Abstract
Background:
In Asia, Solanum nigrum fruit is traditionally used to manage, control, and treat diabetes.
Objective:
This study was carried out to investigate the endothelium and nitric oxide roles in Solanum nigrum-induced vasorelaxation in non-diabetic and diabetic rat vessels.
Materials and Methods:
Diabetes was induced by a single i.p. injection of streptozotocin. Eight weeks later, superior mesenteric arteries of non-diabetic and diabetic groups were isolated and perfused according to the McGregor method. Solanum nigrum fruit extract (SNE) at concentrations of 0.00001 to 0.6 mg/ml was added to the medium and perfusion pressure was recorded.
Results:
Baseline perfusion pressure of diabetic group was significantly higher than non-diabetic rats in both intact and denuded endothelium. The low concentrations of SNE have vasodilatory effect in the diabetic and non-diabetic, but high concentrations of SNE produce initial significant contractions, followed by secondary relaxations in normal and diabetic rats. We observed vasorelaxation at low and high concentrations of SNE in both diabetic and non-diabetic groups after endothelium denudation. SNE-induced vasorelaxation in diabetic group is mediated by both endothelium and smooth muscle, but the relaxatory effect of SNE in non-diabetic group is not mediated by endothelium, and SNE has direct action on the smooth muscle.
Conclusion:
Although the part of SNE-induced relaxation in diabetic vessel was mediated by endothelium, nitric oxide didn’t play any role in this action, and maybe we can use SNE in the management of diabetes vessel complications in future.
Keywords: Diabetes mellitus, mesenteric bed, nitric oxide, Solanum nigrum fruit extract vasorelaxation
INTRODUCTION
Vascular complications are important causes of morbidity and mortality in patients with diabetes. In diabetic patients, previous prospective study has shown an association between the degree of hyperglycemia and increased risk of microvascular complications.[1] Inhibition of nitric oxide and endothelial injury contribute to vascular complications.[2] Endothelial dysfunction is thought to be an early event in the atherosclerotic process and has been implicated in the pathogenesis of diabetic atherosclerotic vascular disease.[3] The search for appropriate hypoglycemic agents has recently been focused on plants, and many herbal medicines have been recommended for the treatment of diabetes.[4] Herbal drugs are frequently considered to be less toxic than their synthetic counterparts, and they have fewer side-effects.[5]
Solanum nigrum Linn. (Sn), commonly known as Black Nightshade, is a dicot weed in the Solanaceae family. Sn (Family: Solanacea) is a thorny shrub widely distributed in Sikkim, Uttar Pradesh, Southern India, Sri Lanka, and Iran in moist environment. Sn is an annual branched herb of up to 90 cm high, with dull dark green leaves, juicy, ovate or lanceolate, and toothless to slightly tooth on the margins. Flowers are small and white with a short pedicellate and five widely spread petals. Fruits are small, black when ripe.[6] Sn is found mainly around waste land, old fields, ditches, and roadsides, fence rows, or edges of woods and cultivated land. The fruit is a round fleshy berry up to 2 cm in diameter and yellowish when ripe. The seeds are brown and numerous. Sn is traditionally used for inflammatory, diuretic, and hepatic disorders.
In Asia, Solanum nigrum fruit is traditionally used to manage, control, and treat diabetes. In our previous study, we showed that Solanum nigrum fruit has anti-diabetic effect.[7] de Melo et al. have reported that the fruit of Solanum nigrum aqueous extract contains acetylcholine. The average content of acetylcholine was found to be 250 micrograms/g of fruit.[8] Nirmal et al. showed that S. nigrum berries inhibited clonidine-induced catalepsy significantly, and it can inhibit parameters linked to the asthma disease.[9] But, there is no more information about the effect of Solanum nigrum fruit on diabetic and normal vessel. This study was carried out to investigate the endothelium and nitric oxide roles in Solanum nigrum-induced vasorelaxation in diabetic rat vessel.
