Abstract
Human mesenchymal stem cells (MSCs) are a rare population of non-hematopoietic stem cells with multilineage potential, originally identified in the bone marrow. Due to the lack of a single specific marker, MSCs can be recognized and isolated by a series of features such as plastic adherence, a panel of surface markers, the clonogenic and the differentiation abilities. The recognized role of MSCs in the regulation of hemopoiesis, in cell-degeneration protection and in the homeostasis of mesodermal tissues through their differentiation properties, justifies the current interest in identifying the biochemical signals produced by MSCs and their active crosstalk in tissue environments. Only recently have extracellular nucleotides (eNTPs) and their metabolites been included among the molecular signals produced by MSCs. These molecules are active on both ionotropic and metabotropic receptors present in most cell types. MSCs possess a significant display of these receptors and of nucleotide processing ectoenzymes on their plasma membrane. Thus, from their niche, MSCs give a significant contribution to the complex signaling network of eNTPs and its derivatives. Recent studies have demonstrated the multifaceted aspects of eNTP metabolism and their signal transduction in MSCs and revealed important roles in specifying differentiation lineages and modulating MSC physiology and communication with other cells. This review discusses the roles of eNTPs, their receptors and ectoenzymes, and the relevance of the signaling network and MSC functions, and also focuses on the importance of this emerging area of interest for future MSC-based cell therapies.
Keywords: Mesenchymal stem cell, Purinergic receptors, Ectoenzymes, ATP, β-NAD, Adenosine, cADPR
Core tip: The multifaceted aspects of extracellular nucleotide metabolism (mainly ATP and β-NAD) on mesenchymal stem cell (MSC) surface has been addressed by basic researchers only recently, sometimes revealing unexpected pivotal roles for these molecules in specifying differentiation lineages and modulating MSC physiology and communication with other cells. This review discusses the roles of extracellular nucleotides, their receptors and ectoenzymes, and the relevance of their signaling network and MSC functions, and also focuses on the importance of this emerging area of interest for future MSC-based cell therapies.
INTRODUCTION
Human mesenchymal stem cells (MSCs, also known as marrow stromal cells) are a rare population of non-hematopoietic stem cells with multilineage potential originally identified in the bone marrow (BM)[1,2]. BM-derived MSCs (BM-MSCs) are still considered the gold standard for MSC applications; nevertheless, the BM has several limitations as a source of MSCs, such as low frequency in this compartment, a painful isolation procedure and the loss of differentiation potential with donor’s increasing age. Thus, there is growing interest in identifying alternative sources for MSCs. To this end, MSCs obtained from the adipose tissue[3], dental pulp[4], placenta and Wharton’s jelly[5] have gained much attention in recent times since they can be easily isolated from tissues without any ethical concerns and which would be otherwise discarded.
Due to the lack of a single specific marker, MSCs can be recognized and isolated by a series of features such as plastic adherence, a panel of surface markers, the clonogenic and differentiation abilities[2,6,7]. They can be expanded in vitro for several passages without losing their lineage properties and are commonly considered the precursors of mesodermal cell types such as osteocytes, adipocytes and chondrocytes. Whether MSCs can differentiate to non-mesodermal cell types such as hepatocytes or neurons is still under debate[8-10].
In the BM, MSCs play a key role in providing hemopoietic progenitors (HPs) with soluble factors essential to their proliferation and differentiation[11]. Furthermore, MSCs possess immunoregulatory functions[12]. Actually, a number of clinical trials are currently exploring the use of MSCs in cell-based therapies of various pathological conditions, such as graft vs host disease, renal, neurological and cardiovascular diseases[13,14]. The clinical benefit of MSC-based cell therapy seems mostly related to MSC-derived soluble factors possessing immunomodulating, growth-supporting and/or antiapoptotic activities, as demonstrated on animal models[12]. Furthermore, their differentiation and tissue regeneration potential have already been used in therapeutic clinical approaches involving tissue engineering and gene therapy[15,16].
In vitro differentiation of MSCs requires the activation of specific transcription factors, regulatory genes and signal cascades[17,18]. Adipogenesis induction gives rise to preadipocytes with cytoplasmic accumulation of lipid droplets and release of adipokines and extracellular matrix-associated proteins[19]. On the other hand, osteogenesis-induced osteoblasts secrete mineralized extracellular matrix, with high levels of calcium phosphate forming hydroxyapatite crystals[20]. Since both osteoblasts and adipocytes originate from a common MSC precursor, it seems obvious that osteoblast and adipocyte differentiation pathways are regulated jointly[21].
Although a plethora of studies[22-24] have shown that many substances, as well as mechanical agents, are causally related to these differentiation processes, the mechanisms involved are not yet completely defined. However, a large body of evidence supports the idea that there is an inverse relationship between the differentiation of MSCs to osteoblasts or to adipocytes, i.e., conditions favoring the differentiation towards one lineage impair the differentiation to the other lineage. This seems to occur during attainment of peak bone mass[25,26] for instance, when adipogenesis in the BM is inhibited, favoring osteogenesis, or in aging population[27], when the BM adipocytes are predominant in respect to other cells of mesodermal origin.
MSCs regulate their fate through the complex integration of autocrine and paracrine extracellular signals (i.e., hormones, cytokines, nucleotides, xenobiotics) enabling the cells to sense the external milieu and to establish a fine communication with the surrounding cell population. Hence, they calibrate their response (differentiation, immunomodulation, proliferation, migration) on the basis of the necessities of the tissue in which they reside or on the organism’s physiopathological conditions.
From an evolutionary point of view, nucleotides are considered among the most ancient molecules with biological activity and they are in fact used by living organisms for many different purposes: energy metabolism, storage of genetic information, signal transduction and extracellular communication. Nucleotides can be released or leaked into the extracellular milieu by virtually every cell in the body. Extracellular nucleotides (eNTPs) comprise both extracellular purines (ATP, ADP, β-NAD, ADPR and cADPR) and extracellular pyrimidines (UTP and UDP). Once outside the cell, they either serve as signaling molecules by binding specific P2 purinergic receptors (P2X or P2Y) or are converted into other active nucleotides[28] and finally degraded to the related nucleosides. Nucleosides, mainly adenosine, can then bind different types of P1 purinergic receptors[29]. Nucleotide extracellular metabolism is mediated by special proteins located on the outer surface of the plasma membrane that possess an enzymatic domain in the extracellular region, called ectoenzymes[30]. Currently, there is an accumulating body of evidence indicating that the various ectoenzymes work in concert to dismantle eNTPs. Thus, in whatsoever milieu, the balance between nucleotides and nucleosides relies on the direct outflow of such molecules from transporters and channels in the plasma membrane[31-33], as well as on the activity of the specific ectoenzymes present on the cell surface.
It is now well established that eNTPs mediate intercellular communication in virtually all tissues. They are one of the most important indicators of cell stress in the pericellular environment[34] and the network of extracellular nucleotides/nucleosides serves multiple functions in a balanced and finely tuned fashion[35-37].
MSCs possess a significant display of purinergic receptors and ectoenzymes on their plasma membrane[38-40] and these cells have been reported to actively release nucleotides such as ATP and β-NAD upon certain stimuli[39-42] (Figure 1). Thus, from their niche, these cell types give a significant contribution to the complex network of signaling involving eNTPs and its derivatives, and accumulating literature indicates that MSC functions are also autocrinally influenced by eNTPs affecting their differentiation properties as well as their immunomodulatory activity.
