Abstract
Adequate sedation and analgesia are considered essential requirements to relieve patient discomfort and pain and ultimately to improve the outcomes of modern gastrointestinal endoscopic procedures. The willingness of patients to undergo sedation during endoscopy has increased steadily in recent years and standard sedation practices are needed for both patient safety and successful procedural outcomes. Therefore, regular training and education of healthcare providers is warranted. However, training curricula and guidelines for endoscopic sedation may have conflicts according to varying legal frameworks and/or social security systems of each country, and well-recognized endoscopic sedation training systems are not currently available in all endoscopy units. Although European and American curricula for endoscopic sedation have been extensively developed, general curricula and guidelines for each country and institution are also needed. In this review, an overview of recent curricula and guidelines for training and basic performance of endoscopic sedation is presented based on the current literature.
Keywords: Endoscopic sedation, Guideline, Education
INTRODUCTION
The rate of endoscopic sedation is increasing, as is the incidence of various sedative-related adverse events. Specialized guidelines and/or curricula for endoscopic sedation are necessary to facilitate safe and successful endoscopic procedures. However, the availability of acceptable guidelines or well-described curricula for endoscopic sedation is limited, and their use differs among countries and institutions. Our domestic circumstance also needs a systematic educational curriculum and guidelines for endoscopic sedation.
Endoscopic sedation curricula and guidelines vary among countries depending on the healthcare system and legal framework. However, standardized sedation techniques and education are required to teach healthcare providers safe and effective diagnostic and therapeutic endoscopy. Quality management requires appropriate pharmacological training for all clinical staff involved in the practice of sedation, regardless of the type of sedation used. Individual qualifications, human resources, and technical requirements are addressed by various guidelines, but may differ according to healthcare circumstances. 1,2,3 Additionally, personnel qualifications for administration of propofol may raise a controversy between anesthesiologists and nonanesthesiologists, in particular endoscopists. Nonanesthesiologist-administered propofol (NAAP) is the direct administration of propofol by trained nurses or endoscopists. NAAP is used regularly in many countries, including Korea. A number of studies of sedation methods and quality have been performed recently,4,5,6,7,8,9,10,11,12 but domestic Korean guidelines and training curricula for endoscopic sedation using propofol have not been established yet.
The European Society of Gastrointestinal Endoscopy (ESGE), the European Society of Anaesthesiology (ESA), and the European Society of Gastroenterology and Endoscopy Nurses and Associates (ESGENA) have formulated an NAAP sedation curriculum and guidelines. However, the ESA withdrew from the final NAAP guideline agreement. The curriculum and guidelines are based on a consensus of physicians from various disciplines including gastroenterology and anesthesiology along with nurses who were involved in the development of the European and National Sedation Guidelines and Curriculum for Endoscopic Sedation and the Organization of National and Local Courses for Endoscopic Sedation. 2,3 In the United States, the Multisocieties Sedation Curriculum for Gastrointestinal Endoscopy (MSCGE) has also presented an endoscopy sedation curriculum and guidelines. The MSCGE grew out of the need for a complete and programmatic approach to sedation training. It provides the knowledge and skills required for the practice of procedural sedation for gastrointestinal endoscopy.1 However, it included institutes for gastroenterologists only, without direct involvement of anesthesiologists.
The aim of this literature review is to describe the established endoscopic sedation guidelines and curriculum, focusing on training and actual performance based on the current European and American guidelines. This review also focuses on propofol sedation and NAAP-associated training and performance.
CURRENT CURRICULA AND GUIDELINES
European curriculum and guidelines
The 2013 European curriculum focuses on training for all types of sedation used in gastrointestinal endoscopy.2 The course structure includes two categories. First, a 3-day introductory course combines theory and practice with a focus on practical training. At least half of the time is spent in practical training sessions in small groups (4 to 8 people), and each unit is followed by a formal test of cognitive or skills competence. Most European countries also recommend that trainees receive periodic basic and/or advanced life support training (i.e., basic life support [BLS] or advanced cardiac life support [ACLS]), according to the national law for sedation in gastrointestinal endoscopy. Next, clinical training will consist of a learning phase of at least 2 weeks with a mentor and will include individual competency assessments. A summative assessment is performed independently by at least three independent supervisors after a minimum of 30 student-documented cases, including diagnostic and therapeutic procedures, or more cases if trustworthy professional performance has not been achieved.
The course based on the European guideline integrates theory with practice and covers the following areas: 1) relevant anatomy and physiology of the heart and respiratory tract, including definitions of hypoxemia, hypocapnia, and hypercapnia, and their relationship to the risk profile of the individual patient; 2) basic pharmacology, pharmacokinetics, indications, and contraindications of drugs commonly used for endoscopy sedation and pain control; 3) different sedation methods, including possibilities and limitations of each, possible side effects, prevention and management of complications; 4) selection of patients appropriate for administration of sedation by nonanesthesiologists, including the use of healthcare questionnaires to assist patient selection; 5) equipment and staff requirements necessary to ensure patient safety before, during, and after endoscopic interventions; 6) the use of different scores to assess patient risk status, such as the American Society of Anesthesiologists (ASA) classification4 and the risk score to predict difficult mask ventilation, along with the relationship of patient risk status, planned sedation technique, and anticipated difficulty of the endoscopic procedure; 7) patient preparation and surveillance, including safe positioning, intravenous access, monitoring, and oxygen administration; 8) sedation stages; 9) management of sedation complications; 10) documentation during sedation, e.g., assessment of oxygen saturation, heart rate, and blood pressure at regular intervals, drugs used (name and dosage), administration of intravenous fluids (type and quantity), and oxygen flow rate, sedation-associated complications and their management, and fulfillment of discharge criteria; 11) discharge criteria and patient instructions following gastrointestinal endoscopy under sedation; 12) national laws and guidelines and European and institutional guidelines and standards; 13) variations in sedation methods, including dosing and drug effects in different types of patients; 14) management of hypoxemic events, apnea, hypertension/hypotension, bradycardia/tachycardia, cardiac arrhythmias and communication in stressful situations (e.g., bleeding plus hypoxia); 15) BLS/ACLS; 16) debriefing in small groups after cases of sedation and endoscopy, including complications and their management.2 After successful completion of all assessments, the trainees receive a certificate from the ESGENA-ESGE.
