Abstract
The primary signal of sex determination in the honeybee, the complementary sex determiner (csd) gene, evolved from a gene duplication event from an ancestral copy of the fem gene. Recently, other paralogs of the fem gene have been identified in several ant and bumblebee genomes. This discovery and the close phylogenetic relationship of the paralogous gene sequences led to the hypothesis of a single ancestry of the csd genetic system of complementary sex determination in the Hymenopteran insects, in which the fem and csd gene copies evolved as a unit in concert with the mutual transfers of sequences (concerted evolution). Here, we show that the paralogous gene copies evolved repeatedly through independent gene duplication events in the honeybee, bumblebee, and ant lineage. We detected no sequence tracts that would indicate a DNA transfer between the fem and the fem1/csd genes between different ant and bee species. Instead, we found tracts of duplication events in other genomic locations, suggesting that gene duplication was a frequent event in the evolution of these genes. These and other evidences suggest that the fem1/csd gene originated repeatedly through gene duplications in the bumblebee, honeybee, and ant lineages in the last 100 million years. Signatures of concerted evolution were not detectable, implicating that the gene tree based on neutral synonymous sites represents the phylogenetic relationships and origins of the fem and fem1/csd genes. Our results further imply that the fem1 and csd gene in bumblebees, honeybees, and ants are not orthologs, because they originated independently from the fem gene. Hence, the widely shared and conserved complementary sex determination mechanism in Hymenopteran insects is controlled by different genes and molecular processes. These findings highlight the limits of comparative genomics and emphasize the requirement to study gene functions in different species and major hymenopteran lineages.
Introduction
Complementary sex determination, in which the heterozygous genotype at a certain locus determines femaleness, is widely shared in hymenopteran insects and has a deep ancestry [1], [2]. Thus far, the underlying gene complementary sex determiner (csd) has been identified in the western honeybee (Apis mellifera) by positional cloning and knockdown studies [3], [4]. The csd gene encodes an SR-type protein. Csd proteins derived from the heterozygous csd genotype induce the female sex pathway by directing the female splicing of the primary transcripts of the fem gene [4], [5]. The resulting female mRNAs subsequently encode the functional Fem proteins. Csd proteins derived from the hemizygous or homozygous genotypes are not required for sex determination. The male splicing of the fem transcripts results by default. The male-specific exons contain a translational stop codon to prematurely stop translation. The absence of functional Fem proteins leads to the development of maleness [5]. More than 14 csd alleles have been identified in local honeybee populations, which show an average of 3% pairwise difference in their entire amino acid encoding sequence [6], [7].
The low divergence of the honeybee csd and fem genes at synonymous sites compared to bumblebee and stingless bee sequences suggests that the csd gene was derived from a gene duplication event of an ancestral copy of the fem gene in the honeybee lineage [4]. The csd gene was shaped by positive selection shortly after it originated [4], [8]. fem is the putative ortholog of the transformer (tra) gene [4], a key sex-determining gene in Drosophila melanogaster. However, unambiguous homology relies on identities in a 30-amino-acid motif deduced from another dipteran ortholog of the tra gene from Ceratitis capitata [9], [10].
A recent study found repeated duplicates of the fem gene in four ant and two bumblebee genomes [11]. The transcripts of these fem genes are sex-specifically spliced, suggesting a conserved sex-determining role of this gene. The function of the duplicated copies are thus far unknown [11]. The wasp Nasonia vitripennis, however, lacks a sister copy of the fem/tra gene [12]. In this study, we named the other copies of the fem gene fem1. This is because we have no functional information as to whether these genes control the complementary sex determination process as in honeybees.
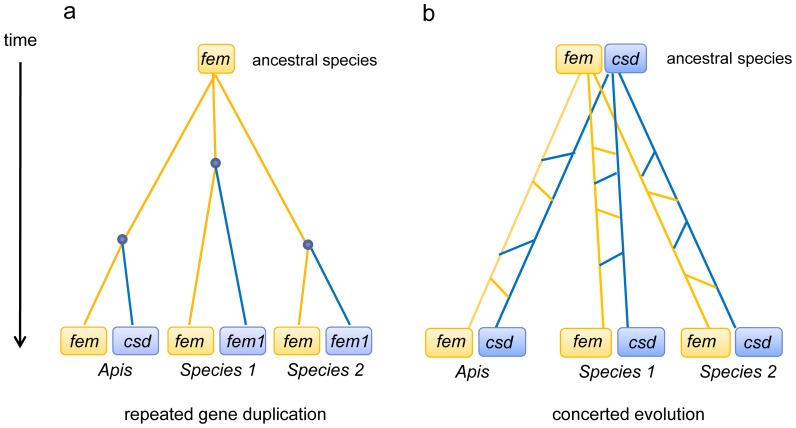
The phylogenetic relationships deduced from coding nucleotide sequences [11] showed that the paralogous gene pairs of fem and csd/fem1 are more closely related in four ant species, the bumblebee and, as previously shown, the honeybee lineage. Figure S1 shows the sequence relationship of the genes for the neutral synonymous sites. Two recent studies [11], [13] have proposed that in contrast to a model of independent gene duplications, the most parsimonious explanation of the close relationship between the fem and fem1/csd sequences is that concerted evolution (either due to repeated unequal crossing-over or gene conversion) homogenized the duplicated copies in the different lineages (Fig. 1). One or a few ancestral duplication events gave rise to the csd gene and complementary sex determination observed in the Hymenoptera order [11], [13]. The process of concerted evolution between the fem and fem1/csd genes repeatedly homogenized the two loci, producing the low divergence in the gene pair that we find today.
Figure 1. Two models for the evolutionary history of fem paralogous genes in ants and bees: (a) repeated gene duplication and (b) concerted evolution.
Points in (a) denote gene duplication events giving rise to two gene copies. Connecting lines in (b) between branches indicate concerted evolution events resulting from unequal crossing over and/or gene conversion.
Here, we readdress the question of whether the fem1/csd copies repeatedly evolved through gene duplication (Fig. 1a) or whether the fem and fem1/csd gene pairs evolved through concerted evolution (Fig. 1b). The clarification of this question will provide fruitful insight into the evolution of paralogous genes and the evolution of a complementary sex determination system.
The arguments given below prompted us to further investigate this question.
Studies at the genome-wide scale showed that concerted evolution only affects 2% of the paralogous gene pairs [14], suggesting that this process rarely acts as a homogenizing force between paralogs.
The rate for the rise of new paralogous gene copies is 0.01 per gene per million years [15], [16]. This suggests that new duplicates of the fem gene can repeatedly originate in different hymenopteran lineages, which have an evolutionary history of more than 120 million years [17]–[20].
The evidence for concerted evolution between the paralogs provided thus far, namely, (i) the alternative tree topologies of the fem and fem1/csd nucleotide sequences [13] and (ii) the putative gene conversion tracts in the nucleotide sequence [11], [13], could also result from a heterogeneity in the sequence divergence, a recombination event between the csd alleles, methodological problems or homoplasic (convergent) nucleotide changes [4], [6], [8], [21].
Here, we present evidence suggesting that the paralogous gene copies fem1/csd in ants, bumblebees and honeybees evolved independently and repeatedly through a series of gene duplication events (Fig. 1a).
