Abstract
MicroRNAs (miRNAs) can control stem cell differentiation by targeting mRNAs. Using 96-well plate electroporation, we screened 466 human miRNA mimics by 4-color flow cytometry to explore differentiation of common myeloid progenitors (CMP) derived from human embryonic stem cells (hESCs). The transfected cells were then cultured in a cytokine cocktail that supported multiple hematopoietic lineages. At 4-5 days post-transfection, flow cytometry of erythroid (CD235+CD41−), megakaryocyte (CD41+CD42+) and myeloid (CD18+CD235−) lineages revealed miR-105 as a novel enhancer of megakaryocyte production during in vitro primitive hematopoiesis. In hESC-derived CMPs, miR-105 caused a 6-fold enhancement in megakaryocyte production. MiR-513a, miR-571 and miR-195 were found to be less potent megakaryocyte enhancers. We confirmed the relevance of miR-105 in adult megakaryopoiesis by demonstrating increased megakaryocyte yield and megakaryocyte colony forming potential in human adult CD34+ cells derived from peripheral blood. In addition, adult CD34+ cells express endogenous miR-105 during megakaryocyte differentiation. SiRNA knockdown of the hematopoietic transcription factor c-Myb caused a similar enhancement of megakaryocyte production as miR-105. Finally, a luciferase/c-Myb-3’UTR construct and western blot analysis demonstrated that the hematopoietic transcription factor c-Myb mRNA was a target of miR-105. We report a novel hESC-based miR screening platform and demonstrate that miR-105 is an enhancer of megakaryopoiesis in both primitive and definitive hematopoiesis.
Keywords: miRNA, megakaryocyte, hematopoiesis, embryonic stem cells
INTRODUCTION
MicroRNAs (miRNAs) are endogenously expressed non-coding RNAs, about 22 base pairs in length when fully processed, which help regulate protein levels in the human body. miRNAs target the 3’ untranslated region (UTR) of mRNAs, leading to translational repression or mRNA degradation depending on the degree of complementarily between the miRNA and its target [1,2]. Small non-coding RNAs have a role in numerous human diseases and biological processes including cell differentiation [3], cancer [4,5,6], and immune response [7].
Increasing evidence supports the role of miRNAs in hematopoiesis. Guo et al. showed that miR-125a can increase hematopoietic stem cell number by 8-fold in vivo.8 Erythropoiesis is promoted by miR-451[9] and inhibited by miR-24 [10], miR-221 [11] and miR-222 [11]. MiR-223 negatively regulates granulocyte differentiation and activation [12], while the miR 17-5p-20a-106a cluster controls monocyte differentiation [13]. MiRNAs can also regulate megakaryopoiesis. K562 cells overexpressing miR-155 showed reduced megakaryocyte differentiation [14]. Also, miR-146a may modulate megakaryopoiesis, but there is conflicting evidence on how it may direct the process [15,16,17]. Overexpression of miR-34a in human CD34+ cells significantly increased megakaryocyte colony forming number [18]. Forced expression of miR-150 enhanced megakaryocyte differentiation both in vitro and in vivo at the expense of erythroid fate [19]. In fact, Barroga et al. showed that thrombopoietin (TPO) increased miR-150 expression in UT7/TPO cells, which supports the critical role of miR-150 in megakaryopoiesis [20].
The identification of candidate miRNAs affecting hematopoiesis have primarily relied on miRNA profiling analyses. We used an alternative approach of transfecting miRNA mimics into human hematopoietic progenitor cells to identify effects on the erythroid, myeloid, and megakaryocyte lineages. By using human embryonic stem cell (hESC)-derived progenitor cells, a library of 466 miRNA mimics was tested in a 96-well format using flow cytometry as a read-out to assess the various differentiated cell lineages. A new enhancer of megakaryopoiesis, miR-105, was identified using this approach. This finding was extended to show that this miR also enhances human CD34+ cell-derived megakaryopoiesis and c-Myb has been identified as a putative downstream target and mechanism for this effect.
MATERIALS AND METHODS
hESC differentiation
A monolayer differentiation protocol, similar to the one used by Mills et al. [21], was used to produce a more synchronous hematopoietic progenitor population from hESCs. Prior to the induction of differentiation, H9 hESCs (2-4 × 105/well) were feeder depleted by culturing on Matrigel-coated wells (BD Biosciences) in 6-well tissue culture plates (Falcon #3046) in human embryonic stem cell (hESC) maintenance medium for 24 to 48 hours or until the cells reached ~70% confluence. When the cells were ready for differentiation, one well was harvested for cell count and pluripotency assessment by analysis of the surface markers, SSEA3 and SSEA4 (>90% co-expression). Three different base media were used in the differentiation protocol and all were supplemented with 2 mM glutamine, 50 μg/mL ascorbic acid (Sigma,), 150 μg/mL transferrin (Roche Diagnostics) and 0.4 μM monothioglycerol (MTG) (Sigma). The base media were RPMI (Invitrogen), StemPro-34 (SP-34) (Invitrogen), and serum free differentiation medium (SFD). SFD [22] contains 75% Iscove's modified Dulbecco's medium (Invitrogen) and 25% Ham's F12 media (Invitrogen) supplemented with 0.5× of both N2 and B27 (without retinoic acid) supplements (Invitrogen), penicillin, streptomycin, 0.05% BSA, 2 mM glutamine (Invitrogen), 0.5 mM ascorbic acid (Sigma) and 4.5 × 10−4 M 1-thioglycerol. Cultures were maintained at 37°C in an environment of 5% CO2, 5% O2, and 90% N2. The medium and cytokines that were used each day of differentiation are shown in supplemental Table 1. Wnt3a was replaced with CHIR (glycogen synthase kinase inhibitor, Cayman Chemical Company, Ann Arbor, MI) in our protocol and similar results in hemotopoietic progenitor formation were observed. CHIR was diluted immediately before use from a 10 mM stock solution. Fresh media mixes (2 mL/well) were added to the 6-well plate cultures daily.
Extensive cell death occurred up to day 2 of differentiation followed by the formation of an endothelial-like stromal layer. By day 6, cellular proliferation was apparent and the volume of medium was increased to 4 mL/well. By day 7, rounded clusters of cells resembling EB-like structures appeared attached to the stromal layer. Between days 7 and 9, single cells shedding off of the adherent layer were collected for further analysis. During differentiation, cells in the adherent layer were analyzed for surface markers by dissociation into single cells using 0.25% trypsin-EDTA (1 mL/well, 5 minutes at room temperature).
