Abstract
More and more attention in the field of drug discovery has been focused on the neuroprotection of natural compounds from traditional medicinal herbs. Cerebral ischemia is a complex pathological process involving a series of mechanisms, and a framework for the development of neuroprotectants from traditional herb medicine is a promising treatment for cerebral ischemia. Natural compounds with the effects of anti-oxidation, anti-inflammation, calcium antagonization, anti-apoptosis, and neurofunctional regulation exhibit preventive or therapeutic effects on experimental ischemic brain injury. According to the pharmacological mechanisms underlying neuroprotection, we evaluated natural products from traditional medicinal herbs that exhibit protective effects on ischemic brain injury and characterized the promising targets.
Keywords: cerebral ischemia, natural compounds, traditional medicinal herb
Introduction
Ischemic brain injury, one of the leading causes of death and adult disability worldwide, has attracted more and more attention in the field of drug discovery. Cerebral ischemia is often caused by transient or permanent reduction of cerebral blood flow initiated by thrombotic or thromboembolic arterial occlusions. Thrombolytic therapy designed to restore cerebral perfusion in a timely fashion is considered the main rational therapeutic strategy for ischemic brain injury1. However, reperfusion after thrombolytic therapy often leads to a series of cellular, biochemical and metabolic consequences of cerebral ischemia, including intracellular reactive oxygen species (ROS) generation, calcium overload, excitotoxic cell injury and inflammation, which ultimately lead to irreversible brain injury. Many neuroprotective agents are designed to protect the brain from irreversible injury after ischemia-reperfusion or to retard the pathological process2, 3.
Recent studies have focused on the possible capacity of natural compounds extracted from fruits, vegetables and beverages to prevent certain age-related neurological disorders. Some beneficial phytochemicals, especially polyphenols such as quercetin, (+)-catechin and resveratrol, display protective abilities in various animal models of neurological disorders. The underlying mechanisms include ischemic preconditioning, antioxidation, anti-inflammation, inhibition of microglia recruitment; in terms of underlying mechanisms, the “preconditioning” or “neurohormone” pathways seem most attractive. Examples of such “preconditioning” or “neurohormone” pathways include those involving cell-survival signaling kinases, the transcription factor NF-E2-related factor 2 (Nrf2) and cAMP-response element binding protein (CREB), as well as histone deacetylases of the sirtuin family. Activation of these pathways might protect neurons against injury by upregulation of antioxidant capacity, neurotrophic factors and protein chaperones to help cells withstand stress4, 5, 6. In fact, many effective components extracted from traditional herbs have been demonstrated to show neuroprotection against ischemic brain injury in experimental studies. According to the pharmacological mechanisms elucidated in numerous reports, we evaluated the natural products that possess protective effects on ischemic brain injury and characterized the promising targets for ischemic brain injury.
Natural compounds with anti-oxidative properties
A growing body of research has reported that a burst of ROS is produced during ischemia/reperfusion, which leads to the oxidation of lipids, proteins and DNA and subsequently cellular damage and apoptosis7, 8. Therefore, much attention has been paid to the rescue of brain injury after ischemia/reperfusion via inhibition of ROS bursts. In fact, many natural compounds with antioxidant ability, such as flavonoids from Scutellaria baicalensis Georgi9, Carnosic acid (CA, found in the herb rosemary obtained from Rosmarinus officinalis)10, Curcuma Oil (isolated from powdered rhizomes of Curcumalonga Linn)11, Ginkgo biloba extract EGb76112, and Cinnamophilin (isolated from Cinnamomum philippinense)13, exhibit significant neuroprotective effects when they are administered before cerebral ischemia occurs, but the related mechanisms or targets have been identified for only a few. For instance, flavonoids from Scutellaria baicalensis Georgi, when either pre-treated or post-treated, are demonstrated to decrease levels of malondialdehyde (MDA) and increase the level of superoxide dismutase (SOD) in the ischemic brains of mice. Aside from the anti-oxidant effects, flavonoids are also found to inhibit platelet aggregation, which is important to improve ischemic brain injury9. Pretreatment with curcuma oil, isolated from powdered rhizomes of Curcuma longa Linn, significantly reduces the levels of NO, ROS, ONOO−, and mitochondrial membrane potential11.
There are more and more reports focusing on the precise mechanisms or direct targets of natural antioxidant compounds protecting against cerebral ischemia. Some natural compounds exhibit direct regulatory effects on endogenous antioxidant enzyme systems. For example, Heme oxygenase (HO) is the rate-limiting enzyme for catabolism of the pro-oxidant heme. Two isoforms of HO exist: an inducible form (HO-1) and a constitutively expressed form (HO-2). HO-1 can be induced in response to various noxious stimuli (such as hypoxia and oxidative stress) and is considered a gene that protects against I/R injury14, 15, 16. CA, a catechol-type electrophilic compound found in the herb rosemary obtained from Rosmarinus officinalis, is shown to be neuroprotective when injected 1 h prior to MCAO in mice. As a representative electrophile, CA can induce the expression of a set of antioxidant enzymes, including heme oxygenase-1 (HO-1), NADPH quinone oxidoreductase 1 (NQO1), and c-glutamyl cysteine ligase (c-GCL). CAs become electrophilic quinones upon oxidation, with protective effects against neuronal oxidative stress and excitotoxicity via binding to specific Keap1 cysteine residues as a direct drug target, and then activating the Keap1/Nrf2 transcriptional pathway. The most attractive advantage of this agent is that it is a pro-electrophilic compound that can be activated by the microenvironment of oxidative stress and only becomes electrophilic at or near the site of ischemic brain tissues, with lower toxicity to normal tissues10. The protective effect of Ginkgo biloba extract on cerebral ischemia can be abolished in HO-1 knockout mice, suggesting that HO-1 is the key target of neuroprotection against free radical damage12. In cerebral ischemia, nitro oxide (NO) plays both a protective and a destructive role at different stages of this complex process. The beneficial effects of NO are related to the small amount of NO produced by endothelial nitric oxide synthase (eNOS), which produces significant effects on the maintenance of cerebral blood flow, prevention of neuronal injury by activation of the GC-cGMP-PKG pathway, and inhibition of platelet as well as leukocyte adhesion, and therefore protects against cerebral ischemia17, 18. Refined Qing Kai Ling (RQKL), an improved injectable multi-component preparation derived from Qing Kai Ling, shows neuroprotection in MCAO rats by relieving vascular endothelial cell damage as well as inhibiting inflammation. More importantly, RQKL was able to stimulate the post-ischemic expression of eNOS, which might be an essential part of the neuroprotective mechanisms of RQKL19. However, the large amount of NO, which is derived from inducible nitric oxide synthase (iNOS), harms neurons by producing peroxynitrite after the reaction with superoxide. Peroxynitrite