Abstract
Objective: To investigate localization and distribution of integrin subunit β1, β2 and β3 and morphological changes of ligand-recepter binding in thrombi of acute pulmonary embolism (PE) patients and explore activation of circulated immune cells, inflammatory immune adherence and coagulation response in acute venous thrombosis. Methods: Thrombi were collected from patients with acute PE. Immunohistochemistry was done to detect the expression and distribution of integrin β1, β2 and β3 in cells within thrombi, and ligands of integrin subunit β1, β2 and β3 were also determined by immunohistochemistry within the thrombi. Results: 1) Acute venous thrombi were red thrombi composed of skeletons and filamentous mesh containing large amounts of red blood cells and white blood cells; 2) Integrin subunit β1, β2 and β3 were expressed on lymphocytes, neutrophils and platelets; 3) No expression of integrin β1 ligands: Laminin, Fibronectin, Collagen I or Collagen-II on lymphocytes; integrin β2 ligands including ICAM, factor X and iC3b are distributed on neutrophils, and ligand fibrinogen bound to neutrophils; integrin β3 was expressed on platelets which form the skeleton of thrombi and bound to fibrinogen to construct mesh structure; 4) Factor Xa was expressed on the filamentous mesh; 5) Filamentous mesh was fully filled with red blood cell dominant blood cells. Conclusion: Acute venous thrombosis is an activation process of circulated lymphocytes, neutrophils and platelets mainly, and a whole process including integrin subunit β2 and β3 binding with their ligands. Activation of immune cells, inflammatory immune adherence and coagulation response are involved in the acute venous thrombosis.
Keywords: Pulmonary embolism, venous thrombosis, integrin
Introduction
Venous thromboembolism (VTE) includes pulmonary embolism (PE) and deep vein thrombosis (DVT). PE has a high morbidity, mortality and misdiagnosis rate and has been an important health problem worldwide [1]. American College of Chest Physicians (ACCP) has published Guidelines for Diagnosis, Treatment and Prevention of VTE, and some risk factors are also proposed in the guideline including advanced age, infection, malignancy, and autoimmune diseases. Moreover, risk stratification is also introduced in this guideline [2]. Although this guideline has been used to guide the clinical practice, the preventive efficacy of VTE is still unsatisfactory [3]. In clinical practice, early, rapid and specific diagnosis of PE/DVT is crucial for its prognosis, and indicators for guidance of individualized anti-coagulation is required to determine the duration of anti-coagulation therapy. However, no specific objective indicators have been identified, due to the unclear understanding of the pathogenesis of acute venous thrombosis.
The authors have reported that the main component of acute venous thrombi is fibrinogen [4]. In our previous study, the thrombi were collected from patients with acute PE, and tandem mass spectrometry and bioinformatics were employed to determine that integrin subunit β1, β2 and β3 are core proteins of acute venous thrombi. In the present study, immunohistochemistry was done to investigate the expression and cell distribution of integrin β1, β2 and β3 in acute venous thrombi and the binding with different ligands in these cells. We aimed to explore role of immune cells in the process of acute venous thrombosis.
Patients and methods
Samples
Thrombi (n=5-8; 5-15 mm in length and 10-20 g in weight) were collected from the pulmonary artery of a patient with acute PE. Preparation of samples for pathological examination was done.
Immunohistochemistry and observation under a light microscope
After preparation of thrombus samples, HE staining, immunohistochemistry and Masson staining were performed. Following reagents were used in this study: integrin β1: 1: 50 (Abcam B3B11), integrin β2: 1: 150 (Abcam MEM-48), integrin β3: 1: 150 (Abcam PM6/13); ligand anti-Laminin antibody 1: 50 (Abcam14055), ligand anti-Fibronectin antibody 1: 50 (Abcam2413), ligand anti-Collagen I antibody 1: 50 (Abcam34710), ligand anti-Collagen II antibody 1: 50 (Abcam34712), ligand anti-fibrinogen antibody 1: 100 (Abcm ab34269), ligand anti-Factor X antibody 1: 50 (Abcam ab11871), ligand anti-Factor Xa heavy chain antibody 1: 50 (Abcam ab140112), ligand anti-C3/C3b antibody 1: 50 (Abcam ab11871), ligand anti-ICAM1 antibody 1: 50 (Abcam124759), ligand Von Will brand Factor antibody 1: 50 (Abcam11713), ligand anti-Vitronectin antibody 1: 50 (Abca m28023).
