Abstract
Psoriatic arthritis (PsA) is an immune-mediated chronic inflammatory disease, affecting both the skin and joints. Disease progression is associated with aberrant cytokine expression, and TNF blockade is the most successful therapy to date. However, not all patients are responsive to anti-TNF treatment, highlighting the need to better understand the cellular and molecular mechanisms that govern the disease. PsA associations with single nucleotide polymorphisms in IL23R as well as TRAF3IP2 (Act1), a molecule downstream of the IL-17 receptor (IL-17R), have linked the IL-23/IL-17 axis to disease pathology. Although both cytokines are implicated in PsA, a full picture of their cellular targets and pathogenic mechanisms has not yet emerged. In this review, we focus on the IL-23/IL-17 axis-elicited responses mediated by osteoclasts, keratinocytes and neutrophils. Expanding our understanding of the cellular and molecular mechanisms that dictate pathogenicity in PsA will contribute to developing novel treatment strategies to combat disease.
Keywords: IL-17, IL-23, psoriatic arthritis, NF-κB
1. Introduction
Psoriatic arthritis (PsA) is an immune-mediated chronic inflammatory disease affecting both the skin and joints in up to 1% of the worldwide population [1]. Psoriatic skin features commonly include epidermal hyperplasia (thickening of the epidermis), hyperkeratosis (thickening of the stratum corneum), parakeratosis (retention of nuclei in the stratum corneum), Munro’s microabscesses (neutrophilic granulocytes in the epidermis) and mixed dermal infiltrates, including T cells, dendritic cells (DCs) and macrophages which together, lead to the clinical features of raised erythematous silvery plaques [2],[3]. The arthritis of PsA targets the spine, peripheral joints and the entheses (attachment sites of ligament to bone) [4]. PsA can lead to destructive bone loss, and 67% of PsA patients exhibit signs of erosive bone disease [1]. Peripheral enthesitis is a feature of a group of arthritides known as spondylarthropathies, which include PsA, but is not commonly observed in other forms of arthritis, such as rheumatoid arthritis (RA) [5]. Other distinguishing features of PsA include elevated numbers of neutrophils and CD163+ macrophages in the synovium and the presence of typical psoriasis nail lesions, not observed in RA [1],[6]. On the contrary, rheumatoid factor and anti-citrullinated peptide antibodies, which are commonly associated with RA, are not typically present in PsA [7],[8]. Despite the differences, increased numbers of osteoclasts have been observed in PsA synovium, similar to RA, suggesting that some molecular mechanisms that contribute to its pathogenicity are shared across these types of inflammatory arthritis [9]. Since osteoclast activation can be achieved through IL-23 and IL-17 as reviewed in [10], it is plausible that the IL-23/IL-17 axis may govern distinct cellular and molecular mechanisms that contribute to bone erosion and epidermal hyperplasia, hallmark features of PsA.
A variety of genetic, immunological and environmental factors have been suggested to contribute to PsA pathogenesis [11]. Single nucleotide polymorphisms in IL23A, IL23R as well as TRAF3IP2 (Act1), a downstream target of the IL-17 receptor (IL-17R), confer susceptibility to PsA, implying a central role of the IL-23/IL-17 axis in PsA disease pathogenesis [12],[13],[14]. In this review, we will address the contribution of the IL-23/IL-17 axis to PsA, with a specific emphasis on the activation of osteoclasts, which are responsible for bone degradation, and of keratinocytes and neutrophils, which have been implicated in IL-23/IL-17-induced PsA pathology.
2. Molecular pathways in PsA pathogenesis
2.1 IL-23 and IL-23 receptor
IL-23 is a heterodimeric cytokine, composed of a p19 and a p40 subunit; it binds IL-23R and IL-12Rβ1, the latter being shared with IL-12 [15]. The p40 subunit can act as a monomer, homodimer or as a heterodimer with p19, and both subunits are secreted predominantly by macrophages and DCs [15]. Genetically engineered IL-23R GFP reporter mice have confirmed that IL-23R is expressed on the surface of lymphoid cells, such as αβ and γδ T cells, innate lymphoid cells and cells of myeloid origin, including DCs, macrophages and monocytes [16]. The human IL-23R cytoplasmic domain has no inherent kinase activity yet contains seven tyrosine residues that can be phosphorylated to initiate downstream signaling [17]. Six of the seven human IL-23R tyrosine residues are conserved in mouse [17]. Three Src homology 2 domain (SH2) binding sites at Y399, Y484 and Y611 recruit SHP2 tyrosine phosphatase and signal transducer and activator of transcription (STAT)4, and STAT1 and STAT3, respectively [17]. Janus kinases, Jak2 and Tyk2, bind directly to IL-23R and IL-12Rβ1, respectively, and induce phosphorylation of their receptors and STAT3 to induce RORγ and RORα, both of which are encoded by the RORC gene to establish Th17-specific cell differentiation, as evidenced by increased gene expression of IL-17, IL-17F and IL-23R [17],[18],[19]. STAT3 regulates an isoform of RORγ, RORγt, and both STAT3 and RORγt bind the IL-17 promoter [20],[21]. Binding of IL-23 to its receptor also stimulates the degradation of inhibitory subunit of nuclear factor kappa B alpha (IκBα) to induce activation of nuclear factor of kappa light chain enhancer of activated B cells (NF-κB) [22] (Fig 1).
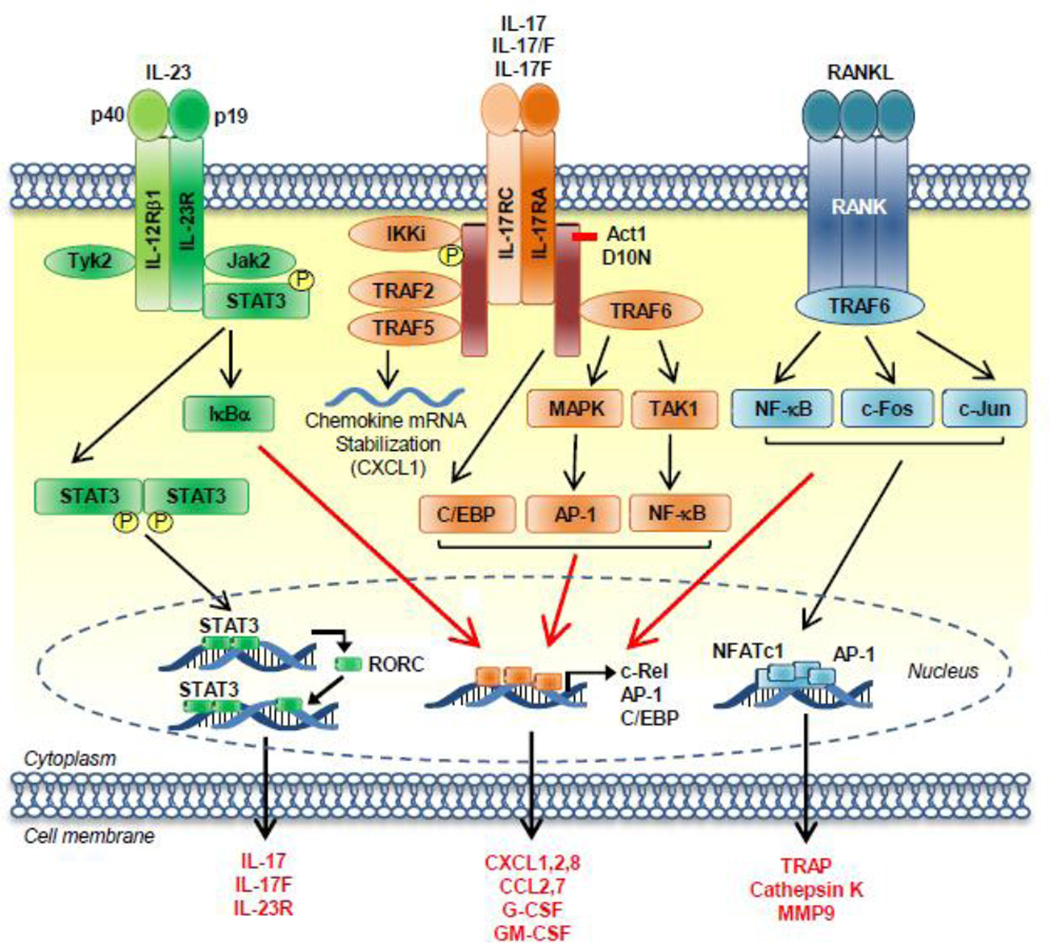
Fig. 1. IL-23, IL-17 and RANK receptor signaling.
