Abstract
SUMMARY
Chlamydia trachomatis is the leading cause of bacterial sexually transmitted disease worldwide, and despite significant advances in chlamydial research, a prophylactic vaccine has yet to be developed. This Gram-negative obligate intracellular bacterium, which often causes asymptomatic infection, may cause pelvic inflammatory disease (PID), ectopic pregnancies, scarring of the fallopian tubes, miscarriage, and infertility when left untreated. In the genital tract, Chlamydia trachomatis infects primarily epithelial cells and requires Th1 immunity for optimal clearance. This review first focuses on the immune cells important in a chlamydial infection. Second, we summarize the research and challenges associated with developing a chlamydial vaccine that elicits a protective Th1-mediated immune response without inducing adverse immunopathologies.
INTRODUCTION
Chlamydia trachomatis is the leading cause of bacterial sexually transmitted diseases (STDs) in humans. According to a 2008 WHO report, there are 105 million new cases of STDs due to C. trachomatis each year, and the infection rate has been increasing steadily (1, 2). When symptomatic, C. trachomatis infection can lead to mucopurulent endocervical discharge, hypertrophic cervix, and postcoital bleeding. In 20 to 40% of untreated women, C. trachomatis may reach the fallopian tubes via the endometrial epithelium and cause pelvic inflammatory disease (PID). However, because patients with C. trachomatis urogenital infections often do not exhibit any symptoms (75 to 90% of patients), they remain undiagnosed and untreated. This can lead to tubal factor infertility, miscarriage, or ectopic pregnancy (3–5), which is a life-threatening condition. Figure 1 shows pathologies caused by C. trachomatis. C. trachomatis is easily treated with antibiotics (i.e., erythromycin, azithromycin, or doxycycline), but several studies indicate that a year after treatment, almost one-fourth of individuals are reinfected with C. trachomatis (6, 7). C. trachomatis can also cause ocular scarring, which often leads to blindness. This disease, known as trachoma, is the leading cause of blindness worldwide (8, 9). As with C. trachomatis genital infection, ocular infections are often asymptomatic but can induce inflammation that leads to conjunctival scarring. Trachoma is prevalent in more than 50 countries, and the WHO estimates that 40 million people worldwide suffer from trachoma and that 1.3 million people are blind as a result of C. trachomatis trachoma infections (10, 11). In addition to causing urogenital and ocular disease, C. trachomatis can also infect the lymph nodes and the lymphatic system. This disease, termed lymphogranuloma venereum (LGV), is mostly caused by C. trachomatis serovars L1 to L3 (12, 13). Therefore, because of the prevalence of asymptomatic infections, recurrent infections, and the severity of genital and ocular pathologies induced by Chlamydia, the development of a vaccine is paramount. This review focuses largely on genital C. trachomatis and C. muridarum (a model organism that naturally infects rodents and is used largely for animal experiments) immunity and the challenges associated with generating a vaccine against these bacteria.
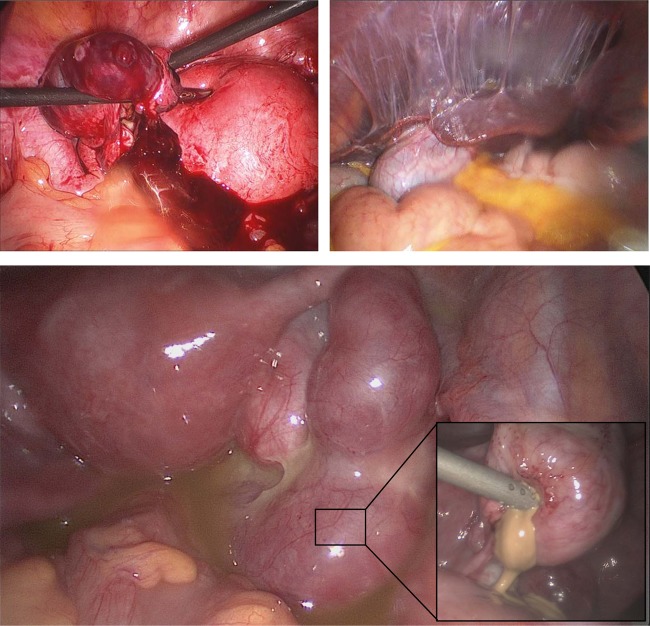
FIG 1.
(Top left) Laparoscopy surgical procedure showing a ruptured left fallopian tube as a result of ectopic pregnancy. The image shows the presence of significant hemoperitoneum. The patient was positive for Chlamydia trachomatis by PCR. (Top right) Fitz-Hugh-Curtis syndrome caused by Chlamydia trachomatis. (Bottom) Pelvic inflammatory disease (PID) caused by Chlamydia trachomatis infection of the uterus, fallopian tubes, and ovaries. The image shows the presence of a large dilation of the right fallopian tube, representing a tubo-ovarian abscess. (Inset) Drainage of the tubo-ovarian abscess.
CHLAMYDIA BIOLOGY
C. trachomatis is a Gram-negative obligate intracellular bacterium, and chlamydial species are able to infect both humans (C. trachomatis and C. pneumoniae) and animals (C. muridarum, C. suis, C. abortus, C. pecorum, C. psittaci, and C. caviae) (14). Presently, there are 18 identified serovars of C. trachomatis (15). Some serovars naturally infect the eye (serovars A to C), while others infect primarily genital tissues (serovars D to K) (16). In the genital tissues, C. trachomatis normally infects the cervical (women) or urethral (men) epithelium layer (17). Chlamydia exists in two developmental forms: the elementary body (EB), which is infectious, nonreplicating, and extracellular; and the reticulate body (RB), which is noninfectious, replicating, and intracellular. The EB displays no metabolic activity and is able to survive for long periods outside the cell. Infection begins when the small (∼0.2 to 0.3 μm) EB is internalized by the cell. After 8 to 10 h, the vesicle-bound EB (termed an inclusion) replicates by binary fission into the larger (∼0.8 μm) RB (18). After replication, the RBs revert back to EBs, which are able to infect neighboring cells (19). C. trachomatis is able to avoid destruction by preventing lysosomal fusion and replicating in an inclusion outside the endocytic pathway (18). Scarring associated with C. trachomatis infections may be the result of increased production of inducible nitric oxide synthase (iNOS) and mediators such as activins (20, 21).
INNATE AND ADAPTIVE IMMUNITY TO CHLAMYDIA
Neutrophils and NK Cells
Innate immunity plays a role in controlling chlamydial infections (22). Natural killer (NK) cells and neutrophils are the first cells that are recruited to the site of a chlamydial infection. These cells are important in innate immunity and have been implicated in the initial control of chlamydial infections. Two early studies demonstrated that human neutrophils were able to inactivate C. trachomatis in vitro (23, 24). Additionally, mice that were neutrophil depleted had a 10-fold greater C. muridarum burden in the female genital tract than neutrophil-competent mice. However, both sets of mice were able to eliminate C. trachomatis within the same time frame (25), suggesting that neutrophils are not critical for the resolution of infection. In fact, neutrophils are usually the first immune cells recruited to an infectious site, and compared to other leukocytes, they are short-lived (26, 27). Therefore, the most likely role for neutrophils is to reduce the initial chlamydial infection and limit it from spreading. However, recent evidence indicates that C. trachomatis may delay neutrophil apoptosis (28). Since neutrophils are a major source of tissue-damaging cytokines, such as matrix metalloproteinase 9 (MMP9), during acute inflammation (29), the prolonged life span of neutrophils may contribute to fibrosis and infertility associated with a chlamydial infection (30).
NK cells are known to be involved primarily in viral infections and cancer but have also been shown to be important in the early elimination of intracellular bacteria (31, 32). A study conducted by Tseng and Rank demonstrated that mice inoculated intravaginally with C. muridarum recruited gamma interferon (IFN-γ)-producing NK cells to the site of infection as early as 12 to 24 h after inoculation (33). Cytokine production by epithelial cells and dendritic cells (DC) has been implicated in NK cell IFN-γ production during a chlamydial challenge. Hook and colleagues demonstrated that C. trachomatis-stimulated human epithelial cells and DC produced interleukin-18 (IL-18) and IL-12, respectively, and that these cytokines induced NK cells to secrete IFN-γ in vitro (34). IFN-γ not only is important in inhibiting the growth of Chlamydia (35) but also is one of the main cytokines important for the induction of a Th1 immune response. Indeed, mice that were depleted of NK cells by treatment with an anti-NK-cell antibody and inoculated intravaginally with C. muridarum had a significant increase in the Th2-associated antibody IgG1. In contrast, Th1-associated IgG2a was the dominant antibody in mice that were not treated with an anti-NK-cell antibody and challenged with Chlamydia (33). A more recent study indicated that NK cells may influence Th1 immunity by modulating DC function. This investigation demonstrated that DC from intranasally C. muridarum-infected and NK-cell-depleted mice produced lower levels of IL-12 and a reduced capacity to stimulate CD4+ T cells in vitro. Furthermore, DC from NK cell knockout (KO) mice that were adoptively transferred into naive mice failed to induce a Th1-mediated immune response after intranasal challenge with C. muridarum (36). These data suggest that early IFN-γ production by NK cells modulates DC to downregulate the Th2 response, thereby allowing expression of strong Th1-mediated immunity, which has been shown to be essential for the resolution of Chlamydia infection.
NK T Cells
Natural killer T cells (NK T cells) are a unique population of T lymphocytes that express typical NK cell markers (NK1.1 and NKR-P1C) and a semivariant T cell receptor (αβT cell receptor; TCR) (37). NK T cells are CD1d restricted, meaning that they are able to recognize lipids and glycolipids presented by antigen-presenting cells on CD1d receptors but not antigens from the classical major histocompatibility complex (MHC) (38). These granular cytolytic lymphocytes are able to destroy infected and cancerous cells without prior sensitization and also secrete cytokines that are important in both innate and adaptive immunity. NK T cells have demonstrated immunomodulatory roles in a wide range of diseases, such as cancer, autoimmunity, allergy, atherosclerosis, and infection (37, 38). Furthermore, these cells have been implicated in regulating both innate (macrophages [Mϕ], natural killer cells, and dendritic cells) and adaptive (B cells and conventional T cells) immune cells (39–41). Zhao et al. demonstrated that NK cells from NK T cell KO mice and from mice that had the CD1d receptors blocked by antibodies exhibited decreased IFN-γ production and proliferation in a C. muridarum lung infection model (37). Another study suggested that natural killer T cells may induce protective Th1 immunity by promoting proliferation, CD40 upregulation, and production of IL-12 in a DC subset (CD8α+) during C. pneumoniae respiratory tract infections (42). However, there is conflicting evidence on whether NK T cells promote protective Th1 cell immunity or a Th2-mediated response that is characterized by bacterial pathogenesis. A study conducted by Bilenki et al. in 2005 (43) examined the role that NK T cells play in C. muridarum pneumonitis infection. This study demonstrated that intranasally infected CD1d-deficient mice lost less weight, exhibited less pathology, and had lower bacterial burdens, IL-14 levels, and IgE titers than wild-type (wt) mice. Additionally, wt mice that were stimulated with a known NK T cell ligand, α-galactosylceramide (α-GalCer), showed induced C. muridarum growth and increased IL-4 and IgE levels, suggesting that NK T cells promote a pathological Th2 response during chlamydial infection (43). However, a more recent study by Wang and colleagues demonstrated that pretreatment with α-GalCer in C. muridarum genital infection reduced bacterial burdens, decreased pathology, and increased the Th1-associated cytokines IFN-γ and IL-12 in both lymph nodes and genital tissues compared with those in non-α-GalCer-pretreated mice. These results suggest a role for NK T cells in protective Th1 immunity against Chlamydia (44).
Mϕ
Studies using both C. trachomatis and C. muridarum have shown that macrophages (Mϕ) migrate to chlamydial infection sites (45), phagocytose bacteria (46), and produce proinflammatory cytokines (47, 48). However, unlike epithelial cells, Mϕ are not a hospitable niche for chlamydial intracellular replication, as illustrated by the fact that compared to the case in epithelial cells, only a small fraction of chlamydial RBs are detected in Mϕ (49). C. trachomatis destruction inside Mϕ has been associated with host cell autophagy, a process by which cells degrade cytoplasmic proteins and organelles (49–51). Also, studies have demonstrated that Mϕ autophagy can enhance antigen presentation to T cells (52). Furthermore, IFN-γ has been shown to enhance both autophagy and upregulation of MHC class II molecules in Mϕ (50, 53). This is relevant because in addition to activating primed T cells, studies indicate that Mϕ can induce a humoral response in naive mice (54). Therefore, enhanced upregulation of MHC molecules containing chlamydial antigens may induce T cells to initiate both cell-mediated and antibody immune responses against Chlamydia. However, Jendro et al. demonstrated that C. trachomatis-infected human Mϕ are able to induce T cell apoptosis (55, 56). In addition to efficiently eliminating Chlamydia and presenting the peptides to T cells, Mϕ may also have an effect on chlamydial infection by inducing T cell death and perpetuating a persistent infection.
DC
Dendritic cells (DC) are known to be the quintessential antigen-presenting cells (APC). Immature DC are highly phagocytic, and after internalization of pathogens, they degrade the components and present the peptides to T cells via MHC receptors. This activates the T cells to initiate a cell-mediated and/or humoral immune response. Numerous investigations have demonstrated the ability of DC to activate T cells through MHC class I/II presentation and to secrete Th1 cytokines in chlamydial infection both in vitro and in vivo (57–61). An early study conducted by Lu and Zhong showed that mice that received heat-killed (HK) C. trachomatis-incubated bone marrow-derived DC (BMDC) were protected against a subsequent nasal challenge with live C. trachomatis (62). The protective response was Th1 mediated, further demonstrating a correlation between Th1-skewed immunity and protection against chlamydial infection. In contrast, DC that were pulsed with recombinant MOMP and adoptively transferred into mice elicited primarily the Th2-associated antibody IgG1 (63). Furthermore, IL-10 (Th2-associated cytokine) knockout DC pulsed with UV-inactivated C. trachomatis and adoptively transferred activated a high frequency of Th1 cells (64). These data have direct relevance to vaccine development because they indicate that the types of cytokines produced and antigens processed by DC and presented to CD4+ T cells mediate the Th1/Th2 balance during a chlamydial infection. There is also evidence that live Chlamydia is required for an optimal and protective immune response. Rey-Ladino and colleagues demonstrated that protection mediated by DC pulsed with UV-inactivated C. trachomatis and adoptively transferred into mice was significantly less than that in mice that were challenged with live EB-pulsed DC (65). A more recent study discovered that murine DC incubated with live C. muridarum presented many more peptides on their MHC class II molecules than DC that were incubated with dead EBs (66). However, C. trachomatis is able to limit MHC class I/II expression in APC (67). C. trachomatis has been shown to inhibit MHC molecules by degrading the MHC class I transcription factor RFX-5 and the MHC class II transcription factor USF-1 by secreting chlamydial protease-activating factor (CPAF) into the cytosol (68–71). DC are important for vaccine research because they are the critical links between innate and adaptive immunity. Two recent studies, using C. trachomatis MOMP transfected into DC (72) and DC that were incubated with recombinant CPAF (rCPAF) in vitro (73), illustrate the ability of DC to induce protective immunity against genital C. trachomatis and C. muridarum challenges, respectively.
T Cells
The involvement of T cells in chlamydial immunity was demonstrated almost 30 years ago, when Rank et al. observed that athymic nude mice established chronic infection with C. muridarum after intravaginal inoculation, but wild-type controls were able to eliminate the infection within 20 days (74). In human and mouse models, CD4+ as well as CD8+ T cells are able to be detected at the site of C. trachomatis infection (75–78). T cells are unable to recognize pathogens or antigens without the help of APC. APC such as DC and Mϕ are able to phagocytose chlamydial EBs in the extracellular space or engulf infected cells harboring RBs. After phagocytosis, APC degrade chlamydial components and present the peptides via MHC class I/II-antigen complexes. CD4+ T cells recognize antigens that are presented on MHC class II, and CD8+ T cells are activated by MHC class I-antigen complexes. In fact, both T cell subsets have been shown to recognize C. trachomatis antigens, such as outer membrane protein 2 (Omp2) (79), polymorphic outer membrane protein D (POMP-D) (80), MOMP (81–83), heat shock protein 60 (hsp60) (81, 84), chlamydial protease activating factor (CPAF) (73), PmpG, PmpF, and RpIF (77, 85). Although Chlamydia is able to induce a Th2-associated response by inducing IL-4 and IgG1 production, a Th1 response predominates. This response is characterized by the production of IL-12 by APC (86) and the subsequent activation of IFN-γ-producing T cells and plasma B cells that secrete Th1-associated antibodies, such as IgG2a and IgG3 (87, 88). However, a recent study demonstrated that previously C. trachomatis-sensitized human CD4+ T cells that were restimulated ex vivo with inactivated (γ-irradiated) EBs secreted significantly more IL-4 than tumor necrosis factor alpha (TNF-α) and IFN-γ. This study suggests that the type of immune response (Th1 versus Th2) to C. trachomatis may be tissue specific (89).
While there is ample evidence that CD4+ T cells play an integral part in C. muridarum and C. trachomatis infection resolution (90–93), the role for CD8+ T cells has been controversial. Indeed, CD8+ T cells are known to migrate to the infection site, and both human and mouse CD8+ T cells have been shown to destroy cells that have been infected with Chlamydia (94). A recent study by Murthy and colleagues showed that wt and CD8+ T knockout mice displayed similar clearances of C. muridarum following vaginal chlamydial challenge (95). These data support previous studies demonstrating that CD8+ T cells are not critical for C. trachomatis clearance (45, 59, 96). Furthermore, compared to wt mice, CD8+ T cell-deficient mice demonstrated less hydrosalpinx, implicating CD8+ T cells in chlamydia-induced pathology (95). A study conducted by Ibana et al. showed that most of the cervical CD8+ T cell populations before and after a C. trachomatis infection do not express the cytolytic protein perforin (97). Therefore, the lack of perforin in endocervix CD8+ T cells may explain why CD8+ T cells are not critical for genital chlamydial infection resolution. Although CD8+ T cells are not critical for chlamydial elimination and may even contribute to chlamydial sequelae, they nonetheless may play a contributory, albeit secondary, role by regulating other cells and by their own production of IFN-γ (94).
B Cells and Antibodies
Previous studies demonstrated that in humans, Chlamydia-specific antibodies play a role in C. trachomatis protective immunity (98, 99), and numerous C. trachomatis proteins have been shown to induce antigen-specific antibodies (91). However, even though anti-Chlamydia antibodies are able to neutralize infection in vitro (100, 101), growing evidence shows that B cells may not be important for initial chlamydial infection but, instead, play an important role in the secondary memory response (102, 103). Several possible mechanisms by which B cells modulate immunity during reinfection include antibody-mediated neutralization and opsonization (100) and antibody-dependent cellular cytotoxicity (ADCC) (a mechanism of cell-mediated immune defense whereby cells that have antibodies attached to their surfaces are targeted for lysis) (104). Another mechanism is the formation of antigen-antibody complexes that bind Fc receptors on APC, which then enhance phagocytosis and antigen presentation to CD4+ T cells (105). A recent study suggests that in humans, antibodies may be more specific for certain chlamydial serovars. Verweij and colleagues demonstrated that in serum samples from 235 C. trachomatis-positive patients, anti-IgG titers specific for C. trachomatis serogroup B (serovars B, Ba, D, Da, E, L1, L2, and L2a) were significantly higher than titers specific for serogroup C (serovars A, C, H, I, Ia, J, K, and L3) and serogroup I (serovars F, G, and Ga) (106).
Heat shock proteins (hsp's) are proteins that influence the correct folding and unfolding of intracellular proteins. C. trachomatis is known to secrete hsp's during an infection, and antigenic epitopes from bacterial hsp's have proven to be strong inducers of cellular and humoral immunity. Chlamydial and human hsp60 proteins are extremely similar, with four defined epitopes having 70% homology and virtually identical amino acid sequences (107). Several studies have suggested that autoimmunity to human hsp60 is a result of cross-reactivity after a chlamydial infection (108, 109). However, a study conducted by Hjelholt and colleagues did not find a correlation between tubal infertility and antibodies specific for human hsp60 in C. trachomatis infections, even though the patients produced antibodies against MOMP and chlamydial hsp60 (110).