Results
1) Acute venous thrombi were red thrombi in which there are cord-like structures, and the spaces were filled with a large amount of aggravated red blood cells and nucleated blood cells (Figure 1).
Figure 1.
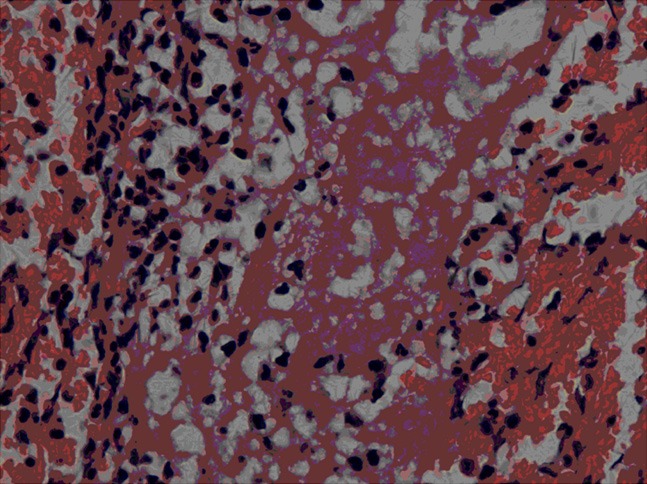
H&E staining of thrombus. HE staining of thrombus showed that venous thrombus was red thrombus in which cord-like structure, massive red blood cells and white blood cells with dark-brown nuclei aggregated (H&E staining, × 400).
2) Immunohistochemistry showed integrin β1 was expressed on the lymphocytes (Figure 2A), but no expression of Laminin, Fibronectin, Collagen-I and Collagen-II (receptors of integrin β1) was observed on the lymphocytes (Figure 2B-E).
Figure 2.
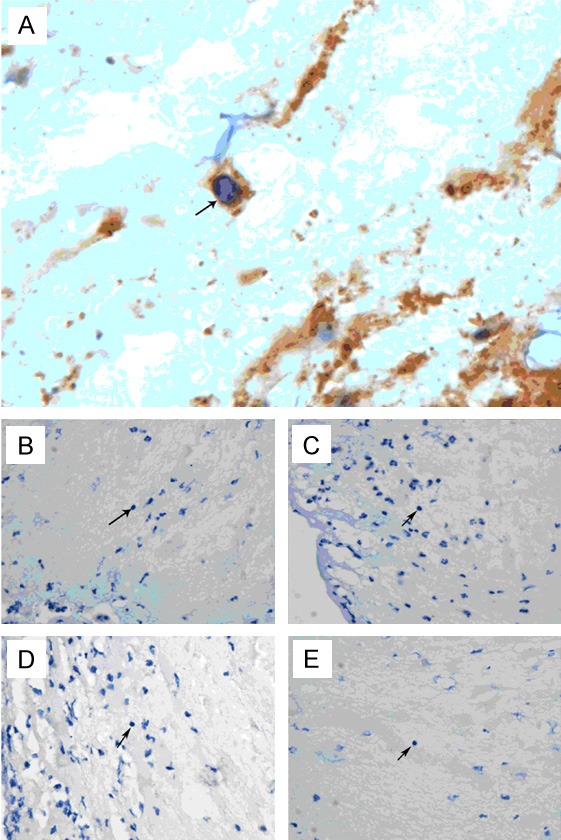
Immunohistochemistry. Arrow: dark-brown integrin β1 was expressed on the lymphocytes (A, × 1000). Expression of integrin β1 ligands (Laminin, B, × 400; Fibronectin, C, × 400; Collagen I, D, × 400; Collagen-II, E, × 400) was not observed on the lymphocytes.
3) Immunohistochemistry showed integrin β2 was expressed on the neutrophils (Figure 3A) which bound to fibrinogen (Figure 3B). The ICAM, factor X and iC3b were expressed on neutrphils (Figure 3C-E).
Figure 3.
