Abstract
Five canonical tastes, bitter, sweet, umami (amino acid), salty and sour (acid) are detected by animals as diverse as fruit flies and humans, consistent with a near universal drive to consume fundamental nutrients and to avoid toxins or other harmful compounds. Surprisingly, despite this strong conservation of basic taste qualities between vertebrates and invertebrates, the receptors and signaling mechanisms that mediate taste in each are highly divergent. The identification over the last two decades of receptors and other molecules that mediate taste has led to stunning advances in our understanding of the basic mechanisms of transduction and coding of information by the gustatory systems of vertebrates and invertebrates. In this review, we discuss recent advances in taste research, mainly from the fly and mammalian systems, and we highlight principles that are common across species, despite stark differences in receptor types.
Introduction
The sense of taste is essential for life – it tells us which prospective foods are nutritious, while warning us of those that are toxic. Five basic tastes are recognized by humans and most other animals - bitter, sweet, sour, salty and umami (the taste of MSG). Natural philosophers as early as Aristotle recognized that “all organisms are nourished by the sweet,” or stated otherwise, calorie-rich sugars taste good. Aristotle also recognized that bitter taste elicits rejection, a feature he ascribed to “its heaviness.” Much has been learned about taste in the intervening time, with spectacular progress in the last 15 years, during which time receptors for many of the canonical tastes have been identified in a variety of vertebrates and invertebrates (Clyne et al., 2000; Dunipace et al., 2001; Robertson et al., 2003; Scott et al., 2001; Yarmolinsky et al., 2009). In this review we compare the peripheral taste systems of two vertebrates mice and humans, with a model invertebrate, the fruit fly, Drosophila melanogaster.
An obvious difference between mice and humans, relative to flies, is that mammals have a tongue and soft palate that contain taste receptor cells (TRCs), while fruit flies distribute their taste receptor cells, referred to as gustatory receptor cells, on a variety of structures on the head, body and legs. Nonetheless, given the observation that these diverse organisms detect virtually the same classes of chemicals, one might have predicted that taste detection would be mediated through evolutionarily conserved receptors. But this is not the case. In mice and humans, bitter, sweet and umami are detected by dedicated G-protein-coupled receptors, which presumably allows for amplification of small sensory responses. In contrast, the major class of taste receptors for bitter and sweet in flies (gustatory receptors; GRs) are unrelated to classical GPCRs and may form ligand-activated ion channels (Sato et al., 2011).
In addition to the five canonical taste qualities, there is growing evidence that many vertebrates and invertebrates use their gustatory systems to detect the presence of other compounds, that may include Ca2+, CO2, water and fats (Fischler et al., 2007; Gaillard et al., 2008; Inoshita and Tanimura, 2006; Masek and Keene, 2013). Interestingly, because taste is tuned to ecological niche, there is considerable variability in the repertoire of receptors expressed by different animals, with some animals missing entire classes of receptors. Adding to this complexity, in many species, receptors originally identified as taste receptors are found in nongustatory tissues, where they have diverse functions. For example, fly GRs sense non-volatile pheromones that help animals select mates and guide aggression. In both flies and mammals, taste receptors are found on internal organs where they can sense ingested compounds and their metabolites, and thus guide post ingestive behaviors.
Here we will survey the wide breadth of research into taste receptors and signaling pathways using examples mainly from the fruit fly, mice and humans. In addition to focusing primarily on their classical roles in taste, we describe recent studies demonstrating roles for taste receptors in cells and tissue external to the mouthparts.
Mammalian taste anatomy
In vertebrates, taste stimuli are detected by taste receptors cells (TRCs), which are located in taste buds on the tongue and palate epithelium (Figures 1A and 1B). Taste buds are found in dense groupings at the back and sides of the tongue, (Figure 1A, circumvallate and foliate papillae, respectively), whereas unitary taste buds are scattered across the front of the tongue and on the palate (Figure 1A, fungiform). TRCs are compact modified epithelial cells that extend a process to the apical surface of the epithelium, where a taste pore allows direct contact with chemicals in the environment.
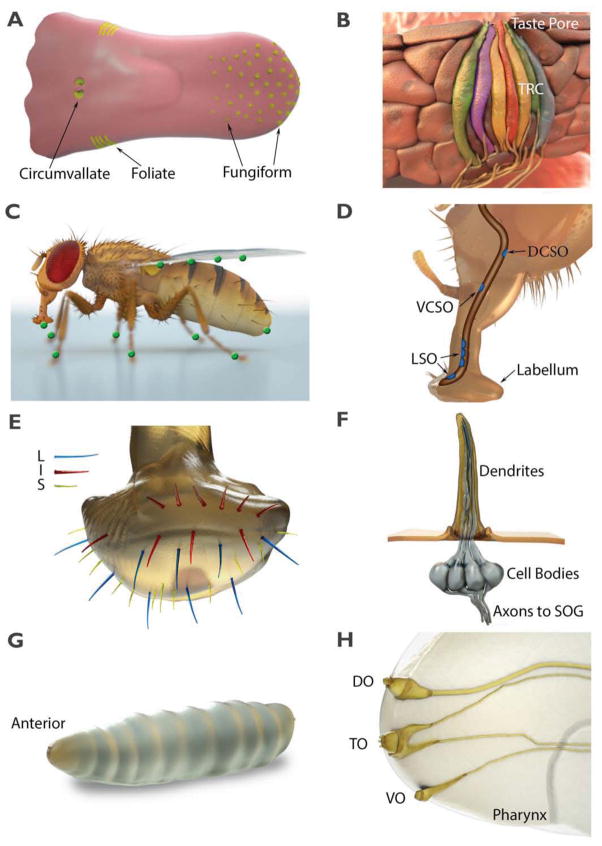
Figure 1.
The taste organs in mammals, such as mice and humans, and in flies. (A) The rodent tongue contains taste buds that are located in three distinct regions. Taste buds are also found on the palate (not shown). (B) The taste bud is composed of 50–100 modified epithelial cells that extend a process to the taste pore, where they come into contact with ingested chemicals. At least five types of sensory cells (depicted in different colors) are found in the taste bud, corresponding to the five canonical tastes. (C) Green circles indicate locations of external gustatory organs distributed on an adult Drosophila female. (D) The Drosophila proboscis. Shown are the labellum, and three internal taste organs indicated in blue: the labral sense organs (LSOs), the dorsal cibarial sense organ (DCSO), and the ventral cibarial sense organ (VCSO). (E) Distribution of the L-, I- and S-type sensilla on a fly labellum. (F) An S- or L-type sensilla containing four GRNs. The accessory cells are not shown. (G) A Drosophila larvae. The external chemosensory organs are located at the anterior. (H) Anterior end of a larvae. The locations of the dorsal organ (DO), the terminal organ (TO), and the ventral organ (VO) are indicated.
In most species the taste bud contains at least three morphologically distinct cell types (Type I, II and III) that constitute at least five functional classes of sensory cells, each specialized to detect one of the five basic taste qualities (bitter, sweet, umami, sour and salty) (Figure 1B). Taste cells are short-lived and are precisely replenished from proliferative basal keratinocytes (Kapsimali and Barlow, 2012). Cells conveying one taste quality can relay information independent of cells relaying other taste qualities, as shown by genetic inactivation of individual cell types (Chandrashekar et al., 2006). Taste cells release numerous neurotransmitters (e.g. serotonin, etc) and express neurotransmitter receptors, suggesting that there is communication among cells in the taste bud, which may shape the output of the bud (Chaudhari and Roper, 2010).
Vertebrate taste cells do not possess an axon, and instead are innervated by pseudo unipolar neurons whose cell bodies reside in the petrosal and geniculate ganglia. Two nerves carry most of the taste information: the chorda tympani nerve, which innervates the anterior tongue, containing the fungiform papillae, and the glossopharyngeal nerve, which innervates the posterior tongue and most of the palate. Neurons from taste ganglia project to the nucleus of the solitary tract, and from there information is relayed to the gustatory cortex (Smith and David, 2000).
Drosophila taste anatomy
A remarkable feature of the taste system in flies and many other insects is that the taste organs are not restricted to the head but are distributed on multiple body parts (Figure 1C) (Stocker, 1994; Vosshall and Stocker, 2007). In adult flies, the closest equivalent to the mammalian tongue is a long appendage extending from the head—the proboscis, which is comprised of external and internal taste organs (Figure 1D). The external taste organ consists of two labella (also referred to as labial palps) that are fused together at the end of the proboscis (Figures 1D and 1E). Internal gustatory structures line the pharynx (Figure 1D), and serve as the final gatekeeper, allowing the fly to make the final decision as to whether to expel the food or allow it to proceed to the digestive system.
Taste sensors are also distributed on the legs and anterior wing margins. While the function of gustatory sensors on the legs and the labellum in sampling foods before ingestion is clear, the role of gustatory cells on the wings remains enigmatic. Another surprising location for taste organs is on the ovipostor, where they can provide information as to the suitability of the environment prior to egg laying (Stocker, 1994).
Unlike their vertebrate counterparts, fly gustatory receptor neurons (GRNs) are bona fide bipolar neurons that extend dendrites into hairlike bristles referred to as sensilla (Figure 1F), and project axons to the central taste center in the subesophageal ganglion (SOG) (Falk et al., 1976). In each labellum there are 31 sensilla, which are subgrouped based on length into nearly equal numbers of long (L), intermediate (I) and short (S) types (Figure 1E). The L- and S-type sensilla each house four GRNs, while the I-type sensilla contain two GRNs. There are also ~30—40 conically shaped taste pegs that house one GRN and one mechanosensory neuron. Each sensillum is bestowed with one mechanosensory cell, and three accessory cells—the trichogen (shaft), the tormogen (socket), and the thecogen (sheath).
Single GRNs tend to respond to either attractive stimuli, such as low salt or sugars or aversive compounds, including high salt and bitter compounds, but not to stimuli of different valence. In general, the four different GRNs in L-type sensilla are most sensitive to attractive stimuli and respond only weakly to aversive stimuli (Hiroi et al., 2002). Each of the four neurons is tuned to different stimuli; one is strongly responsive to low salt, another is strongly responsive to sugars, a third is weakly responsive to high salt, and the fourth is moderately responsive to water. In contrast, the four GRNs in S-type sensilla are most sensitive to bitter compounds and high salt, and respond only weakly to low salt and sugars. The I-type sensilla include two GRNs, one excited by a narrow group of bitter compounds and high, aversive levels of salt (Hiroi et al., 2004; Weiss et al., 2011) and the other activated by sugars and low levels of salt that are attractive.
The taste system in Drosophila larvae includes sensilla located on three organs in the head region (dorsal, terminal and ventral organs) (Figures 1G and 1H), as well as three organs in the pharynx (dorsal, ventral and posterior pharyngeal sense organs) (Stocker, 1994; Vosshall and Stocker, 2007). The cell bodies associated with the dendrites in the external organs are present in three discrete ganglia. The dorsal organ mainly contains olfactory sensilla, and both the internal and external organs may also respond to mechanical, thermosensory and hygrosensory input (Liu et al., 2003b; Stocker, 2008).
Taste Coding
Theories of coding have generally been described as conforming either to a labeled line model, in which each cell represents a distinct taste quality and communicates essentially without interruption to the central nervous system, or to a distributive model, in which cells respond in varying amounts to each taste quality, and the central nervous system makes sense of the chorus of activity. This latter model appears to hold for the olfactory system of vertebrates, where odorants bind to a large number of olfactory receptors that are, in turn, sensitive to a range of odorants (Buck, 1996) Odorant identity is therefore encoded by the relative responses of sensory receptor cells. Chemosensation in the nematode, C. elegans, is, in contrast, a classic example of a labeled line model system (Troemel et al., 1997).
Taste in flies and mammals adheres loosely to a labeled line model of coding. In flies, single neurons can detect multiple taste qualities, but these taste qualities, in general, have the same valence (behavioral output). This “valence labeled line” model is supported by the observation that some GRNs are activated by sugars, and low levels of fatty acids, both of which promote feeding (Wisotsky et al., 2011) while other GRNs are activated by bitter compounds and high concentrations of salt, which suppress feeding (Hiroi et al., 2004). In addition, a subset of bitter GRNs is also activated by low pH carboxylic acids, which are feeding deterrents (Charlu et al., 2013).
The taste system of mice also uses a variant of the labeled line model. In mice, taste receptors are, in general, segregated into distinct populations such that bitter, sweet, sour and low concentrations of salt are detected by non-overlapping sets of cells (Voigt et al., 2012; Yarmolinsky et al., 2009). Whether this principle applies to sweet and umami, such that cells are responsive to one or the other stimulus but not both is presently unclear. An initial study reports that receptors for each are expressed in completely non-overlapping populations of cells (Hoon et al., 1999), but this conclusion is questioned by more recent experiments suggesting that the umami receptor is expressed in sweet-responsive TRCs (Kusuhara et al., 2013). Moreover, reminiscent of the valence label line model in flies, there is recent evidence that aversively high concentrations of salt are not detected by a separate subset of cells, but are instead detected by the populations of cells that detect bitter and sour (Oka et al., 2013). Nevertheless, in an elegant set of experiments, expression of a completely artificial receptor (RASSL) in bitter or sweet responsive cells, makes the previously tasteless ligand, spiradoline, either aversive or attractive, respectively (Mueller et al., 2005). This finding drives home the principle that the mammalian taste system is relatively hard-wired to behavior, as is the case in flies.
Mammalian bitter, sweet and amino acid taste: reception and signaling
The best understood of the five tastes are bitter, sweet and umami – tastes that are generally evoked by organic compounds in the environment. All three are mediated by specialized, taste-specific G protein coupled receptors (GPCRs), which are expressed in distinct subsets of taste receptor cells (Chandrashekar et al., 2006) (Figure 2A). The identification of the taste receptors (TRs) and downstream signaling components for each of the three tastes has precipitated enormous advances in our understanding of the cell biology, genetics and evolution of taste.
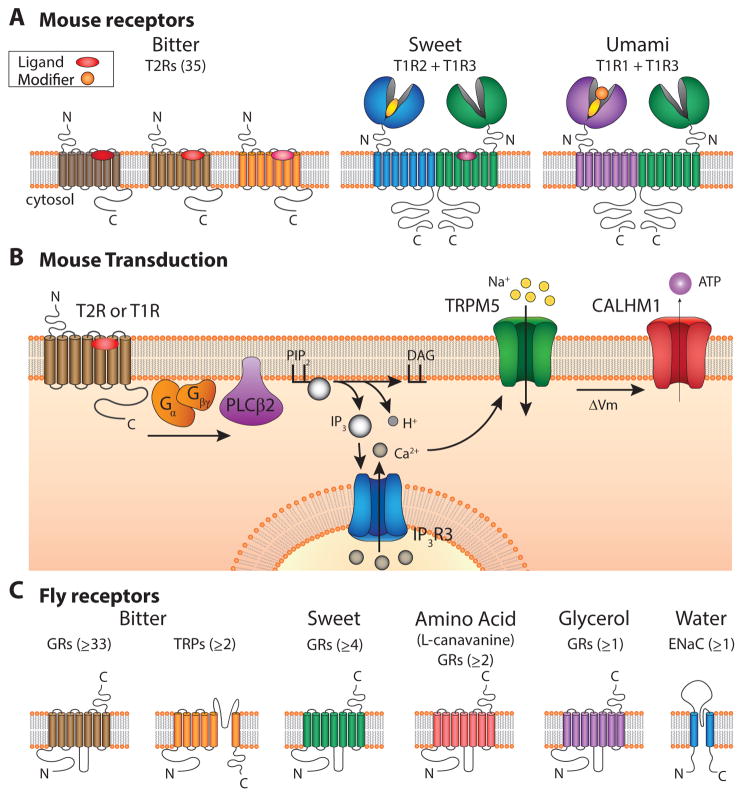
Figure 2.
