Abstract
Aim:
To investigate the efficacy of mitomycin C (MMC) in combination with curcumin in suppressing human breast cancer in vitro and in vivo.
Methods:
Human breast cancer MCF-7 cells were used. Cell viability was measured using MTT assay. The cell cycle phase was detected with flow cytometric analysis. Cell cycle-associated proteins were examined using Western blot analysis. MCF-7 breast cancer xenografts were established to monitor tumor growth and cell cycle-associated protein expression.
Results:
Curcumin inhibited MCF-7 breast cancer cell viability in a concentration-dependent manner (IC50 value=40 μmol/L). Similarly, MMC inhibited the cell viability with an IC50 value of 5 μmol/L. Combined treatment of MMC and curcumin showed a synergistic antiproliferative effect. In the presence of curcumin (40 μmol/L), the IC50 value of MMC was reduced to 5 μmol/L. In MCF-7 xenografts, combined administration of curcumin (100 mg/kg) and MMC (1-2 mg/kg) for 4 weeks produced significantly greater inhibition on tumor growth than either treatment alone. The combined treatment resulted in significantly greater G1 arrest than MMC or curcumin alone. Moreover, the cell cycle arrest was associated with inhibition of cyclin D1, cyclin E, cyclin A, cyclin-dependent kinase 2 (CDK2) and CDK4, along with the induction of the cell cycle inhibitor p21 and p27 both in MCF-7 cells and in MCF-7 xenografts. These proteins were regulated through p38 MAPK pathway.
Conclusion:
The results suggest that the combination of MMC and curcumin inhibits MCF-7 cell proliferation and cell cycle progression in vitro and in vivo via the p38 MAPK pathway.
Keywords: curcumin, mitomycin C, human breast cancer MCF-7 cells, combined chemotherapy, cell cycle, xenografts, p38 MAPK, cyclin, cyclin-dependent kinases (CDKs), p21
Introduction
Cell growth and proliferation are tightly regulated by various interactions between molecules1. Several major checkpoints in the cell cycle are controlled by multiple protein kinases2, 3. The cyclin-dependent kinases (CDKs) join with regulatory proteins called cyclins to drive the cell through the cell cycle4.
Specific complexes regulate each step of the cycle. Cyclin D1/CDK2 and CDK4 drive progression through G1, cyclin E/CDK2 controls entry into S phase, and cyclin A/CDK2 controls S-phase progression. CDK inhibitors (CDKIs) such as p21 and p275 block specific interactions. In many cell types, aberrant expression of cell cycle regulatory proteins can induce cell cycle progression6, 7.
The mitogen-activated protein kinase (MAPK) family is activated in response to proliferative cues8. P38 MAPK is a major subfamily of mammalian MAPK. It has been reported that p38 MAPK activation is involved in the induction of a G1/S checkpoint9 that results in the accumulation of p2110, 11. Moreover, p21 establishes a G1/S checkpoint by inactivating CDK210. In addition, p38 MAPK can stabilize p21 in vivo9 and can down-regulate the level of cyclin D112.
The cell cycle controls cell proliferation and cancer is a disease of inappropriate cell proliferation. The excess cells initiate a vicious cycle where cells are insensitive to the signals that are involved in adhesion, differentiation and death in normal cells13. Modulation of the cell cycle also contributes to oncotherapy. The majority of human breast cancers display deregulated overexpression of cyclin D1, whereas in normal cells its expression is tightly regulated by mitogenic signaling involving the p21 Ras pathway14. Research on cell cycle progression and cell cycle regulatory proteins in human breast cancer cell lines has been performed in recent years15, 16.
Curcumin (diferuloylmethane), a yellow colored polyphenol and a natural plant phenolic food additive, is an active component of the perennial herb Curcuma longa (commonly known as turmeric)17. Curcumin exhibits anti-cancer activities both in vitro and in vivo through a variety of mechanisms. It inhibits proliferation and induces apoptosis in a wide array of cancer cell types in vitro, including cells from cancers of the bladder, breast, lung and other tissues18. It also controls the cell cycle and causes cell cycle arrest in various cancers. Curcumin inhibited cell cycle progression through the downregulation of cyclin D1 in vitro and its transcriptional and translational levels in vivo and by blocking its association with CDK419. It also induced cell cycle arrest through the upregulation of CDKIs such as p21 and p2720, 21.
Mitomycin C (MMC) is currently used as the third-line chemotherapeutic agent for breast cancers22. Previous studies have shown that there are cell cycle alterations in response to MMC treatment23, 24. However, it is not clear whether curcumin in combination with MMC impacts breast cancer cell proliferation and cell cycle progression.
The present study sought to examine the combination treatment of curcumin and MMC-induced inhibition of cell growth and cell cycle arrest in vitro and in vivo. The p38 MAPK pathway was identified as a mechanism through which the combination treatment induced cell growth inhibition that was mediated via p21 and p27-induced cell cycle arrest.
Materials and methods
Materials
MMC was purchased from ICN Company (USA), dissolved in physiological saline as a 1 mmol/L stock solution and stored at 4 °C away from light. Curcumin, with a purity of more than 98%, was obtained from the National Institute for the Control of Pharmaceutical and Biological Products in China. Curcumin was dissolved in dimethyl sulfoxide (DMSO) as 40 mmol/L solution for use in the treatment of cells, and it was dissolved in a solution of physiological saline with 1% DMSO and 10% Tween-80 for use in animal research. SB203580 was obtained from Biomol (Philadelphia, PA, USA). The antibodies against cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21 and p27 were obtained from Cell Signaling Inc (Boston, MA, USA). The antibody against phosphorylated p38 (p-p38) was obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Propidium iodide (PI) was purchased from Sigma (St Louis, MO, USA).
Cell culture
Human breast cancer MCF-7 cells were obtained from the American Type Culture Collection (ATCC) (Manassas, VA, USA) and cultured in RPMI-1640 medium from Gibco (San Francisco, CA, USA) supplemented with 10% heat-inactivated (56 °C, 30 min) fetal calf serum (PAA, Pasching, KA, Austria), 0.01 mg/mL insulin from Sigma (St Louis, MO, USA), 2 mmol/L glutamine from Gibco (San Francisco, CA, USA), penicillin (100 U/mL) and streptomycin (100 μg/mL). The cell culture was maintained at 37 °C with 5% CO2 in a humidified atmosphere.