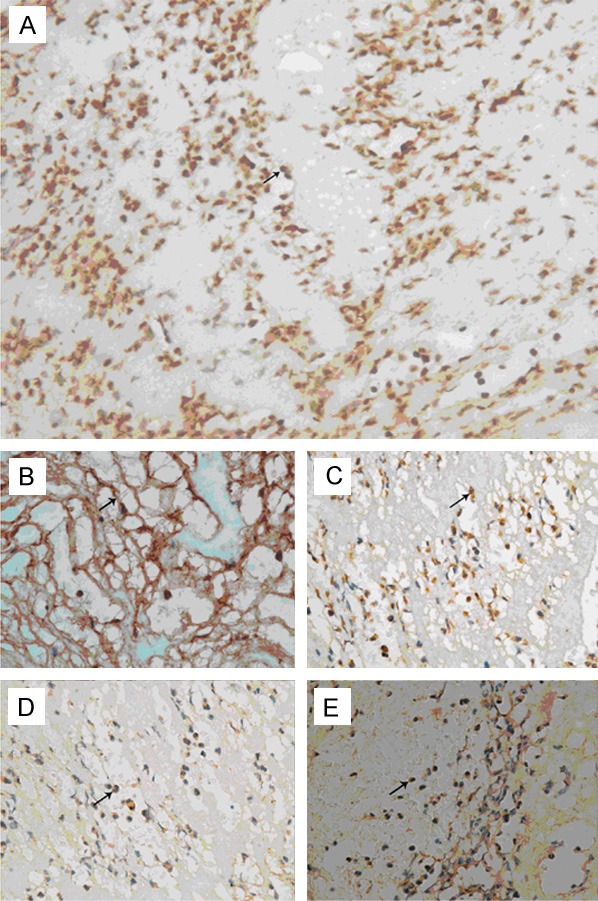
Immunohistochemistry. Arrow: dark-stained integrin β2 was expressed on the neutrophils (A, × 400) and bound fibrinogen (B, × 400). ICAM (C, × 400), factor X (D, × 400), and C3b (E, × 400) were expressed on neutrophils.
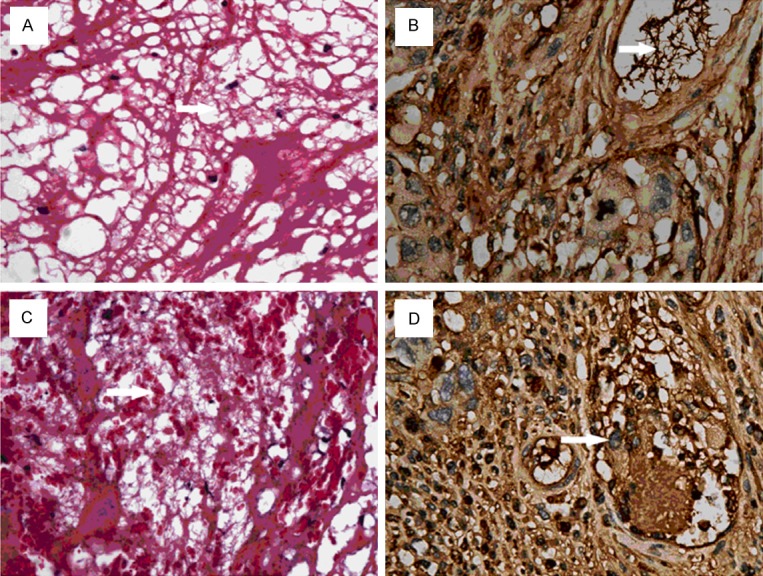
4) Immunohistochemistry showed integrin β3 was expressed on platelets which aggregated to be thrombotic skeleton (Figure 4A) and corallike structure (Figure 4B); these platelets bound fibrinogen to construct mesh structure (Figure 4C). No expression of Fibronectin, Vitronectin or vWF was observed on the platelets (Figure 4D-F).
Figure 4.
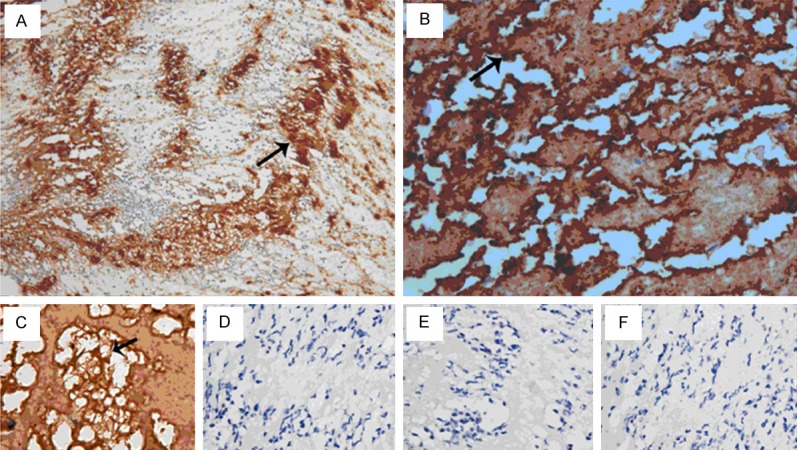
Immunohistochemistry. Arrow: dark-brown integrin β3 was expressed on platelets (A, × 200) and on the coral-like skeleton formed by platelets (B, × 400). Platelets and neutrophils bound fibrinogen to construct mesh-like structure (C, × 400). No expression of Fibronectin (D, × 400), Vitronectin (E, × 400), vWF (F, × 400) was observed on these cells.