Taste receptors and transduction in the mouse and fly. (A) Transmembrane topology of bitter, sweet and umami receptors in mouse. All are G-protein coupled-receptors. Bitter receptors (35 total in mice) are Class A GPCRs while sweet and umami receptors (two each) are Class C receptors, characterized by a large N terminal domain that forms a Venus flytrap structure. Sweet and umami receptors bind both ligands (ovals) and allosteric modifiers (circles) that can increase potency of the agonist. (B) Transduction of bitter, sweet and umami in the vertebrate is mediated by a canonical PLC-signaling cascade, that culminates in the opening of the TRPM5 ion channel. This produces a depolarization that may allow CALMH1 channels to open and release ATP, which serves as a neurotransmitter. (C) Drosophila taste receptors that function in bitter, sweet, amino acid (L-canavanine), glycerol and water detection. The minimum number of receptors are indicated.
Bitter taste receptors
Bitter taste has evolved to allow animals to detect toxins in the environment that are primarily produced by plants. Consequently, a large number of structurally diverse chemicals taste bitter to humans and mice, including caffeine, cycloheximide (a protein synthesis inhibitor), denatonium (added to rubbing alcohol to discourage consumption), and quinine (a component of tonic water). But not all bitter-tasting compounds are toxic, as some plants have subverted this relationship to their advantage to produce bitter tasting compounds that are harmless - Brussels sprouts and cocoa beans among them. Indeed it has been stated that starting from the knowledge that a chemical is bitter provides no clue as to whether it is nutritious or dangerous (Glendinning, 1994).
In vertebrates, bitter chemicals are detected by a small family of receptors (T2Rs), which are structurally related to rhodopsin, and range in number from 3–49, depending on the species (Chandrashekar et al., 2000; Matsunami et al., 2000; Shi and Zhang, 2006). T2Rs are required for bitter taste, as a knockout of a single bitter receptor (e.g. mT2R5) eliminates behavioral and nerve responses to receptor agonists (cycloheximide for mT2R5) at concentrations that evoke strong responses in wild-type animals (Mueller et al., 2005). In general, each bitter responsive taste receptor cell expresses multiple types of bitter receptors (Mueller et al., 2005), such that bitter chemicals cannot be readily distinguished by taste alone. However, not all bitter receptors are expressed by every bitter cell (Voigt et al., 2012), leading formally to the possibility that there are subclasses of bitter cells, as is the case in flies (Weiss et al., 2011).
Mapping the “chemical receptive field” of the bitter receptors has shown that they fall into two classes - “specialists” that detect one or a few bitter chemicals and “generalists” that detect many (Behrens and Meyerhof, 2009). Not surprisingly, receptors that detect many ligands, such as T2R10, do so at the expense of sensitivity. Mutations in the receptor that increase affinity for one agonist, without exception, decrease affinity for others (Born et al., 2013). Thus, evolution has balanced sensitivity with specificity. Adding to the complexity of signaling is the recent observation that some compounds, such as naturally occurring sesquiterpene lactones from plants, can function as both agonists for one set of bitter receptors and antagonists for others. These competing actions may produce responses to complex mixtures of foods that are not the sum of their respective components (Brockhoff et al., 2013).
Sweet and amino acid taste
In contrast to bitter taste, which is used to detect a large repertoire of structurally diverse compounds, sweet and umami are evoked by a relatively small number of molecules that signal either calorie-rich (sweet) or protein-rich foods (umami). Accordingly, sweet and umami are each primarily sensed by a single type of GPCR, a heterodimer of a common subunit (T1R3) and a unique subunit – T1R2 and T1R1, for sweet and umami, respectively (Yarmolinsky et al., 2009) (Figure 2A).
Sweet receptors
Sweet taste is elicited by high concentrations of sugars (100–500 mM), by artificial sweeteners, and by a small number of sweet-tasting proteins. This response profile is fully recapitulated in heterologous cells by co-expression of T1R2 and T1R3, providing strong evidence that the heterodimer constitutes the sweet receptor (Nelson et al., 2001). Moreover, a single knockout of either T1R2 or T1R3 eliminates all behavioral preference for artificial sweeteners (Damak et al., 2003; Zhao et al., 2003). However, it appears that T1R2 is not required for the responses to high concentrations of sucrose and glucose, which are retained in the T1R2 knockout. One possibility is that sensitivity to sugars is maintained by the T1R3 subunit, which can be activated in heterologous cells by high concentrations of sweeteners.
T1Rs are class C GPCRs, which include large N-termini that bind ligands and form structures that resembles a “Venus flytrap” (Figure 2A). This domain is connected to the transmembrane segments by a cysteine-rich domain that couples ligand binding to receptor activation. Somewhat unexpectedly, different sweeteners target distinct domains or different subunits of the T1R2/T1R3 receptor. Natural sweeteners, as well as some artificial sweeteners (e.g. aspartame) bind in the Venus flytrap domain of T1R2. Other artificial sweeteners, such as cyclamate, and the sweet receptor blocker lactisole target the transmembrane segments of T1R3, while sweet proteins target the cysteine-rich domains (Cui et al., 2006; Temussi, 2011) (Figure 2A). These observations raise the possibility that sensory qualities of different sweeteners might be attributed to differences in receptor kinetics or even differences in downstream signaling as a consequence of binding to different sites. The identification of structural determinants for receptor activation has also led to the generation of a new class of sweeteners that target allosteric regulatory sites on the receptor (Servant et al., 2010), in much the same way that nucleotides enhance activity of the umami receptor (see below). These modulators have the potential to more faithfully mimic the sensory qualities of natural sweeteners.
The identification of sweet receptors has also provided the tools to solve one of the mysteries of taste – the ability of miracle fruit (Synepalum dulcificum) to change sour taste to sweet – lemon to lemonade. At neutral pH the active component of the berry, the protein miraculin, binds the T1R2/T1R3 receptor with high affinity, but it does not activate the receptor. A switch to acid pH (4.8–6.5) causes the bound ligand to become a strong agonist, eliciting a sweet sensation (Koizumi et al., 2013). Interestingly, only humans and old world monkeys taste miraculin, at any pH, as sweet, due to a structural determinant in the amino terminal region of T1R2 found only in these species (Koizumi et al., 2013).
In addition to T1R2/T1R3, it has also been proposed that animals sense sugars and other energy-rich foods through a mechanism similar to that used by pancreatic β-cells to detect blood glucose (Yee et al., 2011). According to this hypothesis, the metabolism of sugars by sweet cells produces ATP, which closes ATP-sensitive K+ channels leading to membrane depolarization (Yee et al., 2011). That there is an element of redundancy in the system for detecting nutrients should not be entirely surprising, given the essential nature of sugar metabolism for animal survival.
Amino acid (umami) taste receptors
Umami is the sensation elicited by glutamate, which makes foods taste more “delicious” (for which it is named), without changing the perceived taste. In humans, umami is only elicited by glutamate, while mice are sensitive to a wider range of L-amino acids (Yarmolinsky et al., 2009). In either animal, addition of the nucleotides IMP or GMP potentiates the response, which serves as a hallmark of umami taste, distinguishing it from a more general sensing of glutamate (Yamaguchi, 1970).
T1R1/T1R3 is widely recognized as the umami receptor because, in addition to responding to glutamate, it recapitulates all features of the umami response, including sensitivity to 5’ ribonucleotides, and species differences in tuning (Yarmolinsky et al., 2009). 5’ ribonucleotides are not agonists of the receptor, but rather bind and stabilize the receptor in the glutamate-bound state (Zhang et al., 2008) (Figure 2A). Disruption of T1R1 eliminates nucleotide enhancement of the nerve response to MSG (Kusuhara et al., 2013; Zhao et al., 2003). But the T1R1 knockout does not entirely eliminate taste sensitivity to glutamate, suggesting that there may also be a contribution to amino acid taste from other glutamate receptors in the tongue (Chaudhari et al., 2000; Kusuhara et al., 2013).
Evolution of vertebrate taste receptors and individual differences in taste
Much of the variability in taste across different species and among individuals within a species can be attributed to differences in taste receptor genes. One striking example is the well-known indifference of cats to sweet. In modern cats, the T1R2 (sweet receptor) gene is a pseudogene, having acquired during evolution multiple inactivating mutations (Rohács et al., 2005). Another example is the variation within the human population in sensitivity to phenylthiocarbamide (PTC), which evokes either an intense bitter taste or is tasteless. PTC is detected by the receptor T2R38 for which there are two predominate alleles, one that generates a PTC–sensitive receptor found in individuals who can taste PTC and the other which generates a PTC-insensitive receptor found in nontasters (Kim et al., 2003). Why the nontaster allele has been maintained is a mystery - possibly it is a functional bitter receptor, detecting a still unknown agonist.
Comparison of receptors for bitter, sweet and umami over a wide range of organisms has shown that changes in receptor function and number are not limited to a few examples but are quite common (Shi and Zhang, 2006). In general, receptors tend to be lost when they are not used to make dietary choices, while an increase in receptor number (for bitter receptors) coincides with an expansion in diet. For example, “pseudogenization” of the sugar receptor, T1R2, occurred in the evolution of many carnivores, not just cats, including the spotted hyena and some otters (Jiang et al., 2012). Conversely, the T1R1 subunit, which is a unique component of the umami receptor, is a pseudogene in the giant panda, which feeds exclusively on bamboo (Zhao et al., 2010). In some animals, including the sea lion and dolphin, all three receptors for sweet and umami taste are pseudogenized, a consequence presumably of a feeding pattern in which foods are swallowed whole (Jiang et al., 2012). This variation in receptor numbers is much more dramatic in insects, which must adapt to environments with widely divergent plant fauna, and which use pheromones extensively for modulating social behavior (see below).
Bitter, Sweet, Umami Transduction
Despite the divergent structures of the receptors for sugars, bitter chemicals and amino acids, they couple to elements of a common signaling pathway (Zhang et al., 2003). All employ a canonical phosphoinositide-based pathway, in which receptors activate a taste cell-specific G protein that activates PLCβ2, generating second messengers IP3, DAG and H+. IP3 acts on the IP3 receptor (IP3R3) to release Ca2+ from intracellular stores, and Ca2+ gates the membrane channel TRPM5 (Figure 2B). Support for this model comes from the observations that animals carrying inactivating mutations of PLC β2, IP3R3 and TRPM5 show severely diminished behavioral responses to bitter, sweet and umami (Damak et al., 2006; Tordoff and Ellis, 2013; Zhang et al., 2003).
A central element of the signal transduction cascade is TRPM5 (Perez et al., 2002; Zhang et al., 2003), a member of the TRP family ion channels (Venkatachalam and Montell, 2007). TRPM5 channels in heterologous expression systems (Hofmann et al., 2003; Liu and Liman, 2003; Prawitt et al., 2003) and in native taste cells (Zhang et al., 2007) are activated by intracellular Ca2+ and are permeable to monovalent but not divalent cations. These biophysical properties allow the channel to transduce the elevation of intracellular Ca2+ that results from receptor signaling into a change in membrane potential.
How a change in membrane potential regulates transmitter release in TRPM5-expressing cells, which lack voltage-gated Ca2+ channels and machinery for vesicular release, is a problem that has nagged at the taste field. ATP is a likely transmitter, based on a number of criteria, including the taste-blind phenotype of a P2X2/P2X3 double knockout mouse (Finger et al., 2005). There is pharmacological, but no genetic evidence that ATP is released through pannexin hemichannels (Huang et al., 2007; Murata et al., 2010; Romanov et al., 2007). An alternative is that ATP is released by CALMH1 (Taruno et al., 2013), an intriguing new type of ion channel that was originally identified as a modulator of Ca2+ signaling (Figure 2B) (Dreses-Werringloer et al., 2008). This channel is highly enriched in TRPM5-expressing cells, and is sufficient to mediate ATP release in heterologous cells. Moreover, CALMH1 knockout animals have severely diminished abilities to taste bitter, sweet and umami (Taruno et al., 2013). Whether CALMH1 acts alone or in combination with pannexin channels remains to be determined.
Drosophila bitter, sweet and amino acid taste
Insects, such as fruit flies and mosquitoes detect a repertoire of taste qualities similar to humans. However, the sensation occurs primarily through receptors that bear no sequence relationship to mammalian taste receptors. The majority of bitter and sweet taste receptors in flies, mosquitoes and many other insects are members of a large protein superfamily, called gustatory receptors (GRs), which in Drosophila contains 68 members (Clyne et al., 2000; Dunipace et al., 2001; Robertson et al., 2003; Scott et al., 2001). These proteins have seven transmembrane domains, but they share no sequence relationship to GPCRs. Rather, they are distantly related to Drosophila olfactory receptors (ORs), which have an opposite membrane topology from GPCRs and form ligand-gated ion channels (Benton et al., 2006; Sato et al., 2008; Wicher et al., 2008). Similarly, insect GRs have an inverted topology relative to GPCRs (Xu et al., 2012; Zhang et al., 2011) and may form ionotropic receptors (Sato et al., 2011).
Bitter taste through Gustatory Receptors
In flies, different sets of bitter sensitive GRNs have distinct sensitivities. On the basis of their responsiveness to a panel of 16 bitter compounds, the L-, I and S-type sensilla that decorate the labella are classified into five groups, four of which are sensitive to bitter chemicals (Weiss et al., 2011). Of the four, two groups are narrowly tuned to distinct sets of bitter compounds (I-a and I-b), while the other two groups respond broadly to bitter tastants, but vary in their patterns of activity (S-a, S-b). It remains possible that using a larger panel of bitter compounds, future analyses will reveal yet additional subgroups.
The spatial distribution of Grs has been studied using transgenic flies harboring gene reporters, which show that at least 38 Gr genes are expressed in the labellum, most of which (33) are localized to bitter GRNs (Weiss et al., 2011). Gratifyingly, the expression of these 33 Gr genes fall into four general groups that appear to correspond to the four functional sets of bitter neurons. Moreover, as might be expected, the two sets of GRNs that are broadly tuned (S-a, S-b) express many Grs, while the two narrowly tuned sets of GRNs (I-a and I-b) express fewer Grs. The larval taste organs in the head express a minimum of 39 Grs (Colomb et al., 2007; Kwon et al., 2011), which are found either in the tip of the head only (15 Grs), 2), the pharyngeal taste organs only (11 Grs) or in both locations (13 Grs) (Kwon et al., 2011). Most of these receptors are presumed to be bitter receptors.
