Abstract
All-trans retinoic acid (ATRA) resistance has been a critical problem in acute promyelocytic leukemia (APL) relapsed patients. In ATRA resistant APL cell lines NB4-R1 and NB4-R2, the combination of staurosporine and ATRA synergized to trigger differentiation accompanied by significantly enhanced protein level of CCAAT/enhancer binding protein ε (C/EBPε) and C/EBPβ as well as the phosphorylation of mitogen-activated protein (MEK) and extracellular signal-regulated kinase (ERK). Furthermore, attenuation of the MEK activation blocked not only the differentiation but also the increased protein level of C/EBPε and C/EBPβ. Taken together, we concluded that the combination of ATRA and staurosporine could overcome differentiation block via MEK/ERK signaling pathway in ATRA-resistant APL cell lines.
Since the introduction of all-trans retinoic acid (ATRA) and arsenic trioxide (As2O3) in the conventional chemotherapy of acute promyelocytic leukemia (APL), the prognosis of APL has significantly improved, approximately 90% of patients achieved 5-year disease free remission1. However, ATRA resistance has still been a critical problem in relapsed patients,especially the patients consolidated with ATRA-containing treatment. Several mechanisms of ATRA resistance have been speculated by early studies including an increased oxidative catabolism of ATRA by cytochrome P450 enzymes2 or P-glycoprotein3, reduced cellular ATRA concentration by an enhanced level of cellular RA-binding protein (CRABP)4,5. However, more and more clinical observations and in vitro studies confirmed that the defects of the pathogenic fusion gene of APL, promyelocytic leukemia-retinoid acid receptorα (PML-RARα), particularly genetic mutations in the ligand binding domain (LBD) of the RARα region resulting in deficient ATRA binding was the major cause of ATRA resistance6,7,8,9,10,11,12,13,14,15. Furthermore, altered ligand-induced co-repressor release, co-activator recruitment and impaired transcriptional activation of genes with the retinoic acid response elements (RAREs) sites were associated with ATRA resistance16,17. Besides PML-RARα, mutations in other genes, such as FLT3-ITD or TP53 might also contribute to ATRA resistance18,19.
As2O3 was demonstrated to be effective in the treatment of relapsed APL patients, achieving complete remission (CR) rate of more than 80%20. Although adverse effects of As2O3 were relatively rare, it still had severe side effects with long-term use. Moreover, clinical resistance to As2O3 was also observed21. Histone deacetylase (HDAC) enzymes are the essential components of the co-repressor complex that promoted chromatin condensation and reduced gene transcription. HDAC inhibitor was confirmed to restore sensitivity to ATRA by in vitro study22. However, clinical trial showed that the addition of HDAC inhibitor to ATRA was only effective in very limited number of ATRA-resistant APL patients12,23. Since HDAC has a dual role in tumorigenesis, behaving as oncosuppressor during tumor initiation while performing as oncogene in tumor maintenance, clinical use of HDAC inhibitor should take in caution24. Allogeneic bone marrow transplantation was another therapy choice, but only applicable to small amount of patients. Therefore, new therapy approaches to ATRA resistant APL patients were required to be developed. Indeed, other agents such as cAMP, granulocyte colony-stimulating factor, tumor necrosis factor, oridonin, tyrosine kinase inhibitor STI571 and interferon-γ have been shown to cooperate with ATRA to induce differentiation in ATRA resistant APL cells25,26,27,28,29,30.
Protein kinase C (PKC) is a family of serine/threonine kinases, consisting of 13 isozymes which play a crucial role in signaling transduction of various cell functions including proliferation, differentiation, apoptosis, cell migration and gene expression. Intensive studies has explored its contribution to carcinogenesis and rendered it an attractive target for cancer therapy31. ATRA either activated or suppressed certain PKC isozyme activity in different cell lines32,33,34,35. In APL cells, PKCδ was activated by ATRA. However, the role of activated PKCδ in ATRA-induced differentiation in APL cells was quite controversial36,37. Activated PKCδ was confirmed to be required for ATRA-induced differentiation36. On the contrary, McNamara S et al37 demonstrated that activated PKCδ could increase protein stability and activity of toposomerase IIβ, resulting in RARα transcription repression and RA resistance. Another PKC isoform, PKCα was suggested to negatively modulate terminal neutrophil differentiation38.
To investigate whether inhibition of PKC could reverse ATRA resistance, we explored the effect of the combination of staurosporine, one of the most powerful but less specific PKC inhibitors with ATRA in some ATRA-resistant APL cell lines, NB4-R1 and NB4-R2. Neither staurosporine nor ATRA could trigger differentiation in ATRA-resistant APL cell lines. Surprisingly, the combination of ATRA and staurosporine could overcome retinoid resistance in these cell lines. The protein level of CCAAT/enhancer binding protein ε (C/EBPε) and C/EBPβ as well as the phosphorylation of mitogen-activated protein (MEK) and extracellular signal-regulated kinase (ERK) was enhanced by the combined treatment. Moreover, MEK/ERK signaling pathway was demonstrated to be required for the combination-induced differentiation.
