Abstract
B cells are required for follicular helper T (Tfh) cell development, as is the ligand for ICOS (ICOS-L); however, the separable contributions of Ag and ICOS-L delivery by cognate B cells to Tfh-cell development and function are unknown. We find that Tfh-cell and germinal center differentiation are dependent upon cognate B-cell display of ICOS-L, but only when Ag presentation by the latter is limiting, with the requirement for B-cell expression of ICOS-L overcome by robust Ag delivery. These findings demonstrate that Ag-specific B cells provide different, yet compensatory signals for Tfh-cell differentiation, while reconciling conflicting data indicating a requirement for ICOS-L expression on cognate B cells for Tfh-cell development with those demonstrating this requirement could be bypassed in lieu of that tendered by non-cognate B cells. Our findings clarify the separable roles of delivery of Ag and ICOS-L by cognate B cells for Tfh-cell maturation and function, and have implications for using therapeutic ICOS blockade in settings of abundantly available Ag, such as in systemic autoimmunity.
Keywords: T cells, Costimulation, Autoimmunity
Introduction
Germinal centers (GCs)1 develop within B cell follicles of secondary lymphoid organs (SLOs) in response to immunization with Ag. Follicular helper T (Tfh) cells, a subset of CD4 Th cells that migrate into GCs, are necessary for B cell maturation into memory and long-lived plasma cells (1). Tfh cells provide signals to cognate B cells via CD40 ligand, programmed cell death 1 (PD-1), and IL-21, among other stimuli, promoting B cell proliferation and affinity maturation within the GC (2-6). Tfh cells also secrete IL-4 and IFN-γ, promoting B cell isotype switching appropriate to the invading pathogen (3, 7-9).
The transcription factor B-cell lymphoma 6 (Bcl6) is both necessary and sufficient for the development of Tfh cells leading to expression of transcripts important for their migration and function, while repressing activation of genes critical for development of other T helper subsets (10-12). Initial upregulation of Bcl6 in T cells occurs within the T cell zone of SLOs (13-18) upon activation by dendritic cells (DCs) expressing ICOS ligand (ICOS-L, B7-H2, CD275) signaling via ICOS on nascent Tfh cells (13). Bcl6 protein expression can be detected in T cells as early as the first cell division, indicating that Tfh-cell developmental potential starts concomitantly with T cell activation (17), with polarization between Bcl6+ Tfh cells and Blimp1+ (Prdm1; positive regulatory domain I-binding factor 1) effector T cell populations developing within the first 72 hours after viral infection (13). Following DC-driven activation, Bcl6-expressing nascent Tfh cells downregulate CCR7 and P-selectin glycoprotein ligand 1 (PSGL-1), facilitating their release from the chemokine ligands CCL19 and CCL21 and allowing for emigration out of the T cell zone (19-23) to sites of initial B cell contact in the interfollicular regions of the lymph node (15) and the T-B border of the spleen (24). Bcl6 upregulation also promotes expression of the chemokine receptor CXCR5, with the latter necessary for Tfh-cell entry into the B cell follicle following a gradient of its ligand CXCL13 (21, 25).
Upon contact with B cells, developing Tfh cells receive additional signal(s) that are critical for their continued maturation, including maintenance of Bcl6 expression and progression to functional B cell helpers. For example, SLAM-associated protein (SAP, encoded by Sh2d1a), an intracellular adapter molecule that is important for signaling between SLAM family receptors on B and T lymphocytes, is necessary for formation of stable T-B cell conjugates, with proper Tfh-cell development, GC formation, and T-dependent Ab responses (8, 26, 27).
ICOS-L expressed on activated B cells, in addition to its provision by DCs in the T cell zone, is critical for Tfh-cell development and maintenance. Mice deficient in ICOS fail to develop GCs (28-31) and have decreased numbers of Tfh cells (32, 33), and blocking the function of ICOS-L on B cells by Abs or via conditional genetic deletion abrogates their formation (13, 32, 34). Conversely, signaling via ICOS promotes expansion of this population (35), with its overexpression leading to an increase in Tfh cells and spontaneous GC development (36). ICOS signaling delivers a strong phosphatidylinositol 3-kinase (PI3K) signal that is critical for the induction of their key cytokines, IL-21 and IL-4 (37), with ICOS-driven IL-21 production mediated by the transcription factor c-Maf (38).
These data suggest a model of Tfh-cell development initiated by DC provision of Ag and ICOS-L in T cell zones leading to Bcl6 expression and induction of a Tfh-cell gene activation program, with the latter solidified by subsequent interaction with ICOS-L-bearing cognate B cells in interfollicular regions of lymph nodes or at the splenic T-B cell border. Yet, other data suggest complexities to this relatively straightforward model, with the demonstration that the role of B cells in Tfh-cell development is not necessarily dependent upon delivery of unique B-cell signal, such as ICOS-L; rather, the former are positioned to provide an abundant source of Ag (39). More recent data have revealed a novel role for ICOS-L in Tfh-cell development, with the finding that its expression on non-cognate follicular B cells with signaling via ICOS to PI3K is necessary for optimal positioning of Ag-specific T cells for interaction with cognate B cells and subsequent Tfh cell and GC development (40); this study also indicated that the necessity of ICOS-L on B cells for Tfh cell migration is bypassed upon delivery of Ag by cognate B cells. Nonetheless, as outlined above, the latter are required for functional Tfh cells with GC formation, and these experiments and others to date have not separated the independent roles, if any, of B-cell delivery of Ag and ICOS-L by cognate B cells in antigen-specific Tfh-cell development, migration, and function. Here, we provide a clarification of these roles, demonstrating that while cognate provision of Ag and ICOS-L by B cells are necessary for Tfh-cell maturation and follicular migration with GC formation and generation of Ab, the latter signal is only operative in settings of limited Ag delivery by cognate B cells. These data clarify the separable roles of delivery of Ag and ICOS-L by cognate B cells for Tfh-cell differentiation and subsequent function, and have implications for the role of therapeutic ICOS blockade in the setting of abundantly available Ag, such as in systemic autoimmune diseases.
Materials and Methods
Mice
Mice were housed in pathogen-free conditions at the Yale School of Medicine (New Haven, CT). C57BL/6J (B6), OT-II TCR transgenic (C57BL/6-Tg [TcraTcrb]425Cbn/J), CD19-deficient (CD19−/−, B6.129P2(C)-Cd19tm1(cre)Cgn/J), Ig anti-hen egg lysozyme transgenic MD4, and ICOS-L−/− (B6.129P2-Icosltm1Mak/J) mice were purchased from The Jackson Laboratory. B1-8 mice were a gift from Klaus Rajewsky (41), with B1-8 animals deficient in ICOS-L−/− bred in our colony. Animals were used at 6-8 weeks of age, with approval for procedures given by The Institutional Animal Care and Use Committee of Yale University.
Cell transfers and immunizations
For cell transfers, a total of 0.5 × 106 CD4 OT-II TCR transgenic T cells along with 1 × 106 B1-8 Ig transgenic B cells were delivered to recipient mice via retro-orbital injection unless otherwise stated. Mice were immunized with 100ug of (4-hydroxy-3-nitrophenyl) acetyl (NP)15-OVA (Biosearch Technologies) precipitated in aluminum hydroxide (alum) i.p. 24 hours post-transfer of cells. Seven days later, animals were sacrificed and spleens harvested, with division into portions for microscopy and flow cytometric analysis.
Flow cytometry and cell sorting
Tissues were homogenized by crushing between two frosted glass slides followed by straining through a 40uM nylon filter. Red cell lysis was followed by hypotonic disruption as described previously (22) and remaining cells were counted. Abs used for flow cytometric staining included CD4 (clone RM4-5), CD44 (clone IM7), PD-1 (clone J43), IgD (clone 11-26), IL-4 (clone 8D4-8) (all from eBioscience), B220 (clone RA3-6B2), Thy1.1 (clone OX-7), CXCR5 (2G8), CD62L (clone MEL-14), GL-7 (clone GL7), FITC CD95 (clone Jo2), PE-Cy7 (all from BD Biosciences) and IL-21R-FC (R&D). Anti-PSGL-1 (BD Biosciences) was directly conjugated to Alexa-647 as described previously (23). Staining for CXCR5 was performed at room temperature (25°C) with 30 min incubation. Intracellular staining for cytokines was performed using BD Cytofix/Cytoperm™ kits following the manufacturer's protocol. Stained and rinsed cells were analyzed using an LSRII Multilaser Cytometer (BD Biosciences). For certain experiments (Fig. 4), CD4 T cells were enriched using a biotin-based magnetic separation kit (EasySep™, StemCell Technologies) prior to cell surface staining, with specific populations sorted using a FACSAria™ (BD Bioscience). Biotin-based magnetic separation kits (EasySep™) were used to isolate T and B cells for cell transfer studies.
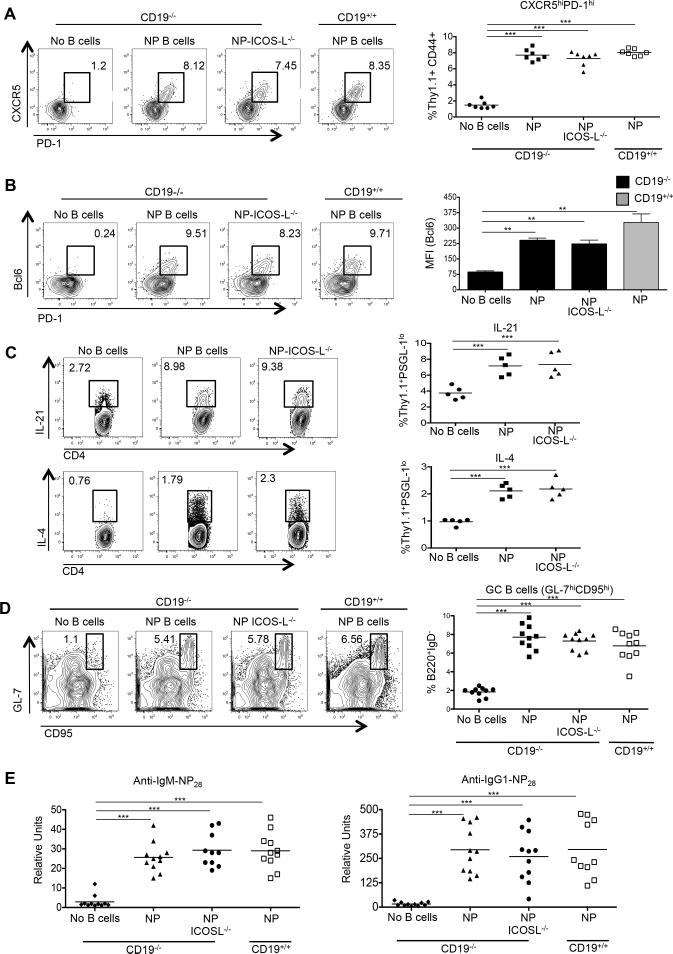
Figure 4.