Results
Amino acid changes in the MRCA ancestral sequences of bees and ants are shared between the Fem and Csd/Fem1 proteins
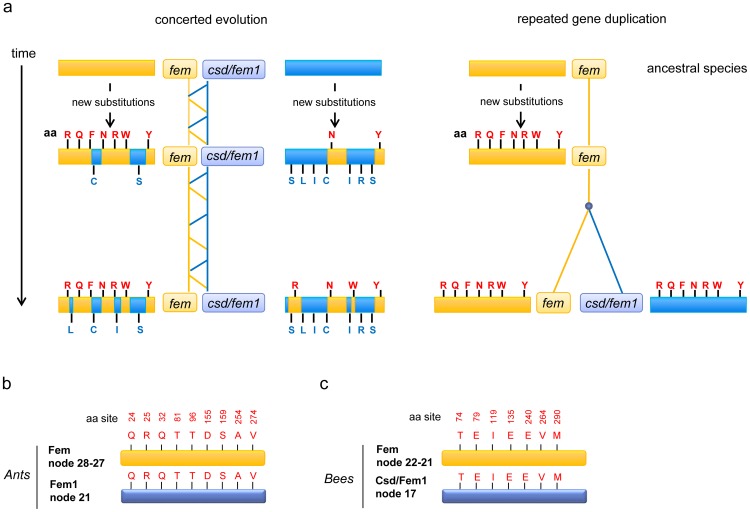
To find further support for either the repeated gene duplication model or the concerted evolution model, we followed the evolutionary trajectory of substitutions that led to amino acid changes in the ancestral sequences of the most recent common ancestor (MRCA) of bees and ants (Fig. 2a). This evolutionary window predates the timing of the different gene duplication events under the repeated duplication model and can therefore provide unique information about the evolutionary history of the sister copies. Under the concerted evolution model, we would expect to find unique substitutions in the ancestral sequences of the MRCA of ants and bees, which are confined to the fem or the csd/fem1 gene (Fig. 2a). This pattern would arise because the two sister copies originated only once in Hymenoptera [11]and accumulated substitutions separately due to their separate evolutionary history, which predates the MRCA of bees and ants [11]. Concerted evolution, the exchange of sequences between evolutionary old paralogous genes, would partly homogenize the sister copy genes, which would thus appear as to have more recent common ancestry in the phylogenetic tree (Fig. S1, S2). Under the repeated gene duplication model, the ancestral sequence in the evolutionary time window that predates the different duplication events should be the same for the fem and csd/fem1 genes because at this time point, only a single copy of the gene existed (Fig. 2a).
Figure 2. The evolutionary fate of fem gene substitutions in an evolutionary window predating the putative repeated gene duplications.
(a) The expected evolutionary fate of fem substitutions in the paralogous genes fem and csd/fem1 under the model of concerted evolution and repeated gene duplication. (b, c) The letters above the yellow boxes show the inferred amino acid changes in the Fem protein tree that evolved during the evolutionary window of the MRCA of ants and bees and the MRCAs of ants (b) and of bees (c). Letters above the blue boxes indicate the amino acid residues that are found at the homologous sites in the ancestral Csd/Fem1 protein sequence of the MRCA of ants (b) and of bees (c). Numbers above the letters designate the homologous sites in the Fem amino acid sequence alignment. Numbers before the boxes indicate the nodes (Fig. S2) used to infer the ancestral sequence information. aa denotes amino acid.
We generated separate phylogenetic trees using the amino acid sequence of the Fem and Fem1/Csd proteins, which allowed us to trace the putative separate evolutionary history of these sister copy genes. We inferred the ancestral amino acid sequences of the MRCAs of bees and of ants (Fig. 2, S2) using the maximum likelihood method [22]. These nodes had high statistical support and represented evolutionary time windows before the putative repeated gene duplication events. For the large evolutionary distances between ants and bees, we analyzed amino acid changes instead of synonymous substitutions, which were saturated, at least for the less degenerate sites. We identified changes in the Fem protein of the MRCA amino acid sequence of bees and ants by comparing the MRCA sequences of bees and of ants. We found 7 changes in the MRCA sequence of bees and 9 in the MRCA sequence of ants. In ants, we found the same 9 amino acid changes in the sister copy sequence of the paralogous Fem1 protein (Fig. 2b, Fig. S3). In bees, we found the same 7 amino acid changes in the MRCA sequence of the paralogous Csd/Fem1 protein (Fig. 2c, Fig. S4). These informative changes in the ant and bee sequences are found in different parts of the protein, suggesting that we obtained information that covered the entire protein. Our study found no amino acid changes that were confined to only one of the sister copies, which would indicate a deeper ancestry of the gene duplication that predated the MRCA of bees and of ants. Therefore, the sequences harbor no information that can provide evidence for a separate history of the fem and csd/fem1 genes that predates those of the ant and bee lineages, or for a single gene duplication event in Hymenoptera.
No evidence of concerted evolution is found in bumblebee sequences
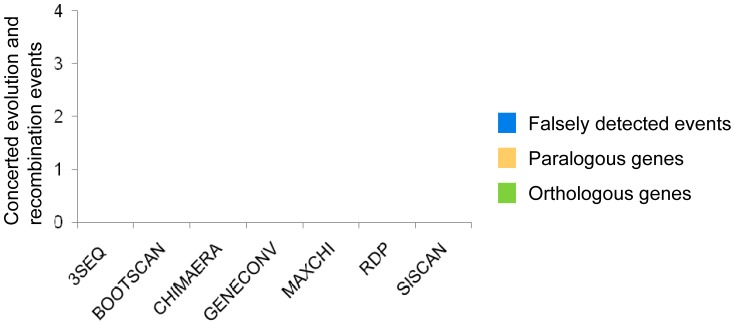
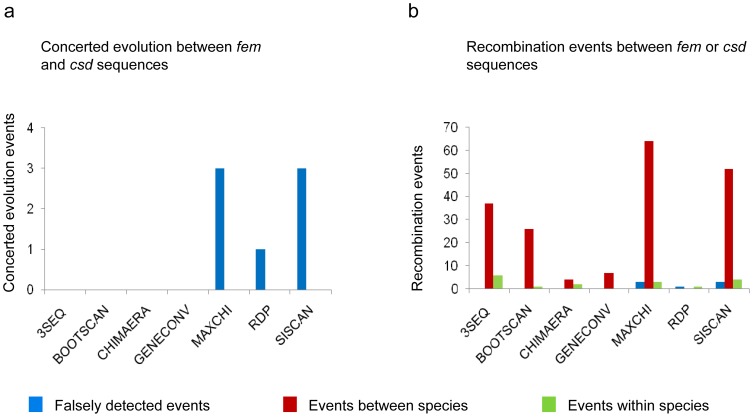
We studied whether we can detect sequence tracts that would indicate a transfer of sequences between paralogous genes (concerted evolution) in the bumblebee lineage. The phylogenetic clustering of the fem and fem1 sequences in the bumblebee lineage (Fig. S1) suggests that concerted evolutionary events should also occur in the bumblebee lineage. We used seven methods (RDP, GENECONV, BootScan, MaxChi, Chimaera, SiScan and 3Seq) designed to detect tracts of recombination events in the nucleotide sequences, which are included in the RDP 3.44 software package [23]. We applied this method to a single sequence alignment, which included the fem and fem1 sequences of the two Bombus species and the fem sequence from A. mellifera as an outgroup reference. We classified the detected transfer events as either (i) concerted evolution events if they occurred between paralogous genes, (ii) recombination events if they occurred between the same gene, and (iii) falsely discovered events (FDE) if these events are biologically implausible. Such biologically implausible events are events in which sequence transfer between orthologous genes should give rise to a paralogous recombinant sequence or are events in which the sequence of the outgroup reference species is involved. The results of the analysis are shown in Figure 3. We found no sequence tracts that were transferred between paralogous genes, suggesting that concerted evolution played no role in the evolution of the paralogous genes of the Bombus lineage.
Figure 3. Number of gene conversion and recombination events in B. terrestris and B. impatiens sequences.
Tracts of putatively recombined sequence were detected by the 7 methods as shown on the x-axis and using RDP 3.44 software program. The analysis was run on a single alignment of the fem and fem1 sequences of the B. terrestris, B. impatiens and A. mellifera fem sequence. Gene conversion events refer to DNA transfers between the paralogous genes fem and fem1. Recombination events indicate transfer events between sequences of the same gene. Falsely detected events (FDE) refer to biologically implausible events (see Materials and Methods).