Results
Combination of staurosporine and ATRA induced granulocytic differentiation accompanied by proliferation inhibition in NB4-R1 and NB4-R2 cells
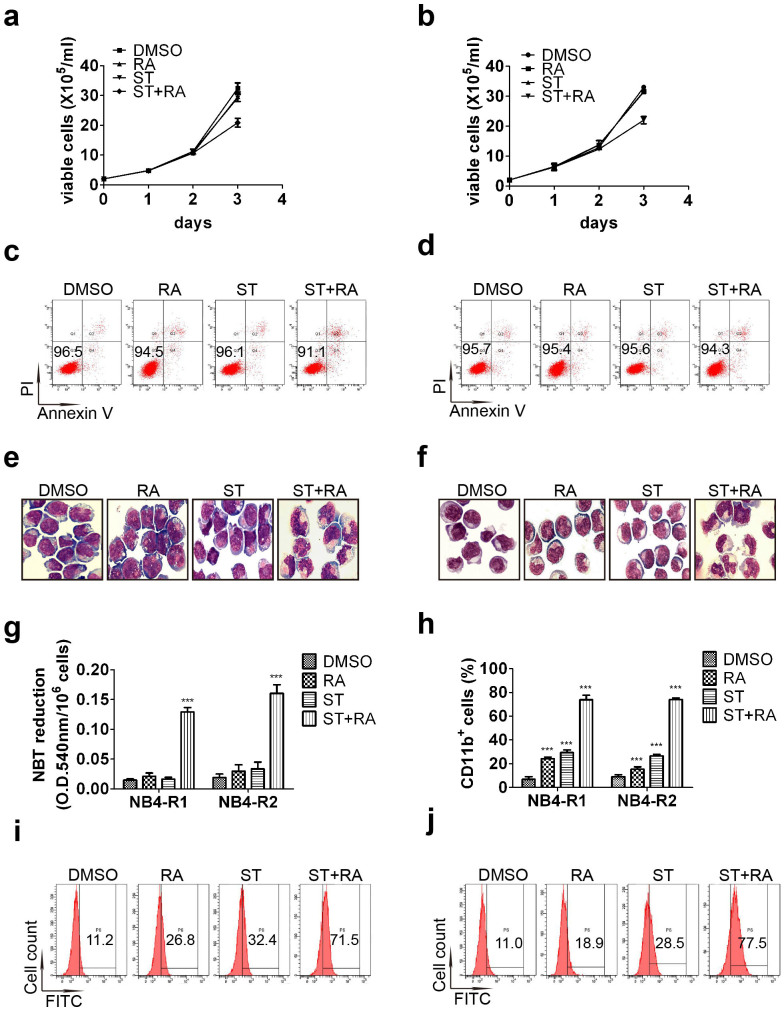
To investigate the effect of combined treatment of staurosporine and ATRA on NB4-R1 and NB4-R2 cells, we first tested the concentration of staurosporine studied in both cell lines. DMSO treatment was regarded as control since both ATRA and staurosporine were dissolved in it. 2 nM staurosporine was determined to be used since there were no obvious effects on cell proliferation (Figure 1a and 1b) and survival at such concentration (Figure 1c and 1d). The combined treatment suppressed cell growth at the third day (Figure 1a and 1b). The proliferation inhibition rate calculated as Methods mentioned was 36.1 ± 3.1% for NB4-R1 cells and 34.5 ± 2.7% for NB4-R2 cells. However, cell viability maintained above 90% in these two cell lines with any treatment for 72 hours. Annexin-V assay also showed that more than 90% cells were PI negative and Annexin-V negative even with combined treatment (Figure 1c and 1d). Thus, the combined treatment had no effect on cell survival but only inhibited proliferation in these two cell lines.
Figure 1. Effects of combined treatment on the growth, survival and differentiation in NB4-R1 and NB4-R2 cells.
Cell growth of NB4-R1 (a) and NB4-R2 (b) treated with 2 nM staurosporine(ST) or/and 1 μM ATRA(RA) was calculated as mentioned in Methods. One representative experiment was shown. Each value represented the mean ± SD of triplicate samples. Similar results were obtained in three independent experiments. Annexin-V assay of NB4-R1 (c) and NB4-R2 cells (d) treated with ST or/and RA for 72 hours. The percentages of viable (PI and Annexin V both negative) cells were shown in the corresponding panels. Results were representative among three independent experiments. Representative morphologic analysis of NB4-R1 (e) and NB4-R2 cells (f) treated with the indicated drugs for 72 hours. Similar results were obtained in three independent experiments. Differentiation was also evaluated by NBT reduction assay (g) and flow cytometric analysis of CD11b expression in NB4-R1 and NB4-R2 cells (h) with the indicated treatment for 72 hours. Each value represented the mean ± SD of three independent measurements. ***, P < 0.001 as compared with DMSO. The representative histogram of flow cytometric analysis of CD11b expression in NB4-R1 (i) and NB4-R2 cells (j) with the indicated treatment for 72 hours were also shown. The percentages of CD11b positive cells were shown in the corresponding panels.
Both cell lines had a characteristic morphology of APL blast such as irregular nucleus and large nuclear/cytoplasm ratio (Figure 1e and 1f). After incubated with 2 nM staurosporine or 1 μM ATRA alone for 72 hours, no significantly morphological change was observed in NB4-R1 cells (Figure 1e). Meanwhile, most of the NB4-R2 cells retained the morphology of APL blast with any single treatment while some cells presented decreased nuclear/cytoplasm ratio with ATRA alone (Figure 1f). However, when simultaneously treated with staurosporine and ATRA for 72 hours, both cell lines displayed appearances of matured granulocytes, such as lobed nuclei accompanied by markedly decreased nuclear/cytoplasm ratio (Figure 1e and 1f). Nitroblue tetrazolium (NBT) reduction activity did not increase remarkably with any single treatment in both cell lines (Figure 1g). In addition, the expression of CD11b was somewhat enhanced with any single treatment in NB4-R1 and NB4-R2 cells (Figure 1h–1j). However, with the combination of staurosporine and ATRA, a more than additive effect was observed. The combined treatment significantly increased the percentage of CD11b positive cells in both cell lines (ST + RA compared with DMSO, 73.9 ± 6.6% vs 6.9 ± 1.3%, x2 = 9321.63, P < 0.001 in NB4-R1 cells and 75.1 ± 2.3% vs 9.0 ± 1.6%, x2 = 8965.07, P < 0.001 in NB4-R2 cells, Figure 1h–1j). Accordingly, prominently enhanced NBT reduction activity was observed in both cell lines with combined treatment (ST + RA compared with DMSO, 0.129 ± 0.006 vs 0.014 ± 0.002, P = 0.0007 in NB4-R1 cells and 0.161 ± 0.014 vs 0.018 ± 0.003, P = 0.0006 in NB4-R2 cells, Figure 1g). Thus, these results demonstrated that the combined treatment induced granulocytic differentiation and indicated that the combination of staurosporine and ATRA could overcome maturation block in ATRA-resistant APL cells.
