Abstract
Nephrotic syndrome (NS) may be complicated by thromboembolism, which occasionally manifests as stroke. Although the optimal, standardized approach to the prophylaxis and management of thromboembolic complications associated with NS has not been established, anticoagulation with heparin and subsequent warfarin is the de facto standard of treatment. Dabigatran, a novel direct thrombin inhibitor, has become a substitute for warfarin and heparin for many indications, including the prophylaxis of stroke associated with nonvalvular atrial fibrillation and postoperative thromboprophylaxis in orthopedic patients. We report a 35-year-old male with NS due to membranous nephropathy (MN) that presented with carotid thromboembolism. Because the patient developed drug-induced hepatitis due to warfarin, we attempted treatment with dabigatran and were successful in continuing the medication without any complications. We also reviewed the literature on stroke associated with NS. Twenty-one prior cases have been reported, and the review of these cases revealed some interesting points. The age of onset ranged from 19 to 59 years. Most of the reported cases sustained a stroke at earlier ages than patients with atherosclerosis and atrial fibrillation, which suggests that NS may independently predispose individuals to arterial and venous thromboses. MN was the most common underlying pathology. Given that a standardized approach to the prophylaxis and management of thrombotic complications associated with NS has not been established, our experience suggests that dabigatran is a valid new treatment option for thrombotic complications of NS.
Key Words: Cerebral infarction, Carotid thromboembolism, Dabigatran, Membranous nephropathy, Nephrotic syndrome
Introduction
Nephrotic syndrome (NS) may be complicated by thromboembolism [1]. Cases of arterial thromboembolism, including cerebrovascular infarctions, are reported far less frequently than those involving venous thromboses [2]. Anticoagulation with sequential heparin and oral warfarin is currently the de facto standard of care [1]. Dabigatran, a novel, direct competitive thrombin inhibitor, has become a substitute for warfarin and heparin for the prophylaxis of stroke associated with nonvalvular atrial fibrillation [3] and postoperative thromboprophylaxis in orthopedic patients [4]. We report a case of NS due to idiopathic membranous nephropathy (MN) that presented with carotid embolism and cerebral infarction that was safely treated with dabigatran. We also review the previously reported cases of cerebral infarctions associated with NS [5, 6, 7, 8, 9, 10].
Case Presentation
A 35-year-old Japanese male presented to the Emergency Department of Okinawa Chubu Hospital with right hemiparesis. He had awakened from sleep with severe, sharp chest pain that was not accompanied by dyspnea. The chest pain subsided within 1 min without treatment, but medical evaluation was sought due to obvious dysarthria and weakness of the right upper and lower limbs. The patient was previously healthy, with no prior hospital admissions, surgeries, medications, or allergies, but bilateral pedal edema had developed 3 months prior to presentation. He had no dyspnea or ambulatory dysfunction and did not seek medical intervention. He worked as a house painter and lived with his wife and children. He had 15 pack-years of exposure to tobacco, and consumed 400 ml of Okinawan spirits nightly. There was no family history of cardiovascular disease, thromboembolism, or chronic kidney disease.
Physical examination revealed that the patient was alert with no distress. He was not obese (BMI 21.4 on admission, when he was edematous), and he was afebrile with a blood pressure of 140/90 mm Hg, a regular pulse of 85 beats/min, and a respiration rate of 25 breaths/min. Oxygen saturation in the room air was 100%. An examination of the head revealed a marked, right-sided facial droop, but was otherwise normal. Carotid upstrokes were symmetric without bruits. The heartbeat was regular without gallops or murmurs, and the lungs were clear; the abdominal examination was also normal. Bilateral pitting edema was present to the knees. There were no rashes or petechiae. Neurological examination revealed intense dysarthria, which made it difficult for him to communicate with others, and right-sided central facial nerve palsy. Both the right arm and leg were at Brunnstrom stage 1, with complete flaccidity and no voluntary movement. Pain and light touch sensation were absent on the right side of his body.
Table 1 contains initial laboratory data. Blood counts revealed leukocytosis and hemoconcentration. Serum albumin was markedly low at 1.8 g/dl. Serum cholesterol, triglyceride, and low-density lipoprotein cholesterol were elevated. The urine protein to creatinine ratio was 7.5 g/g Cr, indicative of high-grade proteinuria. The examination of the urine sediment revealed oval fat bodies, and testing for anti-nuclear antibodies, anti-neutrophil cytoplasmic antibodies, and anti-phospholipid antibodies were all negative. Hepatitis B and C serologies were negative.
