Abstract
Aim:
To investigate the immunosuppressive effects of 1,25-dihydroxyvitamin D3 (1,25-(OH)2VD3) on concanavalin A (ConA)-induced hepatitis and elucidate the action mechanism.
Methods:
Female BALB/C mice were intravenously administered ConA (20 mg/kg) to induce acute immunological liver injury. Liver damage was evaluated in respect to serum alanine transaminase (ALT) level and liver histological changes. The proliferation of splenocytes was measured by using [3H]-thymidine incorporation. The cytokine level in the cultured splenocyte supernatant was determined by using enzyme-linked immunosorbent assays (ELISAs). The percentage of different splenic T cell subtypes was analyzed by using flow cytometry. The expression of splenic vitamin D receptor (VDR) mRNA and protein was detected by using real-time qRT-PCR and Western blot, respectively.
Results:
1,25-(OH)2VD3 (2.5 μg/kg, ip) significantly decreased the serum ALT levels and markedly attenuated the histological liver damage. The beneficial effect of 1,25-(OH)2VD3 was associated with: (i) inhibition of CD4+ T cell activation; (ii) reduction of interferon-γ (IFN-γ) and elevation of both IL-4 and IL-5 in supernatants of cultured splenocytes; and (iii) elimination of activated T cells by increasing VDR mRNA and protein expression in the spleen.
Conclusion:
1,25-(OH)2VD3 had a significant protective effect against ConA-induced hepatitis, and its mechanism of action was associated with down-regulation of T cell-mediated immunity and up-regulation of VDR gene expression.
Keywords: 1,25-dihydroxyvitamin D3; concanavalin A; hepatitis; vitamin D receptor; interferon-γ; interleukin 4; interleukin 5
Introduction
Autoimmune liver disease includes autoimmune hepatitis (AIH), a distinct form of acute and chronic inflammatory liver disease in which immune reactions against host antigens have been found to be the major pathological mechanism1. AIH may lead to severe liver disease such as liver cirrhosis. The frequency of AIH among patients with chronic liver disease in North America is between 11% and 23%2. With improvements in the clinical detection technology, the incidence of autoimmune liver disease has gradually increased in recent years. The major treatment option is immunosuppressive therapy, which is proven to be effective3.
In the pathogenesis of AIH, genetic factors predispose individuals to the development of the disease4. Moreover, the environment contributes to development, and there is reason to believe that vitamin D may be an environmental factor that affects AIH. A previous study demonstrated that calcium and vitamin D supplementation is beneficial to patients with AIH5. Vitamin D inadequacy is common in noncholestatic chronic liver disease such as AIH and correlates with disease severity6. Compared with healthy people, patients with AIH display lower serum levels of vitamin D7.
1,25-(OH)2VD3 is one of the metabolites of vitamin D, and among all vitamin D metabolites, it has the strongest biological activity in vivo with the bio-active domain of non-protein binding free form8. In addition to regulating calcium and phosphorus metabolism, 1,25-(OH)2VD3 was also recently found to have immunosuppressive effects. Its biological effects are primarily mediated by vitamin D receptor (VDR)9. 1,25-(OH)2VD3 selectively inhibits Th1 cell proliferation and function10 by inhibiting interleukin (IL)-2 and interferon (IFN)-γ generation in Th1 cells11, 12] as well as IL-12 production in mononuclear cells13, thereby inhibiting the occurrence and progress of autoimmune diseases14 and organ transplant rejection15, which are caused by excessive activation of T cell-mediated immunity. The immunosuppressive effects of 1,25-(OH)2VD3 have been demonstrated in many animal models such as experimental autoimmune encephalomyelitis16, autoimmune diabetes17, rheumatoid arthritis18, systemic red spot lupus19, inflammatory bowel disease20 and allogeneic transplantation15; however, its role in AIH has not previously been reported.
ConA-induced hepatitis is a commonly used mouse model of T cell immune-mediated liver injury that resembles viral and autoimmune hepatitis in humans21. In the present study, we investigated whether 1,25-(OH)2VD3 could alleviate ConA-induced liver injury of mice in vivo, and we also investigated its immunosuppressive mechanisms of action.