Figure 1.
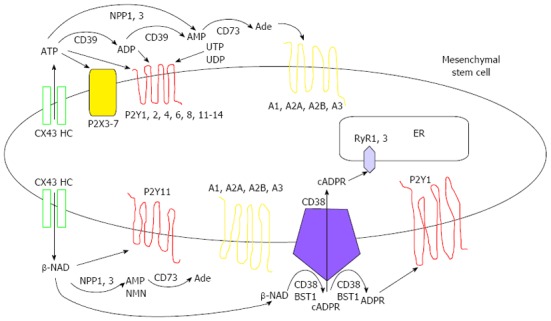
Surface network of purinergic receptors and nucleotide ectoenzymes on mesenchymal stem cells. On the basis of the recent findings, all the purinergic receptors and ectoenzymes whose presence has been ascertained on mesenchymal stem cells through qPCR analyses and/or demonstration of a clear physiological function (see text for references) are shown. Furthermore, both ATP and β-NAD stimulation mechanisms and metabolisms are summarized as an example of the finely tuned extracellular balance between nucleotides and nucleosides and their pleiotropic effects. CX43 HC: CX43 hemichannels; Ade: Adenosine; NMP: Nicotinamide monophosphate; ER: Endoplasmic reticulum; RyR1,3: Ryanodine receptors 1 and 3.
Here, the role of eNTPs, its receptors and converting ectoenzymes and the relevance of this signaling network in MSC functions are discussed, also focusing on the importance of this emerging area of interest for future MSC-based cell therapies.
P1 RECEPTORS IN MSC
Purinergic receptors (PRs) are plasma membrane receptors specific for adenosine, purine and pyrimidine nucleotides, which are expressed throughout the mammalian organism in all cell types. Upon their physiological agonist, Ps can be classified into P1 receptors, whose natural ligand is adenosine, and P2 receptors, whose recognized natural ligands are nucleotides (mainly ATP and UTP, see Figure 1)[29]. The adenosine receptors are G protein-coupled seven-transmembrane proteins, further classified into the A1R, A2AR, A2BR and A3R subtypes[29]. In particular, the P1 signaling pathway involves cyclic adenosine monophosphate (cAMP) synthesis upon A2AR and A2BR activation, or cAMP inhibition upon A1R and A3R activation[29].
Adenosine can be directly released by cells[31,32] or generated by the dephosphorylation of adenine nucleotides, which in many tissues are dephosphorylated to AMP by the ectonucleoside triphosphate phosphohydrolase (CD39). AMP is then further dephosphorylated to adenosine by ecto-5’-nucleotidase (CD73)[30]. The resulting adenosine has an essential role in the attenuation of inflammation and in damaged tissue healing. Furthermore, it mediates diverse cardioprotective, neuroprotective, vasodilatatory and angiogenic responses[43-46], in many cases counteracting the ATP inflammatory/stress signal triggered by P2 purinergic receptor activation.
Several studies in the last decade have established the presence of both P1 and P2 receptor family members on MSC surface (Figure 1), trying to elucidate their role in the homeostasis and differentiation properties of this cell type both in vitro and in vivo.
Adenosine receptor presence and function on MSC surface was first evidenced by Evans and coworkers[47], demonstrating the formation of extracellular adenosine by an osteoprogenitor cell line and by MSCs for the first time. On that occasion, the presence of all four adenosine receptor subtypes, especially A2bR, was ascertained, demonstrating a causal role of their activation in active secretion of the inflammatory cytokine IL-6 and of the osteoclastogenesis inhibitory factor osteoprotegerin. These data indicate that adenosine production, as well as its activity through adenosine receptors, could be a potential target for pharmacological interventions in the bone for many diseases, including osteoporosis[48].
A further study[49] demonstrated that adenosine signaling affects proliferation and development of BM-MSCs. Perhaps the most significant finding of this work is the demonstration that adenosine A2AR deletion or blockade diminishes the number of colony-forming unit-fibroblasts (CFU-F) in cultured BM-MSCs. Thus, the authors speculated that adenosine, targeting the A2AR, could increase the proliferation of MSCs, as also reported for other cell types[50,51]. Alternatively, they suggest that since A2AR stimulation has been shown to diminish apoptosis in other cell types[52,53], an increased survival of MSCs could enhance CFU-F yield from freshly isolated adult stem cells. Interestingly, they confirmed that A2AR and CD73 are coordinately regulated in MSCs as in other cell types[54], strengthening the idea of an active crosstalk in adenosine signaling between the adenosine receptor and the ectoenzymes able to generate the nucleoside in the pericellular space.
More recently, both in vitro[55] and in vivo[56,57] studies have evaluated the contribution of adenosine signaling in MSC differentiation. Gharibi et al[55] in particular investigated the in vitro expression of adenosine receptor subtypes and the adenosine metabolism as they differentiated MSCs into osteoblasts or adipocytes. They found differential expression of the adenosine receptor subtypes during differentiation as well as in mature cells. Differential expression was related both to the progression of lineage specificity (A2BR dominant in osteoblast differentiation; A1R and A2AR in adipogenic differentiation) and to the maintenance of specialized features in the two lineages (A2AR essential to ALP expression in osteoblasts; A1R involved in lipogenic activity in adipocytes).
These data suggest that useful strategies could include the targeting of the adenosine signaling pathway in cases of diseases associated with an imbalance in the differentiation and function of these two lineages. This research will be useful in preventing or treating conditions with insufficient bone or excessive adipocyte formation[25-27].
Finally, an essential role of adenosine signaling through A2BR in in vivo osteoblast differentiation and bone formation seems to be definitely confirmed in recent reports[56,57]. Both studies suggest that the pharmacological stimulation of this signaling pathway may enhance bone density and bone fracture healing in variously compromised situations, such as non-healing fractures in osteoporosis[56] and osteolytic bone lesions in multiple myeloma[57]. In general, all the above-mentioned studies confirm an essential, functional role of extracellular adenosine and its signaling pathway in MSC physiology, homeostasis and intervention in bone and adipose tissue reconstitution, allowing the identification of new pharmacological targets.
P2 RECEPTORS IN MSC
Extracellular nucleotides have been definitely recognized as autocrine/paracrine signaling molecules[58] released from cells in response to physiological and pathological stimulation, such as mechanical stress, hypoxia, inflammation and other agonists. The mechanisms of nucleotide release comprise exocytosis, ATP-binding cassette transporters, connexin hemichannels and voltage-dependent anion channels[33]. Many signaling roles for nucleotides have been demonstrated in several tissues, including: neurotransmission[33]; rhythm regulation in the myocardium[59]; gastrointestinal and liver function[60], regulation of epithelial cell responses[61]; blood flow distribution, oxygen delivery and endothelial barrier integrity[62,63]; immune responses[43,64]; and activation of platelets at sites of vascular injury[65]. Besides acute signaling events, there is increasing evidence that purines and pyrimidines also have potent long-term roles in cell proliferation and growth[34], induction of apoptosis and anticancer activity[43] and atherosclerotic plaque formation[66]. These effects are mediated by extracellular stimulation of P2 purinergic receptors, of which two major subfamilies, P2X and P2Y, have been described. The ionotropic P2X receptors are ligand-gated channels that gate extracellular cations in response to ATP and comprise seven receptor subtypes (P2X1-P2X7)[29]. Conversely, the metabotropic P2Y receptors are G-protein-coupled proteins that alternatively couple to Gq (P2Y1-2, P2Y4, P2Y6 and P2Y11) and therefore activate phospholipase C-β, or to Gi (P2Y12-14), that inhibit adenylyl cyclase and regulate ion channels[29]. Notably, P2Y11 receptor is dually coupled to phospholipase C and adenylyl cyclase stimulation.