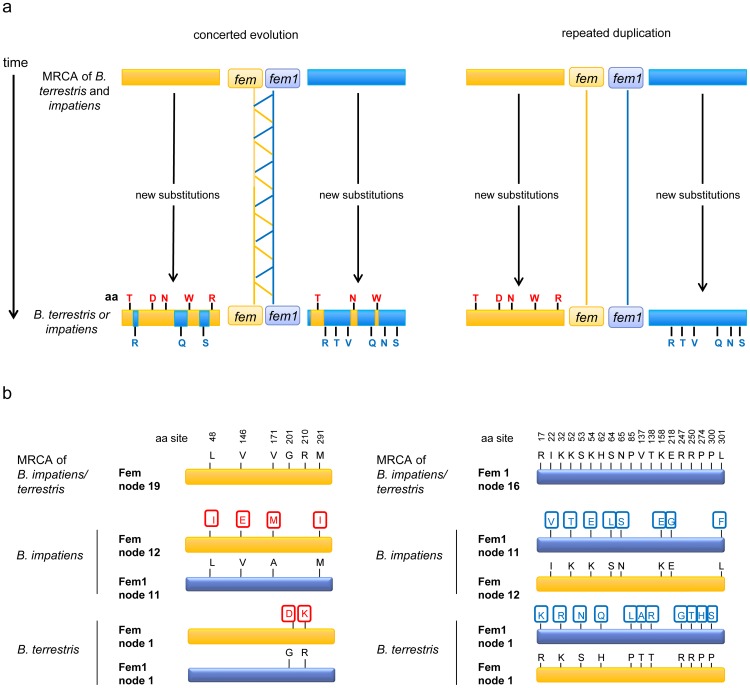
We next studied whether we can identify signatures of concerted evolution by confining our analysis to single amino acid substitutions. We tested whether some fem or fem1 substitutions that newly evolved in each bumblebee species were transferred to its paralogous sister gene (Fig. 4a). Such shared evolved states between the two paralogous genes within each species would indicate a transfer of the corresponding nucleotide sequence by concerted evolution. We determined the ancestral sequences of the Fem and Fem1 protein of the MRCA of Bombus terrestris and Bombus impatiens, identified the evolutionary changes and studied whether these changes were also present in the paralogous sister copy (Fig. 4a). We found 4 newly evolved amino acid changes in the Fem protein of B. impatiens and 2 in that of B. terrestris. All of these newly evolved changes were not present in the sister Fem1 proteins (Fig. 4b, Fig. S5). We detected 8 newly evolved amino acid changes in the Fem1 protein of B. impatiens and 11 in B. terrestris, and these newly evolved changes were not present in the sister Fem protein (Fig. 4b, Fig. S6). Taken together, we found that all 12 newly evolved amino acid changes in B. impatiens and all 13 newly evolved amino acid changes in B. terrestris were absent in their corresponding sister copies. This survey covered different parts of the approximately 400 amino acid (aa)-long protein, providing evidence that concerted evolution events were absent in the Bombus fem and fem1 sequences.
Figure 4. The evolutionary trajectory of fem gene substitutions in the evolutionary window that follows the putative gene duplication event in the Bombus lineage.
(a) The expected evolutionary fate of fem substitutions in the paralogous genes fem and fem1 under the models of concerted evolution and repeated gene duplication. (b) Deduced amino acid changes. The yellow box indicates the Fem protein, and the blue box indicates the Fem1 protein. Black letters above the boxes indicate the ancestral state of the amino acid residues found in the MRCA of B. terrestris and B. impatiens. Red letters in the red frame indicate the amino acid residues that evolved since the MRCA of B. terrestris and B. impatiens in the Fem protein. Blue letters in the blue frame indicate the amino acid residues that evolved since the MRCA of B. terrestris and B. impatiens in the Fem1 protein. Numbers before the boxes indicate the nodes (Fig. S2) used to infer the ancestral sequence information. aa denotes amino acid.
No evidence of concerted evolution in sequences of the honeybee lineage
Next we applied the RDP, GENECONV, BootScan, MaxChi, Chimaera, SiScan and 3Seq methods to identify sequence tracts of concerted evolution between the paralogous fem and csd nucleotide sequences of the honeybee. We included the same coding nucleotide sequences as in a previous study [13], comprised of 36 csd and 1 fem A. mellifera, 16 csd and 1 fem Apis cerana csd, 19 csd and 1 fem Apis dorsata sequence and a fem B. terrestris sequence as an outgroup reference.
In this study, we applied the methods to a single sequence alignment that included all fem and csd sequences. In the previous study from Privman et al., 100 alignments were used, each consisting of the fem sequences and a randomly chosen csd allele sequence from each Apis species and a fem sequence from B. terrestris. The rationale behind our altered experimental design was that the detection methods used in the RDP 3.44 software program are designed for large datasets, to identify the recombinant sequence and the two sequences from which the recombinant sequence was derived [23]. We removed a sequence (GenBank accession #: AY352276) from the analysis because it was a chimeric sequence of the csd and fem gene. This sequence resulted from a misassembly of cDNA sequences derived from the fem and csd gene at a point in time when we had no knowledge about a second gene in the genome and the nature of allelic variation [3]. This sequence entry has now been removed from GenBank. We also updated the sequence (GenBank accession #: AY350616), which is a fem and not a csd sequence. The results of this sequence analysis are shown in (Fig. 5).
Figure 5. Events of gene conversion and recombination in the fem and csd sequences of A. mellifera, A. dorsata and A. cerana.
Tracts of putatively recombined sequences were detected by seven different methods as indicated on the x-axis using the RDP 3.44 software program. The analysis was run on a single alignment of 71 csd and 4 fem sequences. (a) The number of concerted evolution events that refer to the DNA transfer between paralogous genes fem and csd. (b) The number of recombination events that identify events between sequences of the same gene. Falsely detected events (FDE) in (a) and (b) refer to biologically implausible events. The outgroup reference sequence, B. terrestris fem, was never involved in one of the detected events.
We found no tracts of gene conversion (Fig. 5a), suggesting that a DNA transfer between the fem and csd gene did not occur in the honeybee lineage. We observed 3 events in which recombination between the Apis csd ortholog sequences gave rise to a paralogous Apis fem sequence, suggesting falsely detected events (FDE) that were identified by the program. We confirmed this falsely detection by demonstrating that the putatively transferred fragment is indeed fem derived which we showed by the clustering into the fem gene cluster in the phylogenetic tree analysis. However, we detected multiple recombination events between the csd sequences (alleles) derived from the same and different Apis species (Fig. 5b). This suggests that recombination is a regular process between alleles of the csd gene, consistent with previous reports [7], [21].
We next evaluated how the number of sequences affects the false detection of events. We generated 20 sequence alignments, each consisting of two randomly chosen csd sequences from a single Apis species and one fem sequence from B. terrestris, which served as the outgroup reference sequence for the alignment above. For these alignments, the GENECONV method detected 8, the MaxChi method detected 12, and the Chimaera method detected 9 tracts of sequences in which DNA was putatively transferred between the csd sequences of the Apis and fem gene of the B. terrestris sequences (Fig. S7). However, these events are biologically implausible, because the nucleotide differences we observe today in the csd alleles evolved after the split into different Apis species [7]. Hence, the Bombus sequence cannot have contributed through concerted evolutionary events to the csd polymorphism. We confirmed this falsely detection in a sample of detected events by demonstrating that the putatively transferred fragment is not derived from the Bombus sequence, which we showed by the clustering into the csd gene cluster in the phylogenetic tree analysis. We detected no such transfer between the Apis csd alleles and the B. terrestris fem sequences if all sequences are included in a single alignment (Fig. 5) suggesting that having fewer sequences in an alignment increases the rate of falsely detecting DNA transfer between paralogs.