IFN-γ
Production of IFN-γ in response to Chlamydia infection is critical for inhibiting chlamydial growth (17). IFN-γ can affect the survival of Chlamydia by several mechanisms. IFN-γ is able to enhance the phagocytic capabilities of Mϕ (111) and may promote the engulfment and elimination of Chlamydia trachomatis (112). Iron has been shown to be important for Chlamydia survival (113). IFN-γ downregulation of the transferrin receptor (114), which is needed for the import of iron into the cell, may also inhibit Chlamydia growth by limiting the available iron to the bacterium. In fact, IFN-γ has been shown to limit iron availability in Mϕ infected with Salmonella (115). Most Chlamydia species require tryptophan for survival (116). IFN-γ induces the expression of the cellular tryptophan-decyclizing enzyme indoleamine-2,3-dioxygenase (IDO), which degrades tryptophan. The lack of this essential amino acid has also been shown to cause Chlamydia trachomatis death through tryptophan starvation (35). However, there are chlamydial species that have adapted to tryptophan starvation by transforming into nonreplicating but viable persistent forms. After IFN-γ removal and subsequent tryptophan production, these persistent forms quickly differentiate into infectious elementary bodies. Furthermore, a recent study by Zhang and colleagues demonstrated that IFN-γ and IL-17A synergistically inhibit Chlamydia muridarum replication by inducing intracellular iNOS and NO production (117).
In conclusion, cell-mediated immunity that activates Mϕ, NK cells, NK T cells, neutrophils, and mediators such as IL-12 and IFN-γ is required for initial clearance. However, for protective immunity, both cell-mediated immunity and humoral immunity are needed, including antigen-specific T cells and antibodies that enhance phagocytosis and subsequent degradation and presentation of bacterial components by DC for a rapid Th1-mediated immune response. Table 1 summarizes recent developments in chlamydial research, including Chlamydia strains and antigens used, cell types affected, and immune responses elicited.
TABLE 1.
Summary of recent developments in Chlamydia research
| Cell type | Chlamydia species or antigen | Immune response | Reference(s) |
|---|---|---|---|
| Human neutrophil | C. trachomatis | Neutrophils incubated with Chlamydia in vitro were able to decrease viable bacterial count by 3 log after 60 min. | 24 |
| C. psittaci and C. trachomatis | C. psittaci and C. trachomatis cultured with complement or antibody induced neutrophil chemotaxis, whereas reticulate bodies alone did not. | 23 | |
| Neutrophil phagocytosis of C. psittaci and C. trachomatis was antibody and complement independent. | |||
| Mouse neutrophil | C. muridarum | Neutrophil-depleted mice had a 10-fold greater chlamydial burden in the female genital tract than neutrophil-competent mice. | 25 |
| Both wt and neutrophil-depleted mice were able to eliminate C. muridarum within the same time frame. | |||
| Human neutrophil | C. pneumoniae | C. pneumoniae delayed neutrophil apoptosis in vitro. | 28 |
| Guinea pig neutrophil | C. caviae | Chlamydia-infected neutrophil-depleted animals exhibited reduced ocular pathology and decreased T cells in infected conjunctivae. | 232 |
| Mouse NK cell | C. muridarum | Chlamydia induced IFN-γ-producing NK cells at the site of infection within 12–24 h after vaginal challenge. | 33 |
| Mice depleted of NK cells and inoculated intravaginally with C. muridarum had a significant increase in Th2-associated IgG1 antibody, whereas Th1-associated IgG2a was the dominant antibody in nondepleted mice. | |||
| Human NK cell | C. trachomatis | C. trachomatis induced human NK cells to secrete IFN-γ in vitro. | 233 |
| Human NK cell line | C. trachomatis | IL-18 and IL-12 from epithelial cells and DC, respectively, that were incubated with C. trachomatis stimulated NK cell IFN-γ production. | 34 |
| Mouse NK cell | C. muridarum | DC from intranasally C. muridarum-infected, NK cell-depleted mice produced lower levels of IL-12 and a reduced capacity to stimulate CD4+ T cells in vitro. | 36 |
| DC from NK cell-deficient mice that were adoptively transferred failed to induce a Th1-mediated immune response after intranasal challenge with C. muridarum. | |||
| Mouse NK T cell | C. muridarum | NK cells from NK T cell KO and CD1d antibody-blocked mice had decreased IFN-γ production and proliferation in a C. muridarum lung infection. | 37 |
| C. pneumoniae | NK T cells promote proliferation, CD40 upregulation, and production of IL-12 in a DC subset (CD8α+) in C. pneumoniae respiratory tract infection. | 42 | |
| C. muridarum | Infected CD1d-deficient mice were less susceptible to weight loss and had less severe pathology, lower bacterial burdens, lower IL-4 levels, and lower IgE titers than wild-type mice. | 43 | |
| Wild-type mice that were stimulated with a known NK T cell ligand (α-GalCer) had induced C. muridarum growth and increased IL-4 and IgE levels. | |||
| C. muridarum | Pretreatment with α-GalCer in C. muridarum genital infection reduced bacterial burdens, decreased pathology, and increased the Th1-associated cytokines IFN-γ and IL-12 in both lymph nodes and genital tissues compared with the case in non-α-GalCer-pretreated mice. | 44 | |
| Mouse Mϕ cell line (J774) and human Mϕ | C. trachomatis | Live and inactivated Chlamydia induced elevated IL-8, IL-1β, TNF-α, and IL-6. | 47, 48 |
| Mouse (RAW) and human (THP-1) Mϕ cell lines | C. trachomatis | Live Chlamydia induced autophagy. | 49, 50 |
| Human Mϕ | C. trachomatis | Live-Chlamydia-infected Mϕ induced T cell apoptosis. | 55, 56 |
| Mouse BMDC | C. muridarum | DC pulsed with UV-inactivated Chlamydia in vitro secreted elevated levels of IL-12. | 62 |
| DC pulsed with UV-inactivated Chlamydia and adoptively transferred into naive mice induced strong protection against live chlamydial lung infection. | |||
| IL-12−/− DC failed to induce Th1-dominant response and did not induce strong protection against chlamydial infection. | |||
| rMOMP | DC pulsed with rMOMP secreted IL-12 and induced infection-sensitized CD4+ T cells to secrete IFN-γ. | 63 | |
| DC pulsed with rMOMP and adoptively transferred into naive mice generated a Th2 anti-MOMP immune response. | |||
| C. trachomatis | IL-10−/− DC pulsed with UV-inactivated Chlamydia caused early DC maturation and activation and an increased ability to process and present antigens and enhanced the rate of Th1 activation. | 64 | |
| C. muridarum | DC incubated with UV-inactivated Chlamydia expressed low levels of CD40 and CD80, secreted low levels of proinflammatory cytokines, and exhibited reduced recognition by Chlamydia-specific CD4+ T cells. | 65 | |
| Adoptive transfer of live-EB-pulsed DC was more effective than UV-inactivated Chlamydia at protecting mice against a live intranasal chlamydial challenge. | |||
| C. muridarum | DC pulsed with live EBs presented 45 MHC class II C. muridarum peptides mapping to 13 proteins. In contrast, DC pulsed with heat- or UV-inactivated Chlamydia presented only 6 MHC class II chlamydial peptides mapping to 3 proteins. | 66 | |
| Only two epitopes were shared in common between live and inactivated C. muridarum. | |||
| Recombinant adenovirus carrying C. trachomatis MOMP | DC exhibited increased CD80, MHC class II, and IL-12 and were able to stimulate CD4+ T cell proliferation and IFN-γ. | 72 | |
| Adoptively transferred MOMP-transfected DC generated Th1-biased cytokine production and mucosal IgA and protected mice against chlamydial genital tract infection. | |||
| UV-inactivated C. muridarum plus CpG or rCPAF plus CpG | DC pulsed with rCPAF plus CpG exhibited increased CD86, CD80, CD40, MHC class II, and IL-12 but not IL-10 and IL-4. | 73 | |
| Mice adoptively immunized with rCPAF-plus-CpG- or UV-inactivated C. muridarum-plus-CpG-pulsed DC produced elevated IFN-γ, IgG1, and IgG2a and exhibited reduced Chlamydia shedding and reduced oviduct pathology compared to infected mock-immunized mice. | |||
| Mouse T cell | C. muridarum | Athymic nude mice established chronic genital tract infection, whereas wild-type mice resolved infection in 20 days. | 74 |
| C. trachomatis T cell antigens plus AbISCO-100 | A potent CD8+ T cell response, polyfunctional Th1-polarized CD4+ T cell responses (IFN-γ, TNF-α, and IL-2), and a high protein-specific Th1-skewed antibody response (IgG2c) were observed. | 234 | |
| Adoptive transfer of CD4+ T cells and CD8+ T cells to naive nonimmunized mice protected against C. trachomatis vaginal challenge, whereas passive transfer of immune sera did not. | |||
| C. muridarum MOMP plus CpG and Montanide ISA | Vaccinated mice were depleted of CD4+ and CD8+ T cells and challenged vaginally with live C. muridarum. Depletion of CD4+ T cells, but not CD8+ T cells, diminished vaccine-induced protection. | 90 | |
| Mouse CD4+ T cell | C. trachomatis | Genital tract C. trachomatis infection stimulated the activation and memory development of C. trachomatis-specific CD4+ T cells. | 92 |
| CD4+ T cells are necessary to confer protection against C. trachomatis infection. | |||
| C. muridarum | CD4+ T cell clone-induced epithelial NO production was critical for controlling replication. | 93 | |
| Most potent CD4+ T cell clones were dependent on T cell degranulation for chlamydial replication control. | |||
| Human CD4+ T cell | C. trachomatis | CD4+ T cells from women with genital tract infection that were pulsed ex vivo with EBs secreted significantly more IL-4 than TNF-α and IFN-γ | 89 |
| Mouse CD8+ T cell | C. muridarum | TNF-α from CD8+ T cells contributed significantly to oviduct pathological sequelae, but not bacterial clearance, following genital chlamydial challenge. | 95 |
| Human CD8+ T cell | C. trachomatis | Endocervix effector memory CD8+ T cells from C. trachomatis-infected women expressed low perforin levels. | 97 |
| Human B cell/antibody | A total of 21 antibody-inducing antigens were identified from C. trachomatis-infected patient sera. | 91 | |
| Mouse B cell/antibody | Recombinant outer membrane vesicles carrying C. muridarum HtrA, C. muridarum MOMP, or a monoclonal antibody (MAb) | Mice immunized with outer membrane vesicles carrying HtrA induced specific anti-HtrA antibodies that neutralized C. muridarum infectivity in vitro. | 100 |
| Passive immunization with sera from C. muridarum-infected mice conferred a marked level of protection from C. muridarum genital reinfection and shortened the time of infection. | 102 | ||
| MOMP MAbs conferred significant level of immunity to reinfection and reduced shedding. | |||
| rCPAF plus CpG | Wild-type and B cell-deficient (μmT) mice vaccinated intranasally with rCPAF plus CpG and challenged vaginally with live C. muridarum demonstrated comparable clearances and similar reductions in pathology. | 103 |
ANIMAL MODELS
Mouse
The most commonly used animal in chlamydial research is the mouse. There are several advantages in using mice to investigate chlamydial genital infections, including availability of transgenic mouse strains, small size, low cost, and availability of mouse-specific reagents. C. muridarum is utilized for genital studies, because C. muridarum intravaginal infection closely mimics acute C. trachomatis infection in women. Moreover, it can cause hydrosalpinx, fibrosis, infertility, and abortion in mice (118–121). C. muridarum genital infection is usually resolved in 3 to 4 weeks, and the mice are partially protected against subsequent reinfections (122, 123). C. trachomatis is also used, but the infection in mice is less severe and is resolved more quickly than C. muridarum infection. Additionally, a mouse C. trachomatis infection requires a larger number of infectious units (118) and usually causes pathology only when injected directly into the uterus, uterine horn, or ovarian bursa (92, 124). However, C. trachomatis genital infection in women is mostly asymptomatic and often does not induce severe upper tract genital pathology. Thus, as Lyons et al. have argued (125), C. trachomatis is an appropriate model for studying chlamydial urogenital infections in mice.
Pigtailed Macaque
Although several nonhuman primate models have been used in Chlamydia research, including the grivet monkey (126), marmoset (127), and baboon (128), the pigtailed macaque is utilized most frequently for genital research. Indeed, it is naturally infected with C. trachomatis human biovars, and the female anatomy, menstrual cycle, and vaginal microflora are akin to those in humans (129). In fact, repeated C. trachomatis infection of macaque fallopian tubes has been shown to induce a pathology similar to that of pelvic inflammatory disease in women (130). Immune responses include Th1-skewed cytokine production after initial inoculation and systemic and local humoral responses. However, unlike the case in mice, where CD4+ T cells are the dominant T cell subset, CD8+ T cells predominate in macaques after chlamydial infection (118). Although the macaque model is ideal for vaccine and immunology studies, the high cost and need for adequate facilities and expertise limit its use.
Guinea Pig
Guinea pigs are naturally infected with C. caviae. Advantages of this model include the ability to study chlamydial sexual transmission (male guinea pigs are able to be infected with Chlamydia) (131), the transmission of Chlamydia to newborns, and the fact that guinea pig genital tract infection is similar to that by C. trachomatis in humans. Additionally, the guinea pig is a good model for hormonal research because humans and guinea pigs have comparable estrous cycles (118, 132, 133). Studies have indicated that CD8+ T cell genital infiltrates after infection are similar to those of humans and nonhuman primates (134, 135). In contrast to the case in mice, antibodies have been implicated in the resolution of primary infection in chlamydia-infected guinea pigs (118).
Pig
In addition to the mouse, guinea pig, and nonhuman primate models, the pig has also been used for chlamydial studies. Pig and human female reproductive tracts are very similar (136), and studies indicate that the immune systems of humans and pigs are much more related than those of mice and humans (118). Pigs are naturally infected with C. abortus and C. suis, but C. pecorum, C. psittaci, and C. trachomatis are also able to infect pigs (118, 137). However, although C. suis is highly related to C. trachomatis and is a natural pig pathogen, C. suis does not induce tubal infertility and PID. Therefore, it is difficult to use this species as a model for investigating human C. trachomatis urogenital pathology (118). Nevertheless, Schautteet and colleagues have used the pig model to investigate recombinant protein-based and DNA-based vaccine candidates. These investigations demonstrated that both rPmpG and C. trachomatis DNA vaccines provided significant protection against C. trachomatis vaginal challenge. DNA mucosal immunization provided superior protection compared to that in pigs immunized intradermally (138–140), demonstrating the importance of vaccination routes.
Although animal models are extremely useful and necessary for understanding the complex nature of chlamydial infection and pathology, comparing data from different animal models can be difficult. C. muridarum-infected mice and genitally C. caviae-infected guinea pigs are characterized by infections that last roughly 3 to 4 weeks (132). However, pigtailed macaque genital C. trachomatis infections are longer and more persistent, with bacterial shedding still occurring after 4 months (132, 141). In comparison, human studies suggest that after 1 year of untreated genital infections, half of C. trachomatis infections still persist (142, 143). Extrapolating data from animal studies and comparing the results to human correlates are difficult because of limited data on untreated human subjects with chlamydial infections. Nevertheless, there are data indicating that although humans are able to spontaneously clear chlamydial infection without antibiotic intervention, the time frame of such clearance can span several months to years, and the resolution appears to be more robust in older individuals (142, 144). Additionally, epidemiological data indicate that the longer an individual is infected with Chlamydia, the greater are the chances of clearance (142, 144). Finally, a major limitation in comparing animal models of chlamydial genital infection to human C. trachomatis urogenital infection is that the actual amounts of Chlamydia inoculated during sexual intercourse in humans are not known, so it is impossible to approximate similar doses in animal models. Table 2 summarizes the main advantages, disadvantages, and protection in the various animal models discussed above.
TABLE 2.
Advantages, disadvantages, and chlamydial protective immunity of different animal models
| Species | Advantages | Disadvantages | Protective immunity | Reference(s) |
|---|---|---|---|---|
| Mouse | Small size, availability of reagents, low cost | C. muridarum does not infect humans; C. trachomatis infects mice only at high doses; it is difficult to extrapolate C. muridarum and C. trachomatis to human correlates. | C. muridarum primary genital tract infection resolves in 3–4 weeks; primary C. muridarum infection partially protects against reinfection; durations are shorter and bacterial loads are lower in C. muridarum-reinfected mice; C. trachomatis genital infection is milder and shorter than C. muridarum infection. | 118 |
| Guinea pig | Ability to study sexual transmission (female to male) and transmission to newborns; good model for hormonal research (estrous cycle similar to that in women); genital infection with C. caviae closely resembles C. trachomatis infection in women | Limited guinea pig-specific reagents | Primary genital infection with C. caviae is resolved in 3–4 weeks; partial immunity remains for roughly 50% of the animals' life span. | 123, 132 |
| Pig | Reproductive organs and immune system are closely related to those of humans; naturally infected with C. suis, which is closely related to C. trachomatis | Expensive, complicated to work with, lack of reagents | C. trachomatis shedding has been documented for up to 21 days. | 132 |
| Nonhuman primates (pigtailed macaque) | Female anatomy, menstrual cycle, and microflora similar to those in women; naturally infected with C. trachomatis | Expensive, need for special facilities, need for expertise | Secondary cervical challenge with C. trachomatis after initial resolution resulted in either no infection or a shorter and less severe infection. | 141 |
VACCINES
Due to increasing rates of mainly asymptomatic C. trachomatis infections worldwide and the adverse long-term consequences resulting from these infections (ectopic pregnancy, infertility, and preterm birth), developing an antichlamydial vaccine is of paramount importance. However, a human vaccine that elicits both T cell and B cell immunity has been elusive. Lack of knowledge of female genital tract immunity, which is highly regulated by sex hormones during the menstrual cycle (145), a dearth of adjuvants that not only optimize the immune response to Chlamydia antigens but also can target the vaccine-specific immune responses at the infection site, and a limited understanding of the mechanisms by which chlamydial antigens induce protective immunity hinder human C. trachomatis vaccine development. A potential C. trachomatis vaccine ideally will induce both mucosal and systemic immune responses, but autoimmune cross-reactions with human antigens and unregulated inflammation that causes pathology must be avoided. Table 3 summarizes recent chlamydial antigens, delivery systems, routes of vaccination and infection, and the subsequent immune responses elicited.
TABLE 3.