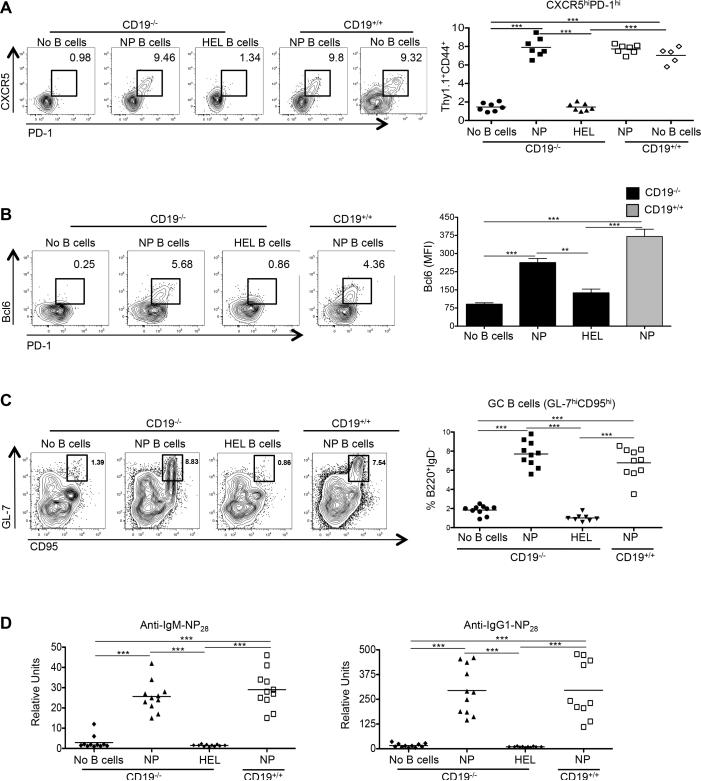
Ag-specific B cells are required for development of functional Tfh cells. CD19−/− or CD19+/+ mice received CD4 Thy1.1+ OT-II cells alone (n ≥ 5), or with NP-specific (n = 7) or HEL-specific MD4 B cells (n = 7), with spleens of recipients harvested 7 days after immunization with NP-OVA and sera collected. (A) Representative flow cytometry plots of CD4+ Thy1.1+ CD44+ CXCR5hi PD-1hi T cells from the recipients, with the graph on the right showing the percentages of such cells among the transferred population. (B) Representative Bcl6 and PD-1 expression in the transferred CD4+ T cells, including cells transferred without B cells (left 4 panels), in conjunction with Bcl6 expression (right panel). (C) Representative flow cytometry plots of B220+ IgD− GL-7hi CD95hi GC B cells taken from the recipients, with the percentages of such cells among B220+ IgD− cells shown on the graph on the right. (D) IgM (left) and IgG (right) anti-NP28 Ab levels in the recipients. Experiments were performed 3 times with n ≥ 3 per group. *** p < 0.001; ** p < 0.003 by Student's t-test comparing cells transferred into CD19−/− or CD19+/+ mice. Error bars represent standard deviation.
Microscopy
Spleens were snap frozen in OCT tissue-freezing solution and stored at −80°C. Tissues were cut into 6um sections and processed as described previously (22). Sections were stained with GFP FITC (Rockland Immunochemicals), CD4 (clone RM4-5) FITC, IgD (clone 11-26) Alexa-647 (both from eBiosciences), PNA biotin (Vector Laboratories), and rabbit IgG anti-FITC 488 and Alexa-555 (both from Invitrogen). Images were obtained from a laser-scanning confocal microscope (model 510 META; Carl Zeiss, Inc.) at 25x magnification. ImageJ software from the National Institutes of Health was used for the measurement of GC and B cell follicle size as well as for T cell counting.
Quantitative PCR
Sorted cell populations were processed for RNA isolation and conversion into cDNA as described previously (23). An MX4005P Thermal Cycler™ and Brilliant SYBER Green Master Mix™ (both from Stratagene) were used for quantitative PCR using the following primers: Bcl6, 5’-CACACTCGAATTCACTCTG-3’ (forward) and 5’-TATTGCACCTTGGTGTTGG-3’ (reverse); Hprt (Superarray, Qiagen). Bcl6 expression was normalized to the Hprt control.
ELISA
For assessment of anti-NP Abs, sera were collected by cardiac puncture 7-14 days following immunization of mice with NP15-OVA in alum. Plates were coated with NP6-CGG or NP28-CGG (Biosearch Technologies) and anti-NP IgM and IgG Abs were detected using HRP-conjugated goat and anti-mouse IgM or IgG1 Abs (Southern Biotechnology Associates). Standard curves were created using sera from B6 mice immunized with NP-OVA15 and used to convert OD values into units using Prism4® (GraphPad Software).
Statistics
Data were analyzed using the Student's t-test with Prism4®. The number of asterisks represents the degree of significance with respect to p value, with the exact value presented within each figure legend.
Results
Expansion of Tfh cells following immunization is B cell-dependent
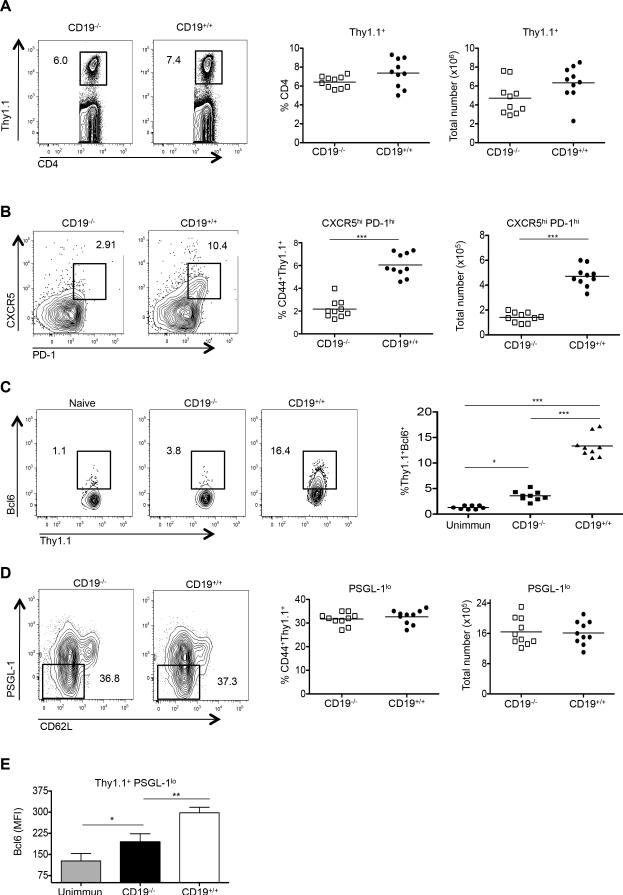
Tfh cells fail to develop in RAG- or B cell-deficient μMT mice (23, 32, 42); however, the absence of mature B cells in the periphery of these animals disrupts secondary lymphoid architecture and hinders CD4 T cell localization (43). To examine Tfh-cell generation in the absence of B-cell help in anatomically intact mice, we used as recipients of adoptive transfers CD19-deficient (CD19−/−) animals (42). While CD19 is crucial for B-cell activation by T-dependent Ags, it is not required for B cell development and normal splenic architecture (44, 45). We adoptively transferred congenically mismatched Thy1.1+ OT-II OVA-specific TCR transgenic CD4 T cells into CD19−/− or, as controls, wild type (WT) CD19-intact (CD19+/+) B6 recipients followed by i.p. challenge with NP-OVA in alum and analysis seven days later. Ag-specific Thy1.1+ CD4+ cells transferred into CD19−/− and WT CD19+/+ mice expanded equivalently (Fig. 1A); however, T cells transferred into the CD19−/− group failed to upregulate the Tfh-cell markers CXCR5 and PD-1 (Fig. 1B), and had greatly diminished expression of Bcl6 protein and Bcl6 mRNA compared to T cells transferred into intact recipients, albeit with amounts higher than in unimmunized controls (Fig. 1C, and data not shown). T cell expansion and residual Bcl6 mRNA and Bcl6 protein upregulation following transfer to CD19-deficient mice were presumably secondary to Ag-specific signals delivered by DCs (13-15, 17, 23, 46). Downregulation of the T zone retention ligand PSGL-1 occurred on T cells transferred into both CD19−/− and WT recipients (Fig. 1D), with the transferred cells that became PSGL-1lo in both groups expressing more Bcl6 than cells adoptively transferred to unimmunized controls (Fig. 1E; MFI 216 ± 28.94 versus MFI 140 ± 19.2, respectively). Thus, in the absence of CD19 signaling in B cells, the Tfh-cell developmental program is initiated by DCs with upregulation of Bcl6 mRNA and protein and downregulation of PSGL-1; however, CD19-bearing B cells are essential for upregulation of CXCR5 and PD-1 and for maximal induction of Bcl6 in antigen-specific Tfh cells.
Figure 1.
