Abstract
The expression of genes encoding components of ABA and metabolic signalling pathways in developing barley endosperm and embryo was investigated. The genes included HvRCAR35_47387 and HvRCAR35_2538 (encoding ABA receptors), HvABI1d (protein phosphatase 2C), HvSnRK2.4, HvSnRK2.6 and HvPKABA1 (SnRK2-type protein kinases) and HvABI5 (ABA response element binding protein; AREBP), as well as two genes encoding SnRK1-type protein kinases. Both SnRK1 and SnRK2 phosphorylate AREBPs, but SnRK2 is activated by ABA whereas SnRK1 may be broken down. Multiple cereal AREBPs with two conserved SnRK1/2 target sites and another class of BZIP transcription factors with SnRK1/2 binding sites, including HvBLZ1, were identified. Barley grain (cv. Triumph) was sampled at 15, 20, 25 and 30 days post-anthesis (dpa). HvRCAR35_47387, HvABI1d, HvSnRK2.4 and HvABI5 were expressed highly in the endosperm but at much lower levels in the embryo. Conversely, HvPKABA1 and HvRCAR35_2538 were expressed at higher levels in the embryo than the endosperm, while HvSnRK2.6 was expressed at similar levels in both. HvRCAR35_47387, HvABI1d, HvSnRK2.4 and HvABI5 all peaked in expression in the endosperm at 20 dpa. A model is proposed in which ABA brings about a transition from a SnRK1-dominated state in the endosperm during grain filling to a SnRK2-dominated state during maturation.
Keywords: Hordeum vulgare, Abscisic acid, Seed development, Signalling
Highlights
-
•
Components of ABA signalling pathway differ between endosperm and embryo in barley.
-
•
ABA signalling pathway components are highly expressed in barley endosperm at 20 dpa.
-
•
ABA may bring about change from SnRK1-dominated state to SnRK2-dominated state.
-
•
This may play key role in transition from grain filling to maturation in barley.
-
•
BLZ1/OHP1 class of bZIP transcription factors contain multiple SnRK1/2 target sites.
1. Introduction
The transition between grain filling and maturation in cereal seed development involves the cessation of starch and storage protein synthesis as well as the expression of genes that initiate processes that prepare the seed for dormancy, and dramatic changes in gene expression in the endosperm have been described (Wan et al., 2008). The hormone abscisic acid (ABA) is one of the major players in this process and the recent elucidation of the ABA signal transduction pathway (Cutler et al., 2010) means that there is now the opportunity to advance our understanding of the mechanisms by which ABA influences seed development.
Integral to ABA signalling is sucrose nonfermenting-1-related protein kinase-2 (SnRK2). In the absence of ABA, SnRK2 is kept in an inactive state through the action of protein phosphatase 2C (PP2C). In the presence of ABA, ABA receptors PYR/PYL/RCAR bind to and inhibit PP2C, allowing the accumulation of active SnRK2 and subsequent phosphorylation of ABA-response element binding proteins (AREBPs) (Cutler et al., 2010). AREBPs (also known as ABFs) are a family of basic leucine zipper (bZIP) transcription factors that recognize the ABA response elements (ABREs) present in some ABA-regulated genes (Cutler et al., 2010).
SnRK2s phosphorylate AREBPs at serine (preferentially) or threonine residues with a basic residue at −3 with respect to the serine/threonine residue (Fujii et al., 2007; Furihata et al., 2006; Yoshida et al., 2010). Two of the SnRK2 target sites in AREBPs are potential targets for phosphorylation by a related protein kinase, SnRK1, which also phosphorylates serine residues with a basic residue at −3, but has an additional requirement for hydrophobic residues at −5 and +4. These SnRK1 target sites are conserved throughout the Arabidopsis AREBP family and in all of the AREBPs that have been identified so far in other species (Zhang et al., 2008). Peptides with amino acid sequences based on these sites have been shown to be phosphorylated by both SnRK1 and 2 (Zhang et al., 2008).
SnRK1 is functionally as well as structurally related to SNF1 of fungi and adenosine monophosphate (AMP)-activated protein kinase (AMPK) of animals, both of which are major regulators of metabolism and a range of cellular processes in their respective systems (reviewed by Hey et al., 2010). SnRK2-type protein kinases, on the other hand, do not have any direct functional counterpart in fungal or animal cells. They are encoded by a medium-sized gene family with, for example, 10 members in Arabidopsis (Arabidopsis thaliana) and are involved with or implicated in responses to abiotic stresses such as drought, salinity, cold and osmotic stress (Hey et al., 2010). Kobayashi et al. (2004) divided Arabidopsis SnRK2s into three classes, based on phylogeny and their response to ABA: Class 1, which are not activated in response to ABA, Class 2, which are activated weakly by ABA and Class 3, which are strongly activated by ABA.