Table 1.
Laboratory data
| First admission | Second admission (day 74) | |
|---|---|---|
| Leukocyte, count/mm3 | 18,000 | 14,000 |
| Stab-formed neutrophil, % | 2 | 4 |
| Segmented neutrophil, % | 76 | 86 |
| Lymphocyte, % | 16 | 9 |
| Monocyte, % | 5 | 1 |
| Hgb, g/dl | 19.9 | 15.5 |
| Hct, % | 56.2 | 43.9 |
| Platelet, ×104/mm3 | 31.4 | 24.7 |
| Total protein, g/dl | 3.6 | 4.3 |
| Albumin, g/dl | 1.8 | 2.5 |
| Sodium, mEq/l | 141 | 143 |
| Potassium, mEq/l | 3.3 | 3.9 |
| Chloride, mEq/l | 105 | 108 |
| Calcium, mg/dl | 7.8 | N/A |
| Magnesium, mg/dl | 1.8 | N/A |
| Glucose, mg/dl | 97 | 145 |
| BUN, mg/dl | 14 | 14 |
| Creatinine, mg/dl | 0.96 | 70.1 |
| AST, IU/l | 19 | 292 |
| ALT, IU/l | 14 | 675 |
| ALP, IU/l | 168 | 371 |
| γ-GTP, IU/l | 27 | 393 |
| T-Bil, mg/dl | 0.4 | 0.8 |
| LDH, IU/l | 294 | 505 |
| CRP, mg/dl | 0.03 | N/A |
| Triglyceride, mg/dl | 266 | 212 |
| Total cholesterol, mg/dl | 311 | 207 |
| HDL-cholesterol, mg/dl | 50 | 78 |
| HbA1c, % (JDS) | 4.7 | 5.6 |
| PT-INR | 0.94 | 3.19 |
| aPTT, s | 23.7 | 31.1 |
| Control aPTT, s | 26.1 | 28.9 |
| Fibrinogen, mg/dl | 538 | N/A |
| D-dimer, μg/ml | 1.5 | N/A |
| Antithrombin activity, % | 115 | N/A |
| Urine findings | ||
| Density | 1.030 | 1.005 |
| Protein-creatinine ratio (g/g Cr) | 7.5 | 5.3 |
| Occult blood | (++) | (±) |
| Leukocyte, count/HPF | <1 | <1 |
| Erythrocyte, count/HPF | 1–5 | <1 |
| Oval fat body | (+) | (–) |
Hgb = Hemoglobin; Hct = hematocrit; BUN = blood urea nitrogen; AST = aspartate aminotransferase; ALT = alanine aminotransferase; ALP = alkaline phosphatase; γ-GTP = γ-glutamyl transpeptidase; T-Bil = total bilirubin, LDH = lactate dehydrogenase; CRP = C-reactive protein; HDL = high-density lipoprotein; JDS = Japan Diabetes Society; PT-INR = prothrombin time-international normalized ratio; HPF = high-power field.