5) The thrombi had mesh-like structure (Figure 5A, Masson staining) in which a large amount of red blood cell dominant blood cells filled (Figure 5C, Masson staining). In colon cancer tissues, there widely presented with dark-brown mesh-like structure in venules (Figure 5B anti-fibrinogen antibody 1: 100) in which a variety of cancer cells filled (Figure 5D).
Figure 5.
Masson staining of mesh-like structure. Arrow: Mesh-like structure was nest-like biological filter (A, × 400, Masson staining), in which red blood cell dominant blood cells filled (C, × 400, Masson staining). In colon cancer, massive mesh-like structure (anti-fibrinogen antibody, 1: 100, × 400) was observed in venules, and cancer cells were also observed in this mesh-like structure (anti-fibrinogen antibody, 1: 100, D, × 400).
6) Dark-brown Factor Xa was distributed on the mesh-like structure which was composed of fibrin/fibrinogen (Figure 6A, 6B).
Figure 6.
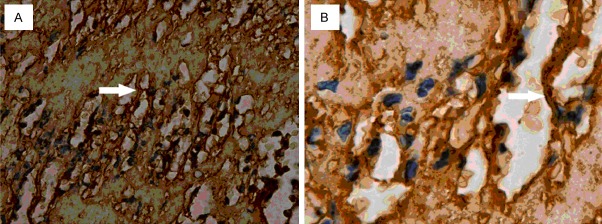
Factor Xa in mesh-like structure. Arrow: dark-brown factor Xa was found on the surface of mesh-like structure (A, × 400; B, × 1000). This suggests factor Xa acts on the fibrinogen/fibrin.
Discussion
Integrin family was initially recognized as adhesion molecules mediating the adhesion between cells and extracellular matrix, which leads to the integration of cells. Integrins are widely distributed in human body. A kind of integrin can be distributed in a variety of types of cells, and one cell may have the expression of several integrins. The expression of integrins varies from activation status and differentiation status of cells [5].
Integrin is a transmembrane heterodimer composed by α and β subunits at a ratio of 1: 1 via the non-covalent bond. A total of 8 β subunits (β1: β8) have been identified in human. Under the quiescent condition, β subunit covers α subunit, and thus the integrin fails to bind ligand. After activation of integrin, the β subunit extends and then α subunit is exposed. α subunit mainly mediates the reversible binding of integrin to its ligand. The β subunit is responsible for signal transduction and regulation of integrin’s affinity [6].
Integrin β1 is mainly expressed on lymphocytes [7], and its ligands include laminin, fibronetin, collagen, thrombospondin and VCAM-1 [8]. The binding of Integrin β1 and ligands is involved in immune cell adherence, which can provide co-stimulation for activation of T cells. Integrin β2 is mainly expressed on the neutrophils and monocytes [9], and its ligand includes Fibrinogen, ICAM, factor X and C3b [10]. The binding of Integrin β2 and ligands is involved in immune cell adherence, inflammation and phagocytosis. Integrin β3 is expressed on the platelets [11] and its ligand includes fibrinogen, fibronetin, vitronectin, VWF and thrombospondin [12]. The binding of Integrin β3 and ligands is involved in activation and aggregation of platelets.
Many cells are involved in inflammatory immune response, including cells locating in tissue, lymphocytes, neutrophiles and platelets. Light microscopy showed the thrombi in acute pulmonary thromboembolism were red thrombi. Immunohistochemistry revealed that integrin β1 was distributed on lymphocytes. Laminin, fibronetin, Collagen I and Collagen II, ligands of integrin β1, were not expressed on these cells. Integrin β2 was mainly distributed on neutrophils. The binding of activated integrin β2 with fibrinogen results in the formation of filamentous mesh. The ligands of integrin β2 (ICAM, factor X and C3b) were expressed on neutrophils, suggesting the binding of integrin β2 with the ligands is involved in the thrombosis. Integrin β3 was distributed on platelets gathered in different shapes, which bound with Fibrinogen to construct the filamentous mesh. No expression of fibronetin, vitronectin and vWF was observed on the platelets. The main protein component of acute venous thrombi is fibrinogen [4]. The result indicates the binding of platelet integrin β3 and neutrophil integrin β2 with ligand fibrinogen in thrombi is the early form of venous thrombosis.