Multisocieties sedation curriculum for gastrointestinal endoscopy
In the United States, the MSCGE is a curriculum for training in endoscopic sedation. The MSCGE includes recommendations from national gastroenterology societies including American Association for the Study of Liver Diseases, the American College of Gastroenterology, the American Gastroenterological Association Institute, the American Society for Gastrointestinal Endoscopy, and the Society for Gastroenterology Nurses and Associates. Other professional non-gastrointestinal societies including the ASA as well ascertain regulatory organizations were also invited to take part in the development of the MSCGE.1
The MSCGE includes the following 14 topics: 1) introduction; 2) sedation pharmacology; 3) informed consent for endoscopic sedation; 4) periprocedural assessment for endoscopic procedures; 5) levels of sedation; 6) training in the administration of specific agents for moderate sedation; 7) training in airway/rescue techniques and management of complications; 8) anesthesiologist assistance for endoscopic procedures; 9) intraprocedural monitoring; 10) postprocedural assessment training; 11) endoscopy in pregnant and lactating females; 12) assessment of competency in endoscopic sedation; 13) bibliography; 14) appendix; and a primer in sedation pharmacology.1 Each topic stresses the importance of training goals, the training process, and a sequential assessment of subject competence.
As with the European curriculum, all training courses are included. Basic and advanced life support training is required for physicians or nurses. Characteristic of the MSCGE are the emphasis on informed consent, inclusion of sedation guidelines for pregnancy, and guidelines for anesthesiologist assistance for cases of risky sedation, usually based on propofol sedation, where in contrast to the European guidelines, NAAP is not routinely permitted for deep sedation by nonanesthesiologists. Moreover, in the European curriculum, anesthesiologists are included as teaching staff (Table 1).1,2,3,13
Table 1.

MSCGE, Multisocieties Sedation Curriculum for Gastrointestinal Endoscopy; ESGE, European Society of Gastrointestinal Endoscopy; ESGENA, European Society of Gastroenterology and Endoscopy Nurses and Associates; ACLS, advanced trauma life support; BLS, basic life support; NAAP, nonanesthesiologist-administered propofol.
TRAINING FOR ENDOSCOPIC SEDATION
Target training group
Published curricula and guidelines are intended for nonanesthesiologists, i.e., physicians working in gastrointestinal endoscopy, nurses, and those who are involved in sedation. 1,2,3,13,14,15 Each country has different local rules or circumstances and healthcare systems. Thus, the target group may differ according to the drugs used, particularly if propofol is used. However, as a rule, physicians and nurses are the main target group in terms of training for performance of endoscopic sedation.
Nonanesthesiologist-administered propofol training guidelines
Currently published guidelines or curricula emphasize the use of propofol by NAAP. All endoscopists and endoscopy nurses who perform NAAP should receive appropriate education and practical training. Self-training in NAAP is strongly discouraged in the guidelines.3 Specific knowledge and skills for endoscopy and sedation are necessary to ensure patient comfort and safety and successful endoscopic outcomes. Previous experience in intensive care medicine is also desirable for physicians responsible for NAAP. European and the American Society of Gastrointestinal Endoscopy guidelines recommend that training courses include theoretical and practical parts, with each being followed by an examination and certification to document successful training.3,13 Training in basic pharmacology, pre-, during-, and post-endoscopic procedure patient care, such as monitoring, and management of complications is needed.
It is important that NAAP providers are able to manage typical adverse effects of propofol as well as other sedatives. They should be trained in life support techniques such as BLS or ACLS, and NAAP training courses teach BLS techniques to all participants and ACLS to healthcare providers who practice in locations where an ACLS provider is not immediately available.1,3,13,16,17 Additionally, BLS techniques may be essential for an adequate response to adverse events during endoscopy sedation using other drugs besides propofol. The European Board of Anesthesiology recommends that NAAP-trained endoscopists who perform patient sedation should be trained in ACLS techniques including endotracheal intubation, and that training of nurses should be similar to the training of medical staff, but focused on BLS.16,18,19,20 However, ACLS training is usually not recommended as a routine course. Practical endotracheal intubation skills are difficult, and while training for extremely low-frequency of adverse events is not recommended, regular participation in education is needed for NAAP.3,13,16,17,21,22 At minimum, a BLS course is warranted in a basic endoscopic sedation curriculum, and more advanced education and training in ACLS may be necessary for prolonged and complex procedures such as endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasound (EUS)-interventional therapy, double-balloon enteroscopy, or endoscopic submucosal dissection (ESD) requiring deep sedation. If an ACLS provider is not present in the endoscopy unit or hospital, use of deep sedation with propofol may be restricted.
Simulator training
Simulator training using a full-scale patient simulator as an adjunct to practical NAAP courses may improve the skills of trainees. The European and MSCGE curricula both recommend the use of such simulator programs. Traditional clinical training does not reliably offer an opportunity for training in crisis management, nor does it address deficiencies in human performance under stress or the challenges of effective teamwork. Simulation-based sedation training includes both technical and human performance aspects of managing adverse events that might occur in the endoscopy unit by using a patient simulator and a simulated clinical environment.1,2,3 Simulator-based education allows training in pharmacology, appropriate selection of sedative drugs for use in endoscopic procedures or according to the needs of individual patient, pertinent monitoring techniques, including level of consciousness, pulse oximetry, frequency of observations, and management of complications of intravenous sedation, including basic to advanced life support and recovery care.
One study performed during a training course for sedation during endoscopy showed significant improvement in examination test scores of attendees after 3 hours of training that included hands-on management on a full-scale patient simulator compared with those before training.23 Direct endoscopy sedation under supervision or observation is also needed following simulator training, and should discuss various unexpected situations and complications. The European guidelines recommend that NAAP should be performed under supervision of an anesthesiologist or another person with previous experience of >300 NAAP cases. One retrospective study has shown that sedation-related complication rates during EUS examinations were significantly lower among advanced nurses (≥100 NAAP procedures) compared with the least-experienced nurses (≤30 NAAP procedures) (17.2% vs. 25.4%; odds ratio [OR], 0.61; 95% confidence interval [CI], 0.41 to 0.92).20 Simulator training can provide trainees with the specified knowledge and skills during a condensed period.