IL-23 subunits, p40 and p19, bind to their respective receptors, IL-12Rβ1 and IL-23R, leading to Tyk2 and Jak2 activation and phosphorylation of STAT3 and IκBα leading to NF-κB activation. STAT3 translocates to the nucleus, where it initiates transcriptional activation of STAT3 and RORC leading to expression of IL-17, IL-17F and IL-23R. IL-17 signals through IL-17RA and IL-17RC receptors to recruit the Act1 adaptor, which in turn recruits TRAF2, TRAF5, and TRAF6. TRAF2 and TRAF5 lead to stabilization of CXCL1 mRNA, whereas TRAF6 leads to activation of NF-κB, AP-1 and C/EBP. TRAF6 is also a critical signaling component of the RANK receptor, which also leads to activation of NF-κB, c-Fos and c-Jun, which induce transcriptional activity of NFATc1 and AP-1 leading to production of TRAP, CatK and MMP9, enzymes required for osteoclast mediated bone destruction.
2.2 IL-17 and IL-17 receptor
IL-17 (IL-17A) is a 15–20-kDa glycoprotein and a member of the IL-17 family of cytokines (IL-17, IL-17B, IL-17C, IL-17D, IL-17E, IL-17F [23]. Biologically active IL-17 is produced as a 35-kDa homodimer or heterodimer with IL-17F by αβ T cells, innate lymphoid cells including γδ T-cells, innate-like lymphoid cells, mast cells and neutrophils [24],[25]. IL-17 binds IL-17R (IL-17RA/IL-17RC), which is expressed by various cells such as monocytes, lymphocytes, lymphoid tissue inducer cells, epithelial cells, synoviocytes, fibroblasts and keratinocytes [26],[27] (Fig. 1).
IL-17RA and IL-17RC interact through specific SEFIR (similar expression to fibroblast growth factor genes and IL-17R) domains with the adaptor protein Act1 [28],[27]. Act1 associates with inducible IκB kinase, IKKi, indispensable for IL-17-induced neutrophilia [29]. Phosphorylation of Act1 (S311) leads to the formation of tumor necrosis factor associated factor (TRAF)2–Act1 and TRAF5–Act1 complexes and stabilization of CXCL1 mRNA, a potent neutrophil chemokine [29]. In addition to TRAF2 and TRAF5, Act1 also binds TRAF6 to activate the NF-κB activator protein 1 (AP-1) or the CCAAT-enhancer-binding protein (C/EBP) cascade [30],[31]. Independently of IKKi, Act1 ubiquinates TRAF6 leading to the activation of NF-κB [32]. Moreover, NF-κB activity can be suppressed through TANK binding kinase 1 (TBK1) phosphorylation of Act1 on additional serine residues in both human (S162, S220, and S233) and mice (S147, S209, and S220) [33]. Thus, the IL-23/IL-17 axis influences activation of the NF-κB pathway in multiple ways.
The relevance of the IL-23/IL-17 axis in PsA is suggested by the elevation of IL-23p19/IL-23R and IL-17/IL-17R in psoriatic skin and synovial fluid from PsA patients [34],[35],[36],[37]. Some findings in rodents argue that IL-17 was not required for IL-23-induced psoriatic-like disease, and IL-17 intradermal delivery failed to induce epidermal hyperplasia [38]. However, others using IL-17 deficient mice have demonstrated that IL-7A, is a downstream mediator for IL-23-induced changes in murine skin, and IL-17A may represent an attractive therapeutic target in psoriatic patients [39]. In support of a prominent role of IL-17 in psoriasis, Cai et al., showed that absence of IL-17 signaling ameliorated IL-23-induced psoriatic-like features in mice [40]. Using an ex vivo human skin model, IL-17 failed to induce epidermal hyperplasia [41]. Collectively these data indicate that our understanding of the role of IL-17 in epidermal hyperplasia is incomplete, and it remains possible that IL-17 may have indirect effects on psoriatic pathology.
Neutralization of IL-23 signaling by antibodies and/or IL-23p19 gene ablation ameliorated collagen-induced arthritis (CIA), whereas overexpression of IL-23 induced arthritis with a severe bone destructive phenotype, independently of IL-17 [42],[43],[44]. However, neutralization of IL-17 signaling and gene ablation also demonstrated protection in the CIA model [45],[46]. Therefore, IL-23 and IL-17 may exert effects in bone loss independent of each other and may induce bone resorption associated with PsA pathology by separate molecular mechanisms.
2.3 RANKL and RANK receptor
Receptor activator of nuclear factor kappa B ligand (RANKL) is a homotrimeric transmembrane protein expressed by bone-forming osteoblasts under physiological conditions; upon proteolytic cleavage or alternative splicing, RANKL is secreted as a soluble protein [47]. RANKL interacts with RANK, a homotrimeric transmembrane receptor expressed commonly on osteoclast precursors and DCs to induce their differentiation into multinucleated, bone-resorbing osteoclasts [48]. Upon binding RANKL, the RANK receptor recruits adaptor molecules, such as TRAF proteins (TRAF2, TRAF5 and TRAF6), to induce NF-κB and mitogen-activated kinases, such as Jun N-terminal kinase (JNK), nuclear factor of activated T-cell cytoplasmic 1 (NFATc1) and AP-1 [49] (Fig. 1). Activation of these transcription factors leads to secretion of bone matrix degradation enzymes, including tartrate resistant acid phosphatase (TRAP), matrix metalloproteinase 9 (MMP9) and cathepsin K (CatK), which are central to the bone destruction process [50].
RANK is elevated in PsA synovial tissue, and both IL-23 and IL-17 upregulate its expression [51],[52],[53]. IL-20, which is secreted by monocytes, keratinocytes and Th17 cells, and IL-20R are also expressed in PsA and RA synovia and have been shown to upregulate RANK and RANKL [54],[55],[56]. IL-20 signaling via IL-20R2/IL-20R1 and IL-22R1/IL-20R2 receptor complexes also promotes keratinocyte proliferation and differentiation in psoriatic skin [57], and it may constitute an alternative pathway whereby the IL-23/17 axis indirectly promotes PsA pathology.
2.4 NF-κB pathways
There are five members of the NF-κB transcription factor family, including RelA (p65), RelB, c-Rel, NF-κB1 (p105) and NF-κB2 (p100). Three main pathways mediated by NF-κB include: 1) the canonical, 2) the p105 and 3) the alternative (p100) pathway, as reviewed in [58]. In the canonical pathway, phosphorylation of inhibitory IκB proteins (IκBα) leads to release of NF-κB (p50/p65 heterodimers) and its nuclear translocation to promote inflammation and cell survival [58]. The p105 pathway is dependent on phosphorylation of p105 proteins, leading to nuclear translocation of p52 heterodimer complexes to promote inflammation. Unlike the canonical and p105 pathways, the alternative p100 pathway does not depend on the NF-κB essential modulator (NEMO)-IKKα-IKKβ (NEMO-IKK) complex for phosphorylation, but rather NF-κB inducing kinase (NIK) and IKKα heterodimers phosphorylate p100 and allow nuclear translocation of p52/RelB heterodimers [58].
Mice deficient in both nfkb1(p105) and nfkb2 (p100) demonstrate an absence of bone-resorbing osteoclasts [59]. In addition, p65 and RelB play important roles in osteoclast differentiation and survival [60],[61]. Immediate early genes associated with NF-κB activation include TRAF6, NIK, and Src [58]. TRAF proteins involved in the canonical and alternative NF-κB pathways also regulate osteoclast differentiation, associated with RANK signaling [62]. IL-17R/Act1/TRAF6 activates the NF-κB pathway (Fig. 1) and regulates bone resorption [63],[64].
