Abstract
Qualitative and quantitative 16S rRNA gene-based real-time PCR and direct sequencing were applied for rapid detection and identification of bacterial DNA (bactDNA) in 356 ascites samples. bactDNA was detected in 35% of samples, with a mean of 3.24 log copies ml−1. Direct sequencing of PCR products revealed 62% mixed chromatograms predominantly belonging to Gram-positive bacteria. Terminal restriction fragment length polymorphism (T-RFLP) results of a sample subset confirmed sequence data showing polymicrobial DNA contents in 67% of bactDNA-positive ascites samples.
TEXT
Spontaneous bacterial peritonitis (SBP) is the most severe bacterial infection in patients with decompensated liver disease (1). As evidenced by culture-based diagnosis, Gram-negative bacteria of the genera Escherichia and Klebsiella are the most frequent cause for SBP (2, 3). However, recent data reveal that also Gram-positive bacteria such as Staphylococcus and Enterococcus species may be responsible for bacterial peritonitis, especially in hospitalized patients (4, 5). Since detection rates of classical culture techniques are low in the routine setting, the causative agent remains unknown in many cases of SBP (3).
Application of culture-independent PCR-based methods for the detection of bacterial DNA (bactDNA) in ascites has been proposed as a suitable tool to improve pathogen identification in patients at risk for SBP (6) and has indicated that bactDNA derives in most cases from a single pathogen (7–9). However, recent studies using 16S rRNA-based fingerprinting analyses have shown that ascites may also be polymicrobial (10, 11) and that the bacterial spectrum is broader than that previously reported in context with SBP (11).
Based on these findings, we analyzed a set of ascites samples using primers targeting the 16S rRNA gene for qualitative and quantitative PCR and further characterized bactDNA-positive samples by direct sequencing and terminal restriction fragment length polymorphism (T-RFLP) analysis in order to determine the frequency of detectable bactDNA in ascites from patients with end-stage liver disease and to identify the corresponding bacterial agents.
A total of 356 ascites samples from 174 patients with liver cirrhosis (77.6% alcoholic, 11.5% cryptogenic, 2.9% viral hepatitis, 1.7% genetic disorders or metabolic diseases, 1.7% autoimmune hepatitis, 0.6% primary biliary cirrhosis, 3.5% nonalcoholic fatty liver disease, and 0.6% cardiac) were obtained at the University Hospital Leipzig, between February 2011 and December 2012. Thirty-seven percent of the patients (n = 61) underwent several diagnostic paracenteses (mean number of paracenteses, 3.9; range, 2 to 11). All patients gave full written informed consent to the study protocol, which was approved by the local ethics committee.
Ascitic fluid samples were routinely analyzed for total leukocyte count using an automated blood cell counter and for the presence of bacterial and fungal pathogens by routine microbiological culture with direct inoculation of agar plates.
For culture-independent PCR analysis, approximately 50 ml ascites was centrifuged, and the pellet was resuspended in 3 ml supernatant with glycerol to a final concentration of 20% and stored at −20°C until sample preparation.
Nucleic acid was isolated from 1 ml resuspended ascites cell pellet with the MolYsis Complete5 DNA isolation kit (Molzym, Bremen, Germany). A negative control with bactDNA-negative ascites supernatant was included in each isolation series, and ascites supernatant spiked with Escherichia coli at a concentration of 100 cells ml−1 served as positive control.
bactDNA was amplified using reagents from the Mastermix 16S Complete kit (Molzym, Bremen, Germany), containing broad-range bacterial primers covering the variable 16S rRNA gene regions V3 and V4. A PCR no-template control and a PCR-positive control containing Escherichia coli DNA were included in each PCR run. With qualitative real-time PCR detection based on Sybr green and subsequent melting-curve analysis, we distinguished bactDNA-positive from bactDNA-negative samples. All samples with appropriate melting-curve temperature and amplicon size after gel electrophoresis were considered positive and subsequently quantified by real-time PCR using a 6-carboxyfluorescein (FAM)-labeled universal hydrolysis probe as previously described by Nadkarni et al. (12). Serial dilutions of a plasmid containing the 16S rRNA gene fragment from Escherichia coli DH5α served as the quantification standard.