PERFORMANCE OF ENDOSCOPIC SEDATION
Conventional endoscopy under moderate sedation is usually performed by endoscopists or trained nurses. However, precisely who is qualified to perform sedation is an important problem particularly during the use of propofol. In the United States, over 75% of endoscopists use a benzodiazepine-plusnarcotic combination, most commonly midazolam and fentanyl, and propofol administration is directed by an anesthesiologist. 24 In Europe, various regimens are used and the administration of propofol differs according to national law.25 European countries in which NAAP has been established because of national legal restrictions include Austria, Denmark, Germany, Greece, The Netherlands, Sweden, and Switzerland.2 Regimens and the use of propofol differ according to national law. The 2008 United States guidelines as well as those of Germany have recommend that NAAP be performed by a person for whom NAAP is the sole task.13,21 However, recent reports have suggested that propofol administration in the presence of an endoscopist and a single nurse is safe, and no severe cardiovascular complications have been reported. In fact, endoscopists or nurses now administer propofol sedation widely and safely,4,5,6,7,8,26,27,28,29 and studies have shown that NAAP does not increase the risk of cardiovascular complications and is more cost-effective than anesthesiologist-administered sedation. There is no evidence to indicate that rapid availability of a life support team is required for propofol administration. However, it is recommended that patients be continuously monitored by a person dedicated to NAAP during endoscopic sedation. The ESGE does not recommend compulsory availability of a life support team if propofol is administered in the presence of a person trained in ACLS.3
Patient preparation and monitoring
Careful monitoring using continuous pulse oximetry and automated noninvasive blood pressure measurement from baseline to the recovery period is recommended for all patients. Continuous electrocardiography is recommended in selected patients who have a history of cardiac and/or pulmonary disease. Baseline, minimum, and maximum heart rate/blood pressure, and baseline and minimum oxygen blood saturation should be recorded. Automatic blood pressure monitors are widely available, relatively reliable, cheap, and easy to use. Although the potential benefit of routine prophylactic oxygen supplementation in terms of decreased cardiopulmonary complications is unclear, oxygen administration is widely recommended because oxygen desaturation is a frequent occurrence during endoscopy.30,31,32,33,34 These basic monitoring steps are recommended for all endoscopic sedation cases.
Visual assessment of respiratory activity during endoscopy procedures that are anticipated to be prolonged is not a reliable method of detecting apnea as most episodes of apnea and disordered respiration remain undetected by visual assessment of respiratory activity during anticipated prolonged endoscopy procedures.35 In a capnography-blinded group, hypoxemia developed significantly more frequently, with no difference in the clinically relevant complication rate, and another study showed that capnography erroneously displayed a flatline for at least 50 seconds in 13% of patients.36,37 Although capnographic monitoring of respiratory activity during NAAP may reduce episodes of hypoxemia during prolonged endoscopic procedures or when visual assessment of patient breathing is impaired, there has been no evidence of clinical impact. The clinical usefulness of capnography should be assessed in patients who are considered to be at high risk for morbidity from hypoxemia, and therefore, capnography is not recommended as a standard monitoring method.
Electroencephalography (EEG)-based monitoring may be used during NAAP to target a sedation level, which may help to reduce propofol consumption during complex endoscopic procedures targeting deep sedation. However, no clinical impact of EEG-based monitoring has been demonstrated, and no specific recommendations have been made due to the paucity of data.3 The bispectral index (BIS) is a variable derived from the EEG that allows quantification of the hypnotic effects of anesthetic drugs on the central nervous system. The usefulness of the BIS during propofol sedation for endoscopy has been evaluated. BIS is not associated with reductions in mean propofol dose or recovery time when used as an adjunct to nurse-administered propofol sedation for colonoscopy, or when used as the primary target for sedation.38 BIS monitoring during an ESD procedure did not lead to a reduction in the dose of propofol required, but did result in higher satisfaction scores among patients and endoscopists.39 There was no difference with regard to propofol consumption or recovery time between these two studies.38,39 On the other hand, randomized controlled trials including patients undergoing ERCP found that the propofol dose was significantly lower and the recovery time shorter in patients randomized to BIS compared with those randomized to clinical parameters for monitoring propofol administration.40,41
In conclusion, neither capnography nor BIS are recommended as routine monitoring tools. Careful inspection by a separate participant and basic monitoring by pulse oximetry, automated noninvasive blood pressure measurement, and electrocardiography are usually sufficient for safe endoscopic sedation. Capnography or BIS should be considered in selected high-risk patients or those undergoing planned deep sedation.
Level of sedation
Simple endoscopic procedures can be performed with moderate sedation using midazolam and maintain a high degree of patient satisfaction. However, prolonged or complex procedures such as EUS, ERCP, or ESD are frequently performed under deep sedation. Propofol is usually used to maintain a deep level of sedation, and it has been recommended that all providers be prepared to rescue patients from deeper levels of sedation than targeted. The level of sedation can be assessed using several scales. Trainees should learn to estimate adequate levels of sedation during endoscopy. The level of sedation can be estimated based on various modalities including the ASA classification, the Ramsay sedation scale, the Modified Richmond Agitation-Sedation score, or the Modified Observer's Assessment of Alertness/Sedation Scale (MOAA/S) (Tables 2,3,4,5).14,42 The Ramsay scale was developed in order to promote adequate sedation in the intensive care unit (ICU). It has six response choices that are quick and easy to assess. The MOAA/S scale is directed at determining the degree of suppression of consciousness and is widely used in the anesthesia research literature for quantifying the hypnotic effects of drugs. The two methods differ in that whereas the Ramsay scale mainly involves a passive approach to the patient and is designed to cause minimal disturbance to sleep, the MOAA/S scale entails a positive action by the observer ab initio in order to determine the patient's responsiveness. However, neither the MOAA/S nor the Ramsay scale takes into account the cardiorespiratory status. The Richmond Agitation-Sedation Scale (RASS), which is also often used for ICU patients, was developed to measure the dynamic concept of sedation and to prevent complications associated with agitation in critically ill adults. Because the RASS measures agitation, the scale may be inappropriate for measuring sedation and guiding adjustment of sedative medications.13,14,16,17,42 Thus, the choice of a sedation scoring system is usually a matter of personal preference. Observers should choose measurement parameters according to the situations of institution, and the Ramsay Sedation Scale and MOAA/S are both widely accepted sedation scoring systems.
