Abstract
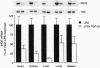
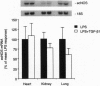
Septic shock is a cytokine-mediated process typically caused by a severe underlying infection. Toxins generated by the infecting organism trigger a cascade of events leading to hypotension, to multiple organ system failure, and frequently to death. Beyond supportive care, no effective therapy is available for the treatment of septic shock. Nitric oxide (NO) is a potent vasodilator generated late in the sepsis pathway leading to hypotension; therefore, NO represents a potential target for therapy. We have previously demonstrated that transforming growth factor (TGF) beta1 inhibits inducible NO synthase (iNOS) mRNA and NO production in vascular smooth muscle cells after its induction by cytokines critical in the sepsis cascade. Thus, we hypothesized that TGF-beta1 may inhibit iNOS gene expression in vivo and be beneficial in the treatment of septic shock. In a conscious rat model of septic shock produced by Salmonella typhosa lipopolysaccharide (LPS), TGF-beta1 markedly reduced iNOS mRNA and protein levels in several organs. In contrast, TGF-beta1 did not decrease endothelium-derived constitutive NOS mRNA in organs of rats receiving LPS. We also performed studies in anesthetized rats to evaluate the effect of TGF-beta1 on the hemodynamic compromise of septic shock; after an initial 25% decrease in mean arterial pressure, TGF-beta1 arrested LPS-induced hypotension and decreased mortality. A decrease in iNOS mRNA and protein levels in vascular smooth muscle cells was demonstrated by in situ hybridization and NADPH diaphorase staining in rats treated with TGF-beta1. Thus these studies suggest that TGF-beta1 inhibits iNOS in vivo and that TGF-beta1 may be of future benefit in the therapy of septic shock.
Full text
PDF

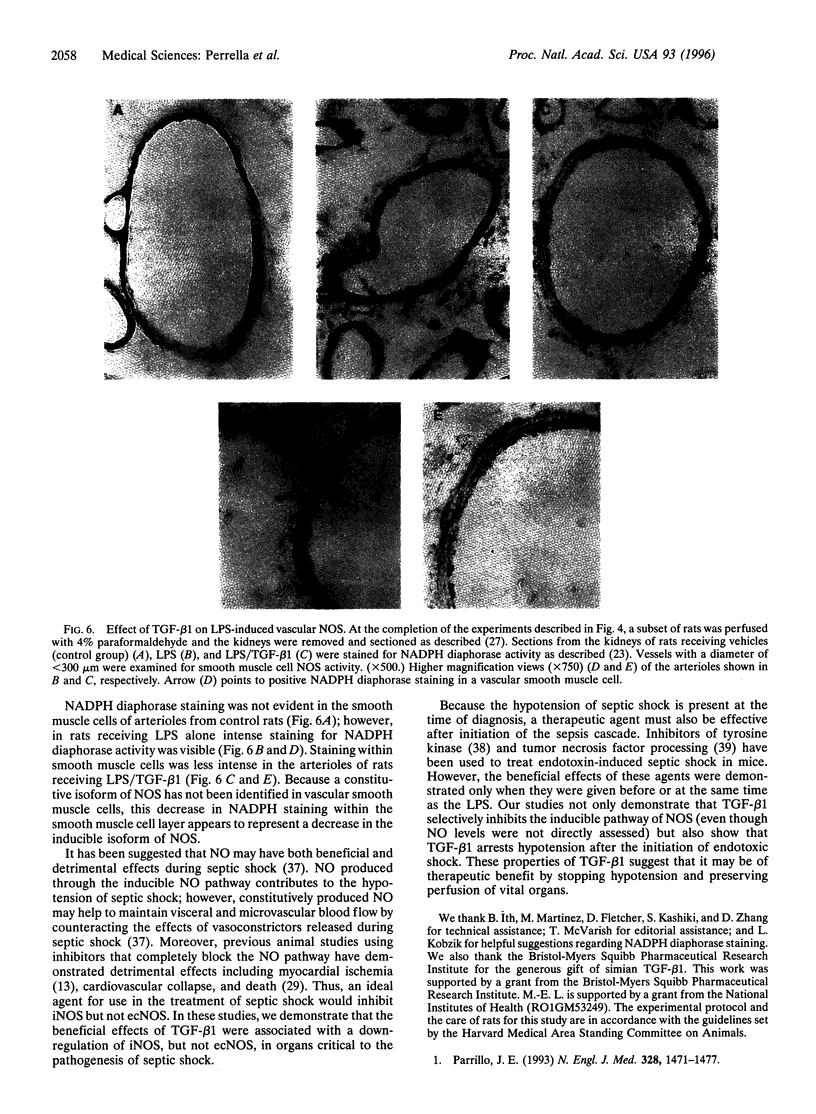

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avontuur J. A., Bruining H. A., Ince C. Inhibition of nitric oxide synthesis causes myocardial ischemia in endotoxemic rats. Circ Res. 1995 Mar;76(3):418–425. doi: 10.1161/01.res.76.3.418. [DOI] [PubMed] [Google Scholar]
- Chantry D., Turner M., Abney E., Feldmann M. Modulation of cytokine production by transforming growth factor-beta. J Immunol. 1989 Jun 15;142(12):4295–4300. [PubMed] [Google Scholar]
- Coffey R. J., Jr, Kost L. J., Lyons R. M., Moses H. L., LaRusso N. F. Hepatic processing of transforming growth factor beta in the rat. Uptake, metabolism, and biliary excretion. J Clin Invest. 1987 Sep;80(3):750–757. doi: 10.1172/JCI113130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corbett J. A., Tilton R. G., Chang K., Hasan K. S., Ido Y., Wang J. L., Sweetland M. A., Lancaster J. R., Jr, Williamson J. R., McDaniel M. L. Aminoguanidine, a novel inhibitor of nitric oxide formation, prevents diabetic vascular dysfunction. Diabetes. 1992 Apr;41(4):552–556. doi: 10.2337/diab.41.4.552. [DOI] [PubMed] [Google Scholar]