Summary of recent developments in Chlamydia vaccine researcha
| Vaccine | Advantages | Disadvantages | Antigen (Ag) and adjuvant | Ag immunization route | Model/Chlamydia infection route | Immune response | Reference(s) |
|---|---|---|---|---|---|---|---|
| Intact Chlamydia | Intact Ag, native configuration, replication, humoral/cellular immunity | Requires refrigeration, potential to revert to virulent strains, large-scale production is difficult, possible transmission to unvaccinated individuals | Plasmid-deficient Chlamydia (CM972, CM3.1) | Mouse/i.v. | Elevated IgG2a (Th1), low levels of IgG1 (Th2). | 87 | |
| Mutants do not stimulate TLR2-dependent cytokine production. | |||||||
| Infected mice with mutant Chlamydia and challenged with wt Chlamydia are protected against oviduct disease. | |||||||
| Plasmid-deficient Chlamydia (L2) | Mouse/i.v. | Elevated IgG2a, low IgG1, no IgA (mucosal). | 88 | ||||
| No pathology in the urogenital tract induced by L2. | |||||||
| Mice vaccinated with plasmid-deficient bacterium were not protected from infection/inflammation with secondary wt chlamydial infection. | |||||||
| Purified subunits | Do not revert to virulent strains, their use avoids undesirable antigens | Expensive to produce, purification not standardized, difficult to maintain native conformation of antigen complex | MOMP plus cholera toxin subunit B conjugated to CpG | i.m. + s.c. | Mouse/i.n. | Elevated IgG2a and IgG3 (Th1), lower IgG1 level, elevated IFN-γ (Th1). | 160 |
| MOMP-ISCOM | i.n. or i.m. | Mouse/i.n. | i.m. route induced highest IFN-γ and IL-4 (Th2) levels. | 159 | |||
| MOMP plus Freund's adjuvant | i.m. + s.c. | Mouse/i.v. | Vortexed MOMP elicited higher IgG2a than IgG1. | 157 | |||
| Sonicated MOMP elicited higher IgG1 than IgG2a. | |||||||
| MOMP plus IC31 | i.m. + s.c. | Mouse/i.n. | Higher IgG1 than IgG2a. | 235 | |||
| MOMP plus CpG/Montanide | i.m. + s.c. | Rhesus macaque | Elevated IgG, IgA, IFN-γ, and TNF-α. | 161 | |||
| Recombinant proteins | High yields, inexpensive | Some proteins require posttranslational modification; if produced in E. coli, possibility of endotoxin contamination | rMOMP plus cholera toxin/CpG or CTA1 | s.l. or t.c. or i.n. | Mouse/i.n. | Elevated IFN-γ and TNF-α. | 167 |
| i.n. immunization with MOMP plus either adjuvant protected mice from infection but not pathology. | |||||||
| t.c. immunization with MOMP and CTA1-DD protected mice from pathology, but Chlamydia burden was same as that in control mice. | |||||||
| rMOMP plus CpG/Montanide | i.m. + s.c. | Mouse/i.n. | Vaccination protected against fibrotic scarring in lungs. | 168 | |||
| Elevated IgG2a and lower levels of IgG1. | |||||||
| rCPAF plus IL-12 | i.n. | Mouse/i.v. | Increased IFN-γ and minimal IL-4. | 170 | |||
| Elevated IgG2a and IgA. | |||||||
| rCPAF plus CpG | i.n. | Mouse/i.v. | Vaccination significantly prevented infertility. | 171 | |||
| rCTH1 plus CAF01 | s.c. | Mouse/i.v. | T cell production of TNF-α, IL-2, and IFN-γ. | 173 | |||
| Anti-CTH1 IgG2a and IgG1. | |||||||
| Protection was solely CD4+ T cell mediated. | |||||||
| rGlgP plus CpG | i.m. | Mouse/i.v. | Th1-dominant T cell response. | 172 | |||
| Reduced hydrosalpinx severity. | |||||||
| rMIP | i.m. | Mouse/i.v. | More IgG2a than IgG1. | 174 | |||
| Elevated IFN-γ and no IL-4. | |||||||
| Reduced hydrosalpinx severity. | |||||||
| rCT043 | i.m. | Mouse/i.n. | rCT043 reduces bacterial load in a mouse model of i.n. infection. | 236 | |||
| rCT823 plus ISCOM and CT144 plus ISCOM | s.c. | Mouse/i.v. | Elevated IFN-γ, TNF-α, and IL-2. | 234 | |||
| No detectable IL-4 and IL-10. | |||||||
| Elevated IgG2c (Th1) but not IgG1. | |||||||
| rPmpG plus GNE (adjuvant) and SctC plus GNE | s.c. | Pig/i.v. | PmpG protected better than SctC. | 140 | |||
| PmpG vaccination did not elicit antibody production. | |||||||
| SctC vaccination elicited high antibody titers. | |||||||
| DNA vaccines | Cheap, easy to produce, can encode multiple epitopes, native conformation of antigenic determinants | Safety, possible genome integration, anti-DNA antibodies, not possible for nonproteins | DNA MOMP | i.m. | Mouse/i.v. | Elevated levels of IgG2a and IgG1. | 177 |
| Priming with MOMP and secondary boost with DNA MOMP-ISCOM | i.m. | Mouse/i.n. | Elevated levels of IgG2a, IgA, and IFN-γ. | 178 | |||
| DNA MOMP plus GM-CSF, enterotoxins (E. coli) A and B | i.n. + i.v. | Pig/i.v. | Vaccination induced significant protection against genital challenge. | 138 | |||
| Protection correlated with efficient T cell priming and elevated IgA. | |||||||
| Anti-MOMP antibodies and low IL-4 production. | |||||||
| OmpA | i.m. | Pig/i.m. | 179 | ||||
| Bacterial ghosts | Inactivation not required, and therefore relevant antigenic determinants are not denatured; easy to produce; require no refrigeration; carriage of different antigens, DNA, and drugs simultaneously; recognition and phagocytosis by APC | Presence of lipopolysaccharide (LPS) | MOMP and PorB DNA plasmid | i.m. | Mouse/i.v. | High levels of IgG2a and IgA. | 182 |
| PmpD and PorB DNA plasmid | i.m. | Mouse/i.v. | High levels of IgG2a, IgA, and IFN-γ and low levels of IL-5 (Th2). | 183 | |||
| Biodegradable | Biodegradable, nontoxic, high | rMOMP encapsulated in PLGA | s.c. | Mouse | Elevated CD4+ and CD8+ T cells. | 189, 190 | |
| polymers | encapsulation capacity, | Elevated IFN-γ and IL-12 and reduced IL-4 and IL-10. | |||||
| PLGAs are efficiently phagocytosed by DC and Mϕ, chitosan has mucosal adhesiveness properties and enhanced penetration across mucosal barrier | Chitosan containing rMOMP DNA | i.m. | Elevated IgG2a and reduced IgG1. | 191 | |||
| Vaccines from transgenic plants | Low-cost production, ease of use | Requirement for strong adjuvant | MOMP introduced into A. thaliana and D. carota | 169 | |||
| Gas vesicles | Able to express peptides from various genes | Gene fragments coding for MOMP, OmcB, and POMP loaded into Halobacteria-derived gas vesicles | Elicited Th1 cytokines in human foreskin fibroblasts. | 237 |
i.v., intravenous; i.n., intranasal; i.m., intramuscular; s.c., subcutaneous; s.l., sublingual; t.c., transcutaneous.
Intact Organisms
Successful vaccines against ovine enzootic abortions have been available for many years (146). These vaccines consist of either live or inactivated C. abortis strains and provide proof of principle that a successful vaccine against Chlamydia is possible in mammals. However, these vaccines are not able to protect against infection, and the vaccines were not designed for use in humans (147, 148). Nonetheless, because of the success of these vaccines, live C. trachomatis bacteria were used as the first human Chlamydia vaccines (149). The first vaccines focused mainly on trachoma rather than genital C. trachomatis infection, with results ranging from limited and short-lived protection to considerable protection against infection and pathology (150, 151). However, some individuals who were challenged with Chlamydia trachomatis developed a pathological response that was worse than that in those who did not receive the vaccine. Notably, Grayston and colleagues vaccinated Taiwanese children at risk for trachoma with formalin-inactivated C. trachomatis and followed their progress for 3 years. Although the children that received the inactivated chlamydial vaccine exhibited partial protection compared to nonimmunized controls, a significant proportion of the immunized individuals developed enhanced disease, ostensibly as a result of delayed-type hypersensitivity after chlamydial infection (152). A vaccine study using two different preparations of live C. trachomatis demonstrated short-lived and modest protection in Gambian children. However, similar to the case in the Taiwanese study, some individuals developed more severe disease after infection (153). Vaccines with live organisms are generally considered optimal because they contain virtually all of the antigenic determinants in the correct three-dimensional conformation. However, using live organisms for vaccines has drawbacks, as growing and purifying Chlamydia on a large scale are extremely complex. Moreover, these vaccines need cold storage, and even more importantly, there is the potential for avirulent strains to revert back to infectious wild-type strains (154).
Because of the safety issues of live vaccines, research switched to organisms that were heat or chemically inactivated. The major disadvantages of these types of vaccines are the absence of replication and a suboptimal immune response, necessitating the need for revaccination and adjuvants. Heat or chemical bacterial inactivation may also release unwanted and detrimental components, which can have deleterious effects or degrade protein antigenic determinants, thereby reducing the degree of protection. Recently, plasmid-deficient Chlamydia strains have been used in vaccine research, with conflicting results. O'Connell et al. demonstrated that a strain of C. muridarum (Nigg) which lacks a plasmid and is defective in the ability to accumulate glycogen did not cause inflammatory pathology in mice. Furthermore, the plasmid-deficient bacterium protected mice against a secondary infection with plasmid-competent virulent C. muridarum (87). However, a different group demonstrated that mice vaccinated with an attenuated plasmidless C. trachomatis strain (L2R) were not protected from colonization and inflammatory pathology after a secondary challenge with wild-type C. trachomatis, although there were reductions in infectious burdens at early time points (88).
Subunit Antigenic Determinants
Another vaccine strategy utilized is the administration of purified antigenic determinants known to elicit an immune response. Subunit vaccines are safer than attenuated or heat- or chemically inactivated organisms because they are unable to cause infection and because virulent components that may cause pathology can be avoided. One of the most well-studied vaccine candidates for C. trachomatis is MOMP. This membrane protein contains several conserved CD4+ T, CD8+ T, and B cell epitopes (155). An early study conducted by Pal and colleagues demonstrated that C. muridarum COMP (chlamydial outer membrane complex), a chlamydial outer membrane with a cysteine-cross-linked protein shell, significantly protected mice against genital challenge, whereas MOMP did not (156). Several years later, the same group administered a different preparation of C. muridarum MOMP along with Freund's adjuvant. This new, purified-and-refolded MOMP–Freund's adjuvant preparation significantly reduced bacterial burdens after a chlamydial genital challenge, demonstrating the importance of adjuvants and a correct MOMP configuration in eliciting a protective immune response (157). Tifrea et al. discovered that a polymer that keeps membrane proteins soluble (Amphipol) in aqueous solution was able to stabilize MOMP (158). Another group immunized mice with a C. trachomatis MOMP-ISCOM vaccine. ISCOM (immune-stimulating complex), which is composed mainly of cholesterol, phospholipids, and saponin, is known to induce both cell-mediated and antibody responses when used as an adjuvant. Inoculation with MOMP-ISCOM was able to elicit a Th1 antigen-specific response, and vaginal infection was cleared within 1 week (159). A C. muridarum native MOMP preparation combined with an adjuvant consisting of the subunit B cholera toxin conjugated to CpG (CTB-CpG) induced significant cell-mediated and antigen-specific antibody responses against intranasal infection with C. muridarum (160). A nonhuman primate model was used to demonstrate the efficacy of a vaccine formulated with native MOMP. Rhesus macaques that were immunized intramuscularly and subcutaneously along with the adjuvants CpG-2395 and Montanide ISA 720 produced high levels of Th1 cytokines (IFN-γ and TNF-α) and C. trachomatis-specific IgG and IgA (161). Drawbacks of subunit vaccines include the facts that extracting, refolding, and purifying protein complexes such as MOMP are very expensive and that purifications are not standardized, so differences in extraction methods may influence the conformation of the protein epitopes and the vaccine efficacy. The advent of protein arrays has aided in the identification of potential immunodominant antigen vaccine candidates. Cruz-Fisher et al. designed a protein chip array that was incubated with sera from mice that were infected with C. muridarum (162). From a total of 909 proteins, 71 were recognized by the array. Another array using sera from C. trachomatis-infected women recognized over 700 chlamydial proteins (163).
Recombinant Proteins
The advent of recombinant DNA technology has made it possible to produce large quantities of bacterial proteins. Thus, different attempts have been made to use rMOMP in C. trachomatis vaccines. Unfortunately, producing rMOMP with its native conformational epitopes intact on a large scale is challenging, and full-length rMOMP is toxic in some expression systems (164, 165). Evidence suggests that differences in MOMP conformation may affect its ability to act as a vaccine. In 2009, a comparison of vaccines using native or recombinant MOMP demonstrated that natural MOMP was superior than rMOMP in its ability to protect against chlamydial challenge (166). However, other studies using rMOMP with and without adjuvants demonstrated protection against Chlamydia (167, 168). In 2011, Kalbina and colleagues designed a chimeric construct containing genes that correlate with two different MOMP regions and introduced the construct into a bacterium (Escherichia coli) and two plants (Arabidopsis thaliana and Daucus carota). The stable integration of the transgene was demonstrated in A. thaliana and D. carota plants over several generations. The rMOMP purified from E. coli was used to produce antibodies in rabbits, and these antibodies recognized the proteins in E. coli, A. thaliana, D. carota, and C. trachomatis. The stability of the construct in the offspring plants suggests that this system may be useful for large-scale production of rMOMP (169).
Recombinant proteins other than MOMP have also been shown to be potential vaccine candidates. In 2007, Murphy and colleagues investigated the potential of rCPAF to induce an immune response that would resolve chlamydial infection. Mice immunized intranasally with rCPAF and IL-12 (a Th1 cytokine) demonstrated increased IFN-γ production and minimal IL-4 (a Th2 cytokine) production and elevated IgG2a (Th1) and IgA (mucosal) antibody levels, displayed markedly reduced bacterial burdens upon C. muridarum genital inoculation, and were protected against pathological consequences of Chlamydia infection compared with mock-immunized mice (170). The same group demonstrated that rCPAF intranasal vaccination may prevent infertility from repeated genital C. muridarum infections in mice (171). Mice immunized with recombinant chlamydial glycogen phosphorylase (GlgP) and intravaginally challenged with live C. muridarum elicited Th1 immunity that included antichlamydial antibodies and reduced hydrosalpinx severity. Additionally, mice that were immunized with GlgP demonstrated less shedding on day 14 post-vaginal challenge (172). Olsen et al. utilized two recombinant proteins in a subunit chlamydial vaccine. The fusion protein CTH1 consisted of CT443 (OmcB), which has been shown to elicit cell-mediated and antibody responses, and CT521 (rl 16), a protein known to be a target during chlamydial infection in humans. Immunization with CTH1 along with the strong Th1-inducing adjuvant CAF01 elicited TNF-α, IL-2, and IFN-γ production from T cells, as well as large amounts of both Th1 (IgG2a) and Th2 (IgG1) CTH1-specific antibodies. The vaccine significantly reduced bacterial burdens after vaginal infections with live C. trachomatis and C. muridarum (173). Lu and colleagues screened 5 recombinant chlamydial antigens that were previously found to react with sera from intravaginally C. muridarum-infected mice as chlamydial vaccine candidates. Only Mip (macrophage inflammatory protein) induced pronounced protection, which was characterized by a Th1-dominant T cell response and anti-Mip antibodies (174).
Plasmid DNA
DNA vaccines work by injecting a plasmid that carries a specific gene of interest within the host. The product of the gene can then be expressed by inducing an immune response. DNA vaccines have several advantages compared with other vaccination strategies. DNA is easy to purify, and plasmids can be constructed relatively quickly (175). Additionally, DNA vaccines can encode multiple epitopes that are in the native three-dimensional configuration and avoid the problem associated with attenuated organisms which are able to revert back to virulent forms. However, as with other vaccine strategies, DNA vaccines have some disadvantages. In autoimmune diseases such as lupus, anti-DNA antibodies are produced, and introduction of a DNA plasmid into the host may result in autoimmunity. Also, because DNA encodes proteins, DNA vaccines are generally used for protein-based antigens (176). In 1999, Pal and colleagues immunized mice with a C. trachomatis MOMP DNA vaccine. When the mice were vaginally challenged with C. trachomatis, the immune response was modest, and immunized mice were not protected against infection (177). The following year, Dong-Ji et al. demonstrated that immunization with DNA-MOMP and boosting with MOMP-ISCOM conferred more protection against C. trachomatis than that in mice that were immunized only with MOMP-ISCOM (178). More recently, two studies using a pig model assessed the efficacy of DNA chlamydial vaccines. Schautteet et al. combined aerosol-vaginal delivery of a DNA vaccine encoding MOMP coadministered with DNAs encoding three different adjuvants (granulocyte-macrophage colony-stimulating factor [GM-CSF] and E. coli enterotoxin subunits A and B). Mice immunized with the DNA vaccine were significantly protected against genital C. trachomatis challenge (138). Ou and colleagues, using a pig model, demonstrated that an OmpA-based DNA vaccine elicited more antigen-specific IgG antibodies and a larger T cell proliferative response than those in controls after a vaginal infection with C. abortus (179). A plasmid encoding MOMP epitopes inserted into a human papillomavirus (HPV) was used to assess the ability of a MOMP DNA vaccine to protect against vaginal C. trachomatis infection. Immunization elicited a Th1 response characterized by low IL-4 production and antibodies against MOMP (180). All of these recent studies demonstrate the feasibility of DNA-based vaccines, and this approach thus deserves further study.
OTHER CHLAMYDIAL VACCINES AND DELIVERY SYSTEMS
BGs
Bacterial ghosts (BGs) are bacterium-based empty shells that do not contain internal components but retain their outer morphological structure and can be loaded with peptides, drugs, or DNA (181). In 2007, a vaccine system in which a DNA plasmid that encoded C. trachomatis MOMP and the porin protein (PorB) inserted into a BG was used. Animals that were immunized intramuscularly with the DNA-bacterial ghost vaccine completely resolved a C. trachomatis genital infection by 2 weeks postinfection. The inflammatory response was Th1 mediated, characterized by high levels of IgA and IgG2a (182). More recently, Eko and colleagues used a BG that contained PorB and chlamydial polymorphic membrane protein D (PmpD) proteins to evaluate its ability to induce chlamydial immunity. Intramuscular immunization elicited high levels of Th1-associated IgG2a antibody, mucosa-associated IgA antibody, and IFN-γ (Th1) and low levels of IL-5 (Th2) in response to an intravaginal C. muridarum infection (183).
Biodegradable Polymers
PLGA (poly-d,l-lactide-coglycolide) is an FDA-approved polysaccharide that can encapsulate peptides, proteins, or DNA. PLGAs are efficiently phagocytosed by DC and Mϕ (184, 185), and PLGA antigens are able to be presented on MHC class I/II molecules, thus activating CD4+ and CD8+ T cells (186, 187). Chitosan is a chitin-derived polysaccharide and has several properties that make it a useful vaccine delivery system, including its mucoadhesiveness and enhanced penetration capacity across mucosal barriers (188). Two recent studies using recombinant MOMP encapsulated in PLGA demonstrated enhanced induction of Th1 cytokines and cellular and antibody immune responses (189, 190). Cambridge et al. demonstrated that MOMP was expressed in the tissues and organs of mice that were intramuscularly injected with chitosan nanoparticles containing recombinant MOMP DNA (191).
Gas Vesicles
Gas vesicles are gas-containing structures that provide buoyancy and are found in some bacteria and archaea. These protein structures are hollow, rigid, and lipid-free and allow diffusion of gases across the membrane. In fact, gas vesicles from Halobacterium spp. have been used in vaccine research (192, 193). Gas vesicles are desirable for use as a delivery system for human vaccines because they are nontoxic to humans and are able to be phagocytosed efficiently by APC (194). Furthermore, exogenous bacterial DNAs that encode particular proteins are able to be inserted into the structure, resulting in expression of these proteins on the gas vesicle surface (192–194). Studies have shown that in the absence of adjuvants, Halobacteria gas vesicles that displayed viral peptides elicited a long-lasting immune response characterized by immunological memory in mice (193). Halobacteria-derived gas vesicles that were loaded with gene fragments coding for MOMP, OmcB (outer membrane complex B), and POMP-B (polymorphic outer membrane B) and expressed on the surface were able to elicit a Th1 cytokine profile in human foreskin fibroblasts in vitro. Furthermore, antibodies specific for the recombinant proteins were confirmed using sera from Chlamydia-positive patients, suggesting that this could be an effective antigen delivery system for a Chlamydia vaccine (192).