CD19-intact B cells are required for Tfh-cell development. CD19−/− (n = 10) or CD19+/+ (n = 10) mice received CD4+ Thy1.1+ OT-II TCR transgenic T cells, with spleens of recipients harvested 7 days after immunization with NP-OVA. (A) Representative flow cytometry plots of splenic cells showing the percentages of transferred Thy1.1 cells among total CD4 T cells (left 2 panels). The graphs (right 2 panels) show percentages and absolute numbers of CD4+ Thy1.1+ T cells following transfer to CD19−/− or CD19+/+ recipients. (B) Representative flow cytometry plots of splenic cells from CD19−/− or CD19+/+ recipients, as gated on CD4+ Thy1.1+CD44+ CXCR5hi PD-1hi T cells (left 2 panels). Graphs on the right show the percentages and total numbers of such cells. (C) Bcl6 expression in the transferred CD4+Thy1.1+ OT-II cells recovered from CD19−/−, CD19+/+, or unimmunized recipients (left 3 panels). Graph on the right shows the mean fluorescent intensity (MFI) of Bcl6 expression among the transferred CD4+ Thy1.1+ OT-II cells. (D) Representative PSGL-1 expression in the transferred CD4+ Thy1.1+ OT-II cells recovered from CD19−/− or CD19+/+ recipients (left 2 panels), with the percentages and numbers of these cells also shown (right 2 graphs). (E) Mean fluorescent intensity (MFI) of PSGL-1 expression among the transferred CD4+ Thy1.1+ OT-II cells, including into unimmunized recipients. All experiments were performed 3 times with n ≥ 5 mice per group. *** p < 0.001; ** p < 0.003; * p < 0.02 by Student's t-test comparing cells transferred into CD19−/− or CD19+/+ mice. Error bars represent standard deviation. Unimmun = unimmunized recipients.
Ag-specific B cells are necessary for antigen-specific Tfh-cell development and function
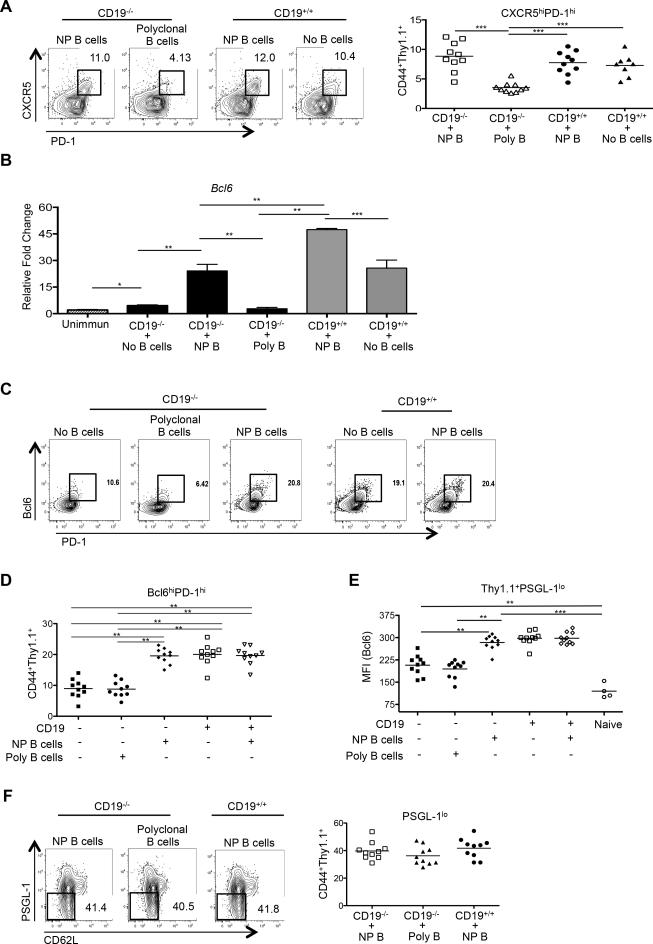
To dissect the factors that B cells use to foster Tfh-cell differentiation, we first examined the requirement for their Ag specificity. Here, we set up a dual T and B cell co-transfer system, transferring Thy1.1+ OT-II OVA-specific TCR transgenic CD4 T cells and either NP-specific B cells from B1-8 Ig transgenic mice (41) or polyclonal B cells from wild type B6 mice into CD19−/− or CD19+/+ hosts, followed by i.p. immunization of recipients with NP-OVA in alum. The B1-8 gene, when paired with endogenous Vλ1 chains, is specific for the hapten NP; approximately 2% of B cells in such animals bind NP (47). Tfh-cell development among the transferred Thy1.1+ OT-II TCR transgenic cells was assessed seven days later. Transfer of NP-specific B cells to CD19-deficient mice restored CXCR5 and PD-1 upregulation on transferred OVA-specific T cells to a degree equivalent to that observed with transfer into CD19+/+ mice (Fig. 2A; 9.6% versus 9.4% respectively). In contrast, co-transfer of polyclonal B cells failed to significantly induce CXCR5 and PD-1 expression on the transferred CD4 T cells (Fig. 2A), a result most likely due to the decreased precursor frequency of NP-specific B cells present in the 1 ×106 transferred polyclonal B cells (48). These data indicate that Ag-specific T-B interactions are essential for the upregulation of the Tfh-cell surface markers CXCR5 and PD-1.
Figure 2.
Ag-specific B cells are required for antigen-specific Tfh-cell development. CD19−/− or CD19+/+ mice received CD4+ Thy1.1+ OT-II cells and NP-specific (n =10), polyclonal B cells (n = 10), or no B cells (n = 10), with spleens of recipients harvested 7 days after immunization with NP-OVA. (A) Representative flow cytometry plots of splenic cells from CD19−/− or CD19+/+ recipients, as gated on CD4+ Thy1.1+ CD44+ CXCR5hi PD-1hi T cells (left 3 panels), with the graph on the right showing the percentages of such cells among the transferred populations. (B) cDNA was synthesized from sorted CD4+ Thy1.1+ OT-II T cells from CD19−/− or CD19+/+ mice that received co-transfers of NP-specific or polyclonal B and CD4+ Thy1.1+ OT-II T cells. qPCR for Bcl6 was compared to that of Hprt. cDNA from sorted naïve CD4+ Thy1.1+ OT-II T cells (CD4+CD44lo) served as a control. (C and D) Representative Bcl6 and PD-1 expression on the transferred CD4+ T cells, including those transferred without B cells, in conjunction with numbers of such cells. (E) Bcl6 MFI from transferred CD4+ Thy1.1+ OT-II PSGL-1lo T cell populations. (F) Representative flow cytometry plots of splenic cells from CD19−/− or CD19+/+ recipients, as gated on CD4+Thy1.1+CD44+ PSGL-1lo T cells (left 3 panels), with the graph on the right showing the percentages of such cells. Experiments were performed 3 times with n ≥ 3 per group. ***p < 0.001; **p < 0.003; * p < 0.02 by Student's t-test comparing cells transferred into CD19−/− or CD19+/+ mice. Error bars represent standard deviation. Poly = polyclonal B cells.
We next examined Bcl6 mRNA and its protein expression in activated OVA-specific T cells with co-transfer of Ag-specific B1-8 or polyclonal B cells. Expression of the mRNA was increased 5-fold in T cells transferred alongside NP-specific B cells relative to the groups receiving polyclonal B cells or T cells alone (Fig. 2B, compare the 2nd, 3rd, and 4th bars) with transfer of NP-specific B cells to CD19+/+ recipients further enhancing Bcl6 upregulation (Fig. 2B; compare the 5th to the 6th bars). Co-transfer of NP-specific B cells to CD19-deficient mice restored normal expansion of Tfh cells in the transferred population, with expression of Bcl6 protein and PD-1 equivalent to that seen following transfer to CD19+/+ recipients, whereas co-transfer with polyclonal B cells did not result in an increase in either compared to transfer of T cells alone (Fig. 2C, 2D, and 2E). By contrast, the presence or absence of NP-specific B cells had no effect on the downregulation of PSGL-1 on the transferred Ag-specific T cells (Fig. 2F), underscoring the observation (Fig. 1D) that initial downregulation of this glycoprotein is independent of T cell interactions with B cells, and precedes induction of CXCR5 and PD-1. Due to the lack of CXCR5 or PD-1 expression on T cells transferred alone or with polyclonal B cells to CD19−/− animals in these co-transfers (Fig. 2A, 2C, and 2D), we determined Bcl6 expression in the total Thy1.1+ PSGL-1lo populations to ensure that we did not bias our analysis of Bcl6-expressing cells by selecting T cells that only upregulated PD-1 and CXCR5.