The roles of only a small handful of the bitter GRs have been dissected genetically using gene knockouts. Nevertheless, a few principles have emerged from the limited functional analyses of bitter taste in the adult fly. First, the repertoire of GRs that contribute to the detection of a bitter compound is large and the requirement for individual subunits is complex. In a survey of five bitter Grs, mutations in any of three impaired caffeine sensing (Gr33a, Gr66a and Gr93a) (Lee et al., 2010; Lee et al., 2009; Moon et al., 2006; Moon et al., 2009), while in a screen of six Grs for roles in sensing DEET, single mutations in any one of three disrupted avoidance of this insect repellent (Lee et al., 2010). However, the combination of all of the GRs that are currently known to be required for responding to any given aversive compound is not sufficient to confer sensitivity to the compound either in a heterologous expression system or after expressing the GRs in vivo in sugar-responsive GRNs. This points to the possibility that a large number of receptor subunits (>3) comprise the functional bitter or DEET receptors. Consistent with this proposal, some GRNs in the labellum express a minimum of 28 Grs, while one of the larval GRN classes expresses at least 17 Grs (Kwon et al., 2011; Weiss et al., 2011).
A second general observation is that some GRs are required for responding to large numbers of aversive chemicals and may act as co-receptors. Gr32a, GrR33a and Gr66a are needed for detection of most bitter chemicals (Lee et al., 2010; Moon et al., 2009). These three Grs, as well as two additional Grs (Gr89a and Gr39a.a) are expressed in all bitter responsive GRNs. Thus, this collection of five GRs has been suggested to be the “core-bitter GRs” (Weiss et al., 2011). Core bitter GRs might function in the bitter response as obligatory co-receptors, analogous to ORCO in the olfactory response (Benton et al., 2006). In larvae, GR33a and GR66a may also be core-bitter GRs since they are the Grs that are the most widely expressed among the larval GRNs (Kwon et al., 2011).
While some GRs contribute broadly to bitter taste detection, other GRs are very narrowly tuned and confer ligand specificity. Examples are Gr8a and Gr93a, which are needed for sensing L-canavanine and caffeine respectively, but are dispensable for all other aversive compounds tested (Lee et al., 2012; Lee et al., 2009). Narrowly tuned GRs might be critical in defining the chemical specificity of the GRs, in combination with other GRs. Different combinations of complex sets of GR receptors may explain how a limited number of bitter GRs confer the capacity to respond to a vast collection of structurally diverse bitter compounds. Thus, in contrast to vertebrate bitter detection, which is mediated by receptors that act largely as homomultimers (Kuhn et al., 2010) flies employ a much more complex strategy to sample foods for bitter chemicals.
Bitter taste, TRP channels and taste plasticity
The responsiveness of the labellum to bitter compounds is not limited to GRs. At least three TRP channels are expressed in the labellum and contribute to the sensation of aversive compounds, most likely through mechanisms that are independent of GRs. One of these channels, TRPA1, is expressed in a subset of bitter GRNs in sensilla on the labellum, and is required for the generation of action potentials and behavioral avoidance to aristolochic acid, but not other bitter chemicals tested (Kim et al., 2010). A contribution of TRPA1 to taste has also been suggested in the caterpillar of the moth, Manduca sexta, where electrophysiological responses of GRNs to aristolochic acid increases at higher temperatures (e.g. 22° versus 30°C) (Afroz et al., 2013). The temperature dependence is consistent with the observation that TRPA1 is both directly and indirectly activated by changes in temperature in Drosophila and other insects such as Anopheles gambiae (Kang et al., 2012; Kwon et al., 2008; Viswanath et al., 2003; Wang et al., 2009; Zhong et al., 2012). The taste responses in the moth to other chemicals that are not mediated by TRPA1, such as sugars, salts and quinine, are not temperature dependent (Afroz et al., 2013).
A related TRPA channel, Painless, is also expressed in GRNs in the labellum and is required for the behavioral avoidance to isothiocyanates (AITC; wasabi) (Al-Anzi et al., 2006). However, it remains unknown if AITC-induced action potentials are affected in painless mutants, leaving open the question of whether Painless is a direct sensor of AITC or serves some other function in GRNs, such as in synaptic transmission.
Another TRP channel member, TRP-Like (TRPL) is also expressed in GRNs, and is both necessary and sufficient to confer sensitivity to camphor (Zhang et al., 2013b). TRPL does not respond to other aversive compounds tested, making it narrowly tuned like TRPA1. Camphor is not harmful to insects and the aversion it elicits may be attributed to trickery on the part of plants to avoid consumption. In turn, flies can adapt to this formerly aversive but nontoxic food, and long-term exposure to a camphor diet greatly increases the fly’s acceptance of camphor-containing foods. This decrease in aversion to camphor occurs through Ube3a-dependent ubiquitination of TRPL, and degradation of the channel (Zhang et al., 2013b). Following a decline in the TRPL protein in the dendrites, there is a moderate elimination of synaptic boutons in the axonal terminals of trpl GRNs in the SOG region of the brain. Thus, a combination of these two events appears to underlie the taste plasticity. Once the flies are returned long-term to a camphor-free diet, the original concentration of the TRPL protein and synaptic boutons returns, and the fly’s aversion to camphor is restored. Thus, reversible changes in the GRNs can form the basis through which an animal adapts to a dynamic food environment.
Sweet receptors
Flies are attracted to many of the same sugars as humans (Gordesky-Gold et al., 2008; Hiroi et al., 2002), although they respond most robustly to disaccharides (such as sucrose and maltose) and oligosaccharides (Dahanukar et al., 2007). The fly sweet receptors belong to the same superfamily of receptors that includes most of the bitter receptors, the GRs. In adult flies the three key receptors required for sensing sugars, except for fructose, are GR5a, GR64a and GR64f (Dahanukar et al., 2001; Dahanukar et al., 2007; Jiao et al., 2007; Jiao et al., 2008; Slone et al., 2007). These three receptors are co-expressed in the sugar-responsive GRNs in the labellum, along with five other related GRs that comprise the Gr-Sugar (Gr-S) clade (Dahanukar et al., 2007; Jiao et al., 2007).
GR5a and GR64a sense structurally different sugars; GR64a participates in the response to sucrose and maltose (Dahanukar et al., 2007; Jiao et al., 2007), while GR5a is needed for detection of trehalose and melezitose (Dahanukar et al., 2001; Dahanukar et al., 2007; Ueno et al., 2001). In addition, GR64f might be a co-receptor since with the exception of fructose, it is required for the responses for all sugars tested, and functions in concert with GR5a and GR64a (Jiao et al., 2008). However, expression of GR64f in combination with either GR64a or GR5a is not sufficient to confer a sugar response to bitter responsive GRNs or tissue culture cells, indicating that additional subunits may be required.
Unlike bitter compounds and most sugars, which are detected by a complex set of GRs, a single GR—GR43a has been reported to detect fructose. Genetic studies demonstrate that Gr43a is required for responding specifically to fructose (Miyamoto et al., 2012). In addition, in vitro expression studies show that the silkworm homolog of GR43a (BmGR-9) is a cation channel that is directly activated by fructose but not other sugars (Sato et al., 2011). Drosophila GR43a also appears to be a fructose-activated channel (Sato et al., 2011), although its substrate specificity remains to be established.
Drosophila larvae sense a similar array of sugars as adult flies (Miyakawa, 1982; Schipanski et al., 2008). However, the genes encoding either the main sugar-sensitive GRs in the adult labellum or the related Gr-S genes are not detected in larval GRNs (Colomb et al., 2007; Kwon et al., 2011). Instead, the fructose receptor, Gr43a, is essential for detecting multiple sugars (Mishra et al., 2013). This is surprising given the narrow response specificity of the silkworm homolog of GR43a (BmGR-9) (Sato et al., 2011). Gr43a is expressed in two locations - in pharyngeal GRNs and in the brain - where two separate mechanisms, acting over different time periods, explain the ability of GR43a to mediate responses to multiple sugars. (Mishra et al., 2013). The more rapid phase, which occurs during the first 2 minutes following contact with a potential food source, is sensitive to fructose and sucrose (a glucose-fructose disaccharide), and might be mediated by GR43a in the pharynx. The slower phase takes ~16 minutes to develop, and is sensitive to most other sugars. This latter response might be delayed due to the additional time necessary for metabolism of the sugars to fructose, and transport of fructose to the brain, where it is detected by GR43a (Mishra et al., 2013).
Suppression of sweet taste by bitter compounds
Many foods are comprised of a combination of sugars and bitter compounds, which stimulate opposing behavior responses that need to be reconciled. Bitter compounds suppress feeding, not just by activating bitter-responsive GRNs, but also by inhibiting sugar-sensitive GRNs (Meunier et al., 2003). The suppression of sugar GRNs depends on a member of the family of “odorant binding proteins” (OBP), OBP49a, which is expressed in gustatory organs (Jeong et al., 2013). OBP49a is synthesized in accessory cells, released into endolymph fluid bathing the GRNs, and then acts non-cell autonomously on sugar activated GRNs. OBP49a binds directly to bitter compounds, and then interacts with the sugar receptor, GR64a, on the cell surface of the GRNs to suppress its activity (Jeong et al., 2013). This non-cell autonomous mechanism for suppression of the sugar response by bitter compounds provides a strategy for ensuring that bitter compounds in sugar-laden foods are not consumed.
Amino acid taste in flies
Fruit flies taste amino acids, although their predilection is enhanced if they are raised on a food source devoid of amino acids. (Toshima and Tanimura, 2012). In females, the preference is greatest for cysteine, phenylalanine, threonine and tyrosine, while males prefer leucine and histidine. However, none of 18 standard amino acids tested stimulates action potentials in GRNs in sugar responsive sensilla (Dahanukar et al., 2007), raising the possibility that taste pegs may sense amino acids. Another amino acid, L-canavanine, which is toxic because it is incorporated into proteins in place of L-arginine, elicits an avoidance response in flies (Mitri et al., 2009) and is sensed by GRNs in a subset of S-type sensilla (Lee et al., 2012). Although taste receptors for standard amino acids are unknown, GR8a and GR66a are both required for L-canavanine avoidance (Lee et al., 2012).
Drosophila sweet, bitter and amino acid transduction
Activation of Drosophila GRNs by sugars, bitter compounds and the amino acid, L-canavanine, appears to occur both through direct activation of ion channels, as well as through G-protein signaling pathways. Multiple signaling pathways are also found in other insects, such as Manduca sexta (Glendinning et al., 2002). A direct activation mechanism may be employed by GRs, as at least one GR (the fructose receptor) appears to be a cation channel (Sato et al., 2011). However, it is not known if other GRs are ionotropic receptors.
Several different G-protein subunits are implicated in sugar signaling, including Gγ, Goα, Gsα and Gqα (Bredendiek et al., 2011; Ishimoto et al., 2005; Kain et al., 2010; Ueno et al., 2006). The effector for Gqα is a PLCβ, and mutation or knockdown in sugar-responsive GRNs of plcβ21c or any of the genes encoding TRPC channels (TRP, TRPL and TRPγ) impairs the behavioral response to trehalose (Kain et al., 2010). However, these studies did not address whether these proteins functioned in GRNs. The effector for Gsα is adenylyl cyclase, and this protein might function in taste transduction in GRNs since RNAi knockdown of AC78C reduces trehalose and sucrose induced action potentials, at low but not high sugar concentrations (Ueno and Kidokoro, 2008).
G-protein coupled signaling pathways may also contribute to the sensation of bitter tastants. AC78C is needed for the response to caffeine (Ueno and Kidokoro, 2008), and the PLCβ encoded by norpA is required in trpA1-expressing GRNs for the behavioral and electrophysiological responses to the bitter compound, aristolochic acid (Kim et al., 2010). These latter results suggest that a Gq/PLC/TRPA1 pathway functions in the detection of aristolochic acid. Another G-protein, Goα47A is necessary for detection of L-canavanine (Devambez et al., 2013).
The G-protein coupled signaling pathways that function in insect taste could serve to enhance the responses to low concentrations of ligands, similar to the function of a phototransduction cascade in amplifying the response to a photon of light. At least in one case, the GPCR signaling appears to be coupled to a TRP channel (TRPA1) (Kim et al., 2010). However, an open question is whether GRs are GPCRs, in addition to ionotropic channels. While this seems unlikely in view of the inverse topology of GRs relative to classical GPCRs (Xu et al., 2012; Zhang et al., 2011), there is controversial evidence that the distantly related ORs might serve as both GPCRs and ionotropic receptors (Nakagawa and Vosshall, 2009; Wicher et al., 2008). Nevertheless, an alternative possibility is that the GPCR signaling pathways in GRNs function in parallel to GRs, or might modulate the activity of GRs through phosphorylation or other regulatory mechanisms.
Diversity in sizes of arthropod GR families
The Dipteran GRs are unrelated to mammalian taste receptors, but share a common ancestor with the insect ORs. Drosophila melanogaster encodes 68 GRs, which is similar to size of the 62–member Drosophila OR family (Robertson et al., 2003). Due to the critical roles of GRs in sensing and adapting to highly variable chemical environments, and in mate selection, the GRs are exceedingly divergent among insects. In fact, only a limited number of GRs have recognizable homologs between different insect species. In some insects, the size of the GR family is relatively large, such as in the mosquito disease vectors, Aedes aegypti and Anopheles gambiae, which include 114 and 90 respectively (Hill et al., 2002; Kent et al., 2008). Conversely, the genome of the honey bee, Apis mellifera, encodes only 10 GRs, as compared to 163 ORs (Robertson and Wanner, 2006; Zhou et al., 2012). This expansion of the ORs likely reflects the importance of olfaction in sensing a wide botanical repertoire and a complexity of volatile pheromones, while the limited number of GRs might be sufficient since bees feed primarily on nectar and pollen. The genomes of ant species such as Camponotus floridanus, Harpegnathos saltator and the Argentine ant, Linepithema humile encode the largest numbers of ORs among insects whose genomes have been sequenced (~350 ORs), but fewer and more variable numbers of GRs (17–97) (Smith et al., 2011; Zhou et al., 2012). These variations in the sizes of the ant GR families might be a consequence of differences in feeding behaviors and the number of pheromones detected by contact chemosensation.
Examination of the distribution of the GRs and ORs among distantly related organisms suggests that that the GRs may be the more ancient chemosensory family. The waterflea, Daphnia pulex, encodes 58 GRs, but is devoid of ORs (Peñalva-Arana et al., 2009). While it is possible that the entire OR family was lost in this crustacean, a more likely scenario is that the GRs are more ancient, and that the ORs emerged with the terrestrial arthropods, such as insects (Peñalva-Arana et al., 2009). In further support of the proposal that the GRs predated ORs, the worm, C. elegans, encodes some GRs, such as LITE-1, but no OR family members (Liu et al., 2010; Robertson et al., 2003). The family of ionotropic receptors IRs, which are distantly to ionotropic glutamate receptors (Benton et al., 2009), may be at least as old as GRs as IRs are encoded in C. elegans (Croset et al., 2010). The original role of IRs may be detection of non-volatile compounds, since IRs are expressed in pharyngeal neurons in worms and flies, and their last common ancestor was probably an aquatic organism (Croset et al., 2010).