MATERIALS AND METHODS
Animals
Animals
The animals were handled in accordance with the criteria outlined in the (NIH publication #85-23, revised in 1985). Male rats (body weight 180-250 gr) were used. All animals were maintained at a constant temperature (23 ± 2°C) with a fixed 12:12-h light-dark cycle. Animals were divided into six groups (n = 6 in each group): Three diabetics and three non-diabetics kept in animal room for eight weeks. Animals were monitored for blood glucose concentrations and body weight every week. Blood glucose was measured with an Ascensia ELITE XL glucometer and Ascensia Elite blood glucose test strips.
Diabetes induction
Diabetes was produced with a single i.p. injection of STZ (60 mg/kg). Ten days after STZ injection, blood glucose levels were determined using a glucometer (Ascensia ELITE XL glucometer). Rats with blood glucose levels of 250 mg/dl were considered to be diabetic.
Preparation of Solanum nigrum fruit extracts
Fresh Solanum nigrum fruit were collected from open grassland in Kerman (Southern Iran). The plant was identified by the Taxonomist at the Shahid Beheshti University of Medical Sciences. A kilogram of fresh Solanum nigrum fruit was air-dried under shade at room temperature (26 ± 1°C) for 2 weeks. The dried fruit were then milled into a fine powder in a Waring commercial blender. The powdered fruit were extracted with deionized water followed by 15 minutes of boiling under continuous stirring. The extract was filtered through filter paper (Whatman No. 1). The dry weight in the extract was evaluated in samples dried at 80°C until constant weight. The percentage yields based on the dried starting material was 21% for dried aqueous. The concentrated extracts and fractions as stock were stored at -20°C in a desiccant before use. One gram of Solanum nigrum fruit extract contained 75.7 mg dried substance.
GC/MS analysis
GC/MS analysis of aqueous extract was performed on a mass spectrometer connected to a gas chromatography system (GC/MSD7890A, 7000 Triple Quad series Agilent), which was equipped with a HP-5MS capillary column (30 m × 0.25 mm; film thickness of 0.25 μm). Helium was used as carrier gas at a rate flow of 1 ml/min. The GC oven temperature was programmed from 50°C to 260°C at a rate of 10°/min and kept at 260°C for 26 min. MS was performed at 1 scan s-1 with ionizing voltage of 70 V and ion source temperature of 280°C. Separated compounds were identified by comparing their retention times with those of authentic standards injected under the same chromatographic conditions and by comparison of their retention indices and their mass spectra of the unknown peaks with the MS Library.
Preparation of mesenteric vascular bed
After eight weeks, all animals were anesthetized by i.p. injection of ketamine HCl 50 mg/kg, and the mesenteric vascular beds was prepared as originally described by McGregor.[10] In brief, abdominal wall was opened, superior mesenteric artery was exposed and cannulated, then gently flushed with modified Krebs Henseleit solution (containing in mM: NaCl: 118, KCl: 4.7, CaCl2: 2.5, MgSO4: 1.2, glucose: 2, NaHCO3: 2.5, NaHPO4: 1.2) concomitantly bubbled with a mixture of 95% O2 and 5% CO2 (final pH 7.4), and warmed to 37°C. The mesentery was isolated from the intestine, and placed in a water-jacked perfusion chamber maintained at 37°C. The preparation was perfused at 1 ml/min with modified Krebs Henseleit solution by a peristaltic pump (Meredos GmbH). The tissue was prevented from drying by superfusion with 0.1 ml/min modified Krebs Henseleit solution. Perfusion pressure was monitored via a T tube inserted between the pump and the inflow cannula. This was connected to a pressure transducer MLT0380 ADInstruments). A Power Lab System recorded the procedure (16SP, ADInstruments). After 30-minute equilibration, the vascular bed was constricted by Krebs-Henseleit solution containing phenylephrine, an α1 -adrenoceptor agonist, (0.001 M for intact and denuded diabetic groups and 0.003 M for intact and denuded non-diabetic groups) to induce 70-75% of maximal vasoconstriction (the doses of phenylephrine were chosen according to the phenylephrine dose response curve, data not shown in the results) then allowed to reach a plateau and stabilize. Solanum nigrum fruit extract at concentrations from 0.00001 to 0.6 mg/ml was added to the medium, and then perfusion pressure was recorded. Drug concentrations increased every 15 minutes.