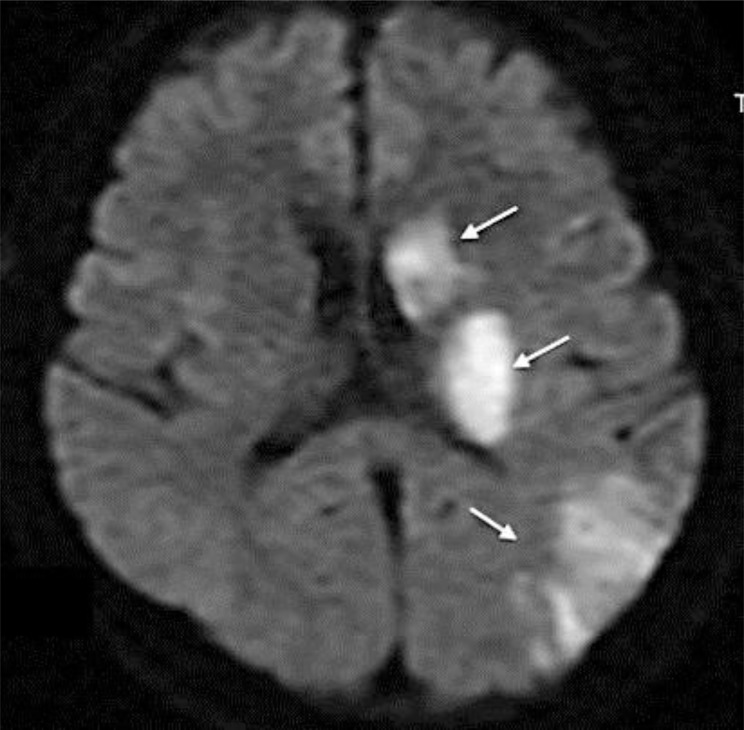
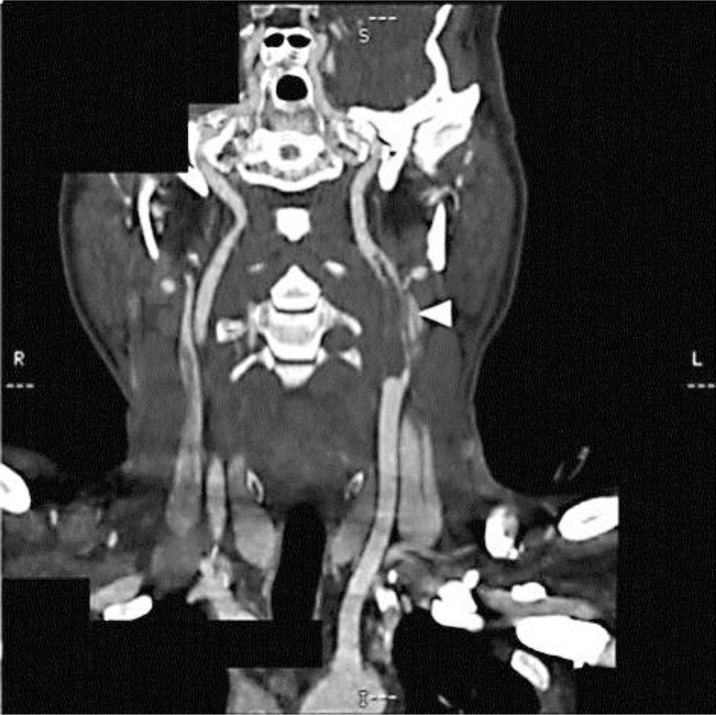
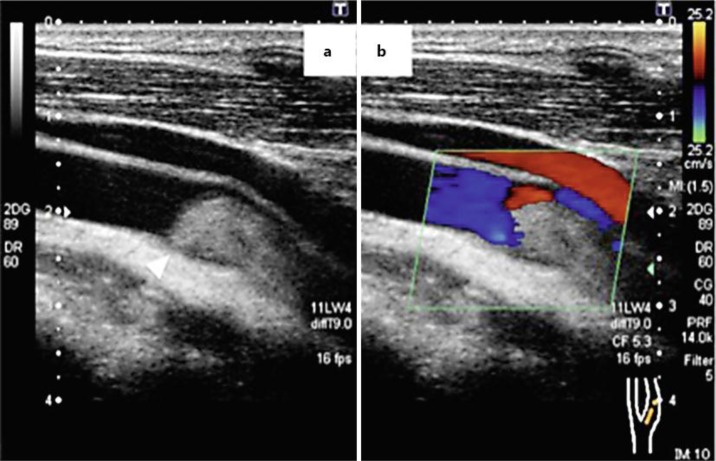
Diffusion-weighted magnetic resonance imaging showed high-intensity areas consistent with acute infarction in the left basal ganglia, left corona radiata, and left cerebral cortex (fig. 1). Contrast-enhanced computed tomography of the neck revealed collateral blood flow around a large embolus in the left carotid artery (fig. 2). Ultrasound of the left carotid revealed the embolus without any plaques or ulcers at the vessel wall (fig. 3). There were no aortic dissections, pulmonary emboli, renal vein thromboses, or deep vein thromboses on the computed tomography. A transthoracic echocardiogram showed normal contractility without dilated chambers, valvular disease, vegetations, intraluminal thrombi, or any findings suggesting pulmonary embolism, such as pulmonary hypertension or right ventricular stress.