Sour and salty
Sour and salty are considered “mineral” tastes, as the effective stimulus is a simple element/ion. Discerning the mechanism of transduction has been more difficult than for the “organic” tastes, partly because the stimulus is so simple and is always present. Sour taste is evoked by acidic pH and by organic acids, such as acetic acid, that penetrate the cell membrane. Salty taste is elicited by Na+ concentrations from 10 mM to 500 mM, and consists of at least two components, with different distributions and pharmacological properties.
Sour taste in mammals
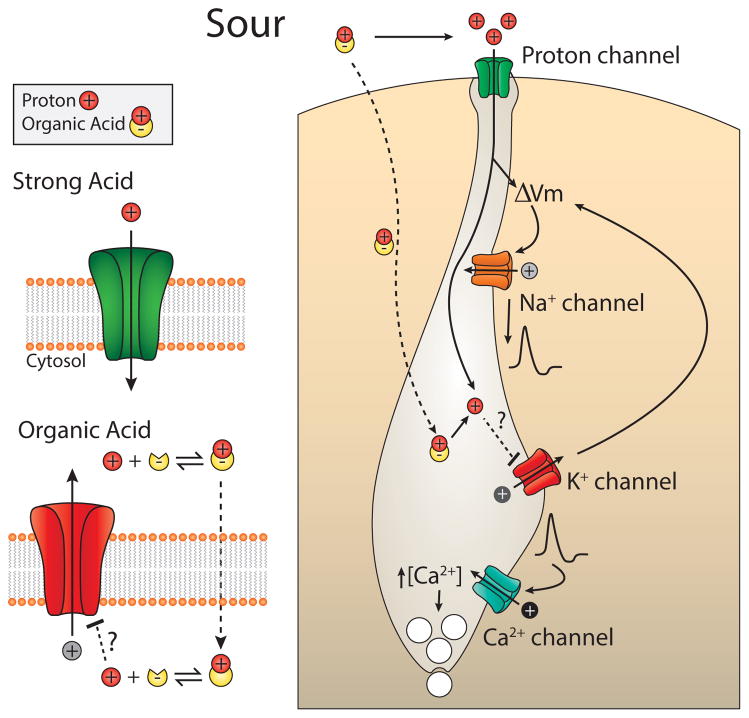
Sour is detected by a subset of taste receptor cells in the tongue and palate epithelium that respond to acidic pH and weak organic acids with electrical activity (Huang et al., 2006; Huang et al., 2008). Over the years, a number of candidates for sour receptors or components of sour signaling have been proposed, including ASICs, HCNs, K+ channels, and most recently the TRP channels, PKD2L1 and PKD1L3 (Roper, 2007). However, presently there is no evidence that any of these proteins mediate sour taste, and knockouts of mouse PKD2L1 or PKD1L3 only slightly attenuate nerve responses to acid stimulation (Horio et al., 2011). Nonetheless, PKD2L1-expressing cells respond to, and are required for, sensory response to acids (Chang et al., 2010; Huang et al., 2006; Oka et al., 2013). The response of PKD2L1-expressing cells to acids is mediated by an unusual proton-selective ion channel (Chang et al., 2010). Proton selectivity allows the cells to respond to acids without interference from Na+, which may vary independently in concentration. The molecular identity of the proton channel is presently unknown.
Entry of protons into sour cells produces cellular acidification, which may affect cell signaling. Notably, several resting two-pore K+ channels are expressed in taste cells (Lin et al., 2004; Richter et al., 2004), which may be blocked by intracellular acidification to produce further depolarization of the cell (Figure 3). The idea that intracellular acidification activates sour cells is attractive, as it could explain why, at the same pH, acetic acid and other weak acids that penetrate the cell membrane taste more sour than strong acids, such as HCl, that do not penetrate the cell membrane (Lyall et al., 2001; Roper, 2007).
Figure 3.
Sour taste. Sour taste in vertebrates is initiated when protons enter through an apically located proton-selective ion channel. Weak acids may also activate sour cells by penetrating the cell membrane and acidifying the cytosol, leading to closure of resting K+ channels and membrane depolarization.
In addition to sour stimuli, PKD2L1-expressing cells are required for the gustatory response to carbonation (CO2). This response is dependent on a membrane anchored carbonic anhydrase isoform 4, Car4 (Chandrashekar et al., 2009), which interconverts CO2 + H2O to H+ + HCO3−. The mechanism by which Car4 contributes to the activation of sour cells is not known. One possibility is that protons generated apically by this enzymatic reaction enter through the proton channel to depolarize the cell.
It should be noted that the response to acids and carbonation is complicated by the fact that the trigeminal system, which heavily innervates the mouth and oral cavity, is also sensitive to these stimuli (Bryant and Silver, 2000). TRPA1 is expressed by nociceptors and can be activated by CO2 and acetic acid (Wang et al., 2010), and the capsaicin receptor, TRPV1, is activated by extracellular protons (Tominaga et al., 1998). Moreover, afferent nerve fibers that innervate the oral cavity retain sensitivity to acids, in otherwise taste-blind mice (Ohkuri et al., 2012). Thus, somatosensory afferents undoubtedly contribute to the burning sensation experienced when ingesting sodas and organic acids.
Taste of sour and carbonation in Drosophila
Fruit flies prefer slightly acidic foods, such as carbonated water, while they reject foods that are too acidic. Carbonated water triggers Ca2+ influx in the region of the SOG innervated by taste peg GRNs, suggesting these neurons are involved in CO2 detection (Fischler et al., 2007). Fruit flies avoid many carboxylic acids with a low pH. Behavioral and physiological analysis reveals that the avoidance to carboxylic acid is mainly mediated by a subset of bitter GRNs (Charlu et al., 2013). In addition, acids also inhibit the activity of sugar GRNs. However, the molecular identities of the taste sensors for both weak carboxylic acids and strong metallic acids are unknown. Nevertheless, since only a subset of bitter GRNs sense acids, this localized expression pattern may contributed to the discrimination of sour versus bitter tastants (Charlu et al., 2013).
Salt taste receptors
The taste of salt is complex from two perspectives. First, while we frequently think of salt taste as the sensation of Na+, other cations such as Li+ or K+ may also be perceived as salty, although less potently than Na+. Second, salt can be attractive or aversive depending on concentration, with lower concentrations (<100 mM) being attractive. This dichotomy reflects the fact that moderate levels of salt are necessary to maintain muscle contraction, action potentials and many other functions while excessive salt intake is deleterious and in humans can lead to hypertension.
Salt taste in mammals
Two distinct mechanisms underlie cellular sensitivity to salt tast in mice and other rodents, one that is sensitive to the diuretic amiloride (Brand et al., 1985; Heck et al., 1984) and another that is insensitive. In humans, salt taste is amiloride-insensitive, reflecting either species-differences in receptor pharmacology or loss of the amiloride-sensitive component. The amiloride sensitive component of salt taste is selective for Na+ and Li+ over other monovalent cations such as K+, is sensitive to low concentrations of salts (<100 mM), and is generally appetitive (Brand et al., 1985). Amiloride-sensitive salt taste occurs only in the front of the tongue (Ninomiya, 1998).
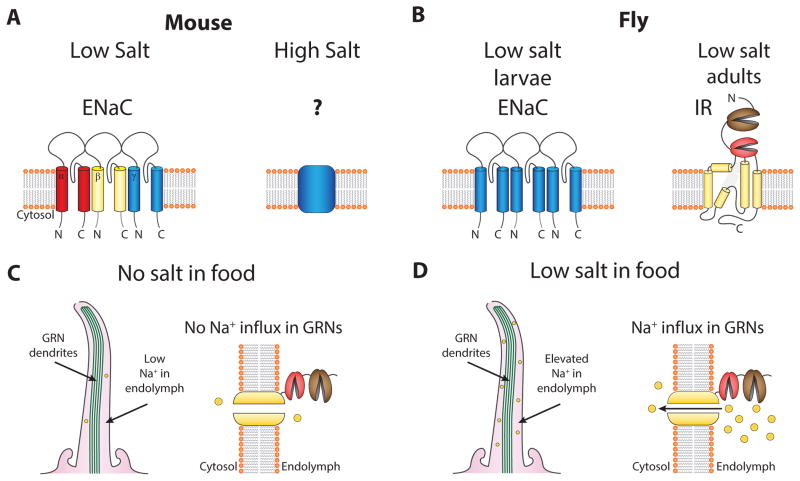
Given the sensitivity of low salt taste to amiloride, which is a relatively specific blocker of epithelial Na+ channels (ENaCs), these channels were considered candidate low salt receptors (Kretz et al., 1999). ENaC is composed of three subunits – α, β and γ, of which the α subunit is absolutely essential and forms part of the pore (Figure 4A) (Canessa et al., 1994). Indeed, ENaC α appears to be a component of the low salt sensor since a taste-cell specific knockout eliminates sensitivity and behavioral attraction to low concentrations of salt (Chandrashekar et al., 2010).
Figure 4.
Salt taste. (A) The mouse low salt sensor is a protypical ENaC channel composed of three subunits. The high salt sensor in TRPM5 or PKD2L1-expressing taste cells is not known. (B) The salt sensors in fly larvae. (C) No Na+ influx through IR76b when adult flies are not exposed to salt containing food, since the Na+ concentration in the endolymph is low. (D) The concentration of Na+ concentration in the endolymph rises when adult flies are exposed to salt containing food, leading to an influx of Na+ through constitutively open IR76b and activation of the GRN.
Identification of the “salty cells” has been more difficult since subunits of ENaC are detected in a wide range of cell types, including PKD2L1-expressing and TRPM5-expressing cells. The observation that only a small fraction of taste cells express all three subunits of the ENaC channel, and that these cells do not express markers for other taste qualities suggests that these “ENaC-alone” cells constitute the sensory cells for amiloride-sensitive salt taste (Chandrashekar et al., 2010).
The cellular basis for sensitivity to high concentrations of salts is comparatively more complicated. Based on taste nerve recordings, there is a population of broadly tuned high-salt fibers that are insensitive to amiloride and activated by KCl and NaCl (Breza and Contreras, 2011). These fibers innervate both the front and back of the tongue, in contrast to the amiloride-sensitive fibers that innervate only the front of the tongue. Currently, the identity of the high salt receptor remains a mystery, as the early proposals that TRPV1 was the high salt sensor (Lyall et al., 2004) are not supported (Breza and Contreras, 2011).
The cells that mediate the behavioral responses to high salts are not specifically dedicated to sensing high salt, but instead comprise at least two populations of cells with previously identified functions in sensing bitter and sour (Oka et al., 2013). Inactivation of TRPM5 or PLCβ2, expressed by bitter cells, eliminates a component of the high salt response, while silencing PKD2L1-expressing sour cells eliminates the remaining components (Oka et al., 2013). Remarkably, mice in which PKD2L1-expressing cells are silenced and TRPM5 is inactivated find high salt concentrations attractive, presumably due to activation of the amiloride-sensitive ENaC channels by high salt (Oka et al., 2013). How high salts activate the two types of cells is not entirely clear. High salts could activate bitter cells through an allosteric effect on the receptors, while a requirement for carbonic anhydrase (CA4) in sour cells suggests that the balance between acid and base may be altered by high salts at the apical surface of the cell (Oka et al., 2013).
Salt taste in Drosophila
Salt taste preferences in Drosophila are similar to those in mammals in that larvae and adult fruit flies also prefer low-salt foods, while they reject high-salt foods. In larvae, two ENaC channels family members, ppk11 and ppk19 are reported to be expressed in the terminal organ and required for sensing low salt (Figure 4B) (Liu et al., 2003a). However, these channels do not appear to function in the salt response in adults (Zhang et al., 2013a).
In adult flies, the attraction to low salt and the aversion to high salt occur through a competition between two types of salt-responsive GRNs. At low and moderate salt concentrations (e.g. ≤100 mM NaCl), GRNs in L-type sensillum are much more robustly activated than the salt-activated GRNs in S- and I-type sensilla. Conversely, at high salt conditions (≥500 mM NaCl), GRNs in several S-type sensilla provide the dominant responses. When the activities of the low salt GRNs associated with the L-type sensilla predominant, feeding is stimulated. However, at high salt concentrations, when the GRNs in S-type sensilla predominate, feeding is suppressed. Thus, the winner of the competition between the two antagonistic pathways dictates the net behavioral output.
A member of the ionotropic glutamate receptor (IR) family member, IR76b, is required for low salt sensing in adult flies (Zhang et al., 2013). IRs were identified originally as a new class of olfactory receptor (Benton et al., 2009). However, several IRs are also expressed in GRNs (Croset et al., 2010). IR76b is expressed in GRNs distinct from those that respond to sugars and bitter compounds, and the IR76b GRNs extend their projections into a unique region of the SOG (Zhang et al., 2013).
The IR76b channel is not gated by voltage or salt, but is continuously open. This feature of IR76b is reminiscent of the mammalian low salt channel (ENaC), which is also a Na+ leak channel (Canessa et al., 1994; McDonald et al., 1995). The open states of IR76b and ENaC are well suited for low-salt sensors given the unusually low Na+ compositions bathing the taste cells in both insects and mammals, relative to the insect hemolymph or mammalian blood. As a result, there is little Na+ conductance when the animals are not exposed to salt-containing food (Figure 4C). When the Na+ concentration outside the taste cells rises following intake of salt, the Na+ flux through the IR76b and ENaC channels depolarize the taste receptor cells (Figure 4D). Thus, despite the lack of relatedness between IRs and ENaC channels, flies and mammals appear to solve the challenge of sensing low salt in food through similar strategies employing Na+ leak channels. A major question is the identity of the sensors in flies and mammals that functions in the perception of high salt. Notably, the TMC-1 (transmembrane channel like) was recently reported to be the Na+ channel that controls high-salt avoidance in C. elegans (Chatzigeorgiou et al., 2013), raising the possibility that related TMC channels function in high salt taste in other animals
Noncanonical taste qualities in mammals
Vertebrates can sense a variety of other important qualities in potential foods, such as wetness and fattiness, but whether they qualify as bona fide tastes is still an open question. Fats are detected by several routes, including olfaction and somatosensation, and they elicit post-ingestive effects that promote consumption. But are they tasted? Several lines of evidence argue that the answer is yes. The observation that mice prefer water spiked with free fatty acids, which are breakdown product of triacylglycerides that are found in vegetable and animal-derived products, supports a role for the taste system in detecting this rich source of calories (Gaillard et al., 2008). Taste cells express putative receptors for fat taste including K+ channels that are sensitive to polyunsaturated fatty acids, a fatty acid transporter (CD36) and two fat-sensitive GPCRs—GPR40 and GPR120 (Liu et al., 2011). The most promising of these candidate receptors is GPR120, which is required for preference to fatty acids in mice (Cartoni et al., 2010) and is expressed in human TRCs (Galindo et al., 2011). Most strikingly, the pharmacological profile of GPR120, which is highly sensitive to long chain fatty acids, matches psychophysical data on taste quality from human subjects (Galindo et al., 2011). Because long chain fatty acids evoke a fatty taste only when dissolved in an otherwise tasteless lipid matrix, input from the somatosensory and taste systems must be integrated, either at the level of the taste cell or higher brain centers (Rolls et al., 1999), to produce the complex attractive sensation associated with fatty foods.