Cell viability assays
MCF-7 cells were seeded in 96-well culture plates (5×104 cells/mL). After overnight incubation, MCF-7 cells were treated with various concentrations of MMC, curcumin, or MMC plus curcumin (the ratio of two compounds was 1:1). DMSO was adjusted to the same final concentration of 0.01%. Following incubation, cell viability was measured at different time points using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT) as described previously25. The formazan product was quantified by measuring the absorbance at 490 nm. The interaction between curcumin and MMC was analyzed by isobologram analysis described by Chou and Talalay26. The interaction of two compounds was quantified by determining the combination index (CI). From this analysis, the combined effects of the two drugs can be summarized as follows: the CI of less than, equal to or more than 1 indicate synergistic, additive or antagonistic effects, respectively.
Cell cycle analysis
MCF-7 cells were seeded into 10-cm dishes and treated with curcumin, MMC or a combination of the agents at the concentrations indicated. Cells were harvested 48 h after treatment. Detached cells were collected by centrifugation (100×g, 5 min). The attached cells were harvested by trypsinization. Cell pellets were washed with PBS and then fixed in ice-cold 70% ethanol. For cell cycle analysis, cells were recentrifuged and stained with propidium iodide (PI). Cell-cycle phase analysis was performed using a Becton Dickinson Facstar flow cytometer (San Jose, CA, USA) equipped with ModFit LT 3.0 software.
Western blot analysis
Whole cell lysate was loaded in each lane and separated by SDS-PAGE. Protein expression was detected using a primary antibody (1:1000, except p-p38 1:200) and secondary antibody (1:800) conjugated with horseradish peroxidase. Levels of cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, p27, p38, p-p38, and GAPDH were analyzed in this manner. Chemiluminescence was observed by ECL (Pharmacia, Buckinghamshire, UK). Quantitative analysis of Western blotting was performed using Alpha Ease FC (FluorChem FC2) software. Using the analysis tools, we calculated the density ratio of each protein to GAPDH, the loading control.
Human tumor xenografts
Female nu/nu athymic mice (7 weeks old) were obtained from Chinese Academy of Sciences. The mice (five per cage) were housed in cages equipped with air filter lids and maintained under pathogen-limiting conditions. MCF-7 cells (1×107/mL) were inoculated into the mammary fat pads of the mice. Before inoculation, 17β-estradiol (1.7 mg) was intraperitoneally injected27. Once palpable tumors developed (approximately 2 weeks), mice were randomized to receive vehicle (physiological saline with 1% DMSO and 10% Tween-80 for curcumin, 100% physiological saline for MMC), curcumin (100 mg/kg), MMC (2 mg/kg), MMC (1.5 mg/kg), MMC (1 mg/kg) and a combination of both curcumin and MMC. All drugs were administered by intraperitoneal injection. There were eight tumor-bearing animals in each group and all tumors were harvested 28 d after treatment. The tumors were immediately removed, freed from connective and adipose tissue, and weighed. The animal protocols were preapproved by the ethical committee of our institution.
Statistical analysis
When appropriate, data were expressed as mean±SD. Data were analyzed by Student's t-test where appropriate. Statistical significance was set at P<0.05.
Results
The combined treatment of curcumin and MMC inhibits proliferation of MCF-7 cells in a synergistic manner
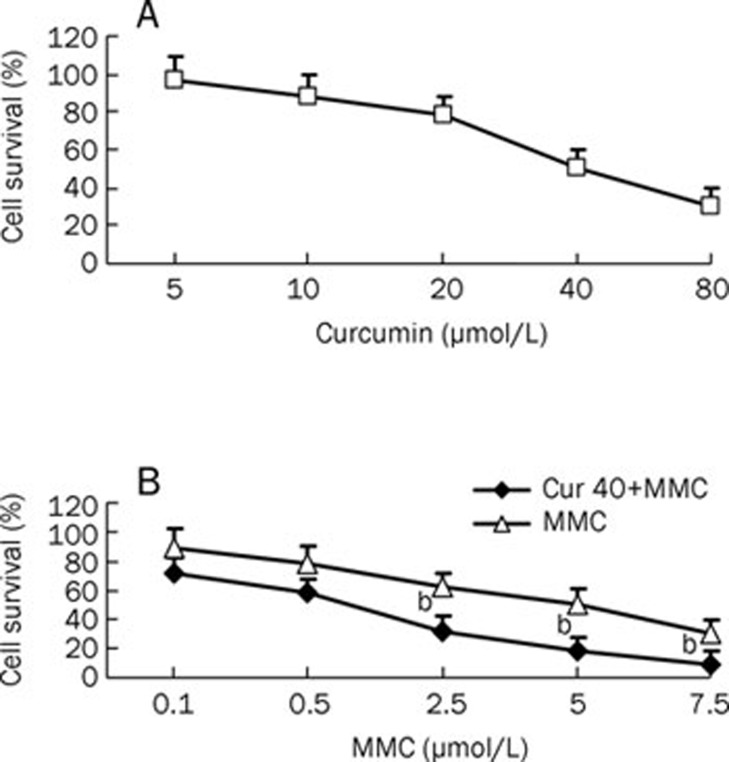
To investigate the inhibitory effect of the combined treatment of curcumin and MMC, MCF-7 cells were treated with or without various concentrations of curcumin or MMC for 48 h. The results of the MTT assay showed that curcumin significantly inhibited cell viability in a concentration-dependent manner; the 50% inhibiting concentration (IC50) for curcumin was 40 μmol/L (Figure 1A). Therefore, the effect of MMC in combination with curcumin was investigated, and the dose of curcumin was fixed at the IC50 40 μmol/L dose. Similarly, MMC inhibited the cell viability with an IC50 value of 5 μmol/L. Increasingdoses of MMC inhibited growth in MCF-7 cells, and the IC50 was shown to be 5 μmol/L (Figure 1B). Isobologram analysis revealed a CI<1 for curcumin at 40 μmol/L in combination with MMC at 2.5, 5, and 7.5 μmol/L, respectively, indicating a synergistic relationship between the treatments. Moreover, the combination treatment of 2.5 μmol/L MMC and 40 μmol/L curcumin reduced the cell viability by 20% as compared to 5 μmol/L MMC alone (P<0.05) (Figure 1B). Therefore, 40 μmol/L curcumin in combination with 2.5 μmol/L MMC was selected for additional experiments.