Several studies have confirmed the presence of NF-κB activity in psoriatic (skin) disease (65),[65],[66]. However, in a human study, elevated active p65 was not reduced to normal baseline levels by TNF blockade, suggesting that TNF-independent NF-κB activity is present in psoriatic pathology [67]. A role for NF-κB pathways in psoriasis and PsA is also implied by findings from genome-wide association scans (GWAS). These analyses revealed several susceptibility genes associated with NF-κB, including TNF induced protein 3 (TNFAIP3), TNIP1 (ABIN-1), TRAF3IP2, NFκBIA and REL (c-Rel) [14],[68],[12],[69]. TNFAIP3 encodes an A20 cytoplasmic zinc finger protein, which induces degradation of NEMO to negatively regulate NF-κB [70]. Myeloid-specific A20 (TNFAIP3) deficient mice exhibit sustained NF-κB activity and enhanced osteoclastogenesis, and A20 deficiency in keratinocytes allows hyperkeratosis but not differentiated psoriasis [71]. Although mice heterozygous for NEMO show skin abnormalities, they do not exhibit psoriatic-like disease, thus the significance of NEMO in psoriasis is unclear [72]. Another gene linked to susceptibility to PsA is TNIP1 which encodes A20 binding and inhibitor of NF-κB-1 (ABIN-1). ABIN-1 expression is protective against psoriatic-like disease and regulates IL-17 and IL-22-producing T cells as well as IL-23 secretion from DCs [73].
Another susceptibility locus identified by GWAS is tumor necrosis factor receptor-associated factor 3-interacting protein Interacting Protein 2 (TRAF3IP2), which encodes Act1 [74]. Act1 deficient mice spontaneously develop skin inflammation and Act1-meditated signaling is required for the pathogenesis of CIA [75],[76]. Therefore Act1 contributes to both skin inflammation and bone destruction, suggesting a plausible role for IL-17 in PsA [75],[63].
3. Cellular mechanisms in PsA pathogenesis
3.1 Neutrophils
The induction of NF-κB activation by the IL-23/IL-17 axis results in the production of granulocyte-colony stimulating factor (G-CSF), granulocyte-macrophage stimulating factor (GM-CSF), and various chemokines (CXCL1, CXCL2, CXCL5 and CXCL8/IL-8) that in turn lead to the recruitment and migration of neutrophils [77],[78],[79]. IL-17 can stimulate endothelial expression of P-selectins, E-selectins and integrin ligands, including ICAM-1 and VCAM-1, to enhance neutrophil mobilization [80]. The presence of neutrophils in psoriasis typically correlates with increased IL-8 in psoriatic lesions, and IL-8 is also known to induce neutrophilia [41],[81]. Furthermore, GM-CSF promotes myelopoiesis to generate monocytes and neutrophils [82],[83]. Whether neutrophils respond directly to IL-17 remains controversial, as human neutrophils were recently shown to lack expression of IL-17RC [84].
A dense accumulation of neutrophils in the upper layers of psoriatic skin (stratum corneum) is commonly identified as Munro’s microabscess formation [85]. In mouse models of psoriatic-like disease, neutrophils are identified by their surface expression of CD11b and Gr-1high and neutrophil depletion using antibodies against Gr-1 reduces epidermal thickening and microabscess formation in flaky skin mice [86],[87]. However, although neutrophils have been recognized to play a role in psoriatic disease pathogenesis, these cells have not been well characterized. New evidence suggests that neutrophils have a much longer lifespan than previously appreciated and have novel roles in regulating the pool size and function of hematopoietic cells in the bone marrow [88],[89]. This suggests that neutrophils also may have the capacity to modulate hematopoietic precursors in tissues outside of the bone marrow microenvironment, potentially regulating cytokine-mediated inflammatory precursors in multiple organ systems during disease progression in inflammatory diseases, such as PsA. Specific subpopulations of neutrophils may initiate or perpetuate PsA pathology.
3.2 Osteoclasts
Myeloid-derived cells differentiate into osteoclasts in the presence of macrophage colony stimulating factor (M-CSF) and RANKL. RANK and colony stimulating factor 1 receptor (CSF-1R/c-fms) are both expressed on osteoclast precursor cells that, upon stimulation with RANKL and M-CSF, develop into mature bone-resorbing cells [90],[91]. In osteoclast precursors, activation of the RANKL-RANK pathway induces calcium oscillations that lead to calcineurin-mediated activation of NFATc1 [92]. NFATc1 regulates the expression of Oscar, Calcr, Itgb3, Rcan2, Myo1d, and Mst1r, and augments the expression of Mmp9, Acp5, Ctsk, Mmmp14, and Clcn7, genes required for bone destruction [92], [93]. Activator protein (AP-1) a transcriptional regulator of cfos, is also required for osteoclast differentiation and has been implicated in PsA [94]. Osteoclasts can be generated from RANKL, RANK or TRAF6 deficient mice-suggesting that RANKL-RANK-independent osteoclast differentiation pathways also exist [64].
The IL-23/IL-17 axis plays a critical role in osteoclastogenesis via a number of direct and indirect effects that both positively and negatively modulate osteoclast formation. Evidence that IL-23 negatively regulates osteoclastogenesis comes from in vitro observations indicating indirect inhibition via T cells [95],[96]. Conversely, IL-23-induced Th17 cell differentiation results in RANKL secretion and thus promotes osteoclastogenesis [97]. Yago and colleagues also demonstrated that IL-23 induces osteoclastogenesis via IL-17 in vitro [43]. In later experiments it was shown that IL-17 dose-dependently induced osteoclastogenesis in human PBMC in the absence of RANKL in a mechanism involving TNF [98]. Additionally, IL-17 upregulated CSF-1R and RANK and promoted osteoclast differentiation [53]. IL-17 also acts on osteoblasts to secrete RANKL to further enhance bone resorption [99]. IL-17 further modulates the expression of the osteoclast fusion protein, DC-STAMP (dendritic cell-specific transmembrane protein), a potential biomarker for early prognosis of PsA [100].
3.3 Cells in skin
Psoriatic skin disease is mediated primarily by immune cells and epidermal keratinocytes [101]. Various triggers of psoriatic disease have been suggested, including the formation of DNA and antimicrobial LL-37 complexes that promote activation of myeloid DCs and their migration to the draining lymph nodes, where they induce differentiation of naïve T cells into effector IL-17-producing cells [102]. Recently, innate lymphoid cells such as γδ T cells, an additional cellular source of IL-17, were shown to be elevated in psoriatic skin [103],[40]. Under homeostatic conditions, γδ T cells and other innate-like lymphoid cells constitutively express IL-23R and the chemokine receptor CCR6, which mediate their recruitment to the skin to interact with keratinocytes and other immune cells [104]. RORγt expressing innate-like lymphocytes and Vγ4+ γδ T cells were shown to initiate Aldara-induced psoriatic-like disease in mice [25]. It is thought that the secretion of IL-17 and IL-22 from IL-23-stimulated cells promotes aberrant keratinocyte differentiation and hyperproliferation, forming the basis of epidermal acanthosis or diffuse hyperplasia, hyperkeratosis, parakeratosis and Munro’s microabscess formation typically observed in PsA [105],[101]. These cytokines then activate keratinocytes via an autocrine or paracrine manner leading to secretion of antimicrobial peptides (e.g. LL-37, cathelcidin, β-defensins), chemokines (e.g. CXCL1, CXCL8), and S100 proteins (e.g. S100A7-9) to mediate inflammation in the skin [106] (Fig. 2). Psoriasis-like skin disease and arthritis was observed in mice with epidermal deletion of Jun proteins [107]. Interestingly, this model suggests that keratinocytes, through the JunB/AP-1 pathway, may underlie both psoriatic-like and arthritic features. However, subsequent reports showed that mRNA and protein levels of JunB are not reduced in human psoriatic plaques, questioning the significance of Jun proteins, at least in human psoriasis [108].