Endothelial denudation
To achieve endothelial denudation, the preparation was perfused with distilled water for 5 minutes.[11]
Nitric oxide inhibition
To inhibit nitric oxide production, L-NAME, a non-selective nitric oxide synthase (NOS) inhibitor at a dose of 0.0001 M, was added to medium 20 min before phenylephrine administration. Then, phenylephrine concentration was adjusted to achieve 70-75% of maximum contractile response.
Drugs
The following drugs were used: STZ was obtained from Sigma (USA) and dissolved in 1 ml normal saline immediately before use. Phenylephrine and L-NAME (non specific NOS synthase inhibitor) were obtained from Sigma (St. Louis, MO, USA), ketamine HCl was obtained from Rotexmedica (Trittau, Germany).
Statistical analysis
Data were expressed as Mean ± SEM. Comparisons between groups were analyzed by two-way analysis of variance followed by Tukey test, using SPSS software. P <0.05 was considered significant.
RESULTS
No significant differences were found between groups before the intervention. Ten days after STZ injection, plasma glucose levels were significantly increased from 107.44 ± 6.93 to 489.33 ± 31.14 mg/dl. Eight weeks after diabetes induction, plasma glucose levels remained significantly elevated in diabetic rats.
Identification of compounds in the extract
RT-1)
RT-2)
RT-3)
RT-4) Hydroxylamine, O-(-2-methylpropyl)-
RT-5) 1,6:3,4-Dianhydro-2-deoxy-β-d-ribo-hexopyranose
RT-6) 13-Docosenamide,(z)-
RT-7) 1,2-Benzenedicarboxylic acid, diisooctyle ester.
Mesenteric bed response
Baseline perfusion pressure of diabetic group was significantly (P < 0.001) higher than non-diabetic one in both intact and denuded endothelium [Figure 1].
Figure 1.
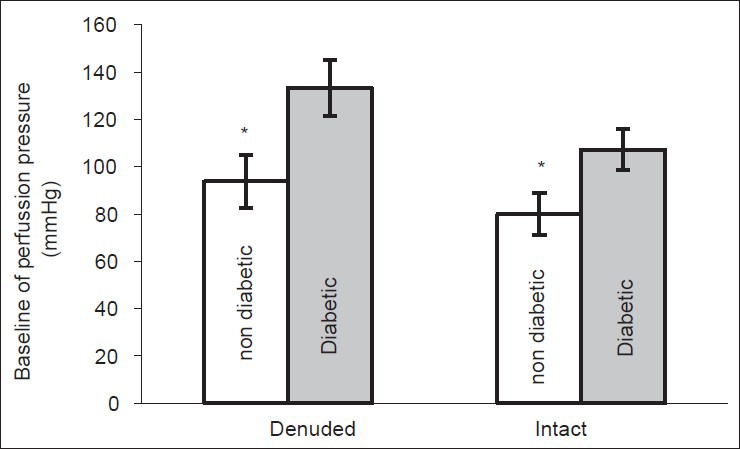
Baseline perfusion pressure (mmHg) of mesenteric vascular bed in non-diabetic and chronic diabetic group with intact and denuded endothelium (6 rats in each group. Data are expressed as mean ± SEM). Significant difference between non-diabetic and chronic diabetic groups (*P< 0.01, #P < 0.0001)
Solanum nigrum fruit extract (SNE) at doses 0.00001 to 0.02 mg/ml caused a significantly dose-depended decrease in perfusion pressure in non-diabetic (ND) (from 83.87 ± 1.26 to 80.98 ± 7.55) [Figure 2a] and chronic diabetic (CD) (from 143.74 ± 5 to 109.23 ± 6.94) [Figure 2b] groups with intact endothelium. But, SNE at dose 0.05 to 0.6 mg/ml initiated a significantly dose-depended vasoconstriction in both ND and CD groups with intact endothelium [Figures 2a and b]. The relaxatory effect of SNE in ND and CD groups with denuded endothelium was started at dose 0.00001 and reached to the steady state at 0.05 mg/ml dose in both ND and CD groups [Figures 2a and b].