P2Y receptors can be divided into: (1) adenine nucleotide-preferring receptors, mainly responding to ATP and ADP (P2Y1, P2Y11-13); (2) uracil nucleotide-preferring receptors (P2Y4 and P2Y6) responding to both UTP and UDP; (3) receptors of mixed selectivity (P2Y2); and (4) nucleotide sugar-preferring P2Y14 receptor responding to UDP-glucose and UDP-galactose[29]. Finally, the P2Y1 and P2Y11 receptors have also been described as β-NAD receptors with diverse functional activities[64,67,68]. In particular, P2Y1 is also a receptor for ADPR, a β-NAD metabolite generated by the cycling/hydrolyzing activity of CD38 and BST1/CD157 ectoenzymes[36,68].
P2 receptors and the related activating nucleotides have been the object of investigation in relation to MSC functions (Figure 1) only recently. In earlier reports[41,42], the spontaneous release of ATP from MSCs via gap junction hemichannels was assessed, on one occasion demonstrating a direct stimulation of P2Y1 receptor triggering intracellular Ca2+ oscillations[41], while showing the concurrent activation of P2X and P2Y receptors by ATP in another, resulting in a modulation of the proliferation rate at early passages of MSC cultivation[42].
The presence of the G-protein coupled P2Y2 receptor has also recently been demonstrated on rat MSCs, as well as its activation by the preferred agonist UTP inducing intracellular Ca2+ oscillations or elevating Ca2+ levels depending on cell density, and suggesting that these different Ca2+ responses in MSCs may be correlated with cell cycle progression[69].
More recently, different investigations have been directed to the pleiotropic effects of P2 receptor activation by ATP, focusing on MSC functionality in the hematopoietic niche and on the differentiation properties of these cells[70-73]. In a recent paper analyzing the effects of ATP on MSC functions, Ferrari and collaborators[70] observed a downregulation of genes related to cell proliferation and anti-inflammatory cytokines and concurrently an upregulation of pro-inflammatory cytokines and cell migration related genes. These data confirm the in vitro inhibitory activity of ATP on MSC proliferation, as already observed in a previous work[42], and demonstrate an in vivo potentiated homing capacity to the BM of ATP-pretreated MSCs that could be useful in supporting therapies for BM engraftment.
The role of ATP during MSC differentiation has also been addressed in the last years[38,71-73]. The related studies indicate that: (1) a variety of metabolically active P2X (P2X3-7) and P2Y (all subtypes) receptors are detectable in MSCs (Figure 1) and are up- or downregulated during adipogenic and osteogenic differentiation. In particular, P2Y4 and P2Y14 seem to be important for the onset of MSC commitment (regulated both in adipogenic and in osteogenic differentiation), P2Y1 and P2Y2 are downregulated in osteogenic differentiation, while P2Y11 is significantly upregulated in adipogenic commitment[38]; (2) significant ATP release by MSCs, especially observed during shockwave treatment, is able to promote osteogenic differentiation through P2X7 receptor activation with a significant positive impact in bone healing[71]; and (3) ATP treatment modulates the expression of several genes governing adipogenic and osteogenic differentiation of MSCs which can be tuned from one lineage to the other by specific culture conditions in the presence of this nucleotide[72]. In addition, evidence from Ciciarello and coworkers[72] seems to indicate that ATP is able to promote adipogenesis through its triphosphate form, while osteogenic differentiation seems to be induced by its nucleoside adenosine, as also proposed by others[55-57], resulting from ATP degradation by the CD39/CD73 system or directly released by cells. Thus, based on these findings, it is proposed that adipogenic differentiation is mainly mediated by activation of P2Y1 and P2Y4 receptors, while stimulation of the adenosine receptor subtype A2BR is involved in osteogenic differentiation. In another recent investigation, P2Y13 receptor has been implicated in in vivo osteogenic differentiation through the study of impaired bone turnover in a P2Y13-KO mouse model[73]. In this study, P2Y13 activation and consequent osteogenic induction, at the expenses of adipocyte differentiation, seems to be orchestrated by ADP stimulation and not ATP, thus complicating the picture of nucleotide involvement in the MSC differentiation process.
Together, all these data provide new insights into the molecular regulation of MSC differentiation and demonstrate the necessity to further deepen this topic of investigation in order to better understand the pleiotropic effects of ATP and its derivatives on MSC differentiating abilities and to finally merge current, sometimes contrasting, observations.
Besides ATP and its derivatives, the dinucleotide β-NAD has also been shown to activate P2 receptors (P2Y1 and P2Y11), its effects mainly investigated in cell types of the immune system and in neuromuscular transmission[64,67,68]. Interestingly, it has been recently demonstrated that this nucleotide also has a significant impact on MSC functions[39]. In particular, β-NAD can be released in the extracellular milieu upon stimuli able to open CX43 hemichannels in MSCs (i.e., low extracellular calcium, shear stress, inflammatory stimuli) and this release is functional to increase MSC proliferation, migration and production of immunomodulatory cytokines without compromising the differentiation abilities of these cells. Such effects are observable in MSCs in the presence of β-NAD, both extracellularly added or autocrinally released, and are dependent on P2Y11 activation (Figure 1). Thus, as for adenosine and its preferential receptors, β-NAD through its specific P2Y11 target can also exert a beneficial role in modulating cell protective functions relevant to MSC-based cell therapies.
NUCLEOTIDE-DEGRADING ECTOENZYMES IN MSC
Ectoenzymes are a family of cell surface molecules whose catalytic domain lies in the extracellular region. A subset of this family, the nucleotide-metabolizing ectoenzymes, are key components in the regulation of the extracellular balance between nucleotides and nucleosides, together with equilibrative transporters and channels enabling direct outflow of these molecules[31-33].
Following the signal transduction, eNTPs need to be rapidly inactivated, mainly to adenosine which in turn has other pharmacological/counteracting properties. Nucleotide hydrolyzing enzymes include the nucleoside triphosphate diphosphohydrolase (NTPDase) family[74], the nucleotide pyrophosphatase/phosphodiesterase (NPP) family[75,76] and ecto-5’-nucleotidase[77].
NTPDases are capable of hydrolyzing a broad range of nucleoside tri and diphosphates, but not monophosphates. Namely, half of the eight different NTPDase genes (NTPDase1, 2, 3 and 8) are expressed as cell surface-located enzymes. The prototypic member of the NTPDase family is the cell activation antigen CD39 (NTPDase1)[78] whose expression has been demonstrated on a variety of cells, vascular endothelial and smooth muscle cells[79], exocrine pancreas[80], dendritic cells[81], lymphocytes[82] and recently MSCs[40] (Figure 1). On the other hand, The NPP family consists of seven related ectoenzymes possessing surprisingly broad substrate specificity capable of hydrolyzing pyrophosphate and phosphodiester bonds generating, for instance, AMP from ATP, or AMP and NMN (nicotinamide monophosphate) from β-NAD[83]. The first three members of this family, NPP1-3, hydrolyze various nucleotides and are therefore relevant in the purinergic signaling cascade[75]. In particular, human NPP1 is highly expressed in bone and cartilage and less in other organs and tissues[75]. In bone tissue, NPP1 acts as a PPi-generating ectoenzyme ensuring normal bone matrix mineralization and soft tissue calcification[84]. The presence and enzymatic activity of NPP1 and NPP3 has been recently demonstrated in MSCs[39] (Figure 1), attesting to the existence of an active and complex extracellular nucleotide metabolism in these cells once more.