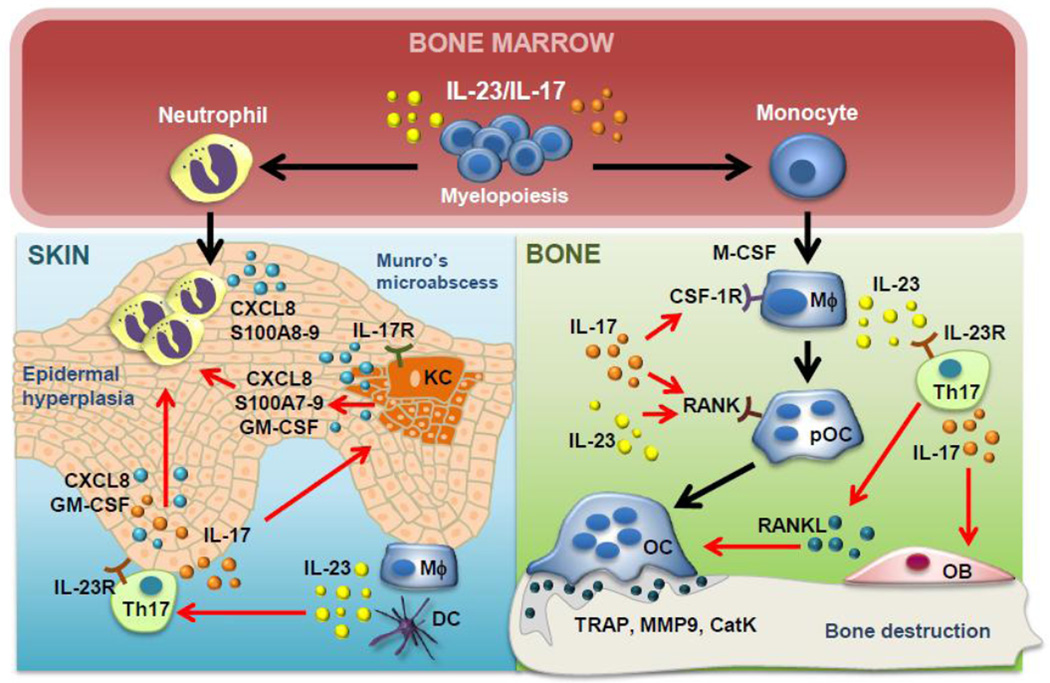
Fig. 2. Schematic of myeloid-derived cellular networks in IL-23/IL-17-mediated psoriatic arthritis.
IL-17 promotes myelopoiesis and induction of neutrophils and monocytes, leading to epidermal hyperplasia and bone destruction, respectively. In the skin, macrophages (Mϕ) and dendritic cells (DCs) secrete IL-23 to induce Th17 cell differentiation and secretion of IL-17, which activates keratinocytes (KC). Keratinocytes have also been shown to express IL-23R. Keratinocytes secrete S100 proteins (S100A7-9), GM-CSF and CXCL8 to recruit neutrophils to the skin. Th17 cells further promote neutrophil recruitment through the secretion of similar cytokines and chemokines. Neutrophils secrete CXCL8 and S100 (S100A8-9) proteins. Together, these cellular networks promote epidermal hyperplasia and Munro’s microabscess formation. In the bone, IL-23 has been shown to upregulate RANK on preosteoclasts (pOC). IL-23 secreted by macrophages induces IL-17 from Th17 cells which has been demonstrated to act on osteoblasts (OB) to secrete RANKL. Th17 cells have also been shown to secrete RANKL directly and further induce osteoclast formation and secretion of bone-degrading enzymes TRAP, CatK and MMP9 leading to bone destruction.
4. Conclusions
The role of IL-23 in inducing IL-17-producing T cells has been well studied; however, the importance of the IL-23/IL-17 axis in myeloid cell populations remains less understood. Nonetheless, sufficient progress has been made in understanding the role of the IL-23/IL-17 axis in psoriatic disease to support initiation of current clinical trials, which are evaluating the efficacy of IL-23 and IL-17 targeted therapy in psoriasis and PsA [109], [110]. More research is required to identify the cellular and molecular mechanisms of IL23/IL17 action, which are only partly known [111]. Elucidating the underlying molecular mechanisms behind these therapeutic targets will help provide insights into PsA disease pathogenesis that can be exploited for the design of more effective treatment strategies.
Take-home messages.
Single nucleotide polymorphisms in IL23R as well as TRAF3IP2, a target downstream of the IL-17 receptor (IL-17R), have linked the IL-23/IL-17 axis to PsA pathology.
The IL-23/IL-17 axis is a modulator of NF-κB activation in inflammation.
NF-κB activation is directly linked with osteoclastogenesis and epidermal hyperplasia, hallmark features of PsA.
Acknowledgements
We thank Thanh Nguyen for help with graphic design. Research reported in this publication was partly supported by the NIAMS/NIH AR62173 and Shriners Hospitals for Children SHC 250862 to IEA. ES is the recipient of a NCATS/NIH #UR1 TR000002 predoctoral fellowship. EDM is partly supported by NIH R01AR061297. FON is partly supported by: EU FP7 grant agreement HEALTH-F2-2011-261366 and the National Institute for Health Research (NIHR) Biomedical Research Centre based at Guy's and St Thomas' NHS Foundation Trust and King's College London. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, SHC, NHS, NIHR or the Department of Health. We apologize to colleagues for omissions imposed by space limitations.
Abbreviations
- IL-17
Interleukin-17A
- PsA
psoriatic arthritis
- NF-κB
nuclear factor kappa-light-chain-enhancer of activated B cells
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures
No conflict of interest disclosed.
References
- 1.Gladman DD, Antoni C, Mease P, Clegg DO, Nash P. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis. 2005;64(Suppl 2):ii14–ii17. doi: 10.1136/ard.2004.032482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nestle FO, Kaplan DH, Barker J. Psoriasis. N Engl J Med. 2009;361:496–509. doi: 10.1056/NEJMra0804595. [DOI] [PubMed] [Google Scholar]
- 3.Terui T, Ozawa M, Tagami H. Role of neutrophils in induction of acute inflammation in T-cell-mediated immune dermatosis, psoriasis: a neutrophil-associated inflammation-boosting loop. Exp Dermatol. 2000;9:1–10. doi: 10.1034/j.1600-0625.2000.009001001.x. [DOI] [PubMed] [Google Scholar]
- 4.McGonagle D, Lories RJ, Tan AL, Benjamin M. The concept of a "synovio-entheseal complex" and its implications for understanding joint inflammation and damage in psoriatic arthritis and beyond. Arthritis Rheum. 2007;56:2482–2491. doi: 10.1002/art.22758. [DOI] [PubMed] [Google Scholar]
- 5.D'Agostino MA, Said-Nahal R, Hacquard-Bouder C, Brasseur JL, Dougados M, Breban M. Assessment of peripheral enthesitis in the spondylarthropathies by ultrasonography combined with power Doppler: a cross-sectional study. Arthritis Rheum. 2003;48:523–533. doi: 10.1002/art.10812. [DOI] [PubMed] [Google Scholar]
- 6.Kruithof E, Baeten D, De Rycke L, Vandooren B, Foell D, Roth J, Canete JD, Boots AM, Veys EM, De Keyser F. Synovial histopathology of psoriatic arthritis, both oligo- and polyarticular, resembles spondyloarthropathy more than it does rheumatoid arthritis. Arthritis Res Ther. 2005;7:R569–R580. doi: 10.1186/ar1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Alenius GM, Berglin E, Rantapaa Dahlqvist S. Antibodies against cyclic citrullinated peptide (CCP) in psoriatic patients with or without joint inflammation. Ann Rheum Dis. 2006;65:398–400. doi: 10.1136/ard.2005.040998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johnson SR, Schentag CT, Gladman DD. Autoantibodies in biological agent naive patients with psoriatic arthritis. Ann Rheum Dis. 2005;64:770–772. doi: 10.1136/ard.2004.031286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dalbeth N, Pool B, Smith T, Callon KE, Lobo M, Taylor WJ, Jones PB, Cornish J, McQueen FM. Circulating mediators of bone remodeling in psoriatic arthritis: implications for disordered osteoclastogenesis and bone erosion. Arthritis Res Ther. 2010;12:R164. doi: 10.1186/ar3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kotake S, Yago T, Kawamoto M, Nanke Y. Role of osteoclasts and interleukin-17 in the pathogenesis of rheumatoid arthritis: crucial 'human osteoclastology'. J Bone Miner Metab. 2012;30:125–135. doi: 10.1007/s00774-011-0321-5. [DOI] [PubMed] [Google Scholar]
- 11.Chimenti MS, Ballanti E, Perricone C, Cipriani P, Giacomelli R, Perricone R. Immunomodulation in psoriatic arthritis: focus on cellular and molecular pathways. Autoimmunity reviews. 2013;12:599–606. doi: 10.1016/j.autrev.2012.10.002. [DOI] [PubMed] [Google Scholar]