Figure 2.
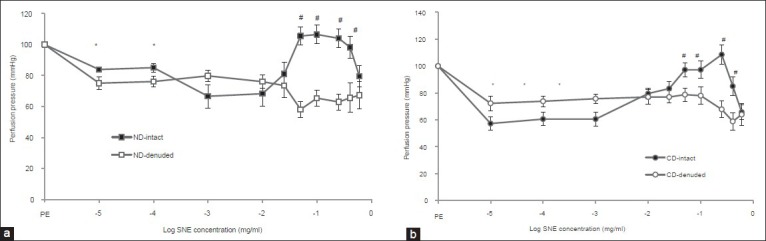
Dose response curves of Solanum nigrum fruit extract (SNE) in mesenteric vascular bed of (a) non-diabetic (ND) and (b) chronic diabetic (CD) animals with intact and denuded endothelium. (PE = phenylephrine, 6 rats in each group. Data were expressed as mean ± SEM). Significant difference between non-diabetic and chronic diabetic groups (*P< 0.01, #P < 0.0001)
In the presence of L-NAME (0.0001 M), SNE - induced relaxation in intact mesenteric beds of non-diabetic and diabetic animals was not suppressed and perfusion pressure in both groups decreased significantly [Figures 3a and b]. Significant differences were observed at SNE concentration of 0.00001 to 0.6 mg/ml with the presence and absence of L-NAME in each group as compared to phenylephrine response.
Figure 3.
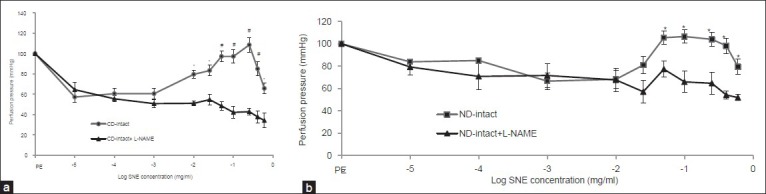
Dose response curve of Solanum nigrum fruit extract (SNE) in mesenteric vascular bed with intact endothelium in non-diabetic (CD) (a) and chronic diabetic (ND) (b) animals with the presence and absence of L-NAME (PE = phenylephrine, 6 rats in each group. Data expressed as Mean ± SEM). Significant difference between intact endothelium group with intact endothelium + L-NAME to Solanum nigrum fruit extract (SNE) (*P< 0.001, #P < 0.0001)
The results of the present study demonstrated that SNE-induced vasorelaxation at low concentrations in CD group with intact endothelium was significantly higher than ND animals with intact endothelium [Figure 4]. But, the vasoconstriction response of ND and CD groups with intact endothelium to high concentrations of SNE was not different [Figure 4].
Figure 4.
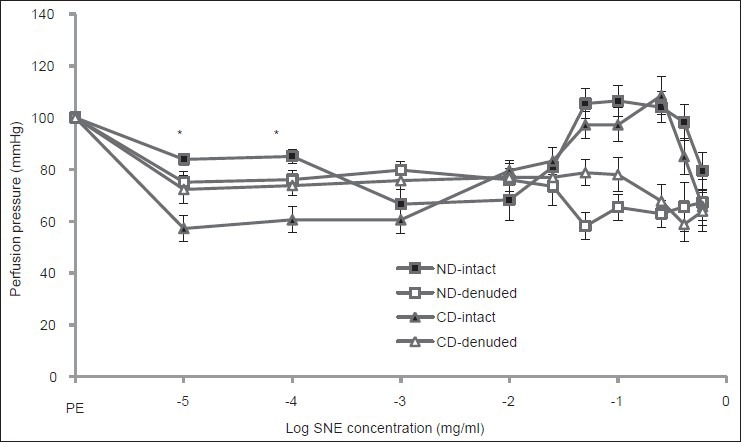
Dose response curve of Solanum nigrum fruit extract (SNE) in mesenteric vascular bed in non-diabetic (ND) and chronic diabetic (CD) animals with intact and denuded endothelium (PE = phenylephrine, 6 rats in each group. Data expressed as Mean ± SEM). Significant difference between chronic diabetic and non-diabetic with intact endothelium (P< 0.001)
DISCUSSION
This study was designed to investigate the endothelium and nitric oxide roles in SNE-induced vasorelaxation in diabetic and non-diabetic rat vessels. The results of the present study indicate that the SNE-induced vasorelaxation in non-diabetic and diabetic animals is mediated by different pathways. The part of this action in diabetic vessel is dependent to the endothelium, but it could not be mediated by nitric oxide system.