In the thrombi, neutrophils and platelets are activated and bind to corresponding ligands, leading to inflammatory immune adhesion, which finally constructs filamentous mesh, a framework of venous thrombus. When the filamentous mesh was fully filled with red blood cell dominant blood cells, red thrombus formed. In the circulation, except for red blood cells, platelets and neutrophils have the largest amount. The binding of integrins on membrane of platelets and neutrophils and their ligands is directly involved in the formation of acute venous thrombus. The binding of neutrophils and factor X can trigger the coagulation process, and activated factor X converted to Xa and distributed on the fibrinogen, promoting soluble fibrinogenic thrombi transformed to fibrinic thrombi. Acute venous thrombosis is a main activation process of circulated neutrophils and platelets, and it is a whole process of integrin subunit β2 and β3 binding with their ligands, and a process of inflammatory immune adherence triggering coagulation reaction.
Thirty years ago, investigators developed and applied transient or permanent inferior vena cava filter in clinical practice to block the flow back of venous thrombi into the pulmonary artery, which may prevent the occurrence of PE [13]. In the study, the mesh-like structure in thrombi is similar to a biological filter, and what is the function of this mesh-like structure?
We have reported that virus-like microorganisms were observed in cytoplasm and intercellular substance of lymphocytes from peripheral venous blood of VTE patients with pulmonary hypertension and T cell immune dysfunction/disorder [14]. We also observed rod-shaped bacteria like microorganisms in apoptotic phagocytes from peripheral venous blood of patients with repeated PE/DVT and T cell immune dysfunction/disorder [15], We also found DVT in the veins of multiple organs (such as pulmonary artery, kidney, liver and pancreas) of a patient who died of SARS [16]. These findings indicate that the onset of VTE has the involvement of infection of microorganisms. Moreover, the mRNA expression of T cells and NK cells was significantly down regulated in patients with symptomatic VTE, as demonstrated by human genomics. The amount of CD3+, CD8+ and CD16+56+ cells reduced significantly, the increased CD4 level in patients with symptomatic VTE, which was consistent with findings in genomics [17]. The increased level of integrin subunit β1 in this study indicates the activation of lymphocytes, suggesting the regulatory function of lymphocytes enhanced. Malignancy is a disease related to immune dysfunction. Figure 5B, 5D showed mesh-like structure in the veins of cancer tissue was similar to that in venous thrombi. Furthermore, a variety of cancer cells were observed in this mesh like structure of veins in cancer tissue.
Acute venous thrombosis is an activation process of circulated lymphocytes, neutrophils and platelets, and it is a whole process of integrin subunit β1, β2 and β3 binding with their ligands, and a process of immune adherence, generating biological sieve and triggering coagulation reaction. Thus, we hypothesize that, when the infected cells or cancer cells can not be effectively and timely cleared in the presence of immune dysfunction/disorder, activated neutrophils and platelets bind to their ligands to construct biological filamentous mesh-like structure, which acts as a barrier to block the flow of infected cells or cancer cells. When the filamentous mesh-like structure was fully filled with red blood cell dominant blood cells, red venous thrombi occurred. The defensive biological filamentous mesh-like structure causes venous thrombosis.
Acknowledgements
This study was supported by “12th five year” National Science and Technology Supporting Program (2011BAI11B16).
Disclosure of conflict of interest
None.