In a recent study, we investigated the interactions between ABA, SnRK1 and SnRK2 in wheat and showed contrasting effects of ABA on SnRK1 and SnRK2 protein levels and phosphorylation state (Coello et al., 2012). Application of ABA to wheat roots brought about a dramatic decrease in SnRK1 protein, and phosphorylation/activation of a 42 kDa SnRK2. Unusually for SnRK2, this member of the family was calcium-dependent. These results imply differential roles for SnRK1 and SnRK2 in ABA signalling and antagonistic effects of SnRK1 and SnRK2 on gene expression. Consistent with this, transient repression of SnRK1 has been shown to repress the activity of an α-amylase gene (α-Amy2) promoter in cultured wheat embryos (Laurie et al., 2003), while a SnRK2, PKABA1, has been shown to down-regulate α-amylase genes in response to ABA (Gomez-Cadenas et al., 1999). Recently, ABA-responsive cis-elements have been identified in the promoters of key genes of starch biosynthesis in barley:HvSUS1, encoding sucrose synthase, and HvAGP-L1, encoding the large subunit of ADP-glucose pyrophosphorylase (AGPase) (Seiler et al., 2011). Homologous genes are up-regulated in response to SnRK1 over-expression in potato tubers (McKibbin et al., 2006), with a concomitant increase in starch content.
In this study, we have investigated the expression of genes encoding components of the ABA sensing and signalling pathway in developing barley endosperm and embryo, compared expression patterns of different SnRK1 and SnRK2 family members, and identified cereal AREBP and other BZIP transcription factors with SnRK target sites. The aim was to provide new knowledge of the interactions between SnRK1, SnRK2, PP2C, ABA and transcription factors in the control of cereal seed development.
2. Materials and methods
2.1. Database screening
BLAST searches of the non-redundant (nr) protein sequence database were performed using the National Center for Biotechnology Information (NCBI) portal (www.ncbi.nlm.nih.gov/). Barley1 22k Microarray data (Dash et al., 2012) was accessed via the PLEXdb portal (www.plexdb.org/modules/tools/plexdb_blast.php).
2.2. Sample preparation
Plants of the two-rowed barley (Hordeum vulgare) cultivar Triumph were grown in 15 cm diameter pots in a glasshouse with a 16 h day-length (supplemental lighting was used as necessary) and a minimum temperature of 14 °C by night and 18 °C by day. There was 1 plant per pot and pots were laid out in a randomised design. Ears were tagged at anthesis, grain sampled at 15, 20, 25 and 30 days post-anthesis (dpa), and caryopses dissected under a dissecting microscope. There were three biological replicates for each of the four time points. Dissected samples were immediately frozen in liquid nitrogen and stored at −80 °C. Images of the dissected grain were obtained with a Leica M205 FA Stereomicroscope (Leica Microsystems, Milton Keynes, UK).
2.3. RNA extraction and cDNA synthesis
Total RNA was isolated using TRIzol® Reagent (Ambion, USA) according to the manufacturer's instructions. The quality and the concentration of RNA were measured by spectrophotometry using a NanoDrop ND-100 (Thermo Fisher, Wilmington, DE, USA; supplied by Labtech International Ltd, Uckfield, UK) and confirmed by electrophoresis through an agarose gel. The RNA was treated with RQ1 RNase-Free DNase (Promega, USA); cDNA was then synthesised using SuperScript® III Reverse Transcriptase (Invitrogen, USA). The cDNA was checked for purity by polymerase chain reaction (PCR) amplification using primers CATCAAGCTCAAGGACGACA and GCCTTGTCCTTGTCAGTGAA, which anneal to sites flanking an intron within the HvGAPDH gene.
2.4. PCR primer design and quantitative real-time PCR
Primers were designed using either Primer Express® Software Version 3.0 (Applied Biosystems, USA) or Primer 3 (http://primer3.wi.mit.edu/). PCR reactions were performed in 96-well plates on a 7500 Real Time PCR System (Applied Biosystems, USA) using SYBR® Select Master Mix (Applied Biosystems, USA). The reactions for biological replicates were separated across three plates, thus forming statistical blocks for subsequent data analysis. Reactions contained 10 μL 2 × mix, 0.6 μL of each primer (10 μM) and 100 ng cDNA template in a final volume of 20 μL. The same thermal profile was used for all PCR reactions: 50 °C for 2 min, 95 °C for 10 min, 45 cycles of 95 °C for 15 s and 60 °C for 1 min; data collection was carried out during the 60 °C step. The dissociation curves/melting curves analyses were conducted after cycle 45.
2.5. Data analysis
The efficiency of the PCR was estimated using the LinReg PCR programme (Ramakers et al., 2003). A Ct value was obtained by 7500 software v2.0.5 (Applied Biosystems, USA) and the Ct and efficiency values were then used to calculate the Relative Quantity (RQ), and the Normalised Relative Quantity (NRQ) of a target gene's expression with respect to three reference genes, HvActin, HvCyclophilin and HvGAPDH. NRQ was calculated using the following formula:
Where Etarget, EHvActin, EHvCyclophilin and EHvGAPDH are the estimated PCR efficiencies for a particular target gene and the two reference genes and where Ct, target, Ct, HvActin, Ct, HvCyclophilin and Ct, HvGAPDH are the corresponding Ct values. The Ct values of each reference gene were found to be stable across the time points, prior to using the geometric mean of the RQ values as shown in the formula.