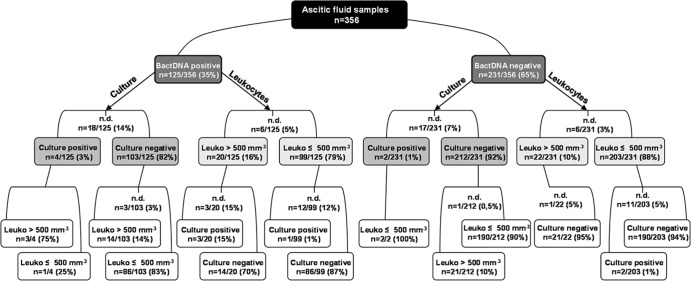
bactDNA was detected in 35% (n = 125) of the ascitic fluid samples with a mean of 3.24 log copies ml−1. Results from routine culture analysis and total leukocyte values were available for 321 (90%) and 344 (97%) ascites samples, respectively, and could be correlated to qualitative (Fig. 1) and quantitative (Table 1) PCR results. Only a small proportion of bactDNA-positive samples showed positive culture results (3%) with detection of Pseudomonas, Staphylococcus, Enterococcus, and Citrobacter species, but mean bactDNA quantities were higher in culture-positive than in culture-negative samples (Table 1). Interestingly, the majority of bactDNA-positive samples (79%) showed a total leukocyte count in ascites below or equal to 500 cells mm−3, a fact that has already been described in context with culture-negative, bactDNA-positive ascites in outpatients and hospitalized patients (6, 13, 14). However, bactDNA detection rates in nonneutrocytic ascites were considerably lower in these studies, ranging from 10% to 35% (4, 6), which might be due to the larger sample volume used in this study in combination with a DNA isolation kit that enhances the bactDNA yield from intact bacterial cells by degrading human DNA prior to nucleic acid purification. Samples with low bactDNA contents could be detected by running a higher number of PCR cycles for the detection of low DNA copy numbers, resulting in a real-time PCR assay with increased sensitivity (detection limit, 2.72 log copies ml−1).
FIG 1.
16S rRNA gene-based PCR results in ascites divided into bactDNA-positive and bactDNA-negative samples and separately analyzed for corresponding culture results and total leukocyte values. Leuko, total leukocyte value; n.d., not determined.
TABLE 1.
Quantitative determination of 16S rRNA gene copies in ascitic fluid samples, grouped according to corresponding culture results and total leukocyte values (n = 104)
| Sample groupa | Mean (range) qPCR result (log copies ml−1) |
|---|---|
| bactDNA+/culture+/leukocyte+ (n = 3) | 5.05 (4.09–6.23) |
| bactDNA+/culture+/leukocyte− (n = 1) | 9.25 |
| bactDNA+/culture−/leukocyte+ (n = 14) | 3.82 (2.72–6.17) |
| bactDNA+/culture−/leukocyte− (n = 86) | 3.05 (2.72–8.94) |
Comparison of mean real-time quantitative PCR (qPCR) results between sample groups and statistical analysis (Mann-Whitney U test): culture+ (n = 4), 6.1 log copies ml−1, versus culture− (n = 100), 3.15 log copies ml−1 (P = 0.001); leukocyte+ (n = 17), 4.04 log copies ml−1, versus leukocyte− (n = 87), 3.12 log copies ml−1 (P = 0.004). Leukocyte+, >500 cells mm−3; leukocyte−, ≤500 cells mm−3.
PCR products from all bactDNA-positive samples were purified and directly sequenced with the sequencing primers SeqGN16 and SeqGP16 (Molzym, Bremen, Germany). Sequence types from nonmixed chromatograms were directly identified up to genus level with sequence identity higher than 98% using the BLAST tool (24) and the RDP Classifier, respectively (16). Mixed chromatograms were present in 62% of the samples (n = 68) and could be analyzed using the web-based software application RipSeq (iSentio, Bergen, Norway) in order to differentiate overlying sequences and to identify the most abundant sequence types in polymicrobial samples (17).