Figure 1.
Survival of MCF-7 cells by combined treatment with curcumin and MMC. (A) MCF-7 cells were incubated with curcumin at doses of 5, 10, 20, 40, or 80 μmol/L for 48 h. (B) MCF-7 cells were treated with the combination of curcumin at 40 μmol/L and MMC at 0.1, 0.5, 2.5, 5, or 7.5 μmol/L for 48 h. bP<0.05 vs 5 μmol/L MMC alone. Values are mean±SD from 3 independent experiments.
The combined treatment with curcumin and MMC induces a G0/G1 arrest in MCF-7 cells
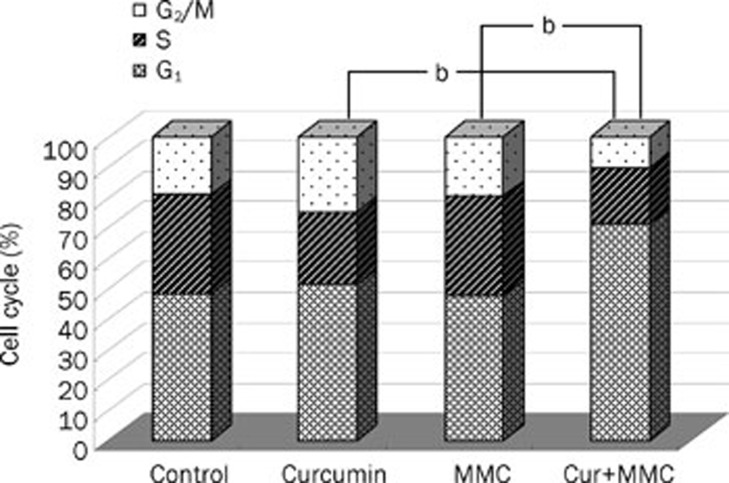
Flow cytometry was performed to determine whether the cell growth inhibition mediated by the combined treatment of curcumin and MMC was due to a cell cycle arrest at a specific point in the cell cycle. Analysis of the untreated control cells showed 32.66% of cells in S phase and 48.36% of cells in G1. Treatment with curcumin at 40 μmol/L inhibited the cells from entering S phase (32.66% to 23.79%) and caused a small increase in the number of cells in G1 (48.36% to 51.51%). MMC at 2.5 μmol/L had a similar effect to untreated control with 33.03% of cells in S phase and 47.47% of cells in G1. The combined treatment, however, resulted in a more robust G1 arrest; only 18.62% of cells were in S phase and 71.17% of cells were in G1. This cell cycle profile is indicative of a G1 cell cycle arrest (Figure 2). The combined treatment of curcumin and MMC had a more complete G0/G1 arrest than curcumin (P<0.05) or MMC (P<0.05) alone in MCF-7 cells.
Figure 2.
Curcumin in combination with MMC suppressed cell proliferation via a G1 cell cycle arrest in MCF-7 cells. MCF-7 cells were treated with the combination of 40 μmol/L curcumin and 2.5 μmol/L MMC for 48 h. Cells were subjected to flow cytometric analysis to determine the effect of the combined treatment on cell cycle distribution. bP<0.05 vs the combination of curcumin and MMC. Values are mean±SD from 3 independent experiments.
The cell cycle arrest is associated with the downregulation of cyclin D1, cyclin E, cyclin A, CDK2, and CDK4 and with the upregulation of p21 and p27 in MCF-7 cells
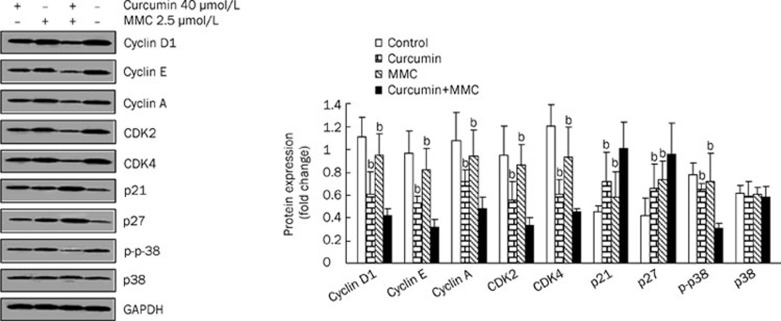
To determine whether the combination-induced growth inhibition of MCF-7 cells was due to the downregulation of cyclins and CDKs and the upregulation of CDKIs, the levels of cell cycle regulatory molecules were examined. The combined treatment of curcumin and MMC in MCF-7 cells for 48 h resulted in 52%–68% reduction in the levels of cyclin D1, cyclin E and cyclin A, as well as CDK2 and CDK4 (Figure 3). The effect of the combined treatment on the induction of p21 and p27, which regulate progression of the cell cycle at the G1-S phase transition checkpoint and are the inhibitors of CDKs, was also examined. The data showed that the combined drug treatment resulted in an approximately 2-fold increase of p21 and p27 as compared to the untreated control (Figure 3). However, the expression of p21 and p27 was increased approximately 1.4-fold by either curcumin or MMC alone.
Figure 3.
The combination treatment-induced cell cycle profile is associated with the downregulation of cyclin D1, cyclin E, cyclin A, CDK2, and CDK4 and upregulation of p21 and p27 in MCF-7 cells. MCF-7 cells were treated with both curcumin and MMC alone or together at the indicated concentrations for 48 h and Western blot analysis was performed with antibodies specific to cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, p27, and p-p38. The density ratio of proteins to GAPDH was shown as a relative expression. bP<0.05 vs the combination of curcumin and MMC. Values are mean±SD from 3 independent experiments.
