is simple, safe, and inexpensive, the vitamin E supplementation may represent a common and easily obtainable treatment for glycaemic control.
The objective of this meta-analysis was to combine evidence from randomised controlled trials (RCTs) to assess the effect of vitamin E supplementation on change in HbA1c, a well-established clinical marker of long-term glycaemic control, and on fasting glucose and fasting insulin levels in subjects with T2DM.
Experimental Methods
Search selection
We searched the PubMed(http://www.ncbi.nlm.nih.gov/pubmed/), EMBASE(http://www.embase.com/) and Cochrane library databases (http://www.cochrane.org/) from their inception until April 2013 for trials that examined the effects of vitamin E on HbA1c, glucose control and insulin sensitivity in humans. The following search terms were used: (vitamin E OR alpha-tocopherol OR tocopherols OR tocotrienols) AND (glucose OR hyperglycaemia OR glyc(a)emia OR insulin OR insulin sensitivity OR insulin resistance OR HbA1c OR glycated protein or glycated albumin OR fructosamine OR diabetes). The search was confined to human studies. Manual recursive searches were also conducted on references from review articles and published randomised controlled trials that met the inclusion criteria. There was no restriction by language or publication status.
Study selection
Potentially relevant studies were selected based on the following inclusion criteria: (1) randomised controlled trials with either a parallel or a crossover design; (2) adults with T2DM supplied with a vitamin E supplementation that is currently FDA-approved for at least 6 weeks treatment; (3) fasting glucose, fasting insulin, HbA1c or glycated haemoglobin levels were describes in the clinical trials; And (4) studies used a concurrent control group, and the difference between the treatment and the control groups was limited to vitamin E. Exclusion criteria were as follows: (1) trials that enrolled children, pregnant women, or patients with type 1 diabetes; (2) patients were supplied with both vitamin E and other antioxidants; and (3) trials with abstracts only.
Data extraction
Two investigators independently selected the trials and extracted the data, and disagreements or uncertainties were resolved by consensus. Extracted data included study characteristics (author, publication status, year, design), treatment characteristics (type of intervention, dosage, frequency, duration), and participant characteristics (sample, mean age, gender, race/ethnicity, serum vitamin E level, mean HbA1c and fasting glucose levels). Primary outcome measures included the net changes in plasma concentrations of glycated haemoglobin (HbA1c), fasting glucose levels, and fasting insulin concentrations after vitamin E supplementation. The secondary outcome included changes in the homeostatic model assessment of insulin resistance (HOMA-IR), postprandial glucose and insulin concentrations, total cholesterol (TC), triglycerides (TG), high density lipoprotein cholesterol (HDL-c), low density lipoprotein cholesterol (LDL-c), systolic blood pressure (SBP) and diastolic blood pressure (DBP).
Quality assessment
The quality of the included studies was assessed using the Jadad scoring system [19]. Trials scored one point for each area addressed in the study (randomisation, double-blinding, description of withdrawals and dropouts, generation of random numbers and allocation concealment), with a possible score between 0 and 5. Trials were subclassified as high-quality (Jadad score ≥4) or low-quality (Jadad score of 2 or 3).
Statistical analyses
The statistical analysis was performed with RevMan software version 5 (Cochrane Collaboration). The mean change in the outcome variables from baseline was treated as a continuous variable, and the weighted mean difference was calculated as the difference between the mean in the vitamin E group and control groups. The significance of any mean changes was evaluated using the WMD and 95% CIs with fixed-effect or random-effect models. If the variances for the mean changes were not reported directly, the variances were imputed from the exact P value, CIs, or individual variances from the vitamin E group and the control group [20]. Missing variances for paired differences were imputed by assuming a correlation coefficient of 0.5 between variances at baselines and ends of trials according to the method of Follmann et al [21]. We assumed equal variances during the trial and between intervention and control groups.
Heterogeneity was assessed using the Cochrane Q statistic and the inconsistency index (I2), where a P value<0.10 was considered significant, and measured in-consistency (I2) statistics, with a measurement >50% taken to indicate substantial heterogeneity [22]. A random-effect model was used to analyse the results with significant heterogeneity. Otherwise, a fixed-effect model was used for outcomes without obvious heterogeneity [23]. Publication bias was assessed using funnel plots and Egger's test [24]. To explore the possible influence of covariates on net changes, subgroup analyses were conducted to evaluate the effects of the following factors on primary outcomes (HbA1c, fasting glucose and insulin concentrations): vitamin E dosages, ethnic groups, vitamin E duration, baseline HbA1c concentrations, baseline fasting glucose concentrations and the baseline serum vitamin E concentrations of the participants. A P value <0.05 was considered to be statistically significant in this trial, unless otherwise specified.