Materials and methods
Mice
Female BALB/c mice (6–8 weeks old, 20–22 g) were purchased from Shanghai Experimental Animal Center of Chinese Academy of Sciences. The animals were housed in specific pathogen-free conditions. All mice were allowed to acclimatize in our facility for 1 week before any experiments were started. All experiments were carried out according to the National Institutes of Health Guide for Care and Use of Laboratory Animals and were approved by the Bioethics Committee of the Shanghai University of Traditional Chinese Medicine.
Induction of ConA-induced hepatitis and drug administration
ConA (Sigma, St Louis, USA) was dissolved in pathogen-free saline and filtered through 0.45 μm filter (Millipore). Mouse hepatitis was induced by injection of the indicated doses of ConA (in 0.2 mL saline) into the tail vein. A stock solution of 1,25-(OH)2VD3 (Sigma, St Louis, USA) was prepared in ethanol and diluted in pathogen-free saline to 0.25 μg/mL. 1,25-(OH)2VD3 was administered intraperitoneally (ip in 0.2 mL solvent) once daily on d -7, -6, -5, -4, -3, -2, -1, and 1 h before ConA injection on d 0.
Liver transaminase activity
Serum from individual mice was obtained 24 h after ConA injection. Alanine transaminase (ALT) activity was measured by the standard photometric method using a commercial kit (Jiancheng, Nanjing, China).
Histological examination
Livers were fixed in 10% formalin, embedded in paraffin, cut into 5 μmol/L sections, deparaffinized in xylene and serially dehydrated in decreasing concentrations of ethanol. Sections were stained with hematoxylin-eosin and examined under light microscopy to evaluate tissue structure and pathological grade. Necrosis was analyzed with a five-point score: 0, none; 1, single cell necrosis; 2, 1%–20% necrosis; 3, 20%–40% necrosis; and 4, >40% necrosis (n= 5 mice/group).
Splenocyte preparation
Mice were killed, and their spleens were removed aseptically. A single spleen cell suspension was prepared, and cell debris and clumps were removed. Erythrocytes were lysed with Tris-buffered ammonium chloride. Mononuclear cells were washed and re-suspended in RPMI-1640 medium supplemented with 10% heat-inactivated fetal bovine serum (FBS), penicillin (100 U/mL) and streptomycin (100 mg/mL).
Flow cytometric analysis
Splenocytes were harvested 24 h after ConA injection. To analyze the splenic T cell subtype percentage, splenocytes were stained with FITC conjugated anti-mouse CD4 and PE conjugated anti-mouse CD8 mAb (eBioscience) and analyzed on a fluorescence activated cell sort (FACS) Aria (Becton Dickinson, CA).
Proliferation assay
Splenocytes (5×105 cells/well) were cultured in triplicate with medium alone or stimulated with ConA (5 μg/mL) for 24 h in 96-well plates in a 37 °C incubator with 5% CO2. Cells were pulsed with 0.25 μCi of [3H]-thymidine for 8 h and harvested onto glass fiber filters. The incorporated radioactivity was then counted using a Beta Scintillation Counter (MicroBeta Trilux, PerkinElmer Life Sciences, Boston, MA).
Cytokine measurement
Splenocytes (5×105 cells/well) were cultured in triplicate in 96-well flat-bottom plates (Corning) in the presence of 5 μg/mL ConA. After 24 h, culture supernatants were harvested and stored at −20 °C. The concentrations of IFN-γ, IL-4, and IL-5 were determined by enzyme-linked immunosorbent assays (ELISAs) (BD PharMingen, San Diego, CA, USA).
Real-time quantitative reverse transcription polymerase chain reaction (real-time qRT-PCR)
Splenocytes (5×106 cells/well) were stimulated with 5 μg/mL ConA in 12-well plates for 24 h and lysed using Trizol reagent (GIBCO). Total RNA was isolated from each cell preparation and reverse transcribed into cDNA. Relative quantitation with real-time PCR was performed with SYBR Green PCR Reagents (Qiagen, Valencia, CA, USA) and a Continuous Fluorescence Detection System (MJ Research, USA) according to the manufacturer's instructions. The mRNA levels were normalized to those of GAPDH.
Gene-specific primers were as follows: VDR: (sense) 5′-AACGCTATGACCTGTGAAGGC-3′, (anti-sense) 5′-CCTGTACTTACGTCTGCACGA-3′. GAPDH: (sense) 5′-AACTTTGGCATTGTGGAAGG-3′, (anti-sense) 5′-GGATGCAGGGATGATGTTCT-3′.