Modulation of C/EBPs and PML-RARα as well as activation of MEK/ERK by the combined treatment
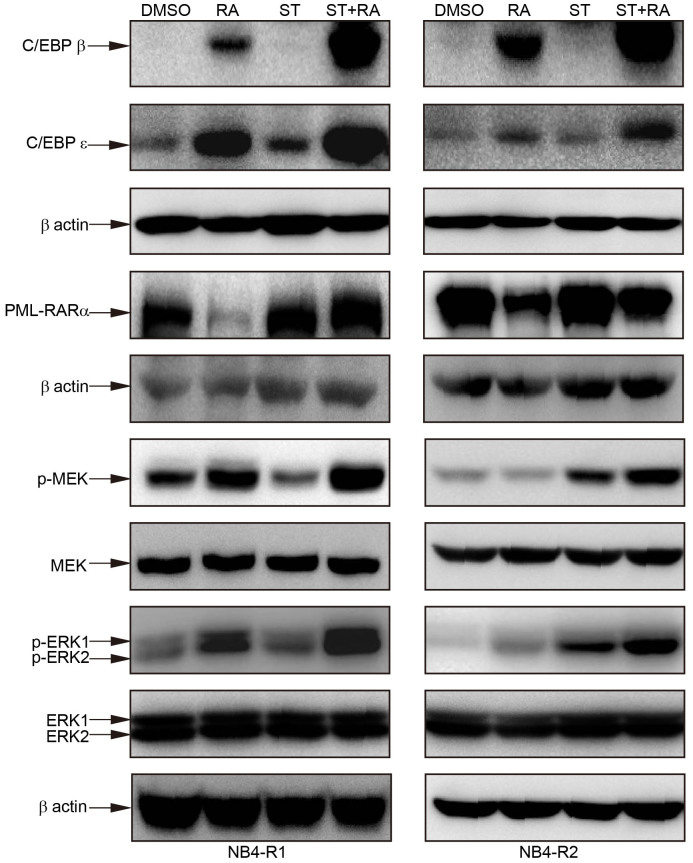
To explore the molecular mechanisms of combined treatment-induced differentiation, we first examined several proteins involved in granulocytic differentiation, especially which are critical for the promyelocyte to granulocyte transition. The induction of CCAAT/enhancer binding protein β (C/EBPβ) and C/EBPε expression is implicated in the later stage of granulocytic differentiation39,40. Therefore, cells were treated with staurosporine, ATRA, or staurosporine plus ATRA for 24 hours and the protein levels of C/EBPβ and C/EBPε were detected by immunoblotting. As shown in Figure 2, in NB4-R1 cells, ATRA but not staurosporine elevated C/EBPβ protein level while the cellular level of C/EBPε was enhanced by both single treatment. In NB4-R2 cells, only ATRA treatment increased the protein level of C/EBPβ and C/EBPε. However, the up-regulation of C/EBPβ and C/EBPε was much more remarkable with the combined treatment in both cell lines (Figure 2). These results suggested that C/EBPβ and C/EBPε may be responsible for the differentiation effect of combined treatment in ATRA-resistant APL cells.
Figure 2. Combined staurosporine and ATRA modulated the protein level of C/EBPβ, C/EBPε, PML-RARα as well as the phosphorylation of MEK and ERK.
Cells were treated with 2 nM staurosporine(ST), 1 μM ATRA(RA) alone and the combined treatment (ST + RA) for 24 hours. The protein level of C/EBPβ and C/EBPε was measured by Western blot analysis using 20 μg protein, while 50 μg protein was loaded for Western blot analysis of the phosphorylation of MEK and ERK1/2. The same membrane incubated with the phosphorylated Erk1/2 or MEK1/2 was stripped and followed by detection of MEK and ERK1/2. For Western blot analysis of PML-RARα, cells were treated with the indicated drugs for 72 hours and 20 μg protein was loaded. Since different amount of protein and diverse time points for collecting protein were used, each has the expression of β-actin as internal control. Similar results were obtained in three independent experiments. The full size blots were shown in the Supplemental Fig. S1 online.
The degradation of PML-RARα has been the crucial mechanism of ATRA-induced granulocytic differentiation of APL cells41. Treated with ATRA for 72 hours, the band corresponding to PML-RARα was almost disappeared in NB4-R1 cells or decreased in NB4-R2 cells (Figure 2). Meanwhile, the protein level of RARα was reduced in both cell lines with ATRA treatment (see Supplemental Fig. S1 online). Unexpectedly, the addition of staurosporine totally restored ATRA-mediated PML-RARα alteration in both cell lines (Figure 2). Interestingly, in NB4-R2 cells, staurosporine alone could increase the protein level of RARα and inhibit ATRA-induced reduction of RARα (see Supplemental Fig. S1 online). These data indicated that the cooperation differentiation effect of ATRA and staurosporine was independent of the regulation of PML-RARα.
Our previous study demonstrated that the non-genomic effect, MEK/ERK signaling pathway could positively regulate ATRA-induced differentiation in APL cells (unpublished data). To elucidate whether MEK/ERK signaling pathway was activated, the phosphorylated MEK and ERK1/2 were assessed by Western blot analysis in cells treated with ATRA or/and staurosporine for 24 hours. As illustrated in Figure 2, the combined treatment increased the amount of phosphorylation of MEK and ERK1/2 more significantly than that with any single treatment in both cell lines. The total amount of MEK and ERK1/2 in both cell lines remained almost unaltered after any treatment (Figure 2).
