Intrinsic and acquired drug resistance is a common problem causing the recurring of malignant tumors, such as breast and liver cancers1, 2. Successful chemotherapy is often limited due to significant side effects and development of drug resistance3. With the rapid development of nanotechnology, overcoming drug resistance and improving the efficacy of antitumor drugs with scalable and biocompatible nanotherapies have been extensively explored.
Up to date many attempts have been made. Nanoparticles that stimulate the endocytosis of drug resistant cells to raise intracellular drug concentration have been proved an effective way to reverse the acquired resistance of cisplatin-resistant cells4. For the resistant tumor cells that pump drugs out through ATP-binding cassette (ABC) transporters, which is considered as a common mechanism for drug resistance5, one of current approaches is to seek nanoparticle-mediated drug delivery system with prolonged retention time to prevent drug efflux and retain therapeutic concentration for maximized tumor inhibition.
Doxorubicin (Dox) has been established as one of the most useful chemotherapeutics with a broad spectrum of activity against human neoplasms. In Dox-resistant cells, the drug can be actively pumped out by ABC transporters6. Nanometer-sized diamonds (NDs) have been used for fluorescent cellular biolabeling7, biomedical imaging8, and as drug carriers9. Chow and colleagues recently developed a biocompatible nanodiamond drug carrier system at 2 to 8 nm in diameter with Dox binding to its surfaces electrostatically, forming NDX complexes with an average size of ∼80 nm. The complexes not only reversed the drug efflux-based chemoresistance to Dox, but also avoided the toxic side effects to healthy tissues often occurred with unconjugated free Dox10.
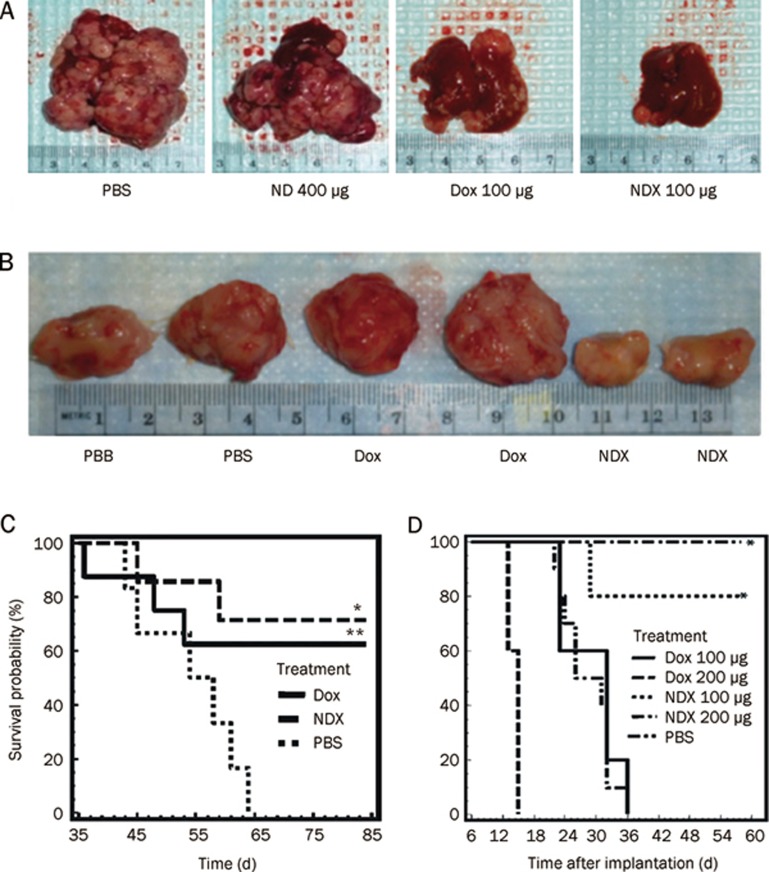
The authors used various Dox-resistant tumor cell lines to determine the ability of NDX in maintaining intracellular drug concentration. The cells retained more Dox when treated with NDX as compared to unmodified Dox treatment. While free Dox was intensively pumped out of these cells by ABC-transporters, sustained release of functional drugs from NDs counteracted the efflux of Dox from the tumor cells. Furthermore, the killing of tumor cells by NDs was not affected by the ABC transporter inhibitor verapamil, suggesting NDs delivery may be used as a nonspecific method for overcoming drug efflux. They also demonstrated in vivo that NDX treatment effectively improved tumor inhibition rate, survival probability and increased apoptosis specifically in murine LT2-Myc liver and 4T1 mammary tumors without affecting adjacent normal tissues (Figure 1).
Figure 1.
ND delivery of Dox inhibits tumor growth in murine liver tumor models and mammary carcinoma model10. (A) Images of livers/tumors from PBS, Dox, ND, or NDX treated LT2-Myc liver tumor bearing mice. (B) Representative images of excised tumors from PBS, Dox, or NDX treated 4T1 mammary tumor bearing mice. (C) Kaplan-Meier survival plot for LT2-Myc mice treated with PBS (n=5), Dox (100 mg) (n=8), or NDX (100 mg of Dox equivalent) (n=7) by tail vein injection every 7 d. *P<0.03; **P<0.06. (D) Kaplan-Meier survival plot for 4T1 mice treated with PBS (n=7), Dox (100 mg) (n=10), NDX (100 mg of Dox equivalent) (n=10), Dox (200 mg) (n=5), or NDX (200 mg of Dox equivalent) (n=5) by tail vein injection every 6 d. *P<0.003. Reprinted with permission from AAAS: Science Translational Medicine, copyright 2011.