Vascular disease is one of the complicating features of diabetes mellitus. Some studies have showed that hypertension in diabetic patients is more than the normal population.[12] The hypertension is also considered an independent risk factor for cardiovascular mortality in patients with diabetes.[12] Our previous studies[13,14] showed that arthrosclerosis in mesenteric bed were performed in the diabetic rats eight weeks after diabetic induction and then the blood pressure increased.[15,16]
As we have expected, baseline perfusion pressure in intact and denuded endothelium of diabetic groups was higher than non-diabetics. This outcome is in agreement with endothelium dysfunction[15] and atherosclerosis in diabetes.[16]
Our findings in the present study indicated that low and high concentrations of SNE had different effects on the perfusion pressure of non-diabetic and diabetic mesentery bed vessels. Low concentrations of SNE have vasodilatory effect in the diabetic and non-diabetic subjects, and this effect of SNE in diabetic vessel is higher than non-diabetic ones. But, high concentrations of SNE produce initial significant contractions, followed by secondary, pronounced relaxations of Phenylephrine-contracted endothelium-intact mesenteric bed preparations in normal and diabetic rats. So, it seems that the endothelial cell produces not only vasodilators such as endothelium-derived relaxing factor (EDRF) and prostacyclin, but also vasoconstrictors such as thromboxane and endothelin (ET). Interestingly, after endothelium denudation, we observed vasorelaxation at low and high concentrations of SNE in both diabetic and non-diabetic groups. However, SNE-induced vasorelaxation in diabetic vessels after endothelium denudation was less than in intact endothelium group. But, SNE-induced vasorelaxation in non-diabetic group after endothelium denudation was higher than in intact endothelium vessels. So, it seems that SNE-induced vasorelaxation in diabetic group is mediated by both endothelium and smooth muscle, but the relaxatory effect of SNE in non-diabetic group is not mediated by endothelium, and SNE has direct action on the smooth muscle. Although the part of SNE-induced relaxation in diabetic vessel was mediated by endothelium, nitric oxide didn’t play any role in this action. Because SNE-induced vasorelaxation was not suppressed in presence of L-NAME, hence the other mechanism maybe involved in this response.
From the results of this study, it may be concluded that SNE-induced relaxation in non-diabetic vessel could not be mediated by endothelium and nitric oxide, but part of SNE-induced relaxation in diabetic vessel depends on the endothelium, but it is not mediated by NO. Our findings support the use of the low concentration of plant's fruit as a natural, adjunct phytomedicine in the management of diabetes vessel complications in future.