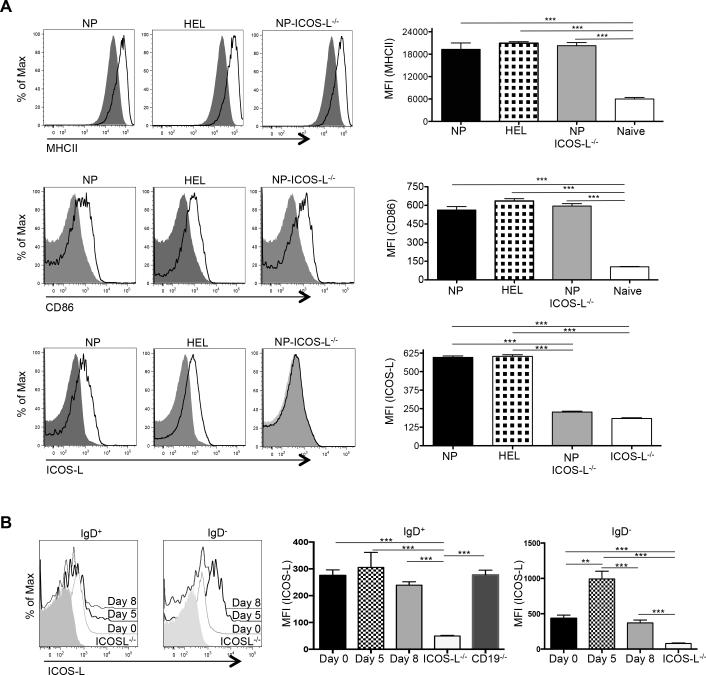
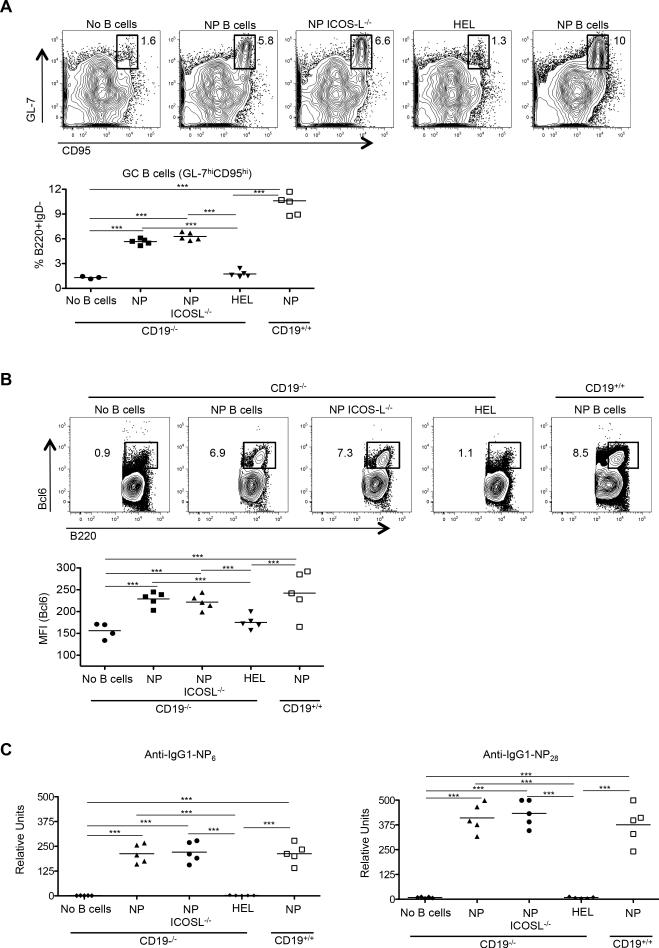
We considered two possible causes for the failure of Tfh-cell differentiation upon co-transfer of TCR transgenic T cells with polyclonal compared to the robust differentiation observed with NP-specific B cells: 1) that the former polyclonal cells were insufficiently activated following NP-OVA challenge, or 2) that they contained a relative lack of Ag-specific B cells compared to the NP-specific transfers. To address the first possibility, we immunized HEL-specific (MD4) (49) or NP-specific B1-8 Ig transgenic mice with either HEL or NP-OVA in alum, respectively, to activate B cells. Forty-eight hours post-immunization MD4 and B1-8 B cells were equivalently activated as assessed by upregulation of class II MHC and CD86 (Fig. 3A). ICOS-L was also robustly expressed on both populations, in a manner analogous to that seen on wild type B cells following their activation (Fig. 3B, compare ICOS-L expression on IgD+ and IgD− B cells to that on ICOS-L deficient animals; ICOS-L is also expressed on IgD+ B cells from CD19−/− mice as expected, given its constitutive expression on naïve B cells (50)). We then transferred purified B cells from these mice together with Thy1.1+ OT-II CD4 T cells into either CD19−/− or CD19+/+ recipients. Activated B1-8 B cells induced upregulation of CXCR5 and PD-1 in co-transferred OT-II T cells roughly equivalently in both CD19−/− and CD19+/+ recipients, whereas transfer of activated MD4 HEL-transgenic B cells failed to induce this Tfh-cell phenotype (Fig. 4A). Likewise, co-transfer of activated ICOS-L-bearing HEL-specific B cells neither induced Bcl6 protein expression in transferred T cells nor enhanced numbers of Bcl6+ PD-1hi OVA-specific T cells (Fig. 4B). Hence, the presence of activated B cells alone is not sufficient to drive antigen-specific Tfh-cell differentiation; rather, activated B cells also need to engage T cells in an Ag-dependent manner (39). To determine if the OVA-specific Tfh cells that developed following co-transfer with B cells were functional, we assessed GC development in recipient mice seven days following transfer and NP-OVA immunization. The percentage of B220+ IgD− CD95hiGL-7hi GC B cells was equivalent between CD19−/− and CD19+/+ recipients that received OT-II TCR transgenic T cells along with NP-specific B cells (Fig. 4C, 7.8% ± 1.7 vs. 7.3% ± 1.3, respectively). At this time point IgM and IgG1 NP-specific Abs are generated by both extrafollicular plasmablasts and GC-derived plasma cells that are dependent upon help provided by Bcl6+ T cells (46). Serum titers of both anti-NP IgM and IgG1 Abs were roughly equivalent between immunized CD19−/− and CD19+/+ mice that had received activated B1-8 B cells, whereas animals that were recipients of HEL-specific MD4 Ig transgenic cells had impaired Ab production in concert with their GC defects (Fig. 4D). Thus, cognate Ag presentation by B cells, not simply their activation, is essential for both full maturation and function of antigen-specific Tfh cells.
Figure 3.
Expression of surface proteins on adoptively transferred B cells. (A) Expression of class II MHC, CD86, and ICOS-L on NP-specific ICOS-L+/+ (n = 7), NP-ICOS-L−/− (n = 7) and HEL-specific MD4 B cells (n = 7) (solid black line) compared to expression of these proteins on naïve B cells (grey shaded area; left), with the MFI expression of these markers from each group of mice graphically displayed (right). (B) Expression of ICOS-L on transferred B cells before (day 0), and at days 5 and 8 after, immunization with NP-OVA (shaded histogram), with its MFI expression graphically displayed (right). ***p < 0.001; ** p < 0.003.
The requirement for B-cell ICOS-L for antigen-specific Tfh-cell development and function can be circumvented in the presence of activated Ag-specific B cells
While B-cell Ag presentation is typically required for the generation of Tfh cells, this does not seem to result from the provision of a unique B-cell derived signal; rather, it appears to be a consequence of responding B cells that rapidly become the primary source of presented Ag (Deenick et al., 2010). Indeed, Tfh cells can initially develop in the absence of Ag-presenting B cells when abundant Ag is presented by DCs (13, 39). Signaling via ICOS-L on DCs is necessary for the initiation of Tfh-cell development and initial Bcl6 upregulation, while its expression on B cells seems to provide signals important for maintenance of Bcl6 levels and Tfh-cell numbers and continued development (13, 34). Its expression on non-cognate follicular B cells is also necessary for optimal positioning of Ag-specific T cells for interaction with cognate B cells and subsequent Tfh-cell and GC development (40). However, the separable relationship between provision of Ag and ICOS costimulation from cognate B cells in antigen-specific Tfh-cell development is unclear.
To address this question, we devised a system to discern the effects of B cell contribution of Ag presentation, costimulation, or both. We intercrossed NP-specific B1-8 with ICOS-L-deficient (ICOS-L−/−) mice (51) yielding Ag-specific B cells deficient in ICOS-L (NP-ICOS-L−/−) that upon antigen stimulation upregulated class II MHC and CD86 equivalently to ICOS-L intact cells (Fig. 3). Because we had shown that robust Tfh-cell development and function was dependent on the presence of Ag-specific B cells (Fig. 4A, 4B, and 4C), co-transferring NP-ICOS-L−/− B cells with T cells allowed us to examine the specific role of ICOS-L in Tfh development. ICOS-L-intact or –deficient NP-specific B cells were co-transferred with OT-II TCR transgenic CD4 T cells into WT CD19+/+ or CD19−/− mice followed by immunization with NP-OVA with analysis 7 days later. OVA-specific T cells upregulated CXCR5 and PD-1, and had equivalent Bcl6 protein expression and IL-21 and IL-4 production, whether they were co-transferred with ICOS-sufficient or –deficient cognate B1-8 B cells (Fig. 5A, 5B, and 5C, respectively)
Figure 5.
ICOS-L on Ag-specific B cells is dispensable for antigen-specific Tfh formation and function. CD19−/− or CD19+/+ mice received CD4 Thy1.1+ OT-II cells alone (n = 7-10), or with ICOS-L+/+ NP-specific (n = 7-10) or ICOS-L−/− NP-specific B cells (n = 7-10), with spleens of recipients harvested 7 days after immunization with NP-OVA and sera collected. (A) Representative flow cytometry plots of CD4+ Thy1.1+ CD44+ CXCR5hi PD-1hi T cells from the recipients, with the graph on the right showing the percentages of such cells among the transferred population. (B) Representative Bcl6 and PD-1 expression in the transferred CD4+ T cells, including cells transferred without B cells (left 4 panels), in conjunction with Bcl6 expression in the Thy1.1+ PSGL-1lo population in the various recipients (right panel). (C) Representative flow cytometry plots of IL-21 and IL-4 expression (top 3 and bottom 3 panels, respectively) in the CD4+ Thy1.1+ CD44+ CXCR5hi PD-1hi T cells from the various recipients, with aggregate totals of cytokine positive cells from Thy1.1+ PSGL-1lo population (right panels, n = 5 in each group). (D) Representative flow cytometry plots of B220+ IgD− GL-7hi CD95hi GC B cells taken from the recipients, with the percentages of such cells among B220+ IgD− cells shown on the graph on the right. (E) IgM (left) and IgG (right) anti-NP28 Ab levels in the recipients. Experiments were performed 3 times with n ≥ 3 per group. ***p < 0.001; **p < 0.003 by Student's t-test comparing cells transferred into CD19−/− or CD19+/+ mice. Error bars represent standard deviation.
We next asked if the OVA-specific Tfh cells that developed following co-transfer with ICOS-L-intact or –deficient NP-specific B cells promoted similar GC responses seven days following transfer and NP-OVA immunization. The percentage of splenic B220+ IgD− CD95hi GL-7hi GC B cells was equivalent between recipients that received ICOS-L-intact or –deficient B cells (Fig. 5D, 7.7% ± 2.1 vs. 7.4% ± 1.8, respectively). Serum titers of both anti-NP IgM and class switched IgG1 Abs were also roughly equivalent among immunized CD19−/− and CD19+/+ mice that had received ICOS-L-intact or –deficient NP-specific B cells (Fig. 5E). Thus, cognate Ag presentation by B cells can bypass the requirement for ICOS signaling for both maturation and function of antigen-specific Tfh cells, the latter measured by GC formation and initiation of isotype switching.