ADJUVANTS
Live attenuated or intact inactivated whole-organism vaccines usually do not require additional components to induce a robust immune response. However, vaccines that comprise subunits of the original organism often induce a suboptimal immune response and therefore require substances, termed adjuvants, that are intended to enhance the immunogenicity of these vaccines. Natural adjuvants can come from the organism itself, such as Toll-like receptor (TLR) ligands, or can be endogenous cytokines/chemokines produced in response to a challenge. The main goal of artificial or naturally derived adjuvants is to induce immunity that closely resembles a natural immune response to the intended pathogen. Therefore, identifying adjuvants that elicit a protective immune response in vivo is going to be one of the main challenges for developing an effective chlamydial vaccine. There are several components that are required for a successful vaccine, including activation of innate immunity, costimulation of immune cells, cytokine production, antigen presentation, and immune modulation, and adjuvants can contribute to all of these signals. Even though various natural and synthetic adjuvants have been utilized in basic research for over 70 years, only a few adjuvants are currently licensed for use in human vaccines. These include alum (aluminum hydroxide), AS04 (monophosphoryl lipid A [MPL]-alum), AS03 and MF59 (squalene-based adjuvants), and liposomes (195). Some adjuvants bind with the antigen and are used as delivery systems. Delivery system adjuvants stabilize the antigen and allow the antigen to be released slowly, thereby contributing to costimulation of immune cells and possible uptake by antigen-presenting cells, such as DC. Examples of antigen delivery system adjuvants include calcium phosphate, tyrosine, liposomes, virosomes, emulsions, nanoparticles, ISCOMs, virus-like particles, and alum (196). However, even though, until recently, alum has been the only FDA-approved adjuvant, it does not induce IL-12 production, weakly activates DC, and induces a Th2-mediated antibody response (197, 198). Therefore, it is a poor adjuvant if the intended outcome is Th1-mediated immunity. Another class of adjuvants influence the immune response by directly activating immune cells. These components are recognized as “danger signals” via receptors, such as TLRs, of innate immune cells. The subsequent cytokine secretion, internalization, and presentation of the antigen to CD4+ T lymphocytes activate the T cells, which can then initiate an adaptive immune response. These adjuvants, termed potentiators, are usually purified bacterial or viral components or synthetic molecules that are structurally similar to the intended natural organism component. Examples of immune potentiators are MPL, MDP (N-acetyl-muramyl-l-alanyl-d-isoglutamine), CpG, bacterial or viral components, lipopeptides, and double-stranded RNA (dsRNA) (196).
Numerous adjuvants, such as those mentioned in this review (e.g., Freund's adjuvant, ISCOMs, CTB-CpG, CpG, and bacterial ghosts), have been used in chlamydial vaccine research, with various results. Recent research has added other new antigen/adjuvant candidates, with encouraging results. A study by Yu and colleagues investigated the ability of liposomes, CpG, alum, and the squalene water-in-oil emulsion adjuvant Montanide coadministered with the chlamydial protein PmpG to mediate protective immunity against C. muridarum. The results demonstrated that two liposomal adjuvants, DDA-MPL and DDa-TDB, were superior compared to the other adjuvants. Additionally, protection against chlamydial infection was better when the liposomal adjuvant DDA-MPL was administered with 7 different T cell antigens compared to immunization with just MOMP (199). This highlights the various opportunities to further improve vaccine candidates by identifying the optimal epitope-adjuvant combination.
VACCINATION ROUTES
Vaccine efficacy is defined not only by the type of antigen and adjuvant used but also by the administration route, since lymphocytes primed by antigens in vivo are endowed with specialized homing programs guiding their migration to specific mucosal sites (200). Once naive T cells are primed in a lymph node, a global switch of their homing program occurs, which enables them, while trafficking through the blood circulation, to detect chemokines and adhesion molecules which direct them to their tissue destination. Furthermore, T cell homing to the genital mucosa involves either α1β1, α4β1 (201), or α4β7/E selectin (202) in Chlamydia-infected mice. Both systemic and mucosal immunization routes have been shown to be able to induce both antibody- and cell-mediated immune responses in the genital tract, with intranasal immunization often being more effective (203, 204). Overall, mucosal immunization routes were more effective at preventing genital challenges with a variety of pathogens (205–209).
Numerous immunization routes have been used for chlamydial vaccinations, including oral (210), intranasal (211), intravaginal (139), subcutaneous (212), intramuscular (213), perivaginal (212), perisacral (212), sublingual (214), and colonic (124) routes. A study using purified MOMP with a Borrelia surface protein as an adjuvant demonstrated that in two different mouse strains, intramuscular-plus-subcutaneous and perivaginal-plus-perisacral immunization elicited high systemic and mucosal serum antibody titers. In contrast, the mice that received the MOMP-adjuvant vaccine intranasally were characterized by low serum titers (212). However, Cunningham et al. showed that intranasal vaccination with rMOMP resulted in antibodies (IgG and IgA) specific for MOMP in the genital tract, demonstrating that intranasal administration may target immunity to the reproductive tract (215). Several studies comparing the protective abilities of various vaccination routes demonstrated that combined mucosal and systemic inoculation may be optimal. Ralli-Jain and colleagues demonstrated that a MOMP-adjuvant combined sublingual (mucosal), intramuscular (systemic), and subcutaneous (systemic) vaccination regimen showed the best protection following intranasal C. trachomatis challenge (214). Another group demonstrated that mice immunized by combined mucosal and systemic routes with C. muridarum recombinant MOMP plus CpG/Montanide not only showed the strongest antibody and cell-mediated responses after vaginal challenge with C. muridarum but also were protected against infertility (124).
POSTVACCINATION PROTECTION
Postvaccination protection can vary depending on the antigen, immunization route, adjuvant, and infection model. Yu and colleagues investigated the ability of live versus inactivated Chlamydia to protect against a subsequent chlamydial vaginal infection. In their studies, the mice were immunized with either live or UV- or heat-inactivated C. muridarum and challenged (at 6 weeks postvaccination for live EBs or 2 weeks postvaccination for inactivated EBs) with live C. muridarum intravaginally. Mice that were vaccinated with inactivated Chlamydia exhibited little to no protection, whereas live-EB-immunized mice had virtually no bacterial titers in cervicovaginal washes at 6 days postchallenge (66). UV-inactivated bacteria are often alive and have their components intact but are unable to replicate (216), whereas heat inactivation kills bacteria and often denatures protein epitopes (217). Therefore, these results indicate a requirement for replicating bacteria that contain nondenatured epitopes in their original conformation to induce protective immunity that significantly reduces or eliminates bacterial shedding at the site of infection. A guinea pig-C. psittaci genital model demonstrated the effectiveness of live chlamydial vaccination and the importance of vaccination routes. Animals were vaccinated by four different routes (intravenous, subcutaneous, oral, and ocular) with either live or UV-inactivated C. psittaci and were challenged intravaginally with live C. psittaci. All immunized animals exhibited a reduction in genital infection, except for guinea pigs that received UV-inactivated Chlamydia orally. Live C. psittaci immunization induced greater resistance to challenge than that with UV-inactivated C. psittaci immunization, and all routes of immunization (intravenous versus subcutaneous versus ocular versus oral) induced similar protective responses (218). Two studies investigating the use of plasmid-deficient C. muridarum and C. trachomatis as attenuated live vaccines demonstrated different results in terms of bacterial burdens and pathology in a genital infection model. Mice vaccinated with mutant C. muridarum strains were protected against oviduct disease but exhibited bacterial burdens similar to those in wild-type C. muridarum-vaccinated controls (87). Plasmid-deficient C. trachomatis (L2)-vaccinated mice were not protected against infection or inflammatory disease but exhibited a reduction in infectious burden 1 to 2 weeks after challenge with wild-type C. trachomatis (88). These results demonstrate the challenges associated with using different chlamydial strains in mouse models to understand protective immunity and pathology during Chlamydia infection. MOMP is one of the most investigated components of Chlamydia in vaccine research, and depending on the source (DNA, purified protein, or recombinant protein), preparation, and serovar, it can have varied results in its efficacy in protection against chlamydial burden and pathology. Shaw et al. demonstrated that mice intravenously receiving rMOMP-pulsed BMDC were not protected against live genital C. muridarum challenge and had vaginal shedding similar to that of unimmunized control mice (63). In contrast, mice immunized intravenously with Ad-MOMP (a recombinant adenovirus carrying the C. trachomatis serovar E MOMP gene)-transfected BMDC exhibited smaller bacterial genital burdens, less pathology, and minimal loss of body weight compared to controls (72). However, mice vaccinated with MOMP DNA and challenged intravaginally with C. muridarum demonstrated vaginal shedding and fertility rates similar to those for mice vaccinated with control plasmids (177). Collectively, these results indicate that the origin of antigen (recombinant MOMP versus a virus carrying the MOMP gene) and the type of chlamydial strain (C. muridarum versus C. trachomatis) may significantly affect how DC present proteins to T cells and modulate protective immunity and pathology in a chlamydial infection. Pal et al. showed that mice immunized intramuscularly and subcutaneously with a vortexed preparation of native MOMP plus Freund's adjuvant were significantly protected against C. muridarum genital challenge in terms of the number of inclusion-forming units (IFUs) and the length of time the mice shed viable organisms compared to sonicated-MOMP-vaccinated mice (157), indicating that the preparation of native MOMP is important for its ability to act as a vaccine antigen. Table 4 summarizes postvaccination protection, including animal models, vaccination antigens, and times of postvaccination chlamydial challenge.
TABLE 4.
Summary of postvaccination protectiona
| Infection model/Chlamydia species | Animal model | Ag/vaccination route | Time (days) of postvaccination challenge | Assessment after postvaccination challenge | Vaccination protection | Reference |
|---|---|---|---|---|---|---|
| Genital (i.v.)/C. muridarum | Mouse | Plasmid-deficient C. muridarum (CM972, CM3.1)/i.v. | 98 | Bacterial burden (4–45 days), pathology (day 42) | Mice vaccinated with mutant C. muridarum strains were protected against oviduct disease but exhibited bacterial burdens similar to those of wt C. muridarum-vaccinated controls. | 87 |
| Genital (i.v.)/C. trachomatis serovar D | Mouse | Plasmid-deficient C. trachomatis (L2)/i.v. | 35 | Bacterial burden (3–28 days), pathology (day 14) | Vaccinated mice were not protected against infection or inflammatory disease but exhibited reductions in infectious burden 3 to 7 days after challenge with wt C. trachomatis. | 88 |
| On days 14 to 28, there was no difference between vaccinated and control groups. | ||||||
| Genital (i.v.)/C. muridarum | Mouse | Purified MOMP (vortexed or sonicated) plus Freund's adjuvant/i.m. and s.c. | 14 | Bacterial burden (day 42) | Mice immunized with vortexed MOMP plus Freund's adjuvant were significantly protected against Chlamydia challenge in terms of the number of IFUs and the length of time the mice shed viable organisms compared to sonicated-MOMP-vaccinated mice. | 157 |
| Mouse | rCPAF plus IL-12/i.n. | 30 | Bacterial burden (4–30 days), pathology (12–80 days) | Vaccinated mice exhibited significant reductions in bacterial shedding as early as 8 days postchallenge compared with controls. | 170 | |
| 80% of vaccinated mice successfully resolved infection by day 15. In contrast, control mice (rCPAF or IL-12) were still heavily infected by day 15. By day 18, 100% of vaccinated mice had resolved the infection. | ||||||
| Vaccination reduced hydrosalpinx, oviduct dilation, and fibrosis at 80 days postchallenge. | ||||||
| Mouse | rCPAF plus CpG/i.n. | 30 | Fertility assessment, breeding (day 80) | Mice vaccinated with rCPAF plus CpG and challenged with Chlamydia exhibited fertility similar to that of nonimmunized controls. | 171 | |
| After a secondary challenge, nonimmunized controls displayed significant reductions in fertility compared to immunized mice. | ||||||
| Mouse | CTH1 plus CAF01 (adjuvant)/s.c. | 42 | Bacterial burden (3–35 days) | Compared to control mice, CTH1-plus-adjuvant-immunized mice exhibited significantly less vaginal bacterial shedding 7, 14, and 21 days after chlamydial challenge. | 173 | |
| Days 14 to 21 showed the most efficient protection. | ||||||
| Mouse | rGlgP plus CpG/i.m. | 30 | Bacterial burden (7–28 days), pathology (day 60) | Immunized mice were characterized by a significant reduction of live organisms in the vagina by day 14 and a reduced severity of hydrosalpinx at 60 days postchallenge. | 172 | |
| Mouse | rMIP/i.m. | 30 | Bacterial burden (4–30 days), pathology (day 60) | Mice immunized with MIP and C. muridarum EBs (positive control) had significantly reduced vaginal bacterial shedding as early as 12 days after intravaginal chlamydial challenge. | 174 | |
| On day 21, most of the MIP-immunized mice exhibited dramatically reduced bacterial burdens compared with negative-control mice vaccinated with glutathione S-transferase (GST). | ||||||
| Hydrosalpinx severity was less severe in MIP-vaccinated mice than in GST-immunized controls. | ||||||
| Genital (i.v.)/C. trachomatis serovar D | Mouse | rCT823 plus AbISCO (adjuvant) or CT144 plus AbISCO/s.c. | 7 | Bacterial burden (3–21 days) | Vaccination with CT823 plus adjuvant reduced cervicovaginal bacterial burdens by 10 log postchallenge compared to controls (UV-inactivated EBs). | 234 |
| Vaccination with CT144 plus adjuvant reduced bacterial burdens by 1/2 log on day 7 and then 2- to 3-fold by days 10 and 14 compared to controls. | ||||||
| Genital (i.v.)/C. muridarum | Mouse | MOMP DNA plasmids/i.m. | 60 | Bacterial burden (7–42 days), fertility assessment, breeding (day 42) | Mice vaccinated with MOMP DNA and challenged with C. muridarum exhibited vaginal shedding and fertility rates similar to those of mice vaccinated with control plasmids. | 177 |
| Genital (i.v.)/C. trachomatis serovar D | Mouse | MOMP plus PorB DNA plasmid plus Vibrio cholerae ghost (VCG)/i.m. | 21 | Bacterial burden (3–24 days) | Compared to controls, VCG-MOMP/PorB-immunized mice exhibited significant differences in vaginal C. trachomatis shedding in terms of duration and intensity, and the infection was completely resolved by 2 weeks postchallenge. | 182 |
| Genital (i.v.)/C. muridarum | Mouse | PmpD and PorB DNA plasmids plus VCG/i.m. | 14 | Bacterial burden (3–24 days) | Mice vaccinated with VCG containing a PmpD or PorB DNA plasmid or both shed 2.5-log fewer vaginal chlamydial IFUs than controls (VCG containing an irrelevant chlamydial antigen [gD2]) at 3 days postchallenge. | 183 |
| Mouse | Adoptively transferred BMDC that were pulsed with rMOMP in vitro/intravenous | 14 | Bacterial burden (day 7) | Vaccinated mice that received BMDC pulsed with rMOMP were not protected against live chlamydial challenge and had vaginal shedding similar to that of unimmunized control mice at 7 days postchallenge. | 63 | |
| Mouse | Live or inactivated (UV or heat) C. muridarum, with or without CpG/i.n. | 42 (live EBs), 14 (UV- or heat-inactivated EBs) | Bacterial burden (day 6) | Vaccination with live EBs significantly reduced bacterial shedding compared to that with both UV- and heat-inactivated C. muridarum at 6 days postchallenge. | 66 | |
| Mouse | Adoptively transferred BMDC that were transfected with a recombinant adenovirus carrying the C. trachomatis serovar E MOMP gene (Ad-MOMP)/intravenous | 21 | Body weight (2–10 days), bacterial burden (2–10 days), pathology (day 11) | Vaccinated mice that received Ad-MOMP-transfected BMDC exhibited lower bacterial genital burdens, less pathology, and minimal loss of body weight compared to controls. | 72 | |
| Mouse | Adoptively transferred BMDC that were pulsed with UV-inactivated C. muridarum plus CpG or rCPAF plus CpG in vitro/s.c. | 30 | Bacterial burden (3–30 days), pathology (day 80) | Mice immunized with adoptively transferred BMDC that were pulsed with either UV-inactivated EBs plus CpG or rCPAF plus CpG demonstrated significantly reduced vaginal shedding at 15 days postchallenge and reduced oviduct pathology at 80 days postinfection compared to mock-immunized mice. | 73 | |
| Mouse | Sera from (convalescent) previously vaginally C. muridarum-infected wt mice; MAb to MOMP | CD4+ T cell- and antibody-deficient mice that had previously resolved a chlamydial infection were immunized with convalescent-phase sera. These mice exhibited decreased vaginal bacterial shedding (from >100,000 IFUs to <10 IFUs) and a shortened duration of infection compared to control mice. | 102 | |||
| Immunization with a MAb specific for MOMP reduced bacterial shedding >100-fold. | ||||||
| Genital (i.v.)/C. trachomatis serovar E | Pig | MOMP DNA plus GM-CSF, enterotoxins (E. coli) A and B/i.n. plus i.v., i.d. | 21 | Bacterial burden (2–25 days), pathology (day 25) | Vaginal shedding was less in i.n. plus i.v. vaccinated pigs than in pigs that were i.d. vaccinated. However, neither group of vaccinated pigs (i.n. plus i.v. or i.d.) completely resolved the infection. | 139 |
| Vaccinated pigs exhibited significantly fewer lesions in urogenital tissues. | ||||||
| Pig | PmpG plus GNE (adjuvant) and SctC plus GNE/s.c. | 41 | Bacterial burden (2–23), pathology (day 23) | PmpG gave better protection than SctC after vaginal challenge with C. trachomatis, characterized by low antibody titers and minor pathology. | 140 | |
| SctC-vaccinated pigs exhibited significant pathology but more antibody production after Chlamydia challenge. | ||||||
| Genital (i.v.)/C. psittaci | Guinea pig | Live or UV-inactivated C. psittaci/intravenous, s.c., oral, or ocular | 14 | Bacterial burden (3–24 days) | All immunized animals exhibited reductions in genital infection, except for guinea pigs that received UV-inactivated Chlamydia orally and nonimmunized controls. | 218 |
| Live compared to UV-inactivated C. psittaci immunization induced more resistance to challenge, and all routes of immunization (i.v. vs s.c. vs ocular) induced similar protective responses. |
Abbreviations: i.v., intravaginal; i.n., intranasal; i.m., intramuscular; s.c., subcutaneous; i.d., intradermal.
Antibiotic intervention studies in Canada, Vietnam, and Africa have yielded important insights into the development of protective immunity to Chlamydia in humans. Collectively, the data suggest that early antibiotic treatment for chlamydial infection may increase the number of individuals who are susceptible to reinfection by disrupting the development of adaptive immunity. Therefore, early antibiotic intervention may paradoxically increase the prevalence of Chlamydia in the population over the long term. For example, an epidemiological study analyzing C. trachomatis urogenital cases discovered that after antibiotic intervention, the incidence of Chlamydia reinfection cases rose by 4.6% (219). These results have also been observed for trachoma (220, 221).
CHALLENGES FOR A CHLAMYDIAL VACCINE
There are many critical questions that still need to be addressed in order to develop a chlamydial vaccine in the future. How does this intracellular bacterium induce pathogenesis in the host? How does Chlamydia mediate the immune response, and by what mechanism does Chlamydia induce sequelae during infection? Why do most patients remain asymptomatic and not develop pathology, whereas others develop severe PID? What type of human genetic polymorphisms may predispose a given individual to a chlamydial infection and pathology?