Sequencing results from 109 of 125 bactDNA-positive samples revealed a variety of sequence types belonging to Gram-positive and Gram-negative bacteria comprising 57 different genera found overall in ascitic fluid samples. Figure 2 shows the distribution of the 12 most frequently identified bacterial sequence types in patients with a positive bactDNA result in at least one of their consecutive samples. bactDNA from the Gram-positive genera Staphylococcus, Streptococcus, and Lactobacillus was predominantly detected, whereas Gram-negative genera were represented by Cloacibacterium spp. and Agrobacterium spp. Genera that occurred only once per patient (n = 26), e.g., Salmonella; twice per patient (n = 11), e.g., Escherichia; or three times per patient (n = 7), e.g., Pseudomonas, are summarized under the term “other genera.”
FIG 2.
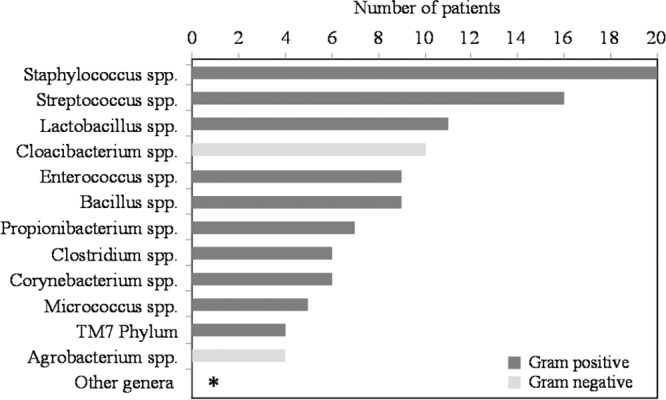
Overview of bactDNA in ascitic fluid samples from cirrhotic patients (n = 174). Bacterial genus results were derived from sequence analysis using direct sequencing of the 16S rRNA gene PCR fragment. The number of patients with detection of the respective sequence type is given on the x axis. Recurring sequence types analyzed in ascitic fluid samples from consecutive paracenteses of the same patient were counted only once. Genera that occurred in three or fewer patients are combined under the term “other genera” and indicated with an asterisk.
bactDNA from Lactobacillus spp. and Propionibacterium spp., which have not been proposed as common SBP-causative organisms so far, could be detected in ascites of 11 and 7 patients, respectively. Lactobacillus spp. and Propionibacterium spp., belonging to the key phyla Firmicutes and Actinobacteria of the natural human gut microbiome (15), respectively, may translocate from the gut into the peritoneal fluid as already described for enteric bacteria in context with SBP (1), and DNA from Propionibacterium spp. has recently been found in a high number of ascitic fluid samples of cirrhotic patients (18).
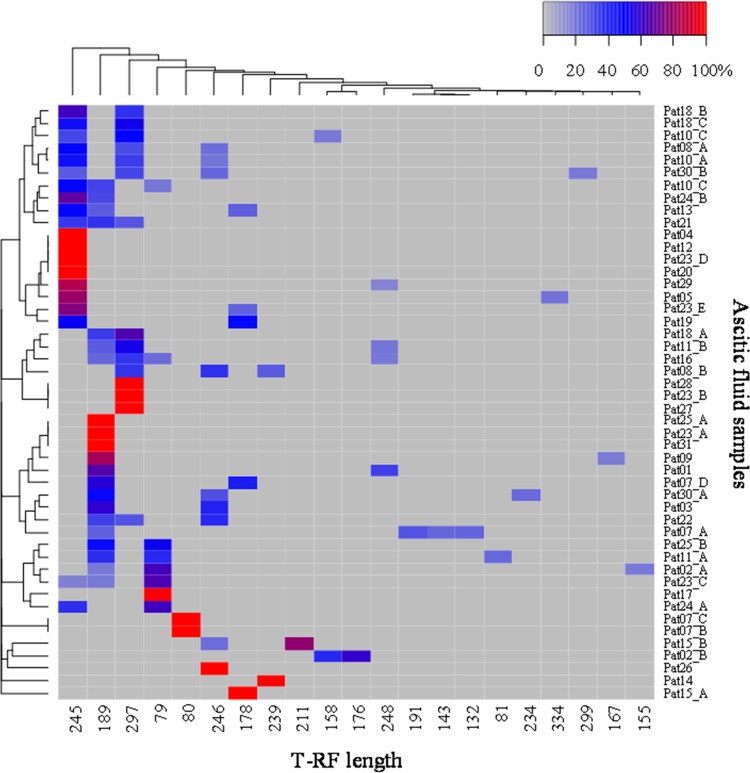
The high proportion of polymicrobial samples and the diversity of bacterial sequence types in ascitic fluid found by direct sequencing could be confirmed with T-RFLP analysis. Bacterial 16S rRNA gene fragments were amplified with the forward primer 27f (19) and the FAM-labeled reverse primer (12), purified, and subsequently digested with the restriction endonuclease MspI. Dried DNA samples were resuspended in HiDi formamide containing 1.5% GeneScan-500 Rox standard (Applied Biosystems, Germany) for a length determination of labeled terminal restriction fragments (T-RF) within the range of 50 to 500 bp. Noise removal, normalization of data, and statistical analyses were performed according to the method in reference 20. For two-way cluster analysis, the Bray-Curtis distance was used and heat maps were constructed using the R package gplots (21, 22).