Fig. 1.
Diffusion-weighted magnetic resonance imaging of the brain on admission. Note the high-intensity areas suggesting acute phase of brain infarction in the left basal ganglia, left corona radiata, and left cerebral cortex (arrows).
Fig. 2.
Computed tomography angiography of the carotid arteries. A large embolus in the left carotid artery (arrowhead) is present.
Fig. 3.
Echocardiogram of the left carotid artery. a An embolus in the left carotid artery (arrowhead). b Color Doppler shows peripheral blood flow of the carotid artery.
A presumptive diagnosis of ischemic cerebrovascular accident caused by a left carotid thromboembolism was attributed to a hypercoagulable state due to NS. Because of the lack of any findings of atherosclerosis or plaques at the left carotid, we believed that embolism was more likely than thrombosis. The origin of the embolus was not detected despite a thorough cardiovascular evaluation. Immediate anticoagulation with unfractionated heparin was employed, and the renal biopsy was deferred. Intravenous methylprednisolone was administered empirically at 1,000 mg/day for 3 days, followed by oral prednisone at 60 mg/day. Cyclosporine was added 30 days later in response to persistent nephrotic-range proteinuria (5.5 g/day on the 28th day). The patient was transferred to a rehabilitation hospital 47 days after first admission.
However, the blood examination performed after transfer revealed abnormal liver function on the 74th day from the first admission, the results of which are detailed in table 1. Serological studies and abdominal ultrasound excluded viral hepatitis or biliary obstruction. He was immediately readmitted under suspicion of drug-induced hepatotoxicity. Medications on admission included prednisolone (50 mg/day), warfarin (2.75 mg/day), cyclosporine (70 mg b.i.d.), sulfamethoxazole/trimethoprim (400/80 mg daily), eicosapentaenoic acid (900 mg b.i.d.), atorvastatin (10 mg/day), lansoprazole (30 mg/day), alfacalcidol (0.25 μg/day), andronate (35 mg/week), and calcium lactate (1 g b.i.d.). All medications except for prednisone were subsequently withheld, and warfarin was replaced by intravenous heparin. The liver test results normalized, but they deteriorated upon rechallenge with warfarin.
At this time, heparin therapy was briefly interrupted to permit a renal biopsy to evaluate refractory NS, with hypoalbuminemia (2.1 g/dl) and proteinuria (5.7 g/day). The biopsy showed MN at stage III of the Ehrenreich and Churg classification. Continued anticoagulation was deemed necessary due to severe persistent hypoalbuminemia and the severe cerebrovascular accident. Warfarin appeared to be clinically intolerable due to idiopathic hepatotoxicity despite a negative lymphocyte stimulation test. Intravenous heparin therapy was incompatible with outpatient rehabilitation.
We discussed the indication of dabigatran, the only available novel oral anticoagulant in Japan at that time, with the patient and also with cardiologists, a neurologist, and the vice-principal of our hospital, all of whom were responsible for the prescription of dabigatran, and we decided to initiate treatment. We also explained and discussed the risks and benefits of dabigatran with the patient and obtained his written and informed consent in advance. Oral dabigatran was administered at a dose of 110 mg b.i.d., and MN was treated with oral prednisone and cyclophosphamide according to Jindal's regimen [11]. Proteinuria decreased from a ratio of 5.1 to 1.6 g/g Cr, and serum albumin rose from 2.3 to 3.7 g/dl over 7 months. He was transferred to the rehabilitation hospital 137 days after stroke onset. At the outpatient clinic, his renal function remained stable, and he did not experience any episodes of edema, bleeding, or thromboembolism. We monitored the activated partial thromboplastin time (aPTT) to predict the risk of bleeding due to excessive anticoagulation. The aPTT values taken 4 h following administration of dabigatran in the morning were stable at around 40 s (standard aPTT was 30 s). A carotid echogram performed 2 years after the initiation of dabigatran revealed a reduced embolus, although it did still occlude the internal carotid.
Literature Review
The PubMed database was searched for related literature. The search parameters were ‘nephrotic syndrome’, ‘membranous nephropathy’, ‘stroke’, and ‘infarction’.