Indeed, regarding genetic susceptibility to chlamydial infection, two good reviews are available, by Morrè et al. and Lal et al. (222, 223). In cases of persistent infection, what are the characteristics of immunity that allow the infection to persist? We still do not completely understand the role that antibodies play in chlamydial infection and how Th1- versus Th2-mediated immunity is regulated at different infection sites (especially in the female genital tract). Furthermore, a better understanding of mucosal immunity may allow the development of a more specific vaccine. What type of chlamydial antigens should be used in a potential vaccine, and how will the vaccine be prepared and delivered? One might need to produce the desired vaccine antigen in a heterologous host, thus bypassing the difficulties of growing an intracellular bacterium or purifying a specific protein. Because APC differentially modulate immunity to Chlamydia depending on the type of antigen (i.e., recombinant, native, from viral transfection, or whole, intact bacteria), antigen origin is important for developing a vaccine to Chlamydia. The fact that most infected patients remain asymptomatic suggests that different strategies of vaccination will be needed. Indeed, both preventing the primary acute infection and curing persistent/chronic infection might be investigated. What type of immunization route(s) and how many vaccinations are optimal? Should mucosal or systemic vaccinations be utilized separately or in combination? How many vaccinations should be administered, and what would be the optimal intervals between vaccinations? In order to enhance or select a specific immune response, novel adjuvants might have to be developed. Although animal experiments cannot replace clinical trials, they are nonetheless necessary, and deciding which animal model to utilize is an important factor in understanding and analyzing protective immunity, pathology, and immunological mechanisms during chlamydial infection. Thus, there is still much research to be done on the biology of C. trachomatis and on the pathogenesis of genital chlamydial infection. Developing a chlamydial vaccine will entail further research on the antigenicity of chlamydial proteins and on novel and more effective vaccine delivery systems.
When should individuals be vaccinated? A prophylactic chlamydial vaccine should be administered before the infection is normally acquired, which usually means the early teens, and preferably before sexual activity. However, the vaccine should be effective enough to provide protection throughout sexual life and may need to be readministered throughout the individual's lifetime to be optimally effective. Who should be vaccinated? Should only men or women be vaccinated, or both sexes? A similar debate occurred when the HPV vaccine Gardasil became available. In Australia, Gardasil was directly approved for both men and women in 2006, whereas the U.S. FDA initially approved it only for women. The reason for approving only women was that the efficacy studies (phase III) were performed in women, and thus no efficacy was assessed at that time for men (protection against genital warts). However, because immunity bridging data were available for both boys and girls, Australia recommended vaccination for both sexes. Computer simulations have demonstrated that more than 80% female vaccination would achieve sufficient coverage and would be more cost-effective than vaccinating both males and females (224, 225). Also, it was calculated that with such high vaccine coverage, herd immunity would be enough to reduce HPV circulation (226). Currently, the U.S. Centers for Disease Control and Prevention (CDC) recommends HPV vaccination for females aged 11 to 12 years of age, with catch-up vaccination at 13 to 16 years of age, and the HPV vaccine was recently approved for use in boys and men aged 9 to 26 years to prevent genital warts (227). If Chlamydia vaccination programs were to be directed mainly at females, the possible rationale would be because Chlamydia-related morbidity and mortality are higher among women (infertility and ectopic pregnancy). A reason for male inclusion would be to further decrease Chlamydia trachomatis prevalence in the population and indirectly improve the protection of women. Since men and women are equally susceptible to genital chlamydial infection (228), a Chlamydia trachomatis vaccine for both sexes should at least be discussed. However, although recent studies have demonstrated that vaccinating both sexes has a beneficial impact on Chlamydia-related morbidity, similar to the case with the HPV vaccine, targeting women is more effective than targeting men (229). Chlamydia vaccination in groups with the highest risks of infection should be prioritized (multiple partners, sex workers, and immunocompromised individuals), independent of gender. Since a large proportion of HIV-positive homosexual men test positive for rectal LGV and non-LGV Chlamydia trachomatis (230), and since Chlamydia trachomatis is associated with an increased risk of HIV transmission (231), this group would also benefit from a Chlamydia vaccine. However, a chlamydial vaccine for these groups would be more for its therapeutic potential than as a prophylactic measure. Finally, who will pay for the cost of vaccination to prevent what is mostly a chronic silent infection? How do we convince decision-makers that the current epidemic of subfertility due to the silent C. trachomatis outbreak will have such a huge negative financial impact at the societal level that vaccine development should now be considered a public health priority?
CONCLUSIONS
Chlamydial infection is a public health concern worldwide, and a vaccine that stimulates multiple arms of the adaptive immune system and avoids immunopathological consequences would be the best solution for controlling this sexually transmitted disease. Unfortunately, a partial or fully protective vaccine has yet to be developed, highlighting the complex nature of the immunobiology mounted against this intracellular parasitic bacterium. The immune response to chlamydial infection is dynamic and involves cells and mediators from both arms of the host's immune system. Clearance of a chlamydial infection requires a coordinated immune response between innate immune cells, such as Mϕ, neutrophils, NK cells, NK T cells, and DC, and cells important in both cell-mediated and humoral adaptive responses, such as CD4+ T cells, CD8+ T cells, and B cells. Activation and clonal expansion of T cells occur through cognate interactions with DC that present chlamydial antigens on their MHC molecules, and B cells produce antichlamydial antibodies through interaction with these clonal T cells. However, persistent infection seems to induce chronic inflammation and tissue damage. A shift from Th1 to Th2 immunity also appears to induce scarring and immunopathology. It is therefore essential to understand these immunological dynamics in order to develop a vaccine that is both effective and long-lasting and does not have the deleterious effects associated with unregulated inflammation. Further research is needed to identify novel adjuvants that enhance the immune response and antigens that induce a protective T cell response and antichlamydial antibodies.
A mathematical model developed by Gray and colleagues demonstrated that a fully protective vaccine administered to adolescents before they are sexually active would be able to significantly decrease Chlamydia trachomatis infection in 20 years. In addition, the model predicted that vaccinating 100% of women would have a greater epidemiological impact than vaccinating both sexes (229). Unfortunately, there are risks and ethical questions associated with vaccination programs, as demonstrated by the first Chlamydia vaccine using a live attenuated bacterium (149). Thus, research is needed to develop an efficient and safe chlamydial vaccine.
ACKNOWLEDGMENT
This work was supported by the Fondation Leenaards through the Bourse pour la Relève Académique.
Biographies

Sam Vasilevsky, Ph.D., is an immunologist currently working at the University State Hospital in Lausanne, Switzerland (CHUV), investigating the roles that Chlamydia and Chlamydia-like bacteria (Waddlia chondrophila and Parachlamydia acanthameobae) play in adverse pregnancy outcomes. He received his Ph.D. at the Uniformed Services University in Bethesda, MD, where his work focused on innate and humoral immunity to Streptococcus pneumoniae. As a postdoctoral fellow at the National Institutes of Health (2009 to 2013), he investigated how phagocyte NADPH oxide regulates innate and humoral immunity in response to Listeria monocytogenes. His interests involve investigating innate and humoral immunity to both intracellular and extracellular bacteria.

Gilbert Greub, M.D., Ph.D., is a chlamydiologist leading a research group focusing on various strictly intracellular bacteria belonging to the Chlamydiales order. He is interested in their pathogenic role and in better understanding the biology of these strictly intracellular bacteria. As an infectious disease specialist and clinical microbiologist, Professor Greub is also Full Professor of Bacteriology at the University of Lausanne and Head of the Medical Diagnostic Microbiology Laboratories of the University Hospital Center, Lausanne, Switzerland.

Denise Nardelli-Haefliger is a Ph.D. researcher currently leading the Research Unit of Urology at the University State Hospital (CHUV) in Lausanne, Switzerland, focusing on immunotherapeutic approaches against urogenital cancers. Over the last 20 years, she has been involved in the development of prophylactic and therapeutic vaccines against genital human papillomavirus infections and associated cancers. In particular, she pioneered the use of mucosal vaccination routes, with a special interest in the induction and analysis of immune responses at mucosal genital sites in both murine models and women.

David Baud, M.D., Ph.D., is a specialist in maternofetal medicine and is presently working as a staff member at the University Hospital in Lausanne, Switzerland. He is board certified in obstetrics and gynecology and in operative gynecology. After completing his Ph.D. on the human papillomavirus vaccine, he trained at various hospitals worldwide, including Mount Sinai Hospital, Toronto, Canada (2010 to 2012), Hopital Necker, Paris, France (2009 to 2010), and St. Mary's Hospital, London, United Kingdom (2004 to 2005), with which he continues active research and clinical collaborations. His current research focuses on emerging infectious causes of adverse pregnancy outcomes, with a special interest in intracellular bacteria such as Chlamydia. Helped by his previous research in vaccine development (for his Ph.D.), his lab is also investigating a potential Chlamydia vaccine.
REFERENCES
- 1.Bebear C, de Barbeyrac B. 2009. Genital Chlamydia trachomatis infections. Clin. Microbiol. Infect. 15:4–10. 10.1111/j.1469-0691.2008.02647.x [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. 2008. Global incidence and prevalence of selected curable sexually transmitted infections. World Health Organization, Geneva, Switzerland [Google Scholar]
- 3.Baud D, Goy G, Jaton K, Osterheld MC, Blumer S, Borel N, Vial Y, Hohlfeld P, Pospischil A, Greub G. 2011. Role of Chlamydia trachomatis in miscarriage. Emerg. Infect. Dis. 17:1630–1635. 10.3201/eid1709.100865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Karaer A, Mert I, Cavkaytar S, Batioglu S. 2013. Serological investigation of the role of selected sexually transmitted infections in the aetiology of ectopic pregnancy. Eur. J. Contracept. Reprod. Health Care 18:68–74. 10.3109/13625187.2012.744818 [DOI] [PubMed] [Google Scholar]
- 5.Kavanagh K, Wallace LA, Robertson C, Wilson P, Scoular A. 2013. Estimation of the risk of tubal factor infertility associated with genital chlamydial infection in women: a statistical modelling study. Int. J. Epidemiol. 42:493–503. 10.1093/ije/dyt011 [DOI] [PubMed] [Google Scholar]
- 6.Gaydos CA, Wright C, Wood BJ, Waterfield G, Hobson S, Quinn TC. 2008. Chlamydia trachomatis reinfection rates among female adolescents seeking rescreening in school-based health centers. Sex. Transm. Dis. 35:233–237. 10.1097/OLQ.0b013e31815c11fe [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hosenfeld CB, Workowski KA, Berman S, Zaidi A, Dyson J, Mosure D, Bolan G, Bauer HM. 2009. Repeat infection with Chlamydia and gonorrhea among females: a systematic review of the literature. Sex. Transm. Dis. 36:478–489. 10.1097/OLQ.0b013e3181a2a933 [DOI] [PubMed] [Google Scholar]
- 8.Mariotti SP, Pascolini D, Rose-Nussbaumer J. 2009. Trachoma: global magnitude of a preventable cause of blindness. Br. J. Ophthalmol. 93:563–568. 10.1136/bjo.2008.148494 [DOI] [PubMed] [Google Scholar]
- 9.Senn L, Hammerschlag MR, Greub G. 2005. Therapeutic approaches to Chlamydia infections. Expert Opin. Pharmacother. 6:2281–2290. 10.1517/14656566.6.13.2281 [DOI] [PubMed] [Google Scholar]
- 10.Burton MJ, Mabey DC. 2009. The global burden of trachoma: a review. PLoS Negl. Trop. Dis. 3:e460. 10.1371/journal.pntd.0000460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hu VH, Holland MJ, Burton MJ. 2013. Trachoma: protective and pathogenic ocular immune responses to Chlamydia trachomatis. PLoS Negl. Trop. Dis. 7:e2020. 10.1371/journal.pntd.0002020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Peuchant O, Baldit C, Le Roy C, Trombert-Paolantoni S, Clerc M, Bebear C, de Barbeyrac B. 2011. First case of Chlamydia trachomatis L2b proctitis in a woman. Clin. Microbiol. Infect. 17:E21–E23. 10.1111/j.1469-0691.2011.03661.x [DOI] [PubMed] [Google Scholar]
- 13.Verweij SP, Ouburg S, de Vries H, Morre SA, van Ginkel CJ, Bos H, Sebens FW. 2012. The first case record of a female patient with bubonic lymphogranuloma venereum (LGV), serovariant L2b. Sex. Transm. Infect. 88:346–347. 10.1136/sextrans-2011-050298 [DOI] [PubMed] [Google Scholar]
- 14.Corsaro D, Greub G. 2006. Pathogenic potential of novel chlamydiae and diagnostic approaches to infections due to these obligate intracellular bacteria. Clin. Microbiol. Rev. 19:283–297. 10.1128/CMR.19.2.283-297.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang SP, Grayston JT. 1991. Three new serovars of Chlamydia trachomatis: Da, Ia, and L2a. J. Infect. Dis. 163:403–405. 10.1093/infdis/163.2.403 [DOI] [PubMed] [Google Scholar]
- 16.Baud D, Regan L, Greub G. 2008. Emerging role of Chlamydia and Chlamydia-like organisms in adverse pregnancy outcomes. Curr. Opin. Infect. Dis. 21:70–76. 10.1097/QCO.0b013e3282f3e6a5 [DOI] [PubMed] [Google Scholar]
- 17.Brunham RC, Rey-Ladino J. 2005. Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nat. Rev. Immunol. 5:149–161. 10.1038/nri1551 [DOI] [PubMed] [Google Scholar]
- 18.Wyrick PB. 2010. Chlamydia trachomatis persistence in vitro: an overview. J. Infect. Dis. 201(Suppl 2):S88–S95. 10.1086/652394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vergara MRC, Buendia Marin AJ, del Rio Alonzo L, Gijon FC, Hernandez NO, Ruiz MCG, Lorente JS. 2005. Chlamydia trachomatis genital infection: immunity and prospects for vaccine development. Immunologia 24:298–312 [Google Scholar]
- 20.Agrawal T, Bhengraj AR, Vats V, Salhan S, Mittal A. 2011. Expression of TLR 2, TLR 4 and iNOS in cervical monocytes of Chlamydia trachomatis-infected women and their role in host immune response. Am. J. Reprod. Immunol. 66:534–543. 10.1111/j.1600-0897.2011.01064.x [DOI] [PubMed] [Google Scholar]
- 21.Refaat B, Al-Azemi M, Geary I, Eley A, Ledger W. 2009. Role of activins and inducible nitric oxide in the pathogenesis of ectopic pregnancy in patients with or without Chlamydia trachomatis infection. Clin. Vaccine Immunol. 16:1493–1503. 10.1128/CVI.00221-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rusconi B, Greub G. 2011. Chlamydiales and the innate immune response: friend or foe? FEMS Immunol. Med. Microbiol. 61:231–244. 10.1111/j.1574-695X.2010.00772.x [DOI] [PubMed] [Google Scholar]
- 23.Register KB, Morgan PA, Wyrick PB. 1986. Interaction between Chlamydia spp. and human polymorphonuclear leukocytes in vitro. Infect. Immun. 52:664–670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yong EC, Klebanoff SJ, Kuo CC. 1982. Toxic effect of human polymorphonuclear leukocytes on Chlamydia trachomatis. Infect. Immun. 37:422–426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Barteneva N, Theodor I, Peterson EM, de la Maza LM. 1996. Role of neutrophils in controlling early stages of a Chlamydia trachomatis infection. Infect. Immun. 64:4830–4833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kolaczkowska E, Kubes P. 2013. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 13:159–175. 10.1038/nri3399 [DOI] [PubMed] [Google Scholar]
- 27.Salamone G, Giordano M, Trevani AS, Gamberale R, Vermeulen M, Schettinni J, Geffner JR. 2001. Promotion of neutrophil apoptosis by TNF-alpha. J. Immunol. 166:3476–3483 [DOI] [PubMed] [Google Scholar]
- 28.van Zandbergen G, Gieffers J, Kothe H, Rupp J, Bollinger A, Aga E, Klinger M, Brade H, Dalhoff K, Maass M, Solbach W, Laskay T. 2004. Chlamydia pneumoniae multiply in neutrophil granulocytes and delay their spontaneous apoptosis. J. Immunol. 172:1768–1776 [DOI] [PubMed] [Google Scholar]
- 29.Bradley LM, Douglass MF, Chatterjee D, Akira S, Baaten BJ. 2012. Matrix metalloprotease 9 mediates neutrophil migration into the airways in response to influenza virus-induced Toll-like receptor signaling. PLoS Pathog. 8:e1002641. 10.1371/journal.ppat.1002641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lee HY, Schripsema JH, Sigar IM, Murray CM, Lacy SR, Ramsey KH. 2010. A link between neutrophils and chronic disease manifestations of Chlamydia muridarum urogenital infection of mice. FEMS Immunol. Med. Microbiol. 59:108–116. 10.1111/j.1574-695X.2010.00668.x [DOI] [PubMed] [Google Scholar]
- 31.Cheng M, Chen Y, Xiao W, Sun R, Tian Z. 2013. NK cell-based immunotherapy for malignant diseases. Cell. Mol. Immunol. 10:230–252. 10.1038/cmi.2013.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shegarfi H, Sydnes K, Lovik M, Inngjerdingen M, Rolstad B, Naper C. 2009. The role of natural killer cells in resistance to the intracellular bacterium Listeria monocytogenes in rats. Scand. J. Immunol. 70:238–244. 10.1111/j.1365-3083.2009.02292.x [DOI] [PubMed] [Google Scholar]
- 33.Tseng CT, Rank RG. 1998. Role of NK cells in early host response to chlamydial genital infection. Infect. Immun. 66:5867–5875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hook CE, Matyszak MK, Gaston JS. 2005. Infection of epithelial and dendritic cells by Chlamydia trachomatis results in IL-18 and IL-12 production, leading to interferon-gamma production by human natural killer cells. FEMS Immunol. Med. Microbiol. 45:113–120. 10.1016/j.femsim.2005.02.010 [DOI] [PubMed] [Google Scholar]