Results
Literature search
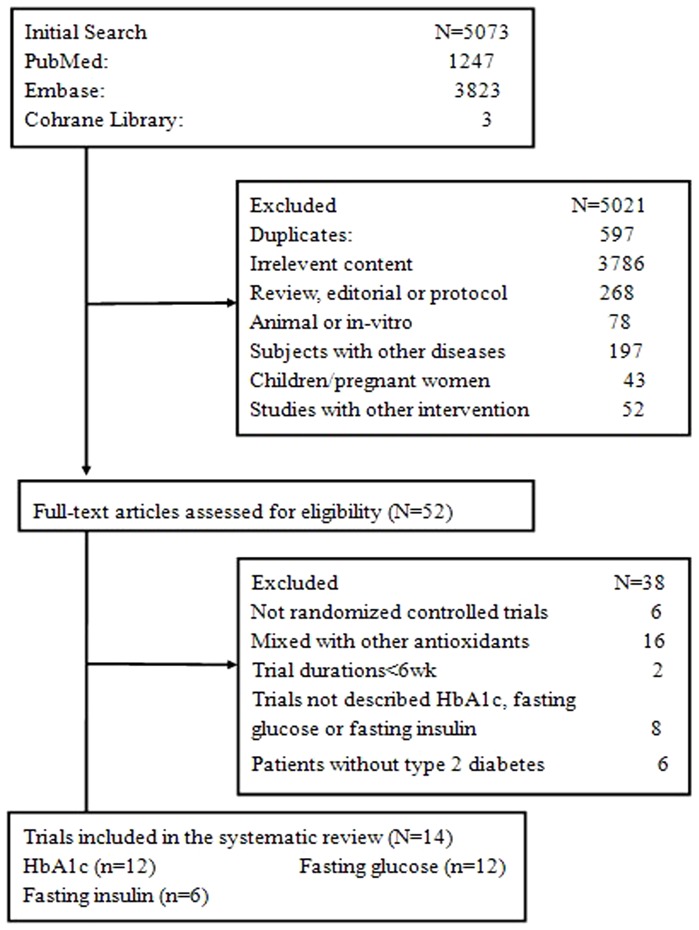
We identified 5073 potentially relevant articles from the PubMed, EMBASE and Cochrane library databases, 5021 of which were determined to be irrelevant after review of the titles and abstracts. 52 trials proceeded to a detailed evaluation of the complete report, and a further 38 were excluded. A total of 14 trials met the selection criteria for the current meta-analysis. A flow describing article selection for this meta-analysis is shown in Figure 1.
Figure 1. Flow diagram of the process of article selection for meta-analysis.
Study description
Fourteen published studies with a total of 714 participants, including 363 subjects in the vitamin E group and 351 patients in the control group, were identified in this meta-analysis [25]–[38]. The characteristics of the studies and participants are outlined in Table 1. Three primary outcomes measures were examined: HbA1c (12 studies) [25]–[36], fasting glucose (12 studies) [25]–[31], [34], [36]–[38] and fasting insulin (6 studies) [29]–[31], [36]–[38]. Among them, one trial examined the effect of vitamin E in both type 1 and type 2 diabetes; only groups with type 2 diabetes were selected for each run of analysis [33]. Twelve of the fourteen included studies were exhibited a parallel design [26]–[34], [36]–[38], whereas the other two were crossover studies [25], [35]. The dosage of vitamin E supplements ranged from 200 to 1600 IU/d, with an intervention duration ranged from 6 to 27 weeks. Nine studies focused on subjects in western countries [25], [26], [28]–[30], [33], [34], [37], [38] and five studies focused on subjects in Asian countries [27], [31], [32], [35], [36]. Most studies enrolled patients with normal serum vitamin E levels [25]–[29], [31]–[35], [37], whereas three other studies enrolled subjects with low serum vitamin E levels [30], [36], [38]. The participants in eight studies exhibited poor fasting glucose control (>8.0 mmol/L) at baseline [25], [26], [28]–[31], [34], [36], and the other four trials exhibited good fasting glucose control at baseline [27], [35], [37], [38]. Four studies exhibited high serum HbA1c (>8.0) levels at baseline [30], [31], [34], [36], whereas the other eight studies exhibited well-controlled serum HbA1c (<8.0) levels at baseline [25]–[29], [32], [33], [35].
Table 1. Characteristic of experimental trials included in the meta-analysis.
| Trial | Country | Vitamin E group intervention | Control group | Duration weeks(n) | Study Size(n) | Mean age (year) | Male(%) | Diabetes duration(years) | Trial design | Baseline HbA1c(%) | Baseline FPG(mmol/l) | Baseline Vit E (mmol/l) | ||||
| T | C | T | C | T | C | T | C | |||||||||
| Paolisso 1993 | Italy | d-α-tocopherol 900 mg/d | Placebo | 12 | 25 | 25 | 71.3 | 71.3 | — | — | 8.4 | 8.4 | C1 | 7.0 | 8.2 | 16.3 |
| Reaven 1995 | USA | dl-α-tocopherol 1600 IU/d | Placebo | 10 | 10 | 11 | 60.8 | 61.8 | 100 | 100 | 8.6 | 8.3 | P | 7.7 | 9.9 | 35 |
| Tutuncu 1998 | Turkey | dl-α-tocopherol acetate 900 mg/d | Placebo | 24 | 11 | 10 | 51.2 | 59.3 | 18.2 | 10 | 8.8 | 8.9 | P | 7.5 | 7.2 | — |
| Gazis 1999 | UK | α-tocopherol 1600 IU/d | Placebo | 8 | 23 | 25 | 56.4 | 57.9 | 65.2 | 84 | 4.3 | 4.8 | P | 6.9 | 9.6 | 27.8 |
| Paolisso 2000 | Italy | Vitamin E 600 mg | Placebo | 8 | 20 | 20 | 58.3 | 56.7 | 45 | 60 | 7.8 | 7.8 | P | 7.7 | 8.3 | 16.9 |
| Manzella 2001 | Italy | All-rac-α-tocopherolAcetate 600 mg/d | Sodium citrate | 16 | 25 | 25 | 64.3 | 65.1 | — | — | — | — | P | 8.2 | 9.1 | 7.1 |