- 12.Bowes J, Orozco G, Flynn E, Ho P, Brier R, Marzo-Ortega H, Coates L, McManus R, Ryan AW, Kane D, Korendowych E, McHugh N, FitzGerald O, Packham J, Morgan AW, Bruce IN, Barton A. Confirmation of TNIP1 and IL23A as susceptibility loci for psoriatic arthritis. Ann Rheum Dis. 2011;70:1641–1644. doi: 10.1136/ard.2011.150102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Filer C, Ho P, Smith RL, Griffiths C, Young HS, Worthington J, Bruce IN, Barton A. Investigation of association of the IL12B and IL23R genes with psoriatic arthritis. Arthritis Rheum. 2008;58:3705–3709. doi: 10.1002/art.24128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huffmeier U, Uebe S, Ekici AB, Bowes J, Giardina E, Korendowych E, Juneblad K, Apel M, McManus R, Ho P, Bruce IN, Ryan AW, Behrens F, Lascorz J, Bohm B, Traupe H, Lohmann J, Gieger C, Wichmann HE, Herold C, Steffens M, Klareskog L, Wienker TF, Fitzgerald O, Alenius GM, McHugh NJ, Novelli G, Burkhardt H, Barton A, Reis A. Common variants at TRAF3IP2 are associated with susceptibility to psoriatic arthritis and psoriasis. Nat Genet. 2010;42:996–999. doi: 10.1038/ng.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Oppmann B, Lesley R, Blom B, Timans JC, Xu Y, Hunte B, Vega F, Yu N, Wang J, Singh K, Zonin F, Vaisberg E, Churakova T, Liu M, Gorman D, Wagner J, Zurawski S, Liu Y, Abrams JS, Moore KW, Rennick D, de Waal-Malefyt R, Hannum C, Bazan JF, Kastelein RA. Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity. 2000;13:715–725. doi: 10.1016/s1074-7613(00)00070-4. [DOI] [PubMed] [Google Scholar]
- 16.Awasthi A, Riol-Blanco L, Jager A, Korn T, Pot C, Galileos G, Bettelli E, Kuchroo VK, Oukka M. Cutting edge: IL-23 receptor gfp reporter mice reveal distinct populations of IL-17-producing cells. J Immunol. 2009;182:5904–5908. doi: 10.4049/jimmunol.0900732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Parham C, Chirica M, Timans J, Vaisberg E, Travis M, Cheung J, Pflanz S, Zhang R, Singh KP, Vega F, To W, Wagner J, O'Farrell AM, McClanahan T, Zurawski S, Hannum C, Gorman D, Rennick DM, Kastelein RA, de Waal Malefyt R, Moore KW. A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rbeta1 and a novel cytokine receptor subunit, IL-23R. J Immunol. 2002;168:5699–5708. doi: 10.4049/jimmunol.168.11.5699. [DOI] [PubMed] [Google Scholar]
- 18.Ivanov, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, Cua DJ, Littman DR. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126:1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 19.Yang XO, Pappu BP, Nurieva R, Akimzhanov A, Kang HS, Chung Y, Ma L, Shah B, Panopoulos AD, Schluns KS, Watowich SS, Tian Q, Jetten AM, Dong C. T helper 17 lineage differentiation is programmed by orphan nuclear receptors ROR alpha and ROR gamma. Immunity. 2008;28:29–39. doi: 10.1016/j.immuni.2007.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen Z, Laurence A, Kanno Y, Pacher-Zavisin M, Zhu BM, Tato C, Yoshimura A, Hennighausen L, O'Shea JJ. Selective regulatory function of Socs3 in the formation of IL-17-secreting T cells. Proc Natl Acad Sci U S A. 2006;103:8137–8142. doi: 10.1073/pnas.0600666103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ruan Q, Kameswaran V, Zhang Y, Zheng S, Sun J, Wang J, DeVirgiliis J, Liou HC, Beg AA, Chen YH. The Th17 immune response is controlled by the Rel-RORgamma-RORgamma T transcriptional axis. J Exp Med. 2011;208:2321–2333. doi: 10.1084/jem.20110462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheung PF, Wong CK, Lam CW. Molecular mechanisms of cytokine and chemokine release from eosinophils activated by IL-17A, IL-17F, and IL-23: implication for Th17 lymphocytes-mediated allergic inflammation. J Immunol. 2008;180:5625–5635. doi: 10.4049/jimmunol.180.8.5625. [DOI] [PubMed] [Google Scholar]
- 23.Moseley TA, Haudenschild DR, Rose L, Reddi AH. Interleukin-17 family and IL-17 receptors. Cytokine & growth factor reviews. 2003;14:155–174. doi: 10.1016/s1359-6101(03)00002-9. [DOI] [PubMed] [Google Scholar]
- 24.Cua DJ, Tato CM. Innate IL-17-producing cells: the sentinels of the immune system. Nature reviews. Immunology. 2010;10:479–489. doi: 10.1038/nri2800. [DOI] [PubMed] [Google Scholar]
- 25.Pantelyushin S, Haak S, Ingold B, Kulig P, Heppner FL, Navarini AA, Becher B. Rorgammat+ innate lymphocytes and gammadelta T cells initiate psoriasiform plaque formation in mice. J Clin Invest. 2012;122:2252–2256. doi: 10.1172/JCI61862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gaffen SL. Structure and signalling in the IL-17 receptor family. Nature reviews. Immunology. 2009;9:556–567. doi: 10.1038/nri2586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Toy D, Kugler D, Wolfson M, Vanden Bos T, Gurgel J, Derry J, Tocker J, Peschon J. Cutting edge: interleukin 17 signals through a heteromeric receptor complex. J Immunol. 2006;177:36–39. doi: 10.4049/jimmunol.177.1.36. [DOI] [PubMed] [Google Scholar]
- 28.Novatchkova M, Leibbrandt A, Werzowa J, Neubuser A, Eisenhaber F. The STIR-domain superfamily in signal transduction, development and immunity. Trends Biochem Sci. 2003;28:226–229. doi: 10.1016/S0968-0004(03)00067-7. [DOI] [PubMed] [Google Scholar]
- 29.Bulek K, Liu C, Swaidani S, Wang L, Page RC, Gulen MF, Herjan T, Abbadi A, Qian W, Sun D, Lauer M, Hascall V, Misra S, Chance MR, Aronica M, Hamilton T, Li X. The inducible kinase IKKi is required for IL-17-dependent signaling associated with neutrophilia and pulmonary inflammation. Nature immunology. 2011;12:844–852. doi: 10.1038/ni.2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sonder SU, Saret S, Tang W, Sturdevant DE, Porcella SF, Siebenlist U. IL-17-induced NF-kappaB activation via CIKS/Act1: physiologic significance and signaling mechanisms. J Biol Chem. 2011;286:12881–12890. doi: 10.1074/jbc.M110.199547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fujioka S, Niu J, Schmidt C, Sclabas GM, Peng B, Uwagawa T, Li Z, Evans DB, Abbruzzese JL, Chiao PJ. NF-kappaB and AP-1 connection: mechanism of NF-kappaB-dependent regulation of AP-1 activity. Mol Cell Biol. 2004;24:7806–7819. doi: 10.1128/MCB.24.17.7806-7819.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu C, Qian W, Qian Y, Giltiay NV, Lu Y, Swaidani S, Misra S, Deng L, Chen ZJ, Li X. Act1, a U-box E3 ubiquitin ligase for IL-17 signaling. Sci Signal. 2009;2:ra63. doi: 10.1126/scisignal.2000382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Qu F, Gao H, Zhu S, Shi P, Zhang Y, Liu Y, Jallal B, Yao Y, Shi Y, Qian Y. TRAF6-dependent Act1 phosphorylation by the IkappaB kinase-related kinases suppresses interleukin-17-induced NF-kappaB activation. Mol Cell Biol. 2012;32:3925–3937. doi: 10.1128/MCB.00268-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Raychaudhuri SP, Raychaudhuri SK, Genovese MC. IL-17 receptor and its functional significance in psoriatic arthritis. Mol Cell Biochem. 2012;359:419–429. doi: 10.1007/s11010-011-1036-6. [DOI] [PubMed] [Google Scholar]
- 35.Mrabet D, Laadhar L, Sahli H, Zouari B, Haouet S, Makni S, Sellami S. Synovial fluid and serum levels of IL-17, IL-23, and CCL-20 in rheumatoid arthritis and psoriatic arthritis: a Tunisian cross-sectional study. Rheumatol Int. 2013;33:265–266. doi: 10.1007/s00296-011-2231-1. [DOI] [PubMed] [Google Scholar]
- 36.Tonel G, Conrad C, Laggner U, Di Meglio P, Grys K, McClanahan TK, Blumenschein WM, Qin JZ, Xin H, Oldham E, Kastelein R, Nickoloff BJ, Nestle FO. Cutting edge: A critical functional role for IL-23 in psoriasis. J Immunol. 2010;185:5688–5691. doi: 10.4049/jimmunol.1001538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wilson NJ, Boniface K, Chan JR, McKenzie BS, Blumenschein WM, Mattson JD, Basham B, Smith K, Chen T, Morel F, Lecron JC, Kastelein RA, Cua DJ, McClanahan TK, Bowman EP, de Waal Malefyt R. Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nature immunology. 2007;8:950–957. doi: 10.1038/ni1497. [DOI] [PubMed] [Google Scholar]