Day 8 cells were transfected with miR mimics. Post transfection, cells were cultured in SFD medium containing 1X penicillin/streptomycin, ascorbic acid (50 μg/mL) (Sigma), monothioglycerol (0.4 μM) (Sigma) and the following human cytokines (all from R&D systems, unless specified otherwise): bFGF (100 ng/ml), SCF (100 ng/ml), Flt3 ligand (5 ng/ml), IL-6 (10 ng/ml) and EPO (5 U/ml) (Amgen).
Human peripheral blood CD34+ differentiation
Peripheral blood (PB) CD34+ cells from healthy adult G-CSF mobilized donors were obtained from Fred Hutchinson Cancer Research Center (Seattle, WA). Prior to transfection, cells were thawed and washed in PBS (containing 1% fetal bovine serum) and maintained overnight in hematopoietic stem/progenitor cell expansion medium [23] comprising StemSpan SFEM (StemCell Technologies), 1X penicillin/streptomycin and the following human cytokines (all from R&D systems): TPO (20 ng/ml), IL-6 (10 ng/ml), IL-3 (10 ng/ml), Flt3 ligand (50 ng/ml) and SCF (100 ng/ml). Immediately post transfection, cells were placed in megakaryocyte differentiation medium [24] containing 80% homemade IMDM, 20% BIT9500 serum substitute (StemCell Technologies), 1X penicillin/streptomycin, human LDL (40 μg/ml) (StemCell Technologies), 0.55 mM β-mercaptoethanol and the following human cytokines (all from R&D systems): SCF (1 ng/ml), TPO (30 ng/ml), IL-9 (13.5 ng/ml) and IL-6 (7.5 ng/ml). 10 L of homemade IMDM was prepared with IMDM powder (Gibco # 12200-069), 100 ml of 100X penicillin/streptomycin, 30.24 g sodium bicarbonate (Sigma) and tissue culture grade water (Mediatech). Medium was replaced on Day 1, 4, 7 and 10. Cells were cultured at 37°C in 5% CO2.
MiRNA 96-well plate screening
The Pre-miR™ miRNA precursor library- Human V3 (Ambion) was used for 96-well plate screening. For further studies, Pre-miR™ miRNA precursors, mirVana™ miRNA mimics, Silencer® Select siRNAs, and negative controls were used (all from Ambion). For the miRNA mimic screen, cells were transfected using the 96-well Shuttle™ System (Lonza) and the P3 Primary Cell 96-well Nucleofactor ™Kit (Lonza). Program 96-E0-100 on the software was used for transfections. Unless specified, the manufacturer's protocol was followed. A total of 2-3 × 104 cells were transfected with 850 nM of miRNA mimic or 0.4 μg pmaxGFP vectors in each well. For further studies, each sample was transfected in a single cuvette using the Nucleofactor™ II device (Amaxa) and the Human CD34 Cell Nucleofector™ Kit (Lonza). For each sample, 1-3 × 105 cells were transfected with 1-2 μM miRNA mimic or 2 μg pmaxGFP vector. At 4-5 days post transfection, cells were stained and directly assayed in 96-well plates using the C6 flow cytometer (Accuri) equipped with a 96-well C-Sampler (Accuri). Data were analyzed on the CFlow Plus software (Accuri). The following antibodies were used for cell surface marker detection: FITC anti-CD18 (Biolegend), FITC anti-CD41 (Biolegend), PE/Cy7 anti-CD41 (Biolegend), Alexa 488® anti-SSEA-3(Biolegend), Alexa Fluor® 647 anti-SSEA-4 (Biolegend), PE anti-CD42a (BD Pharmingen™) and APC anti-CD235a (BD Pharmingen™). Colony forming assays and qPCR were performed as previously described.[12] (see Supplemental Methods)
Luciferase reporter assay
Full length 3’ UTR of MYB was inserted downstream of a Gaussia luciferase reporter gene in the pEZX-MT05 vector (GeneCopoeia). The vector also contained a constitutively expressed secreted alkaline phosphatase (seAP) reporter gene, which served as an internal control for transfection normalization. HEK-293 cells were seeded at 32,000 cells/well in 96-well plates and allowed to spread overnight. Cells were then co-transfected by adding 0.35 μL/well of Lipofectamine 2000 (Invitrogen) complexed with 0.075 μg/well of vector and 6.7 nM of miR mimic or negative controls. Culture medium was assayed for luciferase and alkaline phosphatase 48 hours post transfection using the Secrete Pair™ Dual Luminescence assay kit (GeneCopoeia).
Western Blot Analysis
Cell lysates were extracted using RIPA lysis buffer system (Santa Cruz Biotech.) supplemented with PMSF, protease inhibitor cocktail and sodium orthovanadate. Protein samples were resolved by SDS–PAGE and transferred to nitrocellulose membranes, which were blocked in Odyssey Blocking Buffer (OBB; LI-COR). The blots were visualized using a LI-COR Odyssey Infrared Imaging System. MYB and GAPDH antibodies were from Santa Cruz Biotech.
Statistical analysis
Student's two tailed t-test was used for all statistical analysis. A p-value < 0.05 was considered as statistically significant.
RESULTS
miRNA screen development
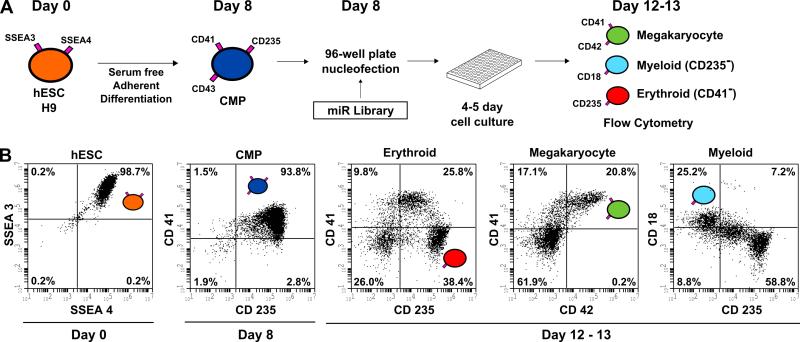
To identify miRNAs that regulate hematopoiesis, a high throughput screening assay was optimized for transfecting siRNAs into human progenitor cells derived from hESCs. Nucleofection was used as the method of transfection since it had been shown to efficiently deliver plasmid vectors and siRNA to hematopoietic cells [25,26]. Progenitor cells derived from hESCs were used because they can easily be transfected, pools of identical cells can be cryopreserved, and these cells can be expanded in liquid culture to the erythroid, myeloid, and megakaryocyte lineages. The high throughput screening assay was performed in a 96-well format using flow cytometry as a read-out to quantify the specific cell lineages. A schematic of the assay and flow profiles of the hESCs during various stages of differentiation are shown in Figure 1. The surface markers used to assess undifferentiated hESCs (SSEA3+, SSEA4+), day 8 progenitor cells (CD41+, CD235+), and day 12-13 megakaryocyte (CD41+, CD42+), myeloid (CD18+), and erythroid (CD235+, CD41−) lineages are shown in Figure 1A along with the 96-well format. The flow profiles of undifferentiated, progenitor, and expanded cell lineages showing expected percentages are shown in Figure 1B. We found that transfection of 2-3 x 104/well progenitor cells (of which >80% co-express the markers CD41+CD235+) resulted in efficiencies of >50% using a GFP-expressing plasmid vector and >80% using fluorescently labeled siRNA, respectively (supplemental Figure 1). The post-transfection cells were differentiated in a cytokine mixture consisting of bFGF, SCF, IL6, Flt3 and EPO for 4-5 days followed by four color 96-well flow cytometry to identify the cell lineages. Using this protocol, we screened 466 miRNA mimics in six 96-well plates. Each miRNA mimic was screened once.