- Dawson T. M., Bredt D. S., Fotuhi M., Hwang P. M., Snyder S. H. Nitric oxide synthase and neuronal NADPH diaphorase are identical in brain and peripheral tissues. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7797–7801. doi: 10.1073/pnas.88.17.7797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas J. A., Khraibi A. A., Perrella M. A., Knox F. G. Role of renal interstitial hydrostatic pressure in natriuresis of systemic nitric oxide inhibition. Am J Physiol. 1993 Mar;264(3 Pt 2):F411–F414. doi: 10.1152/ajprenal.1993.264.3.F411. [DOI] [PubMed] [Google Scholar]
- Inoue N., Venema R. C., Sayegh H. S., Ohara Y., Murphy T. J., Harrison D. G. Molecular regulation of the bovine endothelial cell nitric oxide synthase by transforming growth factor-beta 1. Arterioscler Thromb Vasc Biol. 1995 Aug;15(8):1255–1261. doi: 10.1161/01.atv.15.8.1255. [DOI] [PubMed] [Google Scholar]
- Keaney J. F., Jr, Puyana J. C., Francis S., Loscalzo J. F., Stamler J. S., Loscalzo J. Methylene blue reverses endotoxin-induced hypotension. Circ Res. 1994 Jun;74(6):1121–1125. doi: 10.1161/01.res.74.6.1121. [DOI] [PubMed] [Google Scholar]
- Kilbourn R. G., Gross S. S., Jubran A., Adams J., Griffith O. W., Levi R., Lodato R. F. NG-methyl-L-arginine inhibits tumor necrosis factor-induced hypotension: implications for the involvement of nitric oxide. Proc Natl Acad Sci U S A. 1990 May;87(9):3629–3632. doi: 10.1073/pnas.87.9.3629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobzik L., Bredt D. S., Lowenstein C. J., Drazen J., Gaston B., Sugarbaker D., Stamler J. S. Nitric oxide synthase in human and rat lung: immunocytochemical and histochemical localization. Am J Respir Cell Mol Biol. 1993 Oct;9(4):371–377. doi: 10.1165/ajrcmb/9.4.371. [DOI] [PubMed] [Google Scholar]
- Lee W. S., Berry M. J., Hediger M. A., Larsen P. R. The type I iodothyronine 5'-deiodinase messenger ribonucleic acid is localized to the S3 segment of the rat kidney proximal tubule. Endocrinology. 1993 May;132(5):2136–2140. doi: 10.1210/endo.132.5.8477661. [DOI] [PubMed] [Google Scholar]
- Lee W. S., Kanai Y., Wells R. G., Hediger M. A. The high affinity Na+/glucose cotransporter. Re-evaluation of function and distribution of expression. J Biol Chem. 1994 Apr 22;269(16):12032–12039. [PubMed] [Google Scholar]
- Long N. C., Kunkel S. L., Vander A. J., Kluger M. J. Antiserum against tumor necrosis factor enhances lipopolysaccharide fever in rats. Am J Physiol. 1990 Feb;258(2 Pt 2):R332–R337. doi: 10.1152/ajpregu.1990.258.2.R332. [DOI] [PubMed] [Google Scholar]
- Lowenstein C. J., Glatt C. S., Bredt D. S., Snyder S. H. Cloned and expressed macrophage nitric oxide synthase contrasts with the brain enzyme. Proc Natl Acad Sci U S A. 1992 Aug 1;89(15):6711–6715. doi: 10.1073/pnas.89.15.6711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacMicking J. D., Nathan C., Hom G., Chartrain N., Fletcher D. S., Trumbauer M., Stevens K., Xie Q. W., Sokol K., Hutchinson N. Altered responses to bacterial infection and endotoxic shock in mice lacking inducible nitric oxide synthase. Cell. 1995 May 19;81(4):641–650. doi: 10.1016/0092-8674(95)90085-3. [DOI] [PubMed] [Google Scholar]