- 38.Chan JR, Blumenschein W, Murphy E, Diveu C, Wiekowski M, Abbondanzo S, Lucian L, Geissler R, Brodie S, Kimball AB, Gorman DM, Smith K, de Waal Malefyt R, Kastelein RA, McClanahan TK, Bowman EP. IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2-dependent mechanisms with implications for psoriasis pathogenesis. J Exp Med. 2006;203:2577–2587. doi: 10.1084/jem.20060244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rizzo HL, Kagami S, Phillips KG, Kurtz SE, Jacques SL, Blauvelt A. IL-23-mediated psoriasis-like epidermal hyperplasia is dependent on IL-17A. J Immunol. 2011;186:1495–1502. doi: 10.4049/jimmunol.1001001. [DOI] [PubMed] [Google Scholar]
- 40.Cai Y, Shen X, Ding C, Qi C, Li K, Li X, Jala VR, Zhang HG, Wang T, Zheng J, Yan J. Pivotal role of dermal IL-17-producing gammadelta T cells in skin inflammation. Immunity. 2011;35:596–610. doi: 10.1016/j.immuni.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nograles KE, Zaba LC, Guttman-Yassky E, Fuentes-Duculan J, Suarez-Farinas M, Cardinale I, Khatcherian A, Gonzalez J, Pierson KC, White TR, Pensabene C, Coats I, Novitskaya I, Lowes MA, Krueger JG. Th17 cytokines interleukin (IL)-17 and IL-22 modulate distinct inflammatory and keratinocyte-response pathways. The British journal of dermatology. 2008;159:1092–1102. doi: 10.1111/j.1365-2133.2008.08769.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Murphy CA, Langrish CL, Chen Y, Blumenschein W, McClanahan T, Kastelein RA, Sedgwick JD, Cua DJ. Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J Exp Med. 2003;198:1951–1957. doi: 10.1084/jem.20030896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yago T, Nanke Y, Kawamoto M, Furuya T, Kobashigawa T, Kamatani N, Kotake S. IL-23 induces human osteoclastogenesis via IL-17 in vitro, and anti-IL-23 antibody attenuates collagen-induced arthritis in rats. Arthritis Res Ther. 2007;9:R96. doi: 10.1186/ar2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Adamopoulos IE, Tessmer M, Chao CC, Adda S, Gorman D, Petro M, Chou CC, Pierce RH, Yao W, Lane NE, Laface D, Bowman EP. IL-23 is critical for induction of arthritis, osteoclast formation, and maintenance of bone mass. J Immunol. 2011;187:951–959. doi: 10.4049/jimmunol.1003986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lubberts E, Koenders MI, Oppers-Walgreen B, van den Bersselaar L, Coenen-de Roo CJ, Joosten LA, van den Berg WB. Treatment with a neutralizing anti-murine interleukin-17 antibody after the onset of collagen-induced arthritis reduces joint inflammation, cartilage destruction, and bone erosion. Arthritis Rheum. 2004;50:650–659. doi: 10.1002/art.20001. [DOI] [PubMed] [Google Scholar]
- 46.Nakae S, Nambu A, Sudo K, Iwakura Y. Suppression of immune induction of collagen-induced arthritis in IL-17-deficient mice. J Immunol. 2003;171:6173–6177. doi: 10.4049/jimmunol.171.11.6173. [DOI] [PubMed] [Google Scholar]
- 47.Ikeda T, Kasai M, Utsuyama M, Hirokawa K. Determination of three isoforms of the receptor activator of nuclear factor-kappaB ligand and their differential expression in bone and thymus. Endocrinology. 2001;142:1419–1426. doi: 10.1210/endo.142.4.8070. [DOI] [PubMed] [Google Scholar]
- 48.Theill LE, Boyle WJ, Penninger JM. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu Rev Immunol. 2002;20:795–823. doi: 10.1146/annurev.immunol.20.100301.064753. [DOI] [PubMed] [Google Scholar]
- 49.Yamashita T, Yao Z, Li F, Zhang Q, Badell IR, Schwarz EM, Takeshita S, Wagner EF, Noda M, Matsuo K, Xing L, Boyce BF. NF-kappaB p50 and p52 regulate receptor activator of NF-kappaB ligand (RANKL) and tumor necrosis factor-induced osteoclast precursor differentiation by activating c-Fos and NFATc1. J Biol Chem. 2007;282:18245–18253. doi: 10.1074/jbc.M610701200. [DOI] [PubMed] [Google Scholar]
- 50.Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–342. doi: 10.1038/nature01658. [DOI] [PubMed] [Google Scholar]
- 51.Ritchlin CT, Haas-Smith SA, Li P, Hicks DG, Schwarz EM. Mechanisms of TNF-alpha- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J Clin Invest. 2003;111:821–831. doi: 10.1172/JCI16069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chen L, Wei XQ, Evans B, Jiang W, Aeschlimann D. IL-23 promotes osteoclast formation by up-regulation of receptor activator of NF-kappaB (RANK) expression in myeloid precursor cells. Eur J Immunol. 2008;38:2845–2854. doi: 10.1002/eji.200838192. [DOI] [PubMed] [Google Scholar]
- 53.Adamopoulos IE, Chao CC, Geissler R, Laface D, Blumenschein W, Iwakura Y, McClanahan T, Bowman EP. Interleukin-17A upregulates receptor activator of NF-kappaB on osteoclast precursors. Arthritis Res Ther. 2010;12:R29. doi: 10.1186/ar2936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hsu YH, Li HH, Hsieh MY, Liu MF, Huang KY, Chin LS, Chen PC, Cheng HH, Chang MS. Function of interleukin-20 as a proinflammatory molecule in rheumatoid and experimental arthritis. Arthritis Rheum. 2006;54:2722–2733. doi: 10.1002/art.22039. [DOI] [PubMed] [Google Scholar]
- 55.Lebre MC, Jonckheere CL, Kraan MC, van Kuijk AW, Bos JD, de Rie M, Gerlag DM, Tak PP. Expression of IL-20 in synovium and lesional skin of patients with psoriatic arthritis: differential response to alefacept treatment. Arthritis Res Ther. 2012;14:R200. doi: 10.1186/ar4038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hsu YH, Chen WY, Chan CH, Wu CH, Sun ZJ, Chang MS. Anti-IL-20 monoclonal antibody inhibits the differentiation of osteoclasts and protects against osteoporotic bone loss. J Exp Med. 2011;208:1849–1861. doi: 10.1084/jem.20102234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Blumberg H, Conklin D, Xu WF, Grossmann A, Brender T, Carollo S, Eagan M, Foster D, Haldeman BA, Hammond A, Haugen H, Jelinek L, Kelly JD, Madden K, Maurer MF, Parrish-Novak J, Prunkard D, Sexson S, Sprecher C, Waggie K, West J, Whitmore TE, Yao L, Kuechle MK, Dale BA, Chandrasekher YA. Interleukin 20: discovery, receptor identification, and role in epidermal function. Cell. 2001;104:9–19. doi: 10.1016/s0092-8674(01)00187-8. [DOI] [PubMed] [Google Scholar]
- 58.Beinke S, Ley SC. Functions of NF-kappaB1 and NF-kappaB2 in immune cell biology. Biochem J. 2004;382:393–409. doi: 10.1042/BJ20040544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Iotsova V, Caamano J, Loy J, Yang Y, Lewin A, Bravo R. Osteopetrosis in mice lacking NF-kappaB1 and NF-kappaB2. Nat Med. 1997;3:1285–1289. doi: 10.1038/nm1197-1285. [DOI] [PubMed] [Google Scholar]