| Park 2002 | Korea | α-tocopherol 200 mg/d | Digestive pill | 8 | 48 | 50 | 49.4 | 49.5 | 56.3 | 62.8 | 7.9 | 8.3 | P | 10.9 | 10.4 | 25.5 |
| Chen 2004 | China | Vitamin E 200 mg | Placebo | 52 | 41 | 41 | 52 | 52.4 | 51.2 | 48.8 | <1 | <1 | P | 7.6 | — | — |
| Economides 2005 | USA | All-rac-α-tocopherol Acetate 1800 IU/d | Soybeanoil | 24 | 25 | 20 | 59 | 59 | 56.7 | 56.7 | 9 | 9 | P | 7.2 | — | 28.9 |
| Ble-Castillo 2005 | Mexico | α-tocopherol 800 IU/d | Corn strach | 6 | 13 | 21 | 51.3 | 55.3 | 0 | 0 | <14 | <14 | P | 10.9 | 12.8 | — |
| Baliarsingh 2005 | India | Tocotrienols 6 mg/kg | Rice brain oil | 8 | 10 | 9 | 48.5 | 52.6 | 50 | 55.6 | 5.3 | 4.3 | C1 | 7.6 | 6.3 | — |
| Boshtam 2005 | Iran | Vitamin E 200 IU/d | Placebo | 27 | 50 | 50 | 52.8 | 54.5 | — | — | 5.7 | 13.3 | P | 9.9 | 10.8 | 10.5 |
| Ward 2007 | Australia | RRR-α-tocopherol or mixd tocopherols 500 mg/d | Soy bean oil | 6 | 37 | 18 | 61 | 62 | 67.5 | 88.9 | — | — | P | — | 7.3 | — |
| de Oliveira 2011 | Brazil | α-tocopherol 800 IU/d | Placebo | 16 | 25 | 26 | 62 | 63 | 72 | 57.7 | — | — | P | — | 7.2 | 4.7 |
FPG: fasting plasma glucose; vitE: vitamin E; mixed tocopherols: 60%γ-,25% δ- and 15% α-tocopherol; HbA1c: glycated hemoglobin; USA: United States of America; UK: The United Kingdom; T:treatment group; C: control group; C1: crossover design; P: Parallel design.
Primary outcomes
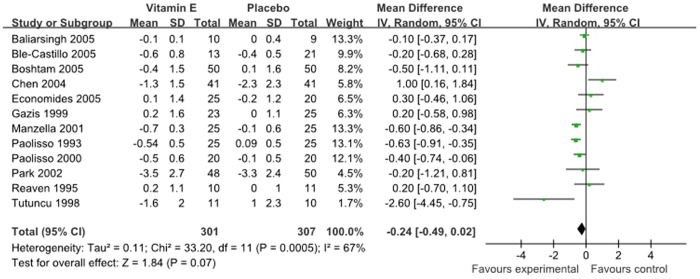
Primary outcome measures included changes in HbA1c, fasting glucose and fasting insulin. Twelve studies with 608 subjects, including 301 subjects in vitamin E group and 307 subjects in placebo group, reported HbA1c at baseline and follow-up. There was no significant reduction in the percentage of HbA1c (−0.24, 95% CI: −0.49 to 0.02]; P = 0.07, Figure 2), with significant evidence of interstudy heterogeneity (I2 = 67%; P = 0.0005) in subjects with vitamin E supplements, so we reported the results from random-effects models.
Figure 2. Forest plot of randomised controlled trials investigate the effect of vitamin E supplementation on HbA1c.
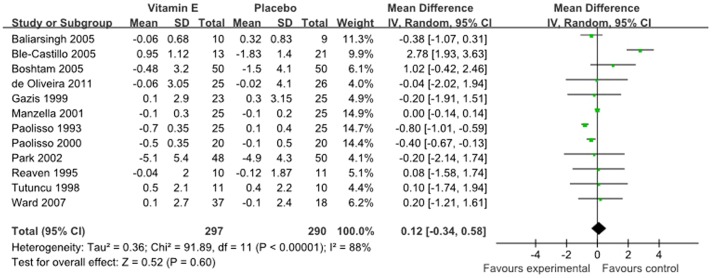
The results for fasting glucose were reported in 12 studies representing 587 participants, including 290 controls. Meta-analyses suggested that vitamin E supplementation, compared with a placebo or control, resulted in no statistically significant improvement (0.12 mmol/l, 95% CI: −0.34 to 0.58 mmol/l, P = 0.60, Figure 3) in fasting glucose status. The degree of heterogeneity was significant (I2 = 88%; P<0.00001), and random-effects models were performed in this meta-analysis.
Figure 3. Forest plot of randomised controlled trials investigate the effect of vitamin E supplementation on fasting glucose.
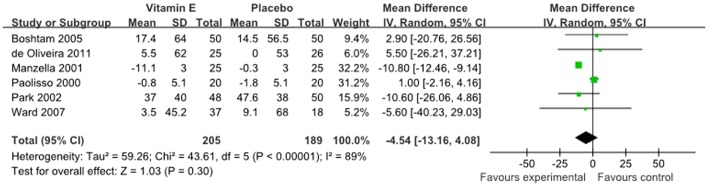
The mean change in fasting insulin concentrations was reported in 6 studies, representing 394 participants, and was not significantly decreased in the intervention groups (−4.54 pmol/l, 95%CI: −13.16 to 4.08 pmol/l, P = 0.30, Figure 4) compared to control arms. Significant heterogeneity (I2 = 89%; P<0.00001) was observed, and the results are reported on the basis of random-effects models.
Figure 4. Forest plot of randomised controlled trials investigate the effect of vitamin E supplementation on fasting insulin.
Subgroup analysis
Subgroup analyses were conducted to explore the dose-effect relationship, study-duration effects, ethnic group variation, and differences in baseline HbA1c, fasting glucose and serum vitamin E concentrations. The results are summarised in Table 2.