References
- 1.Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence) Int Angiol. 2006;25:101–161. [PubMed] [Google Scholar]
- 2.American College of Chest Physicians. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141:e1S–801S. doi: 10.1378/chest.11-2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shackford SR, Rogers FB, Terrien CM, Bouchard P, Ratliff J, Zubis R. A 10-year analysis of venous thromboembolism on the surgical service: the effect of practice guidelines for prophylaxis. Surgery. 2008;144:3–11. doi: 10.1016/j.surg.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 4.Wang L, Gong Z, Jiang J, Xu W, Duan Q, Liu J, Qin C. Confusion of wide thrombolytic time window for acute pulmonary embolism: mass spectrographic analysis for thrombus proteins. Am J Respir Crit Care Med. 2011;184:145–146. doi: 10.1164/ajrccm.184.1.145. [DOI] [PubMed] [Google Scholar]
- 5.Dorner M, Zucol F, Alessi D, Haerle SK, Bossart W, Weber M, Byland R, Bernasconi M, Berger C, Tugizov S, Speck RF, Nadal D. beta1 integrin expression increases susceptibility of memory B cells to Epstein-Barr virus infection. J Virol. 2010;84:6667–6677. doi: 10.1128/JVI.02675-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Van Der Flier A, Sonnenberg A. Function and interactions of integrins. Cell Tissue Res. 2001;305:285–298. doi: 10.1007/s004410100417. [DOI] [PubMed] [Google Scholar]
- 7.Cavers M, Afzali B, Macey M, McCarthy DA, Irshad S, Brown KA. Differential expression of beta1 and beta2 integrins and L-selectin on CD4+ and CD8+ T lymphocytes in human blood: comparative analysis between isolated cells, whole blood samples and cryopreserved preparations. Clin Exp Immunol. 2002;127:60–65. doi: 10.1046/j.1365-2249.2002.01711.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fiorilli P, Partridge D, Staniszewska I, Wang JY, Grabacka M, So K, Marcinkiewicz C, Reiss K, Khalili K, Croul SE. Integrins mediate adhesion of medulloblastoma cells to tenascin and activate pathways associated with survival and proliferation. Lab Invest. 2008;88:1143–1156. doi: 10.1038/labinvest.2008.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rezzonico R, Chicheportiche R, Imbert V, Dayer JM. Engagement of CD11b and CD11c beta2 integrin by antibodies or soluble CD23 induces IL-1beta production on primary human monocytes through mitogen-activated protein kinase-dependent pathways. Blood. 2000;95:3868–3877. [PubMed] [Google Scholar]
- 10.Schwarz M, Nordt T, Bode C, Peter K. The GP IIb/IIIa inhibitor abciximab (c7E3) inhibits the binding of various ligands to the leukocyte integrin Mac-1 (CD11b/CD18, alphaMbeta2) Thromb Res. 2002;107:121–128. doi: 10.1016/s0049-3848(02)00207-4. [DOI] [PubMed] [Google Scholar]
- 11.Fang J, Nurden P, North P, Nurden AT, Du LM, Valentin N, Wilcox DA. C560Rbeta3 caused platelet integrin alphaII b beta3 to bind fibrinogen continuously, but resulted in a severe bleeding syndrome and increased murine mortality. J Thromb Haemost. 2013;11:1163–1171. doi: 10.1111/jth.12209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coburn J, Magoun L, Bodary SC, Leong JM. Integrins alpha(v)beta3 and alpha5beta1 mediate attachment of lyme disease spirochetes to human cells. Infect Immun. 1998;66:1946–1952. doi: 10.1128/iai.66.5.1946-1952.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Athanasoulis CA, Kaufman JA, Halpern EF, Waltman AC, Geller SC, Fan CM. Inferior vena caval filters: review of a 26-year single-center clinical experience. Radiology. 2000;216:54–66. doi: 10.1148/radiology.216.1.r00jl1254. [DOI] [PubMed] [Google Scholar]
- 14.Wang L, Gong Z, Liang A, Xie Y, Liu SL, Yu Z, Wang L, Wang Y. Compromised T-cell immunity and virus-like structure in a patient with pulmonary hypertension. Am J Respir Crit Care Med. 2010;182:434–435. doi: 10.1164/ajrccm.182.3.434. [DOI] [PubMed] [Google Scholar]
- 15.Wang L, Zhang X, Duan Q, Lv W, Gong Z, Xie Y, Liang A, Wang Y. Rod-like bacteria and recurrent venous thromboembolism. Am J Respir Crit Care Med. 2012;186:696. doi: 10.1164/ajrccm.186.7.696. [DOI] [PubMed] [Google Scholar]
- 16.Xiang-Hua Y, Le-Min W, Ai-Bin L, Zhu G, Riquan L, Xu-You Z, Wei-Wei R, Ye-Nan W. Severe acute respiratory syndrome and venous thromboembolism in multiple organs. Am J Respir Crit Care Med. 2010;182:436–437. doi: 10.1164/ajrccm.182.3.436. [DOI] [PubMed] [Google Scholar]
- 17.Duan Q, Gong Z, Song H, Wang L, Yang F, Lv W, Song Y. Symptomatic venous thromboembolism is a disease related to infection and immune dysfunction. Int J Med Sci. 2012;9:453–461. doi: 10.7150/ijms.4453. [DOI] [PMC free article] [PubMed] [Google Scholar]