Statistical analysis of the NRQ data was performed using the GenStat statistical package (2011, 14th edition, © VSN International Ltd, Hemel Hempstead, UK). Taking account of the three plates as statistical blocks, ANOVA was applied to the log (to base 2)-transformed inverse of the NRQ data (Rieu and Powers, 2009). This transformation ensured homogeneity of variance and effectively provided values back on the Ct-scale. Therefore, as for Ct values (Ct = log2(1/NRQ)), a low log2(1/NRQ) indicated a high gene expression, whereas a high log2(1/NRQ) indicated low gene expression. Following a significant (p < 0.05, F-test) result from ANOVA, the analysis assessed the statistical significance of differences in gene expression between time points using the least significant difference (LSD) at the 5% (p < 0.05) level of significance.
3. Results
3.1. Identification of genes involved in ABA signalling in barley
The aim of the project was to analyse expression of genes encoding proteins potentially involved in the abscisic acid (ABA) signalling pathway during barley grain development, including the ABA receptor protein, RCAR, protein phosphatase 2C (PP2C), sucrose nonfermenting-1-related protein kinase 2 (SnRK2) and related protein kinase SnRK1, and ABA response element binding proteins (AREBPs), as well as additional transcription factors that could be substrates for SnRK1 or SnRK2.
Two genes encoding ABA receptors were studied; these were HvRCAR35_47387 and HvRCAR35_2538 (Seiler et al., 2011). The protein phosphatase 2C gene was HvABI1d (Seiler et al., 2011). The ABI1 name derives from the fact that this gene was first identified in Arabidopsis in a screen of mutants that were ABA insensitive (Finkelstein, 1994).
Two SnRK2 genes from barley have been described previously: HvSnRK2.6 (Mangelsen et al., 2011; Sreenivasulu et al., 2006; Seiler et al., 2011) and HvPKABA1 (Yamauchi et al., 2002), and these were both included in the study. A third SnRK2 gene was identified from Barley1 22k Microarray data (Dash et al., 2012) and also included. It has been annotated as HvSnRK2.4 but there is no experimental evidence that the SnRK2 that it encodes is a functional equivalent of Arabidopsis SnRK2.4, which is ABA-independent.
Two SnRK1 genes have been characterised previously in barley:HvSnRK1a (also known as BKIN2) and HvSnRK1b (also known as BKIN12 and related BKIN9) (Halford et al., 1992). HvSnRK1a has been shown to be expressed in all tissues that have been analysed to date, whereas HvSnRK1b is expressed at high levels in the seed but at relatively low levels elsewhere (Hannappel et al., 1995). Despite its relatively low levels of expression in non-seed tissues, antisense inhibition of HvSnRK1b gene expression in anthers has been shown to result in impaired pollen development, a failure of pollen grains to accumulate starch, and pollen inviability (Zhang et al., 2001). Southern analysis of the gene family has suggested that it comprises 10–20 members (Halford et al., 1992).
A BLAST search of the non-redundant (nr) protein sequence database was performed with the HvSnRK1a amino acid sequence using the National Center for Biotechnology Information (NCBI) portal. This identified one barley SnRK1a entry, accession number CAA57898.1, and seven barley SnRK1b entries, accession numbers BAJ95249.1, BAK03923.1, CAA46555.1, CAA46556.1, BAK05223.1, CAA07813.1 and CAA46554.1. Primers with which to amplify HvSnRK1a and HvSnRK1b products were designed to reveal any of these homologues with the aim being to distinguish between the two types. SnRK1 interacts with two non-catalytic subunits to form a heterotrimeric complex and the expression of a barley gene, HvSnRK1β, encoding one of these was identified from Barley1 22k Microarray data (Dash et al., 2012) and this gene was also included in the study.
The screen of ABA-insensitive Arabidopsis mutants that identified ABI1 (Finkelstein, 1994) also identified genes encoding transcription factors of the ABA response element binding protein (AREBP) class. Arabidopsis contains 14 genes encoding AREBP-type transcription factors (Zhang et al., 2008); of these, ABI5 has been the most intensively studied because of its role in germination (abi5 mutant seeds germinate in the presence of ABA at levels that prevent germination of wild-type seeds). However, it is important to note that other AREBPs may have different but equally important roles in controlling plant development. Unfortunately, the prominence of ABI5 in the literature has led to AREBPs from other species being annotated as ABI5 analogues, in some cases without any evidence that they have the same function as ABI5.
SnRK2 phosphorylates AREBPs at multiple sites, requiring only a serine or threonine residue with a basic residue at −3 for substrate recognition (Furihata et al., 2006). Two of the sites in AREBPs that can be phosphorylated by SnRK2 have also been shown to be phosphorylated by SnRK1 (Zhang et al., 2008). SnRK1 phosphorylates target proteins at serine residues with a hydrophobic residue at −5 and +4 with respect to the serine as well as the basic residue (preferably arginine) at −3 or (sub-optimally) at −4.