can inhibit the mitochondrial respiratory chain, which implicates the involvement of ATP loss and eventually leads to irreversible cellular damage20, 21. Tetrahydroxystilbene glucoside (TSG), an active component of the rhizome extract from Polygomum multiflorum, has been reported to attenuate intracellular ROS generation and mitochondrial membrane potential dissipation caused by ischemia/reperfusion. Interestingly, it can directly upregulate the expression of sirt1, which is a class III histone deacetyltransferase that promotes cell survival and subsequently reduces the expression and activity of iNOS. This in turn induces the increase in NO production as well as peroxynitrite formation and results in apoptotic cell death by inhibiting the phosphorylation and subsequent degradation of I-κB, thereby hampering the DNA binding of nuclear factor kappa-B (NF-κB) by sirt1 activation22. More evidence has emphasized the significance of sirt1 in promoting cell survival, regulating lifespan and inhibiting inflammation. Recently, sirt1 has been introduced for the therapy of neurodegenerative diseases23, 24. Many natural compounds such as resveratrol, butein and quercetin, which are known as anti-aging agents, have been found to directly activate sirt125, suggesting that sirt1 may be the direct target of many herbal components that exhibit anti-aging effects. Phosphoinositide 3-kinases (PI3K)/Akt regulate the survival response against oxidative stress-associated neuronal apoptosis, which is determined by the balance between the activity of PI3K and the phosphatase and tensin homolog (PTEN). Activation of Akt promotes cell survival and suppresses apoptosis by inhibition of several downstream substrates, including glycogen synthase kinase-3β (GSK3β). PTEN is a major negative regulator of the PI3K/Akt signaling pathway and has been demonstrated to act as an important mediator of ROS production and mitochondria-dependent apoptosis26. A recent study reveals that Baicalein (Bai), one flavonoid extracted from Scutellaria baicalensis Georgi, when administered either prior to or after ischemia, can significantly protect against brain injury. Similarly, incubation with Bai reverses the rapid PTEN dephosphorylation after oxygen-glucose deprivation (OGD) in cultured hippocampal neurons. PTEN siRNA largely abolished the protection of Bai against OGD-induced cell injury, which highlighted the critical role of PTEN signaling in Bai-mediated effects27. The cytoplasmic enzyme NADPH oxidase is another important target for ROS production in cerebral ischemia and has received increased attention in recent years. Excessive activation of the neuronal N-methyl-D-aspartate receptor (NMDAR) initiates superoxide formation and promotes neuronal death. Previous studies have suggested that mitochondria are the primary source of NMDAR-induced superoxide production, but there is no definitive evidence. A recent study revealed that activation of NADPH oxidase is required for NMDAR-mediated superoxide production28. Interestingly, some natural compounds inhibit NADPH oxidase. For instance, sinomenine, an alkaloid extracted from a Chinese medicinal plant, Sinomenium acutum, which has been used widely in the clinic, apparently inhibits the activation of microglial NADPH oxidase29. The ethanol extract of E rutaecarpa, an anti-inflammatory drug commonly used in traditional Chinese medicine includes four bioactive compounds (dehydroevodiamine, evodiamine, rutaecarpine, and synephrine) and exhibits antioxidative effects by inhibiting NADPH oxidase activity30. These novel medicinal targets and mechanisms provide innovative clues and can help researchers to screen drugs for therapeutic intervention in ischemic brain injury mediated by natural antioxidants.
Some natural antioxidant compounds exhibit therapeutic effects on brain ischemia in the clinic. For example, cinnamophilin, extracted from Cinnamomum philippinense, has been demonstrated to reduce brain infarction and improve neurobehavioral outcome when administered either 15 min before (pretreatment) or 2 h after the onset of middle cerebral artery occlusion (MCAO) (postischemic treatment)13. TSG also protected against brain injury at 2 h after cerebral ischemia22. A significant effect of curcuma oil (500 mg/kg body wt) given 4 h postischemia was also observed in a rat MCAO model31. These findings offer beneficial references for the application of natural antioxidants in clinical treatment of ischemia and related neuronal diseases.
Natural compounds with anti-inflammatory effects
Inflammation is obvious within several hours during ischemia/reperfusion injury; it contributes to secondary damage caused by the microglial activation and resident perivascular and parenchymal macrophages, as well as infiltration of peripheral inflammatory cells. Aside from regulation of inflammation at the molecular level, progressive ischemic brain injury is related to several post-injury inflammatory responses at the cellular level, including neutrophil response as early as 4 h after ischemia/reperfusion and delayed macrophage infiltration, which occurs several days later. Sufficient evidence has indicated that neutrophils play a key role in the development of ischemic brain damage, and the depletion of circulating neutrophils or inhibition of neutrophil infiltration is demonstrated to ameliorate ischemic cerebral injury32. Thus, more and more evidence suggests that anti-inflammatory treatment might reduce ischemic brain injury and facilitate recovery33. Many bioactive components from Chinese medicinal plants exhibit significant anti-inflammatory effects, and recent studies have revealed that some exhibit neuroprotection against cerebral ischemia. Such compounds include theaflavin34, Wogonin (a flavonoid derived from the root of a medicinal herb, Scutellaria baicalensis Georgi)35, and Graptopetalum paraguayense E Walther leaf extracts36. Pretreatment with these bioactive components diminishes microglia-mediated inflammatory activity and neutrophil responses after brain ischemia.
Few studies have elucidated in detail the precise target for natural anti-inflammatory agents used to treat cerebral ischemia, suggesting that its precise mechanism of neuroprotection is still far from definite. Recently, several new targets closely related to inflammation, such as peroxisome proliferator-activated receptor-γ (PPARγ) and NF-κB, have been reported to mediate inflammation in activated macrophages by regulation of gene promoter regions, leading to inflammatory gene transcription37. Some plant-derived compounds exhibit direct regulation of PPARγ and NF-κB. For example, unique polychlorinated compounds named chlorophyllins A-C exhibit potent PPARγ agonistic effects38 and prenyloxycinnamic acid derivative 4′-geranyloxyferulic acid, obtained from Acronychia baueri Schott, also increases PPARγ activity significantly39. Incensole acetate and its nonacetylated form, incensole (IN), isolated from Boswellia Resin, a major anti-inflammatory agent in the herbal medical tradition, inhibits NF-κB activation40. The anti-inflammatory compound parthenolide from the medicinal herb Feverfew (Tanacetum parthenium) directly binds to and inhibits I-κB kinase41. Hesperetin, a flavanone derived from citrus fruits, suppresses NF-κB activation in both young and old rats through multiple signal transduction pathways42. These exciting studies indicate that nuclear transcription factors may serve as the direct target of natural anti-inflammatory compounds. More attention should be paid to the role of these transcription factors in the neuroprotective effects of natural anti-inflammatory compounds.