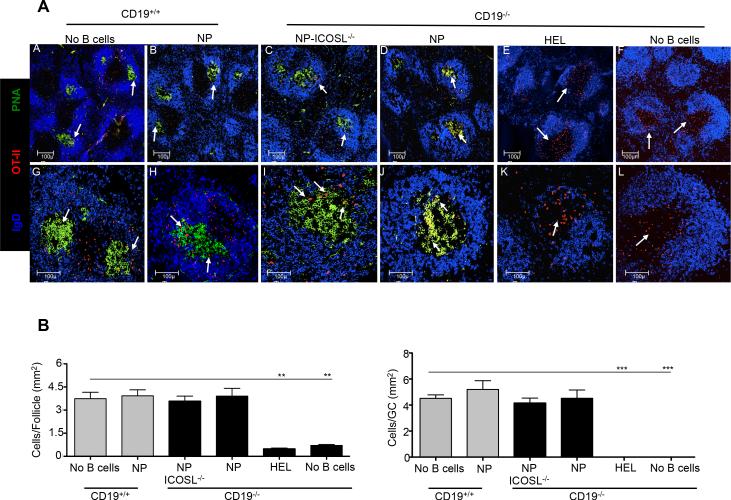
We next assessed the ability of T cells to migrate into the B cell follicle and function in GC responses in the presence or absence of ICOS-L signaling delivered by Ag-specific B cells. HEL- or NP-specific ICOS-L+/+ or NP-specific ICOS-L−/− B cells were co-transferred with OT-II red fluorescent protein (RFP)-expressing CD4 T cells (52) into CD19−/− hosts, with an additional group of recipients receiving only T cells, followed by immunization with NP-OVA in alum and sacrifice 7 days later. Donor RFP+ T cells were present in both the B cell follicle and GCs of CD19−/− mice that received NP-specific ICOS-L+/+ or NP-specific ICOS-L−/− B cells, as in CD19-intact mice (Fig. 6A and 6B). Moreover, CD19−/− recipients that received ICOS-L-sufficient or -deficient donor B1-8 B cells developed similar numbers of GC B cells and anti-NP Ab responses. By contrast, we found an increased number of Ag-specific RFP+ T cells outside of the follicle in CD19−/− mice that received HEL B cells, or T cells alone, with poor GC development. As expected, control animals that received T cells alone failed to generate either GC B cells or Ab responses.
Figure 6.
Ag-specific B cells are required for follicular migration of antigen-specific T cells. CD19−/− or CD19+/+ mice received RFP+ CD4+ Thy1.1+ OT-II cells alone (n = 5), or with NP-specific (n = 5), NP-specific ICOS-L−/− (n = 5), or HEL-specific MD4 B cells (n = 5), with spleens of recipients harvested 7 days after immunization with NP-OVA followed by staining for confocal microscopy. (A) Representative follicles (10×) stained with anti-IgD (blue) or PNA (green), along with transferred RFP+ OT-II T cells (red) (panels A-F) or representative follicles, with the same stains, shown at higher magnification (25× objective) (panels G-L). Bars, 100 μm. (B) T cells localized within follicles (left) and GCs (right) were manually counted (blinded) using Image J software, with numbers of T cells per mm2 of follicle and GCs determined, using all the follicles and GCs examined. Grey and black bars = CD19+/+ and CD19−/− recipients, respectively. ** p < 0.03 and *** p = 0.01.
Since Tfh cells are essential for GC maintenance and B cell affinity maturation, with their upregulation of Bcl6, we also examined the peak of the primary response, assessing the GC phenotype 14 days post-immunization. Consistent with our day 7 results, CD19−/− mice that received adoptively transferred activated HEL B cells, or T cells alone, failed to robustly generate GC B cells, with their B220+IgD− B cells also not significantly upregulating Bcl6 (Fig. 7A and 7B). By contrast, CD19−/− mice that received NP-specific ICOS-L+/+ or ICOS-L−/− B cells developed substantial numbers of GC B cells, with B220+IgD− B cells having enhanced Bcl6 expression similar to that seen in wild type mice. CD19+/+ mice had slightly more such cells, presumably a consequence of maturation of endogenous B cell populations. In addition, sera collected from CD19−/− mice that received NP-specific ICOS-L+/+ or ICOS-L−/− B cells had significantly increased levels of both high (NP6) and low affinity (NP28) NP-specific Ab levels compared to mice receiving HEL B cells or mice that did not receive B cell transfers (Fig. 7C).
Figure 7.
Cognate Tfh-B cell interactions are required for GC B cell development. CD19−/− or CD19+/+ mice received CD4+ Thy1.1+ OT-II cells alone (n = 5), or with NP-specific (n = 5), NP-specific ICOS-L−/− (n = 5), or HEL-specific MD4 B cells (n = 5), with spleens and sera of recipients harvested 14 days after immunization with NP-OVA. (A) Representative flow cytometry plots of B220+ IgD− GL-7hi CD95hi GC B cells from CD19−/− or CD19+/+ recipients (top 5 panels), with their percentages of B220+ IgD− cells (below). (B) Representative flow cytometry plots demonstrating the percentage of Bcl6+ B cells among the total B220+ IgD− population in spleens of recipients (top 5 panels), with aggregate percentages of same (below). (C) High and low affinity anti-NP IgG1 Abs (anti-NP6 and anti-NP28; left and right panels, respectively) were determined from the different recipients. Experiments were performed 3 times with n ≥ 3 per group. ***p < 0.001; **p < 0.003; *p < 0.02 by Student's t-test comparing cells transferred into CD19−/− or CD19+/+ mice. Error bars represent standard deviation.
Taken together, these data indicate that Ag presentation by activated cognate B cells is necessary to promote antigen-specific T cell migration into the B cell follicle with associated GC development and Ig affinity maturation, a phenotype consistent with the deficiency in T-cell specific CXCR5, PD-1, and Bcl6 upregulation in the absence of Ag-specific B cells (Fig. 3 and Fig. 4). They also demonstrate that in the context of robust Ag presentation by activated B cells, the latter's requirement for ICOS-L expression in antigen-specific Tfh-cell development, migration, and proper function can be circumvented.
ICOS-L on B cells is required for Tfh-cell development and GC responses when Ag-presenting B cells are limited
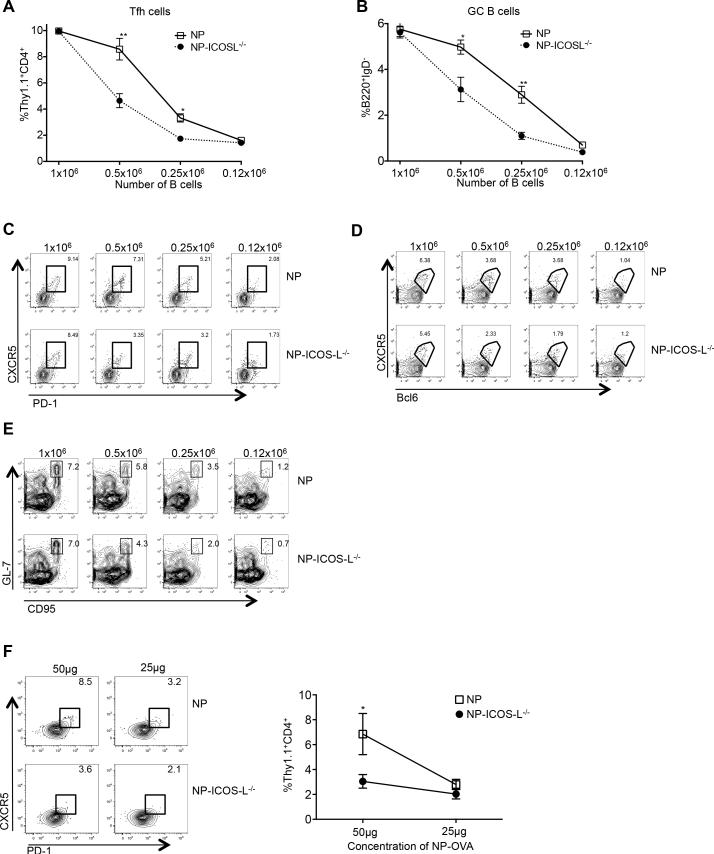
We next addressed the possibility that the transfer of a B cell population highly enriched for Ag-specific cells could overcome the requirement for their provision of ICOS-L in Tfh-cell development. Hence, we transferred decreasing numbers (1 × 106, 0.5 × 106, 0.25 × 106 and 0.12 × 106) of NP-specific B cells sufficient or deficient in ICOS-L, together with a fixed number of OT-II CD4 T cells into CD19−/− mice, followed by immunization with NP-OVA in alum. Seven days later, spleens were harvested and analyzed for Tfh and GC B cells. Upon limiting the transfer of NP-specific B cells, we observed a decline in the development of Ag-specific CXCR5hiPD-1hi OT-II Tfh cells that was proportional to the number of transferred B cells, with this decline significantly accentuated in the absence of ICOS-L on the transferred Ag-specific B cells; for example, compare the numbers of OVA-specific Tfh cells that developed in mice receiving 0.5 × 106 and 0.25 × 106 transferred ICOS-intact versus –deficient NP-specific B cells (Fig. 8A). A 50% decrease in the number of transferred NP-ICOS-L−/− B cells led to an approximately 50% reduction in the development of OT-II Tfh cells with a like reduction in their Bcl6 expression (Fig. 8A, 8C, and 8D). The significant reduction in percentage of Tfh cells that developed in concert with transfer of ICOS-L−/− compared to ICOS-L+/+ NP-specific B cells indicates that when T cells are not in the presence of an excess of Ag-specific B cells, co-stimulation from ICOS-L on B cells becomes essential for efficient OVA-specific Tfh-cell differentiation.
Figure 8.
ICOS-L on B cells is required for Tfh-cell development and GC responses when Ag-presenting B cells or Ag are limited. (A) CD19−/− mice received 1 × 106, 0.5 × 106, 0.25 × 106 or 0.12 × 106 NP-specific or NP-specific ICOS-L−/− B cells co-transferred with 0.5 × 106 CD4+ Thy1.1+ OT-II T cells, with spleens of recipients harvested 7 days after immunization with NP-OVA. Percentages of CD4+ Thy1.1+ CD44+ CXCR5hi PD-1hi T and B220+ IgD− GL-7hi CD95hi B cells from animals receiving various numbers of NP-specific B cells are shown. (C-E) Representative flow cytometry plots of splenic CD4+ Thy1.1+ OT-II CD44+CXCR5hiPD-1hi or CD4+Thy1.1+CD44+CXCR5hiBcl6hi T cells, or B220+IgDloGL-7hiCD95hi B cells. Experiments were performed 3 times with n ≥ 5 per group. (F) CD4+ Thy1.1+ OT-II cells were transferred into CD19-deficient mice, followed by immunization with 100 μg NP-OVA. Two days after priming, 0.05 × 106 OT-II T cells were re-transferred, along with 1 × 106 ICOS-L-intact or –deficient NP-specific B cells, into CD19−/− animals primed 2 days earlier with 25 or 50 ug of NP-OVA, with spleens of recipients harvested 7 days after immunization. Representative flow cytometry plots of splenic CD4+ Thy1.1+ OT-II CD44+CXCR5hiPD-1hi Tfh cells with the percentages of such cells among CD4+ Thy1.1+ cells shown on the graph on the right. n = 3 mice per group. **p < 0.003; *p < 0.02 by Sidak's multiple comparison test comparing the means of Tfh (A and F) and GC B (B) cell percentages (of transferred cells) in recipient mice. Error bars represent standard deviation.