Table 2.
Levels of Sedation according to the American Society of Anesthesiologists
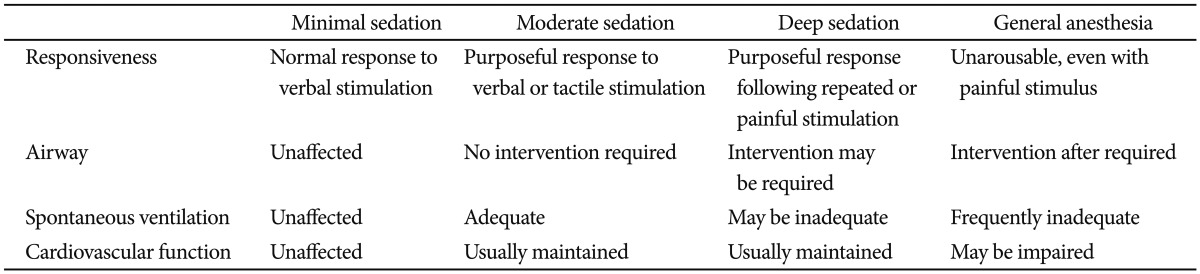
Table 3.
Ramsay Sedation Scale

Table 4.
Modified Observer's Assessment of Alertness/Sedation Scale
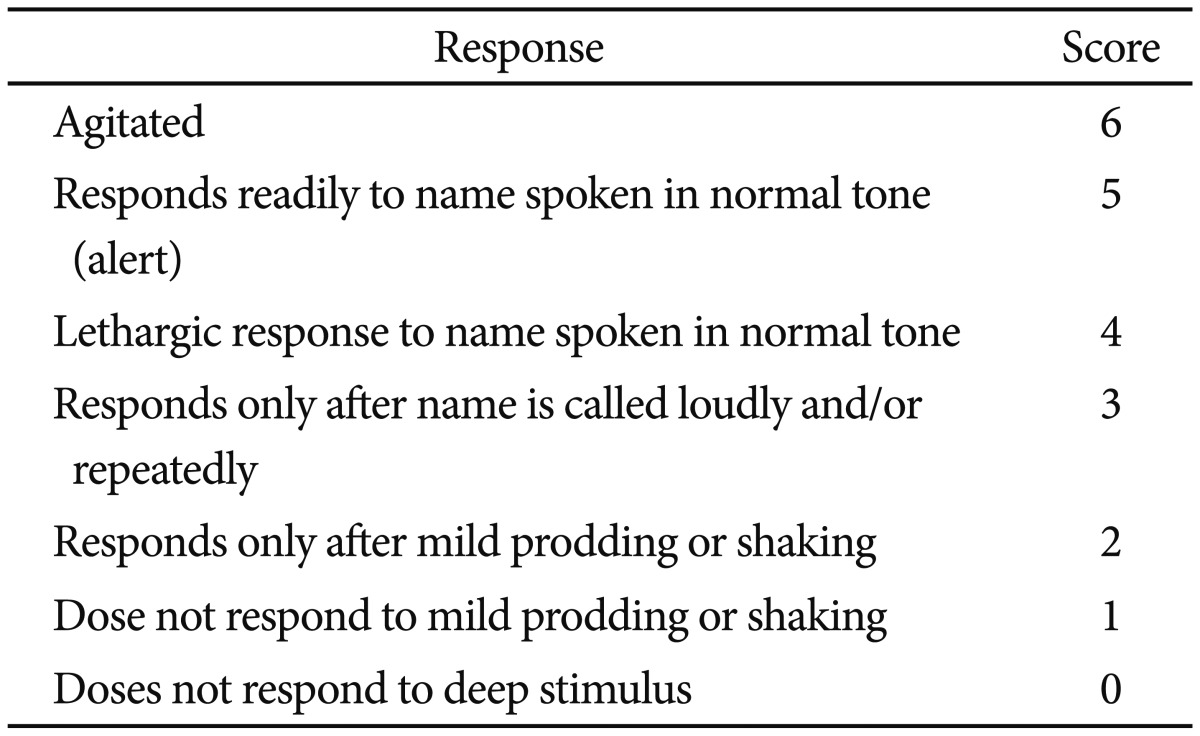
Table 5.
Modified Richmond Agitation-Sedation Score

However, as mentioned above, use of certain monitoring devices and techniques remains controversial, and distinguishing between moderate and deep sedation is frequently difficult because continuous monitoring of the sedation level may be difficult. Bedside observers may occasionally misjudge the level of sedation between monitoring time intervals. A continuous monitoring system has been developed, but no definite guidelines for its application have been introduced, and regular careful monitoring by a separate observer should be performed for deeper levels of sedation.
Management of adverse events
The most common complications during endoscopic sedation are hypoxemia and hypotension. Hypoxemia and hypotension are usually defined as hemoglobin oxygen saturation <90% and systolic blood pressure <90 mm Hg, respectively. Most cardiopulmonary complications can be prevented by careful monitoring of the sedation level and preprocedural risk stratification of patients. Additionally, there are antidotes to some sedative drugs, i.e., flumazenil or naloxone may be administered if benzodiazepines or opioids have been used. However, propofol does not have a pharmacologic antagonist, and it is also contraindicated in patients with a known allergy to soy protein. Propofol may also cause pain at the injection site. On average, 70% of patients report pain on injection, but this can be prevented in 60% of patients by intravenous administration of lidocaine (0.5 mg/kg) with a rubber tourniquet on the forearm.43 Although hypoxemia and hypotension are the most frequent adverse effects of propofol, according to a meta-analysis, the incidence during propofol-based sedation is 11% (95% CI, 7% to 16%) and 5% (95% CI, 2% to 10%), respectively. 44
Both the European curriculum and the MSCGE recommend that healthcare providers have BLS and/or ACLS training, particularly when they are involved in sedation using propofol.1,2 Measures that can be taken if hypoxemia develops include stopping the infusion of the sedative drugs, increasing the oxygen supply, maintaining a patent airway using the jawthrust maneuver, suctioning, and mask ventilation. If the patient does not respond adequately to these measures, the endoscopy should be stopped. If hypoxemia does not reverse, an emergency call must be performed according to local protocols, and ACLS must be initiated. In cases of arterial hypotension, an electrolyte solution should be administered, possibly with catecholamines. In cases of bradycardia, atropine should be administered intravenously. While ACLS training is required for advanced life support, these severe complications are rare, and ACLS is usually not recommended for routine certification. BLS should be sufficient for basic training endoscopy sedation courses, and ACLS can be taught in the more advanced training courses. The curricula for Simulator training includes BLS or ACLS.