MEK/ERK activation was required for the combination-induced differentiation and the up-regulation of C/EBPs
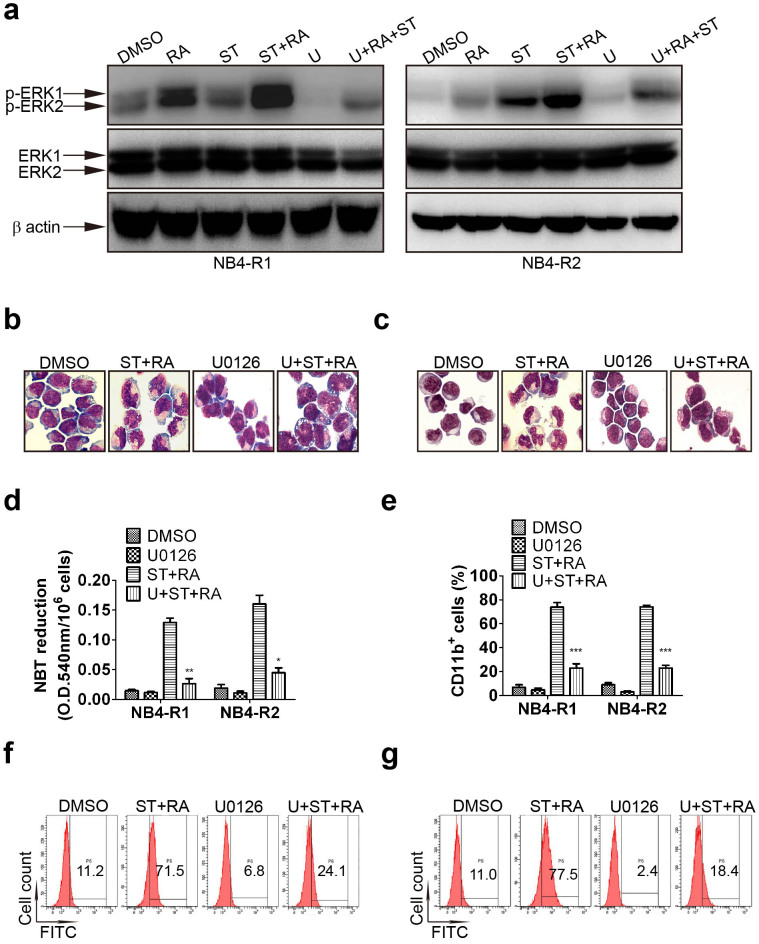
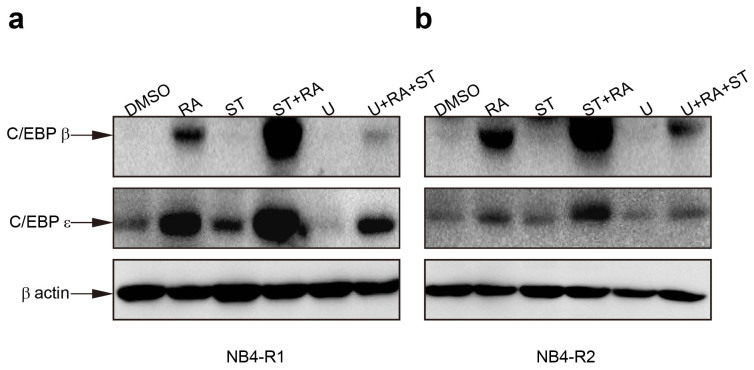
Having shown that the combination phosphorylated MEK and ERK, we tested whether MEK/ERK activation was necessary for ATRA and staurosporine-mediated differentiation. Cells were incubated with 1 μM U0126, a specific inhibitor of MEK for 1 hour prior to other treatments. The effectiveness of U0126 was evaluated by ERK1/2 phosphorylation. U0126 did attenuate ERK1/2 activation in these 2 cell lines (Figure 3a). Moreover, it abrogated cell differentiation triggered by the combined treatment in both cell lines as assessed by morphology (Figure 3b and 3c), NBT reduction assay (U + ST + RA compared with ST + RA, 0.026 ± 0.003 vs 0.129 ± 0.006, P = 0.0076 in NB4-R1 cells, 0.045 ± 0.008 vs 0.161 ± 0.014, P = 0.0199 in NB4-R2 cells Figure 3d) and CD11b expression (U + ST + RA compared with ST + RA, 23.0 ± 2.1% vs 73.9 ± 6.6%, x2 = 5186.60, P < 0.001 in NB4-R1 cells and 21.9 ± 2.6% vs 75.1 ± 2.3%, x2 = 5665.58, P < 0.001 in NB4-R2 cells, Figure 3e–3g). In addition, in the presence of U0126, the combined treatment-enhanced C/EBPβ and C/EBPε protein level were remarkably suppressed in both cell lines (Figure 4a and 4b). These results highlighted a major role of MEK/ERK signal pathway in the differentiation-inducing effect of ATRA and staurosporine in ATRA resistant-APL cells.
Figure 3. Inhibition of MEK activation blocked the combined treatment-induced differentiation.
Cells were exposed to 1 μM U0126 for 1 hour prior to other treatment. The attenuation of MEK activation by U0126 (U) was measured by Western blot analysis of phosphorylated ERK1/2 in NB4-R1 and NB4-R2 cells with indicated treatments for 24 hours (a). Similar results were obtained in three independent experiments. The full size blots were shown in the Supplemental Fig. S2 online. Inhibitory effect of U0126 on morphologic changes in NB4-R1 (b) and NB4-R2 cells (c) incubated with the indicated drugs for 72 hours. One representative experiment among three independent assays was shown. The inhibitory effect of U0126 on differentiation was also confirmed by NBT reduction assay (d) and flow cytometric analysis of CD11b expression in NB4-R1 and NB4-R2 cells (e) with the indicated drugs for 72 hours. Each value represented the mean ± SD of three independent measurements. * P < 0.05, ** P < 0.01, *** P < 0.001 indicated significant difference between U + ST + RA and ST + RA. The representative histograms of flow cytometric analysis of CD11b expression in NB4-R1 (f) and NB4-R2 cells (g) with the indicated drugs for 72 hours were also shown. The percentages of CD11b positive cells were shown in the corresponding panels.
Figure 4. Inhibition of MEK activation restored the protein levels of C/EBPs regulated by combined treatment.
Cells were exposed to 1 μmol/L U0126 for 1 hour prior to other treatments. The protein level of C/EBPβ and C/EBPε in NB4-R1 (a) and NB4-R2 (b) cells with the indicated drugs for 24 hours was determined by Western blot analysis. Expression of β-actin was assessed as internal control. Similar results were obtained in three independent experiments. The full size blots are shown in the Supplemental Fig. S3 online.
Discussion
In the present work, we demonstrated that the combination of staurosporine and ATRA could overcome granulocytic differentiation block in retinoid resistant APL cell lines. Staurosporine has been proven to be not only an effective apoptosis inducing agent but also a potent differentiation inducer in a variety of tumor cell lines42,43,44,45,46,47. To our knowledge, it is the first time to show that staurosporine can restore ATRA sensitivity in retinoid resistant APL cell lines. However, the effect of the combination on primary leukemia cells from retinoid resistant APL patients needs further investigation.