Natural compounds with calcium antagonization
ROS bursts and excitatory amino acid toxicity caused by ischemic reperfusion will lead to intracellular Ca2+ overload. Ca2+ overload in neurons is an essential signal of catastrophic events leading to irreversible neuronal injury. As a result, effects of herb components on Ca2+ overload induced by ischemic reperfusion have been studied extensively. Some herb components, for example, guattegaumerine (bisbenzylisoquinoline alkaloid from Guatteria gaumeri)43 and TSG22, are found to reduce ischemia induced Ca2+ overload in neurons. However, the exact mechanisms have not been elucidated in most studies. The main pathways of Ca2+ overload after cerebral ischemia and reperfusion are as follows: 1) the depletion of ATP leads to inactivation of Na+,K+-ATPase, an enzyme regulating ionic concentration gradients for the generation of action potentials in neurons, with resultant depolarization of cell membrane potential and further opening of Ca2+-permeable cation channels such as voltage-gated calcium channels (VGCCs)44; 2) the abundant release of excitatory amino acid activates α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, thereby further depolarizing the cell membrane potential and resulting in massive activation of NMDAR45; 3) the activation of NMDAR induces NADPH oxidase- or mitochondria pathway-dependent ROS generation, with subsequent Ca2+ release from intracellular calcium stores and influx through transient receptor potential melastatin (TRPM) channels, mediating ROS-dependant Ca2+ overload46; 4) extracellular acidulation could activate some acid-sensing ion channels and receptors, especially the Ca2+ permeable acid-sensing ion channel 1a (ASIC1a)47. Honokiol, a component of the herb Magnolia officinalis, can attenuate the decrease in Na+, K+-ATPase activities induced by MCAO, but it has not been clarified whether the agent exhibits its effect by acting directly on Na+, K+-ATPase or by attenuating the pathological inducer48. Use of VGCC blockers has been considered a therapeutic approach for post-stroke neuroprotection in humans for many years. Previous studies have shown that some isoquinoline alkaloids contained in medicinal herbs such as berberine (an alkaloid derived from the herbal medicine Rhizoma coptidis and palmatine (a flavonoid in propolis) exhibit significant and rapid inhibition of voltage-gated calcium currents in many native cells49, 50. Interestingly, some of these alkaloids also exhibit neuroprotective effects against several central nervous system diseases51.
There are few reports about the direct inhibition of NMDA receptor by natural products. Recently, some cation channels closely related to Ca2+ overload and selectively activated in certain pathological conditions have been a focus of drug development, including ASICs and TRPM. ASICs, especially ASIC1, which has been reported to be activated by extracellular acidosis, play a key role in ischemic cerebral injury47. In recent studies, puerarin, extract from Radix puerariae, has been reported to inhibit ASICs current in both natural cells and transfected cells, indicating the advantages of isolating novel ASIC antagonists with low toxicity from natural products52. Inhibition of TRPM, a Ca2+-permeable nonselective cation channel, increases the resistance of neurons to ischemic death after brain ischemia and then preserves neuronal morphology and function. At present, several lines of evidence have demonstrated that accumulation of ADP-ribose induced by oxidative stress has led to the rapid opening of TRPM2 channels and calcium influx. TRPM2 might be the major pathway of cellular calcium overload induced by oxidation53. Blockade of TRPM2 could reduce the cellular injury induced by oxidation54. However, few research efforts have been conducted to explore compounds isolated from natural products that antagonize ADP-ribose or block TRPM2 channels. Certain natural drugs are able to decrease the intracellular Ca2+ overload induced by oxidation. For example, salvianolic acid B, isolated from Radix Salviae miltiorrhizae, protects PC12 cells against hydrogen peroxide-induced intracellular calcium overload55. More interestingly, the structure-activity relationship of quercetin, which is abundant in various fruits and vegetables such as apple and carrot, as well as red wine and tea, has been revealed to antagonize hydrogen peroxide-induced calcium dysregulation in PC12 cells56. In one study, hydrogen peroxide induced an intracellular calcium elevation that could not recover the Ca2+ levels in PC12 cells, but quercetin antagonized the effects of hydrogen peroxide in that cell model. Structure-activity relationships of five flavonoids were examined and the results indicate that two structural components, including (i) 3′,4′-hydroxyl (OH) groups in the B ring and (ii) a 2,3-double bond in conjugation with a 4-oxo group in the C ring, as well as the polyphenolic structures, are crucial for the protective effect. Based on the significance of TRPM channels in calcium overload induced by oxidation, more attention should be paid to the natural compounds that antagonize hydrogen peroxide-induced calcium dysregulation by regulation of TRPM channels. These studies indicate that the natural products that can antagonize the ischemia-induced calcium overload might act on these novel targets involving ASICs and TRPM channels rather than on traditional targets such as L-type calcium channels and NMDA receptors. Nevertheless, more evidence and further studies are necessary to support this hypothesis.
Natural compounds with anti-apoptotic effects
Ischemic cerebral injury is known to induce histopathological damage and related neurological deficits, leading to the activation of complex neurochemical cascades of cell death, which are primarily expressed as apoptosis. In principle, these apoptotic cascades are reversible and form an important aspect of the penumbra concept, a major target of therapeutic interventions. In general, ischemia and reperfusion induced neuronal apoptosis can be classified into two types: caspase-dependent and caspase-independent pathways. Caspases are intracellular proteases that function as initiators and effectors of apoptosis. When activated, caspases cleave a variety of intracellular proteins, including major structural elements in the cytoplasm and nucleus, components of the DNA repair machinery, and a number of protein kinases57. Thus, caspases may be effective against ischemia-induced neuronal cell apoptosis by blocking apoptotic cascades with appropriate drugs. An alkaloid-free ethyl acetate, extracted from the root of Sophora flavescens, has been reported to protect against focal cerebral ischemia by decreasing DNA fragmentation and inhibiting caspase-3 activity directly58. In that study, it was found that caspase-independent programmed cell death, mediated by the translocation of apoptosis-inducing factor (AIF) from the mitochondria to the nucleus, plays a key role in ischemia. During ischemia and reperfusion injury, in the case of massive and irreparable DNA damage, overactivation of Poly(ADP-ribose) Polymerase-1 (PARP-1) can lead to necrotic cell death caused by the depletion of NAD+ and ATP, as well as enhance AIF release from mitochondria59. A series of PARP-1 antagonists have been developed, some of which have become promising drug candidates60, 61. However, the characterization of natural compounds regulating PARP-1 remains to be elucidated. It is thought that berberine performs its anti-apoptotic effect by inhibiting both caspase-dependent and independent pathways62. Synergistic inhibition of caspase-dependent and independent neuronal cell apoptosis is more likely to be effective as a therapeutic approach for the treatment of ischemia.