- Massagué J. The transforming growth factor-beta family. Annu Rev Cell Biol. 1990;6:597–641. doi: 10.1146/annurev.cb.06.110190.003121. [DOI] [PubMed] [Google Scholar]
- Misko T. P., Moore W. M., Kasten T. P., Nickols G. A., Corbett J. A., Tilton R. G., McDaniel M. L., Williamson J. R., Currie M. G. Selective inhibition of the inducible nitric oxide synthase by aminoguanidine. Eur J Pharmacol. 1993 Mar 16;233(1):119–125. doi: 10.1016/0014-2999(93)90357-n. [DOI] [PubMed] [Google Scholar]
- Mohler K. M., Sleath P. R., Fitzner J. N., Cerretti D. P., Alderson M., Kerwar S. S., Torrance D. S., Otten-Evans C., Greenstreet T., Weerawarna K. Protection against a lethal dose of endotoxin by an inhibitor of tumour necrosis factor processing. Nature. 1994 Jul 21;370(6486):218–220. doi: 10.1038/370218a0. [DOI] [PubMed] [Google Scholar]
- Moncada S., Palmer R. M., Higgs E. A. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991 Jun;43(2):109–142. [PubMed] [Google Scholar]
- Natanson C., Hoffman W. D., Suffredini A. F., Eichacker P. Q., Danner R. L. Selected treatment strategies for septic shock based on proposed mechanisms of pathogenesis. Ann Intern Med. 1994 May 1;120(9):771–783. doi: 10.7326/0003-4819-120-9-199405010-00009. [DOI] [PubMed] [Google Scholar]
- Nathan C., Xie Q. W. Nitric oxide synthases: roles, tolls, and controls. Cell. 1994 Sep 23;78(6):915–918. doi: 10.1016/0092-8674(94)90266-6. [DOI] [PubMed] [Google Scholar]
- Nava E., Palmer R. M., Moncada S. Inhibition of nitric oxide synthesis in septic shock: how much is beneficial? Lancet. 1991 Dec 21;338(8782-8783):1555–1557. doi: 10.1016/0140-6736(91)92375-c. [DOI] [PubMed] [Google Scholar]
- Nava E., Palmer R. M., Moncada S. The role of nitric oxide in endotoxic shock: effects of NG-monomethyl-L-arginine. J Cardiovasc Pharmacol. 1992;20 (Suppl 12):S132–S134. doi: 10.1097/00005344-199204002-00037. [DOI] [PubMed] [Google Scholar]
- Novogrodsky A., Vanichkin A., Patya M., Gazit A., Osherov N., Levitzki A. Prevention of lipopolysaccharide-induced lethal toxicity by tyrosine kinase inhibitors. Science. 1994 May 27;264(5163):1319–1322. doi: 10.1126/science.8191285. [DOI] [PubMed] [Google Scholar]
- Ochoa J. B., Udekwu A. O., Billiar T. R., Curran R. D., Cerra F. B., Simmons R. L., Peitzman A. B. Nitrogen oxide levels in patients after trauma and during sepsis. Ann Surg. 1991 Nov;214(5):621–626. doi: 10.1097/00000658-199111000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parrillo J. E. Pathogenetic mechanisms of septic shock. N Engl J Med. 1993 May 20;328(20):1471–1477. doi: 10.1056/NEJM199305203282008. [DOI] [PubMed] [Google Scholar]
- Perrella M. A., Yoshizumi M., Fen Z., Tsai J. C., Hsieh C. M., Kourembanas S., Lee M. E. Transforming growth factor-beta 1, but not dexamethasone, down-regulates nitric-oxide synthase mRNA after its induction by interleukin-1 beta in rat smooth muscle cells. J Biol Chem. 1994 May 20;269(20):14595–14600. [PubMed] [Google Scholar]