To determine whether the effects of the combined treatment include activation of p38 MAPK, cells were exposed to the combination for 48 h, and an antibody specific for p-p38 was utilized. The results indicated that the level of p-p38 was reduced by 50% in response to the combined treatment. Both curcumin and MMC alone resulted in a nearly 20% decrease in the level of p-p38. These data show that the combined drug treatment affects p-p38.
The combination of curcumin and MMC regulates the expression of cell cycle-related proteins via the p38 MAPK pathway in MCF-7 cells
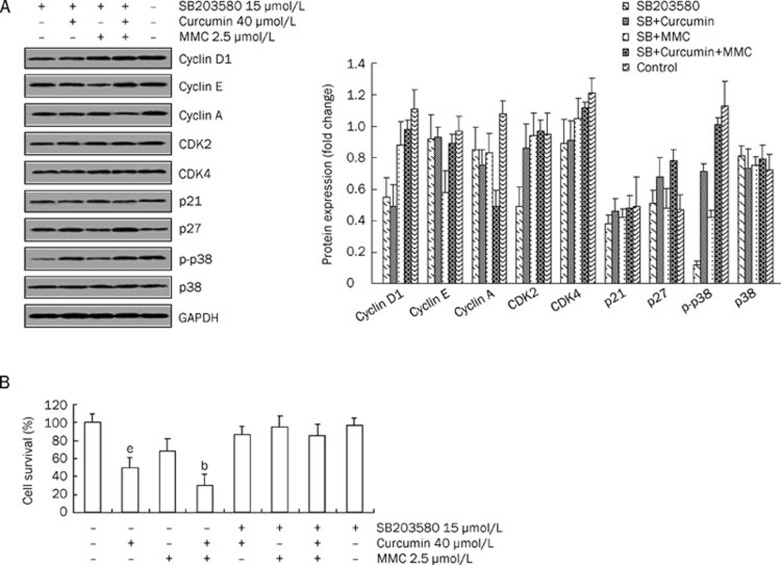
To assess the role of p38 MAPK in the combined treatment, cells were treated with the p38 MAPK-specific inhibitor, SB203580, alone or with additional drugs. The data showed that the combination treatment-decreased p-p38 was reversed by SB203580 (Figure 4A). Because p38 MAPK activity was decreased by the combined drug treatment, the role of cell cycle-related proteins in mediating p38 MAPK activity was investigated.
Figure 4.
SB203580 reversed G1 phase cell cycle-associated protein expression and decreased cell growth inhibition that was induced by the combination of curcumin and MMC. (A) MCF-7 cells were plated into plates and were preincubated for 2 h in the presence of SB203580 (15 μmol/L). Cells were then treated with both 40 μmol/L curcumin and 2.5 μmol/L MMC alone or together for 48 h, followed by Western blot analysis performed with antibodies specific to cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, p27, and p-p38. The density ratio of proteins to GAPDH was shown as a relative expression. (B) MCF-7 cells were pretreated for 2 h with or without 15 μmol/L SB203580 before cells were treated with both 40 μmol/L curcumin and 2.5 μmol/L MMC alone or together for 48 h. The MTT assay was performed as described under materials and methods. Values are mean±SD from 3 independent experiments. bP<0.05 vs untreated control. eP<0.05 vs the combination of curcumin and MMC treatment.
MCF-7 cells were pretreated for 2 h with or without 15 μmol/L SB203580, followed by exposure to 40 μmol/L curcumin in combination with 2.5 μmol/L MMC. As shown in Figure 4A, the combination-induced decrease in cyclin D1, cyclin E, CDK2 and CDK4 and the increase in p21 were reversed to the level of the SB203580 treatment alone. However, under similar experimental conditions, expression of cyclin A was unaffected, suggesting that cyclin A is not likely to be involved in the p38 MAPK-mediated cell cycle arrest that is induced by the combination treatment. Moreover, SB203580 demonstrated a slightly reversed effect on the combination-mediated p27 expression. These results strongly suggest that the p38 MAPK-signaling pathway is required for the G1 phase cell cycle arrest in response to the combination treatment.
To investigate the effect of p38 MAPK activation on the combination-induced cell growth inhibition, MCF-7 cells were pretreated for 2 h with or without 15 μmol/L SB203580, followed by treatment with both curcumin and MMC alone or together. Cell viability was determined by the MTT assay (Figure 4B). After treatment with the combination of curcumin and MMC or curcumin and MMC alone, cell growth inhibition was approximately 70%, 51%, or 32%, respectively. However, this inhibitory effect was reversed by pretreatment with SB203580. These results suggest that the cell growth inhibition caused by the individual or combined treatments is dependent on the p38 MAPK pathway.
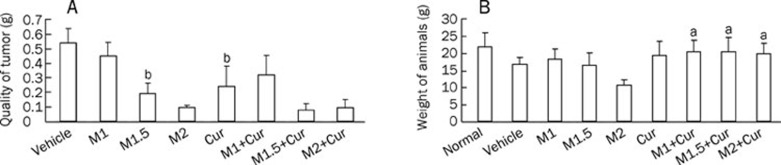
The combination of curcumin and MMC produces greater inhibition of tumor growth than either treatment alone in MCF-7 xenografts
Our in vitro data suggested a synergistic interaction between curcumin and MMC, we studied the antitumor activity of curcumin and MMC in mice bearing MCF-7 breast cancer xenografts. Animals were treated with MMC at a dose of 1, 1.5, or 2 mg/kg ip, with a curcumin dose of 100 mg/kg ip or with curcumin in combination with an MMC dose of 1, 1.5, or 2 mg/kg for 4 weeks. As shown in Figure 5A, the 2 mg/kg dose of MMC treatment alone produced a significant inhibition of tumor growth. The 2 mg/kg dose of MMC in combination with curcumin treatment also revealed a significant reduction in tumor growth. The combined treatment of a 1.5 mg/kg dose of MMC plus curcumin resulted in 60.4% and 68.6% reduction in tumor weight as compared to monotherapy with 1.5 mg/kg MMC and with curcumin, respectively. Treatment with a 1 mg/kg dose of MMC plus curcumin did not show more significant tumor suppression than curcumin alone. Thus, the combination of a 1.5 mg/kg dose of MMC plus a 100 mg/kg dose of curcumin produced a more robust and statistically significant tumor regression when compared with either 1.5 mg/kg MMC (P<0.05) or 100 mg/kg curcumin alone (P<0.05). Moreover, there was no difference between the untreated control and the combined treatment on the body weight of the animals (P>0.05) (Figure 5B). However, the 2 mg/kg MMC treatment resulted in significant weight loss. This weight loss suggests that curcumin probably improves MMC toxicity.