Another noncanonical sensory attribute encoded by taste cells is referred to as “calcium taste.” Ca2+, an ion required for a vast array of cellular functions, is attractive to Ca2+-deprived animals, but is rejected by Ca2+-sated animals. Paradoxically, the aversive response to Ca2+requires a functioning T1R3 receptor, a subunit of the umami and sweet receptor (Tordoff et al., 2008). That T1R3 functions as a Ca2+ receptor in vivo, is further supported by the observation that human subjects report an attenuation of the taste of Ca2+ by the T1R3 blocker lactisole (Tordoff et al., 2012). One explanation for these observations is that that T1R3 functions as an aversive Ca2+ sensor in the subset of cells that that express T1R3 alone, but not other subunits of sweet or umami receptors, which are hardwired to attractive behaviors. In this model, the T1R3-only cells may show a high degree of plasticity in how they signal to afferent nerve fibers, activating higher brain centers that cause rejection of Ca2+ depending on the internal state of the organism.
Last among these noncanonical tastes is the taste of water. Animals can detect wetness across their body by virtue of the somatosensory system, and this system is also likely to contribute to the sensing of aqueous solutions in the oral cavity. In addition, various tastes have been ascribed to distilled water, from bitter to salty and sweet. Notably, application of water after exposure to some artificial sweeteners, such as saccharin, elicits a sweet taste (Galindo-Cuspinera et al., 2006). This has been shown to reflect an inhibitory action on the sweet receptor of high concentrations of some artificial sweeteners, which when removed produces a transient re-activation of the receptor, and a sweet taste (Galindo-Cuspinera et al., 2006). While it would seem beneficial for vertebrates to detect water, no water receptor has been identified.
Noncanonical taste qualities in Drosophila
Flies are attracted by the taste of a variety of long- and short-chain fatty acids, except when concentrations are very high. (Masek and Keene, 2013). The appeal of fatty acids is not due to their acidity, and is mediated through sugar-GRNs. Currently, the receptors for fatty acids are not known, but a requirement for PLC in detection of fatty acids suggest that they might be GPCRs (Masek and Keene, 2013). If so, then fatty acid taste may bear greater similarities between flies and mammals than bitter and sweet taste. The same GRNs that are activated by sugars and low concentrations of fatty acids are also activated by glycerol (Wisotsky et al., 2011). Furthermore, a receptor that is required for sensing glycerol, GR64e, belongs to the GR-S clade that includes the three sugar receptors: GR5a, Gr64a and GR64f.
Flies also use their gustatory system to sense water, employing a GRN that is activated by low osmolarity (Inoshita and Tanimura, 2006). The detection of water depends on a member of the DEG/ENaC (degenerin/epithelial Na+ channel) family of channels, referred to as PPK28 (Cameron et al., 2010; Chen et al., 2010). This channel is activated by low osmolarity, and is required and sufficient for conferring water sensitivity.
Extraoral taste receptors in mammals
Cells outside the taste system such as those in the gut and in the lungs also sense nutrients and toxins and some of the signaling molecules first identified in taste cells may be doing double duty in these organs (Behrens and Meyerhof, 2011). For example, bitter taste receptors are expressed in the airways, where their activation in ciliated epithelial and solitary chemosensory cells leads to changes in respiratory function that protect against inhaled toxins and irritants [Kinnamon, 2011 #3654}. Taste receptors are also expressed by sperm. Most remarkably, in mice carrying a humanized T1R3 gene, infertility is induced by treatment with a human T1R3 specific blocker (Meyer et al., 2012). These studies point to the importance of developing chemicals that target taste receptors for treating conditions ranging from asthma to infertility.
Perhaps more important in the context of trying to understand taste signaling and its relation to behavior is the discovery that taste receptors are expressed in the gastrointestinal tract, where they are positioned to contribute to the regulation of ingestive behavior or satiety (reviewed in (Breer et al., 2012)). Ingested chemicals are detected by a variety of different cell types that line the intestinal lumen, including enterocytes, brush cells and enteroendocrine cells, none of which are neuronal, but which may communicate through vagal afferents or by afferents emanating from the enteric system. Among these, brush cells express gustducin, TRPM5 and T1R3. However, a direct role for these molecules in signaling the caloric quality of ingested foods has not been demonstrated conclusively.
Bitter receptors are also found in enteroendocrine cells in the stomach (Wu et al., 2002), where they may function to elicit protective measures upon ingestion of toxins, such as vomiting. However, this hypothesis has been difficult to test using mice, which do not show a vomiting response. Instead, and somewhat counterintuitively, intragastric infusion of bitter receptor agonists elicit an increase feeding and delayed gastric emptying (Janssen et al., 2011). However, evidence for the involvement of bitter receptors in gastric responses is complicated by the fact that many bitter chemicals are cytotoxic. An alternative might be to use mice that express an unnatural receptor (RASSL) under the promoter of a bitter receptor (Mueller et al., 2005) and that can be challenged by intragastric administration of the inert agonist, spiradoline.
Vertebrates pheromones detected through contact chemosensation
Contact chemosensation is used to detect both tastes and pheromones in vertebrates, and these sensory systems are housed in different organs. Pheromones are detected primarily by the vomeronasal organ (VNO), which lies above the palate and has access to chemicals that enter through the nasal or oral cavity (Dulac and Torello, 2003). Receptors for pheromones fall into two main classes, the V1Rs and the V2Rs, which bear structurally similarity to the T2Rs and T1Rs, respectively. A TRP channel (TRPC2) is an essential downstream element, analogous to the role of TRPM5 in taste cells (Liman et al., 1999; Stowers et al., 2002). In humans and other apes the TRPC2 gene as well as many of the VNO receptors are pseudogenes, indicating that VNO signaling was lost when primates acquired trichromatic color vision (Liman and Innan, 2003). The similarity between components of VNO sensory signaling and those of taste transduction make it tempting to suggest that the two systems evolved from a common precursor with a more general role in contact chemosensation.
Fly taste receptors outside the mouthparts
In flies, there are at least three families of receptors/channels that function in the gustatory response: GRs, IRs and TRPs. Among these families the GRs (gustatory receptors) are so-named since they were originally thought to function exclusively in taste. However, there is now a wealth of evidence demonstrating expression and roles for GRs outside the main mouthparts in the proboscis—the labellum and pharynx. These include expression associated with external taste sensilla in the legs, wing margins, female ovipostor, as well as expression in a segment of the antenna (arista) and in the brain. GRs are also expressed in a variety of afferent neurons associated with other senses, including olfaction, proprioception, hygrosensation and thermosensation (Dunipace et al., 2001; Scott et al., 2001; Thorne and Amrein, 2008).
Expression of GRs in legs accomplishes two tasks. First, it endows the flies with the ability to taste chemicals in the environment without placing the prospective foods in their mouth, and second, it allows flies to directly sample pheromones on prospective mates. The gustatory receptors Gr68a and GR39a are expressed in male forelegs where they may sense female pheromones, as knockdown of the RNAs or mutation of Gr39a decreases mating with females (Bray and Amrein, 2003; Watanabe et al., 2011). In addition to GRNs, Gr68a is expressed in mechanosensory neurons (Ejima and Griffith, 2008; Koganezawa et al., 2010), raising the possibility that it contributes to courtship through a combination of chemo- and mechanosensory functions. Mutation of at least two other Grs that are expressed in forelegs (Gr32 and Gr33a) increases male-male courtship and therefore may be receptors for a male pheromone (Koganezawa et al., 2010; Miyamoto and Amrein, 2008; Moon et al., 2009).
Two Grs function in olfaction, rather than contact chemosensation. These include Gr21a and Gr63a, which are expressed in olfactory receptor neurons in the antenna, and are required and sufficient for the detection of CO2 (Jones et al., 2007; Kwon et al., 2007). Orthologs of Gr21a and Gr63a are expressed in another olfactory organ (maxillary palps) of the mosquito vectors for malaria (Anopheles gambaie), West Nile virus (Culex pipiens) and yellow fever (Aedes aegypti) and knockdown of these RNAs in this latter mosquito impairs CO2 detection (Erdelyan et al., 2012). Thus, two GR family members are actually olfactory receptors and they appear to function as heteromultimers. Additional GRs are expressed in Drosophila olfactory receptor neurons, and may therefore serve as olfactory receptors rather than gustatory receptors (Dunipace et al., 2001; Scott et al., 2001).
Multiple Grs are expressed in tissues outside the gustatory and olfactory systems (Park and Kwon, 2011b; Thorne and Amrein, 2008). These include 18 Gr that are expressed in multidendritic neurons in the body wall and four that that are expressed in neurons that innervate male and female reproductive organs (Park and Kwon, 2011b). Similar to the reports that mammalian taste receptors are expressed in enteroendocrine cells, a survey of gene reporters indicates that at least 12 Grs are expressed in enteroendocrine cells in the midgut of Drosophila (Park and Kwon, 2011a). The fructose receptor, Gr43a, is expressed in many neurons that are not associated with taste, including several in the brain and uterus. In the brain, GR43a senses a rise in fructose in the hemolymph following feeding (Miyamoto et al., 2012). Fructose is a better indicator of food consumption than the main hemolymph sugars glucose and trehalose because it is present only at low levels in hemolymph under starvation conditions and its concentration rises dramatically upon feeding. A homolog of Gr43a (HaGr9a) is expressed in the foregut of the agriculture pest, the cotton bollworm (Helicoverpa armigera) (Xu et al., 2012). These studies imply roles for GRs in sensing internal chemical signals that regulate a host of physiological processes ranging from egg laying to nutrient sensing.
At least three GRs function in detecting sensory inputs other than chemicals. The locus (Gr28b) that encodes these proteins is complex as it includes five transcriptional start sites and encodes five different proteins, all with different N-termini and common C-termini (Robertson et al., 2003; Thorne and Amrein, 2008). One of the genes, Gr28b.d, is expressed in three warm-activated “hot neurons” in the arista—an appendage extending out from the antenna. GR28b(D) helps flies sense a rapid increase in temperature above 26°C, and might be directly heat activated since misexpression of GR28a(D) in sugar-responsive GRNs or motor neurons confers heat sensitivity to these cells (Ni et al., 2013). The function of GR28b(E) is not known, but it might be a thermosensor since it can restore thermosenstivity to hot neurons in flies missing GR28b(D) (Ni et al., 2013). A third member of the Gr28b locus, which remains to be defined, appears to be a light sensor in class IV dendritic arborization neurons in larvae (Xiang et al., 2010). This latter finding is reminiscent of the earlier demonstration that a C. elegans protein that is related to the GR28b proteins (LITE-1) is required for phototaxis (Liu et al., 2010).
The preceding findings demonstrate that GRs function in multiple senses, including taste, smell, light sensation and temperature sensation. The functions of other receptors/channels that contribute to taste, IRs and TRP channels, also have polymodal roles. This is best documented for TRP channels, which contribute to many sensory modalities in flies and mammals (Fowler and Montell, 2013; Venkatachalam and Montell, 2007). Currently, roles for IRs are documented in olfaction and salt taste. Given the broad roles of GRs and TRPs, it would not be surprising if IRs also prove to function in many sensory modalities.
Concluding remarks and future questions
Gustatory receptors were first identified in mammals, leading to the expectation that invertebrates such as Drosophila melanogaster would use related GPCRs to sense the basic qualities of sweet, bitter and umami. Surpisingly, insect taste receptors are not only unrelated to the mammalian receptors, but are mostly inotropic. Such receptors might provide insects with the capacity to quickly sample the chemical environment, while GPCR mediated signaling offers the ability to amplify weak signals and provide for adaptation. Another surprise is that taste receptors are not restricted to the gustatory organs, but are expressed in many cell types, and in some cases contribute to the ability to sense changes in temperature, light and olfactory cues.
Questions for the future include identification of the receptors for high salt, sour taste and for non-canonical tastes such as fatty acids in mammals. The nature of the receptors that allow animals to evaluate food texture are also not known. Also limited is a molecular understanding of taste plasticity, and the mechanisms through which animals integrate chemical taste with temperature, texture and food odors, to decide whether to consume or reject a food. Finally, the dissection of extraoral roles for taste receptors is just beginning, and may to lead to insights into the integration of neuronal activity and metabolism.