Twenty-one prior cases have been reported [5, 6, 7, 8, 9, 10] and are detailed in table 2, along with our case. The age of onset ranged from 19 to 59 years; 16/22 (72.7%) were male. Histopathology was MN in 5 cases (22.7%), minimal change in 4 cases (18.2%), membranoproliferative glomerulonephritis in 3 cases (13.6%), focal segmental sclerosis in 2 cases (9.0%), diabetic nephropathy in 1 case (4.5%), and IgA nephropathy in 1 case (4.5%). Pathologic diagnosis was undetermined in 6 cases (27.2%). The middle cerebral artery territory was most frequently involved; the left and right middle cerebral artery areas were involved in 8 cases (36.4%) and 5 cases (22.7%), respectively. Carotid occlusion, as in our patient, was reported twice.
Table 2.
Characteristics of case reports of cerebral infarction associated with nephrotic syndrome
| Age, years | Gender | Site of infarction | Renal pathology | First author | Year |
|---|---|---|---|---|---|
| 28 | Male | Left MCA area | MN | Fuh [15] | 1992 |
| 34 | Male | Left MCA area | MN | Marsh [16] | 1991 |
| 35 | Male | Left ICA occlusion | MN | Present case | 2013 |
| 37 | Male | Left MCA area | MN | Chauturvedi [17] | 1993 |
| 59 | Male | Bilateral occipital lobe | MN | Ogawa [18] | 1999 |
| 21 | Male | Left basal ggl to internal capsule | MC | Fuh [15] | 1992 |
| 23 | Male | Left MCA area | MC | Parag [19] | 1990 |
| 34 | Male | ICA occlusion | MC | Navascués [9] | 2006 |
| 42 | Female | Right ACA+MCA area | MC | Pandian [20] | 2000 |
| 29 | Female | Right ICA | MPGN | Maruyama [21] | 1995 |
| 36 | Male | Left MCA area | MPGN | Marsh [16] | 1991 |
| 39 | Male | Right frontal lobe | MPGN | Song [22] | 1994 |
| 19 | Female | Right anterior choroidal artery | FSGS | Izumi [5] | 1998 |
| 53 | Male | Right MCA area | FSGS | Yun [6] | 2004 |
| 35 | Female | Left temporal and parietal lobe | IgAN | Lee [23] | 2000 |
| 26 | Female | Multiple emboli | DMN | Huang [24] | 1995 |
| 23 | Male | Right M1 occlusion | Unknown | Sekiguchi [25] | 1990 |
| 51 | Male | Right MCA area | Unknown | Fritz [26] | 1992 |
| 28 | Female | Right MCA area | Unknown | Kotani [7] | 1997 |
| 47 | Male | Left MCA occlusion | Unknown | Naganuma [8] | 2003 |
| 42 | Male | Left ICA occlusion | Unknown | Wiroteurairueng [10] | 2007 |
| 52 | Male | Left corona radiata | Unknown | Miyamoto [27] | 1989 |
The cases are listed in the order of commonly seen underlying renal pathology. AC = Anterior cerebral artery; DMN = diabetic nephropathy; FSGS = focal segmental glomerulosclerosis; ICA = internal carotid artery; IgAN = IgA nephropathy; M1 = M1 portion of middle cerebral artery; MC = minimal change; MCA = middle cerebral artery; MPGN = membranoproliferative glomerulopathy; ggl = ganglia.
Discussion
Hypercoagulability contributes to the predisposition to thromboembolism in NS. Hypercoagulability of NS seems to be caused by an imbalance of prothrombotic and antithrombotic factors [1]. The loss of low molecular weight coagulation factors (factor IX, XI), antithrombin III, plasminogen, and free protein S in the urine, along with inverse increases in high molecular weight procoagulant cofactors (factor V, VII, VIII, and X), fibrinogen, alpha 2-antiplasmin, alpha-2-macroglobulin, diphosphate, and platelet aggregation have been reported as causes of this predisposition [6].
Risk factors for thromboembolic complications include a history of thromboembolism, pre-existing coagulation disorders, genetic predisposition, severity of hypoalbuminemia, volume depletion, use of diuretics or glucocorticoids, venous stasis, immobilization, and underlying renal pathology [1]. The role of conventional atherosclerotic risk factors in the promotion of arterial thromboembolism is not well defined. At any rate, our patient sustained a major stroke with relatively few conventional risk factors. MN is considered to present a particularly high risk for hypercoagulable states, but venous or arterial thromboembolism has also been reported in membranoproliferative glomerulonephritis and minimal change. As with our patient, most of the reported cases sustained stroke at earlier ages than patients with atherosclerosis and atrial fibrillation – this suggests that NS may independently predispose individuals to arterial and venous thromboembolism.