The cDNA was amplified by 45 cycles of denaturation at 95 °C for 5 s, annealing and extension at 60 °C for 20 s and then melting at 65 °C–95 °C.
Western blot
Splenocytes (5×106 cells/well) were stimulated with 5 μg/mL ConA in 12-well plates for 24 h. Stimulated cells were lysed in lysis buffer (25 mmol/L Tris buffer, pH 7.4, containing 150 mmol/L NaCl, 1% Nonidet P-40, 1 mmol/L EDTA, 10 mmol/L NaF, 1 mmol/L dithiothreitol, 50 μg/mL each of leupeptin, aprotinin and phenylmethylsulphonylfluoride) by incubation on ice for 30 min. Lysates were then centrifuged at 13 000×g at 4 °C for 10 min, and the supernatants were transferred to fresh tubes and stored at −80 °C until required. Proteins (50 μg per lane) were electrophoresed on a 10% SDS-PAGE gel and transferred to nitrocellulose membranes (Schleicher & Schuell). The membranes were incubated with 5% non-fat milk for 1 h to block non-specific binding and then rinsed and incubated with a panel of rabbit polyclonal antibodies against VDR (Santa Cruz Biotechnology, Inc) overnight at 4 °C. The membranes were then incubated with a 1:2000 dilution of HRP-conjugated anti-rabbit IgG (H+L) for 1 h. Immune complexes were detected with a chemiluminescence substrate (Pierce) and exposed to Kodak X-ray film (Kodak).
Statistical analysis
Data are expressed as mean±SD of indicated experiments. The Student's t-test was used to determine variances between groups where appropriate. A P value <0.05 was considered significant.
Results
1, 25-(OH)2VD3 effectively attenuated ConA-induced liver damage
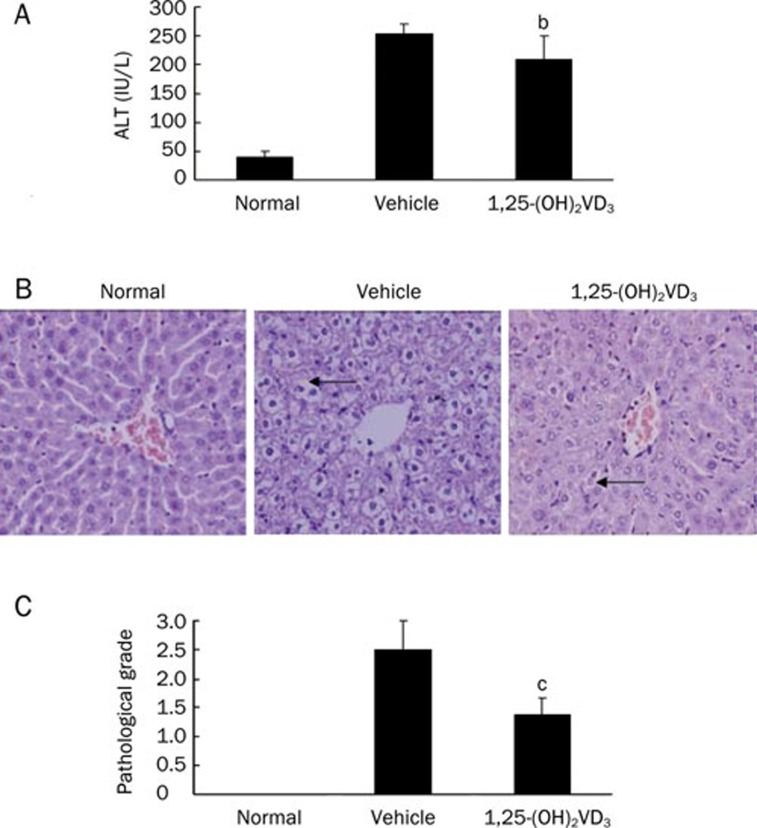
To examine the effects of 1,25-(OH)2VD3 on ConA-induced mouse hepatitis, which engenders a T cell-dependent acute liver-specific injury that closely resembles human autoimmune hepatitis, 1,25-(OH)2VD3 (2.5 μg/kg) was intraperitoneally administered to mice. Serum ALT levels were measured 24 h after ConA injection in both the 1,25-(OH)2VD3 and vehicle-treated groups. As shown in Figure 1A, in the 1,25-(OH)2VD3-treated group, significantly lower serum ALT levels were detected.
Figure 1.