For the molecular mechanisms of the combination–induced differentiation, we focused on certain proteins or signaling pathways involving in granulocytic differentiation rather than PKC. C/EBPs are a family of transcription factors which regulate cell proliferation and differentiation. Among them, C/EBPα, C/EBPβ and C/EBPε are critical mediators of granulopoiesis. C/EBPα is required for the proceeding from myeloblast to promyelocyte while C/EBPβ and C/EBPε play central roles in terminal differentiation of granulocytes48. Moreover, C/EBPβ and C/EBPε were demonstrated to be required for ATRA-mediated differentiation in APL cells49,50. In addition, C/EBPε alone could suppress the leukemia phenotype of APL and imitate the differentiation-induction effect of ATRA in APL mouse model50. Thus, C/EBPβ and C/EBPε are not only the crucial regulators of granulocyte development but also the potential therapy target for leukemia. In this work, compared with any single treatment, the combination of staurosporine and ATRA remarkably increased the protein levels of both C/EBPβ and C/EBPε in NB4-R1 and NB4-R2 cells. Though any single treatment to some extent did up-regulate C/EBPβ or C/EBPε, we speculated that such increase might not be enough to trigger maturation but only combined treatment resulting in significant up-regulation of C/EBPβ and C/EBPε could speed up terminal differentiation.
It was well known that co-repressor release, co-activator recruitment, transcriptional activation of genes with the RAREs and PML-RARα degradation were principal mechanisms of ATRA-induced differentiation of APL cells. However, it was indicated that pre-genomic activation of kinase signaling cascades also contributed to ATRA-induced differentiation51,52. Our previous studies showed that MEK/ERK was activated during ATRA-induced granulocytic differentiation in APL cell line, NB4. Furthermore, inhibition of MEK activation suppressed almost all differentiation induced by ATRA, suggesting the significant role of MEK/ERK signaling pathway in ATRA-dependent NB4 cells granulocytic differentiation (unpublished data). Interestingly, the activation of MEK/ERK signaling pathway was also observed in the combination treatment. Moreover, attenuation of the MEK activation inhibited not only the differentiation but also the increased protein level of C/EBPε and C/EBPβ. Therefore, the protein level of C/EBPε and C/EBPβ was regulated by MEK/ERK signaling pathway and the later was required for the combination-induced differentiation. Accumulating evidence showed that MEK/ERK signaling pathway could promote C/EBPβ expression and modify its activity53,54,55. Moreover, in ATRA-treated APL cells, as the result of increasing protein level of C/EBPβ, enhanced C/EBPβ binding activity preceded that of C/EBPε and C/EBPβ itself could induce the expression of C/EBPε49. Hence, there might be MEK-C/EBPβ-C/EBPε cascade in the combination-induced differentiation.
PML-RARα fusion protein is believed to contribute to the pathogenesis of APL in a dominant negative fashion, disrupting the normal physiologic function of PML and repressing transcription by RARs. Either As2O3 or ATRA is regarded as one of the most successful examples of tumor targeted therapy since they both degraded APL pathogenic protein, PML-RARα. However, in this work, though staurosporine inhibited ATRA-mediated PML-RARα degradation, the combination of staurosporine and ATRA still synergistically induced differentiation. It meant that the combination could induce differentiation independent of PML-RARα degradation, suggesting that the degradation of PML-RARα might not be necessary for APL therapy. In consistent with our observation, other agents such as oridonin, G-CSF, STI571, pharicin B and TNF could cooperate with ATRA to induce differentiation in ATRA-resistant APL cell lines without any effect on PML-RARα26,27,28,29,56. The increased protein level of C/EBPε and C/EBPβ by ATRA treatment was confirmed to depend on PML-RARα expression49,50. Therefore, it remains to be further explored whether staurosporine prevented PML-RARα degradation contributes to the enhanced protein level of C/EBPε and C/EBPβ as well as the combination–induced differentiation.
In conclusion, the combination of staurosporine and ATRA synergized to trigger differentiation in ATRA resistant APL cell lines by MEK/ERK signaling pathway. It remains to investigate whether such combination could induce differentiation in primary leukemia cells from retinoid resistant APL patients. The exact molecular mechanisms of how staurosporine overcomes ATRA resistance as well as more detailed mechanisms of the combination-induced differentiation need to be further elucidated.
Methods
Reagents
ATRA was purchased from Sigma-Aldrich (St Louis, MO) while both staurosporine and U0126 were obtained from EMD Chemicals (San Diego, CA). They were all dissolved by dimethylsilfoxide (DMSO) as a stock solution at 1 mM, 2 μM and 1 mM respectively.
Cell culture, cell viability and cell proliferation
The ATRA resistant cell lines, NB4-R1 and NB4-R2 cells were obtained from Dr Michel Lanotte (Hopital Saint Louis, Paris, France)57 and were cultured in RPMI-1640, supplemented with 10% fetal calf serum (Thermo Fisher Scientific Inc, Waltham, MA) in a humidified atmosphere of 95% air/5% CO2 at 37°C. To avoid possible effects of cell density on cell growth and survival, cells were maintained at less than 5 × 105 cells/ml. Cell viability was assayed by trypan-blue exclusion. Actual viable cell numbers were calculated by multiplying diluted times with counted viable cell numbers. Proliferation inhibition rate (%) = (control group actual viable cell numbers-experimental group actual viable cell numbers)/control group actual viable cell numbers ×100%.
Cell differentiation assays
Cell maturation was evaluated by cellular morphology, NBT reduction assay and the content of cell surface differentiation-related antigen CD11b. Morphology was determined with May-Grunwald-Giemsa's staining of cells centrifuged onto slides by cytospin (Shandon, Runcon, UK; 500 r.p.m., 5 min) and viewed at ×1,000 magnification. NBT reduction was performed as previously described58. Briefly, 1 × 106 cells were collected and incubated with 1 mg/ml NBT (Sigma-Aldrich) solution containing 10 μM phorbol 12-myristate 13-acetate (Sigma-Aldrich) at 37°C for 1 hour. Cells were lysed by 10% sodium dodecyl sulfate (SDS) and 0.04 N hydrochloric acid (HCl). The absorbance at O.D 540 nm was detected by spectrophotometer (Beckman Coulter, Brea, CA). The expression of cell surface differentiation-related antigen CD11b was determined on flow cytometry (EPICS XL, Coulter, Hialeah, FL). Fluorochrome-labeled anti-human CD11b/FITC antibody was purchased from Coulter-Immunotech (Marseilles, France).