Numerous herb drugs have been demonstrated to act on anti-apoptotic pathways, such as Bcl-2 family proteins. The abundant expression of Bcl-xL protein in the adult brain is known to suppress activation of procaspase 9 by forming a complex with Apaf1 and to prevent the release of cytochrome c from mitochondria, thus maintaining cell viability. Therefore, Bcl-xL becomes a promising target for drug intervention to reduce cell apoptosis63. Pretreatment with 4-hydroxybenzyl alcohol 30 min before ischemia, one of the major active phenolic constituents of Gastrodia elata Blume, could antagonize cerebral ischemia by increasing Bcl-2 expression and inhibiting caspase-3 activity, leading to the amelioration of cell apoptosis in ischemic regions64. Direct evidence from preclinical research shows that Ginsenoside Rb1 (gRb1) regulates the anti-apoptosis signaling pathway, which stimulates the expression of mitochondrion-associated anti-apoptotic factor Bcl-xL through the use of reporter plasmids. The transcription factor signal transducer and activator of transcription 5 (Stat5) are known to activate Bcl-xL family proteins via binding to the bcl-xL promoter. The Stat5 responsive element in the bcl-xL promoter becomes active in response to gRb1 treatment, suggesting that the Stat5 pathway participates in anti-apoptotic effects by regulating Bcl-2 indirectly through activation of gRb165. Molecular pharmacology research in this direction should elucidate the direct target of natural drugs that regulate apoptotic signaling pathways.
Natural compounds that affect neurofunctional regulation
Ischemia/reperfusion induces angiogenesis and increases cell proliferation in the subventricular zone (SVZ), which is home to neural stem/progenitor cells66. In order to amplify the intrinsic propensity for neuroplasticity and subsequent neurological recovery, it is necessary to promote basal neurogenesis and synaptogenesis. Several potential therapeutic agents have been shown to promote functional outcome after ischemia, most of which are growth factors such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), and brain-derived neurotrophic factor (BDNF). VEGF is a major mediator of angiogenesis that stimulates revascularization. VEGF receptors have been reported to be upregulated in the ischemic brain. It is believed that VEGF is beneficial for the recovery of neurological function after cerebral ischemia, at least partly because newly formed neuroblasts migrate into the damaged striatum and survive for several months after stroke67. Cornel iridoid glycoside has been reported to provide a better microenvironment for angiogenesis and cell proliferation by increasing VEGF levels68. Treatment with NeuroAid (MLC601, MLC901), a traditional medicine, has been shown to induce neurogenesis in rodent and human cells, promoting cell proliferation as well as neurite outgrowth, and to stimulate the development of dense axonal and dendritic networks. Furthermore, in in vivo experiments, either pre-treatment or post-treatment of MCAO with NeuroAid protects against brain infarction and neurological dysfunction. As in vitro data and other evidence show that MLC901 indeed increases BDNF expression in cortical neurons, it is proposed that BDNF plays an essential role in recovery from ischemia69. The pathological processes of cerebral ischemia ultimately lead to neurofunctional damage, such as learning and memory deficits. The extracellular signal-regulated protein kinase (ERK) cascade is one of the important pathways involved in hippocampal synaptogenesis and synaptic plasticity, which are important for learning and memory70. Natural products may act as neurohormetic phytochemicals to activate ERK and contribute to the restoration of learning and memory after cerebral ischemia. For instance, ERK activation is decreased dramatically after ischemic brain injury. Pamela et al found that bioflavonoid fisetin at the concentration of 1–5 μmol/L significantly promoted ERK activation in the hippocampal CA1 area and enhanced memory for novel objects in mice71. Cholinesterase inhibitors potentiate the function of the cholinergic system by inhibiting cholinesterase activity and thereby enhancing cognition. Some natural products such as (−)Huperzine A are used as cholinesterase inhibitors in the clinic72. As a result, natural drugs with anticholinesterase activity may have potential as treatments to facilitate post-ischemic functional recovery.
Discussion
Since brain ischemia is a complex progressive injury involving a series of mechanisms such as cell survival and apoptosis, ROS accumulation, Ca2+ overload, inflammation, microglia aggregation, etc, it is difficult to describe the mechanisms of protective agents against ischemia. Many agents derived from traditional medicinal herbs act not only on direct targets, but also on multiple pathways. For instance, Paeoniflorin (PF), the principal component of Paeoniae radix (used in traditional Chinese medicine), displays a preconditioning neuroprotective effect against cerebral ischemia in mice. The related mechanism has been shown to involve multiple factors including the adenosine 1 receptor/regulator of G protein/KATP signaling, the arachidonic acid cascade, the nitric oxide system, neuronal damage markers, molecules related to mitochondrial damage, mitogen-activated protein kinase, as well as the nuclear factor-κB pathway73. Pretreatment with another herb extract from Choto-san (Gouteng-san), a traditional Kampo medicine, also exhibits antidementia effects and neuroprotection against ischemia when administered at an appropriate dosage. The compound acts via the following mechanisms: blockade of calcium channels, scavenging of free radicals and inhibition of NMDA receptors. Moreover, some herbal glycoside recipes have been demonstrated to affect nine genes related to spatial learning and memory, thereby alleviating ischemic damage74. Considering that multifactorial and progressive pathophysiological processes are involved in cerebral ischemia therefore suggests that drugs binding to multiple targets or that combinations of drugs that act on single targets may be more effective in curing ischemia and related cerebral injury. Many international pharmaceutical companies have emphasized drugs with multiple targets, defined as drugs that possess at least two definite and independent action targets (eg, PAN-811)75, 76. In contrast to the multiple-target drugs that attracted the attention of several pharmaceutical companies, many natural products regulate multiple signaling pathways without potent effects on direct targets. Thus, it remains to be determined whether these traditional medicines are multiple-targeted or non-targeted agents. Theoretically, there certainly exist exact targets that the drugs act upon directly. For instance, as mentioned above, some important functional proteins including Keap1, Sirt1, NF-κB, and Stat510, 25, 41, 65 can bind directly to natural compounds. However, many key functional proteins that mediate the neuroprotection of natural compounds, such as HO-1 for CA, iNOS for TSG, PTEN for Bai and Bcl-2 for gRb110, 22, 26, 65, may not act as the primary binding targets of these compounds. Multiple signal pathways are involved in the pathways between direct targets and ultimate biological effects, including Nrf2, JNK, Akt, and GSK310, 22, 26. In addition to the discovery of novel functional molecules and signal pathways in ischemic cerebral injury, extending the restrictive traditional definition of drug targets may help to clarify the exact targets of natural products.