- Petros A., Bennett D., Vallance P. Effect of nitric oxide synthase inhibitors on hypotension in patients with septic shock. Lancet. 1991 Dec 21;338(8782-8783):1557–1558. doi: 10.1016/0140-6736(91)92376-d. [DOI] [PubMed] [Google Scholar]
- Rédini F., Mauviel A., Pronost S., Loyau G., Pujol J. P. Transforming growth factor beta exerts opposite effects from interleukin-1 beta on cultured rabbit articular chondrocytes through reduction of interleukin-1 receptor expression. Arthritis Rheum. 1993 Jan;36(1):44–50. doi: 10.1002/art.1780360108. [DOI] [PubMed] [Google Scholar]
- Sporn M. B., Roberts A. B. Transforming growth factor-beta: recent progress and new challenges. J Cell Biol. 1992 Dec;119(5):1017–1021. doi: 10.1083/jcb.119.5.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamler J. S., Singel D. J., Loscalzo J. Biochemistry of nitric oxide and its redox-activated forms. Science. 1992 Dec 18;258(5090):1898–1902. doi: 10.1126/science.1281928. [DOI] [PubMed] [Google Scholar]
- Stone R. Search for sepsis drugs goes on despite past failures. Science. 1994 Apr 15;264(5157):365–367. doi: 10.1126/science.8153620. [DOI] [PubMed] [Google Scholar]
- Stuehr D. J., Cho H. J., Kwon N. S., Weise M. F., Nathan C. F. Purification and characterization of the cytokine-induced macrophage nitric oxide synthase: an FAD- and FMN-containing flavoprotein. Proc Natl Acad Sci U S A. 1991 Sep 1;88(17):7773–7777. doi: 10.1073/pnas.88.17.7773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabó C., Southan G. J., Thiemermann C. Beneficial effects and improved survival in rodent models of septic shock with S-methylisothiourea sulfate, a potent and selective inhibitor of inducible nitric oxide synthase. Proc Natl Acad Sci U S A. 1994 Dec 20;91(26):12472–12476. doi: 10.1073/pnas.91.26.12472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vodovotz Y., Bogdan C., Paik J., Xie Q. W., Nathan C. Mechanisms of suppression of macrophage nitric oxide release by transforming growth factor beta. J Exp Med. 1993 Aug 1;178(2):605–613. doi: 10.1084/jem.178.2.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wahl S. M., Costa G. L., Corcoran M., Wahl L. M., Berger A. E. Transforming growth factor-beta mediates IL-1-dependent induction of IL-1 receptor antagonist. J Immunol. 1993 Apr 15;150(8 Pt 1):3553–3560. [PubMed] [Google Scholar]
- Wakefield L. M., Winokur T. S., Hollands R. S., Christopherson K., Levinson A. D., Sporn M. B. Recombinant latent transforming growth factor beta 1 has a longer plasma half-life in rats than active transforming growth factor beta 1, and a different tissue distribution. J Clin Invest. 1990 Dec;86(6):1976–1984. doi: 10.1172/JCI114932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei X. Q., Charles I. G., Smith A., Ure J., Feng G. J., Huang F. P., Xu D., Muller W., Moncada S., Liew F. Y. Altered immune responses in mice lacking inducible nitric oxide synthase. Nature. 1995 Jun 1;375(6530):408–411. doi: 10.1038/375408a0. [DOI] [PubMed] [Google Scholar]