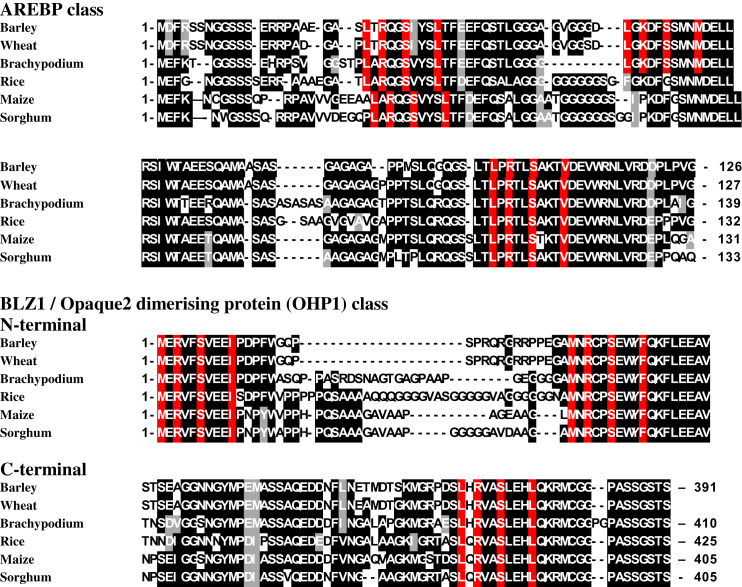
In order to identify cereal AREBPs, BLAST searches were performed of the non-redundant protein sequence database using a barley AREBP amino acid sequence identified previously (Zhang et al., 2008). This identified members of the AREBP family from barley, wheat, Brachypodium, rice, maize and sorghum. The N-terminal regions of one AREBP from each species are shown in Fig. 1, with SnRK1 target sites highlighted in red. Two such sites are present in all six of the AREBPs shown and also in all but one (ABI5 itself) of the AREBPs in Arabidopsis (Zhang et al., 2008). Synthetic peptides based on both sites have been shown to be phosphorylated by partially-purified SnRK1 and SnRK2 from wheat roots (Coello et al., 2012). For the present study, primers for analysis of AREBP gene expression were based on the barley gene annotated as HvABI5 (Casaretto and Ho, 2003).
Fig. 1.
SnRK1/2 target sites in cereal transcription factors Top: Alignment of the N-terminal region of representative ABA-response element binding proteins (AREBPs) from barley (Hordeum vulgare) (GenBank: AAO06115.1); wheat (Triticum aestivum) (GenBank: BAD97366.1); Brachypodium (Brachypodium distachyon) (NCBI Reference Sequence: XP_003578228.1); rice (Oryza sativa) (GenBank: EEC84700.1); maize (Zea mays) (NCBI Reference Sequence: NP_001132507.1); and sorghum (Sorghum bicolor) (NCBI Reference Sequence: XP_002460329.1). Bottom: Alignment of the N- and C-terminal regions of BLZ1 and related transcription factors from barley (Hordeumvulgare) (GenBank: CAA56374.1), wheat (Triticum aestivum), from wheat genome data (Brenchley et al., 2012); Brachypodium (Brachypodium distachyon) (NCBI Reference Sequence: XP_003557420.1); rice (Oryza sativa) (Genbank ABF99347.1); maize (Zea mays) Opaque2 heterodimerizing protein1 (OHP1) (NCBI Reference Sequence: NP_001105687.1); and sorghum (Sorghum bicolor) (NCBI Reference Sequence: XP_002463740.1). Residues that make up SnRK1/2 target sites (a serine residue with a hydrophobic residue at −5 and +4 with respect to the serine, and a basic residue at −3) are highlighted in red. Other conserved residues are highlighted in black, with like-for-like substitutions highlighted in grey. Numbers refer to the position in the amino acid sequence; note that the length of the wheat BLZ1-like transcription factor is not known.
3.2. Identification of additional transcription factors with SnRK target sites
The tolerance of multiple similar amino acids within the SnRK1 target site makes computational identification of putative SnRK1-regulated proteins and protein families difficult. A pipeline has therefore been developed consisting of a basic motif search, PROSITE family searches (Falquet et al., 2002) and multiple alignments within each PROSITE family. AREBPs were identified as potential substrates for SnRK1 using this pipeline (Zhang et al., 2008). For the present study, the database that was compiled using this pipeline was mined for additional bZIP transcription factors that could also be phosphorylated by SnRKs. This process identified another class of cereal bZIPs with N-terminal SnRK1 target sites, comprising Opaque2 dimerising protein (OHP1) from maize (NP_001105687; 542704_ohp1) and sorghum, BLZ1 from barley (HvBLZ1; CAA56374) and multiple uncharacterised BZIPS from rice (NP_001051558; AAP44683; ABF99347; BAF13472; Os03g0796900; BAA11431; EAY92182; BAA36492). These are aligned in Fig. 1. BLZ1 (Vicente-Carbajosa et al., 1997) recognizes the so-called N motif of the prolamin box of wheat, barley and rye storage protein gene promoters and was therefore included in the analysis of gene expression.