Figure 1. MicroRNA screen in differentiating human embryonic stem cells.
(A) A miRNA mimic library was screened in hematopoietic progenitors derived from the H9 human embryonic stem cell line. hESCs (SSEA-3+SSEA-4+) were plated on matrigel on Day 0 and were subjected to a 8-day serum free differentiation protocol in a 6-well plate. CMPs (CD41+CD235+CD43+) emerge from the adherent layer into suspension as single cells on Days 7, 8 and 9. Day 8 CMPs were transfected via electroporation with miRNA mimics in 96-well plates and then cultured for 4 to 5 days after which erythroid (CD235+CD41−), megakaryocyte (CD41+CD42+) and myeloid (CD18+CD235−) populations were assessed by 96-well flow cytometry. (B) Representative flow cytometry plots are shown.
miRNA screen and validation
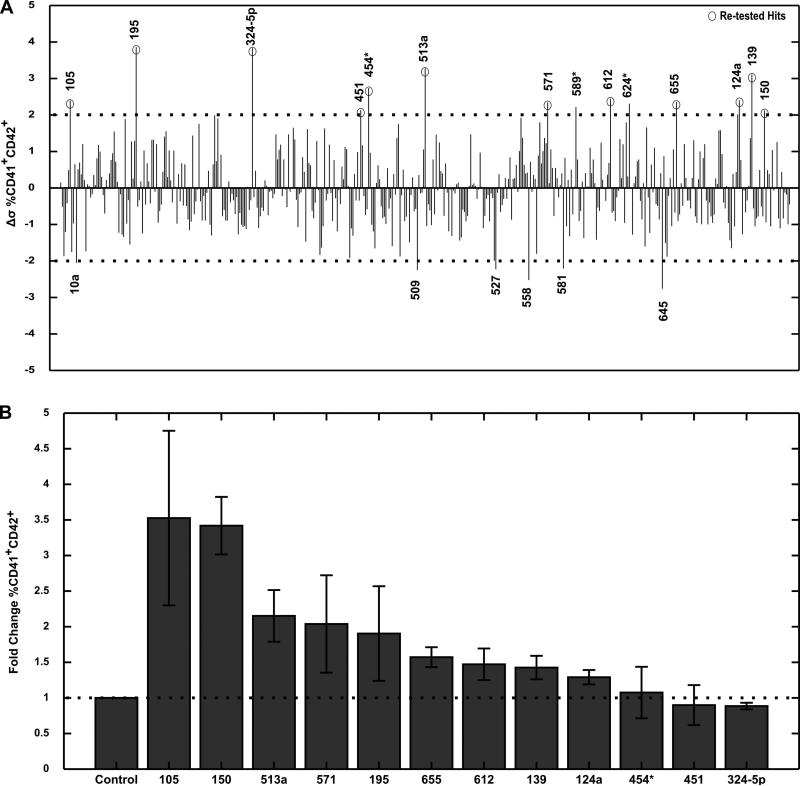
To compare the flow cytometry data obtained from all of the plates, we assessed the data for each hematopoietic lineage population for each miR in terms of deviation (Δσ) from the plate mean. Since each plate was screened on a separate day, an expected degree of plate to plate variability was observed. This analysis, based on individual plate means, enabled us to compare the data from all 466 miRs on a single scale despite a degree of variability between plates in terms of plate mean populations and standard deviation. The deviation for the megakaryocyte population (∆σ %CD41+ CD42+miR) was defined:
∆σ %CD41+ CD42+miR is the deviation in %CD41+ CD42+ population from the mean for a particular miR. %CD41+ CD42+miR is the raw %CD41+ CD42+ population for the miR from flow cytometry. %CD41+ CD42+plate is the mean of %CD41+ CD42+ population for all miRs in the plate. σ %CD41+ CD42+plate is the standard deviation of %CD41+ CD42+ population for all miRs in the plate. Similar analyses were done for erythroid and myeloid populations. Using this metric, we assessed our screen data for miRNAs that were both potential enhancers and inhibitors of all three hematopoietic lineages (Figure 2A, supplemental Figure 2). We next ranked the top 20 miRNAs that either increased or decreased hematopoietic lineage populations (supplemental Table 2 and 3). MiR-150, a well established enhancer of megakaryopoiesis, was amongst the top enhancers of the megakaryocyte lineage. Detection of miR-150 as a hit confirmed that our screen was a valid platform for identifying miRNA regulators of hematopoietic lineages. The top 12 miRNAs that caused the greatest increase in megakaryocyte populations were retested in the 96-well format (Figure 2B). MiR-105 and miR-150 had the greatest effect, enriching for CD41+CD42+ megakaryocytes by greater than 3-fold. MiR-513a, miR-571 and miR-195 had a mild effect on enhancing megakaryocyte population, while the rest of miRNAs were seen to be either very weak enhancers or false positives. The potential miRNA enhancers of erythroid and myeloid populations, as well as the miRNA inhibitors of all three lineages, that were identified as hits from our screen were not further pursued, as those experiments were beyond the scope of the current study. In order to validate and scale up the system, we focused on the newly identified miR-105 and the previously identified miR-150 that enhanced megakaryopoiesis.
Figure 2. microRNA screen results for regulators of megakaryocyte populations.
466 miRNA mimics were screened in hESC derived Day 8 CMPs in six 96-well plates. (A) The deviation for each miRNA mimic from the mean plate value of %megakaryocyte (CD41+CD42+) population is shown. The top miRNA enhancers and inhibitors are labeled. Dotted lines represent 2 standard deviations. (B) The top hits were re-tested in replicates. The fold change in % megakaryocyte population as a result of miRNA mimic transfection is shown. Data are representative of 2 to 5 independent transfections.