Altogether, 21 different T-RF could be found in the DNA fragments derived from a subset of 48 ascites samples from 29 patients (Fig. 3). Only one T-RF was detected in 33% of analyzed samples (n = 16), whereas 67% revealed two to four different T-RF per sample, confirming the high proportion of mixed samples found by direct sequencing. Additional T-RFLP analyses with the restriction endonucleases AluI and CfoI showed 66% and 71% mixed DNA samples, respectively, and confirmed the results obtained with MspI (data not shown).
FIG 3.
Two-way clustering of T-RFLP profiles of bacterial DNA from ascitic fluid samples generated by restriction digest using the restriction endonuclease MspI (deviation = 2) displayed as a heat map. Ascitic fluid samples (n = 48) from 29 patients are represented by rows, whereas T-RF are represented by columns. Each square in the heat map represents the relative abundance of a single T-RF per sample measured by fluorescence intensity and indicated by color. Rows are clustered according to the similarity of the T-RFLP profile from each sample, and columns are clustered by cooccurring T-RF within the sample based on the Bray-Curtis index. The color key for the relative abundance of T-RF is shown in the upper right corner. T-RF length is in base pairs.
The T-RF types with lengths of 245 bp, 189 bp, 297 bp, and 79 bp were dominantly found throughout all samples. Only two of the 11 patients with repeated paracenteses showed a clear clustering of the T-RF in different samples, whereas the remaining patients revealed a shift of the T-RF pattern during follow-up.
In this study, we could show that application of 16S rRNA gene-based real-time PCR with subsequent direct sequencing allows achievement of fast results regarding the bactDNA content and differentiation of several dominant sequence types up to genus level even within polymicrobial ascites samples. Considering the bacterial spectrum found in our study, these data will add to the continuing controversy about the actual impact of bactDNA detection in context with the development of SBP and clinical outcome. Since recent studies have shown that the presence of bactDNA in ascites is associated with a poor prognosis (23) and a higher risk of SBP (14), further prospective studies are needed for a broader evaluation of the predictive value of positive bactDNA results, including samples with low bactDNA levels and multiple 16S rRNA gene sequences.