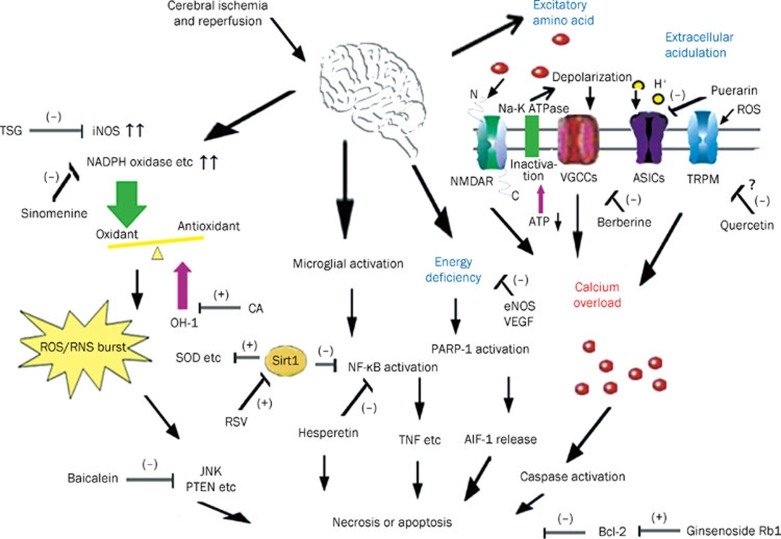
Although more and more agents are reported to be potentially effective for ischemia via numerous pathways, as determined in experimental environments, few display the same effects in clinic77. The reasons may include the following. First, several agents are difficult to transport across the blood brain barrier (BBB); it is therefore difficult to measure their neuroprotective effects. Second, more attention should be paid to the therapeutic time windows of drugs. Currently, most researchers still focus on the “preconditioning” or preventive application rather than post-ischemic treatment, which results in a very narrow therapeutic time window. Post-ischemic treatment is surely more meaningful for patients who have suffered ischemic stroke. Third, some potential agents show dramatic therapeutic effects on ischemia; however, therapeutic dosages may produce cellular toxicity or cause transmitter disorders, leading to severe side effects. Finally, according to the characteristics of many multi-component herb-resource agents, the exact mechanisms and targets remain to be elucidated. It is also difficult to determine whether the coordination- or single-compound strategy is more effective. However, this problem may be resolved by the development of separation techniques. Recent studies have demonstrated that some of these compounds, such as paeoniflorin78, emodin-8-O-β-D-glucoside79, baicalin80, and curcuma oil11, have the ability to penetrate the blood-brain barrier and achieve wide distribution in the brain, which is critical for CNS effects. It is easy to understand that the highly lipophilic structure of curcuma oil greatly enhances its access to the brain. However, the passage of glucosides and flavonoids through the BBB cannot be explained fully by traditional principles, due to their highly polar structures. Despite abundant evidence of their neuroprotective effects, little is known about how these compounds can enter the CNS. A recent study has demonstrated that brain endothelial cells, which form the BBB and are sealed by complex tight junctions that possess few pinocytotic vesicles but express a number of specific uptake and efflux transport systems and metabolic enzymes, may play an important role in the delivery of flavonoids and flavonoid glucosides to the CNS. These brain endothelial cells enable the BBB to restrict the passage of most small polar molecules, but selectively take up important polar molecules including glucose and amino acids. Recent studies showed that the citrus flavonoids (hesperetin, naringenin) and their relevant in vivo metabolites (cyanidin-3-rutinoside and pelargonidin-3-glucoside) can be taken up by two kinds of brain endothelial cell lines from mice (b.END5) and rats (RBE4), demonstrating that flavonoids and some metabolites are able to traverse the BBB81. These phenomena require further research, which may help to resolve the problems related to the extraction of active components from traditional Chinese drugs to treat CNS diseases. The multiple signal pathways and potential targets involved in the neuroprotection of natural compounds are shown in Figure 1.
Figure 1.
Multiple signal pathways and potential targets involved in the neuroprotection of natural compounds.
In conclusion, development of protective agents from traditional herb medicine is a promising direction in the treatment of ischemic cerebral injury and related neurodegenerative diseases. In the future, more attention should be paid to natural compounds that can transverse the BBB and have wide therapeutic time windows, clear pharmacological targets and fewer side effects.
Acknowledgments
The related works were supported by grants from the Key Project of National Natural Science Foundation of China (NSFC) (No 30930104), the National Basic Research Program of China (973 Program) (No 2007CB507404), and the Chang Jiang Scholar Program of the Ministry of Education of China to Dr JG CHEN. The work was also supported by grants from the Program for New Century Excellent Talents in Universities of China (NCET-08-0225) to Dr F WANG.
References
- Stoll G, Kleinschnitz C, Nieswandt B. Molecular mechanisms of thrombus formation in ischemic stroke: novel insights and targets for treatment. Blood. 2008;112:3555–62. doi: 10.1182/blood-2008-04-144758. [DOI] [PubMed] [Google Scholar]
- Green AR. Pharmacological approaches to acute ischaemic stroke: reperfusion certainly, neuroprotection possibly. Br J Pharmacol. 2008;153:S325–38. doi: 10.1038/sj.bjp.0707594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ly JV, Zavala JA, Donnan GA. Neuroprotection and thrombolysis: combination therapy in acute ischaemic stroke. Expert Opin Pharmacother. 2006;7:1571–81. doi: 10.1517/14656566.7.12.1571. [DOI] [PubMed] [Google Scholar]
- Mattson MP, Cheng AW. Neurohormetic phytochemicals: low-dose toxins that induce adaptive neuronal stress responses. Trends Neurosci. 2006;29:632–9. doi: 10.1016/j.tins.2006.09.001. [DOI] [PubMed] [Google Scholar]
- Doré S. Unique properties of polyphenol stilbenes in the brain: more than direct antioxidant actions; gene/protein regulatory activity. Neurosignals. 2005;14:61–70. doi: 10.1159/000085386. [DOI] [PubMed] [Google Scholar]
- van Praag H. Exercise and the brain: something to chew on. Trends Neurosci. 2009;32:283–90. doi: 10.1016/j.tins.2008.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niizuma K, Endo H, Chan PH. Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J Neurochem. 2009;109:133–8. doi: 10.1111/j.1471-4159.2009.05897.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crack PJ, Taylor JM. Reactive oxygen species and the modulation of stroke. Free Radic Biol Med. 2005;38:1433–44. doi: 10.1016/j.freeradbiomed.2005.01.019. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Wang X, Wang X, Xu Z, Liu Z, Ni Q, et al. Protective effect of flavonoids from Scutellaria baicalensis Georgi on cerebral ischemia injury. J Ethnopharmacol. 2006;108:355–60. doi: 10.1016/j.jep.2006.05.022. [DOI] [PubMed] [Google Scholar]
