The increasing aged population poses new challenges to healthcare systems. The elderly frequently suffers from severe infections and end-stage organ disease, which are associated with an age-dependent decline in immune reactivity1, 2. Immunosenescence, defined as the changes in the immune system associated with age, is gathering interest in the scientific and healthcare communities alike.
Immunosenescence is generally related with weaker immune responses, which produce a progressive deterioration in the ability to respond to new stimulants. One might expect the elderly to be less reactive against transplanted organs than younger patients, thus to show better transplant outcomes. It is reasonable to assume that older individuals would be more easily rendered tolerant of transplanted organs3. Paradoxically, experimental studies and clinical data of organ transplantation have shown that the graft is more rapidly rejected in the elderly than in the younger. Thus, the age-related muting of the immune responses does not improve tolerance induction and reduce rejection after cell or organ transplantation. The effects of aging on acute and chronic allograft rejection have been extensively studied4. Many researches showed that the effects of immunosenescence on the induction of tolerance is one of the major factors5.
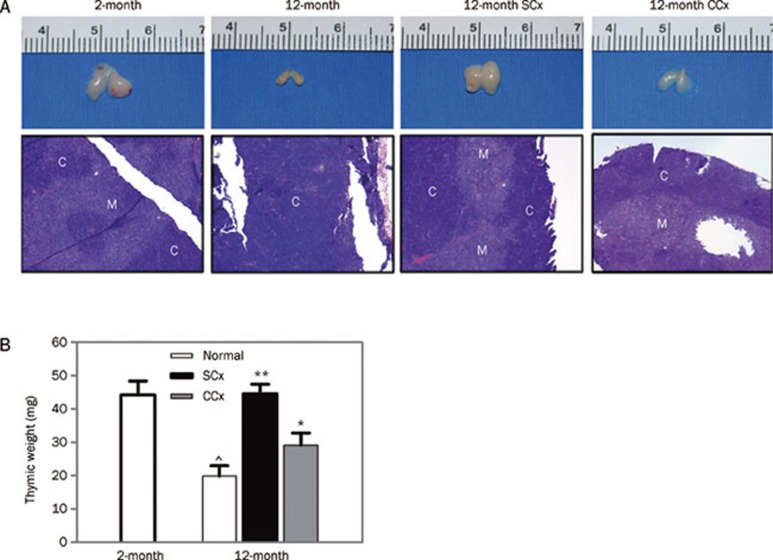
How does immunosenescence affect the transplantation tolerance? Experimental and clinical studies have shown that reversion or blockade of age-related atrophy of the thymus might be one of the promising therapeutic measures to reconstitute immune functions in the elderly. In mice, transplantation of aged thymuses into juvenile recipients led to reconstruction of the structure and function of the thymus6. Recently, Zhao and colleagues7 showed that surgical and chemical castration could restore the induction of transplant tolerance in aged mice. The authors found that old mice (age of 12 months) exhibited resistance to tolerance induction for cardiac transplants even after the anti-CD45RB monoclonal antibody therapy, in contrast to the robust tolerance induced in young mice (2 months). Surgical castration could lead to long-term graft acceptance and restoration of thymic cellularity. Furthermore, they found similar results from prostate cancer patients who received Lupron Depot injections, which could temporarily disrupt gonadal function. Both surgical and chemical castration could restore thymic architecture, increase thymic weight and numbers of developing regulating T cells (Treg) in aged mice (Figure 1). The tolerance-restorative effects of gonadectomy and hormonal suppression (with antiCD25 treatment) would not happen in aged mice with thymectomy or Treg depletion. These results indicated that surgical castration or Lupron Depot injections could restore the thymic function and promote transplant tolerance-induction, reverse the inhibition of transplantation tolerance by immunosenescence in aged mice.
Figure 1.
Surgical and chemical castration enhanced thymic mass and restored thymic architecture. (A) Thymic mass decreased with age. The mass increased after either surgical or chemical castration. (B) Histograms showing the thymic weight in different groups (*P<0.01; **P<0.001 versus 12-month normal; ⁁P<0.01 versus 2-month normal; n=3 to 5 animals per group). The increased thymic mass was associated with a restoration of the normal thymic architecture as shown in the lower row in (A): the recovery of corticomedullary differentiation that was disrupted in the noncastrated 12-month-old animal (H&E staining, ×100). C, cortex; M, medulla; SC, surgical castration; CC, chemical castration. Reprinted with permission from AAAS: Science Translational Medicine, copyright 2011.