We also analyzed GC B cell development after co-transfer of a range of ICOS-intact and–deficient NP B cells. As we observed for Tfh-cell development, GC B cell formation was hindered as the numbers of Ag-specific B cells in co-transfers were limited, in parallel with demonstration of a requirement for ICOS-L on B cells (Fig. 8B and 8E). Our data shown above demonstrated that the NP-ICOS-L−/− B cells do not have an intrinsic defect in GC B cell development or function (Fig. 6 and Fig. 7); thus, the diminished GC response observed in the absence of ICOS-L on B cells when Ag-specific B cells are limited is likely due to defective Tfh-cell differentiation. Hence, when fewer Ag-specific B cells are present, ICOS signaling by Ag-specific B cells is required for robust antigen-specific Tfh-cell development and function.
We next examined the requirement for cognate B-cell expression of ICOS-L for Tfh-cell development in a situation in which Ag is limiting. We transferred congenically mismatched Thy1.1+ OT-II CD4 T cells into CD19−/− mice followed by immunization with 100 μg of NP-OVA to ensure equivalent priming of nascent Tfh cells by DCs. Thy1.1+ CD4 T cells were then sorted 48 hours after immunization and re-transferred with NP-specific B cells sufficient or deficient in ICOS-L into CD19−/− recipients primed two earlier with 25 or 50 μg of NP-OVA. The former Ag dose was not sufficient to engender a robust Tfh-cell response (Fig. 8F, left flow cytometry panels); however, upon immunization of recipients with 50 μg of NP-OVA, CXCR5hiPD-1hi OT-II Tfh cells robustly developed, but were reduced in the absence of ICOS-L on cognate B cells (Fig. 8F, left flow cytometry panels, and graph on right). This finding demonstrates that ICOS-L expression on B cells is required for development of Tfh cells at this dose of Ag, one that is presumably limiting.
Discussion
Cognate B cells are required for functional Tfh-cell development with GC formation, as is B-cell expression of ICOS-L; however, the separable contributions of delivery of Ag and ICOS-L by cognate B cells in antigen-specific Tfh-cell development, migration, and function have not been dissected. Here we do so, finding that robust Tfh-cell and GC B cell formation are dependent upon ICOS-L provided by cognate B cells, but only when Ag presentation from the latter is limiting. The requirement for B-cell delivery of an ICOS costimulatory signal is overcome in circumstances of their robust Ag delivery. These findings demonstrate that Ag-specific B cells provide different, yet compensatory signals for the differentiation of their cognate Tfh cells, while reconciling conflicting data indicating a requirement for ICOS-L expression on cognate B cells for Tfh-cell development that was not necessarily restricted to degree of Ag delivery (13, 32, 34), with those demonstrating this ICOS-L requirement could be bypassed in lieu of that tendered by non-cognate B cells (40). Our data fit the idea that excessive Ag delivery to cognate T cells can circumvent the need for specific costimulatory signals delivered by cognate B cells (39), while extending this latter work with the demonstration that ICOS-L is required for such development when Ag is limiting.
Reports from several groups have established that cognate B cells are required for the differentiation and function of Tfh cells (3, 10, 13, 15, 16, 21, 23). Our data corroborate this conclusion, with the added dissection of the effects of B cells upon Tfh-cell development, follicular migration, and function as determined by GC development and Ig affinity maturation. Co-transfer of activated non-cognate B cells with Ag-specific T cells failed to maintain the Tfh-cell developmental program and drive GC responses. Similar to SAP-deficient B cells which have a defect in their ability to form prolonged immunological synapses with T cells (8, 26, 27), our experiments with highly activated hen egg lysozyme-specific B cells corroborate the need for B cells to provide TCR signaling via cognate interactions for development, follicular migration, and function of Tfh cells. These experiments emphasize the necessity for Tfh cells to interact with B cells in an Ag-specific manner to upregulate Bcl6 and form proper GCs.
In a like manner, several investigators have dissected the role of ICOS-L signaling in Tfh-cell development. Blocking the function of ICOS-L on B cells by Abs or via conditional genetic deletion abrogates formation of Tfh cells (13, 34). Similarly, Tfh-cell development is impaired in NF-κB-inducing kinase-deficient mice, which lack ICOS-L upregulation on B cells (53), although such animals have a kinase deficiency in DCs that could potentially affect ICOS-L expression on this lineage. Yet, recent work found that antigen-specific T cells localize to the follicle even when co-transferred with cognate B cells that are deficient in ICOS-L (40). We too observed migration of T cells into the follicle upon co-transfer with either ICOS-sufficient or – deficient cognate B cells; however, we extended this earlier work with the demonstration that upon B cell co-transfers, Ag-specific T cells upregulated CXCR5 and PD-1 and achieved equivalent Bcl6 protein expression (Fig. 5A and 5B). Importantly, our system also allowed us to define the interacting roles of antigen presentation and ICOS signaling upon Tfh-cell maturation, with our results demonstrating a requirement for the latter in situations in which B-cell Ag presentation is limited (Fig. 8A). Thus our work establishes separable roles for B-cell delivery of Ag and co-stimulation in driving Tfh-cell development.
A hallmark of the NP-hapten primary Ab response is the development of high affinity anti-NP-specific Abs, a product of GC-matured plasma cells (54). Tfh cells developing in immunized CD19−/− mice that received Ag-specific, but ICOS-L-deficient, B cells expressed IL-21 and IL-4 equivalent to that seen in mice following transfer of ICOS-L-intact B cells, along with promoting production of similar amounts of high affinity NP-specific Abs (Fig. 7). IL-4 and IL-21 are necessary for genesis of a normal Ag-specific GC response (4, 5, 46, 55), with the quality of the latter a reflection of the persistence of Tfh cells (4). These data suggest that robust Tfh-cell differentiation can be achieved in the absence of ICOS-ICOS-L signaling from B cells, as long as B-cell display of Ag is not limiting, analogous to other circumstances of Ag excess (39). Thus, as Tfh cells mature, they are able to integrate compensatory signals, including Ag availability and costimulatory ligands, as they promote GC maturation.
Upon transfer to CD19−/− mice and immunization, nascent Tfh cells upregulated Bcl6 mRNA and protein compared to naïve T cells; however, only after transfer of Ag-specific B cells did Bcl6 expression in Tfh cells achieve levels similar to those activated in CD19+/+ recipients. In the absence of CD19-intact, and presumably functional B cells, DCs are the primary Ag-presenting cells leading to CD4 T cell activation; thus, the increased Bcl6 expression and development of the Tfh phenotype is a consequence of their priming by DCs. These findings are consistent with previous reports that DCs initiate Bcl6 upregulation via ICOS-L with initiation of the Tfh-cell developmental program (13-15, 17). While upregulation of Bcl6 protein was impaired in the T cells adoptively transferred to CD19−/− mice, its expression was nonetheless maintained, a finding concomitant with that of PSGL-1 downregulation. The latter appears to be a very early developmental step in Tfh-cell differentiation (23), like that of CCR7 downregulation (21, 39), and in our experiments herein, preceding that of CXCR5 and PD-1 upregulation, which also occur early in Tfh-cell differentiation (13-15, 17). Downregulation of CCR7 and PSGL-1 is permissive for emigration of maturing Tfh cells from the T cell zone following release from their CCL19 and CCL21 tethers (20, 22, 23), with migration to the T-B cell border of the spleen or interfollicular region of the lymph node where they receive further developmental signals from B cells, with CXCR5 expression enabling their migration into the B cell follicle (21). These findings emphasize that during a primary immune response, Tfh-cell development is a sequential process requiring signals from different cell types. It also suggests that PSGL-1 downregulation is part of the Bcl6-driven Tfh-cell developmental program, but one that requires different transcriptional events than other steps in this program, such as CXCR5 upregulation (10-12).
Development of Tfh cells with subsequent GC maturation in the absence of ICOS-L signaling from B cells has implications for the understanding and treatment of autoimmune diseases and chronic infections. In scenarios during which excessive self or non-self Ag is present, the need for temporal signaling to developing Tfh cells may be bypassed with excessive TCR signaling contributing to T cell expansion (39, 56) or differentiation (57). Although ICOS is also important for such expansion (35), its contribution may be relatively less than that of other signals such as CD28 (37), with abundant Ag presentation possibly contributing to the aberrant formation of Tfh cells observed in chronic viral infections in mice, macaques, and humans (56, 58, 59), and in systemic autoimmunity in mice and in humans (60-62). The latter might account for our earlier observation that pathogenic plasmablast and autoantibody formation, while diminished in ICOS-deficient lupus-prone mice compared to ICOS-intact controls, are not completely abrogated (22, 63). Much effort has been devoted to dissecting the steps in Tfh-cell development in the hope that they may be augmented for effective vaccine development or clearance of pathogens in chronic infection, or alternatively, blocked for therapy of autoimmune syndromes. Our work suggests that, at least in terms of the latter, blockade of specific co-stimulatory molecules required for T-B cell collaboration may not be therapeutically sufficient, if other drivers of Tfh-cell development such as Ag compensate. More work is needed to evaluate the relative roles of Ag dose versus costimulatory signaling in Tfh-cell development, and whether these represent possible therapeutic targets for modulating the immunopathology that occurs during autoimmunity and chronic infection.
Acknowledgments
The authors thank the members of the Craft lab for helpful discussions.