Before we will get down to the discussion of anticoagulation with dabigatran, we would like to explain our management of two controversial issues: the use of glucocorticoids and the switching of the immunosuppressants.
First, because the use of glucocorticoids is a well-known risk factor for thromboembolic complications of NS, our early administration of a high-dose glucocorticoid may be somewhat controversial [1]. However, we believed that the early initiation of a glucocorticoid would contribute to a better outcome because the patient might develop further thromboembolic complications as long as he remained nephrotic. The fact that an arterial embolus had developed without the existence of a glucocorticoid seems to support our supposition.
Second, switching cyclosporine to cyclophosphamide may also be considered controversial. Although it may have been too early to regard cyclosporine as ineffective, we thought that the rapid remission of NS was essential for the patient because the efficacy and safety of anticoagulation with dabigatran for an extended period has not been established. We therefore initiated the strongest available treatment option for MN.
The optimal, standardized approach to the prophylaxis and management of thromboembolic complications associated with NS has not been established. Anticoagulation with sequential heparin and oral warfarin may represent the de facto standard of treatment due to the traditional frequency of use [1]. Experience with subcutaneous heparin is limited due to practical issues (e.g., patient inconvenience, difficulty with monitoring), and no alternative oral anticoagulants have yet been established.
Dabigatran is a direct competitive thrombin inhibitor that can be given orally. Dabigatran has been a substitute for warfarin in the prophylaxis of stroke associated with nonvalvular atrial fibrillation [3] and has been used for postoperative thromboprophylaxis in orthopedic patients in Canada and Europe [4]. A recent study showed the noninferiority of dabigatran compared to warfarin for treatment of acute venous thromboses [12].
The dosage of dabigatran is an important point of discussion. Dabigatran is generally used at 150 mg b.i.d. A lower dosage of 110 mg b.i.d. is indicated for patients with a higher risk of bleeding, such as elderly patients, patients with renal impairment (more than 30 ml/min of creatinine clearance), and patients with a previous history of gastrointestinal bleeding. Although our patient was only 35 years old and without any risk for bleeding, we treated him with 110 mg b.i.d., the dosage indicated for patients with a higher risk of bleeding. Maximal caution was taken because of the lack of previous experiences with dabigatran, the limited information on drug interactions (especially between immunosuppressants), and the lack of health insurance coverage available in Japan. While it might be better to use a regular dose (150 mg b.i.d.) of the agent, we considered safety to be the first priority due to the reasons discussed above at the time of initiation. Given that the embolus persists, an increase in the dosage of the agent should be considered.
Usability without regular monitoring is an outstanding feature of novel anticoagulants such as dabigatran. However, the necessity of monitoring has been discussed recently because fatal hemorrhagic complications associated with dabigatran have been reported, especially in elderly patients and patients with renal impairment. A value of aPTT is reportedly well correlated with the risk of bleeding associated with anticoagulation due to dabigatran, although it is not correlated with the efficacy of the agent. An aPTT value of more than 70–80 s measured at the dabigatran trough is associated with major bleeding [3]. In an outpatient situation, the measurement of aPTT at the trough might be difficult to accomplish. In fact, we measured aPTT 4 h following the administration of dabigatran instead of at the trough. We believed that measurement at any time could be tolerated because the difference between the values of aPTT measured at the peak (2 h following oral administration of dabigatran) and the values of aPTT measured at the trough was reportedly less than 10 s [13]. Thus, an aPTT value of less than 80 s at any time would be, to some extent, indicative of safety. Nevertheless, a more effective parameter of excessive anticoagulation is warranted because of the low specificity and the lack of standardized reagents of aPTT measurement [14].