We did not perform single amino acid substitution analysis in the honeybee as we did for Bombus because we have not robustly identified enough newly evolved sites.
dN/dS ratio differences between paralogous genes suggest a directional DNA transfer process
We consistently observed that the dN/dS ratios (nonsynonymous (dN) to synonymous (dS) per site substitutions) of the fem1/csd sequence pairs were higher compared to those of the fem sequence pairs ((Table 1); χ2-test,P<0.05 for all comparisons), suggesting that selection operates differently on the fem and the fem1/csd genes. The ratio of the differences is most pronounced for the Apis sequences (dN/dSfem = 0.1–0.2 versus dN/dScsd = 0.8–1), consistent with previous findings [4], [8], and for the bumblebee sequences (dN/dSfem = 0.16 versus dN/dScsd = 0.56). The difference in the dN/dS ratios is less pronounced in the ants.
Table 1. The dN and dS values and ratios for the interspecies comparisons of the fem and fem1/csd genes.
| fem gene | fem1/csd gene | dN/dSfem | |||||||
| Clade | Species | dN (SE) | dS (SE) | dN/dS | dN (SE) | dS (SE) | dN/dS | <dN/dSfem 1/csd * | |
| Apis | Ador | Amel | 0.02 (0.004) | 0.09 (0.02) | 0.22 | 0.13 (0.01) | 0.13 (0.02) | 1.00 | P<0.0001 |
| Amel | Acer | 0.01 (0.003) | 0.08 (0.02) | 0.13 | 0.12 (0.01) | 0.15 (0.02) | 0.8 | P<0.0001 | |
| Bombus | Bter | Bimp | 0.008 (0.003) | 0.051 (0.013) | 0.16 | 0.05 (0.007) | 0.09 (0.02) | 0.56 | P<0.01 |
| Cflo | Hsal | 0.26 (0.01) | 0.51 (0.029) | 0.51 | 0.35 (0.02) | 0.5 (0.03) | 0.7 | P<0.01 | |
| Cflo | Pbar | 0.19 (0.01) | 0.44 (0.03) | 0.43 | 0.3 (0.01) | 0.43 (0.03) | 0.7 | P<0.01 | |
| Ants | Hsal | Pbar | 0.24 (0.01) | 0.53 (0.03) | 0.45 | 0.26 (0.01) | 0.46 (0.03) | 0.57 | P<0.02 |
| Acep | Pbar | 0.11 (0.01) | 0.37 (0.03) | 0.30 | 0.16 (0.01) | 0.41 (0.03) | 0.39 | P<0.05 | |
| Cflo | Acep | 0.21 (0.01) | 0.46 (0.03) | 0.46 | 0.32 (0.01) | 0.49 (0.03) | 0.65 | P<0.01 | |
| Acep | Hsal | 0.23 (0.01) | 0.5 (0.03) | 0.46 | 0.28 (0.01) | 0.44 (0.03) | 0.64 | P<0.01 | |
Species names: Amel, A. mellifera; Ador, A. dorsata; Acer, A. cerana; Bimp, B. impatiens; Bter, B. terrestris; Hsal, H. saltator; Pbar, P. barbatus; Acep, Atta cephalotes; Cflo, Camponotus floridanus. SE: standard error.
*A one-tailed χ2 - test was conducted using the absolute number of synonymous and nonsynonymous differences.
We further evaluated how differences in the dN/dS ratios are compatible with the mutual transfer of DNA and concerted evolution. The mutual transfer between the paralogous genes would also transfer the differences in the dN/dS ratios between paralogous genes. We assume that the entire sequences are in equilibrium of homogenization through concerted evolution and divergence. This is consistent with the model that concerted evolution is a random mutational and ongoing process that occurs through hymenopteran phylogeny. At this equilibrium, the gene-wide dN/dS values are good approximations for DNA fragments that are, on average, transferred between paralogs.
First, we showed that there are not different rates at synonymous sites in the honeybee and the bumblebee clade (Tajima's relative rate test, P>0.05). This result suggests that the differences in the dN/dS ratios between the genes reflect substitution rate differences at nonsynonymous sites (dN).
DNA fragments transferred from the fem1/csd can only reach the lower dN/dS ratios in the fem gene as evolutionary time progresses if new mutations occur at the neutral synonymous sites along with purifying selection at the nonsynonymous sites. We approximated the mean number of neutral pairwise substitutions per site (dS) that is required to reach the lower dN/dS ratios. The csd sequences in honeybees show an average ratio of dN/dS csd = 0.9 (Table 1), suggesting that during the separation time of the two paralogous genes (in terms of dScsd/fem = 0.18, Table S1), approximately dN csd = 0.16 pairwise substitutions in the csd gene have accumulated. We next estimated dS, which has accumulated so that a transferred csd fragment (that has on average a ratio dN/dS csd = 0.9) can reach the observed dN/dS ratio of the fem gene (in which dN/dS fem = 0.17). We assume the most conservative model, in which all newly arising nonsynonymous mutations were removed by purifying selection and only new synonymous mutations became fixed. A csd fragment can only adjust for the fem's dN/dS ratio if dS x = 0.77 additional synonymous substitutions have accumulated. This result suggests that a transferred csd DNA requires on average dS = 0.77 synonymous substitutions to observe the low dN/dS ratio of the fem gene.
Similarly, we approximated dS x for bumblebee sequences. fem1-derived sequences in the fem sequence (dS fem1/fem = 0.22, dN/dS fem1 = 0.56, dN fem1 = 0.12, dN/dS fem = 0.16) would require dS x = 0.53 pairwise synonymous differences to accumulate in order for the sequence to reach the same dN/dS fem ratio.
dS x = 0.77 in honeybees and dS x = 0.53 in bumblebees, as required for the transferred csd DNA to reach the observed dN/dS ratios of the fem gene are largely exceeding dS = 0.39 that have accumulated between the bumblebee and honeybee species. This suggests that these transfers should predate the MRCA of bees, which is inconsistent with our previous result that there would be an absence of such transfers in this evolutionary window (Fig. 2). In addition, dS x largely exceeds the divergence between paralogous genes in the bumblebee (dS fem1/fem = 0.22) and the honeybee (dS fem/csd = 0.18), suggesting that the divergence between paralogs is too low to be compatible with such transfers and ratios. The results of this simple transfer model imply that the transfer processes cannot be bidirectional between paralogous genes, as predicted under a model of concerted evolution in which sequences are mutual exchanged and can become fixed through positive selection and genetic drift. Only a directional transfer from fem to csd/fem1 would be compatible with data suggesting that relaxed or positive selection could substantially increase the dN/dS ratio. This directional transfer process is consistent with the gene duplication model, in which a new gene copy becomes neofunctionalized [4].