Effects of 1,25-(OH)2VD3 on ConA-induced hepatitis. (A) Effect of 1,25-(OH)2VD3 on ALT level; (B) Histological examination. H&E staining (×200). Necrotic liver injury was indicated by black arrows. (C) Pathological grade. Data were from a representative experiment. Three separate experiments were performed with similar results. n=5 mice/group. Mean±SD. bP<0.05, cP<0.01 vs vehicle-treated mice.
Liver histological studies were also used to determine the effect of 1,25-(OH)2VD3 on ConA-induced liver injury (Figure 1B). Light microscopy examination showed massive necrosis in the livers from ConA-induced hepatitis mice 24 h after ConA injection, whereas mice pretreated with 1,25-(OH)2VD3 displayed only minor liver damage. The pathological grades also showed the significant preventative effects of 1,25-(OH)2VD3 on necrosis (Figure 1C).
1,25-(OH)2VD3 effectively reduced the ratio of CD4+/CD8+ T cells in the spleen
To test whether the inhibitory effect of 1,25-(OH)2VD3 was associated with the variation in percentages of T cell subtypes the spleens, FACS analysis was performed. Compared with vehicle control mice, treatment with 1,25-(OH)2VD3 significantly reduced the percentage of CD4+ T cells and increased the percentage of CD8+ T cells. As a result, 1,25-(OH)2VD3 effectively reduced the ratio of CD4+/CD8+ T cells in the spleen (Table 1).
Table 1. Effect of 1,25-(OH)2VD3 on splenic T-cell subtype percentage. Data were from a representative experiment. Three independent experiments were performed with similar results.
| Normal | Vehicle | 1,25-(OH)2VD3 | |
|---|---|---|---|
| CD4+ T | 20.1% | 26.9% | 22.2% |
| CD8+ T | 10.2% | 5.9% | 8.2% |
| CD4+/CD8+ ratio | 1.97 | 4.56 | 2.71 |
1,25-(OH)2VD3 significantly impaired T lymphocyte proliferation
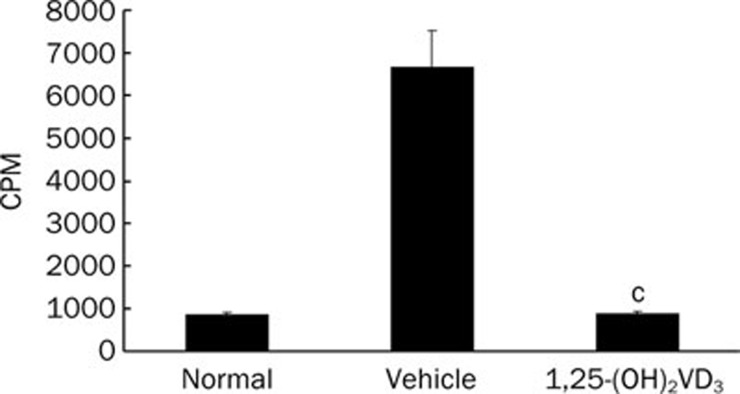
The proliferation response of lymphocytes after in vivo 1,25-(OH)2VD3 treatment was further investigated. When stimulated with ConA ex vivo, the proliferation of splenocytes from 1,25-(OH)2VD3-treated mice was dramatically inhibited compared with splenocytes from vehicle control mice (Figure 2).
Figure 2.
1,25-(OH)2VD3 inhibited ConA-induced splenocytes proliferation. Data were from a representative experiment made in triplicate and expressed as mean±SD. Three independent experiments were performed with similar results. n=5 mice/group. cP<0.01 vs vehicle-treated mice.
1,25-(OH)2VD3 effectively reduced interferon-γ (IFN-γ) and increased IL-4 and IL-5 in culture supernatants of splenocytes
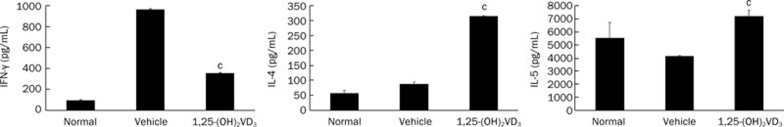
Immune responses are greatly influenced by the balance between Th1- and Th2-type cytokines. Th1 cytokines, including IFN-γ and IL-2, generally promote Th1 cell-mediated immune responses. By contrast, Th2 cytokines, which include IL-4 and IL-5, are generally anti-inflammatory and are involved in Th2 cell-mediated humoral immune responses22, 23. To examine the immunosuppressive effect of 1,25-(OH)2VD3, the cytokine production of the splenocytes was assayed. Splenocytes separated from 1,25-(OH)2VD3-treated and vehicle-treated mice were activated with ConA. After 24 h in culture, the culture supernatant of the splenocytes was harvested, and the levels of IFN-γ, IL-4, and IL-5 were detected by ELISA. Our data show that IFN-γ was markedly reduced in 1,25-(OH)2VD3-treated mice compared with vehicle-treated mice, and the levels of IL-4 and IL-5 were significantly increased in 1,25-(OH)2VD3-treated mice compared with vehicle-treated mice (Figure 3).