In the aged, the thymus undergoes progressive involution, and the output of new cells significantly falls, which results in decreased thymic output of naïve T cells and decreased concentration of these cells in peripheral blood and lymph nodes8. Naïve T cells from aged mice exhibit reduced activation, differentiation and cytokine production following antigen presentation9. These age-associated changes in cell-mediated immunity strongly depend on thymic functions. Transplanting cultured thymic fragments to patients with DiGeorge syndrome who lack a functional thymus has been proved successful6 and may be an approach to restore naïve T lymphocyte numbers in the elderly. Zhao' study showed that surgical and chemical castration could promote the restoration of thymic architecture, increase the thymus weight and the absolute numbers of Treg (CD4+Foxp3+) thymocytes, and reduced the graft rejection. Their results suggest that endocrine modulation could represent a potential therapy to modify age-induced thymic involution.
Sex steroids may have a direct effect on immune function in the periphery by augmenting Treg and other peripheral T cell functions. However, increased levels of sex hormones may cause thymus involution10. Luteinizing hormone-releasing hormone (LHRH) synthesized in the hypothalamus is transported to the anterior pituitary, where it controls the synthesis and release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which govern the synthesis of gonadal steroids. Gonadal steroids have negative effects on the thymus and bone marrow, while LHRH itself is a positive immunomodulator. Using LHRH analogues or gonadectomy could reverse age-related thymic atrophy11. In Zhao's study, the results verified that Lupron-mediated gonadal suppression could restore the transplantation tolerance induction.
The mechanisms underlying immunosenescence is complex and not completely understood. Multiple factors, such as oxidative stress, gender differences, innate immunity, humoral and cellular immunity, thymus atrophy and persistent microbial infections, may contribute to immunosenescence5. Zhao' study has made an important progress in the treatment of immunosenescence, and established an effective therapeutic protocol for restoration of transplantation tolerance in aged mice, and afforded a potential approach to organs transplantation of the elders. Although it's hard to extrapolate a mouse's age to a human's, their results suggest that hormone modification may improve transplant acceptance in older patients by restoring exuberance in the immune system.
References
- Agarwal S, Busse PJ. Innate and adaptive immunosenescence. Ann Allergy Asthma Immunol. 2010;104:183–90. doi: 10.1016/j.anai.2009.11.009. [DOI] [PubMed] [Google Scholar]
- Burns EA, Leventhal EA. Aging, immunity, and cancer. Cancer Control. 2000;7:513–22. doi: 10.1177/107327480000700603. [DOI] [PubMed] [Google Scholar]
- Salama AD, Remuzzi G, Harmon WE, Sayegh MH. Challenges to achieving clinical transplantation tolerance. J Clin Invest. 2001;108:943–8. doi: 10.1172/JCI14142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins PN, Pratschke J, Pascher A, Fritsche L, Frei U, Neuhaus P, et al. Age and immune response in organ transplantation. Transplantation. 2005;79:127–32. doi: 10.1097/01.tp.0000146258.79425.04. [DOI] [PubMed] [Google Scholar]
- Ongrádi J, Kövesdi V. Factors that may impact on immunosenescence: an appraisal. Immun Ageing. 2010;7:7. doi: 10.1186/1742-4933-7-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markert ML, Boeck A, Hale LP, Kloster AL, McLaughlin TM, Batchvarova MN, et al. Transplantation of thymus tissue in complete DiGeorge syndrome. N Engl J Med. 1999;341:1180–9. doi: 10.1056/NEJM199910143411603. [DOI] [PubMed] [Google Scholar]
- Zhao G, Moore DJ, Kim JI, Lee KM, O'Connor MR, Duff PE, et al. Inhibition of transplantation tolerance by immune senescence is reversed by endocrine modulation. Sci Transl Med. 2011;3:87ra52. doi: 10.1126/scitranslmed.3002270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruver AL, Hudson LL, Sempowski GD. Immunosenescence of ageing. J Pathol. 2007;211:144–56. doi: 10.1002/path.2104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haynes L, Eaton SM, Burns EM, Randall TD, Swain SL. Newly generated CD4 T cells in aged animals do not exhibit age-related defects in response to antigen. J Exp Med. 2005;201:845–51. doi: 10.1084/jem.20041933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hince M, Sakkal S, Vlahos K, Dudakov J, Boyd R, Chidgey A. The role of sex steroids and gonadectomy in the control of thymic involution. Cell Immunol. 2008;252:122–38. doi: 10.1016/j.cellimm.2007.10.007. [DOI] [PubMed] [Google Scholar]
- Sutherland JS, Goldberg GL, Hammett MV, Uldrich AP, Berzins SP, Heng TS, et al. Activation of thymic regeneration in mice and humans following androgen blockade. J Immunol. 2005;175:2741–53. doi: 10.4049/jimmunol.175.4.2741. [DOI] [PubMed] [Google Scholar]