Effective chemotherapy requires maintaining chemotherapeutic agents at therapeutic concentration for a prolonged period, maximizing tumor inhibition and minimizing normal tissue toxicity11, 12. The reason for the advantages of NDX over free Dox is likely due to the gradual release of the drug from nanodiamonds, which allows for increased drug retention in blood circulation and tumor, and therefore maintained the Dox concentration below a toxic level to normal tissues. It prevents the “burst effect” — a majority of Dox released in a rapid and uncontrollable fashion, which creates unpredictable pharmacokinetics, and thereby making effective dose regimens difficult to predict and acute toxicity to normal organs/tissues13.
Although high-dosage of chemotherapeutic administered as a single dose may exert some effect on tumor, it is also accompanied by significant toxicity. For a solid tumor with a long doubling time, prolonged exposure to an active drug at a minimally effective concentration can affect a greater number of tumor cells while minimizing toxicity to normal organs11. Chow and colleagues' work demonstrates that nanodiamond delivery systems may greatly increase the efficacy of traditional anticancer drugs by preventing drugs from being pumped out of tumor cells, allowing anticancer drugs to kill drug-resistant cells at a lower dosage, thus lowering the drug toxicity to normal organs and tissues. Compared to other nanoparticles, nanodiamond is small in diameter and can be cleared out of body within a certain period after administration. Meanwhile, no immune response or liver toxicity was observed with NDs even at high dosage, demonstrating its good biocompatibility. Moreover, surface modification with different functionalities would allow NDs binding with different types of therapeutics and biomolecules (such as imaging contrast agent, therapeutic siRNA, proteins and some other hydrophilic/hydrophobic drugs) through either covalent or noncovalent bonds. This implies that NDs have a great potential to be used for a wide range of drug delivery in treatment of a variety of diseases. Diamonds at nanoscale may entertain new expectation for treatment of malignant tumors in future.
References
- Giaccone G, Linn SC, Pinedo HM. Multidrug resistance in breast cancer: mechanisms, strategies. Eur J Cancer. 1995;31A:S15–7. doi: 10.1016/0959-8049(95)00293-r. [DOI] [PubMed] [Google Scholar]
- Chenivesse X, Franco D, Brechot C. MDR1 (multidrug resistance) gene expression in human primary liver cancer and cirrhosis. J Hepatol. 1993;18:168–72. doi: 10.1016/s0168-8278(05)80243-0. [DOI] [PubMed] [Google Scholar]
- Gillet JP, Gottesman MM. Mechanisms of multidrug resistance in cancer. Methods Mol Biol. 2010;596:47–76. doi: 10.1007/978-1-60761-416-6_4. [DOI] [PubMed] [Google Scholar]
- Liang XJ, Meng H, Wang Y, He H, Meng J, Lu J, et al. Metallofullerene nanoparticles circumvent tumor resistance to cisplatin by reactivating endocytosis. Proc Natl Acad Sci USA. 2010;107:7449–54. doi: 10.1073/pnas.0909707107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishna R, Mayer LD. Multidrug resistance (MDR) in cancer. Mechanisms, reversal using modulators of MDR and the role of MDR modulators in influencing the pharmacokinetics of anticancer drugs. Eur J Pharm Sci. 2000;11:265–83. doi: 10.1016/s0928-0987(00)00114-7. [DOI] [PubMed] [Google Scholar]
- Shen F, Chu S, Bence AK, Bailey B, Xue X, Erickson PA, et al. Quantitation of doxorubicin uptake, efflux, and modulation of multidrug resistance (MDR) in MDR human cancer cells. J Pharmacol Exp Ther. 2008;324:95–102. doi: 10.1124/jpet.107.127704. [DOI] [PubMed] [Google Scholar]
- Mkandawire M, Pohl A, Gubarevich T, Lapina V, Appelhans D, Rodel G, et al. Selective targeting of green fluorescent nanodiamond conjugates to mitochondria in HeLa cells. J Biophotonics. 2009;2:596–606. doi: 10.1002/jbio.200910002. [DOI] [PubMed] [Google Scholar]
- Mochalin VN, Gogotsi Y. Wet chemistry route to hydrophobic blue fluorescent nanodiamond. J Am Chem Soc. 2009;131:4594–5. doi: 10.1021/ja9004514. [DOI] [PubMed] [Google Scholar]
- Huang H, Pierstorff E, Osawa E, Ho D. Active nanodiamond hydrogels for chemotherapeutic delivery. Nano Lett. 2007;7:3305–14. doi: 10.1021/nl071521o. [DOI] [PubMed] [Google Scholar]
- Chow EK, Zhang XQ, Chen M, Lam R, Robinson E, Huang H, et al. Nanodiamond therapeutic delivery agents mediate enhanced chemoresistant tumor treatment. Sci Transl Med. 2011;3:73ra21. doi: 10.1126/scitranslmed.3001713. [DOI] [PubMed] [Google Scholar]
- Kamen BA, Rubin E, Aisner J, Glatstein E. High-time chemotherapy or high time for low dose. J Clin Oncol. 2000;18:2935–7. doi: 10.1200/JCO.2000.18.16.2935. [DOI] [PubMed] [Google Scholar]
- Wang YZ, Huang B, Lv J, Liang XJ. Current status of nanotechnology applied in biomedicine. Acta Biophys Sin. 2009;25:168–4. [Google Scholar]
- Lenzhofer R, Ganzinger U, Rameis H, Moser K. Acute cardiac toxicity in patients after doxorubicin treatment and the effect of combined tocopherol and nifedipine pretreatment. J Cancer Res Clin Oncol. 1983;106:143–7. doi: 10.1007/BF00395393. [DOI] [PubMed] [Google Scholar]