Postsedation recovery care
The European curriculum and the MSCGE recommend that patients are observed by someone who is aware of the side effects of the drugs administered using monitoring equipment similar to that used during the procedure. During the recovery period, this observer can perform minor interruptible tasks but should not leave the room. Although it is possible to observe patients in the examination room, a separate room is recommended for practical reasons.1,2,3 A large prospective study has shown that serious adverse effects may occur up to 30 minutes after the administration of benzodiazepines and opioids for sedation, but postprocedural adverse effects represent <10% of per-procedural adverse effects. Serious postprocedural adverse effects are less frequent with propofol compared with the combination of midazolam/meperidine.45,46 A standardized discharge assessment scoring system such as the postanesthetic discharge scoring system or the Aldrete score (Table 6) is used.47
Table 6.
Aldrete Scoring System

Total score is 10. Patients who score ≥8 (and/or are returned to similar preoperative status) are considered fit for transition to phase II recovery.
BP, blood pressure.
Administration of specific drugs for endoscopic sedation
During training, trainees should learn the appropriate selection of patients for moderate or deep sedation based on the intended endoscopic procedure. Then, training in the pharmacological profiles of drugs, dosing regiments for induction and maintenance of sedation according to age and comorbidities, and safe injection practices is needed.1,2,3 As noted, there has been some conflicts between anesthesiologists and endoscopists regarding the use of propofol, and the MSCGE recommends a training process that includes both cognitive and procedural training. The cognitive training is a didactic component that incorporates lectures and independent study of a core of essential literature and the procedural training consists of two processes. Level 1 incorporates a high-fidelity sedation simulator, if available, and observation of a faculty physician managing sedation. Level 2 is the independent ordering and administration of sedation drugs under faculty supervision. After these processes, trainees should participate in a discussion of cases of sedation-related adverse events.1 The European curriculum also recommends that trainees are aware of all sedatives and rescue drugs used for endoscopy sedation, including the pharmacological principles, pharmacokinetics, dosing, application techniques, contraindications, and side effects of individual drugs and the combinations of drugs used for sedation.2
Assessments of competence
Whenever possible, basic knowledge such as pharmacology and the use of physiological monitoring should be established before the trainee is placed in the procedure room environment. Simulators and web-based programs that are designed to assess technical and cognitive abilities should be used whenever possible. The MSCGE uses a web-based objective examination, patient simulation, or direct observation with or without oral testing of trainees.1,2 ESGENA-ESGE and MSCGE provide certificates to each student who successfully completes the training course.1,2 As these certification programs are currently not available in the domestic education program for Korea, regular education and advanced periodic education and certification are needed.
Special considerations for endoscopic sedation
The MSCGE includes indications and contraindications for endoscopy and information on the safety of commonly used medications for endoscopy during pregnancy. This curriculum also recommends that trainees gain an understanding of the clinical conditions, history, and physical findings that may predispose a patient to an increased risk of cardiopulmonary complications with standard sedation. Thus, the training process involves didactic lectures as well as clinical instruction and demonstration. On the basis of this training, an appropriate level of anesthesiologist assistance for an endoscopic procedure is established. Patient- or procedure-related risk factors including prolonged or therapeutic endoscopic procedures requiring deep sedation or general anesthesia, anticipated intolerance, paradoxical reaction or allergy to standard sedation regimens, increased risk of complications because of severe comorbidity (ASA class 4 and higher), increased risk of airway obstruction, history of stridor and severe sleep apnea, dysmorphic facial features, oral abnormalities, neck abnormalities, and jaw abnormalities may contribute to the decision to have an anesthesiologist present.1
Finally, trainees should be aware of legal issues when performing sedation, particularly for the use of propofol. In some countries, all sedation can be administered by all trained clinicians, while in others administration of propofol can be performed only by anesthesiologists. In the majority of European countries, endoscopists administer sedation with support from endoscopy nurses, whereas in some countries, including France, only anesthesiologists administer intravenous sedation.2 The current Food and Drug Administration-approved product label for propofol states that it should be administered only by individuals trained in the administration of general anesthesia.13 The narrow therapeutic window of propofol distinguishes it from the conventional sedative hypnotics that are used for endoscopy and may increase the risk of cardiopulmonary complications if not administered appropriately. An ASA task force has recommended that patients who receive propofol should receive care consistent with deep sedation and that the involved personnel should be capable of rescuing a patient from general anesthesia.48 However, there is now abundant evidence that propofol can be administered safely by nonanesthesiologists, as during the last decade, a number of studies have addressed the safety and efficacy of propofol administration during endoscopy by either physicians or trained nurses.4,5,49,50,51,52,53,54,55,56,57,58,59 Gastroenterologist endoscopists regard NAAP as a safe and cost-effective method for endoscopic sedation and will usually perform it, although the majority opinion of anesthesiology societies concerning the use of propofol by nonanesthesiologists is reportedly negative.
CONCLUSIONS
An adequate level of satisfactory endoscopic sedation is now essential for the safety of patients and for successful therapeutic endoscopy. Current endoscopic sedation curricula and guidelines are well-established in Europe and the United States. Basic knowledge and actual advanced performance should be required particularly in NAAP, and taking into consideration the various legal frameworks and healthcare systems of each country. Moreover, a more systematized curriculum and a regular education system extending from training to expert performance should be established to facilitate safe and successful endoscopic sedation. This should be based on an updated international consensus regarding the use of sedative agents, particularly propofol, and on the current domestic legal framework.