Figure 5.
MMC treatment at 1, 1.5 or 2 mg/kg in combination with curcumin at 100 mg/kg produced effects on reducing tumor quality and body weight compared with either treatment alone. (A) The growth-inhibitory effect of MMC at 1 mg/kg (M1), 1.5 mg/kg (M1.5), and 2 mg/kg (M2), curcumin 100 mg/kg (Cur) and their combination on MCF-7 xenografts is shown. bP<0.05 vs 1.5 mg/kg MMC in combination 100 mg/kg curcumin-treated group. (B) Body weights of MCF-7 xenografts were measured after animals were treated with or without MMC, curcumin or a combination for 4 weeks. aP>0.05 vs normal group. Experiments were repeated with similar results.
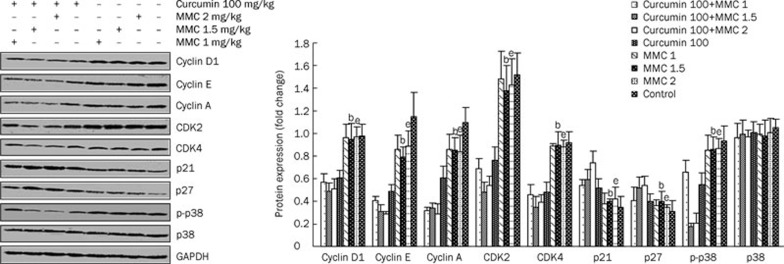
The combination of curcumin and MMC at a dose of 1.5 and 2 mg/kg, respectively, decreases the levels of cyclin D1, cyclin A, cyclin E, CDK2 and CDK4 and increases the expression of p21 and p27 in MCF-7 xenografts
To determine whether the inhibition of tumor growth of MCF-7 xenografts that was mediated by the combined treatment involved the regulation of cell cycle regulatory proteins, the tumors were harvested from each group and then analyzed using Western blot. As shown in Figure 6, the levels of cyclin D1, cyclin E, and cyclin A as well as CDK2 and CDK4 were all reduced by the combined treatment of curcumin and the various concentrations of MMC relative to either curcumin or various concentrations of MMC alone. Moreover, as compared to 100 mg/kg curcumin or 1.5 mg/kg MMC alone, the combined treatment of 100 mg/kg curcumin plus 1.5 mg/kg MMC resulted in a 19.7% or 48.4% decrease in the level of cyclin D1, 36.7% or 60.8% in cyclin E, 44.3% or 60% in cyclin A, 45.3% or 67.4% in CDK2 and 34.5% or 58.2% in CDK4, respectively. Similarly, the combination of 100 mg/kg curcumin plus 2 mg/kg MMC also decreased the levels of cyclin D1, cyclin E, cyclin A, CDK2, and CDK4 significantly. However, the combination of 100 mg/kg curcumin plus 1 mg/kg MMC did not affect the expression of these proteins significantly. Therefore, curcumin at 100 mg/kg in combination with MMC at 1.5 or 2 mg/kg alone both decreased the expression of cell cycle regulatory proteins more significantly than the other combination or single drug therapy.
Figure 6.
MMC treatment at 1, 1.5, or 2 mg/kg in combination with curcumin at 100 mg/kg downregulated the levels of cyclin D, cyclin E, cyclin A, CDK2, and CDK4 and upregulated p21 and p27 levels. The MCF-7 xenografts were treated with either curcumin at 100 mg/kg or MMC at 1, 1.5, and 2 mg/kg alone or in combination, and Western blot analysis was performed with antibodies specific to cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, p27, and p-p38. The density ratio of proteins to GAPDH was shown as a relative expression. bP<0.05 vs 1.5 mg/kg MMC in combination with 100 mg/kg curcumin-treated group. eP<0.05 vs 2 mg/kg MMC in combination with 100 mg/kg curcumin-treated group. Values are mean±SD from 3 independent experiments.
Under similar experimental conditions, treatment with the combination of 100 mg/kg curcumin plus 1.5 mg/kg MMC resulted in a 15.5% or 48.6% increase in the level of p21 and 31.1% or 46.9% in p27 as compared to 100 mg/kg curcumin or 1.5 mg/kg MMC alone, respectively. The level of p21 was increased approximately 42.6% or 74.4%, and p27 was increased approximately 34.9% or 53.9% by the combination of 100 mg/kg curcumin plus 2 mg/kg MMC as compared to 100 mg/kg curcumin or 2 mg/kg MMC alone, respectively. In contrast, the combination of 100 mg/kg curcumin plus 1 mg/kg MMC did not affect the expression of p21 and p27 significantly. These results suggest that the combination of curcumin at 100 mg/kg plus MMC at 1.5 or 2 mg/kg enhances tumor growth inhibition by regulating these cell cycle-associated proteins.
To investigate whether the tumor growth inhibitory effects of the combination treatment include the activation of p38 MAPK, we analyzed the level of p-p38. Results indicated that the expression of p-p38 was significantly suppressed by the combined therapy (Figure 6), suggesting that the combination treatment also affects p-p38.
Discussion
MMC has been already used in the clinic, but it is currently used as the third-line chemotherapeutic agent in breast cancer22. Currently, much research has addressed the chemotherapeutic potential of curcumin (diferuloylmethane), which is a relatively nontoxic plant-derived polyphenol28. It has been reported that curcumin sensitizes cancer cells to cisplatin29. Moreover, our study showed that the combined treatment of curcumin and MMC reduced MMC side effects by inhibiting GRP58-mediated DNA cross-linking through the ERK/p38 MAPK pathway30. The exact mechanisms mediating the cell cycle changes that result from the combined treatment of curcumin and MMC have yet to be fully explained.