Table 2. Subgroup analyses of HbA1c, fasting glucose and fasting insulin stratified by previously defined study characteristics.
| Variables | HbA1c(%) | Fasting glucose(mmol/l) | Fasting insulin(pmol/l) | ||||||
| No.of tirals | Mean difference(95%CI) | P for heterogeneity | No.of tirals | Mean difference(95%CI) | P for heterogeneity | No.of tirals | Mean difference(95%CI) | P for heterogeneity | |
| Subgroup analysis | |||||||||
| Vitamin E dose | |||||||||
| ≤400 mg/d | 4 | −0.0(−0.52,0.51) | 0.04 | 3 | 0.01(−0.85,0.87) | 0.23 | 2 | −6.56(−19.50,6.38) | 0.35 |
| >400 mg/d | 8 | −0.35(−0.63,−0.07) | 0.01 | 9 | 0.15(−0.38,0.68) | <0.0001 | 4 | −4.13(−14.48,6.21) | <0.0001 |
| Duration | |||||||||
| Shorter term (<12 wk) | 6 | −0.17(−0.35,0.01) | 0.62 | 7 | 0.31(−0.68,1.29) | <0.001 | 3 | 0.33(−5.49,4.82) | 0.33 |
| Longer term (≥12 wk) | 6 | −0.33(−0.79,0.13) | 0.0004 | 5 | −0.14(−0.78,0.50) | <0.001 | 3 | −9.0(−15.90, −2.10) | 0.32 |
| BaselineHbA1c (%) | |||||||||
| <8 | 8 | −0.12(−0.49,0.25) | 0.0002 | 6 | −0.55(−0.81, −0.29) | 0.19 | 1 | 1.00(−2.16,4.16) | - |
| ≥8 | 4 | −0.49(−0.71, −0.28) | 0.5 | 4 | 0.95(−0.70,2.59) | <0.001 | 5 | −10.67(−12.31, −9.02) | 0.64 |
| Baseline fasting glucose (mmol/l) | |||||||||
| <8 | 2 | −1.18(−3.60,1.25) | 0.009 | 4 | −0.22(−0.78,0.35) | 0.87 | 2 | 0.44(−22.95,23.82) | 0.64 |
| ≥8 | 8 | −0.44(−0.62, −0.26) | 0.27 | 8 | 0.21(−0.34,0.77) | <0.0001 | 4 | −5.15(−14.48,4.18) | <0.0001 |
| Ethnicity | |||||||||
| Asian | 5 | −0.21(−0.84,0.43) | 0.004 | 4 | −0.10(−0.67,0.46) | 0.39 | 2 | −6.56(−19.50,6.38) | 0.35 |
| Western | 7 | −0.34(−0.58, −0.09) | 0.05 | 8 | 0.15(−0.4,0.7) | <0.0001 | 4 | −4.13(−14.48,6.21) | <0.0001 |
| Baseline serum Vitamin E(mmol/l) | |||||||||
| Normal | 6 | −0.23(−0.56,0.09) | 0.08 | 5 | −0.57(−0.87, −0.28) | 0.16 | 2 | −2.23(−12.42,7.96) | 0.15 |
| Low | 2 | −0.58(−0.83, −0.34) | 0.77 | 3 | 0.01(−0.13,0.15) | 0.39 | 3 | −9.0(−15.90, −2.10) | 0.32 |
In general, the reduction in HbA1c and fasting insulin were greater in studies with low serum vitamin E concentrations, with mean changes of −0.58 (95% CI: −0.83, −0.34) and −9.0 pmol/l (95% CI: −15.90, −2.10 pmol/l) respectively. In contrast, fasting glucose decreased significantly in studies with normal vitamin E concentrations (−0.57 mmol/l; 95% CI: −0.87, −0.28 mmol/l). Trials with low serum vitamin E concentrations were limited in this meta-analysis, so more trials are needed in the following research area.
Subgroup analysis also suggested that HbA1c and fasting insulin were reduced to a greater degree in studies with higher HbA1c concentrations (≥8%), with mean changes of −0.49 (95% CI: −0.71, −0.28) and −10.67 pmol/l (95% CI: −12.31, −9.02 pmol/l). In contrast, there was a negative dose-response relationship between HbA1c concentrations (<8%) and the reduction in fasting glucose. Significant reductions in HbA1c were also observed in studies with poor fasting glucose concentrations (≥8 mmol/l) (−0.44 mmol/l; 95% CI: −0.62, −0.26 mmol/l).
A significant reduction in HbA1c was observed in studies with higher vitamin E doses (>400 mg/d), with a net change of −0.35 (95% CI: −0.63, −0.07). Similarly, concentrations of fasting insulin declined significantly in studies with longer vitamin E treatment durations (>12 wk) (−9.0 pmol/l; 95% CI: −15.90, −2.10 pmol/l). Western population with vitamin supplementation also yielded significant effect on HbA1c (−0.34; 95% CI: −0.58, −0.09) compared with controls. In conclusion, although there are significant results in subgroup analysis, more trials with larger subjects are needed to make a definite conclusion.