Figure 3.
1,25-(OH)2VD3 reduced IFN-γ and increased IL-4 and IL-5 levels in the supernatant of cultured splenocytes. Data of cytokines production were from a representative experiment made in duplicate and expressed as mean±SD. Three independent experiments were performed with similar results. n=5 mice/group. cP<0.01 vs vehicle-treated mice.
1,25-(OH)2VD3 significantly increased the expression of vitamin D receptor mRNA and protein in spleens
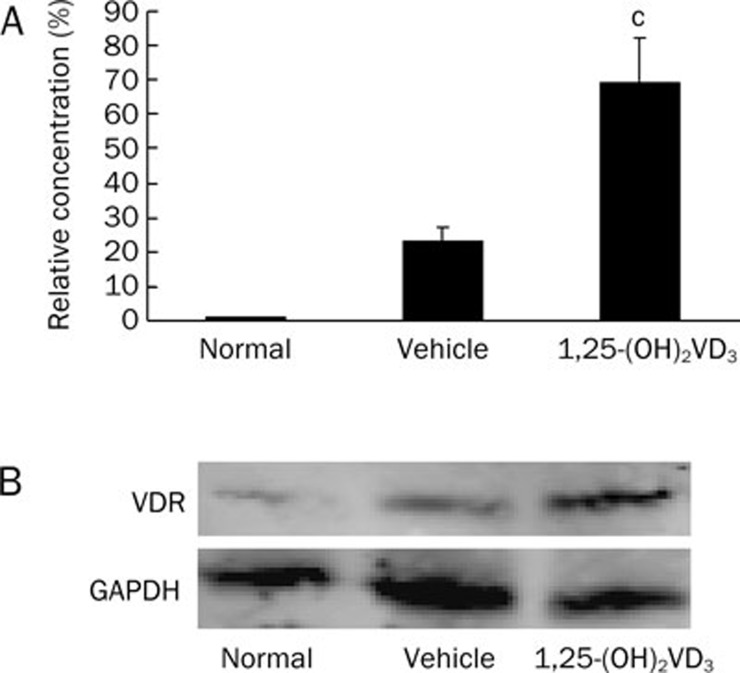
To determine whether the immunosuppressive effect of 1,25-(OH)2VD3 was related to vitamin D receptor (VDR) expression, the effect of 1,25-(OH)2VD3 on VDR mRNA and VDR protein production in splenocytes were examined. Splenocytes separated from 1,25-(OH)2VD3-treated and vehicle-treated mice were activated with ConA (5 μg/mL). After 24 h in culture, total RNA and total protein were extracted, and the VDR mRNA and protein expression were detected by real-time quantitative reverse transcription polymerase chain reaction (real-time qRT-PCR) and Western blot assays, respectively. Our results show that expression of VDR mRNA (Figure 4A) and VDR protein (Figure 4B) in splenocytes was markedly increased in 1,25-(OH)2VD3-treated mice compared with vehicle-treated mice.
Figure 4.
1,25-(OH)2VD3 increased VDR mRNA and protein levels in spleen. (A) The VDR mRNA expression was detected by real-time qRT-PCR assay; (B) The VDR protein production detected by Western blot. Data were from a representative experiment. Three independent experiments were performed with similar results. n=5 mice/group. Mean±SD. cP<0.01 vs vehicle-treated mice.
Discussion
In the present study, we report that 1,25-(OH)2VD3 protected mice from immune-mediated liver injury in ConA-induced hepatitis (CIH), and the mechanism of action of 1,25-(OH)2VD3 was shown to be closely associated with its immunosuppressive effect on T cell-mediated immunity and its role in promoting VDR gene expression.