Treatment of MCF-7 cells with either curcumin (5–80 μmol/L) or MMC (0.1–7.5 μmol/L) alone resulted in cell growth inhibition in a dose-dependent manner (Figure 1), which was consistent with previously published data31. The combination of 40 μmol/L curcumin (IC50) and 2.5 μmol/L MMC (IC50) reduced cell viability approximately 20% as compared to 5 μmol/L MMC (IC50) alone. Furthermore, isobologram analysis showed a strong synergistic interaction between 40 μmol/L curcumin and 2.5 μmol/L MMC. These results suggest that curcumin in combination with MMC (at a reduced dosage) enhances cell growth inhibition more significantly than MMC alone.
Cyclins are a family of proteins that control the progression of cells through the cell cycle by activating CDK enzymes32. Cyclin D1/CDK4 and cyclin E/CDK2 regulate transition in the G1 phase. Cyclin A/CDK2 is activated from the G1 phase to the S phase and from the S phase to the G2 phase33. The results of this study indicate that the combination of curcumin and MMC induced a G1 phase cell cycle arrest (Figure 2) and also led to a decrease in cyclin D1, cyclin E, cyclin A, CDK2, and CDK4, which are involved in cell cycle progression from the G1 phase to the S phase. Moreover, p21 and p27, which regulate progression of the cell cycle at the G1-S transition checkpoint, were significantly upregulated during the G1 phase arrest in MCF-7 cells that were treated with the combination therapy. Notably, the combination therapy significantly inhibited the activation of p38 MAPK, suggesting that the combination treatment-induced suppression of p-p38 may also be responsible for G1 phase arrest in MCF-7 cells.
Previous studies have demonstrated that p38 MAPK is involved in cell growth inhibition and the regulation of the cell cycle34, 35. The p38 MAPK pathway induced a G1 and G2/M cell cycle arrest, which was associated with increased p21 levels and reduced amounts of cyclin A36, 37. In this study, curcumin in combination with MMC resulted in the downregulation of phosphorylated p38 MAPK. Moreover, the combined treatment regulated the expression of cyclin D1, cyclin E, CDK2, CDK4, and p21, and this regulation was reversed by the p38 MAPK-specific inhibitor SB203580. Robust p38 MAPK stimulation has been reported to suppress cell cycle progression via induction of the cell cycle inhibitor protein p2138, 39. The results of the present study suggest that p38 MAPK signaling is involved in the cell growth inhibition that is induced by combined therapy through the regulation of cyclin D1, cyclin E, CDK2, CDK4, and p21 in MCF-7 cells.
It has been reported that both curcumin40, 41 and MMC42, 43 alone inhibit tumor growth in different cancers. This study has presented the first example of the effects of the combined curcumin and MMC therapy in MCF-7 xenografts. In the present study, we confirmed the antitumor effect of MMC when given for a longer period of time and additionally showed that curcumin also led to tumor stasis. These results were consistent with results of previous studies38, 39, 40, 41. Notably, the data also showed that the combination of 100 mg/kg curcumin and 1.5 mg/kg MMC was more effective than either drug alone at inhibiting MCF-7 tumor growth (Figure 5).
However, 100 mg/kg curcumin in combination with 2 mg/kg MMC did not show more significant tumor growth inhibition than 2 mg/kg MMC alone. The most likely cause of this condition was that the effects of 100 mg/kg curcumin were masked by the 2 mg/kg MMC. For the same reason, the combination of 100 mg/kg curcumin and 1 mg/kg MMC was not more effective than 100 mg/kg curcumin alone. The effects of 1 mg/kg MMC might be masked by 100 mg/kg curcumin. Moreover, the body weight of the animals was reverted to the level of the untreated control with the addition of curcumin with a range of concentrations of MMC. In contrast, treatment of MMC alone reduced body weight significantly. These results suggest that the administration of 100 mg/kg curcumin together with 1.5 mg/kg MMC was an equally effective combination.
To explain the effects and the mechanism of action of the combined treatment of curcumin and MMC in vivo, the levels of cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, and p27 were examined in MCF-7 xenografts. The data indicated that the levels of cyclin D1, cyclin E and cyclin A as well as CDK2 and CDK4 were decreased and that the levels of p21 and p27 were increased by the combined treatment with 100 mg/kg curcumin and 1.5 mg/kg MMC (Figure 6). Moreover, the activation of p38 MAPK was significantly suppressed by the combined treatment. Therefore, it further demonstrated that 100 mg/kg curcumin plus 1.5 mg/kg MMC inhibited tumor growth through regulating the levels of cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, and p27. This regulatory effect was involved in the p38 MAPK pathway.
We characterized the effects and the mechanism of action of the combined treatment of curcumin and MMC in breast cancer models in vitro and in vivo. The results in this study suggest the following of the combined treatment: (1) it increases the anti-proliferative ability while reducing the dosage needed of MMC alone; (2) it enhances G1 arrest through the modulation of cyclin D1, cyclin E, cyclin A, CDK2, CDK4, p21, and p27; and (3) it inhibits MCF-7 cell proliferation and cell cycle progression in vitro and in vivo via the p38 MAPK pathway. Therefore, this combined therapy may be beneficial in the treatment of breast cancer.
Author contribution
Shi-bing SU designed the experiments and proofread the manuscript. Qian-mei ZHOU performed the Western blot analysis, apoptosis and proliferation assays, interpreted the results, generated the figures and wrote the manuscript. Xiu-feng WANG, Hui ZHANG and Xin-jun LIU assisted with animal experiments and provided many useful discussions on experiments. Yi-yu LU analyzed data. All authors assisted in revision of and approved the final manuscript.
Acknowledgments
This work was financially supported by the National S&T Major Project of China (No 2009ZX10004-601 and 2009ZX09311-003) and by the Leading Academic Discipline Project of Shanghai Municipal Education Commission (No J50301), E-institutes of Shanghai Municipal Education Commission (No E 03008).