Additional sequence copies are repeatedly found at other genomic loci in bees and ants
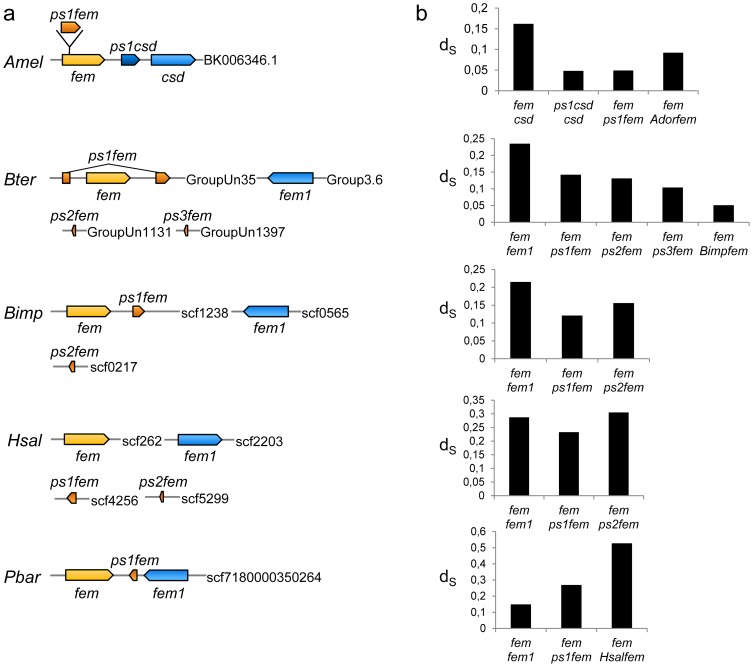
To find further evidence for the repeated duplication model, we searched for other gene duplication tracts of the fem and fem1/csd genes. Using BlastN searches, we found genomic sequences with a high similarity to the coding nucleotide sequences of the fem or csd genes in the western honeybee, two bumblebee species and two ant species at other genomic loci (Fig. 6a). These duplicate genes exist as inactive genes with no complete open reading frame (ORF), suggesting that they are pseudogenes (ps). For the ps1 csd gene of A. mellifera, we confirmed by RT-PCR that this gene is transcriptionally inactive. We estimated the synonymous pairwise divergence (dS) of the pseudogenes and of the fem or fem1/csd genes within each species (Fig. 6b), which we related to the MRCA events by providing the fem dS values of different species. For A. mellifera, we found two pseudogenes, one derived from the csd gene and one derived from the fem gene (Fig. 6). The latter of these gave rise to a new female intron sequence in the fem gene [5]. The dS divergence between pseudogenes and the fem or csd gene is smaller compared to the dS between the csd and fem and the dS between the fem of A. mellifera and A. dorsata (Fig. 6b), suggesting that both sequences were duplicated recently in the A. mellifera lineage. The dS divergence in the bumblebee genomes suggests that the origin of these duplications predates the split between the B. terrestris and B. impatiens species (Fig. 6b) but that it originated after the split of the current functional gene copies of fem and fem1. In the ant Harpegnathos saltator, we observed one pseudogene that originated after and one that arose during the split between the functional fem and fem1/csd gene copies. However, in the ant Pogonomyrmex barbatus, we also found a pseudogene with a much deeper ancestry (Pbarps1 fem; dS = 0.27) than the functional gene pairs. Our results demonstrate that other gene duplication events occurred throughout the phylogeny and even within the A. mellifera lineage.
Figure 6. Pseudogenes (ps) of the fem and csd genes in the ant, bumblebee and western honeybee genomes.
(a) The orientation and location of the pseudogenes (psfem, pscsd). Boxes denote the genes or pseudogenes. The box length of pseudogenes indicates the relative degree of homology to the coding nucleotide sequences of the fem or csd genes. The phylogenetic relationship assignments are based on the lowest dS estimates or the ancestral state. Numbers behind the bars indicate the genomic scaffold, linkage group or the GenBank accession number. (b) Evolutionary distance between duplicated fem and fem1/csd gene copies are presented in terms of pairwise synonymous divergence per synonymous site (dS). Abbreviations: Amel, A. mellifera; Bimp, B. impatiens; Bter, B. terrestris; Hsal, H. saltator; Pbar, P. barbatus.
Discussion
Our study presents several lines of evidence that support the repeated gene duplication model, but reject the concerted evolution model in which the low divergence of paralogs resulted from homogenization. We studied fem and csd/fem1 paralogous genes in several bee and ant species, representing 120 million years of evolution [17]–[20]. We first showed that there were no unique changes in the Fem or Csd/Fem1 proteins at a point in time that would indicate a separate history of the two gene copies, predating the MRCAs of bees and of ants. We detected no sequence tracts that would indicate a DNA transfer between the paralogs in two bumblebee and three honeybee species by using different methods. We also identified other tracts of duplicated copies of the fem and fem1/csd gene at other genomic loci in different ant and bee species, suggesting that repeated gene duplication is a frequent process in the evolution of these genes. Finally, we showed that the major differences in the dN/dS ratio between the fem and fem1/csd genes in bees exclude a mutual transfer of sequences but suggest a directional transfer from fem to the fem1/csd gene, which is consistent with gene duplication and a neofunctionalization model [4] and not with a mutual exchange of sequences under concerted evolution. We conclude from these results that the fem1/csd genes repeatedly originated through gene duplication in the bumblebee, honeybee and ant lineages. Concerted evolution played no detectable role in the evolution of these genes, suggesting that the phylogenetic relationship of the paralogs is represented by a gene tree based on neutral synonymous sites (Fig. S1).
Our finding is consistent with frequency estimates of gene duplication and concerted evolution events. Previous studies estimated that a gene will, on average, duplicate every 100 million years [15], [16], which is consistent with our finding of repeated gene duplications of the fem gene in the phylogeny of ants and bees which split approximately 120 million years ago [17]–[20]. Another study showed that gene conversion is a rare event, detectable in only 2% of duplicated genes, and that this process requires physical distances smaller than 9 kb [14]. Contrary to the latter requirement, fem and csd gene are separated by more than 12 kb in the honeybee (A. mellifera). Studies of genes that have multiple copies in the genome have demonstrated that new copies constantly originate by gene duplication [24]–[35]. Some of the duplicated copies are maintained in the genome for an extended period of time, while other copies were deleted or became nonfunctional through the accumulation of deleterious mutations.
Previous studies [11], [13] proposed that concerted evolution produced the low divergence of the fem and fem1/csd genes. The authors suggested that (i) alternative tree topologies of the nucleotide sequences [13] and (ii) putative gene conversion tracts [11], [13] are evidence for concerted evolution. Schmieder et al. identified gene conversion tracts in the genomic sequences of the paralogs. Our results showed that de novo duplications of the fem and fem1/csd genes generated such tracts. Privman et al. counted more than 100 recombination events in the honeybee fem and csd sequences, which they take as evidence for concerted evolution. In our reanalysis of the same sequences using the same methods (Fig. 5), we distinguished whether the transfer of DNA occurred between paralogous genes (concerted evolution) or between alleles of the same (orthologous) gene (recombination events). We found no tracts of gene conversion events between the fem and csd sequences (Fig. 5a), suggesting the absence of concerted evolution in honeybees. However, we found repeated transfers between alleles of the same (csd) gene (Fig. 5b), a finding which has been repeatedly reported [7], [21].
In our reanalysis, we used the entire 75-sequence data set in a single alignment, in contrast to the Privman et al. study, which used 100 alignments of 7 fem and csd sequences with different sets of csd alleles chosen from each species. We inspected some of the results using the Privman et al. alignments and repeatedly found tracts that suggested recombination events between the csd alleles. We also demonstrated in this study that, as in the work of Privman et al., small alignments of only a few sequences (Fig. S7) can generate an increase of the number of falsely detected events.
Privman et al. also proposed that the differences in the phylogenetic relationships of recombinant and non-recombinant regions provide further evidence for concerted evolution. Because these falsely detected “recombinant” sequence tracts are sequences from the same gene (csd) (see Fig. 5) these inconsistencies in the phylogenetic relationships are no further evidence of concerted evolution events. These “inconsistencies” have been previously reported for csd alleles. The combined forces of meiotic recombination and balancing selection generate a heterogeneity of divergence across the csd gene [6], [7], [21]. Recombination redistributes a small subset of variants of the 5′ region with multiple, highly diverged 3′ variants, which generates inconsistencies in the resulting phylogenetic relationships as previously shown [7], [21], [36].
Privman et al. found fem-specific substitutions for csd allele AY352276. However, this sequence is actually a chimeric sequence that was generated by the misassembly of the fem and csd cDNA sequences. This sequence was generated at a point in time when we searched for a third csd allele and had no knowledge of a second gene or the nature of the allelic diversity [3]. This sequence entry has been removed from GenBank. Privman et al. also suggested that alternative tree topologies of fem and fem1 sequences in the ants indicate gene conversion events [13]. We argue that the divergence of these sequences is too high (dS = 0.5 for most species pairs) to exclude the possibility that ambiguous trees (and split phylogenetic networks) resulted from homoplasic (convergent) substitutions in the sequences.