Acknowledgments
This work was supported by the Soonchunhyang University Research Fund.
Footnotes
The authors have no financial conflicts of interest.
References
- 1.American Association for Study of Liver Diseases; American College of Gastroenterology; American Gastroenterological Association Institute et al. Multisociety sedation curriculum for gastrointestinal endoscopy. Gastrointest Endosc. 2012;76:e1–e25. doi: 10.1016/j.gie.2012.03.001. [DOI] [PubMed] [Google Scholar]
- 2.Dumonceau JM, Riphaus A, Beilenhoff U, et al. European curriculum for sedation training in gastrointestinal endoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastroenterology and Endoscopy Nurses and Associates (ESGENA) Endoscopy. 2013;45:496–504. doi: 10.1055/s-0033-1344142. [DOI] [PubMed] [Google Scholar]
- 3.Dumonceau JM, Riphaus A, Aparicio JR, et al. European Society of Gastrointestinal Endoscopy, European Society of Gastroenterology and Endoscopy Nurses and Associates, and the European Society of Anaesthesiology Guideline: non-anesthesiologist administration of propofol for GI endoscopy. Endoscopy. 2010;42:960–974. doi: 10.1055/s-0030-1255728. [DOI] [PubMed] [Google Scholar]
- 4.Lee CK, Lee SH, Chung IK, et al. Balanced propofol sedation for therapeutic GI endoscopic procedures: a prospective, randomized study. Gastrointest Endosc. 2011;73:206–214. doi: 10.1016/j.gie.2010.09.035. [DOI] [PubMed] [Google Scholar]
- 5.Lee TH, Lee CK, Park SH, et al. Balanced propofol sedation versus propofol monosedation in therapeutic pancreaticobiliary endoscopic procedures. Dig Dis Sci. 2012;57:2113–2121. doi: 10.1007/s10620-012-2234-0. [DOI] [PubMed] [Google Scholar]
- 6.Chun SY, Kim KO, Park DS, et al. Safety and efficacy of deep sedation with propofol alone or combined with midazolam administrated by nonanesthesiologist for gastric endoscopic submucosal dissection. Gut Liver. 2012;6:464–470. doi: 10.5009/gnl.2012.6.4.464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oh JE, Lee HJ, Lee YH. Propofol versus midazolam for sedation during esophagogastroduodenoscopy in children. Clin Endosc. 2013;46:368–372. doi: 10.5946/ce.2013.46.4.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kang SH, Hyun JJ. Preparation and patient evaluation for safe gastrointestinal endoscopy. Clin Endosc. 2013;46:212–218. doi: 10.5946/ce.2013.46.3.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yu YH, Han DS, Kim HS, et al. Efficacy of bispectral index monitoring during balanced propofol sedation for colonoscopy: a prospective, randomized controlled trial. Dig Dis Sci. 2013;58:3576–3583. doi: 10.1007/s10620-013-2833-4. [DOI] [PubMed] [Google Scholar]
- 10.Cha JM, Jeun JW, Pack KM, et al. Risk of sedation for diagnostic esophagogastroduodenoscopy in obstructive sleep apnea patients. World J Gastroenterol. 2013;19:4745–4751. doi: 10.3748/wjg.v19.i29.4745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee SH, Kim SJ, Lee TH, et al. Human applications of submucosal endoscopy under conscious sedation for pure natural orifice transluminal endoscopic surgery. Surg Endosc. 2013;27:3016–3020. doi: 10.1007/s00464-013-2844-4. [DOI] [PubMed] [Google Scholar]
- 12.Park CH, Min JH, Yoo YC, et al. Sedation methods can determine performance of endoscopic submucosal dissection in patients with gastric neoplasia. Surg Endosc. 2013;27:2760–2767. doi: 10.1007/s00464-013-2804-z. [DOI] [PubMed] [Google Scholar]
- 13.Standards of Practice Committee of the American Society for Gastrointestinal Endoscopy. Lichtenstein DR, Jagannath S, et al. Sedation and anesthesia in GI endoscopy. Gastrointest Endosc. 2008;68:815–826. doi: 10.1016/j.gie.2008.09.029. [DOI] [PubMed] [Google Scholar]
- 14.American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology. 2002;96:1004–1017. doi: 10.1097/00000542-200204000-00031. [DOI] [PubMed] [Google Scholar]
- 15.Kuwatani M, Kawakami H, Hayashi T, et al. Carbon dioxide insufflation during endoscopic retrograde cholangiopancreatography reduces bowel gas volume but does not affect visual analogue scale scores of suffering: a prospective, double-blind, randomized, controlled trial. Surg Endosc. 2011;25:3784–3790. doi: 10.1007/s00464-011-1789-8. [DOI] [PubMed] [Google Scholar]
- 16.Knape JT, Adriaensen H, van Aken H, et al. Guidelines for sedation and/or analgesia by non-anaesthesiology doctors. Eur J Anaesthesiol. 2007;24:563–567. doi: 10.1017/S0265021507000452. [DOI] [PubMed] [Google Scholar]
- 17.Cohen LB, Delegge MH, Aisenberg J, et al. AGA institute review of endoscopic sedation. Gastroenterology. 2007;133:675–701. doi: 10.1053/j.gastro.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 18.Rex DK, Heuss LT, Walker JA, Qi R. Trained registered nurses/endoscopy teams can administer propofol safely for endoscopy. Gastroenterology. 2005;129:1384–1391. doi: 10.1053/j.gastro.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 19.Rex DK, Overley CA, Walker J. Registered nurse-administered propofol sedation for upper endoscopy and colonoscopy: why? when? how? Rev Gastroenterol Disord. 2003;3:70–80. [PubMed] [Google Scholar]