- Satoh T, Kosaka K, Itoh K, Kobayashi A, Yamamoto M, Shimojo Y, et al. Carnosic acid, a catechol-type electrophilic compound, protects neurons both in vitro and in vivo through activation of the Keap1/Nrf2 pathway via S-alkylation of targeted cysteines on Keap1. J Neurochem. 2008;104:1116–31. doi: 10.1111/j.1471-4159.2007.05039.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rathore P, Dohare P, Varma S, Ray A, Sharma U, Jagannathan NR, et al. Curcuma oil: reduces early accumulation of oxidative product and is anti-apoptogenic in transient focal ischemia in rat brain. Neurochem Res. 2008;33:1672–82. doi: 10.1007/s11064-007-9515-6. [DOI] [PubMed] [Google Scholar]
- Saleem S, Zhuang H, Biswal S, Christen Y, Doré S. Ginkgo biloba extract neuroprotective action is dependent on heme oxygenase 1 in ischemic reperfusion brain injury. Stroke. 2008;39:3389–96. doi: 10.1161/STROKEAHA.108.523480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee EJ, Chen HY, Lee MY, Chen TY, Hsu YS, Hu YL, et al. Cinnamophilin reduces oxidative damage and protects against transient focal cerebral ischemia in mice. Free Radic Biol Med. 2005;39:495–510. doi: 10.1016/j.freeradbiomed.2005.04.004. [DOI] [PubMed] [Google Scholar]
- Nimura T, Weinstein PR, Massa SM, Panter S, Sharp FR. Heme oxygenase-1 (HO-1) protein induction in rat brain following focal ischemia. Brain Res Mol Brain Res. 1996;37:201–8. doi: 10.1016/0169-328x(95)00315-j. [DOI] [PubMed] [Google Scholar]
- Doré S. Decreased activity of the antioxidant heme oxygenase enzyme: implications in ischemia and in Alzheimer's disease. Free Radic Biol Med. 2002;32:1276–82. doi: 10.1016/s0891-5849(02)00805-5. [DOI] [PubMed] [Google Scholar]
- Li RC, Saleem S, Zhen G, Cao W, Zhuang H, Lee J, et al. Heme-hemopexin complex attenuates neuronal cell death and stroke damage. J Cereb Blood Flow Metab. 2009;29:953–64. doi: 10.1038/jcbfm.2009.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Endres M, Laufs U, Liao JK, Moskowitz MA. Targeting eNOS for stroke protection. Trends Neurosci. 27:283–9. doi: 10.1016/j.tins.2004.03.009. [DOI] [PubMed] [Google Scholar]
- Ha KS, Kim KM, Kwon YG, Bai SK, Nam WD, Yoo YM, et al. Nitric oxide prevents 6-hydroxydopamine-induced apoptosis in PC12 cells through cGMP-dependent PI3 kinase/Akt activation. FASEB J. 2003;17:1036–47. doi: 10.1096/fj.02-0738com. [DOI] [PubMed] [Google Scholar]
- Hua Q, Zhu X, Li P, Tang H, Cai D, Xu Y, et al. Refined Qing Kai Ling, traditional Chinese medicinal preparation, reduces ischemic stroke-induced infarct size and neurological deficits and increases expression of endothelial nitric oxide synthase. Biol Pharm Bull. 2008;31:633–7. doi: 10.1248/bpb.31.633. [DOI] [PubMed] [Google Scholar]
- Bolaños JP, Almeida A. Roles of nitric oxide in brain hypoxia-ischemia. Biochim Biophys Acta. 1999;1411:415–36. doi: 10.1016/s0005-2728(99)00030-4. [DOI] [PubMed] [Google Scholar]
- del Zoppo G, Ginis I, Hallenbeck JM, Iadecola C, Wang X, Feuerstein GZ. Inflammation and stroke: putative role for cytokines, adhesion molecules and iNOS in brain response to ischemia. Brain Pathol. 2000;10:95–112. doi: 10.1111/j.1750-3639.2000.tb00247.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T, Gu J, Wu PF, Wang F, Xiong Z, Yang YJ, et al. Protection by tetrahydroxystilbene glucoside against cerebral ischemia: involvement of JNK, SIRT1, and NF-kappaB pathways and inhibition of intracellular ROS/RNS generation. Free Radic Biol Med. 2009;47:229–40. doi: 10.1016/j.freeradbiomed.2009.02.027. [DOI] [PubMed] [Google Scholar]
- Lavu S, Boss O, Elliott PJ, Lambert PD. Sirtuins — novel therapeutic targets to treat age-associated diseases. Nat Rev Drug Discov. 2008;7:841–53. doi: 10.1038/nrd2665. [DOI] [PubMed] [Google Scholar]
- Tang BL, Chua CE. SIRT1 and neuronal diseases. Mol Aspects Med. 2008;29:187–200. doi: 10.1016/j.mam.2007.02.001. [DOI] [PubMed] [Google Scholar]
- Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG, et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 2003;425:191–6. doi: 10.1038/nature01960. [DOI] [PubMed] [Google Scholar]
- Waite KA, Eng C. Protean PTEN: form and function. Am J Hum Genet. 2002;70:829–44. doi: 10.1086/340026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Wu J, Xu K, Cai F, Gu J, Ma L, et al. Neuroprotection by baicalein in ischemic brain injury involves PTEN/AKT pathway. J Neurochem. 2010;112:1500–12. doi: 10.1111/j.1471-4159.2009.06561.x. [DOI] [PubMed] [Google Scholar]
- Brennan AM, Suh SW, Won SJ, Narasimhan P, Kauppinen TM, Lee H, et al. NADPH oxidase is the primary source of superoxide induced by NMDA receptor activation. Nat Neurosci. 2009;12:857–63. doi: 10.1038/nn.2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian L, Xu Z, Zhang W, Wilson B, Hong JS, Flood PM. Sinomenine, a natural dextrorotatory morphinan analog, is anti-inflammatory and neuroprotective through inhibition of microglial NADPH oxidase. J Neuroinflammation. 2007;4:23. doi: 10.1186/1742-2094-4-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko HC, Wang YH, Liou KT, Chen CM, Chen CH, Wang WY, et al. Anti-inflammatory effects and mechanisms of the ethanol extract of Evodia rutaecarpa and its bioactive components on neutrophils and microglial cells. Eur J Pharmacol. 2007;555:211–7. doi: 10.1016/j.ejphar.2006.10.002. [DOI] [PubMed] [Google Scholar]
- Dohare P, Garg P, Sharma U, Jagannathan NR, Ray M. Neuroprotective efficacy and therapeutic window of curcuma oil: in rat embolic stroke model. BMC Complement Altern Med. 2008;8:55. doi: 10.1186/1472-6882-8-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordán J, Segura T, Brea D, Galindo MF, Castillo J. Inflammation as therapeutic objective in stroke. Curr Pharm Des. 2008;14:3549–64. doi: 10.2174/138161208786848766. [DOI] [PubMed] [Google Scholar]
- Barone FC, Feuerstein GZ. Inflammatory mediators and stroke: new opportunities for novel therapeutics. J Cereb Blood Flow Metab. 1999;19:819–34. doi: 10.1097/00004647-199908000-00001. [DOI] [PubMed] [Google Scholar]