Our results imply that the fem1 and csd genes in the ant, bumblebee and honeybee species are not orthologs because they originated independently through gene duplications (Fig. S1). The csd gene originated in the honeybee lineage [4], [5]. Hence, complementary sex determination in bumblebee and ant species [1], [2] is regulated by other genes and not by the orthologs of the csd gene. Consistent with this conclusion, the csd alleles of honeybees share a hypervariable region of asparagine- and tyrosine-enriched repeats [7] that are consistently absent in the fem1 genes of ant and bumblebee species.
Our results show that a new gene for complementary sex determination originated in honeybees [4], while the phylogenetic distribution of complementary sex determination indicates a deep ancestry in Hymenopteran insects [1], [2]. One explanation for the replacement of a complementary sex determination gene is that ancestral complementary sex determiner genes degenerate over evolutionary time [4], [10]. This is because meiotic recombination is suppressed at the sex determiner gene locus [21], allowing more deleterious mutations to accumulate over time [37]–[39]. This process could generate an adaptive advantage for evolving new sex determination genes that would eventually replace the older, malfunctioning, complementary sex determination gene. Such a degeneration process has been proposed for sex chromosomal systems [40]–[45] and may also explain the rapid evolution of the complementary sex determination system.
Characterizing the gene functions of the fem1 genes and the molecular basis of complementary sex determination in ants and bumblebees would provide interesting insights into the evolution of this sex determination system. Our results imply that the conserved phenotype (complementary sex determination) is only loosely evolutionary associated with the controlling molecular process. These findings highlight the limits of comparative genomics and emphasize the requirement to study gene functions in different species and major hymenopteran lineages.
Materials and Methods
The fem paralogous sequences defined in this study as fem1 (in ants and bumblebees) or csd (in honeybees) were taken from Schmieder et al. [11] and were provided by Schmieder, S. and Poirie, M. The genomic locus of the fem/fem1 gene of Solenopsis invicta was not accessible in public libraries. The honeybee fem and csd sequences that we used to detect tracts of DNA transfers were kindly provided by Privman, E. The coding sequences were aligned based on the deduced amino acids, assuming a standard genetic code table. We used either the Clustal or the Muscle program that was implemented in the MEGA5 program suite [46] to align the coding nucleotide sequences according to the deduced amino acid sequences. The alignments were edited manually. Nucleotide and amino acid substitution analyses were conducted using the MEGA5 program. Maximum likelihood fits of the substitution models with the lowest Bayesian information criterion (BIC) score were used to choose a substitution model for amino acids and nucleotides when possible. Pairwise gaps in the alignment were deleted.
Ancestral sequences
Ancestral amino acid sequences were inferred using the maximum likelihood method [22] under the Jones-Taylor-Thornton (JTT) matrix-based model [47]. The rates between sites were treated as a gamma distribution. The ancestral sequences were inferred from separate fem and fem1/csd sequence alignments, and Figure S2 shows the gene tree used. All informative changes used had a probability P>0.5. We had to exclude some sites where the homology between sites of the Fem protein and the Csd/Fem1 alignment were ambiguous. In three cases of the bumblebee analysis, we changed the ancestral state we obtained from the MEGA analysis, because it contradicted the parsimony evolution of the sequence in the Fem and Fem1 tree (Fig. S5 and Fig. S6).
Evolutionary divergence between sequences
The dN/dS ratios of the interspecies comparisons between the ants, bumblebees and honeybees were inferred from sequence alignments that included either the bee or ant sequences, which greatly improved the number of identical positions in the alignments. The evolutionary distances between the ants and bees suggested that the less degenerated synonymous sites were saturated, making the dS estimates between the bee and ant sequences less reliable. Analyses were conducted using the Nei-Gojobori model to estimate either the nonsynonymous and synonymous substitutions per site or the absolute numbers. A χ2-square test was used to test the ratio differences in terms of absolute numbers. We tested the equality of the evolutionary rate at the most degenerate third codon position (which is presumably largely synonymous) by using Tajima's relative rate [48], which we performed using MEGA5 software [46]. We tested equality among the Apis csd and fem sequences by using the Bombus fem sequence as the outgroup. Similarly, we tested equality among Bombus fem1 and fem sequences by using the Apis fem sequence as an outgroup.
RT-PCR analysis
We performed RT-PCR experiments on embryonic and larval RNA [5] to identify possible splice products of mRNA in regions of the identified pseudogenes. PCR fragments were sequenced and compared with the genomic data.
Phylogenetic relationships
The phylogenetic relationship of the fem and fem1/csd sequences was determined based on presumably neutral synonymous differences by excluding the tra sequences of Ceratitis capitata and Nasonia vitripennis (because dS could not be estimated for these species) using the neighbor-joining method. The confidence probability (multiplied by 100) that the interior branch length is greater than 0 was estimated using the bootstrap test with 10,000 replicates. The evolutionary distances were computed using the Pamilo-Bianchi-Li method and are displayed in the units of the number of synonymous substitutions per synonymous site. For each sequence pair, all ambiguous positions were removed.
Sequence tracts of DNA transfers
Tracts of DNA transfers in the honeybee fem and csd sequences were identified using the RDP 3.44 software program [23]. We used the following tests: RDP [49] with internal and external references, GENECONV [50], BootScan [51], MaxChi [52], Chimaera [53], SiScan [54] and 3Seq [55]. The methods implemented in the RDP 3.44 software program relied on the identification of recombinant sequences and the parental sequences from which these recombinant sequences were derived, which is facilitated by having a large set of sequences [23]. We thus generated a single alignment. Our alignment included 36 A. mellifera, 16 A. cerana and 19 A. dorsata csd coding nucleotide sequences, the single fem nucleotide sequence from each Apis species, and the fem nucleotide sequence of B. terrestris as an outgroup reference sequence. We used the same program settings as described in a previous study [13].We removed sequence GenBank accession #: AY352276 from the analysis, as this is a chimeric sequence of the fem and the csd gene (the entry has now been deleted). We also updated the sequence (GenBank accession #: AY350616) because it is not a csd but a fem-derived sequence. To evaluate the effect of the number of sequences in the alignment, we generated 20 sequence alignments consisting of three sequences. Each alignment included two randomly chosen csd sequences from a single Apis species as well as the fem sequence from B. terrestris. We classified the detected transfer events as (i) concerted evolution events if they occurred between paralogous genes, (ii) recombination events if they occurred between the same gene and (iii) falsely discovered events (FDE) if they were biologically implausible. Such biologically implausible events are events in which the sequence transfer between orthologous genes should give rise to a paralogous recombinant sequence or events in which the sequence of the outgroup reference species was involved. We confirmed falsely detection by inferring the clustering of the sequence tracts into the gene cluster in phylogenetic tree analyses.
Supporting Information
Gene tree of the fem and fem1/csd sister copies in ants and bees, which were inferred from synonymous differences. The evolutionary history was inferred using the neighbor-joining method. The confidence probability (multiplied by 100) that the interior branch length is greater than 0 was estimated using the bootstrap test (10000 replicates are shown next to the branches). The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Pamilo-Bianchi-Li method [1] and are in the units of the number of synonymous substitutions per synonymous site. All ambiguous positions were removed for each sequence pair. There were a total of 575 positions in the final dataset. Evolutionary analyses were conducted in MEGA 5 [2]. The sequences of Nasonia and Ceratitis were excludeto estimate dS. Abbreviations: Acep, Atta cephalotes; Acer, Apis cerana; Aech, Acromyrmex echinatior; Ador, Apis dorsata; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris; Cflo, Camponotus floridanus; Hsal, Harpegnathos saltator; Mcom, Melipona compressipes; Pbar, Pogonomyrmex barbatus; Sinv, Solenopsis invicta.