3.3. Expression analyses
Plants of barley (Hordeum vulgare) cultivar Triumph were grown in a glasshouse and grain sampled at 15, 20, 25 and 30 days post-anthesis (dpa) (Fig. 2). Caryopses were dissected to separate the embryo (including the scutellum) and endosperm. For the endosperm analysis, only material from the central, starchy endosperm was analysed (Fig. 2). RNA was purified from the samples and used as a template for quantitative, real-time polymerase chain reaction (qRT-PCR) analysis of gene expression for the HvRCAR35_47387, HvRCAR35_2538, HvABI1d, HvSnRK2.4, HvSnRK2.6, HvPKABA1, HvABI5, HvSnRK1a, HvSnRK1b, HvSnRK1β, and HvBLZ1 genes described above. The primers used for the PCR reactions, mean log2(1/NRQ) values and results of ANOVA to consider significance of differences in gene expression between time points for the genes of interest are given in Supplementary Information. For comparison, expression of a gene encoding a late embryogenesis abundant protein, HvLEA3, was also included. Each analysis included three biological replicates from different pots and the expression level of a gene was calculated as the Normalised Relative Quantity (NRQ) with respect to three reference genes, HvActin, HvCyclophilin and HvGAPDH. The results for expression in the endosperm are shown in Fig. 3 and the embryo in Fig. 4. The differences in scales on the graphs within and between Figs. 3 and 4 should be considered when comparing the expression levels of different genes or the same gene in different tissues.
Fig. 2.

Seeds and samples at 15, 20, 25 and 30 days post anthesis (dpa). The scale beneath each picture shows 1 mm. A. Whole developing seeds. B. Whole developing seeds without pericarp. C. Longitudinal sections of developing seeds. D. Sections of embryo. E. Seed transection; rectangles indicate sections manually dissected to give endosperm samples.
Fig. 3.
Differential expression of genes encoding components of ABA and metabolic signalling pathways in endosperm of barley (Hordeum vulgare) cv. Triumph analysed by qRT-PCR. Seeds were sampled for analysis at 15, 20, 25 and 30 days post-anthesis (dpa), as indicated. Expression is represented as the Normalised Relative Quantity (NRQ) of a target gene's expression with respect to three reference genes: HvActin, HvCyclophilin and HvGAPDH. Means and standard errors are shown from the analysis of three biological replicates. See Supplementary Information for results of statistical comparisons using the log2(1/NRQ) transformed data.
Fig. 4.
Differential expression of genes encoding components of the ABA and metabolic signalling pathways in embryo of barley (Hordeum vulgare) cv. Triumph analysed by qRT-PCR. Seeds were sampled for analysis at 15, 20, 25 and 30 days post-anthesis (dpa), as indicated. Expression is represented as the Normalised Relative Quantity (NRQ) of a target gene's expression with respect to three reference genes, HvActin, HvCyclophilin and HvGAPDH. Means and standard errors are shown from the analysis of three biological replicates. See Supplementary Information for results of statistical comparisons using the log2(1/NRQ) transformed data.
The expression of the HvLEA3 gene was as expected, rising through mid-development to its highest level in the endosperm at 30 dpa and the embryo at 25–30 dpa. HvRCAR35_47387 gene expression, in contrast, was readily detectable in the endosperm, with expression peaking at 20 dpa (Fig. 3), but almost undetectable in the embryo (not shown), while HvRCAR35_2538 was expressed at low but detectable levels in the embryo, with expression declining through seed development (Fig. 4), but not in the endosperm (not shown). The expression of the PP2C-encoding gene, HvABI1d, showed a similar pattern to HvRCAR35_47387 in the endosperm, with a peak at 20 dpa. However, it was also expressed in the embryo, with expression increasing through development to its highest point at 30 dpa. At 20 dpa (the peak of its expression in the endosperm), the level of expression in the endosperm was approximately six times that in the embryo, but by 30 dpa, with expression declining in the endosperm but increasing in the embryo, there was only a two-fold difference.
Analysis of expression of the three SnRK2 genes, HvSnRK2.6 (Mangelsen et al., 2011; Sreenivasulu et al., 2006; Seiler et al., 2011), HvPKABA1 (Yamauchi et al., 2002) and HvSnRK2.4 (Barley1 22k Microarray data; Dash et al., 2012), revealed clear differences. HvSnRK2.4 was expressed at the highest levels of the three in the endosperm, with expression peaking at 20 dpa. It was also detectable in the embryo but at levels 20 times or more lower than in the endosperm, with expression showing no change through development. HvSnRK2.6 showed a similar pattern of expression in the endosperm, peaking at 20 dpa, but its expression levels were ten times lower than HvSnRK2.4. It showed comparable levels of expression in the embryo, but in contrast to HvSnRK2.4, its expression increased from 15 dpa through to 30 dpa, so that by 30 dpa it was expressed more highly than HvSnRK2.4. SnRK2.4 is considered to be ABA-independent in Arabidopsis, but the similar expression pattern of HvSnRK2.4 to other genes encoding components of the ABA signalling pathway suggests that this is not the case in barley.
HvPKABA1 was not expressed at detectable levels in the endosperm, but was expressed in the embryo, with expression increasing from low levels at 15 dpa to its highest at 30 dpa. In other words, expression of this SnRK2 was associated with mid- to late-development in the embryo. Previous studies in wheat and barley have implicated this SnRK2 with a role in controlling the onset of seed dormancy, ABA-induction of gene expression and the suppression of genes that are induced by GA, as well as responses to cold and drought (Gómez-Cadenas et al., 1999; Yamauchi et al., 2002).