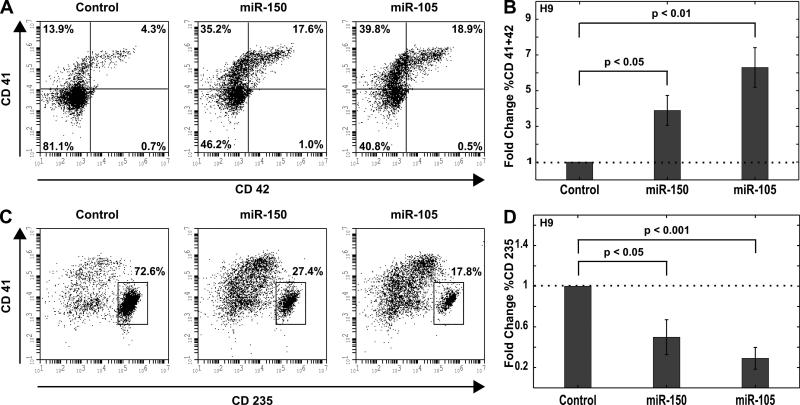
To validate that miR-105 affects megakaryopoiesis, the system was scaled up in which cells were plated in a 24-well plate and transfected with 2 μM miRNA mimic. Control cells were transfected with a non-specific miRNA mimic. Cells were cultured for 4 days post-transfection followed by flow cytometry to determine the percent of CD41+CD42+ cells. Cells transfected with either miR-105 or miR-150 generated higher percentages of CD41+CD42+ cells with a significant fold-increase over the control cell population (Figure 3A and B). Cells transfected with miR-105 showed a 6-fold increase in enrichment for megakaryocytes compared to cells transfected with a non-specific control. Interestingly, both miR-105 and miR-150 diminished the respective erythroid populations by greater than 50% (Figure 3C and D), suggesting that both miR-105 and miR-150 may play a role in reciprocally regulating the megakaryocyte and erythroid lineages in this progenitor population.
Figure 3. miR-105 and miR-150 enhance megakaryocyte population from H9 hESC derived hematopoietic progenitors.
Day 8 CMPs were transfected with 2 μM miR-105 mimic, miR-150 mimic, or a non-specific negative control. Cell populations were analyzed by flow cytometry on Day 12. (A) Representative flow cytometry plots for megakaryocyte (CD41+CD42+) population are shown. (B) The fold change in % megakaryocyte population as a result of miR mimic transfection is shown. Data are representative of 3 to 4 independent transfections. (C) Representative flow cytometry plots for erythroid (CD235+CD41−) population are shown. (D) The fold change in % erythroid population as a result of miR mimic transfection is shown. Data are representative of 3 to 4 independent transfections.
The effect of miR-150 on enriching the megakaryocyte lineage was expected, as it was previously shown to have a similar effect in adult hematopoiesis [19]. Our observations extend this result to in vitro yolk sac hematopoiesis. MiR-105 (distinct from miR-150) emerged as a novel enhancer of the megakaryocyte lineage from our results. In fact, miR-105 overexpression yielded a higher enriched population of megakaryocytes as well as a lower erythroid population compared to miR-150 (Figure 3).
MiR-105 enhances megakaryocyte production in adult CD34+ cells
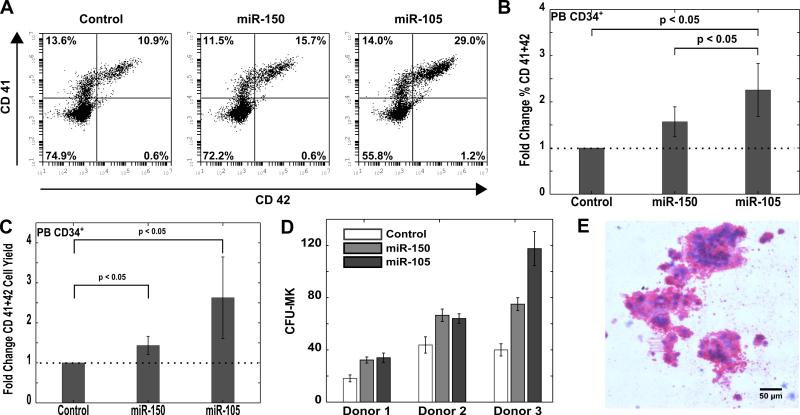
We next investigated whether miR-105 could enhance the megakaryocyte lineage in adult CD34+ cells. Cells from mobilized adult donors enriched in CD34+ population (>90%) were nucleofected with the miR-105 mimic, miR-150 mimic, or a non-specific negative control. Cells were cultured for 14 days in medium that had previously been shown to induce megakaryocyte differentiation [24]. Using flow cytometry, we assayed the samples for CD41+CD42+ megakaryocyte-like cells on days 4, 7 and 10. Cells transfected with miR-150 and miR-105 showed an enhancement in the megakaryocyte population on day 4 (data not shown) and day 7 (Figure 4A and B). In addition to determining megakaryocyte enrichment, the yield of megakaryocytes was also determined. To eliminate effects of cell death from nucleofection on the yield calculations, cells were counted and plated post transfection. We found a 1.5-fold and 2-fold enhancement of megakaryocyte yield from cells transfected with miR-150 and miR-105, respectively (p < 0.05) (Figure 4C). These data confirm that expression of miR-105, like miR-150, increases megakayocyte number, and that enrichment of megakaryocytes was not simply a result of diminished proliferation or increased cell death of other lineages.
Figure 4. miR-105 enhances megakaryocyte production in vitro from adult CD34+ cells.
CD34+ cells isolated from the peripheral blood of mobilized adult donors were transfected with 2 μM miR-105 mimic, miR-150 mimic or a negative control. Cells were cultured for 14 days post transfection in a megakaryocyte lineage promoting medium containing TPO, SCF, IL-6 and IL-9. Cells were assayed for colony forming units 4 days post transfection. (A) Representative flow cytometry plots for megakaryocyte (CD41+CD42+) population on Day 7 are shown. (B) The fold change in % megakaryocyte population as a result of miR mimic transfection is shown. Data are representative of 5 independent transfections. (C) The fold change in megakaryocyte yield as a result of miR mimic transfection is shown. Megakayocyte yield is defined as the number of CD41+CD42+ in culture on Day 7 per CD34+ cell seeded within 24 hrs post transfection. Data are representative of 5 independent transfections. (D) Megakaryocyte colony forming units per 2500 transfected cells for 3 separate donors are shown. Data are representative of 3-4 plates of colony assay. (E) Representative megakaryocyte colony image of miR-105 transfected cells is shown.