References
- Choudhuri T, Pal S, Das T, Sa G. Curcumin selectively induces apoptosis in deregulated cyclin D1-expressed cells at G2 phase of cell cycle in a p53-dependent manner. J Biol Chem. 2005;280:20059–68. doi: 10.1074/jbc.M410670200. [DOI] [PubMed] [Google Scholar]
- Sherr CJ. Cancer cell cycles. Science. 1996;274:1672–7. doi: 10.1126/science.274.5293.1672. [DOI] [PubMed] [Google Scholar]
- Hall M, Peters G. Genetic alterations of cyclins, cyclin-dependent kinases, and Cdk inhibitors in human cancer. Adv Cancer Res. 1996;68:67–108. doi: 10.1016/s0065-230x(08)60352-8. [DOI] [PubMed] [Google Scholar]
- Dickson MA, Schwartz GK. Development of cell-cycle inhibitors for cancer therapy. Curr Oncol. 2009;16:36–43. doi: 10.3747/co.v16i2.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherr CJ, Roberts JM. cdk inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 1999;13:1501–12. doi: 10.1101/gad.13.12.1501. [DOI] [PubMed] [Google Scholar]
- Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M, et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell. 1992;69:119–28. doi: 10.1016/0092-8674(92)90123-t. [DOI] [PubMed] [Google Scholar]
- Sofer-Levi Y, Resnitzky D. Apoptosis induced by ectopic expression of cyclin D1 but not cyclin E. Oncogene. 1996;13:2431–7. [PubMed] [Google Scholar]
- Karin M, Hunter T. Transcriptional control by protein phosphorylation: signal transmission from the cell surface to the nucleus. Curr Biol. 1995;5:747–57. doi: 10.1016/s0960-9822(95)00151-5. [DOI] [PubMed] [Google Scholar]
- Tina M, Mercedes T. RinconNon-classical P38 map kinase functions: cell cycle checkpoints and survival. Int J Biol Sci. 2009;5:44–52. doi: 10.7150/ijbs.5.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim GY, Mercer SE, Ewton DZ, Yan Z, Jin K, Friedman E. The stress-activated protein kinases p38 alpha and JNK1 stabilize p21(Cip1) by phosphorylation. J Biol Chem. 2002;277:29792–802. doi: 10.1074/jbc.M201299200. [DOI] [PubMed] [Google Scholar]
- Kishi H, Nakagawa K, Matsumoto M, Suga M, Ando M, Taya Y, Yamaizumi M. Osmotic shock induces G1 arrest through p53 phosphorylation at Ser33 by activated p38MAPK without phosphorylation at Ser15 and Ser20. J Biol Chem. 2001;276:39115–22. doi: 10.1074/jbc.M105134200. [DOI] [PubMed] [Google Scholar]
- Lavoie JN, L''Allemain G, Brunet A, Müller R, Pouysségur J. Cyclin D1 expression is regulated positively by the p42/p44MAPK and negatively by the p38/HOGMAPK pathway. J Biol Chem. 1996;271:20608–16. doi: 10.1074/jbc.271.34.20608. [DOI] [PubMed] [Google Scholar]
- Collins K, Jacks T, Pavletich NP. The cell cycle and cancer. Proc Natl Acad Sci USA. 1997;94:2776–8. doi: 10.1073/pnas.94.7.2776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Q, Geng Y, Sicinski P. Specific protection against breast cancers by cyclin D1 ablation. Nature. 2001;411:1017–21. doi: 10.1038/35082500. [DOI] [PubMed] [Google Scholar]
- Martin LA, Head JE, Pancholi S, Salter J, Quinn E, Detre S, et al. The farnesyltransferase inhibitor R115777 (tipifarnib) in combination with tamoxifen acts synergistically to inhibit MCF-7 breast cancer cell proliferation and cell cycle progression in vitro and in vivo. Mol Cancer Ther. 2007;6:2458–67. doi: 10.1158/1535-7163.MCT-06-0452. [DOI] [PubMed] [Google Scholar]
- Sun J, Hai Liu R. Cranberry phytochemical extracts induce cell cycle arrest and apoptosis in human MCF-7 breast cancer cells. Cancer Lett. 2006;241:124–34. doi: 10.1016/j.canlet.2005.10.027. [DOI] [PubMed] [Google Scholar]
- Kunnumakkara AB, Anand P, Aggarwal BB. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008;269:199–225. doi: 10.1016/j.canlet.2008.03.009. [DOI] [PubMed] [Google Scholar]
- Aggarwal BB, Kumar A, Bharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 2003;23:363–98. [PubMed] [Google Scholar]
- Choudhuri PS, Das T, Sa G. Curcumin selectively induces apoptosis in deregulated cyclin D1-expressed cells at G2 phase of cell cycle in a p53-dependent manner. J Biol Chem. 2005;20:20059–68. doi: 10.1074/jbc.M410670200. [DOI] [PubMed] [Google Scholar]
- Park MJ, Kim EH, Park IC, Lee HC, Woo SH, Lee JY, et al. Curcumin inhibits cell cycle progression of immortalized human umbilical vein endothelial (ECV304) cells by up-regulating cyclin-dependent kinase inhibitor, p21WAF1/CIP1, p27KIP1 and p53. Int J Oncol. 2002;21:379–83. [PubMed] [Google Scholar]
- Srivastava RK, Chen Q, Siddiqui I, Sarva K, Shankar S. Linkage of curcumin-induced cell cycle arrest and apoptosis by cyclin-dependent kinase inhibitor p21(/WAF1/CIP1) Cell Cycle. 2007;6:2953–61. doi: 10.4161/cc.6.23.4951. [DOI] [PubMed] [Google Scholar]
- Chalasani P, Kurtin S, Dragovich T. Response to a third-Line mitomycin C (MMC)-based chemotherapy in a patient with metastatic pancreatic adenocarcinoma carrying germline BRCA2 mutation. JOP. 2008;9:305–8. [PubMed] [Google Scholar]
- Kang SG, Chung H, Yoo YD, Lee JG, Choi YI, Yu YS. Mechanism of growth inhibitory effect of mitomycin-C on cultured human retinal pigment epithelial cells: apoptosis and cell cycle arrest. Curr Eye Res. 2001;22:174–81. doi: 10.1076/ceyr.22.3.174.5513. [DOI] [PubMed] [Google Scholar]