ConA-induced mouse hepatitis is due to T cell-mediated liver damage. Intravenous injection of ConA in mice can selectively cause liver injury; T helper (Th) cells and macrophages are the major effector cells in this pathological process24. A large number of splenic T lymphocytes are activated by ConA and produce cytokines; activated T cells and cytokines can damage liver cells directly or injure liver cells indirectly through activating macrophages. When mice are given 20 mg/kg ConA, the liver is almost entirely edematous and necrotic 24 h later. 1,25-(OH)2VD3 can significantly reduce the extent of liver necrosis, indicating its protective effect in immunological liver injury.
In the ConA-induced mouse hepatitis model, the activation of CD4+ T cell is closely related to liver injury. The research of Tiegs et al has shown that pretreatment with an antibody against T lymphocytes can fully protect mice against ConA-induced liver injury similar to a monoclonal anti-mouse CD4 antibody. However, monoclonal anti-mouse CD8 antibodies failed to protect against damage21. Our study demonstrated that 1,25-(OH)2VD3 significantly reduced T cell proliferation in vitro and significantly reduced the generation of CD4+ T cells, thereby reducing the CD4+/CD8+ ratio. This indicates that 1,25-(OH)2VD3 can prevent mice from ConA-induced hepatitis by inhibiting CD4+ T cell activation.
CD4+ T cells are functionally classified into functional Th1 cells, Th2 cells and others (undifferentiated, nonfunctional T cells). Th1 cells induce T cell-mediated immune responses25. Th1-type cytokines and pro-inflammatory cytokines play a major role in ConA-induced liver injury26, and IFN-γ and TNF-α are the most important cytokines that induce liver injury27, 28. Th2 cells inhibit the generation and effector functions of Th1 cells via the release of cytokines such as IL-4 and IL-5, and consequently, both the reduction in Th1 and an increase in Th2 cell activity are associated with resolution of inflammation in autoimmune diseases29. 1,25-(OH)2VD3 significantly reduced IFN-γ expression and increased IL-4 and IL-5 production, indicating that it can inhibit T cell-mediated immunity in ConA-induced mouse hepatitis by inhibiting Th1-cell function and enhancing Th2-cell function.
Vitamin D achieves its biological effects through the vitamin D receptor (VDR). Most cells in the immune system contain VDR, such as monocytes, tissue macrophages and activated T and B lymphocytes30. Of all the vitamin D metabolites, 1,25-(OH)2VD3 has the strongest biological activity in vivo, and it can directly inhibit T cell proliferation and IL-2 and IFN-γ secretion31. Our results show that 1,25-(OH)2VD3 significantly increased VDR mRNA and protein levels, and it also inhibited the production of IFN-γ and increased the generation of IL-4 and IL-5, increasing the proportion of CD8+ T cells and reducing the proportion of CD4+ T cells. These results indicate that 1,25-(OH)2VD3 can inhibit T cell-mediated immunity by increasing VDR gene expression.
In conclusion, 1,25-(OH)2VD3 has a strong inhibitory effect on T cell-mediated immunity. It may inhibit T cell immune responses partially through promoting VDR gene expression, thereby playing a role in the prevention and treatment of ConA-induced acute immunological liver injury. However, the inhibition mechanism of VDR gene expression against T cell activation needs to be further studied.
Author contribution
Xu-dong HU and Ping LIU designed research; Xu-dong HU and Shi-li JIANG performed research; Cheng LIU, Ming-yu SUN and Gao-feng CHEN contributed new analytical tools and reagents; Xu-dong HU, Cheng-hai LIU and Yi-yang HU analyzed data; Xu-dong HU wrote the paper.
Acknowledgments
This research work was supported by the Shanghai Postdoctoral Science Foundation (A class) (No 08R214146), Shanghai Leading Academic Discipline Project (Y0302), E-institute of Shanghai Municipal Education Commission (No E03008), Shanghai Municipal Education Commission Innovation Team (Phase I) and Shanghai Municipal Education Commission Key Subject (No J50301).