Footnotes
Published ahead of print 12 March 2014
REFERENCES
- 1.Wongcharatrawee S, Garcia-Tsao G. 2001. Clinical management of ascites and its complications. Clin. Liver Dis. 5:833–850. 10.1016/S1089-3261(05)70194-X [DOI] [PubMed] [Google Scholar]
- 2.Căruntu FA, Benea L. 2006. Spontaneous bacterial peritonitis: pathogenesis, diagnosis, treatment. J. Gastrointestin. Liver Dis. 15:51–56 [PubMed] [Google Scholar]
- 3.Wiest R, Krag A, Gerbes A. 2012. Spontaneous bacterial peritonitis: recent guidelines and beyond. Gut 61:297–310. 10.1136/gutjnl-2011-300779 [DOI] [PubMed] [Google Scholar]
- 4.El Sayed Zaki M, El Shabrawy WO, El-Eshmawy MM, Aly Eletreby S. 2011. The high prevalence of Listeria monocytogenes peritonitis in cirrhotic patients of an Egyptian medical center. J. Infect. Public Health 4:211–216. 10.1016/j.jiph.2011.06.002 [DOI] [PubMed] [Google Scholar]
- 5.Garcia-Tsao G, Wiest R. 2004. Gut microflora in the pathogenesis of the complications of cirrhosis. Best Pract. Res. Clin. Gastroenterol. 18:353–372. 10.1016/j.bpg.2003.10.005 [DOI] [PubMed] [Google Scholar]
- 6.Bruns T, Sachse S, Straube E, Assefa S, Herrmann A, Hagel S, Lehmann M, Stallmach A. 2009. Identification of bacterial DNA in neutrocytic and non-neutrocytic cirrhotic ascites by means of a multiplex polymerase chain reaction. Liver Int. 29:1206–1214. 10.1111/j.1478-3231.2009.02073.x [DOI] [PubMed] [Google Scholar]
- 7.Soriano G, Esparcia Ó, Montemayor M, Guarner-Argente C, Pericas R, Torras X, Calvo N, Román E, Navarro F, Guarner C, Coll P. 2011. Bacterial DNA in the diagnosis of spontaneous bacterial peritonitis. Aliment. Pharmacol. Ther. 33:275–284. 10.1111/j.1365-2036.2010.04506.x [DOI] [PubMed] [Google Scholar]
- 8.Such J, Francés R, Muños C, Zapater P, Casellas J, Cifuentes A, Rodríguez-Valera F, Pascual S, Sola-Vera J, Carnicer F, Uceda F, Palazón JM, Pérez-Mateo M. 2002. Detection and identification of bacterial DNA in patients with cirrhosis and culture-negative nonneutrocytic ascites. Hepatology 36:135–141. 10.1053/jhep.2002.33715 [DOI] [PubMed] [Google Scholar]
- 9.Sugihara T, Koda M, Maeda Y, Matono T, Nagahara T, Mandai M, Ueki M, Murawaki Y. 2009. Rapid identification of bacterial species with bacterial DNA microarray in cirrhotic patients with spontaneous bacterial peritonitis. Intern. Med. 48:3–10. 10.2169/internalmedicine.48.1539 [DOI] [PubMed] [Google Scholar]
- 10.Hardick J, Won H, Jeng K, Hsieh Y, Gaydos CA, Rothman RE, Yang S. 2012. Identification of bacterial pathogens in ascitic fluids from patients with suspected spontaneous bacterial peritonitis by use of broad-range PCR (16S PCR) coupled with high-resolution melt analysis. J. Clin. Microbiol. 50:2428–2432. 10.1128/JCM.00345-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rogers GB, Russell LE, Preston PG, Marsh P, Collins JE, Saunders J, Sutton J, Fine D, Bruce KD, Wright M. 2010. Characterisation of bacteria in ascites—reporting the potential of culture-independent, molecular analysis. Eur. J. Clin. Microbiol. Infect. Dis. 29:533–541. 10.1007/s10096-010-0891-5 [DOI] [PubMed] [Google Scholar]
- 12.Nadkarni MA, Martin FE, Jacques NA, Hunter N. 2002. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology 148:257–266 [DOI] [PubMed] [Google Scholar]
- 13.Appenrodt B, Lehmann LE, Thyssen L, Gentemann M, Rabe C, Molitor E, Trebicka J, Stüber F, Sauerbruch T. 2010. Is detection of bacterial DNA in ascitic fluid of clinical relevance? Eur. J. Gastroenterol. Hepatol. 22:1487–1494. 10.1097/MEG.0b013e328340c43a [DOI] [PubMed] [Google Scholar]