Annexin-V analysis
Annexin-V assay was performed according to instructions provided in the Annexin V-PI Apoptosis Detection Kit (BD Biosciences Pharmingen, San Diego, CA). Briefly, 5 × 105 cells were harvested and washed with binding buffer provided in the kit. Then, cells were incubated with 5 μl annexin-V and 10 μl propidium iodide (PI) at room temperature in the dark for 15 min. Fluorescent intensities was determined on flow cytometry (Coulter).
Western blot analysis
Cells were washed with phosphate-buffered saline (PBS) and lysed with RIPA buffer (Sigma-Aldrich). Cell lysates were centrifuged at 13,000 rpm for 10 minutes at 4°C. Supernants were collected and quantified by Bio-Rad Dc protein assay (Bio-Rad Laboratories, Hercules, CA). 20 or 50 μg protein extracts were loaded on 8% SDS-polyacrylamide gel, subjected to electrophoresis, and transferred to polyvinylidene difluoride membranes (GE Healthcare UK Ltd, Buckinghamshire, UK). After blocking with 5% nonfat milk in PBS, the membranes were probed with antibodies against RARα (Santa Cruz Biotech, Santa Cruz, CA), C/EBPβ (Santa Cruz Biotech), C/EBPε (Santa Cruz Biotech), anti-β-actin (Santa Cruz Biotech), phosphor-p44/42 Erk1/2 (Thr202/Try 204) (Cell Signaling Technology, Beverly, MA) and phosphor-MEK1/2 (Ser217/Try 221) (Cell Signaling Technology). Subsequently, membranes were incubated with horseradish peroxidase (HRP)-conjugated secondary antibody (GE Healthcare UK Ltd). Immunocomplexes were visualized by chemiluminescence kit (GE Healthcare UK Ltd) according to the manufacturer's instruction. To detect Erk1/2 and MEK1/2, the same membrane incubated with the phosphorylated Erk1/2 or MEK1/2 was stripped with stripping buffer (2% SDS, 100 mM beta-mercaptoethanol, 50 mM Tris, pH6.8) followed by blocking and probing with anti-Erk1/2 (Cell Signaling Technology) or anti-MEK1/2 (Cell Signaling Technology).
Statistical analysis
For NBT reduction, two-tailed paired Student's t test was used, n value was 3. The flow cytometric analysis of CD11b was analyzed by chi-square test, n value was 20,000.
Author Contributions
D.Z.G. and Y.S. carried out the experiments. D.Z.G. prepared all the figures. X.C designed the study and wrote the manuscript. All authors reviewed the manuscript.
Supplementary Material
Combined staurosporine and retinoic acid induces differentiation in retinoic acid resistant acute promyelocytic leukemia cell lines
Acknowledgments
This work is supported, in part, by the grants from the National Natural Science Foundation of China (NSFC: 30871105) and the Science and Technology Commission of Shanghai (13ZR1425400).
References
- Shen Z. X. et al. All-trans retinoic acid/As2O3 combination yields a high quality remission and survival in newly diagnosed acute promyelocytic leukemia. Proc Natl Acad Sci U S A. 101, 5328–5335 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonneveld E. et al. Human retinoic acid (RA) 4-hydroxylase (CYP26) is highly specific for all-trans-RA and can be induced through RA receptors in human breast and colon carcinoma cells. Cell Growth Differ. 9, 629–637 (1998). [PubMed] [Google Scholar]
- Michieli M. et al. P-glycoprotein (PGP), lung resistance-related protein (LRP) and multidrug resistance-associated protein (MRP) expression in acute promyelocytic leukaemia. Br J Haematol. 108, 703–709 (2000). [DOI] [PubMed] [Google Scholar]
- Delva L. et al. Resistance to all-trans retinoic acid (ATRA) therapy in relapsing acute promyelocytic leukemia: study of in vitro ATRA sensitivity and cellular retinoic acid binding protein levels in leukemic cells. Blood 82, 2175–2181 (1993). [PubMed] [Google Scholar]
- Cornic M. et al. Induction of retinoic acid-binding protein in normal and malignant human myeloid cells by retinoic acid in acute promyelocytic leukemia patients. Cancer Res. 52, 3329–3334 (1992). [PubMed] [Google Scholar]
- Duprez E. et al. A retinoid acid ‘resistant' t(15;17) acute promyelocytic leukemia cell line: isolation, morphological, immunological, and molecular features. Leukemia 6, 1281–1287 (1992). [PubMed] [Google Scholar]
- Shao W., Benedetti L., Lamph W. W., Nervi C. & Miller W. H. Jr A retinoid-resistant acute promyelocytic leukemia subclone expresses a dominant negative PML-RAR alpha mutation. Blood 89, 4282–4289 (1997). [PubMed] [Google Scholar]
- Kitamura K. et al. Mutant AF-2 domain of PML-RARalpha in retinoic acid-resistant NB4 cells: differentiation induced by RA is triggered directly through PML-RARalpha and its down-regulation in acute promyelocytic leukemia. Leukemia 11, 1950–1956 (1997). [DOI] [PubMed] [Google Scholar]
- Nason-Burchenal K. et al. Targeting of PML/RARalpha is lethal to retinoic acid-resistant promyelocytic leukemia cells. Blood 92, 1758–1767 (1998). [PubMed] [Google Scholar]
- Marasca R. et al. Missense mutations in the PML/RARalpha ligand binding domain in ATRA-resistant As(2)O(3) sensitive relapsed acute promyelocytic leukemia. Haematologica 84, 963–968 (1999). [PubMed] [Google Scholar]
- Duprez E., Benoit G., Flexor M., Lillehaug J. R. & Lanotte M. A mutated PML/RARA found in the retinoid maturation resistant NB4 subclone, NB4-R2, blocks RARA and wild-type PML/RARA transcriptional activities. Leukemia 14, 255–261 (2000). [DOI] [PubMed] [Google Scholar]