References
- Meyer zum Büschenfelde KH, Dienes HP. Autoimmune hepatitis. Definition-classification-histopathology-immunopathogenesis. Virchows Arch. 1996;429:1–12. doi: 10.1007/BF00196814. [DOI] [PubMed] [Google Scholar]
- Czaja AJ. Autoimmune hepatitis--approach to diagnosis. Med Gen Med. 2006;8:55. [PMC free article] [PubMed] [Google Scholar]
- Maggs J, Cullen S. Management of autoimmune liver disease. Minerva Gaastroenterol Dietol. 2009;55:173–206. [PubMed] [Google Scholar]
- Béland K, Lapierre P, Alvarez F. Influence of genes, sex, age and environment on the onset of autoimmune hepatitis. World J Gastroenterol. 2009;15:1025–34. doi: 10.3748/wjg.15.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn J, Flamm SL. Autoimmune hepatitis. Curr Treat Options Gastroenterol. 2005;8:481–92. doi: 10.1007/s11938-005-0035-7. [DOI] [PubMed] [Google Scholar]
- Fisher L, Fisher A. Vitamin D and parathyroid hormone in outpatients with noncholestatic chronic liver disease. Clin Gastroenterol Hepatol. 2007;5:513–20. doi: 10.1016/j.cgh.2006.10.015. [DOI] [PubMed] [Google Scholar]
- Saron ML, Godoy HT, Hessel G. Nutritional status of patients with biliary atresia and autoimmune hepatitis related to serum levels of vitamins A, D, and E. Arq Gastroenterol. 2009;46:62–8. doi: 10.1590/s0004-28032009000100016. [DOI] [PubMed] [Google Scholar]
- Van Hoof HJ, Sninkele LM, Ross HA, Sweep CG, Benraad TJ. Determination of on-protein-bound plasma 1,25-dihydroxyvitamin D by symmetric (rate) dialysis. Anal Biochem. 1998;258:176–83. doi: 10.1006/abio.1998.2586. [DOI] [PubMed] [Google Scholar]
- Deluca HF, Cantorna MT. Vitamin D: its role and uses in immunology. FASEB J. 2001;15:2579–85. doi: 10.1096/fj.01-0433rev. [DOI] [PubMed] [Google Scholar]
- Lemire JM. Immunomodulatory actions of 1,25-dihydroxyvitamin D3. J Steroid Biochem Mol Biol. 1995;53:599–602. doi: 10.1016/0960-0760(95)00106-a. [DOI] [PubMed] [Google Scholar]
- Lemire JM, Archer DC, Beck L, Spiegelberg HL. Immunosuppressive actions of 1,25-dihydroxyvitamin D3: Preferential inhibition of Th1 functions. J Nutr. 1995;125:1704S–8S. doi: 10.1093/jn/125.suppl_6.1704S. [DOI] [PubMed] [Google Scholar]
- Teodora P, Staeva V, Leonard PF. 1,25-Dihydroxyvitamin D3 inhibits IFN-γ and IL-4 levels during in vitro polarization of primary murine CD4 T cells. J Immunol. 2002;168:1181–9. doi: 10.4049/jimmunol.168.3.1181. [DOI] [PubMed] [Google Scholar]
- D'Ambrosio D, Cippitelli M, Cocciolo MG, Mazzeo D, Di Lucia P, Lang R, et al. Inhibition of IL-12 production by 1,25-dihydroxyvitamin D3. Involvement of NF-kappaB downregulation in transcriptional repression of the p40 gene. J Clin Invest. 1998;101:252–62. doi: 10.1172/JCI1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luciano A. Immunomodulatory effects of vitamin D receptor ligands in autoimmune diseases. Int Immunopharmacol. 2002;2:1017–28. doi: 10.1016/s1567-5769(02)00049-8. [DOI] [PubMed] [Google Scholar]
- Zhang AB, Zheng SS, Jia CK, Wang Y. Effect of 1,25-dihydroxyvitamin D3 on preventing allograft from acute rejection following rat orthotopic liver transplantation. World J Gastroenterol. 2003;9:1067–71. doi: 10.3748/wjg.v9.i5.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nashold FE, Miller DJ, Hayes CE. 1,25-dihydroxyvitamin D3 treatment decreases macrophage accumulation in the CNS of mice with experimental autoimmune encephalomyelitis. J Neuroimmunol. 2000;103:171–9. doi: 10.1016/s0165-5728(99)00247-7. [DOI] [PubMed] [Google Scholar]
- Zella JB, DeLuca HF. Vitamin D and autoimmune diabetes. J Cell Biochem. 2003;88:216–22. doi: 10.1002/jcb.10347. [DOI] [PubMed] [Google Scholar]