A sensitivity analysis was performed by excluding three trials (Economides 2005, Baliarsingh 2005, Ward 2007) which used an oil for placebo. The exclusion of trials resulted in greater reductions in HbA1c (−0.30, 95% CI: −0.59, −0.02; P = 0.04), which significantly influenced the results. While, after exclusion these trials, the pooled reductions in fasting glucose and fasting insulin were not significantly influenced, with 0.19 mmol/l (95% CI: −0.33, 0.71 mmol/l; P = 0.47) and −4.33 pmol/l (95% CI: −13.06, 4.41 pmol/l; P = 0.33), respectively. In a sensitivity analysis, after exclusion the trial (Park 2002) with digestive pill, the pooled reductions in HbA1c, fasting glucose and fasting insulin were not significantly influenced, with −0.24 (95% CI: −0.50, 0.03; P = 0.08), 0.12 mmol/l (95% CI: −0.35, 0.59 mmol/l; P = 0.62) and −3.06 pmol/l (95% CI: −12.82, 6.70 pmol/l; P = 0.54), respectively.
Secondary outcomes
Pooled analysis of the changes in secondary outcomes was performed by calculating or estimating the weighted averages. These changes are summarised in Tables 3. Vitamin E resulted in no significant decrease in HOMA-IR (−0.32, 95% CI:−0.65 to 0.01, P = 0.06) or postprandial glucose (−0.20, 95% CI: −1.07 to 0.67, P = 0.65) compared with controls. Vitamin E supplementation also appeared to have no statistically significant effect on lipid metabolism, including triglyceride (TG), total cholesterol (TC), low-density lipoprotein-cholesterol (LDL-C), and high-density lipoprotein-cholesterol (HDL-C), compared with placebo. Furthermore, vitamin E supplementation failed to significantly alter either systolic blood pressure (SBP) or diastolic blood pressure (DBP) compared with the control group.
Table 3. Summary of effect sizes (weighted mean difference) for secondary outcomes.
| Variables | No. of comparison | Sample size | Net change(95%CI) | P | Test for heterogenenity | ||
| T | C | I2(%) | P | ||||
| HOMA index | 3 | 70 | 71 | −0.32(−0.65,0.01) | 0.06 | 89 | 0.0001 |
| Postprandial glucose | 3 | 69 | 69 | −0.20(−1.07,0.67) | 0.65 | 0 | 0.77 |
| Triglyceride(mmol/l) | 8 | 201 | 212 | −0.02(−0.29,0.25) | 0.90 | 96 | <0.0001 |
| Total-C(mmol/l) | 9 | 254 | 252 | −0.29(−0.85,0.27) | 0.31 | 99 | <0.0001 |
| HDL-c(mmol/l) | 9 | 217 | 223 | −0.00(−0.03,0.02) | 0.70 | 20 | 0.27 |
| LDL-c(mmol/l) | 8 | 192 | 203 | −0.24(−0.78,0.29) | 0.37 | 98 | <0.0001 |
HOMA, homeostasis model assessment; Total-C, total cholesterol; HDL-C, high-density lipoprotein-cholesterol;LDL-C, low-density lipoprotein-cholesterol; CI, confidence interval. T: treatment group; C: control group.
Study quality and publication bias
The quality of these fourteen randomised controlled trials was variable (Table 4). Only one study was classified as high quality (Jadad score of 4) [37], and thirteen studies were classified as low quality (Jadad score of 2 or 3) [25]–[36], [38]. Randomisation was a prerequisite for inclusion in this meta-analysis, so all studies were randomised, but only two trails mentioned the method for sequence generation [32], [37]. Thirteen studies were double-blind [25]–[31], [33]–[38], and one study did not describe blinding [32]. Ten trials presented a clear explanation for withdrawals and dropouts in each group [25], [26], [28], [29], [31]–[35], [37].
Table 4. Quality assessment of included studies.
| Authors | Randomisation | Allocation concealment | Random sequence generation | Blinding | Reporing of withdrawals | Jaded score |
| Paolisso 1993 | Y | U | U | Y | Y | 3 |
| Reaven 1995 | Y | U | U | Y | Y | 3 |
| Tutuncu 1998 | Y | U | U | Y | U | 2 |
| Gazis 1999 | Y | U | U | Y | Y | 3 |
| Paolisso 2000 | Y | U | U | Y | Y | 3 |
| Manzella 2001 | Y | U | U | Y | U | 2 |
| Park 2002 | Y | U | U | Y | Y | 3 |
| Chen 2004 | Y | U | Y | U | Y | 3 |
| Economides 2005 | Y | U | U | Y | Y | 3 |
| Ble-Castillo 2005 | Y | U | U | Y | Y | 3 |
| Baliarsingh 2005 | Y | U | U | Y | Y | 3 |
| Boshtam 2005 | Y | U | U | Y | U | 2 |
| Ward 2007 | Y | U | Y | Y | Y | 4 |
| Oliveira 2011 | Y | U | U | Y | U | 2 |
Y, yes; U, unclear; Randomisation:the study described as randomized; Random sequence generation: the correct method for generation of random numbers computer random numbers table, shuffled cards or tossed coins, and minimization; Allocation concealment: Adequate concealment was that up to the point of treatment (eg, central randomisation); Double-blinding: masking to both researchers and patients; Reporting of withdrawals: The numbers and reasons for withdrawal in each group had to be stated for a point to be awarded.
The funnel plots of the studies were symmetric for HbA1c, fasting glucose and fasting insulin on visual examination. Furthermore, the results of the Egger's test did not support the existence of publication bias for HbA1c (P = 0.46), fasting glucose (P = 0.87), or fasting insulin (P = 0.27).
Discussion
This meta-analysis of fourteen RCTs with 714 subjects demonstrated that vitamin E supplementation was not associated with the reduction in HbA1C, fasting glucose and fasting insulin. However, significant heterogeneity was detected in all three pooled analyses. Therefore, further subgroup analysis was performed to identify the source of heterogeneity. The subgroup analyses indicated that vitamin E supplementation significantly decreased both HbA1c and fasting insulin in studies with low serum vitamin E concentrations and poorer glycaemic control. Furthermore, larger vitamin E doses and longer study durations also benefited HbA1c and fasting insulin concentrations. Although this meta-analysis failed to identify significant correlations between vitamin E supplements and improvements in glycaemic control, our results suggest that T2DM subjects with low serum vitamin E concentrations or poorer glycaemic control may experience a positive change in glycaemic control after vitamin E supplementation.