Expression of HvSnRK1a (also known as BKIN2;Halford et al., 1992; Hannapel et al., 1995) increased (p < 0.05, LSD) between 15 and 20 days post-anthesis, then did not change significantly (Fig. 3). This gene was also expressed in the embryo (Fig. 4), but levels increased steadily after 15 dpa and were at their highest at 30 dpa. At this point, transcription in the endosperm was still approximately four times higher than in the embryo. HvSnRK1b (also known as BKIN12) showed a similar pattern of expression in the endosperm, although its level was about half that of HvSnRK1a. However, its expression in the embryo, while detectable, was approximately 100 times lower (Fig. 4). This is broadly consistent with the findings of Hannappel et al. (1995), although that study suggested that HvSnRK1b was expressed more highly in the endosperm than HvSnRK1a.
Currently there is insufficient knowledge of the different roles of SnRK1a and SnRK1b to assess the significance of their different expression patterns. However, specific down-regulation of HvSnRK1b in anthers has been shown to have profound effects on pollen development, including a failure to accumulate starch, arrested development and inviability (Zhang et al., 2001). SnRK1bhomologues have also been implicated in controlling starch accumulation in sorghum and rice grain (Jain et al., 2008; Kanegae et al., 2005).
The gene encoding the SnRK1 non-catalytic subunit, HvSnRK1β, had a similar pattern of expression in the endosperm to HvSnRK1a and HvSnRK1b, with the peak at 20 dpa. In contrast, its expression in the embryo, while similar to that of HvSnRK1a at 15 dpa, did not rise in the way that HvSnRK1a gene expression did, resulting in it being substantially lower than HvSnRK1a by 30 dpa.
The AREBP-encoding gene, annotated as HvABI5, showed a similar expression pattern to HvRCAR35_47387, HvSnRK2.4 and HvSnRK2.6 in the endosperm, with expression peaking at 20 dpa. Expression was also detectable in the embryo but at much lower levels that remained constant throughout development. The expression of HvBLZ1 was also more than ten times higher in the endosperm than the embryo, with expression in the endosperm rising significantly (p < 0.05, LSD) from 15 to 20 dpa and then remaining relatively high.
4. Discussion
The different events that occur in the endosperm and embryo during the onset of seed dormancy have been reviewed by Sreenivasulu et al. (2006), who considered the cells of the endosperm to undergo a form of programmed cell death, while those of the embryo enter true dormancy, from which they emerge at germination. ABA is implicated in bringing about both of these very different developmental programmes and an important conclusion of the present study is that different ABA receptors and SnRK2s are involved in the different tissues. HvRCAR35_47387 (encoding an ABA receptor), HvABI1d (PP2C), HvSnRK2.4 and the AREBP-encoding gene annotated as HvABI5 were expressed highly in the endosperm but at much lower levels in the embryo. Conversely, a second ABA receptor-encoding gene, HvRCAR35_2538, and the SnRK2-encoding gene, HvPKABA1, were expressed at higher levels in the embryo than the endosperm, while HvSnRK2.6 was expressed at similar levels in both (although HvSnRK2.4 was the predominant SnRK2 transcript in endosperm). This adds a level of complexity to what otherwise might be regarded as a relatively simple ABA signalling pathway. It should be noted here that while differential regulation of gene expression is the most likely explanation of these results, post-transcriptional effects, such as alternative splicing resulting in the loss of regions spanning primer-binding sites, could explain why a transcript was detected in one tissue but not the other. It should also be noted that there is no contradiction in the high expression of HvABI1 (PP2C) in the endosperm, or the fact that its expression pattern closely matches that of HvSnRK2.4 and HvRCAR35_47387. The presence of PP2C would ensure that SnRK2 would only be activated in the presence of ABA.
HvRCAR35_47387, HvABI1d, HvSnRK2.4 and the AREBP-encoding gene annotated as HvABI5 all peaked in expression in the endosperm at 20 dpa. ABA is required for both storage compound accumulation and the transition from filling to maturation, and we hypothesise that high levels of the signalling components are present at this time to enable these processes to be initiated. SnRK1 on the other hand, has been implicated in the initiation and control of starch biosynthesis in seeds, tubers and pollen (Jain et al., 2008; Kanegae et al., 2005; McKibbin et al., 2006; Zhang et al., 2001). In this study, we found SnRK1 to be expressed in both endosperm and embryo throughout development, with sub-type SnRK1b showing much higher levels of expression in the endosperm than the embryo, while SnRK1a was expressed at more similar levels in both tissues. However, SnRK1 has been shown to be broken down in wheat roots in the presence of ABA, while at least one SnRK2 is activated (Coello et al., 2012). We propose a possible model for the interaction between ABA and the two SnRK families in which the hormone brings about a transition from a SnRK1-dominated state in early grain filling to a SnRK2-dominated state during maturation (Fig. 5).
Fig. 5.
Diagram summarising a model in which there is a switch in the barley endosperm from a SnRK1-dominated system to one controlled by SnRK2, brought about by the action of ABA, during the transition from grain filling to maturation.
Acknowledgements
ZC was the recipient of an overseas visiting grant from Shanghai Academy of Agricultural Sciences, Shanghai, P. R. China, funded from the Basic Research of Shanghai Municipal S&T Commission (12JC1407800) and the earmarked fund for Modern Agro-industry Technology Research System (No. CARS-05). Rothamsted Research receives grant-aided support from the Biotechnology and Biological Sciences Research Council of the UK.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Supplementary data related to this article can be found at http://dx.doi.org/10.1016/j.jcs.2013.06.009.