Extracellular AMP, generated either from ATP or from β-NAD degradation, can be further metabolized by the ecto-5’-nucleotidase CD73 releasing adenosine[77]. CD73 is expressed to a variable extent in different tissues, with abundant expression in the colon, kidney, brain, liver, heart, lung and large vessel endothelium[77,85,86]. Notably, CD73 is coexpressed with CD39 on the surface of CD4+ Treg cells, being an important constituent of the suppressive machinery that converts ATP to the anti-inflammatory mediator adenosine with subsequent inhibition of T cell proliferation and cytokine secretion[82]. Interestingly, this situation closely resembles that of MSCs whose immunomodulatory activity has also been recently related to the CD39/CD73 enzymatic axis actively producing extracellular adenosine, also with paracrine/immunosuppressive effects in these cells[40] (Figure 1). These data may indicate a key role of adenosine in switching the stem cell properties of MSCs towards an immunomodulatory/pro-healing phenotype which in so many occasions has demonstrated its utility[14], suggesting a possible pharmacological use of adenosine in potentiating these features in cell-based therapies.
Although CD73 is one major cell surface marker defining MSCs according to the International Society for Cellular Therapy (ISCT), it is surprising how little is known about the enzymatic function of CD73 in these cells[87]. Notably, CD73 expression is regulated by Wnt-β-catenin signaling, one of the major pathways in stem cell and bone homeostasis[88]. Recently, CD73 has been reported to be involved in osteogenic differentiation where loss of this ectoenzyme causes a lower bone mineral content in mouse trabecular bone with decreased osteocalcin serum levels and reduced expression of osteogenic mRNA markers[89]. Little is known about the role of CD73 in chondrogenesis, except that CD73 is downregulated during differentiation[90,91]. In a recent investigation, further insights into CD73 in relation to osteogenic/chondrogenic differentiation have been added to the literature[92] using an in vitro model of MSCs differentiated after cyclic-compressive loading. In these conditions, Ode et al[92] observed increased chondrogenic differentiation accompanied by a decreased CD73 expression; in addition to that, they found that inhibition of CD73 enzymatic activity further increased chondrogenic matrix deposition. In contrast, in the same experimental setting but in conditions of osteogenic induction and in the presence of a CD73 inhibitor, MSCs showed a reduction of osteogenic marker expression and of mineral matrix deposition, suggesting that CD73 and its metabolite adenosine, as well as P1 receptors, belong to alternative differentiation pathways in MSCs whose expression enhance (osteogenic) or inhibit (chondrogenic) specific cell lineages. So far, and to our knowledge, no investigations have been undertaken to test the role of CD73 as an ectoenzyme during adipocyte differentiation in MSCs. Since it is known that this protein is expressed on mature adipocytes and that CD73-derived adenosine is functionally involved in body fat homeostasis, mainly inhibiting lipolysis[93], it is highly probable that this topic will be eventually addressed in the near future, hopefully adding new bricks to the comprehension of adipose tissue formation mechanism and complex homeostasis.
Another well-known class of ectoenzymes are β-NAD-consuming surface proteins, primarily represented by the CD38-BST1 system[36]. The CD38 gene codes for a type II transmembrane protein distributed in a broad range of cell types[36]. The other member of the family is BST1/CD157, which differs in structure and tissue distribution[36]. The dual cycling/hydrolyzing metabolism of β-NAD by CD38 leads to the generation of potent intracellular Ca2+ mobilizing compounds, including cADPR (from cycling activity) and ADPR (from both cycling and hydrolyzing activities)[94].
It has been recently demonstrated that MSCs show both a significant β-NAD release from CX43 hemichannels and an active extracellular metabolism of this dinucleotide due not only to NPP1/3 and CD73 degradation to adenosine, but also to CD38-BST1 secondary metabolite production[39] (Figure 1). The release of β-NAD in the BM milieu from MSCs is essential not only for autocrine physiological and immunomodulatory functions[39], but also for HP proliferation and stem cell niche maintenance[95-97]. Thus, the bilateral nucleotide network generated upon β-NAD release from MSCs in the BM comprises the following enzymatic steps and functional effects: (1) β-NAD released in the BM milieu directly stimulates MSC and HP functions through the purinergic receptor P2Y11[39,98]; (2) extracellular β-NAD can be a substrate of various ectoenzymes present either on MSCs, possessing both NPP-CD73 and CD38-BST1 ectoenzymes, or on HP displaying the CD38 activity[39,99-101]; and (3) these enzymatic activities are able to release secondary metabolites in the BM milieu, namely adenosine, ADPR and cADPR, which again can exert autocrine and paracrine regulatory effects on MSCs and HPs[28,39,99-102]. Indeed, nanomolar/low micromolar concentrations of cADPR, such as those produced by variously stimulated CD38-BST1 positive BM cells[99,100], significantly increase the in vitro[99-102] and in vivo[96,103] proliferation and engraftment of human HPs and MSCs, indicating a relevant role for this network of nucleotide-responding and nucleotide-metabolizing proteins in the BM.
CONCLUSION
The increasingly recognized role of MSCs in the homeostasis of mesodermal tissues through their proliferation/differentiation properties and in the regulation of hemopoiesis and cell-degeneration protection through the production of paracrine signals justifies the current interest in identifying the biochemical signals produced by MSCs and their active crosstalk in tissue environments. Only recently, such signals have been shown to also belong to the network of eNTPs and their metabolites produced by specialized ectoenzymes[39,40,87,89-92,99] and active on both ionotropic[41,71] and metabotropic receptors[38,39,42,69,70,72,73] in MSCs (Figure 1). Researchers have just begun to uncover the multifaceted aspects of the eNTP network on MSCs, sometimes revealing unexpected pivotal roles for these molecules and their derivatives in specifying differentiation lineages and in modulating MSC physiology and signaling towards other cells.
Thus, while extracellular β-NAD and cADPR signaling seem to be more related to MSC homeostasis/proliferation and to the maintenance of an optimal stem cell niche for the harmonious growth of HPs and MSCs in the BM[39,95-97,99-103], ATP and adenosine demonstrate more pleiotropic roles affecting both the immunomodulatory properties of these cells and their lineage commitment. In particular, the nucleotide has been more frequently associated with inhibition of proliferation[42,69], pro-inflammatory and cell migration properties[70], as well as to an enhancement of both adipogenic and osteogenic differentiation[38,71-73] in MSCs. Conversely, adenosine has been associated with an autocrine protective[49] as well as a paracrine immunosuppressive[40] activity counteracting ATP stimulation. Furthermore, in MSCs, adenosine seems to have a significant role in alternative lineage specification by concomitant promotion of bone formation[55-57,72,90-92] and inhibition of cartilage production[92]. In agreement with this, it has been suggested that the positive effect of ATP on osteocyte differentiation could be just a consequence of adenosine production on MSCs through surface activity of degrading ectoenzymes[72].
The prosecution of these studies, on the basis of what has been discovered until now and is summarized in this review, seems to be essential for a thorough comprehension of MSC physiology and in the future will enable researchers to precisely define the involvement of these cells in tissue repair and to finally address the current clinical issues related to their use in cell-based therapies.