- Cai F, Li CR, Wu JL, Chen JG, Liu C, Min Q, et al. Theaflavin ameliorates cerebral ischemia-reperfusion injury in rats through its anti-inflammatory effect and modulation of STAT-1. Mediators Inflamm. 2006;2006:30490. doi: 10.1155/MI/2006/30490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho J, Lee HK. Wogonin inhibits ischemic brain injury in a rat model of permanent middle cerebral artery occlusion. Biol Pharm Bull. 2004;27:1561–4. doi: 10.1248/bpb.27.1561. [DOI] [PubMed] [Google Scholar]
- Kao TK, Ou YC, Raung SL, Chen WY, Yen YJ, Lai CY, et al. Graptopetalum paraguayense E. Walther leaf extracts protect against brain injury in ischemic rats. Am J Chin Med. 2010;38:495–516. doi: 10.1142/S0192415X10008019. [DOI] [PubMed] [Google Scholar]
- Yi JH, Park SW, Kapadia R, Vemuganti R. Role of transcription factors in mediating post-ischemic cerebral inflammation and brain damage. Neurochem Int. 2007;50:1014–27. doi: 10.1016/j.neuint.2007.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee IY, Lee JH, Yun BS. Polychlorinated compounds with PPAR-gamma agonistic effect from the medicinal fungus Phellinus ribis. Bioorg Med Chem Lett. 2008;18:4566–8. doi: 10.1016/j.bmcl.2008.07.034. [DOI] [PubMed] [Google Scholar]
- Genovese S, Foreman JE, Borland MG, Epifano F, Gonzalez FJ, Curini M, et al. A natural propenoic acid derivative activates peroxisome proliferator-activated receptor-β/δ (PPARβ/δ) Life Sci. 2010;86:493–8. doi: 10.1016/j.lfs.2010.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moussaieff A, Shohami E, Kashman Y, Fride E, Schmitz ML, Renner F, et al. Incensole acetate, a novel anti-inflammatory compound isolated from Boswellia resin, inhibits nuclear factor-kappa B activation. Mol Pharmacol. 2007;72:1657–64. doi: 10.1124/mol.107.038810. [DOI] [PubMed] [Google Scholar]
- Kwok BH, Koh B, Ndubuisi MI, Elofsson M, Crews CM. The anti-infammatory natural product parthenolide from the medicinal herb Feverfew directly binds to and inhibits Ikappa kinase. Chem Biol. 2001;8:759–766. doi: 10.1016/s1074-5521(01)00049-7. [DOI] [PubMed] [Google Scholar]
- Kim JY, Jung KJ, Choi JS, Chung HY. Modulation of the age-related nuclear factor- kappaB(NF-kappaB) pathway by hesperetin. Aging Cell. 2006;5:401–11. doi: 10.1111/j.1474-9726.2006.00233.x. [DOI] [PubMed] [Google Scholar]
- Lü Q, Xu XL, He Z, Huang XJ, Guo LJ, Wang HX. Guattegaumerine protects primary cultured cortical neurons against oxidative stress injury induced by hydrogen peroxide concomitant with serum deprivation. Cell Mol Neurobiol. 2009;29:355–64. doi: 10.1007/s10571-008-9327-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decollogne S, Bertrand IB, Ascensio M, Drubaix I, Lelièvre LG. Na+, K+-ATPase and Na+/Ca2+ exchange isoforms: physiological and physiopathological relevance. J Cardiovasc Pharmacol. 1993;22:S96–8. [PubMed] [Google Scholar]
- Salińska E, Lazarewicz JW. NMDA receptor-mediated calcium fluxes in the hippocampus: relevance to ischemic brain pathology. Neurol Neurochir Pol. 1996;30:35–42. [PubMed] [Google Scholar]
- Simard JM, Tarasov KV, Gerzanich V. Non-selective cation channels, transient receptor potential channels and ischemic stroke. Biochim Biophys Acta. 2007;1772:947–57. doi: 10.1016/j.bbadis.2007.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong ZG, Chu XP, Simon RP. Ca2+-permeable acid-sensing ion channels and ischemic brain injury. J Membr Biol. 2006;209:59–68. doi: 10.1007/s00232-005-0840-x. [DOI] [PubMed] [Google Scholar]
- Chen CM, Liu SH, Lin-Shiau SY. Honokiol, a neuroprotectant against mouse cerebral ischemia, mediated by preserving Na+, K+-ATPase activity and mitochondrial functions. Basic Clin Pharmacol Toxicol. 2007;101:108–16. doi: 10.1111/j.1742-7843.2007.00082.x. [DOI] [PubMed] [Google Scholar]
- Wang F, Zhou HY, Cheng L, Zhao G, Zhou J, Fu LY, et al. Effects of palmatine on potassium and calcium currents in isolated rat hepatocytes. World J Gastroenterol. 2003;9:329–33. doi: 10.3748/wjg.v9.i2.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F, Zhou HY, Zhao G, Fu LY, Cheng L, Chen JG, et al. Inhibitory effects of berberine on ion channels of rat hepatocytes. World J Gastroenterol. 2004;10:2842–5. doi: 10.3748/wjg.v10.i19.2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulkarni SK, Dhir A. Berberine: a plant alkaloid with therapeutic potential for central nervous system disorders. Phytother Res. 2010;24:317–24. doi: 10.1002/ptr.2968. [DOI] [PubMed] [Google Scholar]
- Gu L, Yang Y, Sun Y, Zheng X. Puerarin inhibits acid-sensing ion channels and protects against neuron death induced by acidosis. Planta Med. 2010;76:583–8. doi: 10.1055/s-0029-1240583. [DOI] [PubMed] [Google Scholar]
- Kühn FJ, Heiner I, Lückhoff A. TRPM2: a calcium influx pathway regulated by oxidative stress and the novel second messenger ADP-ribose. Pflugers Arch. 2005;451:212–9. doi: 10.1007/s00424-005-1446-y. [DOI] [PubMed] [Google Scholar]
- Yang KT, Chang WL, Yang PC, Chien CL, Lai MS, Su MJ, et al. Activation of the transient receptor potential M2 channel and poly(ADP-ribose)polymerase is involved in oxidative stress-induced cardiomyocyte death. Cell Death Differ. 2006;13:1815–26. doi: 10.1038/sj.cdd.4401813. [DOI] [PubMed] [Google Scholar]
- Liu CS, Chen NH, Zhang JT. Protection of PC12 cells from hydrogen peroxide-induced cytotoxicity by salvianolic acid B, a new compound isolated from Radix Salviae miltiorrhizae. Phytomedicine. 2007;14:492–7. doi: 10.1016/j.phymed.2006.11.002. [DOI] [PubMed] [Google Scholar]
- Wang H, Joseph JA. Structure-activity relationships of quercetin in antagonizing hydrogen peroxide-induced calcium dysregulation in PC12 cells. Free Radic Biol Med. 1999;27:683–94. doi: 10.1016/s0891-5849(99)00119-7. [DOI] [PubMed] [Google Scholar]
- Broughton BR, Reutens DC, Sobey CG. Apoptotic mechanisms after cerebral ischemia. Stroke. 2009;40:e331–9. doi: 10.1161/STROKEAHA.108.531632. [DOI] [PubMed] [Google Scholar]