Contributor Information
Zhiwei Chen, Email: czw1900@yahoo.com.cn.
Jianhua Huang, Email: sw1@saas.sh.cn.
Nira Muttucumaru, Email: nira.muttucumaru@rothamsted.ac.uk.
Stephen J. Powers, Email: Stephen.powers@rothamsted.ac.uk.
Nigel G. Halford, Email: nigel.halford@rothamsted.ac.uk.
Appendix A. Supplementary data
References
- Brenchley R., Spannagl M., Pfeifer M., Barker G.L.A., D’Amore R., Allen A.M., McKenzie N., Kramer M., Kerhornou A., Bolser D., Kay S., Waite D., Trick M., Bancroft I., Gu Y., Huo N., Luo M.-C., Sehgal S., Gill B., Kianian S., Anderson O., Kersey P., Dvorak J., McCombie W.R., Hall A., Mayer K.F.X., Edwards K.J., Bevan M.W., Hall N. Analysis of the bread wheat genome using whole-genome shotgun sequencing. Nature. 2012;491:705–710. doi: 10.1038/nature11650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casaretto J., Ho T.H. The transcription factors HvABI5 and HvVP1 are required for the abscisic acid induction of gene expression in barley aleurone cells. Plant Cell. 2003;15:271–284. doi: 10.1105/tpc.007096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coello P., Hirano E., Hey S.J., Muttucumaru N., Martinez-Barajas E., Parry M.A.J., Halford N.G. Evidence that abscisic acid promotes degradation of SNF1-related protein kinase (SnRK) 1 in wheat and activation of a putative calcium-dependent SNRK2. Journal of Experimental Botany. 2012;63:913–924. doi: 10.1093/jxb/err320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutler S.R., Rodriguez P.L., Finkelstein R.R., Abrams S.R. Abscisic acid: emergence of a core signaling network. Annual Review of Plant Biology. 2010;61:651–679. doi: 10.1146/annurev-arplant-042809-112122. [DOI] [PubMed] [Google Scholar]
- Dash S., Van Hemert J., Hong L., Wise R.P., Dickerson J.A. PLEXdb: gene expression resources for plants and plant pathogens. Nucleic Acids Research. 2012;40(D1):D1194–D1201. doi: 10.1093/nar/gkr938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falquet L., Pagni M., Bucher P., Hulo N., Sigrist C.J., Hofmann K., Bairoch A. The PROSITE database, its status in 2002. Nucleic Acids Research. 2002;30:235–238. doi: 10.1093/nar/30.1.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelstein R.R. Mutations at two new Arabidopsis ABA response loci are similar to the ABI3 mutations. Plant Journal. 1994;5:765–771. [Google Scholar]
- Fujii H., Verslues P.E., Zhu J.K. Identification of two protein kinases required for abscisic acid regulation of seed germination, root growth, and gene expression in Arabidopsis. Plant Cell. 2007;19:485–494. doi: 10.1105/tpc.106.048538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furihata T., Maruyama K., Fujita Y., Umezawa T., Yoshida R., Shinozaki K., Yamaguchi-Shinozaki K. Abscisic acid-dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proceedings of the National Academy of Sciences USA. 2006;103:1988–1993. doi: 10.1073/pnas.0505667103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Cadenas A., Verhey S.D., Holappa L.D., Shen Q.X., Ho T.H.D., Walker-Simmons M.K. An abscisic acid-induced protein kinase, PKABA1, mediates abscisic acid-suppressed gene expression in barley aleurone layers. Proceedings of the National Academy of Sciences USA. 1999;96:1767–1772. doi: 10.1073/pnas.96.4.1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halford N.G., Vicente-Carbajosa J., Sabelli P.A., Shewry P.R., Hannappel U., Kreis M. Molecular analyses of a barley multigene family homologous to the yeast protein kinase gene SNF1. Plant Journal. 1992;2:791–797. [PubMed] [Google Scholar]
- Hannappel U., Vicentei-Carbajosa J., Barker J.H., Shewry P.R., Halford N.G. Differential expression of two barley SNF1-related protein kinase genes. Plant Molecular Biology. 1995;27:1235–1240. doi: 10.1007/BF00020898. [DOI] [PubMed] [Google Scholar]
- Hey S.J., Byrne E., Halford N.G. The interface between metabolic and stress signalling. Annals of Botany. 2010;105:197–203. doi: 10.1093/aob/mcp285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain M., Li Q.-B., Chourey P.S. Cloning and expression analyses of sucrose non-fermenting-1-related kinase 1(SnRK1b) gene during development of sorghum and maize endosperm and its implicated role in sugar-to-starch metabolic transition. Physiologia Plantarum. 2008;134:161–173. doi: 10.1111/j.1399-3054.2008.01106.x. [DOI] [PubMed] [Google Scholar]
- Kanegae H., Miyoshi K., Hirose T., Tsuchimoto S., Mori M., Nagato Y., Takano M. Expressions of rice sucrosenon-fermenting-1 related protein kinase 1 genes are differently regulated during the caryopsis development. Plant Physiology and Biochemistry. 2005;43:669–679. doi: 10.1016/j.plaphy.2005.06.004. [DOI] [PubMed] [Google Scholar]