- 60.Vaira S, Alhawagri M, Anwisye I, Kitaura H, Faccio R, Novack DV. RelA/p65 promotes osteoclast differentiation by blocking a RANKL-induced apoptotic JNK pathway in mice. J Clin Invest. 2008;118:2088–2097. doi: 10.1172/JCI33392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Vaira S, Johnson T, Hirbe AC, Alhawagri M, Anwisye I, Sammut B, O'Neal J, Zou W, Weilbaecher KN, Faccio R, Novack DV. RelB is the NF-kappaB subunit downstream of NIK responsible for osteoclast differentiation. Proc Natl Acad Sci U S A. 2008;105:3897–3902. doi: 10.1073/pnas.0708576105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kadono Y, Okada F, Perchonock C, Jang HD, Lee SY, Kim N, Choi Y. Strength of TRAF6 signalling determines osteoclastogenesis. EMBO Rep. 2005;6:171–176. doi: 10.1038/sj.embor.7400345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.DeSelm CJ, Takahata Y, Warren J, Chappel JC, Khan T, Li X, Liu C, Choi Y, Kim YF, Zou W, Teitelbaum SL. IL-17 mediates estrogen-deficient osteoporosis in an Act1-dependent manner. J Cell Biochem. 2012;113:2895–2902. doi: 10.1002/jcb.24165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kim N, Kadono Y, Takami M, Lee J, Lee SH, Okada F, Kim JH, Kobayashi T, Odgren PR, Nakano H, Yeh WC, Lee SK, Lorenzo JA, Choi Y. Osteoclast differentiation independent of the TRANCE-RANK-TRAF6 axis. J Exp Med. 2005;202:589–595. doi: 10.1084/jem.20050978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Abdou AG, Hanout HM. Evaluation of survivin and NF-kappaB in psoriasis, an immunohistochemical study. J Cutan Pathol. 2008;35:445–451. doi: 10.1111/j.1600-0560.2007.00841.x. [DOI] [PubMed] [Google Scholar]
- 66.Moorchung N, Kulaar JS, Chatterjee M, Vasudevan B, Tripathi T, Dutta V. Role of NF-kappaB in the pathogenesis of psoriasis elucidated by its staining in skin biopsy specimens. Int J Dermatol. 2013 doi: 10.1111/ijd.12050. [DOI] [PubMed] [Google Scholar]
- 67.Lizzul PF, Aphale A, Malaviya R, Sun Y, Masud S, Dombrovskiy V, Gottlieb AB. Differential expression of phosphorylated NF-kappaB/RelA in normal and psoriatic epidermis and downregulation of NF-kappaB in response to treatment with etanercept. The Journal of investigative dermatology. 2005;124:1275–1283. doi: 10.1111/j.0022-202X.2005.23735.x. [DOI] [PubMed] [Google Scholar]
- 68.Stuart PE, Nair RP, Ellinghaus E, Ding J, Tejasvi T, Gudjonsson JE, Li Y, Weidinger S, Eberlein B, Gieger C, Wichmann HE, Kunz M, Ike R, Krueger GG, Bowcock AM, Mrowietz U, Lim HW, Voorhees JJ, Abecasis GR, Weichenthal M, Franke A, Rahman P, Gladman DD, Elder JT. Genome-wide association analysis identifies three psoriasis susceptibility loci. Nat Genet. 2010;42:1000–1004. doi: 10.1038/ng.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ellinghaus E, Stuart PE, Ellinghaus D, Nair RP, Debrus S, Raelson JV, Belouchi M, Tejasvi T, Li Y, Tsoi LC, Onken AT, Esko T, Metspalu A, Rahman P, Gladman DD, Bowcock AM, Helms C, Krueger GG, Koks S, Kingo K, Gieger C, Wichmann HE, Mrowietz U, Weidinger S, Schreiber S, Abecasis GR, Elder JT, Weichenthal M, Franke A. Genome-wide meta-analysis of psoriatic arthritis identifies susceptibility locus at REL. The Journal of investigative dermatology. 2012;132:1133–1140. doi: 10.1038/jid.2011.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Vereecke L, Beyaert R, van Loo G. The ubiquitin-editing enzyme A20 (TNFAIP3) is a central regulator of immunopathology. Trends Immunol. 2009;30:383–391. doi: 10.1016/j.it.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 71.Martin F, Dixit VM. A20 edits ubiquitin and autoimmune paradigms. Nat Genet. 2011;43:822–823. doi: 10.1038/ng.916. [DOI] [PubMed] [Google Scholar]
- 72.Tsuruta D. NF-kappaB links keratinocytes and lymphocytes in the pathogenesis of psoriasis. Recent Pat Inflamm Allergy Drug Discov. 2009;3:40–48. doi: 10.2174/187221309787158399. [DOI] [PubMed] [Google Scholar]
- 73.Callahan JA, Hammer GE, Agelides A, Duong BH, Oshima S, North J, Advincula R, Shifrin N, Truong HA, Paw J, Barrera J, Defranco A, Rosenblum MD, Malynn BA, Ma A. Cutting Edge: ABIN-1 Protects against Psoriasis by Restricting MyD88 Signals in Dendritic Cells. J Immunol. 2013;191:535–539. doi: 10.4049/jimmunol.1203335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chang SH, Park H, Dong C. Act1 adaptor protein is an immediate and essential signaling component of interleukin-17 receptor. J Biol Chem. 2006;281:35603–35607. doi: 10.1074/jbc.C600256200. [DOI] [PubMed] [Google Scholar]
- 75.Wang C, Wu L, Bulek K, Martin BN, Zepp JA, Kang Z, Liu C, Herjan T, Misra S, Carman JA, Gao J, Dongre A, Han S, Bunting KD, Ko JS, Xiao H, Kuchroo VK, Ouyang W, Li X. The psoriasis-associated D10N variant of the adaptor Act1 with impaired regulation by the molecular chaperone hsp90. Nature immunology. 2013;14:72–81. doi: 10.1038/ni.2479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pisitkun P, Claudio E, Ren N, Wang H, Siebenlist U. The adaptor protein CIKS/ACT1 is necessary for collagen-induced arthritis, and it contributes to the production of collagen-specific antibody. Arthritis Rheum. 2010;62:3334–3344. doi: 10.1002/art.27653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Laan M, Cui ZH, Hoshino H, Lotvall J, Sjostrand M, Gruenert DC, Skoogh BE, Linden A. Neutrophil recruitment by human IL-17 via C-X-C chemokine release in the airways. J Immunol. 1999;162:2347–2352. [PubMed] [Google Scholar]
- 78.Biasi D, Carletto A, Caramaschi P, Bellavite P, Maleknia T, Scambi C, Favalli N, Bambara LM. Neutrophil functions and IL-8 in psoriatic arthritis and in cutaneous psoriasis. Inflammation. 1998;22:533–543. doi: 10.1023/a:1022354212121. [DOI] [PubMed] [Google Scholar]
- 79.von Vietinghoff S, Ley K. Homeostatic regulation of blood neutrophil counts. J Immunol. 2008;181:5183–5188. doi: 10.4049/jimmunol.181.8.5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Griffin GK, Newton G, Tarrio ML, Bu DX, Maganto-Garcia E, Azcutia V, Alcaide P, Grabie N, Luscinskas FW, Croce KJ, Lichtman AH. IL-17 and TNF-alpha sustain neutrophil recruitment during inflammation through synergistic effects on endothelial activation. J Immunol. 2012;188:6287–6299. doi: 10.4049/jimmunol.1200385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Duan H, Koga T, Kohda F, Hara H, Urabe K, Furue M. Interleukin-8-positive neutrophils in psoriasis. J Dermatol Sci. 2001;26:119–124. doi: 10.1016/s0923-1811(00)00167-5. [DOI] [PubMed] [Google Scholar]
- 82.Liu B, Tan W, Barsoum A, Gu X, Chen K, Huang W, Ramsay A, Kolls JK, Schwarzenberger P. IL-17 is a potent synergistic factor with GM-CSF in mice in stimulating myelopoiesis, dendritic cell expansion, proliferation, and functional enhancement. Exp Hematol. 2010;38:877 e1–884 e1. doi: 10.1016/j.exphem.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 83.Stark MA, Huo Y, Burcin TL, Morris MA, Olson TS, Ley K. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity. 2005;22:285–294. doi: 10.1016/j.immuni.2005.01.011. [DOI] [PubMed] [Google Scholar]
- 84.Pelletier M, Maggi L, Micheletti A, Lazzeri E, Tamassia N, Costantini C, Cosmi L, Lunardi C, Annunziato F, Romagnani S, Cassatella MA. Evidence for a cross-talk between human neutrophils and Th17 cells. Blood. 2010;115:335–343. doi: 10.1182/blood-2009-04-216085. [DOI] [PubMed] [Google Scholar]
- 85.Steffen C. William John Munro and Munro's abscess, and Franz Kogoj and Kogoj's spongiform pustule. Am J Dermatopathol. 2002;24:364–368. doi: 10.1097/00000372-200208000-00016. [DOI] [PubMed] [Google Scholar]
- 86.van der Fits L, Mourits S, Voerman JS, Kant M, Boon L, Laman JD, Cornelissen F, Mus AM, Florencia E, Prens EP, Lubberts E. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol. 2009;182:5836–5845. doi: 10.4049/jimmunol.0802999. [DOI] [PubMed] [Google Scholar]