Footnotes
P- Reviewers: Li GR, Pedata F, Yue JB S- Editor: Song XX L- Editor: Roemmele A E- Editor: Zhang DN
References
- 1.Friedenstein AJ, Chailakhjan RK, Lalykina KS. The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Tissue Kinet. 1970;3:393–403. doi: 10.1111/j.1365-2184.1970.tb00347.x. [DOI] [PubMed] [Google Scholar]
- 2.Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 3.Mosna F, Sensebé L, Krampera M. Human bone marrow and adipose tissue mesenchymal stem cells: a user’s guide. Stem Cells Dev. 2010;19:1449–1470. doi: 10.1089/scd.2010.0140. [DOI] [PubMed] [Google Scholar]
- 4.Huang GT, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res. 2009;88:792–806. doi: 10.1177/0022034509340867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Batsali AK, Kastrinaki MC, Papadaki HA, Pontikoglou C. Mesenchymal stem cells derived from Wharton’s Jelly of the umbilical cord: biological properties and emerging clinical applications. Curr Stem Cell Res Ther. 2013;8:144–155. doi: 10.2174/1574888x11308020005. [DOI] [PubMed] [Google Scholar]
- 6.Wagner W, Ho AD. Mesenchymal stem cell preparations--comparing apples and oranges. Stem Cell Rev. 2007;3:239–248. doi: 10.1007/s12015-007-9001-1. [DOI] [PubMed] [Google Scholar]
- 7.Ho AD, Wagner W, Franke W. Heterogeneity of mesenchymal stromal cell preparations. Cytotherapy. 2008;10:320–330. doi: 10.1080/14653240802217011. [DOI] [PubMed] [Google Scholar]
- 8.Phinney DG, Prockop DJ. Concise review: mesenchymal stem/multipotent stromal cells: the state of transdifferentiation and modes of tissue repair--current views. Stem Cells. 2007;25:2896–2902. doi: 10.1634/stemcells.2007-0637. [DOI] [PubMed] [Google Scholar]
- 9.Bianco P, Riminucci M, Gronthos S, Robey PG. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells. 2001;19:180–192. doi: 10.1634/stemcells.19-3-180. [DOI] [PubMed] [Google Scholar]
- 10.Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund T, Blackstad M, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418:41–49. doi: 10.1038/nature00870. [DOI] [PubMed] [Google Scholar]
- 11.Dazzi F, Ramasamy R, Glennie S, Jones SP, Roberts I. The role of mesenchymal stem cells in haemopoiesis. Blood Rev. 2006;20:161–171. doi: 10.1016/j.blre.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 12.Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8:726–736. doi: 10.1038/nri2395. [DOI] [PubMed] [Google Scholar]
- 13.Salem HK, Thiemermann C. Mesenchymal stromal cells: current understanding and clinical status. Stem Cells. 2010;28:585–596. doi: 10.1002/stem.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.English K. Mechanisms of mesenchymal stromal cell immunomodulation. Immunol Cell Biol. 2013;91:19–26. doi: 10.1038/icb.2012.56. [DOI] [PubMed] [Google Scholar]
- 15.Satija NK, Singh VK, Verma YK, Gupta P, Sharma S, Afrin F, Sharma M, Sharma P, Tripathi RP, Gurudutta GU. Mesenchymal stem cell-based therapy: a new paradigm in regenerative medicine. J Cell Mol Med. 2009;13:4385–4402. doi: 10.1111/j.1582-4934.2009.00857.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baksh D, Song L, Tuan RS. Adult mesenchymal stem cells: characterization, differentiation, and application in cell and gene therapy. J Cell Mol Med. 2004;8:301–316. doi: 10.1111/j.1582-4934.2004.tb00320.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nakashima K, de Crombrugghe B. Transcriptional mechanisms in osteoblast differentiation and bone formation. Trends Genet. 2003;19:458–466. doi: 10.1016/S0168-9525(03)00176-8. [DOI] [PubMed] [Google Scholar]
- 18.Komori T. Regulation of osteoblast differentiation by transcription factors. J Cell Biochem. 2006;99:1233–1239. doi: 10.1002/jcb.20958. [DOI] [PubMed] [Google Scholar]
- 19.MacDougald OA, Mandrup S. Adipogenesis: forces that tip the scales. Trends Endocrinol Metab. 2002;13:5–11. doi: 10.1016/s1043-2760(01)00517-3. [DOI] [PubMed] [Google Scholar]
- 20.Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem. 1997;64:295–312. [PubMed] [Google Scholar]
- 21.Rodríguez JP, Astudillo P, Ríos S, Pino AM. Involvement of adipogenic potential of human bone marrow mesenchymal stem cells (MSCs) in osteoporosis. Curr Stem Cell Res Ther. 2008;3:208–218. doi: 10.2174/157488808785740325. [DOI] [PubMed] [Google Scholar]
- 22.Blair HC, Zaidi M, Schlesinger PH. Mechanisms balancing skeletal matrix synthesis and degradation. Biochem J. 2002;364:329–341. doi: 10.1042/BJ20020165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang W, Yang S, Shao J, Li YP. Signaling and transcriptional regulation in osteoblast commitment and differentiation. Front Biosci. 2007;12:3068–3092. doi: 10.2741/2296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Otto TC, Lane MD. Adipose development: from stem cell to adipocyte. Crit Rev Biochem Mol Biol. 2005;40:229–242. doi: 10.1080/10409230591008189. [DOI] [PubMed] [Google Scholar]
- 25.Di Iorgi N, Rosol M, Mittelman SD, Gilsanz V. Reciprocal relation between marrow adiposity and the amount of bone in the axial and appendicular skeleton of young adults. J Clin Endocrinol Metab. 2008;93:2281–2286. doi: 10.1210/jc.2007-2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Di Iorgi N, Mo AO, Grimm K, Wren TA, Dorey F, Gilsanz V. Bone acquisition in healthy young females is reciprocally related to marrow adiposity. J Clin Endocrinol Metab. 2010;95:2977–2982. doi: 10.1210/jc.2009-2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Verma S, Rajaratnam JH, Denton J, Hoyland JA, Byers RJ. Adipocytic proportion of bone marrow is inversely related to bone formation in osteoporosis. J Clin Pathol. 2002;55:693–698. doi: 10.1136/jcp.55.9.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rossi L, Salvestrini V, Ferrari D, Di Virgilio F, Lemoli RM. The sixth sense: hematopoietic stem cells detect danger through purinergic signaling. Blood. 2012;120:2365–2375. doi: 10.1182/blood-2012-04-422378. [DOI] [PubMed] [Google Scholar]
- 29.Ralevic V, Burnstock G. Receptors for purines and pyrimidines. Pharmacol Rev. 1998;50:413–492. [PubMed] [Google Scholar]
- 30.Goding JW, Howard MC. Ecto-enzymes of lymphoid cells. Immunol Rev. 1998;161:5–10. doi: 10.1111/j.1600-065x.1998.tb01567.x. [DOI] [PubMed] [Google Scholar]