- Zhou D. C. et al. Frequent mutations in the ligand-binding domain of PML-RARalpha after multiple relapses of acute promyelocytic leukemia: analysis for functional relationship to response to all-trans retinoic acid and histone deacetylase inhibitors in vitro and in vivo. Blood 99, 1356–1363 (2002). [DOI] [PubMed] [Google Scholar]
- Ding W. et al. Leukemic cellular retinoic acid resistance and missense mutations in the PML-RARalpha fusion gene after relapse of acute promyelocytic leukemia from treatment with all-trans retinoic acid and intensive chemotherapy. Blood 92, 1172–1183 (1998). [PubMed] [Google Scholar]
- Takayama N., Kizaki M., Hida T., Kinjo K. & Ikeda Y. Novel mutation in the PML/RARalpha chimeric gene exhibits dramatically decreased ligand-binding activity and confers acquired resistance to retinoic acid in acute promyelocytic leukemia. Exp Hematol. 29, 864–872 (2001). [DOI] [PubMed] [Google Scholar]
- Imaizumi M. et al. Mutations in the E-domain of RAR portion of the PML/RAR chimeric gene may confer clinical resistance to all-trans retinoic acid in acute promyelocytic leukemia. Blood 92, 374–382 (1998). [PubMed] [Google Scholar]
- Cote S. et al. Altered ligand binding and transcriptional regulation by mutations in the PML/RARalpha ligand-binding domain arising in retinoic acid-resistant patients with acute promyelocytic leukemia. Blood 96, 3200–3208 (2000). [PubMed] [Google Scholar]
- Farris M., Lague A., Manuelyan Z., Statnekov J. & Francklyn C. Altered nuclear cofactor switching in retinoic-resistant variants of the PML-RARalpha oncoprotein of acute promyelocytic leukemia. Proteins 80, 1095–1109 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallagher R. E. et al. Treatment-influenced associations of PML-RARalpha mutations, FLT3 mutations, and additional chromosome abnormalities in relapsed acute promyelocytic leukemia. Blood 120, 2098–2108 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welch J. S. et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 150, 264–278 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mi J. Q., Li J. M., Shen Z. X., Chen S. J. & Chen Z. How to manage acute promyelocytic leukemia. Leukemia 26, 1743–1751 (2012). [DOI] [PubMed] [Google Scholar]
- Goto E. et al. Missense mutations in PML-RARA are critical for the lack of responsiveness to arsenic trioxide treatment. Blood 118, 1600–1609 (2011). [DOI] [PubMed] [Google Scholar]
- Cote S. et al. Response to histone deacetylase inhibition of novel PML/RARalpha mutants detected in retinoic acid-resistant APL cells. Blood 100, 2586–2596 (2002). [DOI] [PubMed] [Google Scholar]
- Warrell R. P. Jr, He L. Z., Richon V., Calleja E. & Pandolfi P. P. Therapeutic targeting of transcription in acute promyelocytic leukemia by use of an inhibitor of histone deacetylase. J Natl Cancer Inst. 90, 1621–1625 (1998). [DOI] [PubMed] [Google Scholar]
- Santoro F. et al. A dual role for Hdac1: oncosuppressor in tumorigenesis, oncogene in tumor maintenance. Blood 121, 3459–3468 (2013). [DOI] [PubMed] [Google Scholar]
- Duprez E., Lillehaug J. R., Gaub M. P. & Lanotte M. Differential changes of retinoid-X-receptor (RXR alpha) and its RAR alpha and PML-RAR alpha partners induced by retinoic acid and cAMP distinguish maturation sensitive and resistant t(15;17) promyelocytic leukemia NB4 cells. Oncogene 12, 2443–2450 (1996). [PubMed] [Google Scholar]
- Higuchi T., Kizaki M. & Omine M. Induction of differentiation of retinoic acid-resistant acute promyelocytic leukemia cells by the combination of all-trans retinoic acid and granulocyte colony-stimulating factor. Leuk Res. 28, 525–532 (2004). [DOI] [PubMed] [Google Scholar]
- Witcher M., Shiu H. Y., Guo Q. & Miller W. H. Jr Combination of retinoic acid and tumor necrosis factor overcomes the maturation block in a variety of retinoic acid-resistant acute promyelocytic leukemia cells. Blood 104, 3335–3342 (2004). [DOI] [PubMed] [Google Scholar]
- Gao F. et al. Apoptosis inducing and differentiation enhancement effect of oridonin on the all-trans-retinoic acid-sensitive and -resistant acute promyelocytic leukemia cells. Int J Lab Hematol. 32, e114–122 (2010). [DOI] [PubMed] [Google Scholar]
- Gianni M. et al. Tyrosine kinase inhibitor STI571 potentiates the pharmacologic activity of retinoic acid in acute promyelocytic leukemia cells: effects on the degradation of RARalpha and PML-RARalpha. Blood 97, 3234–3243 (2001). [DOI] [PubMed] [Google Scholar]
- He P. et al. Interferon-gamma enhances promyelocytic leukemia protein expression in acute promyelocytic cells and cooperates with all-trans-retinoic acid to induce maturation of NB4 and NB4-R1 cells. Exp Ther Med. 3, 776–780 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leskow F. C., Krasnapolski M. A. & Urtreger A. J. The pros and cons of targeting protein kinase C (PKC) in the management of cancer patients. Curr Pharm Biotechnol. 12, 1961–1973 (2011). [DOI] [PubMed] [Google Scholar]
- Miloso M. et al. Retinoic acid-induced neuritogenesis of human neuroblastoma SH-SY5Y cells is ERK independent and PKC dependent. J Neurosci Res. 75, 241–252 (2004). [DOI] [PubMed] [Google Scholar]
- Bertolaso L. et al. Accumulation of catalytically active PKC-zeta into the nucleus of HL-60 cell line plays a key role in the induction of granulocytic differentiation mediated by all-trans retinoic acid. Br J Haematol. 100, 541–549 (1998). [DOI] [PubMed] [Google Scholar]
- Gruber J. R., Desai S., Blusztajn J. K. & Niles R. M. Retinoic acid specifically increases nuclear PKC alpha and stimulates AP-1 transcriptional activity in B16 mouse melanoma cells. Exp Cell Res. 221, 377–384 (1995). [DOI] [PubMed] [Google Scholar]