- 87.Schon M, Denzer D, Kubitza RC, Ruzicka T, Schon MP. Critical role of neutrophils for the generation of psoriasiform skin lesions in flaky skin mice. The Journal of investigative dermatology. 2000;114:976–983. doi: 10.1046/j.1523-1747.2000.00953.x. [DOI] [PubMed] [Google Scholar]
- 88.Beyrau M, Bodkin JV, Nourshargh S. Neutrophil heterogeneity in health and disease: a revitalized avenue in inflammation and immunity. Open Biol. 2012;2:120134. doi: 10.1098/rsob.120134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Casanova-Acebes M, Pitaval C, Weiss LA, Nombela-Arrieta C, Chevre R, N AG, Kunisaki Y, Zhang D, van Rooijen N, Silberstein LE, Weber C, Nagasawa T, Frenette PS, Castrillo A, Hidalgo A. Rhythmic Modulation of the Hematopoietic Niche through Neutrophil Clearance. Cell. 2013;153:1025–1035. doi: 10.1016/j.cell.2013.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tanaka S, Takahashi N, Udagawa N, Tamura T, Akatsu T, Stanley ER, Kurokawa T, Suda T. Macrophage Colony-Stimulating Factor Is Indispensable for Both Proliferation and Differentiation of Osteoclast Progenitors. J Clin Invest. 1993;91:257–263. doi: 10.1172/JCI116179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Nakagawa N, Kinosaki M, Yamaguchi K, Shima N, Yasuda H, Yano K, Morinaga T, Higashio K. RANK is the essential signaling receptor for osteoclast differentiation factor in osteoclastogenesis. Biochem Bioph Res Co. 1998;253:395–400. doi: 10.1006/bbrc.1998.9788. [DOI] [PubMed] [Google Scholar]
- 92.Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H, Saiura A, Isobe M, Yokochi T, Inoue J, Wagner EF, Mak TW, Kodama T, Taniguchi T. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell. 2002;3:889–901. doi: 10.1016/s1534-5807(02)00369-6. [DOI] [PubMed] [Google Scholar]
- 93.Aliprantis AO, Ueki Y, Sulyanto R, Park A, Sigrist KS, Sharma SM, Ostrowski MC, Olsen BR, Glimcher LH. NFATc1 in mice represses osteoprotegerin during osteoclastogenesis and dissociates systemic osteopenia from inflammation in cherubism. J Clin Invest. 2008;118:3775–3789. doi: 10.1172/JCI35711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zenz R, Eferl R, Scheinecker C, Redlich K, Smolen J, Schonthaler HB, Kenner L, Tschachler E, Wagner EF. Activator protein 1 (Fos/Jun) functions in inflammatory bone and skin disease. Arthritis Res Ther. 2008;10:201. doi: 10.1186/ar2338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kamiya S, Nakamura C, Fukawa T, Ono K, Ohwaki T, Yoshimoto T, Wada S. Effects of IL-23 and IL-27 on osteoblasts and osteoclasts: inhibitory effects on osteoclast differentiation. J Bone Miner Metab. 2007;25:277–285. doi: 10.1007/s00774-007-0766-8. [DOI] [PubMed] [Google Scholar]
- 96.Quinn JM, Sims NA, Saleh H, Mirosa D, Thompson K, Bouralexis S, Walker EC, Martin TJ, Gillespie MT. IL-23 inhibits osteoclastogenesis indirectly through lymphocytes and is required for the maintenance of bone mass in mice. J Immunol. 2008;181:5720–5729. doi: 10.4049/jimmunol.181.8.5720. [DOI] [PubMed] [Google Scholar]
- 97.Sato K, Suematsu A, Okamoto K, Yamaguchi A, Morishita Y, Kadono Y, Tanaka S, Kodama T, Akira S, Iwakura Y, Cua DJ, Takayanagi H. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J Exp Med. 2006;203:2673–2682. doi: 10.1084/jem.20061775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Yago T, Nanke Y, Ichikawa N, Kobashigawa T, Mogi M, Kamatani N, Kotake S. IL-17 induces osteoclastogenesis from human monocytes alone in the absence of osteoblasts, which is potently inhibited by anti-TNF-alpha antibody: a novel mechanism of osteoclastogenesis by IL-17. J Cell Biochem. 2009;108:947–955. doi: 10.1002/jcb.22326. [DOI] [PubMed] [Google Scholar]
- 99.Kotake S, Udagawa N, Takahashi N, Matsuzaki K, Itoh K, Ishiyama S, Saito S, Inoue K, Kamatani N, Gillespie MT, Martin TJ, Suda T. IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis. The Journal of clinical investigation. 1999;103:1345–1352. doi: 10.1172/JCI5703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Chiu YH, Mensah KA, Schwarz EM, Ju Y, Takahata M, Feng C, McMahon LA, Hicks DG, Panepento B, Keng PC, Ritchlin CT. Regulation of human osteoclast development by dendritic cell-specific transmembrane protein (DC-STAMP) . J Bone Miner Res. 2012;27:79–92. doi: 10.1002/jbmr.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lowes MA, Russell CB, Martin DA, Towne JE, Krueger JG. The IL-23/T17 pathogenic axis in psoriasis is amplified by keratinocyte responses. Trends Immunol. 2013;34:174–181. doi: 10.1016/j.it.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Di Meglio P, Nestle FO. The role of IL-23 in the immunopathogenesis of psoriasis. F1000 Biol Rep. 2010;2 doi: 10.3410/B2-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Laggner U, Di Meglio P, Perera GK, Hundhausen C, Lacy KE, Ali N, Smith CH, Hayday AC, Nickoloff BJ, Nestle FO. Identification of a novel proinflammatory human skin-homing Vgamma9Vdelta2 T cell subset with a potential role in psoriasis. J Immunol. 2011;187:2783–2793. doi: 10.4049/jimmunol.1100804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Guo L, Junttila IS, Paul WE. Cytokine-induced cytokine production by conventional and innate lymphoid cells. Trends Immunol. 2012;33:598–606. doi: 10.1016/j.it.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385–422. doi: 10.1146/annurev-pathol-011811-132448. [DOI] [PubMed] [Google Scholar]
- 106.Onishi RM, Gaffen SL. Interleukin-17 and its target genes: mechanisms of interleukin-17 function in disease. Immunology. 2010;129:311–321. doi: 10.1111/j.1365-2567.2009.03240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zenz R, Eferl R, Kenner L, Florin L, Hummerich L, Mehic D, Scheuch H, Angel P, Tschachler E, Wagner EF. Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature. 2005;437:369–375. doi: 10.1038/nature03963. [DOI] [PubMed] [Google Scholar]
- 108.Haider AS, Duculan J, Whynot JA, Krueger JG. Increased JunB mRNA and protein expression in psoriasis vulgaris lesions. The Journal of investigative dermatology. 2006;126:912–914. doi: 10.1038/sj.jid.5700183. [DOI] [PubMed] [Google Scholar]
- 109.McInnes IB, Sieper J, Braun J, Emery P, van der Heijde D, Isaacs JD, Dahmen G, Wollenhaupt J, Schulze-Koops H, Kogan J, Ma S, Schumacher MM, Bertolino AP, Hueber W, Tak PP. Efficacy and safety of secukinumab, a fully human anti-interleukin-17A monoclonal antibody, in patients with moderate-to-severe psoriatic arthritis: a 24-week, randomised, double-blind, placebo-controlled, phase II proof-of-concept trial. Ann Rheum Dis. 2013 doi: 10.1136/annrheumdis-2012-202646. [DOI] [PubMed] [Google Scholar]
- 110.Garber K. Anti-IL-17 mAbs herald new options in psoriasis. Nat Biotechnol. 2012;30:475–477. doi: 10.1038/nbt0612-475. [DOI] [PubMed] [Google Scholar]
- 111.Kupetsky EA, Mathers AR, Ferris LK. Anti-cytokine therapy in the treatment of psoriasis. Cytokine. 2013;61:704–712. doi: 10.1016/j.cyto.2012.12.027. [DOI] [PubMed] [Google Scholar]