- 31.Latini S, Pedata F. Adenosine in the central nervous system: release mechanisms and extracellular concentrations. J Neurochem. 2001;79:463–484. doi: 10.1046/j.1471-4159.2001.00607.x. [DOI] [PubMed] [Google Scholar]
- 32.Heinrich A, Andó RD, Túri G, Rózsa B, Sperlágh B. K+ depolarization evokes ATP, adenosine and glutamate release from glia in rat hippocampus: a microelectrode biosensor study. Br J Pharmacol. 2012;167:1003–1020. doi: 10.1111/j.1476-5381.2012.01932.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Burnstock G. Historical review: ATP as a neurotransmitter. Trends Pharmacol Sci. 2006;27:166–176. doi: 10.1016/j.tips.2006.01.005. [DOI] [PubMed] [Google Scholar]
- 34.Burnstock G. Pathophysiology and therapeutic potential of purinergic signaling. Pharmacol Rev. 2006;58:58–86. doi: 10.1124/pr.58.1.5. [DOI] [PubMed] [Google Scholar]
- 35.Salmi M, Jalkanen S. Cell-surface enzymes in control of leukocyte trafficking. Nat Rev Immunol. 2005;5:760–771. doi: 10.1038/nri1705. [DOI] [PubMed] [Google Scholar]
- 36.Malavasi F, Deaglio S, Funaro A, Ferrero E, Horenstein AL, Ortolan E, Vaisitti T, Aydin S. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol Rev. 2008;88:841–886. doi: 10.1152/physrev.00035.2007. [DOI] [PubMed] [Google Scholar]
- 37.Burnstock G, Verkhratsky A. Evolutionary origins of the purinergic signalling system. Acta Physiol (Oxf) 2009;195:415–447. doi: 10.1111/j.1748-1716.2009.01957.x. [DOI] [PubMed] [Google Scholar]
- 38.Zippel N, Limbach CA, Ratajski N, Urban C, Luparello C, Pansky A, Kassack MU, Tobiasch E. Purinergic receptors influence the differentiation of human mesenchymal stem cells. Stem Cells Dev. 2012;21:884–900. doi: 10.1089/scd.2010.0576. [DOI] [PubMed] [Google Scholar]
- 39.Fruscione F, Scarfì S, Ferraris C, Bruzzone S, Benvenuto F, Guida L, Uccelli A, Salis A, Usai C, Jacchetti E, et al. Regulation of human mesenchymal stem cell functions by an autocrine loop involving NAD+ release and P2Y11-mediated signaling. Stem Cells Dev. 2011;20:1183–1198. doi: 10.1089/scd.2010.0295. [DOI] [PubMed] [Google Scholar]
- 40.Chen M, Su W, Lin X, Guo Z, Wang J, Zhang Q, Brand D, Ryffel B, Huang J, Liu Z, et al. Adoptive transfer of human gingiva-derived mesenchymal stem cells ameliorates collagen-induced arthritis via suppression of Th1 and Th17 cells and enhancement of regulatory T cell differentiation. Arthritis Rheum. 2013;65:1181–1193. doi: 10.1002/art.37894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kawano S, Otsu K, Kuruma A, Shoji S, Yanagida E, Muto Y, Yoshikawa F, Hirayama Y, Mikoshiba K, Furuichi T. ATP autocrine/paracrine signaling induces calcium oscillations and NFAT activation in human mesenchymal stem cells. Cell Calcium. 2006;39:313–324. doi: 10.1016/j.ceca.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 42.Coppi E, Pugliese AM, Urbani S, Melani A, Cerbai E, Mazzanti B, Bosi A, Saccardi R, Pedata F. ATP modulates cell proliferation and elicits two different electrophysiological responses in human mesenchymal stem cells. Stem Cells. 2007;25:1840–1849. doi: 10.1634/stemcells.2006-0669. [DOI] [PubMed] [Google Scholar]
- 43.Bours MJ, Swennen EL, Di Virgilio F, Cronstein BN, Dagnelie PC. Adenosine 5’-triphosphate and adenosine as endogenous signaling molecules in immunity and inflammation. Pharmacol Ther. 2006;112:358–404. doi: 10.1016/j.pharmthera.2005.04.013. [DOI] [PubMed] [Google Scholar]
- 44.Shryock JC, Belardinelli L. Adenosine and adenosine receptors in the cardiovascular system: biochemistry, physiology, and pharmacology. Am J Cardiol. 1997;79:2–10. doi: 10.1016/s0002-9149(97)00256-7. [DOI] [PubMed] [Google Scholar]
- 45.Spychala J. Tumor-promoting functions of adenosine. Pharmacol Ther. 2000;87:161–173. doi: 10.1016/s0163-7258(00)00053-x. [DOI] [PubMed] [Google Scholar]
- 46.Jacobson KA, Gao ZG. Adenosine receptors as therapeutic targets. Nat Rev Drug Discov. 2006;5:247–264. doi: 10.1038/nrd1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Evans BA, Elford C, Pexa A, Francis K, Hughes AC, Deussen A, Ham J. Human osteoblast precursors produce extracellular adenosine, which modulates their secretion of IL-6 and osteoprotegerin. J Bone Miner Res. 2006;21:228–236. doi: 10.1359/JBMR.051021. [DOI] [PubMed] [Google Scholar]
- 48.Fredholm BB. Adenosine receptors as drug targets. Exp Cell Res. 2010;316:1284–1288. doi: 10.1016/j.yexcr.2010.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Katebi M, Soleimani M, Cronstein BN. Adenosine A2A receptors play an active role in mouse bone marrow-derived mesenchymal stem cell development. J Leukoc Biol. 2009;85:438–444. doi: 10.1189/jlb.0908520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sun LL, Xu LL, Nielsen TB, Rhee P, Burris D. Cyclopentyladenosine improves cell proliferation, wound healing, and hair growth. J Surg Res. 1999;87:14–24. doi: 10.1006/jsre.1999.5716. [DOI] [PubMed] [Google Scholar]
- 51.Shimegi S. ATP and adenosine act as a mitogen for osteoblast-like cells (MC3T3-E1) Calcif Tissue Int. 1996;58:109–113. doi: 10.1007/BF02529732. [DOI] [PubMed] [Google Scholar]
- 52.Walker BA, Rocchini C, Boone RH, Ip S, Jacobson MA. Adenosine A2a receptor activation delays apoptosis in human neutrophils. J Immunol. 1997;158:2926–2931. [PubMed] [Google Scholar]
- 53.Zhao ZQ, Budde JM, Morris C, Wang NP, Velez DA, Muraki S, Guyton RA, Vinten-Johansen J. Adenosine attenuates reperfusion-induced apoptotic cell death by modulating expression of Bcl-2 and Bax proteins. J Mol Cell Cardiol. 2001;33:57–68. doi: 10.1006/jmcc.2000.1275. [DOI] [PubMed] [Google Scholar]
- 54.Napieralski R, Kempkes B, Gutensohn W. Evidence for coordinated induction and repression of ecto-5’-nucleotidase (CD73) and the A2a adenosine receptor in a human B cell line. Biol Chem. 2003;384:483–487. doi: 10.1515/BC.2003.054. [DOI] [PubMed] [Google Scholar]
- 55.Gharibi B, Abraham AA, Ham J, Evans BA. Adenosine receptor subtype expression and activation influence the differentiation of mesenchymal stem cells to osteoblasts and adipocytes. J Bone Miner Res. 2011;26:2112–2124. doi: 10.1002/jbmr.424. [DOI] [PubMed] [Google Scholar]