- 35.Beatty WL, Belanger TA, Desai AA, Morrison RP, Byrne GI. 1994. Tryptophan depletion as a mechanism of gamma interferon-mediated chlamydial persistence. Infect. Immun. 62:3705–3711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jiao L, Gao X, Joyee AG, Zhao L, Qiu H, Yang M, Fan Y, Wang S, Yang X. 2011. NK cells promote type 1 T cell immunity through modulating the function of dendritic cells during intracellular bacterial infection. J. Immunol. 187:401–411. 10.4049/jimmunol.1002519 [DOI] [PubMed] [Google Scholar]
- 37.Zhao L, Gao X, Peng Y, Joyee AG, Bai H, Wang S, Yang J, Zhao W, Yang X. 2011. Differential modulating effect of natural killer (NK) T cells on interferon-gamma production and cytotoxic function of NK cells and its relationship with NK subsets in Chlamydia muridarum infection. Immunology 134:172–184. 10.1111/j.1365-2567.2011.03477.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Godfrey DI, Stankovic S, Baxter AG. 2010. Raising the NKT cell family. Nat. Immunol. 11:197–206. 10.1038/ni.1841 [DOI] [PubMed] [Google Scholar]
- 39.Carnaud C, Lee D, Donnars O, Park SH, Beavis A, Koezuka Y, Bendelac A. 1999. Cutting edge: cross-talk between cells of the innate immune system: NKT cells rapidly activate NK cells. J. Immunol. 163:4647–4650 [PubMed] [Google Scholar]
- 40.Joyee AG, Qiu H, Fan Y, Wang S, Yang X. 2008. Natural killer T cells are critical for dendritic cells to induce immunity in chlamydial pneumonia. Am. J. Respir. Crit. Care Med. 178:745–756. 10.1164/rccm.200804-517OC [DOI] [PubMed] [Google Scholar]
- 41.Tonti E, Galli G, Malzone C, Abrignani S, Casorati G, Dellabona P. 2009. NKT-cell help to B lymphocytes can occur independently of cognate interaction. Blood 113:370–376. 10.1182/blood-2008-06-166249 [DOI] [PubMed] [Google Scholar]
- 42.Joyee AG, Uzonna J, Yang X. 2010. Invariant NKT cells preferentially modulate the function of CD8 alpha+ dendritic cell subset in inducing type 1 immunity against infection. J. Immunol. 184:2095–2106. 10.4049/jimmunol.0901348 [DOI] [PubMed] [Google Scholar]
- 43.Bilenki L, Wang S, Yang J, Fan Y, Joyee AG, Yang X. 2005. NK T cell activation promotes Chlamydia trachomatis infection in vivo. J. Immunol. 175:3197–3206 [DOI] [PubMed] [Google Scholar]
- 44.Wang H, Zhao L, Peng Y, Liu J, Qi M, Chen Q, Yang X, Zhao W. 2012. Protective role of alpha-galactosylceramide-stimulated natural killer T cells in genital tract infection with Chlamydia muridarum. FEMS Immunol. Med. Microbiol. 65:43–54. 10.1111/j.1574-695X.2012.00939.x [DOI] [PubMed] [Google Scholar]
- 45.Morrison SG, Morrison RP. 2000. In situ analysis of the evolution of the primary immune response in murine Chlamydia trachomatis genital tract infection. Infect. Immun. 68:2870–2879. 10.1128/IAI.68.5.2870-2879.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Beagley KW, Huston WM, Hansbro PM, Timms P. 2009. Chlamydial infection of immune cells: altered function and implications for disease. Crit. Rev. Immunol. 29:275–305. 10.1615/CritRevImmunol.v29.i4.10 [DOI] [PubMed] [Google Scholar]
- 47.Bas S, Neff L, Vuillet M, Spenato U, Seya T, Matsumoto M, Gabay C. 2008. The proinflammatory cytokine response to Chlamydia trachomatis elementary bodies in human macrophages is partly mediated by a lipoprotein, the macrophage infectivity potentiator, through TLR2/TLR1/TLR6 and CD14. J. Immunol. 180:1158–1168 [DOI] [PubMed] [Google Scholar]
- 48.Yilma AN, Singh SR, Fairley SJ, Taha MA, Dennis VA. 2012. The anti-inflammatory cytokine, interleukin-10, inhibits inflammatory mediators in human epithelial cells and mouse macrophages exposed to live and UV-inactivated Chlamydia trachomatis. Mediators Inflamm. 2012:520174. 10.1155/2012/520174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sun HS, Eng EW, Jeganathan S, Sin AT, Patel PC, Gracey E, Inman RD, Terebiznik MR, Harrison RE. 2012. Chlamydia trachomatis vacuole maturation in infected macrophages. J. Leukoc. Biol. 92:815–827. 10.1189/jlb.0711336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Al-Zeer MA, Al-Younes HM, Lauster D, Abu Lubad M, Meyer TF. 2013. Autophagy restricts Chlamydia trachomatis growth in human macrophages via IFNG-inducible guanylate binding proteins. Autophagy 9:50–62. 10.4161/auto.22482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yasir M, Pachikara ND, Bao X, Pan Z, Fan H. 2011. Regulation of chlamydial infection by host autophagy and vacuolar ATPase-bearing organelles. Infect. Immun. 79:4019–4028. 10.1128/IAI.05308-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Crotzer VL, Blum JS. 2009. Autophagy and its role in MHC-mediated antigen presentation. J. Immunol. 182:3335–3341. 10.4049/jimmunol.0803458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cao H, Wolff RG, Meltzer MS, Crawford RM. 1989. Differential regulation of class II MHC determinants on macrophages by IFN-gamma and IL-4. J. Immunol. 143:3524–3531 [PubMed] [Google Scholar]
- 54.Vasilevsky S, Colino J, Puliaev R, Canaday DH, Snapper CM. 2008. Macrophages pulsed with Streptococcus pneumoniae elicit a T cell-dependent antibody response upon transfer into naive mice. J. Immunol. 181:1787–1797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jendro MC, Deutsch T, Korber B, Kohler L, Kuipers JG, Krausse-Opatz B, Westermann J, Raum E, Zeidler H. 2000. Infection of human monocyte-derived macrophages with Chlamydia trachomatis induces apoptosis of T cells: a potential mechanism for persistent infection. Infect. Immun. 68:6704–6711. 10.1128/IAI.68.12.6704-6711.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jendro MC, Fingerle F, Deutsch T, Liese A, Kohler L, Kuipers JG, Raum E, Martin M, Zeidler H. 2004. Chlamydia trachomatis-infected macrophages induce apoptosis of activated T cells by secretion of tumor necrosis factor-alpha in vitro. Med. Microbiol. Immunol. 193:45–52. 10.1007/s00430-003-0182-1 [DOI] [PubMed] [Google Scholar]
- 57.Jiang X, Shen C, Rey-Ladino J, Yu H, Brunham RC. 2008. Characterization of murine dendritic cell line JAWS II and primary bone marrow-derived dendritic cells in Chlamydia muridarum antigen presentation and induction of protective immunity. Infect. Immun. 76:2392–2401. 10.1128/IAI.01584-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Matyszak MK, Young JL, Gaston JS. 2002. Uptake and processing of Chlamydia trachomatis by human dendritic cells. Eur. J. Immunol. 32:742–751. [DOI] [PubMed] [Google Scholar]
- 59.Morrison RP, Feilzer K, Tumas DB. 1995. Gene knockout mice establish a primary protective role for major histocompatibility complex class II-restricted responses in Chlamydia trachomatis genital tract infection. Infect. Immun. 63:4661–4668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ojcius DM, Bravo de Alba Y, Kanellopoulos JM, Hawkins RA, Kelly KA, Rank RG, Dautry-Varsat A. 1998. Internalization of Chlamydia by dendritic cells and stimulation of Chlamydia-specific T cells. J. Immunol. 160:1297–1303 [PubMed] [Google Scholar]
- 61.Su H, Messer R, Whitmire W, Fischer E, Portis JC, Caldwell HD. 1998. Vaccination against chlamydial genital tract infection after immunization with dendritic cells pulsed ex vivo with nonviable Chlamydiae. J. Exp. Med. 188:809–818. 10.1084/jem.188.5.809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lu H, Zhong G. 1999. Interleukin-12 production is required for chlamydial antigen-pulsed dendritic cells to induce protection against live Chlamydia trachomatis infection. Infect. Immun. 67:1763–1769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shaw J, Grund V, Durling L, Crane D, Caldwell HD. 2002. Dendritic cells pulsed with a recombinant chlamydial major outer membrane protein antigen elicit a CD4(+) type 2 rather than type 1 immune response that is not protective. Infect. Immun. 70:1097–1105. 10.1128/IAI.70.3.1097-1105.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.He Q, Moore TT, Eko FO, Lyn D, Ananaba GA, Martin A, Singh S, James L, Stiles J, Black CM, Igietseme JU. 2005. Molecular basis for the potency of IL-10-deficient dendritic cells as a highly efficient APC system for activating Th1 response. J. Immunol. 174:4860–4869 [DOI] [PubMed] [Google Scholar]
- 65.Rey-Ladino J, Koochesfahani KM, Zaharik ML, Shen C, Brunham RC. 2005. A live and inactivated Chlamydia trachomatis mouse pneumonitis strain induces the maturation of dendritic cells that are phenotypically and immunologically distinct. Infect. Immun. 73:1568–1577. 10.1128/IAI.73.3.1568-1577.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yu H, Karunakaran KP, Kelly I, Shen C, Jiang X, Foster LJ, Brunham RC. 2011. Immunization with live and dead Chlamydia muridarum induces different levels of protective immunity in a murine genital tract model: correlation with MHC class II peptide presentation and multifunctional Th1 cells. J. Immunol. 186:3615–3621. 10.4049/jimmunol.1002952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ibana JA, Schust DJ, Sugimoto J, Nagamatsu T, Greene SJ, Quayle AJ. 2011. Chlamydia trachomatis immune evasion via downregulation of MHC class I surface expression involves direct and indirect mechanisms. Infect. Dis. Obstet. Gynecol. 2011:420905. 10.1155/2011/420905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Dong F, Pirbhai M, Zhong Y, Zhong G. 2004. Cleavage-dependent activation of a chlamydia-secreted protease. Mol. Microbiol. 52:1487–1494. 10.1111/j.1365-2958.2004.04072.x [DOI] [PubMed] [Google Scholar]
- 69.Peschel G, Kernschmidt L, Cirl C, Wantia N, Ertl T, Durr S, Wagner H, Miethke T, Rodriguez N. 2010. Chlamydophila pneumoniae downregulates MHC-class II expression by two cell type-specific mechanisms. Mol. Microbiol. 76:648–661. 10.1111/j.1365-2958.2010.07114.x [DOI] [PubMed] [Google Scholar]
- 70.Zhang XL, Tsui IS, Yip CM, Fung AW, Wong DK, Dai X, Yang Y, Hackett J, Morris C. 2000. Salmonella enterica serovar Typhi uses type IVB pili to enter human intestinal epithelial cells. Infect. Immun. 68:3067–3073. 10.1128/IAI.68.6.3067-3073.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhong G, Fan P, Ji H, Dong F, Huang Y. 2001. Identification of a chlamydial protease-like activity factor responsible for the degradation of host transcription factors. J. Exp. Med. 193:935–942. 10.1084/jem.193.8.935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lu H, Wang H, Zhao HM, Zhao L, Chen Q, Qi M, Liu J, Yu H, Yu XP, Yang X, Zhao WM. 2010. Dendritic cells (DCs) transfected with a recombinant adenovirus carrying chlamydial major outer membrane protein antigen elicit protective immune responses against genital tract challenge infection. Biochem. Cell Biol. 88:757–765. 10.1139/O10-011 [DOI] [PubMed] [Google Scholar]
- 73.Li W, Murthy AK, Chaganty BK, Guentzel MN, Seshu J, Chambers JP, Zhong G, Arulanandam BP. 2011. Immunization with dendritic cells pulsed ex vivo with recombinant chlamydial protease-like activity factor induces protective immunity against genital Chlamydia muridarum challenge. Front. Immunol. 2:73. 10.3389/fimmu.2011.00073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Rank RG, Soderberg LS, Barron AL. 1985. Chronic chlamydial genital infection in congenitally athymic nude mice. Infect. Immun. 48:847–849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Johansson M, Lycke N. 2001. Immunological memory in B-cell-deficient mice conveys long-lasting protection against genital tract infection with Chlamydia trachomatis by rapid recruitment of T cells. Immunology 102:199–208. 10.1046/j.1365-2567.2001.01167.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kelly KA, Walker JC, Jameel SH, Gray HL, Rank RG. 2000. Differential regulation of CD4 lymphocyte recruitment between the upper and lower regions of the genital tract during Chlamydia trachomatis infection. Infect. Immun. 68:1519–1528. 10.1128/IAI.68.3.1519-1528.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Olive AJ, Gondek DC, Starnbach MN. 2011. CXCR3 and CCR5 are both required for T cell-mediated protection against C. trachomatis infection in the murine genital mucosa. Mucosal Immunol. 4:208–216. 10.1038/mi.2010.58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Van Voorhis WC, Barrett LK, Sweeney YT, Kuo CC, Patton DL. 1996. Analysis of lymphocyte phenotype and cytokine activity in the inflammatory infiltrates of the upper genital tract of female macaques infected with Chlamydia trachomatis. J. Infect. Dis. 174:647–650. 10.1093/infdis/174.3.647 [DOI] [PubMed] [Google Scholar]
- 79.Goodall JC, Beacock-Sharp H, Deane KH, Gaston JS. 2001. Recognition of the 60 kilodalton cysteine-rich outer membrane protein OMP2 by CD4(+) T cells from humans infected with Chlamydia trachomatis. Clin. Exp. Immunol. 126:488–493. 10.1046/j.1365-2249.2001.01709.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Goodall JC, Yeo G, Huang M, Raggiaschi R, Gaston JS. 2001. Identification of Chlamydia trachomatis antigens recognized by human CD4+ T lymphocytes by screening an expression library. Eur. J. Immunol. 31:1513–1522. [DOI] [PubMed] [Google Scholar]
- 81.Holland MJ, Conway DJ, Blanchard TJ, Mahdi OM, Bailey RL, Whittle HC, Mabey DC. 1997. Synthetic peptides based on Chlamydia trachomatis antigens identify cytotoxic T lymphocyte responses in subjects from a trachoma-endemic population. Clin. Exp. Immunol. 107:44–49. 10.1046/j.1365-2249.1997.2511129.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kim SK, Devine L, Angevine M, DeMars R, Kavathas PB. 2000. Direct detection and magnetic isolation of Chlamydia trachomatis major outer membrane protein-specific CD8+ CTLs with HLA class I tetramers. J. Immunol. 165:7285–7292 [DOI] [PubMed] [Google Scholar]
- 83.Ortiz L, Angevine M, Kim SK, Watkins D, DeMars R. 2000. T-cell epitopes in variable segments of Chlamydia trachomatis major outer membrane protein elicit serovar-specific immune responses in infected humans. Infect. Immun. 68:1719–1723. 10.1128/IAI.68.3.1719-1723.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Deane KH, Jecock RM, Pearce JH, Gaston JS. 1997. Identification and characterization of a DR4-restricted T cell epitope within chlamydia heat shock protein 60. Clin. Exp. Immunol. 109:439–445. 10.1046/j.1365-2249.1997.4711371.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Johnson RM, Yu H, Kerr MS, Slaven JE, Karunakaran KP, Brunham RC. 2012. PmpG303-311, a protective vaccine epitope that elicits persistent cellular immune responses in Chlamydia muridarum-immune mice. Infect. Immun. 80:2204–2211. 10.1128/IAI.06339-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chen L, Lei L, Zhou Z, He J, Xu S, Lu C, Chen J, Yang Z, Wu G, Yeh IT, Zhong G, Wu Y. 2013. Contribution of interleukin-12 p35 (IL-12p35) and IL-12p40 to protective immunity and pathology in mice infected with Chlamydia muridarum. Infect. Immun. 81:2962–2971. 10.1128/IAI.00161-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.O'Connell CM, Ingalls RR, Andrews CW, Jr, Scurlock AM, Darville T. 2007. Plasmid-deficient Chlamydia muridarum fail to induce immune pathology and protect against oviduct disease. J. Immunol. 179:4027–4034 [DOI] [PubMed] [Google Scholar]
- 88.Olivares-Zavaleta N, Whitmire W, Gardner D, Caldwell HD. 2010. Immunization with the attenuated plasmidless Chlamydia trachomatis L2(25667R) strain provides partial protection in a murine model of female genitourinary tract infection. Vaccine 28:1454–1462. 10.1016/j.vaccine.2009.11.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Vicetti Miguel RD, Harvey SA, LaFramboise WA, Reighard SD, Matthews DB, Cherpes TL. 2013. Human female genital tract infection by the obligate intracellular bacterium Chlamydia trachomatis elicits robust type 2 immunity. PLoS One 8:e58565. 10.1371/journal.pone.0058565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Farris CM, Morrison SG, Morrison RP. 2010. CD4+ T cells and antibody are required for optimal major outer membrane protein vaccine-induced immunity to Chlamydia muridarum genital infection. Infect. Immun. 78:4374–4383. 10.1128/IAI.00622-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Finco O, Frigimelica E, Buricchi F, Petracca R, Galli G, Faenzi E, Meoni E, Bonci A, Agnusdei M, Nardelli F, Bartolini E, Scarselli M, Caproni E, Laera D, Zedda L, Skibinski D, Giovinazzi S, Bastone R, Ianni E, Cevenini R, Grandi G, Grifantini R. 2011. Approach to discover T- and B-cell antigens of intracellular pathogens applied to the design of Chlamydia trachomatis vaccines. Proc. Natl. Acad. Sci. U. S. A. 108:9969–9974. 10.1073/pnas.1101756108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gondek DC, Olive AJ, Stary G, Starnbach MN. 2012. CD4+ T cells are necessary and sufficient to confer protection against Chlamydia trachomatis infection in the murine upper genital tract. J. Immunol. 189:2441–2449. 10.4049/jimmunol.1103032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jayarapu K, Kerr M, Ofner S, Johnson RM. 2010. Chlamydia-specific CD4 T cell clones control Chlamydia muridarum replication in epithelial cells by nitric oxide-dependent and -independent mechanisms. J. Immunol. 185:6911–6920. 10.4049/jimmunol.1002596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wizel B, Nystrom-Asklin J, Cortes C, Tvinnereim A. 2008. Role of CD8(+)T cells in the host response to Chlamydia. Microbes Infect. 10:1420–1430. 10.1016/j.micinf.2008.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Murthy AK, Li W, Chaganty BK, Kamalakaran S, Guentzel MN, Seshu J, Forsthuber TG, Zhong G, Arulanandam BP. 2011. Tumor necrosis factor alpha production from CD8+ T cells mediates oviduct pathological sequelae following primary genital Chlamydia muridarum infection. Infect. Immun. 79:2928–2935. 10.1128/IAI.05022-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Su H, Caldwell HD. 1995. CD4+ T cells play a significant role in adoptive immunity to Chlamydia trachomatis infection of the mouse genital tract. Infect. Immun. 63:3302–3308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ibana JA, Myers L, Porretta C, Lewis M, Taylor SN, Martin DH, Quayle AJ. 2012. The major CD8 T cell effector memory subset in the normal and Chlamydia trachomatis-infected human endocervix is low in perforin. BMC Immunol. 13:66. 10.1186/1471-2172-13-66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Barenfanger J, MacDonald AB. 1974. The role of immunoglobulin in the neutralization of trachoma infectivity. J. Immunol. 113:1607–1617 [PubMed] [Google Scholar]
- 99.Jawetz E, Rose L, Hanna L, Thygeson P. 1965. Experimental inclusion conjunctivitis in man: measurements of infectivity and resistance. JAMA 194:620–632 [PubMed] [Google Scholar]
- 100.Bartolini E, Ianni E, Frigimelica E, Petracca R, Galli G, Scorza FB, Norais N, Laera D, Giusti F, Pierleoni A, Donati M, Cevenini R, Finco O, Grandi G, Grifantini R. 2013. Recombinant outer membrane vesicles carrying Chlamydia muridarum HtrA induce antibodies that neutralize chlamydial infection in vitro. J. Extracell. Vesicles 2013:2. 10.3402/jev.v2i0.20181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Byrne GI, Stephens RS, Ada G, Caldwell HD, Su H, Morrison RP, Van der Pol B, Bavoil P, Bobo L, Everson S, Ho Y, Hsia RC, Kenendy K, Kuo C-C, Montgomery PC, Peterson E, Swanson A, Whitaker C, Whittum Hudson J, Yang CL, Zhang Y-X, Zhong GM. 1993. Workshop on in vitro neutralization of Chlamydia trachomatis: summary of proceedings. J. Infect. Dis. 168:415–420. 10.1093/infdis/168.2.415 [DOI] [PubMed] [Google Scholar]