(DOCX)
The initial tree of the Fem (a) and Fem1/Csd (b) proteins that were used to infer the ancestral amino acid sequences. The evolutionary history was inferred by using the maximum likelihood method based on the JFF model [1]. Numbers in the tree assign the different nodes for which the ancestral sequence were obtained. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. Abbreviations: Acep, Atta cephalotes; Acer, Apis cerana; Aech, Acromyrmex echinatior; Ador, Apis dorsata; Aflo, Apis florea; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris; Ccap, Ceratitis capitata; Cflo, Camponotus floridanus; Hsal, Harpegnathos saltator; Mcom, Melipona compressipes; Nvit, Nasonia vitripennis; Pbar, Pogonomyrmex barbatus; Sinv, Solenopsis invicta.
(DOCX)
The informative substitutions found in the ant lineage that were used in Figure 2 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicates the positions in the Fem and in the Csd/Fem1 protein sequence alignment.
(DOCX)
The informative substitutions found in the bee lineage that were used in Figure 2 . The identity of the different species and nodes of the Fem and Csd/Fem1 protein tree is shown. Site number (#) indicates the positions in the Fem and in the Csd/Fem1 protein sequence alignment.
(DOCX)
The informative substitutions found in the Bombus lineage (Fem tree) that were used in Figure 3 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicates the position in the alignment of the Fem and of the Csd/Fem1 protein sequences.
(DOCX)
The informative substitutions found in the Bombus lineage (Fem1 tree) that were used in Figure 3 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicate the position in the alignment of the Fem and of the Csd/Fem1 protein sequences.
(DOCX)
The number of falsely detected events (FDE) using the methods as indicated on the X axis. These programs were implemented in the RDP 3.44 software program. The methods were run on each of the 20 alignments which consisted of two randomly chosen csd sequences from a single Apis species and one fem sequence from B. terrestris. These events are falsely detected as this transfer involve the outgroup Bombus sequence and the polymorphism between csd alleles which newly evolved in the different Apis species.
(DOCX)
The dS i values for the paralogous gene pairs fem and csd/fem1 within each species (Ador, Apis dorsata; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris).
(DOCX)
Funding Statement
This work was supported by grants from the Deutsche Forschungsgemeinschaft, DFG. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Heimpel GE, de Boer JG (2008) Sex determination in the hymenoptera. Annu Rev Entomol 53: 209–230. [DOI] [PubMed] [Google Scholar]
- 2. Cook JM (1993) Sex determination in the hymenoptera: A review of models and evidence. Heredity (Edinburgh) 71: 421–435. [Google Scholar]
- 3. Beye M, Hasselmann M, Fondrk MK, Page RE, Omholt SW (2003) The gene csd is the primary signal for sexual development in the honeybee and encodes an SR-type protein. Cell 114: 419–429. [DOI] [PubMed] [Google Scholar]
- 4. Hasselmann M, Gempe T, Schiott M, Nunes-Silva CG, Otte M, et al. (2008) Evidence for the evolutionary nascence of a novel sex determination pathway in honeybees. Nature 454: 519–522. [DOI] [PubMed] [Google Scholar]
- 5. Gempe T, Hasselmann M, Schiott M, Hause G, Otte M, et al. (2009) Sex determination in honeybees: two separate mechanisms induce and maintain the female pathway. PLoS Biol 7: e1000222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hasselmann M, Beye M (2004) Signatures of selection among sex-determining alleles of the honey bee. Proc Natl Acad Sci U S A 101: 4888–4893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hasselmann M, Vekemans X, Pflugfelder J, Koeniger N, Koeniger G, et al. (2008) Evidence for convergent nucleotide evolution and high allelic turnover rates at the complementary sex determiner gene of Western and Asian honeybees. Mol Biol Evol 25: 696–708. [DOI] [PubMed] [Google Scholar]
- 8. Hasselmann M, Lechner S, Schulte C, Beye M (2010) Origin of a function by tandem gene duplication limits the evolutionary capability of its sister copy. Proc Natl Acad Sci USA 107: 13378–13383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pane A, Salvemini M, Bovi PD, Polito C, Saccone G (2002) The transformer gene in Ceratitis capitata provides a genetic basis for selecting and remembering the sexual fate. Development 129: 3715–3725. [DOI] [PubMed] [Google Scholar]
- 10. Gempe T, Beye M (2011) Function and evolution of sex determination mechanisms, genes and pathways in insects. Bioessays 33: 52–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Schmieder S, Colinet D, Poirie M (2012) Tracing back the nascence of a new sex-determination pathway to the ancestor of bees and ants. Nat Commun 3: 895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Verhulst EC, Beukeboom LW, van de Zand L (2010) Maternal control of haplodiploid sex determination in the wasp Nasonia . Science 328: 620–623. [DOI] [PubMed] [Google Scholar]
- 13. Privman E, Wurm Y, Keller L (2013) Duplication and concerted evolution in a master sex determiner under balancing selection. Proc Biol Sci 280: 20122968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Semple C, Wolfe KH (1999) Gene duplication and gene conversion in the Caenorhabditis elegans genome. J Mol Evol 48: 555–564. [DOI] [PubMed] [Google Scholar]
- 15. Lipinski KJ, Farslow JC, Fitzpatrick KA, Lynch M, Katju V, et al. (2011) High spontaneous rate of gene duplication in Caenorhabditis elegans . Curr Biol 21: 306–310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lynch M, Conery JS (2000) The evolutionary fate and consequences of duplicate genes. Science 290: 1151–1155. [DOI] [PubMed] [Google Scholar]
- 17.Grimaldi D, Engel MS (2005) Evolution of the Insects. Cambridge: University Press Cambridge, UK. [Google Scholar]
- 18. Ramirez SR, Nieh JC, Quental TB, Roubik DW, Imperatriz-Fonseca VL, et al. (2010) A molecular phylogeny of the stingless bee genus Melipona (Hymenoptera: Apidae). Mol Phylogenet Evol 56: 519–525. [DOI] [PubMed] [Google Scholar]
- 19. Wilson EO, Holldobler B (2005) The rise of the ants: a phylogenetic and ecological explanation. Proc Natl Acad Sci U S A 102: 7411–7414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Brady SG, Schultz TR, Fisher BL, Ward PS (2006) Evaluating alternative hypotheses for the early evolution and diversification of ants. Proc Natl Acad Sci U S A 103: 18172–18177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hasselmann M, Beye M (2006) Pronounced differences of recombination activity at the sex determination locus (SDL) of the honey bee, a locus under strong balancing selection. Genetics 174: 1469–1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nei M, Kumar S (2000) Molecular Evolution and Phylogenetics. New York: Oxford University Press. [Google Scholar]
- 23. Martin DP, Lemey P, Lott M, Moulton V, Posada D, et al. (2010) RDP3: a flexible and fast computer program for analyzing recombination. Bioinformatics 26: 2462–2463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Su C, Nei M (2001) Evolutionary dynamics of the T-cell receptor VB gene family as inferred from the human and mouse genomic sequences. Mol Biol Evol 18: 503–513. [DOI] [PubMed] [Google Scholar]
- 25. Rooney AP, Piontkivska H, Nei M (2002) Molecular evolution of the nontandemly repeated genes of the histone 3 multigene family. Mol Biol Evol 19: 68–75. [DOI] [PubMed] [Google Scholar]
- 26. Piontkivska H, Rooney AP, Nei M (2002) Purifying selection and birth-and-death evolution in the histone H4 gene family. Mol Biol Evol 19: 689–697. [DOI] [PubMed] [Google Scholar]
- 27. Ota T, Nei M (1995) Evolution of immunoglobulin VH pseudogenes in chickens. Mol Biol Evol 12: 94–102. [DOI] [PubMed] [Google Scholar]
- 28. Ota T, Nei M (1994) Divergent evolution and evolution by the birth-and-death process in the immunoglobulin VH gene family. Mol Biol Evol 11: 469–482. [DOI] [PubMed] [Google Scholar]