Footnotes
Abbreviations: B6, C57BL/6J; Bcl6, B-cell lymphoma 6; DC, dendritic cell; CD19−/−, CD19-deficient; CD19+/+, CD19-intact; GC, germinal center; ICOS-L, ICOS ligand; ICOS-L−/−, ICOS-L-deficient; NP, (4-hydroxy-3-nitrophenyl)acetyl; PD-1, programmed cell death 1; PI3K, phosphatidylinositol 3-kinase; PSGL-1, P-selectin glycoprotein ligand 1; RFP, red fluorescent protein; SLOs, secondary lymphoid organs; Tfh cell, follicular helper T cell; WT, wild type
References
- 1.King C, Tangye SG, Mackay CR. T follicular helper (TFH) cells in normal and dysregulated immune responses. Annu Rev Immunol. 2008;26:741–766. doi: 10.1146/annurev.immunol.26.021607.090344. [DOI] [PubMed] [Google Scholar]
- 2.Xu J, Foy TM, Laman JD, Elliott EA, Dunn JJ, Waldschmidt TJ, Elsemore J, Noelle RJ, Flavell RA. Mice deficient for the CD40 ligand. Immunity. 1994;1:423–431. doi: 10.1016/1074-7613(94)90073-6. [DOI] [PubMed] [Google Scholar]
- 3.Zaretsky AG, Taylor JJ, King IL, Marshall FA, Mohrs M, Pearce EJ. T follicular helper cells differentiate from Th2 cells in response to helminth antigens. J Exp Med. 2009;206:991–999. doi: 10.1084/jem.20090303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zotos D, Coquet JM, Zhang Y, Light A, D'Costa K, Kallies A, Corcoran LM, Godfrey DI, Toellner KM, Smyth MJ, Nutt SL, Tarlinton DM. IL-21 regulates germinal center B cell differentiation and proliferation through a B cell-intrinsic mechanism. J Exp Med. 2010;207:365–378. doi: 10.1084/jem.20091777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Linterman MA, Beaton L, Yu D, Ramiscal RR, Srivastava M, Hogan JJ, Verma NK, Smyth MJ, Rigby RJ, Vinuesa CG. IL-21 acts directly on B cells to regulate Bcl-6 expression and germinal center responses. J Exp Med. 2010;207:353–363. doi: 10.1084/jem.20091738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Good-Jacobson KL, Szumilas CG, Chen L, Sharpe AH, Tomayko MM, Shlomchik MJ. PD-1 regulates germinal center B cell survival and the formation and affinity of long-lived plasma cells. Nat Immunol. 2010;11:535–542. doi: 10.1038/ni.1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Reinhardt RL, Liang HE, Locksley RM. Cytokine-secreting follicular T cells shape the antibody repertoire. Nat Immunol. 2009;10:385–393. doi: 10.1038/ni.1715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yusuf I, Kageyama R, Monticelli L, Johnston RJ, DiToro D, Hansen K, Barnett B, Crotty S. Germinal Center T Follicular Helper Cell IL-4 Production Is Dependent on Signaling Lymphocytic Activation Molecule Receptor (CD150). J Immunol. 2010;185:190–202. doi: 10.4049/jimmunol.0903505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.King IL, Mohrs M. IL-4-producing CD4+ T cells in reactive lymph nodes during helminth infection are T follicular helper cells. J Exp Med. 2009;206:1001–1007. doi: 10.1084/jem.20090313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Johnston RJ, Poholek AC, DiToro D, Yusuf I, Eto D, Barnett B, Dent AL, Craft J, Crotty S. Bcl6 and Blimp-1 Are Reciprocal and Antagonistic Regulators of T Follicular Helper Cell Differentiation. Science. 2009:1175870. doi: 10.1126/science.1175870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nurieva RI, Chung Y, Martinez GJ, Yang XO, Tanaka S, Matskevitch TD, Wang YH, Dong C. Bcl6 mediates the development of T follicular helper cells. Science. 2009;325:1001–1005. doi: 10.1126/science.1176676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yu D, Rao S, Tsai LM, Lee SK, He Y, Sutcliffe EL, Srivastava M, Linterman M, Zheng L, Simpson N, Ellyard JI, Parish IA, Ma CS, Li QJ, Parish CR, Mackay CR, Vinuesa CG. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity. 2009;31:457–468. doi: 10.1016/j.immuni.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 13.Choi YS, Kageyama R, Eto D, Escobar TC, Johnston RJ, Monticelli L, Lao C, Crotty S. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity. 2011;34:932–946. doi: 10.1016/j.immuni.2011.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goenka R, Barnett LG, Silver JS, O'Neill PJ, Hunter CA, Cancro MP, Laufer TM. Cutting edge: dendritic cell-restricted antigen presentation initiates the follicular helper T cell program but cannot complete ultimate effector differentiation. J Immunol. 2011;187:1091–1095. doi: 10.4049/jimmunol.1100853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kerfoot SM, Yaari G, Patel JR, Johnson KL, Gonzalez DG, Kleinstein SH, Haberman AM. Germinal center B cell and T follicular helper cell development initiates in the interfollicular zone. Immunity. 2011;34:947–960. doi: 10.1016/j.immuni.2011.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kitano M, Moriyama S, Ando Y, Hikida M, Mori Y, Kurosaki T, Okada T. Bcl6 protein expression shapes pre-germinal center B cell dynamics and follicular helper T cell heterogeneity. Immunity. 2011;34:961–972. doi: 10.1016/j.immuni.2011.03.025. [DOI] [PubMed] [Google Scholar]
- 17.Baumjohann D, Okada T, Ansel KM. Cutting Edge: Distinct waves of BCL6 expression during T follicular helper cell development. J Immunol. 2011;187:2089–2092. doi: 10.4049/jimmunol.1101393. [DOI] [PubMed] [Google Scholar]
- 18.Pepper M, Pagan AJ, Igyarto BZ, Taylor JJ, Jenkins MK. Opposing signals from the Bcl6 transcription factor and the interleukin-2 receptor generate T helper 1 central and effector memory cells. Immunity. 2011;35:583–595. doi: 10.1016/j.immuni.2011.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Debes GF, Arnold CN, Young AJ, Krautwald S, Lipp M, Hay JB, Butcher EC. Chemokine receptor CCR7 required for T lymphocyte exit from peripheral tissues. Nat Immunol. 2005;6:889–894. doi: 10.1038/ni1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Veerman KM, Williams MJ, Uchimura K, Singer MS, Merzaban JS, Naus S, Carlow DA, Owen P, Rivera-Nieves J, Rosen SD, Ziltener HJ. Interaction of the selectin ligand PSGL-1 with chemokines CCL21 and CCL19 facilitates efficient homing of T cells to secondary lymphoid organs. Nat Immunol. 2007;8:532–539. doi: 10.1038/ni1456. [DOI] [PubMed] [Google Scholar]
- 21.Haynes NM, Allen CD, Lesley R, Ansel KM, Killeen N, Cyster JG. Role of CXCR5 and CCR7 in follicular Th cell positioning and appearance of a programmed cell death gene-1high germinal center-associated subpopulation. J Immunol. 2007;179:5099–5108. doi: 10.4049/jimmunol.179.8.5099. [DOI] [PubMed] [Google Scholar]
- 22.Odegard JM, Marks BR, DiPlacido LD, Poholek AC, Kono DH, Dong C, Flavell RA, Craft J. ICOS-dependent extrafollicular helper T cells elicit IgG production via IL-21 in systemic autoimmunity. J Exp Med. 2008;205:2873–2886. doi: 10.1084/jem.20080840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Poholek AC, Hansen K, Hernandez SG, Eto D, Chandele A, Weinstein JS, Dong X, Odegard JM, Kaech SM, Dent AL, Crotty S, Craft J. In vivo regulation of Bcl6 and T follicular helper cell development. J Immunol. 2010;185:313–326. doi: 10.4049/jimmunol.0904023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Coffey F, Alabyev B, Manser T. Initial clonal expansion of germinal center B cells takes place at the perimeter of follicles. Immunity. 2009;30:599–609. doi: 10.1016/j.immuni.2009.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ansel KM, McHeyzer-Williams LJ, Ngo VN, McHeyzer-Williams MG, Cyster JG. In vivo-activated CD4 T cells upregulate CXC chemokine receptor 5 and reprogram their response to lymphoid chemokines. J Exp Med. 1999;190:1123–1134. doi: 10.1084/jem.190.8.1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kamperschroer C, Roberts DM, Zhang Y, Weng NP, Swain SL. SAP enables T cells to help B cells by a mechanism distinct from Th cell programming or CD40 ligand regulation. J Immunol. 2008;181:3994–4003. doi: 10.4049/jimmunol.181.6.3994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Qi H, Cannons JL, Klauschen F, Schwartzberg PL, Germain RN. SAP-controlled T-B cell interactions underlie germinal centre formation. Nature. 2008;455:764–769. doi: 10.1038/nature07345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McAdam AJ, Greenwald RJ, Levin MA, Chernova T, Malenkovich N, Ling V, Freeman GJ, Sharpe AH. ICOS is critical for CD40-mediated antibody class switching. Nature. 2001;409:102–105. doi: 10.1038/35051107. [DOI] [PubMed] [Google Scholar]
- 29.Dong C, Juedes AE, Temann UA, Shresta S, Allison JP, Ruddle NH, Flavell RA. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature. 2001;409:97–101. doi: 10.1038/35051100. [DOI] [PubMed] [Google Scholar]
- 30.Tafuri A, Shahinian A, Bladt F, Yoshinaga SK, Jordana M, Wakeham A, Boucher LM, Bouchard D, Chan VS, Duncan G, Odermatt B, Ho A, Itie A, Horan T, Whoriskey JS, Pawson T, Penninger JM, Ohashi PS, Mak TW. ICOS is essential for effective T-helper-cell responses. Nature. 2001;409:105–109. doi: 10.1038/35051113. [DOI] [PubMed] [Google Scholar]
- 31.Dong C, Temann UA, Flavell RA. Cutting edge: critical role of inducible costimulator in germinal center reactions. J Immunol. 2001;166:3659–3662. doi: 10.4049/jimmunol.166.6.3659. [DOI] [PubMed] [Google Scholar]
- 32.Akiba H, Takeda K, Kojima Y, Usui Y, Harada N, Yamazaki T, Ma J, Tezuka K, Yagita H, Okumura K. The Role of ICOS in the CXCR5+ Follicular B Helper T Cell Maintenance In Vivo. J Immunol. 2005;175:2340–2348. doi: 10.4049/jimmunol.175.4.2340. [DOI] [PubMed] [Google Scholar]