Our patient was safely treated for carotid thromboembolism with dabigatran rather than warfarin. Although the embolus persisted, we believe that dabigatran worked to diminish the embolus and, at the very least, contributed as thromboprophylaxis without any complications. This indicates the potential utility of dabigatran in patients with thromboembolic complications associated with NS. We need to carefully consider the indication of dabigatran for thromboembolic complications associated with NS because the indication is currently off-label in all countries. Appropriate explanation and discussion with the patients and careful monitoring for the risk of hemorrhagic complications due to excessive anticoagulation such as regular aPTT measurement will also be required in clinical settings. Although warfarin will remain the first-line anticoagulant for this situation, dabigatran offers a good treatment option for patients who are intolerant of warfarin. Further research is needed to clarify the safety and efficacy of dabigatran for patients with NS.
Disclosure Statement
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Acknowledgments
We thank Dr. Rita Lynn McGill (Department of Nephrology, Allegheny General Hospital, Pittsburgh, Pa., USA), Dr. Tomoatsu Sakuda (Sakuda Medical Clinic, Okinawa, Japan) and Dr. Takuji Seo (Department of Medicine, Kameda Medical Center, Chiba, Japan) for their significant contributions to this paper.
References
- 1.Glassock RJ. Prophylactic anticoagulation in nephrotic syndrome: a clinical conundrum. J Am Soc Nephrol. 2007;18:2221–2225. doi: 10.1681/ASN.2006111300. [DOI] [PubMed] [Google Scholar]
- 2.Kodner C. Nephrotic syndrome in adults: diagnosis and management. Am Fam Physician. 2009;80:1129–1134. [PubMed] [Google Scholar]
- 3.Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgre J, Prekh A. Dabigtran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139–1151. doi: 10.1056/NEJMoa0905561. [DOI] [PubMed] [Google Scholar]
- 4.Gomez-Outes A, Terleira-Fernandez AI, Suarez-Gea ML, Vargas-Castriiion E. Dabigatran, rivaroxaban, or apixaban versus enoxaparin for thromboprophylaxis after total hip or knee replacement: systematic review, meta-analysis, and indirect treatment comparisons. BMJ. 2012;14:e3675. doi: 10.1136/bmj.e3675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Izumi M, Terao S, Nakamori T, Inoue H, Mitsuma T, Yamada H, Nakayama M. Cerebral infarction associated with nephrotic syndrome in a young adult: a case report (in Japanese) No To Shinkei. 1998;50:1119–1124. [PubMed] [Google Scholar]
- 6.Yun YW, Chung S, You S-J, Lee DK, Lee K-Y, Han S-W, Jee HO, Kim HJ. Cerebral infarction as a complication of nephrotic syndrome: a case report with a review of the literature. J Korean Med Sci. 2004;19:315–319. doi: 10.3346/jkms.2004.19.2.315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kotani K, Kawano M. A young female with marked hyperlipoprotein(a)emia associated with nephrotic syndrome and stroke. J Atheroscler Thromb. 1997;12:234. doi: 10.5551/jat.12.234. [DOI] [PubMed] [Google Scholar]
- 8.Naganuma M, Sugimoto R, Hashimoto Y, Matsuura Y, Terasaki S, Uchino M. A case of brain infarction with nephrotic syndrome (in Japanese) Rinsho Shinkeigaku. 2003;43:126–129. [PubMed] [Google Scholar]
- 9.Navascués RÁ, Ojea BD, Marín R. Trombosis carotída como primera manifestacion de un sindrome nefrotico a cambios minimos. Nefrologia. 2006;26:497–498. [PubMed] [Google Scholar]
- 10.Wiroteurairueng T, Poungvarin N. Orbital infarction syndrome in nephrotic syndrome patient with extensive carotid arteries occlusion. J Med Assoc Thai. 2007;90:2499–2505. [PubMed] [Google Scholar]