ACKNOWLEDGMENT
This research is supported by the Deputy of Research of Hormozgan University of Medical Sciences.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Jenkins AJ, Klein RL, Chassereau CN, Hermayer KL, Lopes-Virella MF. LDL from patient with well-controlled IDDM is not more susceptible to in vitro oxidation. Diabetes. 1996;45:762–7. doi: 10.2337/diab.45.6.762. [DOI] [PubMed] [Google Scholar]
- 2.Singleton JR, Smith AG, Russell JW, Feldman EL. Microvascular complications of impaired glucose tolerance. Diabetes. 2003;52:2867–73. doi: 10.2337/diabetes.52.12.2867. [DOI] [PubMed] [Google Scholar]
- 3.Järvisalo MJ, Raitakari M, Toikka JO, Putto-Laurila A, Rontu R, Laine S, et al. Endothelial Dysfunction and Increased Arterial Intima-Media Thickness in Children With Type 1 Diabetes. Circulation. 2004;109:1750–5. doi: 10.1161/01.CIR.0000124725.46165.2C. [DOI] [PubMed] [Google Scholar]
- 4.Marles RJ, Farnsworth NR. Antidiabetic plants and their active constituens. Phytomedicine. 1995;2:137–89. doi: 10.1016/S0944-7113(11)80059-0. [DOI] [PubMed] [Google Scholar]
- 5.Pari L, Umamaheswari J. Antihyperglycaemic activity of Musa Sapientum flowers: Effect on lipid peroxidation in alloxan diabetic rats. Phytother Res. 2000;14:136–8. doi: 10.1002/(sici)1099-1573(200003)14:2<136::aid-ptr607>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 6.Poongothai K, Syed Zameer Ahmed K, Ponmurugan P, Jayanthi M. Assessment of antidiabetic and antihyperlipidemic potential of Solanum nigrum and Musa paradisiaca in alloxan induced diabetic rats. J Pharmacy Res. 2010;3:2203–5. [Google Scholar]
- 7.Sohrabipour S, Kharazmi F, Soltani N, Kamalinejad M. Effect of the administration of Solanum nigrum fruit on blood glucose, lipid profiles, and sensitivity of the vascular mesenteric bed to phenylephrine in streptozotocin-induced diabetic rats. Med Sci Monit Basic Res. 2013;22:133–40. doi: 10.12659/MSMBR.883892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Melo AC, Perec CJ, Rubio MC. Acetylcholine-like activity in the fruit of the black nightshade (Solanaceae) Acta Physiol Lat Am. 1978;28:19–26. [PubMed] [Google Scholar]
- 9.Nirmal SA, Patel AP, Bhawar SB, Pattan SR. Antihistaminic and antiallergic actions of extracts of Solanum nigrum berries: Possible role in the treatment of asthma. J Ethnopharmacol. 2012;142:91–7. doi: 10.1016/j.jep.2012.04.019. [DOI] [PubMed] [Google Scholar]
- 10.McGregor DD. The effect of sympathetic nerve stimulation of vasoconstrictor responses in perfused mesenteric blood vessels of the rat. J Physiol. 1965;177:21–30. doi: 10.1113/jphysiol.1965.sp007572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wagner A, Varga K, Jarai Z, Kunos G. Mesenteric vasodilation mediated by endothelium anandamide receptors. Hypertension. 1999;33:429–34. doi: 10.1161/01.hyp.33.1.429. [DOI] [PubMed] [Google Scholar]
- 12.Ozcelikay AT, Tay A, Guner S, Tasyaran V, Yildizoglu-Ari N, Dincer UD, et al. Reversal effects of L-arginine treatment on blood pressure and vascular responsiveness of streptozotocin-diabetic rats. Pharmacol Res. 2000;41:201–9. doi: 10.1006/phrs.1999.0576. [DOI] [PubMed] [Google Scholar]
- 13.Farsi L, Keshavarz M, Soltani N. Relaxatory effect of GABA mediated by same pathway in diabetic and normal rat vessel. Iran Basic Med Sci. 2010;4:94–8. [Google Scholar]
- 14.Soltani N, Keshavarz M, Sohanaki H, Zahed Asl SZ, Dehpour AR. Relaxatory effect of magnesium on mesenteric vascular beds differs from normal and streptozotocin induced diabetic rats. Eur J Pharmacol. 2005;31:177–81. doi: 10.1016/j.ejphar.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 15.Laight DW, Carrier MJ, Anggard EE. Antioxidant, diabetes and endothelial dysfunction. Cardiovasc Res. 2000;47:457–64. doi: 10.1016/s0008-6363(00)00054-7. [DOI] [PubMed] [Google Scholar]
- 16.Diederich D, Skopec J, Diederich A, Dai FX. Endothelial dysfunction in mesenteric resistance arteries of diabetic rat: Role of free radical. Am J Physiol. 1994;266:H1153–61. doi: 10.1152/ajpheart.1994.266.3.H1153. [DOI] [PubMed] [Google Scholar]