- 20.Fatima H, DeWitt J, LeBlanc J, Sherman S, McGreevy K, Imperiale TF. Nurse-administered propofol sedation for upper endoscopic ultrasonography. Am J Gastroenterol. 2008;103:1649–1656. doi: 10.1111/j.1572-0241.2008.01906.x. [DOI] [PubMed] [Google Scholar]
- 21.Riphaus A, Wehrmann T, Weber B, et al. S3-guidelines: sedation in gastrointestinal endoscopy. Z Gastroenterol. 2008;46:1298–1330. doi: 10.1055/s-2008-1027850. [DOI] [PubMed] [Google Scholar]
- 22.Byrne MF, Chiba N, Singh H, Sadowski DC Clinical Affairs Committee of the Canadian Association of Gastroenterology. Propofol use for sedation during endoscopy in adults: a Canadian Association of Gastroenterology position statement. Can J Gastroenterol. 2008;22:457–459. doi: 10.1155/2008/268320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.DeMaria S, Jr, Levine AI, Cohen LB. Human patient simulation and its role in endoscopic sedation training. Gastrointest Endosc Clin N Am. 2008;18:801–813. doi: 10.1016/j.giec.2008.06.010. [DOI] [PubMed] [Google Scholar]
- 24.Cohen LB, Wecsler JS, Gaetano JN, et al. Endoscopic sedation in the United States: results from a nationwide survey. Am J Gastroenterol. 2006;101:967–974. doi: 10.1111/j.1572-0241.2006.00500.x. [DOI] [PubMed] [Google Scholar]
- 25.Riphaus A, Rabofski M, Wehrmann T. Endoscopic sedation and monitoring practice in Germany: results from the first nationwide survey. Z Gastroenterol. 2010;48:392–397. doi: 10.1055/s-0028-1109765. [DOI] [PubMed] [Google Scholar]
- 26.Heuss LT, Schnieper P, Drewe J, Pflimlin E, Beglinger C. Risk stratification and safe administration of propofol by registered nurses supervised by the gastroenterologist: a prospective observational study of more than 2000 cases. Gastrointest Endosc. 2003;57:664–671. doi: 10.1067/mge.2003.191. [DOI] [PubMed] [Google Scholar]
- 27.Cohen LB, Dubovsky AN, Aisenberg J, Miller KM. Propofol for endoscopic sedation: a protocol for safe and effective administration by the gastroenterologist. Gastrointest Endosc. 2003;58:725–732. doi: 10.1016/s0016-5107(03)02010-8. [DOI] [PubMed] [Google Scholar]
- 28.Külling D, Orlandi M, Inauen W. Propofol sedation during endoscopic procedures: how much staff and monitoring are necessary? Gastrointest Endosc. 2007;66:443–449. doi: 10.1016/j.gie.2007.01.037. [DOI] [PubMed] [Google Scholar]
- 29.Yusoff IF, Raymond G, Sahai AV. Endoscopist administered propofol for upper-GI EUS is safe and effective: a prospective study in 500 patients. Gastrointest Endosc. 2004;60:356–360. doi: 10.1016/s0016-5107(04)01711-0. [DOI] [PubMed] [Google Scholar]
- 30.Bell GD, Quine A, Antrobus JH, et al. Upper gastrointestinal endoscopy: a prospective randomized study comparing continuous supplemental oxygen via the nasal or oral route. Gastrointest Endosc. 1992;38:319–325. doi: 10.1016/s0016-5107(92)70424-6. [DOI] [PubMed] [Google Scholar]
- 31.Bowling TE, Hadjiminas CL, Polson RJ, Baron JH, Foale RA. Effects of supplemental oxygen on cardiac rhythm during upper gastrointestinal endoscopy: a randomised controlled double blind trial. Gut. 1993;34:1492–1497. doi: 10.1136/gut.34.11.1492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Crantock L, Cowen AE, Ward M, Roberts RK. Supplemental low flow oxygen prevents hypoxia during endoscopic cholangiopancreatography. Gastrointest Endosc. 1992;38:418–420. doi: 10.1016/s0016-5107(92)70468-4. [DOI] [PubMed] [Google Scholar]
- 33.Haines DJ, Bibbey D, Green JR. Does nasal oxygen reduce the cardiorespiratory problems experienced by elderly patients undergoing endoscopic retrograde cholangiopancreatography? Gut. 1992;33:973–975. doi: 10.1136/gut.33.7.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rozario L, Sloper D, Sheridan MJ. Supplemental oxygen during moderate sedation and the occurrence of clinically significant desaturation during endoscopic procedures. Gastroenterol Nurs. 2008;31:281–285. doi: 10.1097/01.SGA.0000334034.94370.bf. [DOI] [PubMed] [Google Scholar]
- 35.Vargo JJ, Zuccaro G, Jr, Dumot JA, Conwell DL, Morrow JB, Shay SS. Automated graphic assessment of respiratory activity is superior to pulse oximetry and visual assessment for the detection of early respiratory depression during therapeutic upper endoscopy. Gastrointest Endosc. 2002;55:826–831. doi: 10.1067/mge.2002.124208. [DOI] [PubMed] [Google Scholar]
- 36.Qadeer MA, Vargo JJ, Dumot JA, et al. Capnographic monitoring of respiratory activity improves safety of sedation for endoscopic cholangiopancreatography and ultrasonography. Gastroenterology. 2009;136:1568–1576. doi: 10.1053/j.gastro.2009.02.004. [DOI] [PubMed] [Google Scholar]
- 37.Lightdale JR, Goldmann DA, Feldman HA, Newburg AR, DiNardo JA, Fox VL. Microstream capnography improves patient monitoring during moderate sedation: a randomized, controlled trial. Pediatrics. 2006;117:e1170–e1178. doi: 10.1542/peds.2005-1709. [DOI] [PubMed] [Google Scholar]
- 38.Drake LM, Chen SC, Rex DK. Efficacy of bispectral monitoring as an adjunct to nurse-administered propofol sedation for colonoscopy: a randomized controlled trial. Am J Gastroenterol. 2006;101:2003–2007. doi: 10.1111/j.1572-0241.2006.00806.x. [DOI] [PubMed] [Google Scholar]