- 29. Niimura Y, Nei M (2005) Evolutionary changes of the number of olfactory receptor genes in the human and mouse lineages. Gene 346: 23–28. [DOI] [PubMed] [Google Scholar]
- 30. Niimura Y, Nei M (2003) Evolution of olfactory receptor genes in the human genome. Proc Natl Acad Sci U S A 100: 12235–12240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Eirin-Lopez JM, Gonzalez-Tizon AM, Martinez A, Mendez J (2004) Birth-and-death evolution with strong purifying selection in the histone H1 multigene family and the origin of orphon H1 genes. Mol Biol Evol 21: 1992–2003. [DOI] [PubMed] [Google Scholar]
- 32. Robertson HM, Gadau J, Wanner KW (2010) The insect chemoreceptor superfamily of the parasitoid jewel wasp Nasonia vitripennis . Insect Mol Biol 19 Suppl 1: 121–136. [DOI] [PubMed] [Google Scholar]
- 33. Robertson HM, Wanner KW (2006) The chemoreceptor superfamily in the honey bee, Apis mellifera: expansion of the odorant, but not gustatory, receptor family. Genome Res 16: 1395–1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Nei M, Gu X, Sitnikova T (1997) Evolution by the birth-and-death process in multigene families of the vertebrate immune system. Proc Natl Acad Sci U S A 94: 7799–7806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Nei M, Rooney AP (2005) Concerted and birth-and-death evolution of multigene families. Annu Rev Genet 39: 121–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Beye M, Seelmann C, Gempe T, Hasselmann M, Vekemans X, et al. (2013) Gradual molecular evolution of a sex determination switch through incomplete penetrance of femaleness. Curr Biol 23: 2559–2564. [DOI] [PubMed] [Google Scholar]
- 37. Graves JA (2006) Sex chromosome specialization and degeneration in mammals. Cell 124: 901–914. [DOI] [PubMed] [Google Scholar]
- 38. Charlesworth B, Charlesworth D (1978) A model for the evolution of dioecy and gynodioecy. Am Nat 112: 975–997. [Google Scholar]
- 39. Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95: 118–128. [DOI] [PubMed] [Google Scholar]
- 40. Marshall Graves JA (2008) Weird animal genomes and the evolution of vertebrate sex and sex chromosomes. Annu Rev Genet 42:565–86 565–586. [DOI] [PubMed] [Google Scholar]
- 41. Mank JE, Avise JC (2009) Evolutionary diversity and turn-over of sex determination in teleost fishes. Sex Dev 3: 60–67. [DOI] [PubMed] [Google Scholar]
- 42.Bull JJ (1983) Evolution of Sex Determining Mechanisms. Menlo Park,Calif.: Benjamin/Cummings Publishing Company. [Google Scholar]
- 43. Sanchez L (2008) Sex-determining mechanisms in insects. Int J Dev Biol 52: 837–856. [DOI] [PubMed] [Google Scholar]
- 44. Schutt C, Nothiger R (2000) Structure, function and evolution of sex-determining systems in Dipteran insects. Development 127: 667–677. [DOI] [PubMed] [Google Scholar]
- 45. Marin I, Baker BS (1998) The evolutionary dynamics of sex determination. Science 281: 1990–1994. [DOI] [PubMed] [Google Scholar]
- 46. Tamura K, Peterson D, Peterson N, Stecher G, Nei M, et al. (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28: 2731–2739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8: 275–282. [DOI] [PubMed] [Google Scholar]
- 48. Tajima F (1993) Simple methods for testing the molecular evolutionary clock hypothesis. Genetics 135: 599–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Martin D, Rybicki E (2000) RDP: detection of recombination amongst aligned sequences. Bioinformatics 16: 562–563. [DOI] [PubMed] [Google Scholar]
- 50. Padidam M, Sawyer S, Fauquet CM (1999) Possible emergence of new geminiviruses by frequent recombination. Virology 265: 218–225. [DOI] [PubMed] [Google Scholar]
- 51. Martin DP, Posada D, Crandall KA, Williamson C (2005) A modified bootscan algorithm for automated identification of recombinant sequences and recombination breakpoints. AIDS Res Hum Retroviruses 21: 98–102. [DOI] [PubMed] [Google Scholar]
- 52. Smith JM (1992) Analyzing the mosaic structure of genes. J Mol Evol 34: 126–129. [DOI] [PubMed] [Google Scholar]
- 53. Posada D, Crandall KA (2001) Evaluation of methods for detecting recombination from DNA sequences: computer simulations. Proc Natl Acad Sci U S A 98: 13757–13762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Gibbs MJ, Armstrong JS, Gibbs AJ (2000) Sister-scanning: a Monte Carlo procedure for assessing signals in recombinant sequences. Bioinformatics 16: 573–582. [DOI] [PubMed] [Google Scholar]
- 55. Boni MF, Posada D, Feldman MW (2007) An exact nonparametric method for inferring mosaic structure in sequence triplets. Genetics 176: 1035–1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Gene tree of the fem and fem1/csd sister copies in ants and bees, which were inferred from synonymous differences. The evolutionary history was inferred using the neighbor-joining method. The confidence probability (multiplied by 100) that the interior branch length is greater than 0 was estimated using the bootstrap test (10000 replicates are shown next to the branches). The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Pamilo-Bianchi-Li method [1] and are in the units of the number of synonymous substitutions per synonymous site. All ambiguous positions were removed for each sequence pair. There were a total of 575 positions in the final dataset. Evolutionary analyses were conducted in MEGA 5 [2]. The sequences of Nasonia and Ceratitis were excludeto estimate dS. Abbreviations: Acep, Atta cephalotes; Acer, Apis cerana; Aech, Acromyrmex echinatior; Ador, Apis dorsata; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris; Cflo, Camponotus floridanus; Hsal, Harpegnathos saltator; Mcom, Melipona compressipes; Pbar, Pogonomyrmex barbatus; Sinv, Solenopsis invicta.
(DOCX)
The initial tree of the Fem (a) and Fem1/Csd (b) proteins that were used to infer the ancestral amino acid sequences. The evolutionary history was inferred by using the maximum likelihood method based on the JFF model [1]. Numbers in the tree assign the different nodes for which the ancestral sequence were obtained. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. Abbreviations: Acep, Atta cephalotes; Acer, Apis cerana; Aech, Acromyrmex echinatior; Ador, Apis dorsata; Aflo, Apis florea; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris; Ccap, Ceratitis capitata; Cflo, Camponotus floridanus; Hsal, Harpegnathos saltator; Mcom, Melipona compressipes; Nvit, Nasonia vitripennis; Pbar, Pogonomyrmex barbatus; Sinv, Solenopsis invicta.
(DOCX)
The informative substitutions found in the ant lineage that were used in Figure 2 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicates the positions in the Fem and in the Csd/Fem1 protein sequence alignment.
(DOCX)
The informative substitutions found in the bee lineage that were used in Figure 2 . The identity of the different species and nodes of the Fem and Csd/Fem1 protein tree is shown. Site number (#) indicates the positions in the Fem and in the Csd/Fem1 protein sequence alignment.
(DOCX)
The informative substitutions found in the Bombus lineage (Fem tree) that were used in Figure 3 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicates the position in the alignment of the Fem and of the Csd/Fem1 protein sequences.
(DOCX)
The informative substitutions found in the Bombus lineage (Fem1 tree) that were used in Figure 3 . The identity of the different species and nodes of the Fem and Fem1 protein tree is shown. Site number (#) indicate the position in the alignment of the Fem and of the Csd/Fem1 protein sequences.
(DOCX)
The number of falsely detected events (FDE) using the methods as indicated on the X axis. These programs were implemented in the RDP 3.44 software program. The methods were run on each of the 20 alignments which consisted of two randomly chosen csd sequences from a single Apis species and one fem sequence from B. terrestris. These events are falsely detected as this transfer involve the outgroup Bombus sequence and the polymorphism between csd alleles which newly evolved in the different Apis species.
(DOCX)
The dS i values for the paralogous gene pairs fem and csd/fem1 within each species (Ador, Apis dorsata; Amel, Apis mellifera; Bimp, Bombus impatiens; Bter, Bombus terrestris).
(DOCX)