- Zorn N. E. & Sauro M. D. Retinoic acid induces translocation of protein kinase C (PKC) and activation of nuclear PKC (nPKC) in rat splenocytes. Int J Immunopharmacol. 17, 303–311 (1995). [DOI] [PubMed] [Google Scholar]
- Kambhampati S. et al. Activation of protein kinase C delta by all-trans-retinoic acid. J Biol Chem. 278, 32544–32551 (2003). [DOI] [PubMed] [Google Scholar]
- McNamara S., Nichol J. N., Wang H. & Miller W. H. Jr Targeting PKC delta-mediated topoisomerase II beta overexpression subverts the differentiation block in a retinoic acid-resistant APL cell line. Leukemia 24, 729–739 (2010). [DOI] [PubMed] [Google Scholar]
- Devalia V., Thomas N. S., Roberts P. J., Jones H. M. & Linch D. C. Down-regulation of human protein kinase C alpha is associated with terminal neutrophil differentiation. Blood 80, 68–76 (1992). [PubMed] [Google Scholar]
- Scott L. M., Civin C. I., Rorth P. & Friedman A. D. A novel temporal expression pattern of three C/EBP family members in differentiating myelomonocytic cells. Blood 80, 1725–1735 (1992). [PubMed] [Google Scholar]
- Lekstrom-Himes J. A. The role of C/EBP(epsilon) in the terminal stages of granulocyte differentiation. Stem Cells 19, 125–133 (2001). [DOI] [PubMed] [Google Scholar]
- Ohnishi K. PML-RARalpha inhibitors (ATRA, tamibaroten, arsenic troxide) for acute promyelocytic leukemia. Int J Clin Oncol. 12, 313–317 (2007). [DOI] [PubMed] [Google Scholar]
- Peng H. Y. & Liao H. F. Staurosporine induces megakaryocytic differentiation through the upregulation of JAK/Stat3 signaling pathway. Ann Hematol. 90, 1017–1029 (2011). [DOI] [PubMed] [Google Scholar]
- Mollereau C., Zajac J. M. & Roumy M. Staurosporine differentiation of NPFF2 receptor-transfected SH-SY5Y neuroblastoma cells induces selectivity of NPFF activity towards opioid receptors. Peptides 28, 1125–1128 (2007). [DOI] [PubMed] [Google Scholar]
- Zhang H., Vollmer M., De Geyter M., Durrenberger M. & De Geyter C. Apoptosis and differentiation induced by staurosporine in granulosa tumor cells is coupled with activation of JNK and suppression of p38 MAPK. Int J Oncol. 26, 1575–1580 (2005). [PubMed] [Google Scholar]
- Zhao C. et al. Down-regulation of DNA methyltransferase 3B in staurosporine-induced apoptosis and its mechanism in human hepatocarcinoma cell lines. Mol Cell Biochem. 376, 111–119 (2013). [DOI] [PubMed] [Google Scholar]
- Li G., Gleinich A., Lau H. & Zimmermann M. Staurosporine-induced apoptosis presents with unexpected cholinergic effects in a differentiated neuroblastoma cell line. Neurochem Int. 61, 1011–1020 (2012). [DOI] [PubMed] [Google Scholar]
- Nicolier M., Decrion-Barthod A. Z., Launay S., Pretet J. L. & Mougin C. Spatiotemporal activation of caspase-dependent and -independent pathways in staurosporine-induced apoptosis of p53wt and p53mt human cervical carcinoma cells. Biol Cell. 101, 455–467 (2009). [DOI] [PubMed] [Google Scholar]
- Friedman A. D. Transcriptional control of granulocyte and monocyte development. Oncogene 26, 6816–6828 (2007). [DOI] [PubMed] [Google Scholar]
- Duprez E., Wagner K., Koch H. & Tenen D. G. C/EBPbeta: a major PML-RARA-responsive gene in retinoic acid-induced differentiation of APL cells. EMBO J 22, 5806–5816 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truong B. T. et al. CCAAT/Enhancer binding proteins repress the leukemic phenotype of acute myeloid leukemia. Blood 101, 1141–1148 (2003). [DOI] [PubMed] [Google Scholar]
- Barbarroja N. et al. MEK inhibition induces caspases activation, differentiation blockade and PML/RARalpha degradation in acute promyelocytic leukaemia. Br J Haematol. 142, 27–35 (2008). [DOI] [PubMed] [Google Scholar]
- Scholl S. et al. Additive effects of PI3-kinase and MAPK activities on NB4 cell granulocyte differentiation: potential role of phosphatidylinositol 3-kinase gamma. J Cancer Res Clin Oncol. 134, 861–872 (2008). [DOI] [PubMed] [Google Scholar]
- Lu J. et al. Troxerutin counteracts domoic acid-induced memory deficits in mice by inhibiting CCAAT/enhancer binding protein beta-mediated inflammatory response and oxidative stress. J Immunol. 190, 3466–3479 (2013). [DOI] [PubMed] [Google Scholar]
- Murakami M. et al. Sphingosine kinase 1/S1P pathway involvement in the GDNF-induced GAP43 transcription. J Cell Biochem. 112, 3449–3458 (2011). [DOI] [PubMed] [Google Scholar]
- Lee S. et al. RSK-mediated phosphorylation in the C/EBP{beta} leucine zipper regulates DNA binding, dimerization, and growth arrest activity. Mol Cell Biol. 30, 2621–2635 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu Z. M. et al. Pharicin B stabilizes retinoic acid receptor-alpha and presents synergistic differentiation induction with ATRA in myeloid leukemic cells. Blood 116, 5289–5297 (2010). [DOI] [PubMed] [Google Scholar]
- Nason-Burchenal K. et al. Common defects of different retinoic acid resistant promyelocytic leukemia cells are persistent telomerase activity and nuclear body disorganization. Differentiation 61, 321–331 (1997). [DOI] [PubMed] [Google Scholar]
- Zhu J. et al. Effect of retinoic acid isomers on proliferation, differentiation and PML relocalization in the APL cell line NB4. Leukemia 9, 302–309 (1995). [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Combined staurosporine and retinoic acid induces differentiation in retinoic acid resistant acute promyelocytic leukemia cell lines