Recent mechanistic studies have examined the effects of vitamin E on glycaemic control and provide further evidence for the biological plausibility of these findings. There is now considerable evidence that oxidative stress plays an important role in the glycation of haemoglobin [39] and beta-cell dysfunction [6] in T2DM. Vitamin E, a common antioxidant, suppresses ROS generation in the pancreas and maintains the structural integrity of pancreatic islets in experimental diabetes [8]. Furthermore, there is evidence that vitamins E supplementation inhibits the glycation of haemoglobin, a biomarker for the diagnosis of diabetes in clinical settings, by interrupting glycosylation at an early step in the Maillard reaction [40] or by partially inhibiting the formation of AGEs [11]. Moreover, in addition to its beneficial effects on the preservation of pancreatic beta cell function, vitamin E partially reverses the beta-cell apoptosis caused by oxidative stress [8], [12].
Observational prospective cohorts and case-control studies have been performed to determine the effect of vitamin E supplementation on glycaemic control, although the results are conflicting. Observational prospective cohorts have indicated that low serum vitamin E concentrations are associated with a lower risk of diabetes. A large, 23-year population-based study involving 4304 middle-aged individuals from Finland revealed that, vitamin E intake, including α-tocopherol γ-tocopherol, δ-tocopherol, and β-tocotrienol were significantly associated with a reduced risk of T2DM [9]. In contrast, some RCTs have demonstrated no association between vitamin E supplementation and the risk of T2DM [41]. Case-control trials also demonstrated that vitamin E supplementation exerts beneficial effects on glycaemic control, including serum HbA1c [27], fasting glucose [29] and fasting insulin [30] in T2DM subjects. In contrast, several studies have reported no positive correlations between vitamin E intake and glycaemic control in subjects with T2DM [33].
In this meta-analysis, subgroup analysis was performed on the basis of our predefined variances to identify sources of heterogeneity. It is reasonable to speculate that the treatment effects of vitamin E are influenced by its baseline levels. Subgroup analyses revealed that vitamin E supplementation has a more pronounced effect on HbA1c and fasting insulin in studies with low baseline serum vitamin E concentrations, which is consistent with the findings of an earlier meta-analysis by N. Suksomboon et. al [42]. This increased effect may indicate that extra vitamin E is needed for subjects with vitamin E deficiencies to antagonise oxidative stress, inhibit glycation of haemoglobin and reverse beta cell dysfunction. Our findings also suggest that subjects with poor HbA1c and fasting glucose control would experience the maximum benefit of vitamin E on reducing HbA1c and fasting insulin concentrations, perhaps by controlling excess oxidative stress secondary to hyperglycaemia, inhibiting the glycosylation of haemoglobin, and / or protecting beta cells function in those patients with poor glycaemic control. Subjects with larger doses of vitamin E and longer treatment periods may experience increased benefits of vitamin E supplementation on HbA1c or fasting insulin control. It is possible that shorter T2DM trials may have been insufficient to detect true HbA1c and insulin changes; in addition, smaller doses of vitamin E may not sufficiently elevate vitamin E levels.
The analysis of our secondary outcomes revealed a tendency but no significant improvement in HOMA-IR, postprandial glucose and blood pressure in the vitamin E group compared with controls. In addition, vitamin E supplementation had no significant effect on lipid metabolism, which was consistent with the findings of an earlier meta-analysis by Shekelle PG [43].
The strength of this study was that trials included in this meta-analysis were all RCTs, which allow reliable inferences about causality. In addition, this meta-analysis assessed the effects of vitamin E supplementation on plasma concentrations of fasting glucose and fasting insulin for the first time. Furthermore, subgroup analyses were performed to detect sources of heterogeneity for primary outcomes.
Although we believe that the current meta-analysis provided useful information, some potential limitations should be addressed. First, in this meta-analysis, small trials with limited subjects were collected, which limited the ability to extrapolate the outcomes of this review toward realistic public health recommendations for vitamin E supplementation for glycaemic control in T2DM. Second, there was considerable evidence of trial heterogeneity. Subgroup analyses revealed that vitamin E dosage, trial duration, glycaemic control, ethnic group and serum vitamin E levels may contribute to the variation in trial effects. Therefore, future studies focusing on the effects of different subject characteristics on changes in glycaemic control after vitamin E supplementation are needed to confirm our results. Third, the quality of RCTs included in this meta-analysis was varied. Of the fourteen trials, only one trial was a high-quality study; the other studies were of low quality, which may affect the confidence of this meta-analysis. Publication bias is also a potential limitation. We explored the possibility of publication bias using a funnel plot and Egger's test and found that publication bias was unlikely to have significantly affected our study results. Fourth, our meta-analysis did not recognise a safety margin of vitamin E supplementation because no serious side effects were reported in the included trials. However, concern has been raised as to the safety of high-dose vitamin E supplementation. A meta-analysis by Miller ER demonstrated that vitamin E doses >400 IU per day in patients with chronic diseases may increase all-cause mortality by increasing the risk of prolonged bleeding time and antagonising vitamin K functions in individuals routinely ingesting various drugs [44].