- 11.Jindal K, West M, Bear R, Goldstein M. Long-term benefits of therapy with cyclophosphamide and prednisone in patients with membranous glomerulonephritis and impaired renal function. Am J Kidney Dis. 1992;19:61–67. doi: 10.1016/s0272-6386(12)70204-4. [DOI] [PubMed] [Google Scholar]
- 12.Schulman S, Kearon C, Kakkar AK, Mismetti P, Schellong S, Eriksson H, Baanstra D, Schnee J, Goldhaber SZ. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361:2342–2352. doi: 10.1056/NEJMoa0906598. [DOI] [PubMed] [Google Scholar]
- 13.Suzuki S, Otsuka T, Sagara K, Matsuno S, Funada R, Uejima T, Oikawa Y, Yajima J, Koike A, Nagashima K, Kirigaya H, Sawada H, Aizawa T, Yamashita T. Dabigatran in clinical practice for atrial fibrillation with special reference to activated partial thromboplastin time. Circ J. 2012;76:755–757. doi: 10.1253/circj.cj-11-1335. [DOI] [PubMed] [Google Scholar]
- 14.Stangier J, Rathgen K, Stahle Hildegard, Gansser D, Roth W. The pharmacokinetics, pharmacodynamics and tolerability of dabigatran etexilate, a new oral direct thrombin inhibitor, in healthy male subjects. Br J Clin Pharmacol. 2007;64:292–303. doi: 10.1111/j.1365-2125.2007.02899.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fuh JL, Teng MM, Yang WC, Liu HC. Cerebral infarction in young men with nephrotic syndrome. Stroke. 1992;23:295–297. doi: 10.1161/01.str.23.2.295. [DOI] [PubMed] [Google Scholar]
- 16.Marsh EE, 3rd, Biller J, Adams HP, Kaplan JM. Cerebral infarction in patients with nephrotic syndrome. Stroke. 1991;22:90–93. doi: 10.1161/01.str.22.1.90. [DOI] [PubMed] [Google Scholar]
- 17.Chaturvedi S. Fulminant cerebral infarctions with membranous nephropathy. Stroke. 1993;24:473–475. doi: 10.1161/01.str.24.3.473. [DOI] [PubMed] [Google Scholar]
- 18.Ogawa M, Tsukahara T, Saisho H. Nephrotic syndrome with acute renal failure and cerebral infarction in a patient with myasthenia gravis. Am J Nephrol. 1999;19:622–623. doi: 10.1159/000013512. [DOI] [PubMed] [Google Scholar]
- 19.Parag KB, Somers SR, Seedat YK, Byrne S, Da Cruz CM, Kenoyer G. Arterial thrombosis in nephrotic syndrome. AM J Kidney Dis. 1990;15:176–177. doi: 10.1016/s0272-6386(12)80516-6. [DOI] [PubMed] [Google Scholar]
- 20.Pandian JD, Sarada C, Elizabeth J, Visweswaran RK. Fulminant cerebral infarction in a patient with nephrotic syndrome. Neurol India. 2000;48:179–181. [PubMed] [Google Scholar]
- 21.Maruyama M, Yanagimoto S, Mruyama T, Hayakawa I, Kamijima G. Bilateral internal carotid artery occlusion in a young patient with nephrotic syndrome. Acta Med Biol. 1995;43:117–120. [Google Scholar]
- 22.Song KS, Won DI, Lee AN, Kim CH, Kim JS. A case of nephrotic syndrome associated with protein S deficiency and cerebral thrombosis. J Korean Sci. 1994;9:347–350. doi: 10.3346/jkms.1994.9.4.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee CH, Chen KS, Tsai FC, Chien YY, Lee N. Concurrent thrombosis of cerebral and femoral arteries in a patient with nephrotic syndrome. Am J Nephrol. 2000;20:483–486. doi: 10.1159/000046204. [DOI] [PubMed] [Google Scholar]
- 24.Huang TY, Chau KM. Biventricular thrombi in diabetic nephrotic syndrome complicatedby cerebral embolism. Int J Cardiol. 1995;150:193–196. doi: 10.1016/0167-5273(95)02366-5. [DOI] [PubMed] [Google Scholar]
- 25.Sekiguchi Y, Senoo H, Gomyo Y, Yada K. A case of cerebral infarction in a young patient with nephrotic syndrome (in Japanese) Gencho Hiroshima Igaku. 1990;43:791–794. [Google Scholar]
- 26.Fritz C, Braune HJ. Cerebral infarction and nephrotic syndrome. Stroke. 1992;23:1380–1381. doi: 10.1161/01.str.23.9.1380. [DOI] [PubMed] [Google Scholar]
- 27.Miyamoto T, Iwamoto T, Katsunuma H, Yada H, Nukaga S, Ibukiyama C. A case of cerebral thrombosis occurred during the course of nephrotic syndrome (in Japanese) Tokyo Ikadaigaku Zasshi. 1969;47:1067–1070. [Google Scholar]