- 56.Carroll SH, Wigner NA, Kulkarni N, Johnston-Cox H, Gerstenfeld LC, Ravid K. A2B adenosine receptor promotes mesenchymal stem cell differentiation to osteoblasts and bone formation in vivo. J Biol Chem. 2012;287:15718–15727. doi: 10.1074/jbc.M112.344994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.He W, Mazumder A, Wilder T, Cronstein BN. Adenosine regulates bone metabolism via A1, A2A, and A2B receptors in bone marrow cells from normal humans and patients with multiple myeloma. FASEB J. 2013;27:3446–3454. doi: 10.1096/fj.13-231233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Schwiebert EM, Fitz JG. Purinergic signaling microenvironments: An introduction. Purinergic Signal. 2008;4:89–92. doi: 10.1007/s11302-007-9091-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vassort G. Adenosine 5’-triphosphate: a P2-purinergic agonist in the myocardium. Physiol Rev. 2001;81:767–806. doi: 10.1152/physrev.2001.81.2.767. [DOI] [PubMed] [Google Scholar]
- 60.Roman RM, Fitz JG. Emerging roles of purinergic signaling in gastrointestinal epithelial secretion and hepatobiliary function. Gastroenterology. 1999;116:964–979. doi: 10.1016/s0016-5085(99)70081-8. [DOI] [PubMed] [Google Scholar]
- 61.Schwiebert EM, Zsembery A. Extracellular ATP as a signaling molecule for epithelial cells. Biochim Biophys Acta. 2003;1615:7–32. doi: 10.1016/s0005-2736(03)00210-4. [DOI] [PubMed] [Google Scholar]
- 62.Sprague RS, Stephenson AH, Ellsworth ML. Red not dead: signaling in and from erythrocytes. Trends Endocrinol Metab. 2007;18:350–355. doi: 10.1016/j.tem.2007.08.008. [DOI] [PubMed] [Google Scholar]
- 63.Umapathy NS, Zemskov EA, Gonzales J, Gorshkov BA, Sridhar S, Chakraborty T, Lucas R, Verin AD. Extracellular beta-nicotinamide adenine dinucleotide (beta-NAD) promotes the endothelial cell barrier integrity via PKA- and EPAC1/Rac1-dependent actin cytoskeleton rearrangement. J Cell Physiol. 2010;223:215–223. doi: 10.1002/jcp.22029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Klein C, Grahnert A, Abdelrahman A, Müller CE, Hauschildt S. Extracellular NAD(+) induces a rise in [Ca(2+)](i) in activated human monocytes via engagement of P2Y(1) and P2Y(11) receptors. Cell Calcium. 2009;46:263–272. doi: 10.1016/j.ceca.2009.08.004. [DOI] [PubMed] [Google Scholar]
- 65.Gachet C. Regulation of platelet functions by P2 receptors. Annu Rev Pharmacol Toxicol. 2006;46:277–300. doi: 10.1146/annurev.pharmtox.46.120604.141207. [DOI] [PubMed] [Google Scholar]
- 66.Di Virgilio F, Solini A. P2 receptors: new potential players in atherosclerosis. Br J Pharmacol. 2002;135:831–842. doi: 10.1038/sj.bjp.0704524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Moreschi I, Bruzzone S, Nicholas RA, Fruscione F, Sturla L, Benvenuto F, Usai C, Meis S, Kassack MU, Zocchi E, et al. Extracellular NAD+ is an agonist of the human P2Y11 purinergic receptor in human granulocytes. J Biol Chem. 2006;281:31419–31429. doi: 10.1074/jbc.M606625200. [DOI] [PubMed] [Google Scholar]
- 68.Durnin L, Hwang SJ, Ward SM, Sanders KM, Mutafova-Yambolieva VN. Adenosine 5-diphosphate-ribose is a neural regulator in primate and murine large intestine along with β-NAD(+) J Physiol. 2012;590:1921–1941. doi: 10.1113/jphysiol.2011.222414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ichikawa J, Gemba H. Cell density-dependent changes in intracellular Ca2+ mobilization via the P2Y2 receptor in rat bone marrow stromal cells. J Cell Physiol. 2009;219:372–381. doi: 10.1002/jcp.21680. [DOI] [PubMed] [Google Scholar]
- 70.Ferrari D, Gulinelli S, Salvestrini V, Lucchetti G, Zini R, Manfredini R, Caione L, Piacibello W, Ciciarello M, Rossi L, et al. Purinergic stimulation of human mesenchymal stem cells potentiates their chemotactic response to CXCL12 and increases the homing capacity and production of proinflammatory cytokines. Exp Hematol. 2011;39:360–74, 374.e1-5. doi: 10.1016/j.exphem.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 71.Sun D, Junger WG, Yuan C, Zhang W, Bao Y, Qin D, Wang C, Tan L, Qi B, Zhu D, et al. Shockwaves induce osteogenic differentiation of human mesenchymal stem cells through ATP release and activation of P2X7 receptors. Stem Cells. 2013;31:1170–1180. doi: 10.1002/stem.1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ciciarello M, Zini R, Rossi L, Salvestrini V, Ferrari D, Manfredini R, Lemoli RM. Extracellular purines promote the differentiation of human bone marrow-derived mesenchymal stem cells to the osteogenic and adipogenic lineages. Stem Cells Dev. 2013;22:1097–1111. doi: 10.1089/scd.2012.0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Biver G, Wang N, Gartland A, Orriss I, Arnett TR, Boeynaems JM, Robaye B. Role of the P2Y13 receptor in the differentiation of bone marrow stromal cells into osteoblasts and adipocytes. Stem Cells. 2013;31:2747–2758. doi: 10.1002/stem.1411. [DOI] [PubMed] [Google Scholar]
- 74.Robson SC, Sévigny J, Zimmermann H. The E-NTPDase family of ectonucleotidases: Structure function relationships and pathophysiological significance. Purinergic Signal. 2006;2:409–430. doi: 10.1007/s11302-006-9003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Goding JW, Grobben B, Slegers H. Physiological and pathophysiological functions of the ecto-nucleotide pyrophosphatase/phosphodiesterase family. Biochim Biophys Acta. 2003;1638:1–19. doi: 10.1016/s0925-4439(03)00058-9. [DOI] [PubMed] [Google Scholar]
- 76.Stefan C, Jansen S, Bollen M. Modulation of purinergic signaling by NPP-type ectophosphodiesterases. Purinergic Signal. 2006;2:361–370. doi: 10.1007/s11302-005-5303-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Colgan SP, Eltzschig HK, Eckle T, Thompson LF. Physiological roles for ecto-5’-nucleotidase (CD73) Purinergic Signal. 2006;2:351–360. doi: 10.1007/s11302-005-5302-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wang TF, Guidotti G. CD39 is an ecto-(Ca2+,Mg2+)-apyrase. J Biol Chem. 1996;271:9898–9901. [PubMed] [Google Scholar]
- 79.Marcus AJ, Broekman MJ, Drosopoulos JH, Islam N, Pinsky DJ, Sesti C, Levi R. Heterologous cell-cell interactions: thromboregulation, cerebroprotection and cardioprotection by CD39 (NTPDase-1) J Thromb Haemost. 2003;1:2497–2509. doi: 10.1111/j.1538-7836.2003.00479.x. [DOI] [PubMed] [Google Scholar]
- 80.Sørensen CE, Amstrup J, Rasmussen HN, Ankorina-Stark I, Novak I. Rat pancreas secretes particulate ecto-nucleotidase CD39. J Physiol. 2003;551:881–892. doi: 10.1113/jphysiol.2003.049411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Mizumoto N, Kumamoto T, Robson SC, Sévigny J, Matsue H, Enjyoji K, Takashima A. CD39 is the dominant Langerhans cell-associated ecto-NTPDase: modulatory roles in inflammation and immune responsiveness. Nat Med. 2002;8:358–365. doi: 10.1038/nm0402-358. [DOI] [PubMed] [Google Scholar]