We next examined whether the miR-105 was directing hematopoietic progenitor differentiation towards megakaryocytes, or if its effect on enhanced megakaryocyte production was a result of increased proliferation or survival of committed megakaryocyte progenitors. To investigate this, we used a collagen-based assay to determine megakaryocyte colony forming potential of transfected cells. Transfection with miR-105 resulted in a 1.5 to 3-fold enhancement on megakaryocyte colony numbers compared to the control in all 3 donors (Figure 4D). MiR-150 served as a positive control and increased megakaryocyte numbers as previously reported [19]. Polyploid megakaryocytes were produced from CD34+ cells transfected with miR-105 (Figure 4E). The ploidy of megakaryocytes generated in culture from miR-105 transfected PB CD34+ cells was similar to that of the control (supplemental Figure 3).
To test if miR-105 had a reciprocal effect on commitment to other hematopoietic lineages, we performed colony forming assays on transfected samples to detect erythroid and myeloid colony forming units. Transfection with miR-105 or miR-150 did not significantly alter erythroid or granulocyte-macrophage colony numbers (supplemental Figure 4). Interestingly, our findings differ from those of a previous study, which suggested that miR-150 reduces erythroid colony forming potential [19]. However, that study was conducted with mouse bone marrow cells transduced with miR-150 retrovirus. Transient transfection of miR-105 and miR-150 did not affect erythroid and myeloid commitment in human CD34+ cells.
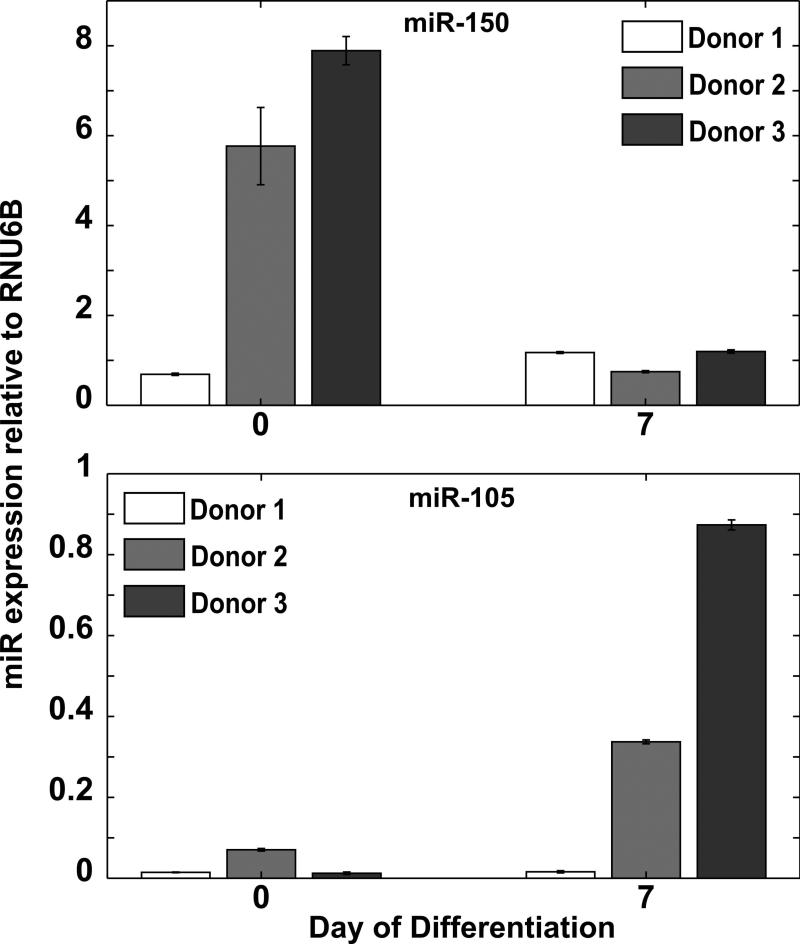
MiR-105 is endogenously expressed in differentiating hematopoietic cells
To determine if miR-105 is present during normal megakaryopoiesis, we examined its expression in hematopoietic cells. CD34+ cells from mobilized donors were cultured in megakaryocyte conditions and data from three separate donors showed that miR-105 was endogenously expressed upon megakaryocyte induction with variable timing depending on the particular donor sample (Figure 5). For two of the donors, miR-105 expression levels were higher on Day 7 compared to Day 0. Interestingly, for the same two donors, miR-150 expression was higher on Day 0. These observations suggest that miR-105 and miR-150 may be regulating two separate stages of megakaryopoiesis.
Figure 5. miR-105 is expressed endogenously in differentiating CD34+ cells.
CD34+ cells isolated from the peripheral blood of mobilized adult donors were cultured in megakaryocyte conditions. Endogenous miRNA expression was assayed by quantitative RT-PCR on Day 0 and Day 7 for 3 different donors. Expression levels were measured relative to RNU6B endogenous control.
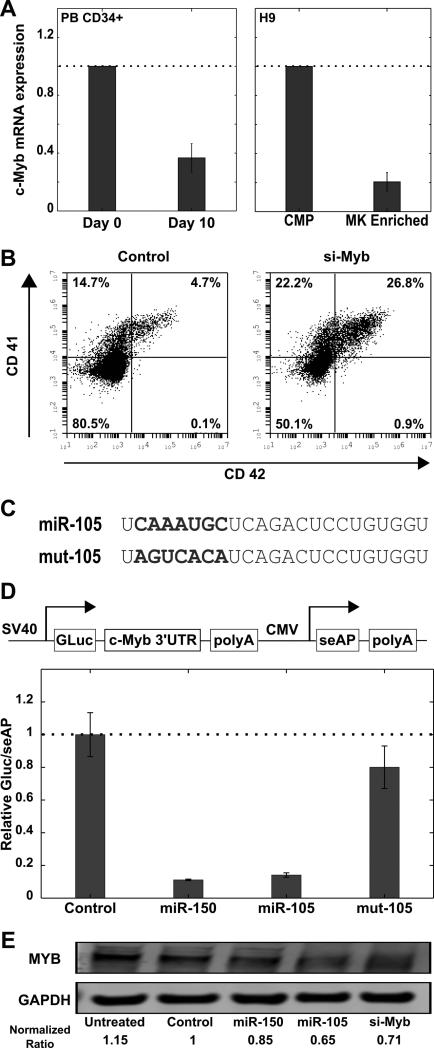
MiR-105 regulates MYB expression
In silico predictions based on miRNA seed match with 3’ UTR of mRNA identified the hematopoietic transcription factor MYB as a potential target of miR-105.27,28 The 3’UTR of the MYB messenger RNA has a single site that is complementary to the seed region of miR-105 (supplemental Figure 5). Previous studies have shown that miR-150 enhances megakaryopoiesis in part by targeting MYB [19]. c-Myb mRNA levels were down-regulated > 60% in CD34+ cells and hESC derived CMPs that were cultured in megakaryocyte conditions (Figure 6A). SiRNA mediated knockdown of c-Myb mRNA in hESC derived CMPs led to an enrichment of megakaryocyte populations comparable to that of miR-105 and miR-150 (Figure 6B).To validate that MYB was indeed a direct target of miR-105, we used a dual reporter assay to investigate the interaction of miR-105 with the MYB 3’UTR. The full length MYB 3’UTR was inserted downstream of Gaussia luciferase (GLuc) in the pEZX-MT05 vector (Figure 6D). The vector also expressed secreted alkaline phosphotase (seAP) as an internal control for transfection normalization. To further verify the specificity of the miR-105 interaction with MYB, we custom designed a miR-105 mimic with the seed region scrambled (Figure 6C). HEK-293 cells were cotransfected with the vector and miR mimics, and the GLuc/seAP activity was measured 48 hr post-transfection. MiR-105 reduced GLuc activity by more than 80%, comparable to that of miR-150 which served as a positive control (Figure 6D). However, the mutated miR-105 mimic (mut-105) did not have an effect on the signal. Additionally, MYB protein knockdown was observed in K-562 human leukemia cells transfected with miR-105 (Figure 6E). These results indicate MYB is a direct target of miR-105.