- 14.El-Naggar MM, Khalil el-SA, El-Daker MA, Salama MF. 2008. Bacterial DNA and its consequences in patients with cirrhosis and culture-negative, non-neutrocytic ascites. J. Med. Microbiol. 57:1533–1538. 10.1099/jmm.0.2008/001867-0 [DOI] [PubMed] [Google Scholar]
- 15.Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, Fernandes GR, Tap J, Bruls T, Batto JM, Bertalan M, Borruel N, Casellas F, Fernandez L, Gautier L, Hansen T, Hattori M, Hayashi T, Kleerebezem M, Kurokawa K, Leclerc M, Levenez F, Manichanh C, Nielsen HB, Nielsen T, Pons N, Poulain J, Qin J, Sicheritz-Ponten T, Tims S, Torrents D, Ugarte E, Zoetendal EG, Wang J, Guarner F, Pedersen O, de Vos WM, Brunak S, Doré J, MetaHIT Consortium. Antolín M, Artiguenave F, Blottiere HM, Almeida M, Brechot C, Cara C, Chervaux C, Cultrone A, Delorme C, Denariaz G, Dervyn R, Foerstner KU, Friss C, van de Guchte M, Guedon E, Haimet F, Huber W, van Hylckama-Vlieg J, Jamet A, Juste C, Kaci G, Knol J, Lakhdari O, Layec S, Le Roux K, Maguin E, Mérieux A, Melo Minardi R, M'rini C, Muller J, Oozeer R, Parkhill J, Renault P, Rescigno M, Sanchez N, Sunagawa S, Torrejon A, Turner K, Vandemeulebrouck G, Varela E, Winogradsky Y, Zeller G, Weissenbach J, Ehrlich SD, Bork P. 2011. Enterotypes of the human gut microbiome. Nature 473:174–180. 10.1038/nature09944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang QG, Garrity M, Tiedje JM, Cole JR. 2007. Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73:5261–5267. 10.1128/AEM.00062-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kommedal O, Karlsen B, Saebo O. 2008. Analysis of mixed sequencing chromatograms and its application in direct 16S rRNA gene sequencing of polymicrobial samples. J. Clin. Microbiol. 46:3766–3771. 10.1128/JCM.00213-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rogers GB, van der Gast CJ, Bruce KD, Marsh P, Collins JE, Sutton J, Wright M. 2013. Ascitic microbiota composition is correlated with clinical severity in cirrhosis with portal hypertension. PLoS One 8:e74884. 10.1371/journal.pone.0074884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lane DJ. 1991. 16S/23S rRNA sequencing, p 115–175 In Stackebrandt E, Goodfellow M. (ed), Nucleic acid techniques in bacterial systematics. John Wiley & Sons, Chichester, United Kingdom [Google Scholar]
- 20.Sträuber H, Schröder M, Kleinsteuber S. 2012. Metabolic and microbial community dynamics during the hydrolytic and acidogenic fermentation in a leach-bed process. Energy Sustain. Soc. 2:13. 10.1186/2192-0567-2-13 [DOI] [Google Scholar]
- 21.R Development Core Team. 2011. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria: http://www.R-project.org/ [Google Scholar]
- 22.Warnes GR, Bolker B, Bonebakker L, Gentleman R, Huber W, Liaw A, Lumley T, Maechler M, Magnusson A, Moeller S, Schwartz M, Venables B. 2011. gplots: various R programming tools for plotting data. R package version 2.10.1. http://CRAN.R-project.org/package=gplots [Google Scholar]
- 23.Zapater P, Francés R, Gonzáles-Navajas JM, de la Hoz MA, Moreu R, Pascual S, Monfort D, Montoliu S, Vila C, Escudero A, Torras X, Cirera I, Llanos L, Guarner-Argente C, Palazón JM, Carnicer F, Bellot P, Guarner C, Planas R, Solá R, Serra MA, Muñoz C, Pérez-Mateo M, Such J. 2008. Serum and ascitic fluid bacterial DNA: a new independent prognostic factor in noninfected patients with cirrhosis. Hepatology 48:1924–1931. 10.1002/hep.22564 [DOI] [PubMed] [Google Scholar]
- 24.Altschul SF, Gish W, Miller W, Myers EW, Lipman D. 1990. Basic local alignment search tool. J. Mol. Biol. 215:403–410 [DOI] [PubMed] [Google Scholar]