Acknowledgments
We thank Peter and Ryan Allen (UCSB) for preparing Figure 1 and Don Arnold (USC) for careful reading of the manuscript. The research in the E.R.L. laboratory is supported by the NIDCD (R21DC012747 and R01DC004564) and the research in the C.M. laboratory is supported by the NIDCD (R01DC007864), NIGMS (R01GM085335) NEI (RO1EY08117-25 and R01EY10852) and the March of Dimes (FY12-423).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errorsmaybe discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Afroz A, Howlett N, Shukla A, Ahmad F, Batista E, Bedard K, Payne S, Morton B, Mansfield JH, Glendinning JI. Gustatory receptor neurons in Manduca sexta contain a TrpA1-dependent signaling pathway that integrates taste and temperature. Chem Senses. 2013;38:605–617. doi: 10.1093/chemse/bjt032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Anzi B, Tracey WD, Jr, Benzer S. Response of Drosophila to wasabi is mediated by painless, the fly homolog of mammalian TRPA1/ANKTM1. Curr Biol. 2006;16:1034–1040. doi: 10.1016/j.cub.2006.04.002. [DOI] [PubMed] [Google Scholar]
- Behrens M, Meyerhof W. Mammalian bitter taste perception. Results Probl Cell Differ. 2009;47:203–220. doi: 10.1007/400_2008_5. [DOI] [PubMed] [Google Scholar]
- Behrens M, Meyerhof W. Gustatory and extragustatory functions of mammalian taste receptors. Physiol Behav. 2011;105:4–13. doi: 10.1016/j.physbeh.2011.02.010. [DOI] [PubMed] [Google Scholar]
- Benton R, Sachse S, Michnick SW, Vosshall LB. Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo. PLoS Biol. 2006;4:e20. doi: 10.1371/journal.pbio.0040020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benton R, Vannice KS, Gomez-Diaz C, Vosshall LB. Variant ionotropic glutamate receptors as chemosensory receptors in Drosophila. Cell. 2009;136:149–162. doi: 10.1016/j.cell.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Born S, Levit A, Niv MY, Meyerhof W, Behrens M. The human bitter taste receptor TAS2R10 is tailored to accommodate numerous diverse ligands. J Neurosci. 2013;33:201–213. doi: 10.1523/JNEUROSCI.3248-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brand JG, Teeter JH, Silver WL. Inhibition by amiloride of chorda tympani responses evoked by monovalent salts. Brain Res. 1985;334:207–214. doi: 10.1016/0006-8993(85)90212-4. [DOI] [PubMed] [Google Scholar]
- Bray S, Amrein H. A putative Drosophila pheromone receptor expressed in male-specific taste neurons is required for efficient courtship. Neuron. 2003;39:1019–1029. doi: 10.1016/s0896-6273(03)00542-7. [DOI] [PubMed] [Google Scholar]
- Bredendiek N, Hutte J, Steingraber A, Hatt H, Gisselmann G, Neuhaus EM. Go alpha is involved in sugar perception in Drosophila. Chem. Senses. 2011;36:69–81. doi: 10.1093/chemse/bjq100. [DOI] [PubMed] [Google Scholar]
- Breer H, Eberle J, Frick C, Haid D, Widmayer P. Gastrointestinal chemosensation: chemosensory cells in the alimentary tract. Histochem Cell Biol. 2012;138:13–24. doi: 10.1007/s00418-012-0954-z. [DOI] [PubMed] [Google Scholar]
- Breza JM, Contreras RJ. Anion size modulates salt taste in rats. J Neurophysiol. 2011;107:1632–1648. doi: 10.1152/jn.00621.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockhoff A, Behrens M, Roudnitzky N, Appendino G, Avonto C, Meyerhof W. Receptor agonism and antagonism of dietary bitter compounds. J Neurosci. 2013;31:14775–14782. doi: 10.1523/JNEUROSCI.2923-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryant B, Silver W. Chemesthesis: the common chemical sense. In: Finger WSTE, Restrepo D, editors. In The Neurobiology of Taste and Smell. 2. John Wiley and Sons, Inc; 2000. pp. 73–100. [Google Scholar]
- Buck LB. Information coding in the olfactory system. Ann Rev Neurosci. 1996;19:517–544. doi: 10.1146/annurev.ne.19.030196.002505. [DOI] [PubMed] [Google Scholar]
- Cameron P, Hiroi M, Ngai J, Scott K. The molecular basis for water taste in Drosophila. Nature. 2010;465:91–95. doi: 10.1038/nature09011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canessa CM, Schild L, Buell G, Thorens B, Gautschi I, Horisberger JD, Rossier BC. Amiloride-sensitive epithelial Na+ channel is made of three homologous subunits. Nature. 1994;367:463–467. doi: 10.1038/367463a0. [DOI] [PubMed] [Google Scholar]
- Cartoni C, Yasumatsu K, Ohkuri T, Shigemura N, Yoshida R, Godinot N, le Coutre J, Ninomiya Y, Damak S. Taste preference for fatty acids is mediated by GPR40 and GPR120. J Neurosci. 2010;30:8376–8382. doi: 10.1523/JNEUROSCI.0496-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandrashekar J, Hoon MA, Ryba NJ, Zuker CS. The receptors and cells for mammalian taste. Nature. 2006;444:288–294. doi: 10.1038/nature05401. [DOI] [PubMed] [Google Scholar]
- Chandrashekar J, Kuhn C, Oka Y, Yarmolinsky DA, Hummler E, Ryba NJ, Zuker CS. The cells and peripheral representation of sodium taste in mice. Nature. 2010;464:297–301. doi: 10.1038/nature08783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandrashekar J, Mueller K, Hoon M, Adler E, Feng L, Guo W, Zuker C, Ryba N. T2Rs function as bitter taste receptors. Cell. 2000;100:703– 711. doi: 10.1016/s0092-8674(00)80706-0. [DOI] [PubMed] [Google Scholar]
- Chandrashekar J, Yarmolinsky D, von Buchholtz L, Oka Y, Sly W, Ryba NJ, Zuker CS. The taste of carbonation. Science. 2009;326:443–445. doi: 10.1126/science.1174601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang RB, Waters H, Liman ER. A proton current drives action potentials in genetically identified sour taste cells. Proc Natl Acad Sci U S A. 2010;107:22320–22325. doi: 10.1073/pnas.1013664107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Charlu S, Wisotsky Z, Medina A, Dahanukar A. Acid sensing by sweet and bitter taste neurons in Drosophila melanogaster. Nat Commun. 2013;4:2042. doi: 10.1038/ncomms3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatzigeorgiou M, Bang S, Hwang SW, Schafer WR. tmc-1 encodes a sodium-sensitive channel required for salt chemosensation in C. elegans. Nature. 2013;494:95–99. doi: 10.1038/nature11845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhari N, Landin AM, Roper SD. A metabotropic glutamate receptor variant functions as a taste receptor. Nat Neurosci. 2000;3:113–119. doi: 10.1038/72053. [DOI] [PubMed] [Google Scholar]
- Chaudhari N, Roper SD. The cell biology of taste. J Cell Biol. 2010;190:285–296. doi: 10.1083/jcb.201003144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z, Wang Q, Wang Z. The amiloride-sensitive epithelial Na+ channel PPK28 is essential for Drosophila gustatory water reception. J Neurosci. 2010;30:6247–6252. doi: 10.1523/JNEUROSCI.0627-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clyne PJ, Warr CG, Carlson JR. Candidate taste receptors in Drosophila. Science. 2000;287:1830–1834. doi: 10.1126/science.287.5459.1830. [DOI] [PubMed] [Google Scholar]
- Colomb J, Grillenzoni N, Ramaekers A, Stocker RF. Architecture of the primary taste center of Drosophila melanogaster larvae. J Comp Neurol. 2007;502:834–847. doi: 10.1002/cne.21312. [DOI] [PubMed] [Google Scholar]
- Croset V, Rytz R, Cummins SF, Budd A, Brawand D, Kaessmann H, Gibson TJ, Benton R. Ancient protostome origin of chemosensory ionotropic glutamate receptors and the evolution of insect taste and olfaction. PLoS Genet. 2010;6:e1001064. doi: 10.1371/journal.pgen.1001064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui M, Jiang P, Maillet E, Max M, Margolskee RF, Osman R. The heterodimeric sweet taste receptor has multiple potential ligand binding sites. Curr Pharm Des. 2006;12:4591–4600. doi: 10.2174/138161206779010350. [DOI] [PubMed] [Google Scholar]
- Dahanukar A, Foster K, van der Goes van Naters WM, Carlson JR. A Gr receptor is required for response to the sugar trehalose in taste neurons of Drosophila. Nat Neurosci. 2001;4:1182–1186. doi: 10.1038/nn765. [DOI] [PubMed] [Google Scholar]
- Dahanukar A, Lei YT, Kwon JY, Carlson JR. Two Gr genes underlie sugar reception in Drosophila. Neuron. 2007;56:503–516. doi: 10.1016/j.neuron.2007.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damak S, Rong M, Yasumatsu K, Kokrashvili Z, Perez CA, Shigemura N, Yoshida R, Mosinger B, Jr, Glendinning JI, Ninomiya Y, Margolskee RF. Trpm5 null mice respond to bitter, sweet, and umami compounds. Chem Senses. 2006;31:253–264. doi: 10.1093/chemse/bjj027. [DOI] [PubMed] [Google Scholar]
- Damak S, Rong M, Yasumatsu K, Kokrashvili Z, Varadarajan V, Zou S, Jiang P, Ninomiya Y, Margolskee RF. Detection of sweet and umami taste in the absence of taste receptor T1r3. Science. 2003;301:850–853. doi: 10.1126/science.1087155. [DOI] [PubMed] [Google Scholar]
- Devambez I, Ali Agha M, Mitri C, Bockaert J, Parmentier ML, Marion-Poll F, Grau Y, Soustelle L. Gαo is required for L-canavanine detection in Drosophila. PLoS ONE. 2013;8:e63484. doi: 10.1371/journal.pone.0063484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dreses-Werringloer U, Lambert JC, Vingtdeux V, Zhao H, Vais H, Siebert A, Jain A, Koppel J, Rovelet-Lecrux A, Hannequin D, et al. A polymorphism in CALHM1 influences Ca2+ homeostasis, Abeta levels, and Alzheimer’s disease risk. Cell. 2008;133:1149–1161. doi: 10.1016/j.cell.2008.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dulac C, Torello AT. Molecular detection of pheromone signals in mammals: from genes to behaviour. Nat Rev Neurosci. 2003;4:551–562. doi: 10.1038/nrn1140. [DOI] [PubMed] [Google Scholar]
- Dunipace L, Meister S, McNealy C, Amrein H. Spatially restricted expression of candidate taste receptors in the Drosophila gustatory system. Curr Biol. 2001;11:822–835. doi: 10.1016/s0960-9822(01)00258-5. [DOI] [PubMed] [Google Scholar]
- Ejima A, Griffith LC. Courtship initiation is stimulated by acoustic signals in Drosophila melanogaster. PLoS ONE. 2008;3:e3246. doi: 10.1371/journal.pone.0003246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erdelyan CN, Mahood TH, Bader TS, Whyard S. Functional validation of the carbon dioxide receptor genes in Aedes aegypti mosquitoes using RNA interference. Insect Mol Biol. 2012;21:119–127. doi: 10.1111/j.1365-2583.2011.01120.x. [DOI] [PubMed] [Google Scholar]
- Falk R, Bleiser-Avivi N, Atidia J. Labellar taste organs of Drosophila melanogaster. J Morph. 1976;150:327–341. doi: 10.1002/jmor.1051500206. [DOI] [PubMed] [Google Scholar]
- Finger TE, Danilova V, Barrows J, Bartel DL, Vigers AJ, Stone L, Hellekant G, Kinnamon SC. ATP signaling is crucial for communication from taste buds to gustatory nerves. Science. 2005;310:1495–1499. doi: 10.1126/science.1118435. [DOI] [PubMed] [Google Scholar]
- Fischler W, Kong P, Marella S, Scott K. The detection of carbonation by the Drosophila gustatory system. Nature. 2007;448:1054–1057. doi: 10.1038/nature06101. [DOI] [PubMed] [Google Scholar]
- Fowler MA, Montell C. Drosophila TRP channels and animal behavior. Life Sci. 2013;92:394–403. doi: 10.1016/j.lfs.2012.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaillard D, Laugerette F, Darcel N, El-Yassimi A, Passilly-Degrace P, Hichami A, Khan NA, Montmayeur JP, Besnard P. The gustatory pathway is involved in CD36-mediated orosensory perception of long-chain fatty acids in the mouse. FASEB J. 2008;22:1458–1468. doi: 10.1096/fj.07-8415com. [DOI] [PubMed] [Google Scholar]
- Galindo MM, Voigt N, Stein J, van Lengerich J, Raguse JD, Hofmann T, Meyerhof W, Behrens M. G protein-coupled receptors in human fat taste perception. Chem Senses. 2011;37:123–139. doi: 10.1093/chemse/bjr069. [DOI] [PubMed] [Google Scholar]
- Galindo-Cuspinera V, Winnig M, Bufe B, Meyerhof W, Breslin PA. A TAS1R receptor-based explanation of sweet ‘water-taste’. Nature. 2006;441:354–357. doi: 10.1038/nature04765. [DOI] [PubMed] [Google Scholar]
- Glendinning JI. Is the bitter rejection response always adaptive? Physiol Behav. 1994;56:1217–1227. doi: 10.1016/0031-9384(94)90369-7. [DOI] [PubMed] [Google Scholar]
- Glendinning JI, Davis A, Ramaswamy S. Contribution of different taste cells and signaling pathways to the discrimination of “bitter” taste stimuli by an insect. J Neurosci. 2002;22:7281–7287. doi: 10.1523/JNEUROSCI.22-16-07281.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordesky-Gold B, Rivers N, Ahmed OM, Breslin PA. Drosophila melanogaster prefers compounds perceived sweet by humans. Chem. Senses. 2008;33:301–309. doi: 10.1093/chemse/bjm088. [DOI] [PubMed] [Google Scholar]
- Heck GL, Mierson S, DeSimone JA. Salt taste transduction occurs through an amiloride-sensitive sodium transport pathway. Science. 1984;223:403–405. doi: 10.1126/science.6691151. [DOI] [PubMed] [Google Scholar]
- Hill CA, Fox AN, Pitts RJ, Kent LB, Tan PL, Chrystal MA, Cravchik A, Collins FH, Robertson HM, Zwiebel LJ. G protein-coupled receptors in Anopheles gambiae. Science. 2002;298:176–178. doi: 10.1126/science.1076196. [DOI] [PubMed] [Google Scholar]
- Hiroi M, Marion-Poll F, Tanimura T. Differentiated response to sugars among labellar chemosensilla in Drosophila. Zoolog Sci. 2002;19:1009–1018. doi: 10.2108/zsj.19.1009. [DOI] [PubMed] [Google Scholar]
- Hiroi M, Meunier N, Marion-Poll F, Tanimura T. Two antagonistic gustatory receptor neurons responding to sweet-salty and bitter taste in Drosophila. J Neurobiol. 2004;61:333–342. doi: 10.1002/neu.20063. [DOI] [PubMed] [Google Scholar]
- Hofmann T, Chubanov V, Gudermann T, Montell C. TRPM5 is a voltage-modulated and Ca2+-activated monovalent selective cation channel. Curr Biol. 2003;13:1153–1158. doi: 10.1016/s0960-9822(03)00431-7. [DOI] [PubMed] [Google Scholar]
- Hoon MA, Adler E, Lindemeier J, Battey JF, Ryba NJ, Zuker CS. Putative mammalian taste receptors: a class of taste-specific GPCRs with distinct topographic selectivity. Cell. 1999;96:541–551. doi: 10.1016/s0092-8674(00)80658-3. [DOI] [PubMed] [Google Scholar]