- Park SJ, Nam KW, Lee HJ, Cho EY, Koo U, Mar W. Neuroprotective effects of an alkaloid-free ethyl acetate extract from the root of Sophora flavescens Ait. against focal cerebral ischemia in rats. Phytomedicine. 2009;16:1042–51. doi: 10.1016/j.phymed.2009.03.017. [DOI] [PubMed] [Google Scholar]
- van Wijk SJ, Hageman GJ. Poly(ADP-ribose) polymerase-1 mediated caspase-independent cell death after ischemia/reperfusion. Free Radic Biol Med. 2005;39:81–90. doi: 10.1016/j.freeradbiomed.2005.03.021. [DOI] [PubMed] [Google Scholar]
- Zhang WT, Ruan JL, Wu PF, Jiang FC, Zhang LN, Fang W, et al. Design, synthesis, and cytoprotective effect of 2-aminothiazole analogues as potent poly(adp-ribose) polymerase-1 inhibitors. J Med Chem. 2009;52:718–25. doi: 10.1021/jm800902t. [DOI] [PubMed] [Google Scholar]
- Ferraris D, Ficco RP, Dain D, Ginski M, Lautar S, Lee-Wisdom K, et al. Design and synthesis of poly(ADP-ribose) polymerase-1 (PARP-1) inhibitors. Part 4: Biological evaluation of imidazobenzodiazepines as potent PARP-1 inhibitors for treatment of ischemic injuries. Bioorg Med Chem. 2003;11:3695–707. doi: 10.1016/s0968-0896(03)00333-x. [DOI] [PubMed] [Google Scholar]
- Ye M, Fu S, Pi R, He F. Neuropharmacological and pharmacokinetic properties of berberine: a review of recent research. J Pharm Pharmacol. 2009;61:831–7. doi: 10.1211/jpp/61.07.0001. [DOI] [PubMed] [Google Scholar]
- Rami A, Bechmann I, Stehle JH. Exploiting endogenous anti-apoptotic proteins for novel therapeutic strategies in cerebral ischemia. Prog Neurobiol. 2008;85:273–96. doi: 10.1016/j.pneurobio.2008.04.003. [DOI] [PubMed] [Google Scholar]
- Yu SS, Zhao J, Zheng WP, Zhao Y. Neuroprotective effect of 4-hydroxybenzyl alcohol against transient focal cerebral ischemia via anti-apoptosis in rats. Brain Res. 2010;1308:167–75. doi: 10.1016/j.brainres.2009.10.037. [DOI] [PubMed] [Google Scholar]
- Zhang B, Hata R, Zhu P, Sato K, Wen TC, Yang L, et al. Prevention of ischemic neuronal death by intravenous infusion of a ginseng saponin, ginsenoside Rb(1), that upregulates Bcl-xL expression. J Cereb Blood Flow Metab. 2006;26:708–21. doi: 10.1038/sj.jcbfm.9600225. [DOI] [PubMed] [Google Scholar]
- Teng H, Zhang ZG, Wang L, Zhang RL, Zhang L, Morris D, et al. Coupling of angiogenesis and neurogenesis in cultured endothelial cells and neural progenitor cells after stroke. J Cereb Blood Flow Metab. 2008;28:764–71. doi: 10.1038/sj.jcbfm.9600573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Jin K, Xie L, Childs J, Mao XO, Logvinova A, et al. VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. J Clin Invest. 2003;111:1843–51. doi: 10.1172/JCI17977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao RQ, Zhang L, Wang W, Li L. Cornel iridoid glycoside promotes neurogenesis and angiogenesis and improves neurological function after focal cerebral ischemia in rats. Brain Res Bull. 2009;79:69–76. doi: 10.1016/j.brainresbull.2008.12.010. [DOI] [PubMed] [Google Scholar]
- Heurteaux C, Gandin C, Borsotto M, Widmann C, Brau F, Lhuillier M, et al. Neuroprotective and neuroproliferative activities of NeuroAid (MLC601, MLC901), a Chinese medicine, in vitro and in vivo. Neuropharmacology. 2010;58:987–1001. doi: 10.1016/j.neuropharm.2010.01.001. [DOI] [PubMed] [Google Scholar]
- Thiels E, Klann E. Extracellular signal-regulated kinase, synaptic plasticity, and memory. Rev Neurosci. 2001;12:327–45. doi: 10.1515/revneuro.2001.12.4.327. [DOI] [PubMed] [Google Scholar]
- Maher P, Akaishi T, Abe K. Flavonoid fisetin promotes ERK-dependent long-term potentiation and enhances memory. Proc Natl Acad Sci USA. 2006;103:16568–73. doi: 10.1073/pnas.0607822103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baskys A, Hou AC. Vascular dementia: pharmacological treatment approaches and perspectives. Clin Interv Aging. 2007;2:327–35. [PMC free article] [PubMed] [Google Scholar]
- Chen DM, Xiao L, Cai X, Zeng R, Zhu XZ. Involvement of multitargets in paeoniflorin-induced preconditioning. J Pharmacol Exp Ther. 2006;319:165–80. doi: 10.1124/jpet.106.104380. [DOI] [PubMed] [Google Scholar]
- Wang Z, Du Q, Wang F, Liu Z, Li B, Wang A, et al. Microarray analysis of gene expression on herbal glycoside recipes improving deficient ability of spatial learning memory in ischemic mice. J Neurochem. 2004;88:1406–15. doi: 10.1046/j.1471-4159.2003.02258.x. [DOI] [PubMed] [Google Scholar]
- Jiang ZG, Lu XC, Nelson V, Yang X, Pan W, Chen RW, et al. A multifunctional cytoprotective agent that reduces neurodegeneration after ischemia. Proc Natl Acad Sci USA. 2006;103:1581–6. doi: 10.1073/pnas.0510573103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youdim MB, Buccafusco JJ. Multi-functional drugs for various CNS targets in the treatment of neurodegenerative disorders. Trends Pharmacol Sci. 2005;26:27–35. doi: 10.1016/j.tips.2004.11.007. [DOI] [PubMed] [Google Scholar]
- Mantz J, Degos V, Laigle C. Recent advances in pharmacologic neuroprotection. Eur J Anaesthesiol. 2010;27:6–10. doi: 10.1097/EJA.0b013e32832fa606. [DOI] [PubMed] [Google Scholar]
- Cao C, He X, Wang W, Zhang L, Lin H, Du L. Kinetic distribution of paeoniflorin in cortex of normal and cerebral ischemia-reperfusion rats after intravenous administration of Paeoniae Radix extract. Biomed Chromatogr. 2006;20:1283–8. doi: 10.1002/bmc.658. [DOI] [PubMed] [Google Scholar]
- Wang C, Zhang D, Ma H, Liu J. Neuroprotective effects of emodin-8-O-beta-D-glucoside in vivo and in vitro. Eur J Pharmacol. 2007;577:58–63. doi: 10.1016/j.ejphar.2007.08.033. [DOI] [PubMed] [Google Scholar]
- Tarragó T, Kichik N, Claasen B, Prades R, Teixidó M, Giralt E. Baicalin, a prodrug able to reach the CNS, is a prolyl oligopeptidase inhibitor. Bioorg Med Chem. 2008;16:7516–24. doi: 10.1016/j.bmc.2008.04.067. [DOI] [PubMed] [Google Scholar]
- Youdim KA, Dobbie MS, Kuhnle G, Proteggente AR, Abbott NJ, Rice-Evans C. Interaction between flavonoids and the blood-brain barrier: in vitro studies. J Neurochem. 2003;85:180–92. doi: 10.1046/j.1471-4159.2003.01652.x. [DOI] [PubMed] [Google Scholar]