- 102.Morrison SG, Morrison RP. 2005. A predominant role for antibody in acquired immunity to chlamydial genital tract reinfection. J. Immunol. 175:7536–7542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Murthy AK, Chaganty BK, Li W, Guentzel MN, Chambers JP, Seshu J, Zhong G, Arulanandam BP. 2009. A limited role for antibody in protective immunity induced by rCPAF and CpG vaccination against primary genital Chlamydia muridarum challenge. FEMS Immunol. Med. Microbiol. 55:271–279. 10.1111/j.1574-695X.2008.00517.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Moore T, Ananaba GA, Bolier J, Bowers S, Belay T, Eko FO, Igietseme JU. 2002. Fc receptor regulation of protective immunity against Chlamydia trachomatis. Immunology 105:213–221. 10.1046/j.0019-2805.2001.01354.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Igietseme JU, Eko FO, He Q, Black CM. 2004. Antibody regulation of T cell immunity: implications for vaccine strategies against intracellular pathogens. Expert Rev. Vaccines 3:23–34. 10.1586/14760584.3.1.23 [DOI] [PubMed] [Google Scholar]
- 106.Verweij SP, Bax CJ, Quint KD, Quint WG, van Leeuwen AP, Peters RP, Oostvogel PM, Mutsaers JA, Dorr PJ, Pleijster J, Ouburg S, Morre SA. 2009. Significantly higher serologic responses of Chlamydia trachomatis B group serovars versus C and I serogroups. Drugs Today (Barc.) 45(Suppl B):135–140 [PubMed] [Google Scholar]
- 107.Bachmaier K, Neu N, de la Maza LM, Pal S, Hessel A, Penninger JM. 1999. Chlamydia infections and heart disease linked through antigenic mimicry. Science 283:1335–1339. 10.1126/science.283.5406.1335 [DOI] [PubMed] [Google Scholar]
- 108.Domeika M, Domeika K, Paavonen J, Mardh PA, Witkin SS. 1998. Humoral immune response to conserved epitopes of Chlamydia trachomatis and human 60-kDa heat-shock protein in women with pelvic inflammatory disease. J. Infect. Dis. 177:714–719. 10.1086/514218 [DOI] [PubMed] [Google Scholar]
- 109.Witkin SS. 2002. Immunological aspects of genital chlamydia infections. Best Pract. Res. Clin. Obstet. Gynaecol. 16:865–874. 10.1053/beog.2002.0326 [DOI] [PubMed] [Google Scholar]
- 110.Hjelholt A, Christiansan G, Johansson TG, Ingerslev HJ, Birkelund S. 2011. Tubal factor infertility is associated with antibodies against Chlamydia trachomatis heat shock protein 60 (HSP60) but not human HSP60. Human Reprod. 26:2069–2076. 10.1093/humrep/der167 [DOI] [PubMed] [Google Scholar]
- 111.Fernandez-Boyanapalli R, McPhillips KA, Frasch SC, Janssen WJ, Dinauer MC, Riches DW, Henson PM, Byrne A, Bratton DL. 2010. Impaired phagocytosis of apoptotic cells by macrophages in chronic granulomatous disease is reversed by IFN-gamma in a nitric oxide-dependent manner. J. Immunol. 185:4030–4041. 10.4049/jimmunol.1001778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhong GM, de la Maza LM. 1988. Activation of mouse peritoneal macrophages in vitro or in vivo by recombinant murine gamma interferon inhibits the growth of Chlamydia trachomatis serovar L1. Infect. Immun. 56:3322–3325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Al-Younes HM, Rudel T, Brinkmann V, Szczepek AJ, Meyer TF. 2001. Low iron availability modulates the course of Chlamydia pneumoniae infection. Cell. Microbiol. 3:427–437. 10.1046/j.1462-5822.2001.00125.x [DOI] [PubMed] [Google Scholar]
- 114.Ryu SY, Jeong KS, Kang BN, Park SJ, Yoon WK, Kim SH, Kim TH. 2000. Modulation of transferrin synthesis, transferrin receptor expression, iNOS expression and NO production in mouse macrophages by cytokines, either alone or in combination. Anticancer Res. 20:3331–3338 [PubMed] [Google Scholar]
- 115.Nairz M, Fritsche G, Brunner P, Talasz H, Hantke K, Weiss G. 2008. Interferon-gamma limits the availability of iron for intramacrophage Salmonella typhimurium. Eur. J. Immunol. 38:1923–1936. 10.1002/eji.200738056 [DOI] [PubMed] [Google Scholar]
- 116.Akers JC, Tan M. 2006. Molecular mechanism of tryptophan-dependent transcriptional regulation in Chlamydia trachomatis. J. Bacteriol. 188:4236–4243. 10.1128/JB.01660-05 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang Y, Wang H, Ren J, Tang X, Jing Y, Xing D, Zhao G, Yao Z, Yang X, Bai H. 2012. IL-17A synergizes with IFN-gamma to upregulate iNOS and NO production and inhibit chlamydial growth. PLoS One 7:e39214. 10.1371/journal.pone.0039214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.De Clercq E, Kalmar I, Vanrompay D. 2013. Animal models for studying female genital tract infection with Chlamydia trachomatis. Infect. Immun. 81:3060–3067. 10.1128/IAI.00357-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Riley MM, Zurenski MA, Frazer LC, O'Connell CM, Andrews CW, Jr, Mintus M, Darville T. 2012. The recall response induced by genital challenge with Chlamydia muridarum protects the oviduct from pathology but not from reinfection. Infect. Immun. 80:2194–2203. 10.1128/IAI.00169-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Shah AA, Schripsema JH, Imtiaz MT, Sigar IM, Kasimos J, Matos PG, Inouye S, Ramsey KH. 2005. Histopathologic changes related to fibrotic oviduct occlusion after genital tract infection of mice with Chlamydia muridarum. Sex. Transm. Dis. 32:49–56. 10.1097/01.olq.0000148299.14513.11 [DOI] [PubMed] [Google Scholar]
- 121.Tang L, Zhang H, Lei L, Gong S, Zhou Z, Baseman J, Zhong G. 2013. Oviduct infection and hydrosalpinx in DBA1/j mice is induced by intracervical but not intravaginal inoculation with Chlamydia muridarum. PLoS One 8:e71649. 10.1371/journal.pone.0071649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Farris CM, Morrison RP. 2011. Vaccination against Chlamydia genital infection utilizing the murine C. muridarum model. Infect. Immun. 79:986–996. 10.1128/IAI.00881-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rank RG, Whittum-Hudson JA. 2010. Protective immunity to chlamydial genital infection: evidence from animal studies. J. Infect. Dis. 201(Suppl 2):S168–S177. 10.1086/652399 [DOI] [PubMed] [Google Scholar]
- 124.Carmichael JR, Pal S, Tifrea D, de la Maza LM. 2011. Induction of protection against vaginal shedding and infertility by a recombinant Chlamydia vaccine. Vaccine 29:5276–5283. 10.1016/j.vaccine.2011.05.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lyons JM, Ito JI, Jr, Pena AS, Morre SA. 2005. Differences in growth characteristics and elementary body associated cytotoxicity between Chlamydia trachomatis oculogenital serovars D and H and Chlamydia muridarum. J. Clin. Pathol. 58:397–401. 10.1136/jcp.2004.021543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ripa KT, Moller BR, Mardh PA, Freundt EA, Melsen F. 1979. Experimental acute salpingitis in grivet monkeys provoked by Chlamydia trachomatis. Acta Pathol. Microbiol. Scand. B 87B:65–70 [DOI] [PubMed] [Google Scholar]
- 127.Johnson AP, Hetherington CM, Osborn MF, Thomas BJ, Taylor-Robinson D. 1980. Experimental infection of the marmoset genital tract with Chlamydia trachomatis. Br. J. Exp. Pathol. 61:291–295 [PMC free article] [PubMed] [Google Scholar]
- 128.Bell JD, Bergin IL, Harris LH, Chai D, Mullei I, Mwenda J, Dalton VK, Vahratian A, Lebar W, Zochowski MK, Kiulia N, Aronoff DM, Patton DL. 2011. The effects of a single cervical inoculation of Chlamydia trachomatis on the female reproductive tract of the baboon (Papio anubis). J. Infect. Dis. 204:1305–1312. 10.1093/infdis/jir541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Patton DL, Sweeney YT, Paul KJ. 2009. A summary of preclinical topical microbicide rectal safety and efficacy evaluations in a pigtailed macaque model. Sex. Transm. Dis. 36:350–356. 10.1097/OLQ.0b013e318195c31a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Patton DL, Kuo CC, Wang SP, Halbert SA. 1987. Distal tubal obstruction induced by repeated Chlamydia trachomatis salpingeal infections in pig-tailed macaques. J. Infect. Dis. 155:1292–1299. 10.1093/infdis/155.6.1292 [DOI] [PubMed] [Google Scholar]
- 131.Cunningham KA, Beagley KW. 2008. Male genital tract chlamydial infection: implications for pathology and infertility. Biol. Reprod. 79:180–189. 10.1095/biolreprod.108.067835 [DOI] [PubMed] [Google Scholar]
- 132.Miyairi I, Ramsey KH, Patton DL. 2010. Duration of untreated chlamydial genital infection and factors associated with clearance: review of animal studies. J. Infect. Dis. 201(Suppl 2):S96–S103. 10.1086/652393 [DOI] [PubMed] [Google Scholar]
- 133.Rank RG, Sanders MM, Kidd AT. 1993. Influence of the estrous cycle on the development of upper genital tract pathology as a result of chlamydial infection in the guinea pig model of pelvic inflammatory disease. Am. J. Pathol. 142:1291–1296 [PMC free article] [PubMed] [Google Scholar]
- 134.Ficarra M, Ibana JS, Poretta C, Ma L, Myers L, Taylor SN, Greene S, Smith B, Hagensee M, Martin DH, Quayle AJ. 2008. A distinct cellular profile is seen in the human endocervix during Chlamydia trachomatis infection. Am. J. Reprod. Immunol. 60:415–425. 10.1111/j.1600-0897.2008.00639.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Rank RG, Bowlin AK, Kelly KA. 2000. Characterization of lymphocyte response in the female genital tract during ascending chlamydial genital infection in the guinea pig model. Infect. Immun. 68:5293–5298. 10.1128/IAI.68.9.5293-5298.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tuggle CK, Green JA, Fitzsimmons C, Woods R, Prather RS, Malchenko S, Soares BM, Kucaba T, Crouch K, Smith C, Tack D, Robinson N, O'Leary B, Scheetz T, Casavant T, Pomp D, Edeal BJ, Zhang Y, Rothschild MF, Garwood K, Beavis W. 2003. EST-based gene discovery in pig: virtual expression patterns and comparative mapping to human. Mamm. Genome 14:565–579. 10.1007/s00335-002-2263-7 [DOI] [PubMed] [Google Scholar]
- 137.Vanrompay D, Hoang TQ, De Vos L, Verminnen K, Harkinezhad T, Chiers K, Morre SA, Cox E. 2005. Specific-pathogen-free pigs as an animal model for studying Chlamydia trachomatis genital infection. Infect. Immun. 73:8317–8321. 10.1128/IAI.73.12.8317-8321.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Schautteet K, De Clercq E, Jonsson Y, Lagae S, Chiers K, Cox E, Vanrompay D. 2012. Protection of pigs against genital Chlamydia trachomatis challenge by parenteral or mucosal DNA immunization. Vaccine 30:2869–2881. 10.1016/j.vaccine.2012.02.044 [DOI] [PubMed] [Google Scholar]
- 139.Schautteet K, Stuyven E, Beeckman DS, Van Acker S, Carlon M, Chiers K, Cox E, Vanrompay D. 2011. Protection of pigs against Chlamydia trachomatis challenge by administration of a MOMP-based DNA vaccine in the vaginal mucosa. Vaccine 29:1399–1407. 10.1016/j.vaccine.2010.12.042 [DOI] [PubMed] [Google Scholar]
- 140.Schautteet K, Stuyven E, Cox E, Vanrompay D. 2011. Validation of the Chlamydia trachomatis genital challenge pig model for testing recombinant protein vaccines. J. Med. Microbiol. 60:117–127. 10.1099/jmm.0.024448-0 [DOI] [PubMed] [Google Scholar]
- 141.Wolner-Hanssen P, Patton DL, Holmes KK. 1991. Protective immunity in pig-tailed macaques after cervical infection with Chlamydia trachomatis. Sex. Transm. Dis. 18:21–25. 10.1097/00007435-199101000-00005 [DOI] [PubMed] [Google Scholar]
- 142.Geisler WM. 2010. Duration of untreated, uncomplicated Chlamydia trachomatis genital infection and factors associated with chlamydia resolution: a review of human studies. J. Infect. Dis. 201(Suppl 2):S104–S113. 10.1086/652402 [DOI] [PubMed] [Google Scholar]
- 143.Molano M, Meijer CJ, Weiderpass E, Arslan A, Posso H, Franceschi S, Ronderos M, Munoz N, van den Brule AJ. 2005. The natural course of Chlamydia trachomatis infection in asymptomatic Colombian women: a 5-year follow-up study. J. Infect. Dis. 191:907–916. 10.1086/428287 [DOI] [PubMed] [Google Scholar]
- 144.Batteiger BE, Xu F, Johnson RE, Rekart ML. 2010. Protective immunity to Chlamydia trachomatis genital infection: evidence from human studies. J. Infect. Dis. 201(Suppl 2):S178–S189. 10.1086/652400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Huston WM, Harvie M, Mittal A, Timms P, Beagley KW. 2012. Vaccination to protect against infection of the female reproductive tract. Expert Rev. Clin. Immunol. 8:81–94. 10.1586/eci.11.80 [DOI] [PubMed] [Google Scholar]
- 146.Entrican G, Wheelhouse N, Wattegedera SR, Longbottom D. 2012. New challenges for vaccination to prevent chlamydial abortion in sheep. Comp. Immunol. Microbiol. Infect. Dis. 35:271–276. 10.1016/j.cimid.2011.12.001 [DOI] [PubMed] [Google Scholar]
- 147.Igietseme JU, Black CM. 2013. Chlamydial infection: a clinical and public health perspective. S. Karger AG, Basel, Switzerland [Google Scholar]
- 148.Meeusen EN, Walker J, Peters A, Pastoret PP, Jungersen G. 2007. Current status of veterinary vaccines. Clin. Microbiol. Rev. 20:489–510. 10.1128/CMR.00005-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Grayston JT, Wang SP. 1978. The potential for vaccine against infection of the genital tract with Chlamydia trachomatis. Sex. Transm. Dis. 5:73–77. 10.1097/00007435-197804000-00011 [DOI] [PubMed] [Google Scholar]
- 150.Brunham RC. 2013. Immunity to Chlamydia trachomatis. J. Infect. Dis. 207:1796–1797. 10.1093/infdis/jit095 [DOI] [PubMed] [Google Scholar]
- 151.Schautteet K, De Clercq E, Vanrompay D. 2011. Chlamydia trachomatis vaccine research through the years. Infect. Dis. Obstet. Gynecol. 2011:963513. 10.1155/2011/963513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Grayston JT, Woolridge RL, Wang SP, Yen CH, Yang CY, Cheng KH, Chang IH. 1963. Field studies of protection from infection by experimental trachoma virus vaccine in preschool-aged children on Taiwan. Proc. Soc. Exp. Biol. Med. 112:589–595. 10.3181/00379727-112-28112 [DOI] [PubMed] [Google Scholar]
- 153.Sowa S, Sowa J, Collier LH, Blyth WA. 1969. Trachoma vaccine field trials in The Gambia. J. Hyg. (Lond.) 67:699–717. 10.1017/S0022172400042157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Detmer A, Glenting J. 2006. Live bacterial vaccines—a review and identification of potential hazards. Microb. Cell Fact. 5:23. 10.1186/1475-2859-5-23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Nunes A, Nogueira PJ, Borrego MJ, Gomes JP. 2010. Adaptive evolution of the Chlamydia trachomatis dominant antigen reveals distinct evolutionary scenarios for B- and T-cell epitopes: worldwide survey. PLoS One 5:e13171. 10.1371/journal.pone.0013171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Pal S, Theodor I, Peterson EM, de la Maza LM. 1997. Immunization with an acellular vaccine consisting of the outer membrane complex of Chlamydia trachomatis induces protection against a genital challenge. Infect. Immun. 65:3361–3369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Pal S, Theodor I, Peterson EM, de la Maza LM. 2001. Immunization with the Chlamydia trachomatis mouse pneumonitis major outer membrane protein can elicit a protective immune response against a genital challenge. Infect. Immun. 69:6240–6247. 10.1128/IAI.69.10.6240-6247.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Tifrea DF, Sun G, Pal S, Zardeneta G, Cocco MJ, Popot JL, de la Maza LM. 2011. Amphipols stabilize the Chlamydia major outer membrane protein and enhance its protective ability as a vaccine. Vaccine 29:4623–4631. 10.1016/j.vaccine.2011.04.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Igietseme JU, Murdin A. 2000. Induction of protective immunity against Chlamydia trachomatis genital infection by a vaccine based on major outer membrane protein-lipophilic immune response-stimulating complexes. Infect. Immun. 68:6798–6806. 10.1128/IAI.68.12.6798-6806.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Cheng C, Bettahi I, Cruz-Fisher MI, Pal S, Jain P, Jia Z, Holmgren J, Harandi AM, de la Maza LM. 2009. Induction of protective immunity by vaccination against Chlamydia trachomatis using the major outer membrane protein adjuvanted with CpG oligodeoxynucleotide coupled to the nontoxic B subunit of cholera toxin. Vaccine 27:6239–6246. 10.1016/j.vaccine.2009.07.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Cheng C, Pal S, Bettahi I, Oxford KL, Barry PA, de la Maza LM. 2011. Immunogenicity of a vaccine formulated with the Chlamydia trachomatis serovar F, native major outer membrane protein in a nonhuman primate model. Vaccine 29:3456–3464. 10.1016/j.vaccine.2011.02.057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Cruz-Fisher MI, Cheng C, Sun G, Pal S, Teng A, Molina DM, Kayala MA, Vigil A, Baldi P, Felgner PL, Liang X, de la Maza LM. 2011. Identification of immunodominant antigens by probing a whole Chlamydia trachomatis open reading frame proteome microarray using sera from immunized mice. Infect. Immun. 79:246–257. 10.1128/IAI.00626-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Wang J, Zhang Y, Lu C, Lei L, Yu P, Zhong G. 2010. A genome-wide profiling of the humoral immune response to Chlamydia trachomatis infection reveals vaccine candidate antigens expressed in humans. J. Immunol. 185:1670–1680. 10.4049/jimmunol.1001240 [DOI] [PubMed] [Google Scholar]
- 164.Longbottom D. 2003. Chlamydial vaccine development. J. Med. Microbiol. 52:537–540. 10.1099/jmm.0.05093-0 [DOI] [PubMed] [Google Scholar]
- 165.Zhu S, Chen J, Zheng M, Gong W, Xue X, Li W, Zhang L. 2010. Identification of immunodominant linear B-cell epitopes within the major outer membrane protein of Chlamydia trachomatis. Acta Biochim. Biophys. Sin. (Shanghai) 42:771–778. 10.1093/abbs/gmq087 [DOI] [PubMed] [Google Scholar]
- 166.Sun G, Pal S, Weiland J, Peterson EM, de la Maza LM. 2009. Protection against an intranasal challenge by vaccines formulated with native and recombinant preparations of the Chlamydia trachomatis major outer membrane protein. Vaccine 27:5020–5025. 10.1016/j.vaccine.2009.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.O'Meara CP, Armitage CW, Harvie MC, Timms P, Lycke NY, Beagley KW. 2013. Immunization with a MOMP-based vaccine protects mice against a pulmonary Chlamydia challenge and identifies a disconnection between infection and pathology. PLoS One 8:e61962. 10.1371/journal.pone.0061962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Tifrea DF, Ralli-Jain P, Pal S, de la Maza LM. 2013. Vaccination with the recombinant major outer membrane protein elicits antibodies to the constant domains and induces cross-serovar protection against intranasal challenge with Chlamydia trachomatis. Infect. Immun. 81:1741–1750. 10.1128/IAI.00734-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Kalbina I, Wallin A, Lindh I, Engstrom P, Andersson S, Strid K. 2011. A novel chimeric MOMP antigen expressed in Escherichia coli, Arabidopsis thaliana, and Daucus carota as a potential Chlamydia trachomatis vaccine candidate. Protein Expr. Purif. 80:194–202. 10.1016/j.pep.2011.08.010 [DOI] [PubMed] [Google Scholar]