- Horio N, Yoshida R, Yasumatsu K, Yanagawa Y, Ishimaru Y, Matsunami H, Ninomiya Y. Sour taste responses in mice lacking PKD channels. PLoS One. 2011;6:e20007. doi: 10.1371/journal.pone.0020007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang AL, Chen X, Hoon MA, Chandrashekar J, Guo W, Trankner D, Ryba NJ, Zuker CS. The cells and logic for mammalian sour taste detection. Nature. 2006;442:934–938. doi: 10.1038/nature05084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang YA, Maruyama Y, Stimac R, Roper SD. Presynaptic (Type III) cells in mouse taste buds sense sour (acid) taste. J Physiol. 2008;586:2903–2912. doi: 10.1113/jphysiol.2008.151233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang YJ, Maruyama Y, Dvoryanchikov G, Pereira E, Chaudhari N, Roper SD. The role of pannexin 1 hemichannels in ATP release and cell-cell communication in mouse taste buds. Proc Natl Acad Sci U S A. 2007;104:6436–6441. doi: 10.1073/pnas.0611280104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoshita T, Tanimura T. Cellular identification of water gustatory receptor neurons and their central projection pattern in Drosophila. Proc Natl Acad Sci USA. 2006;103:1094–1099. doi: 10.1073/pnas.0502376103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishimoto H, Takahashi K, Ueda R, Tanimura T. G-protein gamma subunit 1 is required for sugar reception in Drosophila. EMBO J. 2005;24:3259–3265. doi: 10.1038/sj.emboj.7600796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen S, Laermans J, Verhulst PJ, Thijs T, Tack J, Depoortere I. Bitter taste receptors and alpha-gustducin regulate the secretion of ghrelin with functional effects on food intake and gastric emptying. Proc Natl Acad Sci U S A. 2011;108:2094–2099. doi: 10.1073/pnas.1011508108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong YT, Shim J, Oh SR, Yoon HI, Kim CH, Moon SJ, Montell C. An Odorant-Binding Protein required for suppression of sweet taste by bitter chemicals. Neuron. 2013;79:725–737. doi: 10.1016/j.neuron.2013.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang P, Josue J, Li X, Glaser D, Li W, Brand JG, Margolskee RF, Reed DR, Beauchamp GK. Major taste loss in carnivorous mammals. Proc Natl Acad Sci U S A. 2012;109:4956–4961. doi: 10.1073/pnas.1118360109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiao Y, Moon SJ, Montell C. A Drosophila gustatory receptor required for the responses to sucrose, glucose, and maltose identified by mRNA tagging. Proc Natl Acad Sci USA. 2007;104:14110–14115. doi: 10.1073/pnas.0702421104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiao Y, Moon SJ, Wang X, Ren Q, Montell C. Gr64f is required in combination with other gustatory receptors for sugar detection in Drosophila. Curr Biol. 2008;18:1797–1801. doi: 10.1016/j.cub.2008.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones WD, Cayirlioglu P, Grunwald Kadow I, Vosshall LB. Two chemosensory receptors together mediate carbon dioxide detection in Drosophila. Nature. 2007;445:86–90. doi: 10.1038/nature05466. [DOI] [PubMed] [Google Scholar]
- Kain P, Badsha F, Hussain SM, Nair A, Hasan G, Rodrigues V. Mutants in phospholipid signaling attenuate the behavioral response of adult Drosophila to trehalose. Chem. Senses. 2010;35:663–673. doi: 10.1093/chemse/bjq055. [DOI] [PubMed] [Google Scholar]
- Kang K, Panzano VC, Chang EC, Ni L, Dainis AM, Jenkins AM, Regna K, Muskavitch MA, Garrity PA. Modulation of TRPA1 thermal sensitivity enables sensory discrimination in Drosophila. Nature. 2012;481:76–80. doi: 10.1038/nature10715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kapsimali M, Barlow LA. Developing a sense of taste. Semin Cell Dev Biol. 2012;24:200–209. doi: 10.1016/j.semcdb.2012.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kent LB, Walden KK, Robertson HM. The Gr family of candidate gustatory and olfactory receptors in the yellow-fever mosquito Aedes aegypti. Chem. Senses. 2008;33:79–93. doi: 10.1093/chemse/bjm067. [DOI] [PubMed] [Google Scholar]
- Kim SH, Lee Y, Akitake B, Woodward OM, Guggino WB, Montell C. Drosophila TRPA1 channel mediates chemical avoidance in gustatory receptor neurons. Proc Natl Acad Sci USA. 2010;107:8440–8445. doi: 10.1073/pnas.1001425107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim UK, Jorgenson E, Coon H, Leppert M, Risch N, Drayna D. Positional cloning of the human quantitative trait locus underlying taste sensitivity to phenylthiocarbamide. Science. 2003;299:1221–1225. doi: 10.1126/science.1080190. [DOI] [PubMed] [Google Scholar]
- Koganezawa M, Haba D, Matsuo T, Yamamoto D. The shaping of male courtship posture by lateralized gustatory inputs to male-specific interneurons. Curr Biol. 2010;20:1–8. doi: 10.1016/j.cub.2009.11.038. [DOI] [PubMed] [Google Scholar]
- Koizumi A, Tsuchiya A, Nakajima K, Ito K, Terada T, Shimizu-Ibuka A, Briand L, Asakura T, Misaka T, Abe K. Human sweet taste receptor mediates acid-induced sweetness of miraculin. Proc Natl Acad Sci U S A. 2013;108:16819–16824. doi: 10.1073/pnas.1016644108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kretz O, Barbry P, Bock R, Lindemann B. Differential expression of RNA and protein of the three pore-forming subunits of the amiloride-sensitive epithelial sodium channel in taste buds of the rat. J Histochem Cytochem. 1999;47:51–64. doi: 10.1177/002215549904700106. [DOI] [PubMed] [Google Scholar]
- Kuhn C, Bufe B, Batram C, Meyerhof W. Oligomerization of TAS2R bitter taste receptors. Chem Senses. 2010;35:395–406. doi: 10.1093/chemse/bjq027. [DOI] [PubMed] [Google Scholar]
- Kusuhara Y, Yoshida R, Ohkuri T, Yasumatsu K, Voigt A, Hubner S, Maeda K, Boehm U, Meyerhof W, Ninomiya Y. Taste responses in mice lacking taste receptor subunit T1R1. J Physiol. 2013;591:1967–1985. doi: 10.1113/jphysiol.2012.236604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon JY, Dahanukar A, Weiss LA, Carlson JR. The molecular basis of CO2 reception in Drosophila. Proc Natl Acad Sci USA. 2007;104:3574–3578. doi: 10.1073/pnas.0700079104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon JY, Dahanukar A, Weiss LA, Carlson JR. Molecular and cellular organization of the taste system in the Drosophila larva. J Neurosci. 2011;31:15300–15309. doi: 10.1523/JNEUROSCI.3363-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon Y, Shim HS, Wang X, Montell C. Control of thermotactic behavior via coupling of a TRP channel to a phospholipase C signaling cascade. Nat Neurosci. 2008;11:871–873. doi: 10.1038/nn.2170. [DOI] [PubMed] [Google Scholar]
- Lee Y, Kang MJ, Shim J, Cheong CU, Moon SJ, Montell C. Gustatory receptors required for avoiding the insecticide L-canavanine. J Neurosci. 2012;32:1429–1435. doi: 10.1523/JNEUROSCI.4630-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y, Kim SH, Montell C. Avoiding DEET through insect gustatory receptors. Neuron. 2010;67:555–561. doi: 10.1016/j.neuron.2010.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y, Moon SJ, Montell C. Multiple gustatory receptors required for the caffeine response in Drosophila. Proc Natl Acad Sci USA. 2009;106:4495–4500. doi: 10.1073/pnas.0811744106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liman ER, Corey DP, Dulac C. TRP2: a candidate transduction channel for mammalian pheromone sensory signaling. Proc Natl Acad Sci U S A. 1999;96:5791–5796. doi: 10.1073/pnas.96.10.5791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liman ER, Innan H. Relaxed selective pressure on an essential component of pheromone transduction in primate evolution. Proc Natl Acad Sci U S A. 2003;100:3328–3332. doi: 10.1073/pnas.0636123100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin W, Burks CA, Hansen DR, Kinnamon SC, Gilbertson TA. Taste receptor cells express pH-sensitive leak K+ channels. J Neurophysiol. 2004;92:2909–2919. doi: 10.1152/jn.01198.2003. [DOI] [PubMed] [Google Scholar]
- Liu D, Liman ER. Intracellular Ca2+ and the phospholipid PIP2 regulate the taste transduction ion channel TRPM5. Proc Natl Acad Sci U S A. 2003;100:15160–15165. doi: 10.1073/pnas.2334159100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Ward A, Gao J, Dong Y, Nishio N, Inada H, Kang L, Yu Y, Ma D, Xu T, et al. C. elegans phototransduction requires a G protein-dependent cGMP pathway and a taste receptor homolog. Nat Neurosci. 2010;13:715–722. doi: 10.1038/nn.2540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Leonard AS, Motto DG, Feller MA, Price MP, Johnson WA, Welsh MJ. Contribution of Drosophila DEG/ENaC genes to salt taste. Neuron. 2003a;39:133–146. doi: 10.1016/s0896-6273(03)00394-5. [DOI] [PubMed] [Google Scholar]
- Liu L, Yermolaieva O, Johnson WA, Abboud FM, Welsh MJ. Identification and function of thermosensory neurons in Drosophila larvae. Nat Neurosci. 2003b;6:267–273. doi: 10.1038/nn1009. [DOI] [PubMed] [Google Scholar]
- Liu P, Shah BP, Croasdell S, Gilbertson TA. Transient receptor potential channel type M5 is essential for fat taste. J Neurosci. 2011;31:8634–8642. doi: 10.1523/JNEUROSCI.6273-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyall V, Alam RI, Phan DQ, Ereso GL, Phan TH, Malik SA, Montrose MH, Chu S, Heck GL, Feldman GM, DeSimone JA. Decrease in rat taste receptor cell intracellular pH is the proximate stimulus in sour taste transduction. Am J Physiol Cell Physiol. 2001;281:C1005–1013. doi: 10.1152/ajpcell.2001.281.3.C1005. [DOI] [PubMed] [Google Scholar]
- Lyall V, Heck GL, Vinnikova AK, Ghosh S, Phan TH, Alam RI, Russell OF, Malik SA, Bigbee JW, DeSimone JA. The mammalian amilorideinsensitive non-specific salt taste receptor is a vanilloid receptor-1 variant. J Physiol. 2004;558:147–159. doi: 10.1113/jphysiol.2004.065656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masek P, Keene AC. Drosophila fatty acid taste signals through the PLC pathway in sugar-sensing nurons. PLoS Genet. 2013;9:e1003710. doi: 10.1371/journal.pgen.1003710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsunami H, Montmayeur JP, Buck LB. A family of candidate taste receptors in human and mouse. Nature. 2000;404:601–604. doi: 10.1038/35007072. [DOI] [PubMed] [Google Scholar]
- McDonald FJ, Price MP, Snyder PM, Welsh MJ. Cloning and expression of the β- and ɣ-subunits of the human epithelial sodium channel. Am J Physiol. 1995;268:C1157–1163. doi: 10.1152/ajpcell.1995.268.5.C1157. [DOI] [PubMed] [Google Scholar]
- Meunier N, Marion-Poll F, Rospars JP, Tanimura T. Peripheral coding of bitter taste in Drosophila. J Neurobiol. 2003;56:139–152. doi: 10.1002/neu.10235. [DOI] [PubMed] [Google Scholar]
- Meyer D, Voigt A, Widmayer P, Borth H, Huebner S, Breit A, Marschall S, de Angelis MH, Boehm U, Meyerhof W, et al. Expression of Tas1 taste receptors in mammalian spermatozoa: functional role of Tas1r1 in regulating basal Ca(2)(+) and cAMP concentrations in spermatozoa. PLoS One. 2012;7:e32354. doi: 10.1371/journal.pone.0032354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra D, Miyamoto T, Rezenom YH, Broussard A, Yavuz A, Slone J, Russell DH, Amrein H. The molecular basis of sugar sensing in Drosophila larvae. Curr Biol. 2013;23:1466–1471. doi: 10.1016/j.cub.2013.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitri C, Soustelle L, Framery B, Bockaert J, Parmentier ML, Grau Y. Plant insecticide L-canavanine repels Drosophila via the insect orphan GPCR DmX. PLoS Biol. 2009;7:e1000147. doi: 10.1371/journal.pbio.1000147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyakawa Y. Behavioural evidence for the existence of sugar, salt and amino acid taste receptor cells and some of their properties in Drosophila larvae. J Insect Physiol. 1982;28:405–410. [Google Scholar]
- Miyamoto T, Amrein H. Suppression of male courtship by a Drosophila pheromone receptor. Nat Neurosci. 2008;11:874–876. doi: 10.1038/nn.2161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto T, Slone J, Song X, Amrein H. A fructose receptor functions as a nutrient sensor in the Drosophila brain. Cell. 2012;151:1113–1125. doi: 10.1016/j.cell.2012.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moon SJ, Köttgen M, Jiao Y, Xu H, Montell C. A taste receptor required for the caffeine response in vivo. Curr Biol. 2006;16:1812–1817. doi: 10.1016/j.cub.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Moon SJ, Lee Y, Jiao Y, Montell C. A Drosophila gustatory receptor essential for aversive taste and inhibiting male-to-male courtship. Curr Biol. 2009;19:1623–1627. doi: 10.1016/j.cub.2009.07.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller KL, Hoon MA, Erlenbach I, Chandrashekar J, Zuker CS, Ryba NJ. The receptors and coding logic for bitter taste. Nature. 2005;434:225–229. doi: 10.1038/nature03352. [DOI] [PubMed] [Google Scholar]
- Murata Y, Yasuo T, Yoshida R, Obata K, Yanagawa Y, Margolskee RF, Ninomiya Y. Action potential-enhanced ATP release from taste cells through hemichannels. J Neurophysiol. 2010;104:896–901. doi: 10.1152/jn.00414.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa T, Vosshall LB. Controversy and consensus: noncanonical signaling mechanisms in the insect olfactory system. Curr Opin Neurobiol. 2009;19:284–292. doi: 10.1016/j.conb.2009.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson G, Hoon MA, Chandrashekar J, Zhang Y, Ryba NJ, Zuker CS. Mammalian sweet taste receptors. Cell. 2001;106:381–390. doi: 10.1016/s0092-8674(01)00451-2. [DOI] [PubMed] [Google Scholar]
- Ni L, Bronk P, Chang EC, Lowell AM, Flam JO, Panzano VC, Theobald DL, Griffith LC, Garrity PA. A gustatory receptor paralogue controls rapid warmth avoidance in Drosophila. Nature. 2013;500:580–584. doi: 10.1038/nature12390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ninomiya Y. Reinnervation of cross-regenerated gustatory nerve fibers into amiloride-sensitive and amiloride-insensitive taste receptor cells. Proc Natl Acad Sci U S A. 1998;95:5347–5350. doi: 10.1073/pnas.95.9.5347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohkuri T, Horio N, Stratford JM, Finger TE, Ninomiya Y. Residual Chemoresponsiveness to Acids in the Superior Laryngeal Nerve in “Taste-Blind” (P2X2/P2X3 Double-KO) Mice. Chem Senses. 2012;37:523–532. doi: 10.1093/chemse/bjs004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka Y, Butnaru M, von Buchholtz L, Ryba NJ, Zuker CS. High salt recruits aversive taste pathways. Nature. 2013;494:472–475. doi: 10.1038/nature11905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JH, Kwon JY. Heterogeneous expression of Drosophila gustatory receptors in enteroendocrine cells. PLoS ONE. 2011a;6:e29022. doi: 10.1371/journal.pone.0029022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JH, Kwon JY. A systematic analysis of Drosophila gustatory receptor gene expression in abdominal neurons which project to the central nervous system. Mol Cells. 2011b;32:375–381. doi: 10.1007/s10059-011-0128-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peñalva-Arana DC, Lynch M, Robertson HM. The chemoreceptor genes of the waterflea Daphnia pulex: many Grs but no Ors. BMC Evol Biol. 2009;9:79. doi: 10.1186/1471-2148-9-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perez CA, Huang L, Rong M, Kozak JA, Preuss AK, Zhang H, Max M, Margolskee RF. A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci. 2002;5:1169–1176. doi: 10.1038/nn952. [DOI] [PubMed] [Google Scholar]