- 33.Bossaller L, Burger J, Draeger R, Grimbacher B, Knoth R, Plebani A, Durandy A, Baumann U, Schlesier M, Welcher AA, Peter HH, Warnatz K. ICOS Deficiency Is Associated with a Severe Reduction of CXCR5+CD4 Germinal Center Th Cells. J Immunol. 2006;177:4927–4932. doi: 10.4049/jimmunol.177.7.4927. [DOI] [PubMed] [Google Scholar]
- 34.Nurieva RI, Chung Y, Hwang D, Yang XO, Kang HS, Ma L, Wang YH, Watowich SS, Jetten AM, Tian Q, Dong C. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity. 2008;29:138–149. doi: 10.1016/j.immuni.2008.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Burmeister Y, Lischke T, Dahler AC, Mages HW, Lam KP, Coyle AJ, Kroczek RA, Hutloff A. ICOS controls the pool size of effector-memory and regulatory T cells. J Immunol. 2008;180:774–782. doi: 10.4049/jimmunol.180.2.774. [DOI] [PubMed] [Google Scholar]
- 36.Vinuesa CG, Cook MC, Angelucci C, Athanasopoulos V, Rui L, Hill KM, Yu D, Domaschenz H, Whittle B, Lambe T, Roberts IS, Copley RR, Bell JI, Cornall RJ, Goodnow CC. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature. 2005;435:452–458. doi: 10.1038/nature03555. [DOI] [PubMed] [Google Scholar]
- 37.Gigoux M, Shang J, Pak Y, Xu M, Choe J, Mak TW, Suh WK. Inducible costimulator promotes helper T-cell differentiation through phosphoinositide 3-kinase. Proc Natl Acad Sci U S A. 2009;106:20371–20376. doi: 10.1073/pnas.0911573106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bauquet AT, Jin H, Paterson AM, Mitsdoerffer M, Ho IC, Sharpe AH, Kuchroo VK. The costimulatory molecule ICOS regulates the expression of c-Maf and IL-21 in the development of follicular T helper cells and TH-17 cells. Nat Immunol. 2009;10:167–175. doi: 10.1038/ni.1690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deenick EK, Chan A, Ma CS, Gatto D, Schwartzberg PL, Brink R, Tangye SG. Follicular Helper T Cell Differentiation Requires Continuous Antigen Presentation that Is Independent of Unique B Cell Signaling. Immunity. 2010;33:241–253. doi: 10.1016/j.immuni.2010.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xu H, Li X, Liu D, Li J, Zhang X, Chen X, Hou S, Peng L, Xu C, Liu W, Zhang L, Qi H. Follicular T-helper cell recruitment governed by bystander B cells and ICOS-driven motility. Nature. 2013;496:523–527. doi: 10.1038/nature12058. [DOI] [PubMed] [Google Scholar]
- 41.Taki S, Meiering M, Rajewsky K. Targeted insertion of a variable region gene into the immunoglobulin heavy chain locus. Science. 1993;262:1268–1271. doi: 10.1126/science.8235657. [DOI] [PubMed] [Google Scholar]
- 42.Haynes NM, Allen CDC, Lesley R, Ansel KM, Killeen N, Cyster JG. Role of CXCR5 and CCR7 in Follicular Th Cell Positioning and Appearance of a Programmed Cell Death Gene-1High Germinal Center-Associated Subpopulation. J Immunol. 2007;179:5099–5108. doi: 10.4049/jimmunol.179.8.5099. [DOI] [PubMed] [Google Scholar]
- 43.Ngo VN, Cornall RJ, Cyster JG. Splenic T zone development is B cell dependent. J Exp Med. 2001;194:1649–1660. doi: 10.1084/jem.194.11.1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rickert RC, Rajewsky K, Roes J. Impairment of T-cell-dependent B-cell responses and B-1 cell development in CD19-deficient mice. Nature. 1995;376:352–355. doi: 10.1038/376352a0. [DOI] [PubMed] [Google Scholar]
- 45.Bai L, Constantinides MG, Thomas SY, Reboulet R, Meng F, Koentgen F, Teyton L, Savage PB, Bendelac A. Distinct APCs explain the cytokine bias of alpha-galactosylceramide variants in vivo. J Immunol. 2012;188:3053–3061. doi: 10.4049/jimmunol.1102414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lee SK, Rigby RJ, Zotos D, Tsai LM, Kawamoto S, Marshall JL, Ramiscal RR, Chan TD, Gatto D, Brink R, Yu D, Fagarasan S, Tarlinton DM, Cunningham AF, Vinuesa CG. B cell priming for extrafollicular antibody responses requires Bcl-6 expression by T cells. J Exp Med. 2011;208:1377–1388. doi: 10.1084/jem.20102065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sonoda E, Pewzner-Jung Y, Schwers S, Taki S, Jung S, Eilat D, Rajewsky K. B Cell Development under the Condition of Allelic Inclusion. Immunity. 1997;6:225–233. doi: 10.1016/s1074-7613(00)80325-8. [DOI] [PubMed] [Google Scholar]
- 48.Lalor PA, Nossal GJ, Sanderson RD, McHeyzer-Williams MG. Functional and molecular characterization of single, (4-hydroxy-3-nitrophenyl)acetyl (NP)-specific, IgG1+ B cells from antibody-secreting and memory B cell pathways in the C57BL/6 immune response to NP. Eur J Immunol. 1992;22:3001–3011. doi: 10.1002/eji.1830221136. [DOI] [PubMed] [Google Scholar]
- 49.Goodnow CC, Crosbie J, Adelstein S, Lavoie TB, Smith-Gill SJ, Brink RA, Pritchard-Briscoe H, Wotherspoon JS, Loblay RH, Raphael K, Trent RJ, Basten A. Altered immunoglobulin expression and functional silencing of self-reactive B lymphocytes in transgenic mice. Nature. 1988;334:676–682. doi: 10.1038/334676a0. [DOI] [PubMed] [Google Scholar]
- 50.Yoshinaga SK, Whoriskey JS, Khare SD, Sarmiento U, Guo J, Horan T, Shih G, Zhang M, Coccia MA, Kohno T, Tafuri-Bladt A, Brankow D, Campbell P, Chang D, Chiu L, Dai T, Duncan G, Elliott GS, Hui A, McCabe SM, Scully S, Shahinian A, Shaklee CL, Van G, Mak TW, Senaldi G. T-cell co-stimulation through B7RP-1 and ICOS. Nature. 1999;402:827–832. doi: 10.1038/45582. [DOI] [PubMed] [Google Scholar]
- 51.Mak TW, Shahinian A, Yoshinaga SK, Wakeham A, Boucher LM, Pintilie M, Duncan G, Gajewska BU, Gronski M, Eriksson U, Odermatt B, Ho A, Bouchard D, Whorisky JS, Jordana M, Ohashi PS, Pawson T, Bladt F, Tafuri A. Costimulation through the inducible costimulator ligand is essential for both T helper and B cell functions in T cell-dependent B cell responses. Nat Immunol. 2003;4:765–772. doi: 10.1038/ni947. [DOI] [PubMed] [Google Scholar]
- 52.Zhu H, Wang G, Li G, Han M, Xu T, Zhuang Y, Wu X. Ubiquitous expression of mRFP1 in transgenic mice. Genesis. 2005;42:86–90. doi: 10.1002/gene.20129. [DOI] [PubMed] [Google Scholar]
- 53.Hu H, Wu X, Jin W, Chang M, Cheng X, Sun SC. Noncanonical NF-kappaB regulates inducible costimulator (ICOS) ligand expression and T follicular helper cell development. Proc Natl Acad Sci U S A. 2011;108:12827–12832. doi: 10.1073/pnas.1105774108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Han S, Zheng B, Dal Porto J, Kelsoe G. In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. IV. Affinity-dependent, antigen-driven B cell apoptosis in germinal centers as a mechanism for maintaining self-tolerance. J Exp Med. 1995;182:1635–1644. doi: 10.1084/jem.182.6.1635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kuhn R, Rajewsky K, Muller W. Generation and analysis of interleukin-4 deficient mice. Science. 1991;254:707–710. doi: 10.1126/science.1948049. [DOI] [PubMed] [Google Scholar]
- 56.Fahey LM, Wilson EB, Elsaesser H, Fistonich CD, McGavern DB, Brooks DG. Viral persistence redirects CD4 T cell differentiation toward T follicular helper cells. J Exp Med. 2011;208:987–999. doi: 10.1084/jem.20101773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tubo NJ, Pagan AJ, Taylor JJ, Nelson RW, Linehan JL, Ertelt JM, Huseby ES, Way SS, Jenkins MK. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell. 2013;153:785–796. doi: 10.1016/j.cell.2013.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Petrovas C, Yamamoto T, Gerner MY, Boswell KL, Wloka K, Smith EC, Ambrozak DR, Sandler NG, Timmer KJ, Sun X, Pan L, Poholek A, Rao SS, Brenchley JM, Alam SM, Tomaras GD, Roederer M, Douek DC, Seder RA, Germain RN, Haddad EK, Koup RA. CD4 T follicular helper cell dynamics during SIV infection. J Clin Invest. 2012;122:3281–3294. doi: 10.1172/JCI63039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lindqvist M, van Lunzen J, Soghoian DZ, Kuhl BD, Ranasinghe S, Kranias G, Flanders MD, Cutler S, Yudanin N, Muller MI, Davis I, Farber D, Hartjen P, Haag F, Alter G, Schulze zur Wiesch J, Streeck H. Expansion of HIV-specific T follicular helper cells in chronic HIV infection. J Clin Invest. 2012;122:3271–3280. doi: 10.1172/JCI64314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bubier JA, Sproule TJ, Foreman O, Spolski R, Shaffer DJ, Morse HC, 3rd, Leonard WJ, Roopenian DC. A critical role for IL-21 receptor signaling in the pathogenesis of systemic lupus erythematosus in BXSB-Yaa mice. Proc Natl Acad Sci U S A. 2009;106:1518–1523. doi: 10.1073/pnas.0807309106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Simpson N, Gatenby PA, Wilson A, Malik S, Fulcher DA, Tangye SG, Manku H, Vyse TJ, Roncador G, Huttley GA, Goodnow CC, Vinuesa CG, Cook MC. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 2010;62:234–244. doi: 10.1002/art.25032. [DOI] [PubMed] [Google Scholar]
- 62.Weinstein JS, Hernandez SG, Craft J. T cells that promote B-Cell maturation in systemic autoimmunity. Immunol Rev. 2012;247:160–171. doi: 10.1111/j.1600-065X.2012.01122.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Odegard JM, DiPlacido LD, Greenwald L, Kashgarian M, Kono DH, Dong C, Flavell RA, Craft J. ICOS Controls Effector Function but Not Trafficking Receptor Expression of Kidney-Infiltrating Effector T Cells in Murine Lupus. J Immunol. 2009;182:4076–4084. doi: 10.4049/jimmunol.0800758. [DOI] [PMC free article] [PubMed] [Google Scholar]