Figure 6. miR-105 regulates c-myb expression.
(A) CD34+ cells isolated from the peripheral blood of mobilized adult donors and CMPs derived from hESCs were cultured in megakaryocyte conditions. C-Myb expression was assessed by quantitative RT-PCR. 18s rRNA was used as the endogenous control (B) Day 8 hESC derived CMPs were transfected with 2 μM c-Myb siRNA or a negative control siRNA. Cells were analyzed by flow cytometry 72 hours post transfection. Representative flow cytometry plots for megakaryocyte (CD41+CD42+) population are shown (C) Mature sequences of miR-105 and miR-105 with its seed region scrambled are shown (D) HEK-293 cells were cotransfected with 0.075 μg/ml reporter plasmid and 6.7 nM miR mimic or non-specific control in 96 well plates. Culture medium was assayed 48 hr post transfection. The dual reporter vector construct is shown. The relative gaussia luciferase to secreted alkaline phosphotase signal is shown. The signal for luciferase/alkaline phosphotase for the control mimic was set to a value of 1. Data are representative of 4 separate transfections in 96-well plate assay. (E) K-562 cells were transfected with control miRNA mimic, miR-150, miR-105 and siRNA against c-Myb. MYB and GAPDH protein levels were detected with western blot analysis.
DISCUSSION
In this study, we have identified miR-105 as a novel enhancer of megakaryopoiesis. Our findings are the first to show that this miRNA is functional in hematopoiesis. Transfection of miR-105 led to enrichment of megakaryocyte populations from hematopoietic progenitors derived from hESCs and adult CD34+ cells, showing that miR-105 is functional in both primitive and definitive hematopoiesis. In addition to enriching for megakaryocytes, miR-105 increased megakaryocyte production and megakaryocyte colony formation in adult CD34+ cells. Previous studies have shown that miR-150 and miR-34a enhance megakaryopoiesis in part by targeting MYB [18,19]. In silico target predictions along with 3’ UTR reporter and western blot assays confirmed that miR-105 may in part drive megakaryopoiesis by silencing translation of the MYB mRNA. To identify additional mechanisms of miR-105 mediated regulation of megakaryopoiesis, we conducted siRNA mediated knockdown of some of the top predicted targets of miR-105 (supplemental Table 4) from multiple databases[27,28,29,30]: TJP1, FLRT2, SUZ12 and TBL1XR1 in hESC derived hematopoietic progenitors. However, we did not observe any significant change in megakaryocyte populations as a result of siRNA transfection (data not shown). MiR-105 has been shown to play a role in modulating TLR2 protein expression in human oral keratinocytes [31], and controlling human ovarian cell proliferation [32]. Megias et al. showed that stimulation of hematopoietic progenitors with TLR2 agonists promotes their differentiation towards macrophages [33]. However, TLR2 mRNA expression did not change significantly in adult CD34+ cells transfected with miR-105 (supplemental Figure 6). Additionally surface protein levels of TLR2 were not observed in these cells (data not shown).
Throughout our study, we compared the effect of miR-105 in enhancing megakaryopoiesis to that of the well established miR-150. MiR-105 surpassed miR-150 in terms of enriching for megakaryocytes, megakaryocyte yield and megakaryocyte colony formation. Interestingly, reporter assays confirmed that both these miRNAs may in part function through the same mechanism by silencing translation of the MYB mRNA. The MYB 3’UTR has four complementary sites for miR-150, but only a single site for miR-105 (supplemental Figure 5). This suggests that miR-105 could be down regulating other unidentified targets. We showed that miR-150 is highly expressed in early hematopoietic progenitors while miR-105 expression increases at a later stage as the mature megakaryocyte population starts increasing. These observations indicate miR-150 may serve as the primary regulator of MYB at an early stage of megakaryopoiesis, and this role may switch over to miR-105 as the cells start maturing. Interestingly, our observations are different from those of Lu et al [19] who showed increased expression of miR-150 in megakaryocyte populations relative to megakaryocyte-erythroid progenitors from sorted cord blood samples at harvest. In contrast, our work used peripheral blood adult CD34+ cells that were cultured in the presence of megakaryocyte lineage promoting cytokines. Our results indicate that transient overexpression of either miR-105 or miR-150 in adult CD34+ cells is sufficient to cause increased megakaryocyte production and colony forming potential. These observations indicate that miR-105 and miR-150 may direct lineage fate in a switch like manner. Further studies comparing transient and stable overexpression of these miRNAs are needed to confirm this hypothesis.
Our study demonstrates that a miRNA mimic screen is a valuable technique to identify regulators of stem cell differentiation. Previous studies have primarily relied on miRNA profiling in different lineage populations to identify regulators of lineage fate [17,19,34]. These studies overlook miRNAs whose expression levels do not change significantly as the cell differentiates as well as miRNAs that are expressed at very low levels or absent at stages of differentiation. MiRNA functional screens have been performed in cancer biology to discover candidate miRNAs that can be used as therapeutic tools [35,36]. To our knowledge, we report the first gain of function miRNA screen to identify regulators of hematopoietic differentiation. We conducted our screen in multipotent hematopoietic progenitors derived from hESCs primarily because (1) these cells can differentiate into mature lineages within a relatively short time period (4-5 days) allowing transient miRNA overexpression to have an effect on fate determination and (2) there have been no previous reports on miRNAs regulating megakaryocyte differentiation in yolk sac hematopoiesis. We also demonstrated that these CMPs can be transfected efficiently with plasmid and small RNAs using nucleofection, in both a single cuvette and a high throughput 96-well format. Utilizing a simple but robust four color 96-well flow cytometry assay that we had previously developed [37], we were able to rapidly assess mature hematopoietic lineage specific surface marker expression. Using our optimized high throughput screening protocol, we generated a dataset showing the effect of 466 miRNAs on the erythroid, myeloid and megakaryocyte lineage potential of multipotent progenitors in a model of yolk sac hematopoiesis. In addition to identifying enhancers, our data also reveal miRNAs that inhibit the differentiation of progenitors towards the three hematopoietic populations. Some of the previously established miRNA regulators of hematopoiesis were identified in the screen (supplemental Table 5), thereby validating the effectiveness of our screening platform. Interestingly, most of these previous studies were done in CD34+ cells. These similarities imply that some miRNA functions may translate between hematopoietic systems. However, this may not be true for all miRNAs. For example, miR-221 or miR-155 overexpression did not decrease erythroid and megakaryocyte populations respectively in our system (data not shown) as previously reported [18,14]. These inconsistencies can be attributed to the differences between primitive and definitive hematopoiesis. It is also possible, that some miRNAs could have been unidentified, as they may have been false negatives in our screen.