- Prawitt D, Monteilh-Zoller MK, Brixel L, Spangenberg C, Zabel B, Fleig A, Penner R. TRPM5 is a transient Ca2+-activated cation channel responding to rapid changes in [Ca2+]i. Proc Natl Acad Sci U S A. 2003;100:15166–15171. doi: 10.1073/pnas.2334624100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richter TA, Dvoryanchikov GA, Chaudhari N, Roper SD. Acidsensitive two-pore domain potassium (K2P) channels in mouse taste buds. J Neurophysiol. 2004;92:1928–1936. doi: 10.1152/jn.00273.2004. [DOI] [PubMed] [Google Scholar]
- Robertson HM, Wanner KW. The chemoreceptor superfamily in the honey bee, Apis mellifera: expansion of the odorant, but not gustatory, receptor family. Genome Res. 2006;16:1395–1403. doi: 10.1101/gr.5057506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson HM, Warr CG, Carlson JR. Molecular evolution of the insect chemoreceptor gene superfamily in Drosophila melanogaster. Proc Natl Acad Sci USA. 2003;100(Suppl 2):14537–14542. doi: 10.1073/pnas.2335847100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohács T, Lopes CM, Michailidis I, Logothetis DE. PI(4,5)P2 regulates the activation and desensitization of TRPM8 channels through the TRP domain. Nat Neurosci. 2005;8:626–634. doi: 10.1038/nn1451. [DOI] [PubMed] [Google Scholar]
- Rolls ET, Critchley HD, Browning AS, Hernadi I, Lenard L. Responses to the sensory properties of fat of neurons in the primate orbitofrontal cortex. J Neurosci. 1999;19:1532–1540. doi: 10.1523/JNEUROSCI.19-04-01532.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romanov RA, Rogachevskaja OA, Bystrova MF, Jiang P, Margolskee RF, Kolesnikov SS. Afferent neurotransmission mediated by hemichannels in mammalian taste cells. EMBO J. 2007;26:657–667. doi: 10.1038/sj.emboj.7601526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roper SD. Signal transduction and information processing in mammalian taste buds. Pflugers Arch. 2007;454:759–776. doi: 10.1007/s00424-007-0247-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato K, Pellegrino M, Nakagawa T, Nakagawa T, Vosshall LB, Touhara K. Insect olfactory receptors are heteromeric ligand-gated ion channels. Nature. 2008;452:1002–1006. doi: 10.1038/nature06850. [DOI] [PubMed] [Google Scholar]
- Sato K, Tanaka K, Touhara K. Sugar-regulated cation channel formed by an insect gustatory receptor. Proc Natl Acad Sci USA. 2011;108:11680–11685. doi: 10.1073/pnas.1019622108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schipanski A, Yarali A, Niewalda T, Gerber B. Behavioral analyses of sugar processing in choice, feeding, and learning in larval Drosophila. Chem. Senses. 2008;33:563–573. doi: 10.1093/chemse/bjn024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott K, Brady R, Jr, Cravchik A, Morozov P, Rzhetsky A, Zuker C, Axel R. A chemosensory gene family encoding candidate gustatory and olfactory receptors in Drosophila. Cell. 2001;104:661–673. doi: 10.1016/s0092-8674(01)00263-x. [DOI] [PubMed] [Google Scholar]
- Servant G, Tachdjian C, Tang XQ, Werner S, Zhang F, Li X, Kamdar P, Petrovic G, Ditschun T, Java A, et al. Positive allosteric modulators of the human sweet taste receptor enhance sweet taste. Proc Natl Acad Sci U S A. 2010;107:4746–4751. doi: 10.1073/pnas.0911670107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi P, Zhang J. Contrasting modes of evolution between vertebrate sweet/umami receptor genes and bitter receptor genes. Mol Biol Evol. 2006;23:292–300. doi: 10.1093/molbev/msj028. [DOI] [PubMed] [Google Scholar]
- Slone J, Daniels J, Amrein H. Sugar receptors in Drosophila. Curr Biol. 2007;17:1809–1816. doi: 10.1016/j.cub.2007.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith CD, Zimin A, Holt C, Abouheif E, Benton R, Cash E, Croset V, Currie CR, Elhaik E, Elsik CG, et al. Draft genome of the globally widespread and invasive Argentine ant (Linepithema humile) Proc Natl Acad Sci U S A. 2011;108:5673–5678. doi: 10.1073/pnas.1008617108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DV, David BJ. Neural Representation of Taste. In: Finger TE, Silver WL, Restrepo D, editors. The neurobiology of taste and smell. Wiley; 2000. pp. 353–394. [Google Scholar]
- Stocker RF. The organization of the chemosensory system in Drosophila melanogaster: a review. Cell Tissue Res. 1994;275:3–26. doi: 10.1007/BF00305372. [DOI] [PubMed] [Google Scholar]
- Stocker RF. Design of the larval chemosensory system. Adv Exp Med Biol. 2008;628:69–81. doi: 10.1007/978-0-387-78261-4_5. [DOI] [PubMed] [Google Scholar]
- Stowers L, Holy TE, Meister M, Dulac C, Koentges G. Loss of sex discrimination and male-male aggression in mice deficient for TRP2. Science. 2002;295:1493–1500. doi: 10.1126/science.1069259. [DOI] [PubMed] [Google Scholar]
- Taruno A, Vingtdeux V, Ohmoto M, Ma Z, Dvoryanchikov G, Li A, Adrien L, Zhao H, Leung S, Abernethy M, et al. CALHM1 ion channel mediates purinergic neurotransmission of sweet, bitter and umami tastes. Nature. 2013;495:223–226. doi: 10.1038/nature11906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temussi PA. New insights into the characteristics of sweet and bitter taste receptors. Int Rev Cell Mol Biol. 2011;291:191–226. doi: 10.1016/B978-0-12-386035-4.00006-9. [DOI] [PubMed] [Google Scholar]
- Thorne N, Amrein H. Atypical expression of Drosophila gustatory receptor genes in sensory and central neurons. J Comp Neurol. 2008;506:548–568. doi: 10.1002/cne.21547. [DOI] [PubMed] [Google Scholar]
- Tominaga M, Caterina MJ, Malmberg AB, Rosen TA, Gilbert H, Skinner K, Raumann BE, Basbaum AI, Julius D. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron. 1998;21:531–543. doi: 10.1016/s0896-6273(00)80564-4. [DOI] [PubMed] [Google Scholar]
- Tordoff MG, Alarcon LK, Valmeki S, Jiang P. T1R3: a human calcium taste receptor. Sci Rep. 2012;2:496. doi: 10.1038/srep00496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tordoff MG, Ellis HT. Taste dysfunction in BTBR mice due to a mutation of Itpr3, the inositol triphosphate receptor 3 gene. Physiol Genomics. 2013;45:834–855. doi: 10.1152/physiolgenomics.00092.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tordoff MG, Shao H, Alarcon LK, Margolskee RF, Mosinger B, Bachmanov AA, Reed DR, McCaughey S. Involvement of T1R3 in calcium-magnesium taste. Physiol Genomics. 2008;34:338–348. doi: 10.1152/physiolgenomics.90200.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toshima N, Tanimura T. Taste preference for amino acids is dependent on internal nutritional state in Drosophila melanogaster. J Exp Biol. 2012;215:2827–2832. doi: 10.1242/jeb.069146. [DOI] [PubMed] [Google Scholar]
- Troemel ER, Kimmel BE, Bargmann CI. Reprogramming chemotaxis responses: sensory neurons define olfactory preferences in C. elegans. Cell. 1997;91:161–169. doi: 10.1016/s0092-8674(00)80399-2. [DOI] [PubMed] [Google Scholar]
- Ueno K, Kidokoro Y. Adenylyl cyclase encoded by AC78C participates in sugar perception in Drosophila melanogaster. Eur J Neurosci. 2008;28:1956–1966. doi: 10.1111/j.1460-9568.2008.06507.x. [DOI] [PubMed] [Google Scholar]
- Ueno K, Kohatsu S, Clay C, Forte M, Isono K, Kidokoro Y. Gsα is involved in sugar perception in Drosophila melanogaster. J Neurosci. 2006;26:6143–6152. doi: 10.1523/JNEUROSCI.0857-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueno K, Ohta M, Morita H, Mikuni Y, Nakajima S, Yamamoto K, Isono K. Trehalose sensitivity in Drosophila correlates with mutations in and expression of the gustatory receptor gene Gr5a. Curr Biol. 2001;11:1451–1455. doi: 10.1016/s0960-9822(01)00450-x. [DOI] [PubMed] [Google Scholar]
- Venkatachalam K, Montell C. TRP channels. Annu Rev Biochem. 2007;76:387–417. doi: 10.1146/annurev.biochem.75.103004.142819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viswanath V, Story GM, Peier AM, Petrus MJ, Lee VM, Hwang SW, Patapoutian A, Jegla T. Opposite thermosensor in fruitfly and mouse. Nature. 2003;423:822–823. doi: 10.1038/423822a. [DOI] [PubMed] [Google Scholar]
- Voigt A, Hubner S, Lossow K, Hermans-Borgmeyer I, Boehm U, Meyerhof W. Genetic labeling of Tas1r1 and Tas2r131 taste receptor cells in mice. Chem Senses. 2012;37:897–911. doi: 10.1093/chemse/bjs082. [DOI] [PubMed] [Google Scholar]
- Vosshall LB, Stocker RF. Molecular architecture of smell and taste in Drosophila. Annu Rev Neurosci. 2007;30:505–533. doi: 10.1146/annurev.neuro.30.051606.094306. [DOI] [PubMed] [Google Scholar]
- Wang G, Qiu YT, Lu T, Kwon HW, Pitts RJ, Van Loon JJ, Takken W, Zwiebel LJ. Anopheles gambiae TRPA1 is a heat-activated channel expressed in thermosensitive sensilla of female antennae. Eur J Neurosci. 2009;30:967–974. doi: 10.1111/j.1460-9568.2009.06901.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YY, Chang RB, Liman ER. TRPA1 is a component of the nociceptive response to CO2. J Neurosci. 2010;30:12958–12963. doi: 10.1523/JNEUROSCI.2715-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe K, Toba G, Koganezawa M, Yamamoto D. Gr39a, a highly diversified gustatory receptor in Drosophila, has a role in sexual behavior. Behavior genetics. 2011;41:746–753. doi: 10.1007/s10519-011-9461-6. [DOI] [PubMed] [Google Scholar]
- Weiss LA, Dahanukar A, Kwon JY, Banerjee D, Carlson JR. The molecular and cellular basis of bitter taste in Drosophila. Neuron. 2011;69:258–272. doi: 10.1016/j.neuron.2011.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wicher D, Schafer R, Bauernfeind R, Stensmyr MC, Heller R, Heinemann SH, Hansson BS. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature. 2008;452:1007–1011. doi: 10.1038/nature06861. [DOI] [PubMed] [Google Scholar]
- Wisotsky Z, Medina A, Freeman E, Dahanukar A. Evolutionary differences in food preference rely on Gr64e, a receptor for glycerol. Nat Neurosci. 2011;14:1534–1541. doi: 10.1038/nn.2944. [DOI] [PubMed] [Google Scholar]
- Wu SV, Rozengurt N, Yang M, Young SH, Sinnett-Smith J, Rozengurt E. Expression of bitter taste receptors of the T2R family in the gastrointestinal tract and enteroendocrine STC-1 cells. Proc Natl Acad Sci U S A. 2002;99:2392–2397. doi: 10.1073/pnas.042617699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiang Y, Yuan Q, Vogt N, Looger LL, Jan LY, Jan YN. Light-avoidance-mediating photoreceptors tile the Drosophila larval body wall. Nature. 2010;468:921–926. doi: 10.1038/nature09576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Zhang HJ, Anderson A. A sugar gustatory receptor identified from the foregut of cotton bollworm Helicoverpa armigera. J Chem Ecol. 2012;38:1513–1520. doi: 10.1007/s10886-012-0221-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi S. The synergistic taste effect of monosodium glutamate and disodium 5′-inosinate. J Food Sci. 1970;32:473–478. [Google Scholar]
- Yarmolinsky DA, Zuker CS, Ryba NJ. Common sense about taste: from mammals to insects. Cell. 2009;139:234–244. doi: 10.1016/j.cell.2009.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yee KK, Sukumaran SK, Kotha R, Gilbertson TA, Margolskee RF. Glucose transporters and ATP-gated K+ (KATP) metabolic sensors are present in type 1 taste receptor 3 (T1r3)-expressing taste cells. Proc Natl Acad Sci U S A. 2011;108:5431–5436. doi: 10.1073/pnas.1100495108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang F, Klebansky B, Fine RM, Xu H, Pronin A, Liu H, Tachdjian C, Li X. Molecular mechanism for the umami taste synergism. Proc Natl Acad Sci U S A. 2008;105:20930–20934. doi: 10.1073/pnas.0810174106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang HJ, Anderson AR, Trowell SC, Luo AR, Xiang ZH, Xia QY. Topological and functional characterization of an insect gustatory receptor. PLoS ONE. 2011;6:e24111. doi: 10.1371/journal.pone.0024111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Hoon M, Chandrashekar J, Mueller K, Cook B, Wu D, Zuker C, Ryba N. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell. 2003;112:293– 301. doi: 10.1016/s0092-8674(03)00071-0. [DOI] [PubMed] [Google Scholar]
- Zhang YV, Ni J, Montell C. The molecular basis for attractive salt-taste coding in Drosophila. Science. 2013a;340:1334–1338. doi: 10.1126/science.1234133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YV, Raghuwanshi RP, Shen WL, Montell C. Food-experience induced taste desensitization modulated by the Drosophila TRPL channel. Nat Neurosci. 2013b;16:1468–1476. doi: 10.1038/nn.3513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z, Zhao Z, Margolskee R, Liman E. The transduction channel TRPM5 is gated by intracellular calcium in taste cells. J Neurosci. 2007;27:5777–5786. doi: 10.1523/JNEUROSCI.4973-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao GQ, Zhang Y, Hoon MA, Chandrashekar J, Erlenbach I, Ryba NJ, Zuker CS. The receptors for mammalian sweet and umami taste. Cell. 2003;115:255–266. doi: 10.1016/s0092-8674(03)00844-4. [DOI] [PubMed] [Google Scholar]
- Zhao H, Yang JR, Xu H, Zhang J. Pseudogenization of the umami taste receptor gene Tas1r1 in the giant panda coincided with its dietary switch to bamboo. Mol Biol Evol. 2010;27:2669–2673. doi: 10.1093/molbev/msq153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong L, Bellemer A, Yan H, Honjo K, Robertson J, Hwang RY, Pitt GS, Tracey WD. Thermosensory and non-thermosensory isoforms of Drosophila melanogaster TRPA1 reveal heat sensor domains of a thermoTRP channel. Cell Rep. 2012;1:43–55. doi: 10.1016/j.celrep.2011.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou X, Slone JD, Rokas A, Berger SL, Liebig J, Ray A, Reinberg D, Zwiebel LJ. Phylogenetic and transcriptomic analysis of chemosensory receptors in a pair of divergent ant species reveals sex-specific signatures of odor coding. PLoS Genet. 2012;8:e1002930. doi: 10.1371/journal.pgen.1002930. [DOI] [PMC free article] [PubMed] [Google Scholar]