In conclusion, to date, evidence from randomised, controlled trials suggests that vitamin E supplementation is insufficient to decrease serum HbA1c, fasting glucose and fasting insulin concentrations in subjects with T2DM. Subgroup analyses support the hypothesis that vitamin E supplementation significantly reduces HbA1c and fasting insulin concentrations in subjects with low serum vitamin E concentrations and poor glycaemic control. Future large-scale, randomised trials are needed to investigate the effect of vitamin E supplementation on glycaemic control and the potential benefits and toxicities in T2DM subjects.
Supporting Information
PRISMA Checklist.
(DOC)
PRISMA 2009 Flow Diagram.
(DOC)
Funding Statement
This work was supported by a grant from the National Natural Science Foundation of China (30971248). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Whiting DR, Guariguata L, Weil C, Shaw J (2011) IDF diabetes atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract 94: 311–321. [DOI] [PubMed] [Google Scholar]
- 2. Arredondo A (2013) Diabetes: a global challenge with high economic burden for public health systems and society. Am J Public Health 103: e1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Paolisso G, Esposito R, D'Alessio MA, Barbieri M (1999) Primary and secondary prevention of atherosclerosis: is there a role for antioxidants? Diabetes Metab 25: 298–306. [PubMed] [Google Scholar]
- 4. Madian AG, Myracle AD, Diaz-Maldonado N, Rochelle NS, Janle EM, et al. (2011) Differential carbonylation of proteins as a function of in vivo oxidative stress. J Proteome Res 10: 3959–3972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Karimi J, Goodarzi MT, Tavilani H, Khodadadi I, Amiri I (2011) Relationship between advanced glycation end products and increased lipid peroxidation in semen of diabetic men. Diabetes Res Clin Pract 91: 61–66. [DOI] [PubMed] [Google Scholar]
- 6. Robertson RP (2004) Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J Biol Chem 279: 42351–42354. [DOI] [PubMed] [Google Scholar]
- 7. Robertson RP, Harmon JS (2006) Diabetes, glucose toxicity, and oxidative stress: A case of double jeopardy for the pancreatic islet beta cell. Free Radic Biol Med 41: 177–184. [DOI] [PubMed] [Google Scholar]
- 8. Pazdro R, Burgess JR (2010) The role of vitamin E and oxidative stress in diabetes complications. Mech Ageing Dev 131: 276–286. [DOI] [PubMed] [Google Scholar]
- 9. Montonen J, Knekt P, Jarvinen R, Reunanen A (2004) Dietary antioxidant intake and risk of type 2 diabetes. Diabetes Care 27: 362–366. [DOI] [PubMed] [Google Scholar]
- 10. Farvid MS, Jalali M, Siassi F, Hosseini M (2005) Comparison of the effects of vitamins and/or mineral supplementation on glomerular and tubular dysfunction in type 2 diabetes. Diabetes Care 28: 2458–2464. [DOI] [PubMed] [Google Scholar]
- 11. Minamiyama Y, Takemura S, Bito Y, Shinkawa H, Tsukioka T, et al. (2008) Supplementation of alpha-tocopherol improves cardiovascular risk factors via the insulin signalling pathway and reduction of mitochondrial reactive oxygen species in type II diabetic rats. Free Radic Res 42: 261–271. [DOI] [PubMed] [Google Scholar]
- 12. Jin L, Xue HY, Jin LJ, Li SY, Xu YP (2008) Antioxidant and pancreas-protective effect of aucubin on rats with streptozotocin-induced diabetes. Eur J Pharmacol 582: 162–167. [DOI] [PubMed] [Google Scholar]
- 13. Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, et al. (2010) Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med 362: 1675–1685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Cui YH, Jing CX, Pan HW (2013) Association of blood antioxidants and vitamins with risk of age-related cataract: a meta-analysis of observational studies. Am J Clin Nutr 98: 778–786. [DOI] [PubMed] [Google Scholar]
- 15. Myung SK, Ju W, Cho B, Oh SW, Park SM, et al. (2013) Efficacy of vitamin and antioxidant supplements in prevention of cardiovascular disease: systematic review and meta-analysis of randomised controlled trials. BMJ 346: f10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Bin Q, Hu X, Cao Y, Gao F (2011) The role of vitamin E (tocopherol) supplementation in the prevention of stroke. A meta-analysis of 13 randomised controlled trials. Thromb Haemost 105: 579–585. [DOI] [PubMed] [Google Scholar]
- 17. Clarke MW, Burnett JR, Croft KD (2008) Vitamin E in human health and disease. Crit Rev Clin Lab Sci 45: 417–450. [DOI] [PubMed] [Google Scholar]
- 18. Radimer K, Bindewald B, Hughes J, Ervin B, Swanson C, et al. (2004) Dietary supplement use by US adults: data from the National Health and Nutrition Examination Survey, 1999–2000. Am J Epidemiol 160: 339–349. [DOI] [PubMed] [Google Scholar]
- 19. Moher D, Pham B, Jones A, Cook DJ, Jadad AR, et al. (1998) Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta-analyses? Lancet 352: 609–613. [DOI] [PubMed] [Google Scholar]
- 20. Anzures-Cabrera J, Sarpatwari A, Higgins JP (2011) Expressing findings from meta-analyses of continuous outcomes in terms of risks. Stat Med 30: 2967–2985. [DOI] [PubMed] [Google Scholar]
- 21. Follmann D, Elliott P, Suh I, Cutler J (1992) Variance imputation for overviews of clinical trials with continuous response. J Clin Epidemiol 45: 769–773. [DOI] [PubMed] [Google Scholar]