Human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs) are a potentially valuable source for large-scale production of megakaryocytes and platelets, which can be used in platelet transfusions [38,39]. Recent studies have shown that functional megakaryocytes and platelets can be generated from hESCs and hiPSCs by optimizing culture conditions [40,41,42]. Currently, yields of ~ 60 megakaryocytes per input hESC have been reported [40]. Our work supplements these established protocols by suggesting that overexpression of miR-105 and miR-150 could further increase megakaryocyte production from hESCs. However, our approach of introducing these small RNAs by electroporation is not feasible for large scale production of megakaryocytes. Employing established viral vector delivery systems for stable or inducible overexpression of these miRNAs in hESCs may serve as more robust alternatives. Another approach could involve developing hESC or hiPSC lines that can be reversibly induced to overexpress miR-105 or miR-150, as the cells start differentiating towards the hematopoietic lineage. Such studies will help us move closer towards the development of donor-independent platelet transfusions.
CONCLUSION
miR-105 is an enhancer of megakaryopoiesis in both hESC derived hematopoietic progenitors and adult CD34+ cells. The hematopoietic transcription factor c-Myb is downregulated by miR-105. High-throughput miRNA transfection combined with 96-well flow cytometry is a novel and effective system to identify small RNA regulators of stem cell differentiation.
Supplementary Material
ACKNOWLEDGEMENTS
We thank Lonza Inc. and Schellenberg lab for the use of the Nucleofactor™ II device and 96-well Shuttle™ System respectively for the purpose of our screen. We thank the Lazzara lab for their help with western blot analysis. This work was supported by the National Institutes of Health (R01 HL-103419 (S.L.D.)) and NIDDK Hematopoietic Center of Excellence.
This work was supported by the National Institutes of Health (R01 HL-103419 (S.L.D.)) and NIDDK Hematopoietic Center of Excellence (P30DK090969).
Footnotes
Author contributions: V.K.: Conception and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing; P.P.: Collection and/or assembly of data; M.M.: Collection and/or assembly of data; D.L.F.: Provision of study materials, discussion of results, manuscript writing; P.G.: Provision of study materials, discussion of results, manuscript writing; S.L.D.: Conception and design, manuscript writing, financial support, final approval of manuscript
Conflict-of-interest disclosure: The authors declare no competing financial interests.
REFERENCES
- 1.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 2.Valencia-Sanchez MA, Liu J, Hannon GJ, Parker R. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev. 2006;20:515–524. doi: 10.1101/gad.1399806. [DOI] [PubMed] [Google Scholar]
- 3.Gangaraju VK, Lin H. MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol. 2009;10:116–125. doi: 10.1038/nrm2621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Calin GA, Dumitru CD, Shimizu M, et al. Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci U S A. 2002;99:15524–15529. doi: 10.1073/pnas.242606799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.He L, Thomson JM, Hemann MT, et al. A microRNA polycistron as a potential human oncogene. Nature. 2005;435:828–833. doi: 10.1038/nature03552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lu J, Getz G, Miska EA, et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435:834–838. doi: 10.1038/nature03702. [DOI] [PubMed] [Google Scholar]
- 7.Gantier MP, Sadler AJ, Williams BR. Fine-tuning of the innate immune response by microRNAs. Immunol Cell Biol. 2007;85:458–462. doi: 10.1038/sj.icb.7100091. [DOI] [PubMed] [Google Scholar]
- 8.Guo S, Lu J, Schlanger R, et al. MicroRNA miR-125a controls hematopoietic stem cell number. Proc Natl Acad Sci U S A. 2010;107:14229–14234. doi: 10.1073/pnas.0913574107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dore LC, Amigo JD, Dos Santos CO, et al. A GATA-1-regulated microRNA locus essential for erythropoiesis. Proc Natl Acad Sci U S A. 2008;105:3333–3338. doi: 10.1073/pnas.0712312105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Q, Huang Z, Xue H, et al. MicroRNA miR-24 inhibits erythropoiesis by targeting activin type I receptor ALK4. Blood. 2008;111:588–595. doi: 10.1182/blood-2007-05-092718. [DOI] [PubMed] [Google Scholar]
- 11.Felli N, Fontana L, Pelosi E, et al. MicroRNAs 221 and 222 inhibit normal erythropoiesis and erythroleukemic cell growth via kit receptor down-modulation. Proc Natl Acad Sci U S A. 2005;102:18081–18086. doi: 10.1073/pnas.0506216102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Johnnidis JB, Harris MH, Wheeler RT, et al. Regulation of progenitor cell proliferation and granulocyte function by microRNA-223. Nature. 2008;451:1125–1129. doi: 10.1038/nature06607. [DOI] [PubMed] [Google Scholar]
- 13.Fontana L, Pelosi E, Greco P, et al. MicroRNAs 17-5p-20a-106a control monocytopoiesis through AML1 targeting and M-CSF receptor upregulation. Nat Cell Biol. 2007;9:775–787. doi: 10.1038/ncb1613. [DOI] [PubMed] [Google Scholar]
- 14.Georgantas RW, III, Hildreth R, Morisot S, et al. CD34+ hematopoietic stem-progenitor cell microRNA expression and function: a circuit diagram of differentiation control. Proc Natl Acad Sci U S A. 2007;104:2750–2755. doi: 10.1073/pnas.0610983104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Labbaye C, Spinello I, Quaranta MT, et al. A three-step pathway comprising PLZF/miR-146a/CXCR4 controls megakaryocytopoiesis. Nat Cell Biol. 2008;10:788–801. doi: 10.1038/ncb1741. [DOI] [PubMed] [Google Scholar]
- 16.Starczynowski DT, Kuchenbauer F, Argiropoulos B, et al. Identification of miR-145 and miR-146a as mediators of the 5q- syndrome phenotype. Nat Med. 2010;16:49–58. doi: 10.1038/nm.2054. [DOI] [PubMed] [Google Scholar]