- Sánchez P, Llorente MT, Castaño A. Flow cytometric detection of micronuclei and cell cycle alterations in fish-derived cells after exposure to three model genotoxic agents: mitomycin C, vincristine sulfate and benzo(a)pyrene. Mutat Res. 2000;465:113–22. doi: 10.1016/s1383-5718(99)00218-1. [DOI] [PubMed] [Google Scholar]
- Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunological Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- Chen J, Hui E, Ip T, Thompson LU. Dietary flaxseed enhances the inhibitory effect of tamoxifen on the growth of estrogen-dependent human breast cancer (MCF-7) in nude mice. Clin Cancer Res. 2004;10:7703–11. doi: 10.1158/1078-0432.CCR-04-1130. [DOI] [PubMed] [Google Scholar]
- Campbell FC, Collett GP. Chemopreventive properties of curcumin. Future Oncol. 2005;1:405–14. doi: 10.1517/14796694.1.3.405. [DOI] [PubMed] [Google Scholar]
- Chanvorachote P, Pongrakhananon V, Wannachaiyasit S, Luanpitpong S, Rojanasakul Y, Nimmannit U. Curcumin sensitizes lung cancer cells to cisplatin-induced apoptosis through superoxide anion-mediated Bcl-2 degradation. Cancer Invest. 2009;27:624–35. doi: 10.1080/07357900802653472. [DOI] [PubMed] [Google Scholar]
- Zhou QM, Zhang H, Lu YY, Wang XF, Su SB. Curcumin reduced the side effects of mitomycin C by inhibiting GRP58-mediated DNA cross-linking in MCF-7 breast cancer xenografts. Cancer Sci. 2009;100:2040–5. doi: 10.1111/j.1349-7006.2009.01297.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choudhuri T, Pal S, Agwarwal ML, Das T, Sa G. Curcumin induces apoptosis in human breast cancer cells througeh p53-dpendent Bax induction. FEBS Lett. 2002;12:334–40. doi: 10.1016/s0014-5793(02)02292-5. [DOI] [PubMed] [Google Scholar]
- Galderisi U, Jori FP, Giordano A. Cell cycle regulation and neural differentiation. Oncogene. 2003;22:5208–19. doi: 10.1038/sj.onc.1206558. [DOI] [PubMed] [Google Scholar]
- Fung TK, Poon RY. A roller coaster ride with the mitotic cyclins. Semin Cell Dev Biol. 2005;16:335–42. doi: 10.1016/j.semcdb.2005.02.014. [DOI] [PubMed] [Google Scholar]
- Neve RM, Holbro T, Hynes NE. Distinct roles for phosphoinositide 3-kinase, mitogen-activated protein kinase and p38 MAPK in mediating cell cycle progression of breast cancer cells. Oncogene. 2002;21:4567–76. doi: 10.1038/sj.onc.1205555. [DOI] [PubMed] [Google Scholar]
- Hsu YL, Kuo PL, Lin LT, Lin CC. Asiatic acid, a triterpene, induces apoptosis and cell cycle arrest through activation of extracellular signal-regulated kinase and p38 mitogen-activated protein kinase pathways in human breast cancer cells. J Pharmacol Exp Ther. 2005;313:333–44. doi: 10.1124/jpet.104.078808. [DOI] [PubMed] [Google Scholar]
- Lavelle D, DeSimone J, Hankewych M, Kousnetzova T, Chen YH. Decitabine induces cell cycle arrest at the G1 phase via p21(WAF1) and the G2/M phase via the p38 MAP kinase pathway. Leuk Res. 2003;27:999–1007. doi: 10.1016/s0145-2126(03)00068-7. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Leonard SS, Huang C, Vallyathan V, Castranova V, Shi X. Role of reactive oxygen species and MAPKs in vanadate-induced G(2)/M phase arrest. Free Radic Biol Med. 2003;34:1333–42. doi: 10.1016/s0891-5849(03)00145-x. [DOI] [PubMed] [Google Scholar]
- Todd DE, Densham RM, Molton SA, Balmanno K, Newson C, Weston CR, Garner AP, Scott L, Cook SJ. ERK1/2 and p38 cooperate to induce a p21CIP1-dependent G1 cell cycle arrest. Oncogene. 2004;23:3284–95. doi: 10.1038/sj.onc.1207467. [DOI] [PubMed] [Google Scholar]
- Lee B, Kim CH, Moon SK. Honokiol causes the p21WAF1-mediated G(1)-phase arrest of the cell cycle through inducing p38 mitogen activated protein kinase in vascular smooth muscle cells. FEBS Lett. 2006;580:5177–84. doi: 10.1016/j.febslet.2006.08.064. [DOI] [PubMed] [Google Scholar]
- Milacic V, Banerjee S, Landis-Piwowar KR, Sarkar FH, Majumdar AP, Dou QP. Curcumin inhibits the proteasome activity in human colon cancer cells in vitro and in vivo. Cancer Res. 2008;68:7283–92. doi: 10.1158/0008-5472.CAN-07-6246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somers-Edgar TJ, Scandlyn MJ, Stuart EC, Le Nedelec MJ, Valentine SP, Rosengren RJ. The combination of epigallocatechin gallate and curcumin suppresses ER alpha-breast cancer cell growth in vitro and in vivo. Int J Cancer. 2008;122:1966–71. doi: 10.1002/ijc.23328. [DOI] [PubMed] [Google Scholar]
- Dobrowsky W, Dobrowsky E, Wilson GD. In vivo cell kinetic measurements in a randomized trial of continuous hyperfractionated accelerated radiotherapy with or without mitomycin C in head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2003;55:576–82. doi: 10.1016/s0360-3016(02)04121-4. [DOI] [PubMed] [Google Scholar]
- Ishihara M, Kubota T, Watanabe M, Kawano Y, Narai S, Yasui N, et al. Interferon gamma increases the antitumor activity of mitomycin C against human colon cancer cells in vitro and in vivo. Oncol Rep. 1999;6:621–5. doi: 10.3892/or.6.3.621. [DOI] [PubMed] [Google Scholar]