- Cantorna MT, Hayes CE, DeLuca HF. 1,25-Dihydroxycholecalciferol inhibits the progression of arthritis in murine models of human arthritis. J Nutr. 1998;128:68–72. doi: 10.1093/jn/128.1.68. [DOI] [PubMed] [Google Scholar]
- Lemire JM, Ince A, Takashima M. 1,25-dihydroxyvitamin D3 attenuates the expression of experimental murine lupus of MRL/1 mice. Autoimmunity. 1992;12:143–8. doi: 10.3109/08916939209150321. [DOI] [PubMed] [Google Scholar]
- Cantorna MT, Munsick C, Bemiss C, Mahon BD. 1,25-Dihydroxycholecalciferol prevents and ameliorates symptoms of experimental murine inflammatory bowel disease. J Nutr. 2000;130:2648–52. doi: 10.1093/jn/130.11.2648. [DOI] [PubMed] [Google Scholar]
- Tiegs G, Hentschel J, Wendel A. A T cell-dependent experimental liver injury in mice inducible by concanavalin A. J Clin Invest. 1992;90:196–203. doi: 10.1172/JCI115836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott MJ, Godshall CJ, Cheadle WG. Jaks, STATs, Cytokines, and Sepsis. Clin Diagn Lab Immunol. 2002;9:1153–9. doi: 10.1128/CDLI.9.6.1153-1159.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coffman RL, Weid T von der, Coffman RL, T von der Weid. Multiple pathways for the initiation of T helper 2 (Th2) responses. J Exp Med. 1997;185:373–5. doi: 10.1084/jem.185.3.373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gantner F, Leist M, Lohse AW, Germann PG, Tiegs G. Concanavalin A induced T-cell-mediated hepatic injury in mice: The role of tumor necrosis factor. Hepatology. 1995;21:190–8. doi: 10.1016/0270-9139(95)90428-x. [DOI] [PubMed] [Google Scholar]
- Tsuchiya T, Ohshima K, Karube K, Yamaguchi T, Suefuji H, Hamasaki M, et al. Th1, Th2, and activated T-cell marker and clinical prognosis in peripheral T-cell lymphoma, unspecified: comparison with AILD, ALCL, lymphoblastic lymphoma, and ATLL. Blood. 2004;103:236–41. doi: 10.1182/blood-2002-05-1352. [DOI] [PubMed] [Google Scholar]
- Kremer M, Hines IN, Milton RJ, Wheeler MD. Favored T helper 1 response in a mouse model of hepatosteatosis is associated with enhanced T cell-mediated hepatitis. Hepatology. 2006;44:216–27. doi: 10.1002/hep.21221. [DOI] [PubMed] [Google Scholar]
- Trautwein C, Rakemann T, Brenner DA, Streetz K, Licato L, Manns MP, et al. Concanavalin A induced liver cell damage: activation of intracellular pathways triggered by tumor necrosis factor in mice. Gastroenterology. 1998;114:1035–45. doi: 10.1016/s0016-5085(98)70324-5. [DOI] [PubMed] [Google Scholar]
- Mizuhara H, Uno M, Seki Y, Yamashita M, Yamaoka M, Ogawa T, et al. Critical involvement of interferon gamma in the pathogenesis of T-cell activation-associated hepatitis and regulatory mechanisms of' interleukin-6 for the manifestations of hepatitis H1. Hepatology. 1996;23:1608–15. doi: 10.1053/jhep.1996.v23.pm0008675184. [DOI] [PubMed] [Google Scholar]
- Dimitrova P, Skapenko A, Herrmann ML, Schleyerbach R, Kalden JR, Schulze-Koops H. Restriction of de novo pyrimidine biosynthesis inhibits Th1 cell activation and promotes Th2 cell differentiation. J Immunol. 2002;169:3392–9. doi: 10.4049/jimmunol.169.6.3392. [DOI] [PubMed] [Google Scholar]
- Norman AW. Minireview: vitamin D receptor: new assignments for an already busy receptor. Endocrinology. 2006;147:5542–8. doi: 10.1210/en.2006-0946. [DOI] [PubMed] [Google Scholar]
- Cippitelli M, Santoni A. Vitamin D3: a transcriptional modulator of the interferon -gamma gene. Eur J Immunol. 1998;28:3017–30. doi: 10.1002/(SICI)1521-4141(199810)28:10<3017::AID-IMMU3017>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]