- 17.Opalinska JB, Bersenev A, Zhang Z, et al. MicroRNA expression in maturing murine megakaryocytes. Blood. 2010;116:e128–138. doi: 10.1182/blood-2010-06-292920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Navarro F, Gutman D, Meire E, et al. miR-34a contributes to megakaryocytic differentiation of K562 cells independently of p53. Blood. 2009;114:2181–2192. doi: 10.1182/blood-2009-02-205062. [DOI] [PubMed] [Google Scholar]
- 19.Lu J, Guo S, Ebert BL, et al. MicroRNA-mediated control of cell fate in megakaryocyte-erythrocyte progenitors. Dev Cell. 2008;14:843–853. doi: 10.1016/j.devcel.2008.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Barroga CF, Pham H, Kaushansky K. Thrombopoietin regulates c-Myb expression by modulating micro RNA 150 expression. Exp Hematol. 2008;36:1585–1592. doi: 10.1016/j.exphem.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mills JA, Wang K, Paluru P, et al. Clonal genetic and hematopoietic heterogeneity among human-induced pluripotent stem cell lines. Blood. 2013;122:2047–2051. doi: 10.1182/blood-2013-02-484444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gadue P, Huber TL, Paddison PJ, Keller GM. Wnt and TGF-beta signaling are required for the induction of an in vitro model of primitive streak formation using embryonic stem cells. Proc Natl Acad Sci USA. 2006;103:16806–16811. doi: 10.1073/pnas.0603916103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Boitano AE, Wang J, Romeo R, et al. Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science. 2010;329:1345–1348. doi: 10.1126/science.1191536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cortin V, Pineault N, Garnier A. Ex vivo megakaryocyte expansion and platelet production from human cord blood stem cells. Methods Mol Biol. 2009;482:109–126. doi: 10.1007/978-1-59745-060-7_7. [DOI] [PubMed] [Google Scholar]
- 25.von Levetzow G, Spanholtz J, Beckmann J, et al. Nucleofection, an efficient nonviral method to transfer genes into human hematopoietic stem and progenitor cells. Stem Cells Dev. 2006;15:278–285. doi: 10.1089/scd.2006.15.278. [DOI] [PubMed] [Google Scholar]
- 26.Banzon V, Ibanez V, Vaitkus K, et al. siDNMT1 increases γ-globin expression in chemical inducer of dimerization (CID)-dependent mouse βYAC bone marrow cells and in baboon erythroid progenitor cell cultures. Exp Hematol. 2011;39:26–36. doi: 10.1016/j.exphem.2010.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang X, El Naqa IM. Prediction of both conserved and nonconserved microRNA targets in animals. Bioinformatics. 2008;24:325–332. doi: 10.1093/bioinformatics/btm595. [DOI] [PubMed] [Google Scholar]
- 28.Wang X. miRDB: a microRNA target prediction and functional annotation database with a wiki interface. RNA. 2008;14:1012–1017. doi: 10.1261/rna.965408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120:15–20. doi: 10.1016/j.cell.2004.12.035. [DOI] [PubMed] [Google Scholar]
- 30.John B, Enright AJ, Aravin A, et al. Human MicroRNA Targets. PLoS Biol. 2004;2:e363. doi: 10.1371/journal.pbio.0020363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Benakanakere MR, Li Q, Eskan MA, et al. Modulation of TLR2 protein expression by miR-105 in human oral keratinocytes. J Biol Chem. 2009;284:23107–23115. doi: 10.1074/jbc.M109.013862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sirotkin AV, Laukova M, Ovcharenko D, Brenaut P, Mlyncek M. Identification of microRNAs controlling human ovarian cell proliferation and apoptosis. J Cell Physiol. 2010;223:49–56. doi: 10.1002/jcp.21999. [DOI] [PubMed] [Google Scholar]
- 33.Megias J, Yanez A, Moriano S, et al. Direct Toll-like receptor-mediated stimulation of hematopoietic stem and progenitor cells occurs in vivo and promotes differentiation toward macrophages. Stem Cells. 2012;30:1486–1495. doi: 10.1002/stem.1110. [DOI] [PubMed] [Google Scholar]
- 34.Garzon R, Pichiorri F, Palumbo T, et al. MicroRNA fingerprints during human megakaryocytopoiesis. Proc Natl Acad Sci U S A. 2006;103:5078–5083. doi: 10.1073/pnas.0600587103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nakano H, Miyazawa T, Kinoshita K, Yamada Y, Yoshida T. Functional screening identifies a microRNA, miR-491 that induces apoptosis by targeting Bcl-X(L) in colorectal cancer cells. Int J Cancer. 2010;127:1072–1080. doi: 10.1002/ijc.25143. [DOI] [PubMed] [Google Scholar]
- 36.Nakano H, Yamada Y, Miyazawa T, Yoshida T. Gain-of-function microRNA screens identify miR-193a regulating proliferation and apoptosis in epithelial ovarian cancer cells. Int J Oncol. 2013;42:1875–1882. doi: 10.3892/ijo.2013.1896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Outten JT, Cheng X, Gadue P, French DL, Diamond SL. A high-throughput multiplexed screening assay for optimizing serum-free differentiation protocols of human embryonic stem cells. Stem Cell Res. 2011;6:129–142. doi: 10.1016/j.scr.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 38.Lambert MP, Sullivan SK, Fuentes R, French DL, Poncz M. Challenges and promises for the development of donor-independent platelet transfusions. Blood. 2013;121:3319–3324. doi: 10.1182/blood-2012-09-455428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Takayama N, Eto K. Pluripotent stem cells reveal the developmental biology of human megakaryocytes and provide a source of platelets for clinical application. Cell Mol Life Sci. 2012;69(20):3419–3428. doi: 10.1007/s00018-012-0995-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lu SJ, Li F, Yin H, et al. Platelets generated from human embryonic stem cells are functional in vitro and in the microcirculation of living mice. Cell Res. 2011;21:530–545. doi: 10.1038/cr.2011.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Takayama N, Nishikii H, Usui J, et al. Generation of functional platelets from human embryonic stem cells in vitro via ES-sacs, VEGF-promoted structures that concentrate hematopoietic progenitors. Blood. 2008;111:5298–5306. doi: 10.1182/blood-2007-10-117622. [DOI] [PubMed] [Google Scholar]
- 42.Pick M, Azzola L, Osborne E, Stanley EG, Elefanty AG. Generation of megakaryocytic progenitors from human embryonic stem cells in a feeder- and serum-free medium. PloS One. 2013;8:e55530. doi: 10.1371/journal.pone.0055530. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.