- 82.Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, Chen JF, Enjyoji K, Linden J, Oukka M, et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204:1257–1265. doi: 10.1084/jem.20062512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Stefan C, Jansen S, Bollen M. NPP-type ectophosphodiesterases: unity in diversity. Trends Biochem Sci. 2005;30:542–550. doi: 10.1016/j.tibs.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 84.Terkeltaub RA. Inorganic pyrophosphate generation and disposition in pathophysiology. Am J Physiol Cell Physiol. 2001;281:C1–C11. doi: 10.1152/ajpcell.2001.281.1.C1. [DOI] [PubMed] [Google Scholar]
- 85.Thompson LF, Eltzschig HK, Ibla JC, Van De Wiele CJ, Resta R, Morote-Garcia JC, Colgan SP. Crucial role for ecto-5’-nucleotidase (CD73) in vascular leakage during hypoxia. J Exp Med. 2004;200:1395–1405. doi: 10.1084/jem.20040915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Moriwaki Y, Yamamoto T, Higashino K. Enzymes involved in purine metabolism--a review of histochemical localization and functional implications. Histol Histopathol. 1999;14:1321–1340. doi: 10.14670/HH-14.1321. [DOI] [PubMed] [Google Scholar]
- 87.Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 88.Spychala J, Kitajewski J. Wnt and beta-catenin signaling target the expression of ecto-5’-nucleotidase and increase extracellular adenosine generation. Exp Cell Res. 2004;296:99–108. doi: 10.1016/j.yexcr.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 89.Takedachi M, Oohara H, Smith BJ, Iyama M, Kobashi M, Maeda K, Long CL, Humphrey MB, Stoecker BJ, Toyosawa S, et al. CD73-generated adenosine promotes osteoblast differentiation. J Cell Physiol. 2012;227:2622–2631. doi: 10.1002/jcp.23001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Song L, Webb NE, Song Y, Tuan RS. Identification and functional analysis of candidate genes regulating mesenchymal stem cell self-renewal and multipotency. Stem Cells. 2006;24:1707–1718. doi: 10.1634/stemcells.2005-0604. [DOI] [PubMed] [Google Scholar]
- 91.Delorme B, Ringe J, Gallay N, Le Vern Y, Kerboeuf D, Jorgensen C, Rosset P, Sensebé L, Layrolle P, Häupl T, et al. Specific plasma membrane protein phenotype of culture-amplified and native human bone marrow mesenchymal stem cells. Blood. 2008;111:2631–2635. doi: 10.1182/blood-2007-07-099622. [DOI] [PubMed] [Google Scholar]
- 92.Ode A, Schoon J, Kurtz A, Gaetjen M, Ode JE, Geissler S, Duda GN. CD73/5’-ecto-nucleotidase acts as a regulatory factor in osteo-/chondrogenic differentiation of mechanically stimulated mesenchymal stromal cells. Eur Cell Mater. 2013;25:37–47. doi: 10.22203/ecm.v025a03. [DOI] [PubMed] [Google Scholar]
- 93.Burghoff S, Flögel U, Bongardt S, Burkart V, Sell H, Tucci S, Ikels K, Eberhard D, Kern M, Klöting N, et al. Deletion of CD73 promotes dyslipidemia and intramyocellular lipid accumulation in muscle of mice. Arch Physiol Biochem. 2013;119:39–51. doi: 10.3109/13813455.2012.755547. [DOI] [PubMed] [Google Scholar]
- 94.Lee HC. Multiplicity of Ca2+ messengers and Ca2+ stores: a perspective from cyclic ADP-ribose and NAADP. Curr Mol Med. 2004;4:227–237. doi: 10.2174/1566524043360753. [DOI] [PubMed] [Google Scholar]
- 95.Podestà M, Zocchi E, Pitto A, Usai C, Franco L, Bruzzone S, Guida L, Bacigalupo A, Scadden DT, Walseth TF, et al. Extracellular cyclic ADP-ribose increases intracellular free calcium concentration and stimulates proliferation of human hemopoietic progenitors. FASEB J. 2000;14:680–690. doi: 10.1096/fasebj.14.5.680. [DOI] [PubMed] [Google Scholar]
- 96.Podestà M, Pitto A, Figari O, Bacigalupo A, Bruzzone S, Guida L, Franco L, De Flora A, Zocchi E. Cyclic ADP-ribose generation by CD38 improves human hemopoietic stem cell engraftment into NOD/SCID mice. FASEB J. 2003;17:310–312. doi: 10.1096/fj.02-0520fje. [DOI] [PubMed] [Google Scholar]
- 97.Podestà M, Benvenuto F, Pitto A, Figari O, Bacigalupo A, Bruzzone S, Guida L, Franco L, Paleari L, Bodrato N, et al. Concentrative uptake of cyclic ADP-ribose generated by BST-1+ stroma stimulates proliferation of human hematopoietic progenitors. J Biol Chem. 2005;280:5343–5349. doi: 10.1074/jbc.M408085200. [DOI] [PubMed] [Google Scholar]
- 98.Wang L, Jacobsen SE, Bengtsson A, Erlinge D. P2 receptor mRNA expression profiles in human lymphocytes, monocytes and CD34+ stem and progenitor cells. BMC Immunol. 2004;5:16. doi: 10.1186/1471-2172-5-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Scarfì S, Ferraris C, Fruscione F, Fresia C, Guida L, Bruzzone S, Usai C, Parodi A, Millo E, Salis A, et al. Cyclic ADP-ribose-mediated expansion and stimulation of human mesenchymal stem cells by the plant hormone abscisic acid. Stem Cells. 2008;26:2855–2864. doi: 10.1634/stemcells.2008-0488. [DOI] [PubMed] [Google Scholar]
- 100.Scarfì S, Fresia C, Ferraris C, Bruzzone S, Fruscione F, Usai C, Benvenuto F, Magnone M, Podestà M, Sturla L, et al. The plant hormone abscisic acid stimulates the proliferation of human hemopoietic progenitors through the second messenger cyclic ADP-ribose. Stem Cells. 2009;27:2469–2477. doi: 10.1002/stem.173. [DOI] [PubMed] [Google Scholar]
- 101.Zocchi E, Podestà M, Pitto A, Usai C, Bruzzone S, Franco L, Guida L, Bacigalupo A, De Flora A. Paracrinally stimulated expansion of early human hemopoietic progenitors by stroma-generated cyclic ADP-ribose. FASEB J. 2001;15:1610–1612. doi: 10.1096/fj.00-0803fje. [DOI] [PubMed] [Google Scholar]
- 102.Tao R, Sun HY, Lau CP, Tse HF, Lee HC, Li GR. Cyclic ADP ribose is a novel regulator of intracellular Ca2+ oscillations in human bone marrow mesenchymal stem cells. J Cell Mol Med. 2011;15:2684–2696. doi: 10.1111/j.1582-4934.2011.01263.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Burastero G, Scarfì S, Ferraris C, Fresia C, Sessarego N, Fruscione F, Monetti F, Scarfò F, Schupbach P, Podestà M, et al. The association of human mesenchymal stem cells with BMP-7 improves bone regeneration of critical-size segmental bone defects in athymic rats. Bone. 2010;47:117–126. doi: 10.1016/j.bone.2010.03.023. [DOI] [PubMed] [Google Scholar]