- 170.Murthy AK, Chambers JP, Meier PA, Zhong G, Arulanandam BP. 2007. Intranasal vaccination with a secreted chlamydial protein enhances resolution of genital Chlamydia muridarum infection, protects against oviduct pathology, and is highly dependent upon endogenous gamma interferon production. Infect. Immun. 75:666–676. 10.1128/IAI.01280-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Murthy AK, Li W, Guentzel MN, Zhong G, Arulanandam BP. 2011. Vaccination with the defined chlamydial secreted protein CPAF induces robust protection against female infertility following repeated genital chlamydial challenge. Vaccine 29:2519–2522. 10.1016/j.vaccine.2011.01.074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Li Z, Lu C, Peng B, Zeng H, Zhou Z, Wu Y, Zhong G. 2012. Induction of protective immunity against Chlamydia muridarum intravaginal infection with a chlamydial glycogen phosphorylase. PLoS One 7:e32997. 10.1371/journal.pone.0032997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Olsen AW, Theisen M, Christensen D, Follmann F, Andersen P. 2010. Protection against Chlamydia promoted by a subunit vaccine (CTH1) compared with a primary intranasal infection in a mouse genital challenge model. PLoS One 5:e10768. 10.1371/journal.pone.0010768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Lu C, Peng B, Li Z, Lei L, Li Z, Chen L, He Q, Zhong G, Wu Y. 2013. Induction of protective immunity against Chlamydia muridarum intravaginal infection with the chlamydial immunodominant antigen macrophage infectivity potentiator. Microbes Infect. 15:329–338. 10.1016/j.micinf.2013.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Longbottom D, Livingstone M. 2006. Vaccination against chlamydial infections of man and animals. Vet. J. 171:263–275. 10.1016/j.tvjl.2004.09.006 [DOI] [PubMed] [Google Scholar]
- 176.Kumar U, Kumar S, Varghese S, Chamoli R, Barthwal P. 2013. DNA vaccine: a modern biotechnological approach towards human welfare and clinical trials. Int. J. Res. Biomed. Biotechnol. 3:17–20 [Google Scholar]
- 177.Pal S, Barnhart KM, Wei Q, Abai AM, Peterson EM, de la Maza LM. 1999. Vaccination of mice with DNA plasmids coding for the Chlamydia trachomatis major outer membrane protein elicits an immune response but fails to protect against a genital challenge. Vaccine 17:459–465. 10.1016/S0264-410X(98)00219-9 [DOI] [PubMed] [Google Scholar]
- 178.Dong-Ji Z, Yang X, Shen C, Lu H, Murdin A, Brunham RC. 2000. Priming with Chlamydia trachomatis major outer membrane protein (MOMP) DNA followed by MOMP ISCOM boosting enhances protection and is associated with increased immunoglobulin A and Th1 cellular immune responses. Infect. Immun. 68:3074–3078. 10.1128/IAI.68.6.3074-3078.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Ou C, Tian D, Ling Y, Pan Q, He Q, Eko FO, He C. 2013. Evaluation of an ompA-based phage-mediated DNA vaccine against Chlamydia abortus in piglets. Int. Immunopharmacol. 16:505–510. 10.1016/j.intimp.2013.04.027 [DOI] [PubMed] [Google Scholar]
- 180.Xu W, Liu J, Gong W, Chen J, Zhu S, Zhang L. 2011. Protective immunity against Chlamydia trachomatis genital infection induced by a vaccine based on the major outer membrane multi-epitope human papillomavirus major capsid protein L1. Vaccine 29:2672–2678. 10.1016/j.vaccine.2010.12.132 [DOI] [PubMed] [Google Scholar]
- 181.Langemann T, Koller VJ, Muhammad A, Kudela P, Mayr UB, Lubitz W. 2010. The bacterial ghost platform system: production and applications. Bioeng. Bugs 1:326–336. 10.4161/bbug.1.5.12540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Ifere GO, He Q, Igietseme JU, Ananaba GA, Lyn D, Lubitz W, Kellar KL, Black CM, Eko FO. 2007. Immunogenicity and protection against genital Chlamydia infection and its complications by a multisubunit candidate vaccine. J. Microbiol. Immunol. Infect. 40:188–200 [PubMed] [Google Scholar]
- 183.Eko FO, Okenu DN, Singh UP, He Q, Black C, Igietseme JU. 2011. Evaluation of a broadly protective Chlamydia-cholera combination vaccine candidate. Vaccine 29:3802–3810. 10.1016/j.vaccine.2011.03.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Luzardo-Alvarez A, Blarer N, Peter K, Romero JF, Reymond C, Corradin G, Gander B. 2005. Biodegradable microspheres alone do not stimulate murine macrophages in vitro, but prolong antigen presentation by macrophages in vitro and stimulate a solid immune response in mice. J. Control Release 109:62–76. 10.1016/j.jconrel.2005.09.015 [DOI] [PubMed] [Google Scholar]
- 185.Waeckerle-Men Y, Groettrup M. 2005. PLGA microspheres for improved antigen delivery to dendritic cells as cellular vaccines. Adv. Drug Deliv. Rev. 57:475–482. 10.1016/j.addr.2004.09.007 [DOI] [PubMed] [Google Scholar]
- 186.Hamdy S, Molavi O, Ma Z, Haddadi A, Alshamsan A, Gobti Z, Elhasi S, Samuel J, Lavasanifar A. 2008. Co-delivery of cancer-associated antigen and Toll-like receptor 4 ligand in PLGA nanoparticles induces potent CD8+ T cell-mediated anti-tumor immunity. Vaccine 26:5046–5057. 10.1016/j.vaccine.2008.07.035 [DOI] [PubMed] [Google Scholar]
- 187.Waeckerle-Men Y, Gander B, Groettrup M. 2005. Delivery of tumor antigens to dendritic cells using biodegradable microspheres. Methods Mol. Med. 109:35–46 [DOI] [PubMed] [Google Scholar]
- 188.Boyoglu S, Vig K, Pillai S, Rangari V, Dennis VA, Khazi F, Singh SR. 2009. Enhanced delivery and expression of a nanoencapsulated DNA vaccine vector for respiratory syncytial virus. Nanomedicine 5:463–472. 10.1016/j.nano.2009.02.004 [DOI] [PubMed] [Google Scholar]
- 189.Fairley SJ, Singh SR, Yilma AN, Waffo AB, Subbarayan P, Dixit S, Taha MA, Cambridge CD, Dennis VA. 2013. Chlamydia trachomatis recombinant MOMP encapsulated in PLGA nanoparticles triggers primarily T helper 1 cellular and antibody immune responses in mice: a desirable candidate nanovaccine. Int. J. Nanomedicine 8:2085–2099. 10.2147/IJN.S44155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Taha MA, Singh SR, Dennis VA. 2012. Biodegradable PLGA85/15 nanoparticles as a delivery vehicle for Chlamydia trachomatis recombinant MOMP-187 peptide. Nanotechnology 23:325101. 10.1088/0957-4484/23/32/325101 [DOI] [PubMed] [Google Scholar]
- 191.Cambridge CD, Singh SR, Waffo AB, Fairley SJ, Dennis VA. 2013. Formulation, characterization, and expression of a recombinant MOMP Chlamydia trachomatis DNA vaccine encapsulated in chitosan nanoparticles. Int. J. Nanomedicine 8:1759–1771. 10.2147/IJN.S42723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Childs TS, Webley WC. 2012. In vitro assessment of halobacterial gas vesicles as a Chlamydia vaccine display and delivery system. Vaccine 30:5942–5948. 10.1016/j.vaccine.2012.07.038 [DOI] [PubMed] [Google Scholar]
- 193.Sremac M, Stuart ES. 2008. Recombinant gas vesicles from Halobacterium sp. displaying SIV peptides demonstrate biotechnology potential as a pathogen peptide delivery vehicle. BMC Biotechnol. 8:9. 10.1186/1472-6750-8-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Sremac M, Stuart ES. 2010. SIVsm Tat, Rev, and Nef1: functional characteristics of r-GV internalization on isotypes, cytokines, and intracellular degradation. BMC Biotechnol. 10:54. 10.1186/1472-6750-10-54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Mbow ML, De Gregorio E, Valiante NM, Rappuoli R. 2010. New adjuvants for human vaccines. Curr. Opin. Immunol. 22:411–416. 10.1016/j.coi.2010.04.004 [DOI] [PubMed] [Google Scholar]
- 196.O'Hagan DT. 2007. New generation vaccine adjuvants, p 1–7 In Encyclopedia of life sciences. John Wiley and Sons, Hoboken, NJ. 10.1002/9780470015902.a0020177 [DOI] [Google Scholar]
- 197.O'Hagan DT, Rappuoli R. 2004. Novel approaches to vaccine delivery. Pharm. Res. 21:1519–1530. 10.1023/B:PHAM.0000041443.17935.33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Sokolovska A, Hem SL, HogenEsch H. 2007. Activation of dendritic cells and induction of CD4(+) T cell differentiation by aluminum-containing adjuvants. Vaccine 25:4575–4585. 10.1016/j.vaccine.2007.03.045 [DOI] [PubMed] [Google Scholar]
- 199.Yu H, Karunakaran KP, Jiang X, Shen C, Andersen P, Brunham RC. 2012. Chlamydia muridarum T cell antigens and adjuvants that induce protective immunity in mice. Infect. Immun. 80:1510–1518. 10.1128/IAI.06338-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Nolz JC, Starbeck-Miller GR, Harty JT. 2011. Naive, effector and memory CD8 T-cell trafficking: parallels and distinctions. Immunotherapy 3:1223–1233. 10.2217/imt.11.100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Perry LL, Feilzer K, Portis JL, Caldwell HD. 1998. Distinct homing pathways direct T lymphocytes to the genital and intestinal mucosae in Chlamydia-infected mice. J. Immunol. 160:2905–2914 [PubMed] [Google Scholar]
- 202.Kelly KA, Chan AM, Butch A, Darville T. 2009. Two different homing pathways involving integrin beta7 and E-selectin significantly influence trafficking of CD4 cells to the genital tract following Chlamydia muridarum infection. Am. J. Reprod. Immunol. 61:438–445. 10.1111/j.1600-0897.2009.00704.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Brandtzaeg P. 2009. Mucosal immunity: induction, dissemination, and effector functions. Scand. J. Immunol. 70:505–515. 10.1111/j.1365-3083.2009.02319.x [DOI] [PubMed] [Google Scholar]
- 204.Naz RK. 2012. Female genital tract immunity: distinct immunological challenges for vaccine development. J. Reprod. Immunol. 93:1–8. 10.1016/j.jri.2011.09.005 [DOI] [PubMed] [Google Scholar]
- 205.Gallichan WS, Rosenthal KL. 1998. Long-term immunity and protection against herpes simplex virus type 2 in the murine female genital tract after mucosal but not systemic immunization. J. Infect. Dis. 177:1155–1161. 10.1086/515286 [DOI] [PubMed] [Google Scholar]
- 206.Gupta S, Janani R, Bin Q, Luciw P, Greer C, Perri S, Legg H, Donnelly J, Barnett S, O'Hagan D, Polo JM, Vajdy M. 2005. Characterization of human immunodeficiency virus Gag-specific gamma interferon-expressing cells following protective mucosal immunization with alphavirus replicon particles. J. Virol. 79:7135–7145. 10.1128/JVI.79.11.7135-7145.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Jiang JQ, Patrick A, Moss RB, Rosenthal KL. 2005. CD8+ T-cell-mediated cross-clade protection in the genital tract following intranasal immunization with inactivated human immunodeficiency virus antigen plus CpG oligodeoxynucleotides. J. Virol. 79:393–400. 10.1128/JVI.79.1.393-400.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Lindqvist M, Persson J, Thorn K, Harandi AM. 2009. The mucosal adjuvant effect of alpha-galactosylceramide for induction of protective immunity to sexually transmitted viral infection. J. Immunol. 182:6435–6443. 10.4049/jimmunol.0900136 [DOI] [PubMed] [Google Scholar]
- 209.Zhang X, Chentoufi AA, Dasgupta G, Nesburn AB, Wu M, Zhu X, Carpenter D, Wechsler SL, You S, BenMohamed L. 2009. A genital tract peptide epitope vaccine targeting TLR-2 efficiently induces local and systemic CD8+ T cells and protects against herpes simplex virus type 2 challenge. Mucosal Immunol. 2:129–143. 10.1038/mi.2008.81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Hickey DK, Aldwell FE, Beagley KW. 2010. Oral immunization with a novel lipid-based adjuvant protects against genital Chlamydia infection. Vaccine 28:1668–1672. 10.1016/j.vaccine.2009.12.010 [DOI] [PubMed] [Google Scholar]
- 211.He Q, Martinez-Sobrido L, Eko FO, Palese P, Garcia-Sastre A, Lyn D, Okenu D, Bandea C, Ananaba GA, Black CM, Igietseme JU. 2007. Live-attenuated influenza viruses as delivery vectors for Chlamydia vaccines. Immunology 122:28–37. 10.1111/j.1365-2567.2007.02608.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Pal S, Luke CJ, Barbour AG, Peterson EM, de la Maza LM. 2003. Immunization with the Chlamydia trachomatis major outer membrane protein, using the outer surface protein A of Borrelia burgdorferi as an adjuvant, can induce protection against a chlamydial genital challenge. Vaccine 21:1455–1465. 10.1016/S0264-410X(02)00680-1 [DOI] [PubMed] [Google Scholar]
- 213.Eko FO, Ekong E, He Q, Black CM, Igietseme JU. 2011. Induction of immune memory by a multisubunit chlamydial vaccine. Vaccine 29:1472–1480. 10.1016/j.vaccine.2010.12.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Ralli-Jain P, Tifrea D, Cheng C, Pal S, de la Maza LM. 2010. Enhancement of the protective efficacy of a Chlamydia trachomatis recombinant vaccine by combining systemic and mucosal routes for immunization. Vaccine 28:7659–7666. 10.1016/j.vaccine.2010.09.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Cunningham KA, Carey AJ, Hafner L, Timms P, Beagley KW. 2011. Chlamydia muridarum major outer membrane protein-specific antibodies inhibit in vitro infection but enhance pathology in vivo. Am. J. Reprod. Immunol. 65:118–126. 10.1111/j.1600-0897.2010.00894.x [DOI] [PubMed] [Google Scholar]
- 216.Goosen N, Moolenaar GF. 2008. Repair of UV damage in bacteria. DNA Repair (Amst.) 7:353–379. 10.1016/j.dnarep.2007.09.002 [DOI] [PubMed] [Google Scholar]
- 217.Ding Y, Liu R, Rong J, Xiong S. 2014. Heat-induced denaturation and aggregation of actomyosin and myosin from yellowcheek carp during setting. Food Chem. 149:237–243. 10.1016/j.foodchem.2013.10.123 [DOI] [PubMed] [Google Scholar]
- 218.Rank RG, Batteiger BE, Soderberg LS. 1990. Immunization against chlamydial genital infection in guinea pigs with UV-inactivated and viable chlamydiae administered by different routes. Infect. Immun. 58:2599–2605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Brunham RC, Pourbohloul B, Mak S, White R, Rekart ML. 2005. The unexpected impact of a Chlamydia trachomatis infection control program on susceptibility to reinfection. J. Infect. Dis. 192:1836–1844. 10.1086/497341 [DOI] [PubMed] [Google Scholar]
- 220.Atik B, Thanh TT, Luong VQ, Lagree S, Dean D. 2006. Impact of annual targeted treatment on infectious trachoma and susceptibility to reinfection. JAMA 296:1488–1497. 10.1001/jama.296.12.1488 [DOI] [PubMed] [Google Scholar]
- 221.Chidambaram JD, Alemayehu W, Melese M, Lakew T, Yi E, House J, Cevallos V, Zhou Z, Maxey K, Lee DC, Shapiro BL, Srinivasan M, Porco T, Whitcher JP, Gaynor BD, Lietman TM. 2006. Effect of a single mass antibiotic distribution on the prevalence of infectious trachoma. JAMA 295:1142–1146. 10.1001/jama.295.10.1142 [DOI] [PubMed] [Google Scholar]
- 222.Lal JA, Malogajski J, Verweij SP, de Boer P, Ambrosino E, Brand A, Ouburg S, Morre SA. 2013. Chlamydia trachomatis infections and subfertility: opportunities to translate host pathogen genomic data into public health. Public Health Genomics 16:50–61. 10.1159/000346207 [DOI] [PubMed] [Google Scholar]
- 223.Morré SA, Karimi O, Ouburg S. 2009. Chlamydia trachomatis: identification of susceptibility markers for ocular and sexually transmitted infection by immunogenetics. FEMS Immunol. Med. Microbiol. 55:140–153. 10.1111/j.1574-695X.2009.00536.x [DOI] [PubMed] [Google Scholar]
- 224.Bogaards JA, Kretzschmar M, Xiridou M, Meijer CJ, Berkhof J, Wallinga J. 2011. Sex-specific immunization for sexually transmitted infections such as human papillomavirus: insights from mathematical models. PLoS Med. 8:e1001147. 10.1371/journal.pmed.1001147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Jit M, Choi YH, Edmunds WJ. 2008. Economic evaluation of human papillomavirus vaccination in the United Kingdom. BMJ 337:a769. 10.1136/bmj.a769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Brisson M, van de Velde N, Franco EL, Drolet M, Boily MC. 2011. Incremental impact of adding boys to current human papillomavirus vaccination programs: role of herd immunity. J. Infect. Dis. 204:372–376. 10.1093/infdis/jir285 [DOI] [PubMed] [Google Scholar]
- 227.FDA. Accessed 19 October 2009 Gardasil approval letter. FDA, Rockville, MD: http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm186991.htm [Google Scholar]
- 228.Quinn TC, Gaydos C, Shepherd M, Bobo L, Hook EW, 3rd, Viscidi R, Rompalo A. 1996. Epidemiologic and microbiologic correlates of Chlamydia trachomatis infection in sexual partnerships. JAMA 276:1737–1742 [PubMed] [Google Scholar]
- 229.Gray RT, Beagley KW, Timms P, Wilson DP. 2009. Modeling the impact of potential vaccines on epidemics of sexually transmitted Chlamydia trachomatis infection. J. Infect. Dis. 199:1680–1688. 10.1086/598983 [DOI] [PubMed] [Google Scholar]
- 230.Dang T, Jaton-Ogay K, Flepp M, Kovari H, Evison JM, Fehr J, Schmid P, Boffi El Amari E, Cavassini M, Odorico M, Tarr PE, Greub G. 2009. High prevalence of anorectal chlamydial infection in HIV-infected men who have sex with men in Switzerland. Clin. Infect. Dis. 49:1532–1535. 10.1086/644740 [DOI] [PubMed] [Google Scholar]
- 231.Malhotra M, Sood S, Mukherjee A, Muralidhar S, Bala M. 2013. Genital Chlamydia trachomatis: an update. Indian J. Med. Res. 138:303–316 [PMC free article] [PubMed] [Google Scholar]
- 232.Lacy HM, Bowlin AK, Hennings L, Scurlock AM, Nagarajan UM, Rank RG. 2011. Essential role for neutrophils in pathogenesis and adaptive immunity in Chlamydia caviae ocular infections. Infect. Immun. 79:1889–1897. 10.1128/IAI.01257-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Gall A, Horowitz A, Joof H, Natividad A, Tetteh K, Riley E, Bailey RL, Mabey DC, Holland MJ. 2011. Systemic effector and regulatory immune responses to chlamydial antigens in trachomatous trichiasis. Front. Microbiol. 2:10. 10.3389/fmicb.2011.00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Picard MD, Cohane KP, Gierahn TM, Higgins DE, Flechtner JB. 2012. High-throughput proteomic screening identifies Chlamydia trachomatis antigens that are capable of eliciting T cell and antibody responses that provide protection against vaginal challenge. Vaccine 30:4387–4393. 10.1016/j.vaccine.2012.01.017 [DOI] [PubMed] [Google Scholar]
- 235.Cheng C, Cruz-Fisher MI, Tifrea D, Pal S, Wizel B, de la Maza LM. 2011. Induction of protection in mice against a respiratory challenge by a vaccine formulated with the Chlamydia major outer membrane protein adjuvanted with IC31(R). Vaccine 29:2437–2443. 10.1016/j.vaccine.2011.01.031 [DOI] [PubMed] [Google Scholar]
- 236.Meoni E, Faenzi E, Frigimelica E, Zedda L, Skibinski D, Giovinazzi S, Bonci A, Petracca R, Bartolini E, Galli G, Agnusdei M, Nardelli F, Buricchi F, Norais N, Ferlenghi I, Donati M, Cevenini R, Finco O, Grandi G, Grifantini R. 2009. CT043, a protective antigen that induces a CD4+ Th1 response during Chlamydia trachomatis infection in mice and humans. Infect. Immun. 77:4168–4176. 10.1128/IAI.00344-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Clifton DR, Fields KA, Grieshaber SS, Dooley CA, Fischer ER, Mead DJ, Carabeo RA, Hackstadt T. 2004. A chlamydial type III translocated protein is tyrosine-phosphorylated at the site of entry and associated with recruitment of actin. Proc. Natl. Acad. Sci. U. S. A. 101:10166–10171. 10.1073/pnas.0402829101 [DOI] [PMC free article] [PubMed] [Google Scholar]