- 22. Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21: 1539–1558. [DOI] [PubMed] [Google Scholar]
- 23. DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7: 177–188. [DOI] [PubMed] [Google Scholar]
- 24. Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315: 629–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Paolisso G, D'Amore A, Galzerano D, Balbi V, Giugliano D, et al. (1993) Daily vitamin E supplements improve metabolic control but not insulin secretion in elderly type II diabetic patients. Diabetes Care 16: 1433–1437. [DOI] [PubMed] [Google Scholar]
- 26. Reaven PD, Herold DA, Barnett J, Edelman S (1995) Effects of Vitamin E on susceptibility of low-density lipoprotein and low-density lipoprotein subfractions to oxidation and on protein glycation in NIDDM. Diabetes Care 18: 807–816. [DOI] [PubMed] [Google Scholar]
- 27. Tutuncu NB, Bayraktar M, Varli K (1998) Reversal of defective nerve conduction with vitamin E supplementation in type 2 diabetes: a preliminary study. Diabetes Care 21: 1915–1918. [DOI] [PubMed] [Google Scholar]
- 28. Gazis A, White DJ, Page SR, Cockcroft JR (1999) Effect of oral vitamin E (alpha-tocopherol) supplementation on vascular endothelial function in Type 2 diabetes mellitus. Diabet Med 16: 304–311. [DOI] [PubMed] [Google Scholar]
- 29. Paolisso G, Tagliamonte MR, Barbieri M, Zito GA, Gambardella A, et al. (2000) Chronic vitamin E administration improves brachial reactivity and increases intracellular magnesium concentration in type II diabetic patients. J Clin Endocrinol Metab 85: 109–115. [DOI] [PubMed] [Google Scholar]
- 30. Manzella D, Barbieri M, Ragno E, Paolisso G (2001) Chronic administration of pharmacologic doses of vitamin E improves the cardiac autonomic nervous system in patients with type 2 diabetes. Am J Clin Nutr 73: 1052–1057. [DOI] [PubMed] [Google Scholar]
- 31. Park S, Choi SB (2002) Effects of alpha-tocopherol supplementation and continuous subcutaneous insulin infusion on oxidative stress in Korean patients with type 2 diabetes. Am J Clin Nutr 75: 728–733. [DOI] [PubMed] [Google Scholar]
- 32. Son JW, Jang EH, Kim MK, Kim IT, Roh YJ, et al. (2011) Diabetic retinopathy is associated with subclinical atherosclerosis in newly diagnosed type 2 diabetes mellitus. Diabetes Res Clin Pract 91: 253–259. [DOI] [PubMed] [Google Scholar]
- 33. Economides PA, Khaodhiar L, Caselli A, Caballero AE, Keenan H, et al. (2005) The effect of vitamin E on endothelial function of micro- and macrocirculation and left ventricular function in type 1 and type 2 diabetic patients. Diabetes 54: 204–211. [DOI] [PubMed] [Google Scholar]
- 34. Ble-Castillo JL, Carmona-Diaz E, Mendez JD, Larios-Medina FJ, Medina-Santillan R, et al. (2005) Effect of alpha-tocopherol on the metabolic control and oxidative stress in female type 2 diabetics. Biomed Pharmacother 59: 290–295. [DOI] [PubMed] [Google Scholar]
- 35. Baliarsingh S, Beg ZH, Ahmad J (2005) The therapeutic impacts of tocotrienols in type 2 diabetic patients with hyperlipidemia. Atherosclerosis 182: 367–374. [DOI] [PubMed] [Google Scholar]
- 36. Boshtam M, Rafiei M, Golshadi ID, Ani M, Shirani Z, et al. (2005) Long term effects of oral vitamin E supplement in type II diabetic patients. Int J Vitam Nutr Res 75: 341–346. [DOI] [PubMed] [Google Scholar]
- 37. Ward NC, Wu JH, Clarke MW, Puddey IB, Burke V, et al. (2007) The effect of vitamin E on blood pressure in individuals with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. J Hypertens 25: 227–234. [DOI] [PubMed] [Google Scholar]
- 38. de Oliveira AM, Rondo PH, Luzia LA, D'Abronzo FH, Illison VK (2011) The effects of lipoic acid and alpha-tocopherol supplementation on the lipid profile and insulin sensitivity of patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled trial. Diabetes Res Clin Pract 92: 253–260. [DOI] [PubMed] [Google Scholar]
- 39. Monnier L, Mas E, Ginet C, Michel F, Villon L, et al. (2006) Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 295: 1681–1687. [DOI] [PubMed] [Google Scholar]
- 40. Ceriello A, Giugliano D, Quatraro A, Donzella C, Dipalo G, et al. (1991) Vitamin E reduction of protein glycosylation in diabetes. New prospect for prevention of diabetic complications? Diabetes Care 14: 68–72. [DOI] [PubMed] [Google Scholar]
- 41. Liu S, Lee IM, Song Y, Van Denburgh M, Cook NR, et al. (2006) Vitamin E and risk of type 2 diabetes in the women's health study randomized controlled trial. Diabetes 55: 2856–2862. [DOI] [PubMed] [Google Scholar]
- 42. Suksomboon N, Poolsup N, Sinprasert S (2011) Effects of vitamin E supplementation on glycaemic control in type 2 diabetes: systematic review of randomized controlled trials. J Clin Pharm Ther 36: 53–63. [DOI] [PubMed] [Google Scholar]
- 43. Shekelle PG, Morton SC, Jungvig LK, Udani J, Spar M, et al. (2004) Effect of supplemental vitamin E for the prevention and treatment of cardiovascular disease. J Gen Intern Med 19: 380–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, et al. (2005) Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med 142: 37–46. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PRISMA Checklist.
(DOC)
PRISMA 2009 Flow Diagram.
(DOC)