- 39.Imagawa A, Fujiki S, Kawahara Y, et al. Satisfaction with bispectral index monitoring of propofol-mediated sedation during endoscopic submucosal dissection: a prospective, randomized study. Endoscopy. 2008;40:905–909. doi: 10.1055/s-2008-1077641. [DOI] [PubMed] [Google Scholar]
- 40.Paspatis GA, Chainaki I, Manolaraki MM, et al. Efficacy of bispectral index monitoring as an adjunct to propofol deep sedation for ERCP: a randomized controlled trial. Endoscopy. 2009;41:1046–1051. doi: 10.1055/s-0029-1215342. [DOI] [PubMed] [Google Scholar]
- 41.Wehrmann T, Grotkamp J, Stergiou N, et al. Electroencephalogram monitoring facilitates sedation with propofol for routine ERCP: a randomized, controlled trial. Gastrointest Endosc. 2002;56:817–824. doi: 10.1067/mge.2002.129603. [DOI] [PubMed] [Google Scholar]
- 42.Ely EW, Truman B, Shintani A, et al. Monitoring sedation status over time in ICU patients: reliability and validity of the Richmond Agitation-Sedation Scale (RASS) JAMA. 2003;289:2983–2991. doi: 10.1001/jama.289.22.2983. [DOI] [PubMed] [Google Scholar]
- 43.Picard P, Tramèr MR. Prevention of pain on injection with propofol: a quantitative systematic review. Anesth Analg. 2000;90:963–969. doi: 10.1097/00000539-200004000-00035. [DOI] [PubMed] [Google Scholar]
- 44.McQuaid KR, Laine L. A systematic review and meta-analysis of randomized, controlled trials of moderate sedation for routine endoscopic procedures. Gastrointest Endosc. 2008;67:910–923. doi: 10.1016/j.gie.2007.12.046. [DOI] [PubMed] [Google Scholar]
- 45.Newman DH, Azer MM, Pitetti RD, Singh S. When is a patient safe for discharge after procedural sedation? The timing of adverse effect events in 1367 pediatric procedural sedations. Ann Emerg Med. 2003;42:627–635. doi: 10.1016/s0196-0644(03)00446-3. [DOI] [PubMed] [Google Scholar]
- 46.Jung M, Hofmann C, Kiesslich R, Brackertz A. Improved sedation in diagnostic and therapeutic ERCP: propofol is an alternative to midazolam. Endoscopy. 2000;32:233–238. doi: 10.1055/s-2000-96. [DOI] [PubMed] [Google Scholar]
- 47.Aldrete JA, Kroulik D. A postanesthetic recovery score. Anesth Analg. 1970;49:924–934. [PubMed] [Google Scholar]
- 48.Zakko SF, Seifert HA, Gross JB. A comparison of midazolam and diazepam for conscious sedation during colonoscopy in a prospective double-blind study. Gastrointest Endosc. 1999;49:684–689. doi: 10.1016/s0016-5107(99)70282-8. [DOI] [PubMed] [Google Scholar]
- 49.Tohda G, Higashi S, Wakahara S, Morikawa M, Sakumoto H, Kane T. Propofol sedation during endoscopic procedures: safe and effective administration by registered nurses supervised by endoscopists. Endoscopy. 2006;38:360–367. doi: 10.1055/s-2005-921192. [DOI] [PubMed] [Google Scholar]
- 50.Training Committee of the American Society for Gastrointestinal Endoscopy. Vargo JJ, Ahmad AS, et al. Training in patient monitoring and sedation and analgesia. Gastrointest Endosc. 2007;66:7–10. doi: 10.1016/j.gie.2007.02.028. [DOI] [PubMed] [Google Scholar]
- 51.Cohen LB, Ladas SD, Vargo JJ, et al. Sedation in digestive endoscopy: the Athens international position statements. Aliment Pharmacol Ther. 2010;32:425–442. doi: 10.1111/j.1365-2036.2010.04352.x. [DOI] [PubMed] [Google Scholar]
- 52.Dinis-Ribeiro M, Vargo JJ. Sedation by non-anesthesiologists: are opioids and benzodiazepines outdated? Digestion. 2010;82:100–101. doi: 10.1159/000287215. [DOI] [PubMed] [Google Scholar]
- 53.Levitzky BE, Lopez R, Dumot JA, Vargo JJ. Moderate sedation for elective upper endoscopy with balanced propofol versus fentanyl and midazolam alone: a randomized clinical trial. Endoscopy. 2012;44:13–20. doi: 10.1055/s-0031-1291421. [DOI] [PubMed] [Google Scholar]
- 54.Vargo JJ, Cohen LB, Rex DK, et al. Position statement: nonanesthesiologist administration of propofol for GI endoscopy. Gastroenterology. 2009;137:2161–2167. doi: 10.1053/j.gastro.2009.09.050. [DOI] [PubMed] [Google Scholar]
- 55.Chen SC, Rex DK. Review article: registered nurse-administered propofol sedation for endoscopy. Aliment Pharmacol Ther. 2004;19:147–155. doi: 10.1111/j.0269-2813.2004.01833.x. [DOI] [PubMed] [Google Scholar]
- 56.Cohen LB, Cattau E, Goetsch A, et al. A randomized, double-blind, phase 3 study of fospropofol disodium for sedation during colonoscopy. J Clin Gastroenterol. 2010;44:345–353. doi: 10.1097/MCG.0b013e3181c2987e. [DOI] [PubMed] [Google Scholar]
- 57.Rex DK, Deenadayalu VP, Eid E, et al. Endoscopist-directed administration of propofol: a worldwide safety experience. Gastroenterology. 2009;137:1229–1237. doi: 10.1053/j.gastro.2009.06.042. [DOI] [PubMed] [Google Scholar]
- 58.Ulmer BJ, Hansen JJ, Overley CA, et al. Propofol versus midazolam/fentanyl for outpatient colonoscopy: administration by nurses supervised by endoscopists. Clin Gastroenterol Hepatol. 2003;1:425–432. doi: 10.1016/s1542-3565(03)00226-x. [DOI] [PubMed] [Google Scholar]
- 59.VanNatta ME, Rex DK. Propofol alone titrated to deep sedation versus propofol in combination with opioids and/or benzodiazepines and titrated to moderate sedation for colonoscopy. Am J Gastroenterol. 2006;101:2209–2217. doi: 10.1111/j.1572-0241.2006.00760.x. [DOI] [PubMed] [Google Scholar]