- Kobayashi Y., Yamamoto S., Minami H., Kagaya Y., Hattori T. Differential activation of the rice sucrose nonfermenting1-related protein kinase 2 family by hyperosmotic stress and abscisic acid. Plant Cell. 2004;16:1163–1177. doi: 10.1105/tpc.019943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurie S., McKibbin R.S., Halford N.G. Antisense SNF1-Related (SnRK1) protein kinase gene represses transient activity of an α-amylase (α-Amy2) gene promoter in cultured wheat embryos. Journal of Experimental Botany. 2003;54:739–747. doi: 10.1093/jxb/erg085. [DOI] [PubMed] [Google Scholar]
- Mangelsen E., Kilian J., Herter K., Jansson C., Wanke D., Sundberg E. Transcriptome analysis of high-temperature stress in developing barley caryopses: early stress responses and effects on storage compound biosynthesis. Molecular Plant. 2011;4:97–115. doi: 10.1093/mp/ssq058. [DOI] [PubMed] [Google Scholar]
- McKibbin R.S., Muttucumaru N., Paul M.J., Powers S.J., Burrell M.M., Coates S., Purcell P.C., Tiessen A., Geigenberger P., Halford N.G. Production of high-starch, low-glucose potatoes through over-expression of the metabolic regulator SNRK1. Plant Biotechnology Journal. 2006;4:409–418. doi: 10.1111/j.1467-7652.2006.00190.x. [DOI] [PubMed] [Google Scholar]
- Ramakers C., Ruijter J.M., Deprez R.H., Moorman A.F. Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neuroscience Letters. 2003;339:62–66. doi: 10.1016/s0304-3940(02)01423-4. [DOI] [PubMed] [Google Scholar]
- Rieu I., Powers S.J. Real-time quantitative RT-PCR: design, calculations and statistics. Plant Cell. 2009;21:1031–1033. doi: 10.1105/tpc.109.066001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiler C., Harshavardhan V.T., Rajesh K., Reddy P.S., Strickert M., Rolletschek H., Scholz U., Wobus U., Sreenivasulu N. ABA biosynthesis and degradation contributing to ABA homeostasis during barley seed development under control and terminal drought-stress conditions. Journal of Experimental Botany. 2011;62:2615–2632. doi: 10.1093/jxb/erq446. [DOI] [PubMed] [Google Scholar]
- Sreenivasulu N., Radchuk V., Strickert M., Miersch O., Weschke W., Wobus U. Gene expression patterns reveal tissue-specific signalling networks controlling programmed cell death and ABA-regulated maturation in developing barley seeds. Plant Journal. 2006;47:310–327. doi: 10.1111/j.1365-313X.2006.02789.x. [DOI] [PubMed] [Google Scholar]
- Vicente-Carbajosa J., Moose S.P., Parsons R., Schmidt R.J. A maize zinc-finger protein binds the prolamin box in zein gene promoters and interacts with the basic leucine zipper transcriptional activator Opaque 2. Proceedings of the National Academy of Sciences USA. 1997;94:7685–7690. doi: 10.1073/pnas.94.14.7685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan Y., Poole R.L., Huttly A.K., Toscano-Underwood C., Feeney K., Welham S., Gooding M.J., Mills C., Eewards K.J., Shewry P.R., Mitchell R.A. Transcriptome analysis of grain development in hexaploid wheat. BMC Genomics. 2008;9:121. doi: 10.1186/1471-2164-9-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamauchi D., Zentella R., Ho T.D. Molecular analysis of the barley (Hordeum vulgare) gene encoding the protein kinase PKABA1 capable of suppressing gibberellin action in aleurone layers. Planta. 2002;215:319–326. doi: 10.1007/s00425-002-0740-6. [DOI] [PubMed] [Google Scholar]
- Yoshida T., Fujita Y., Sayama H., Kidokoro S., Maruyama K., Mizoi J., Shinozaki K., Yamaguchi-Shinozaki K. AREB1, AREB2, andABF3 are master transcription factors that co-operatively regulate ABRE-dependent ABA signalling involved in drought stress tolerance and require ABA for full activation. Plant Journal. 2010;61:672–685. doi: 10.1111/j.1365-313X.2009.04092.x. [DOI] [PubMed] [Google Scholar]
- Zhang Y., Shewry P.R., Jones H., Barcelo P., Lazzeri P.A., Halford N.G. Expression of antisense SnRK1 protein kinase sequence causes abnormal pollen development and male sterility in transgenic barley. Plant Journal. 2001;28:431–442. doi: 10.1046/j.1365-313x.2001.01167.x. [DOI] [PubMed] [Google Scholar]
- Zhang Y., Andralojc P.J., Hey S.J., Primavesi L.F., Specht M., Koehler J., Parry M.A.J., Halford N.G. Arabidopsis sucrose non-fermenting-1-related protein kinase-1 and calcium-dependent protein kinase phosphorylate conserved target sites in Aba response element binding proteins. Annals of Applied Biology. 2008;153:401–409. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.