Abstract
Background
Stimulation of the nervous system plays a central role in brain development and neurodevelopmental outcome. Thalamocortical and corticocortical development is diminished in premature infants and correlated to electroencephalography (EEG) progression. The purpose of this study was to determine the effects of orocutaneous stimulation on the modulation of spectral edge frequency, fc=90% (SEF-90) derived from EEG recordings in preterm infants.
Methods
Twenty two preterm infants were randomized to experimental and control conditions. Pulsed orocutaneous stimulation was presented during gavage feedings begun at around 32 weeks postmenstrual age (PMA). The SEF-90 was derived from 2-channel EEG recordings.
Results
Compared to the control condition, the pulsed orocutaneous stimulation produced a significant reorganization of SEF-90 in the left (p = 0.005) and right (p < 0.0001) hemispheres. Notably, the left and right hemisphere showed a reversal in the polarity of frequency shift, demonstrating hemispheric asymmetry in the frequency domain. Pulsed orocutaneous stimulation also produced a significant pattern of short term cortical adaptation and a long term neural adaptation manifest as a 0.5 Hz elevation in SEF-90 after repeated stimulation sessions.
Conclusion
This is the first study to demonstrate the modulating effects of a servo-controlled oral somatosensory input on the spectral features of EEG activity in preterm infants.
INTRODUCTION
The astonishing capabilities of the human brain arise from intricate connectivity patterns among its billions of interacting brain cells. The specific patterns of connectivity are forged by the unique interaction between a person's genes and the local environment (1). Injury or deprivation of environmental input occurring at specific stages of postnatal life can dramatically reshape brain circuit development. For the premature infant, the perinatal insults associated with premature birth and extrauterine environment significantly disrupts normal thalamocortical and corticocortical development (2). Extremely premature infants endure an extensive course of respiratory support and frequent adverse stimulation during their neonatal intensive care unit (NICU) stay. The invasiveness of intubation, oxygen supplementation procedures, and prolonged nasogastric feeding tube placement disrupt the preterm baby precious sensory and motor experiences during a critical period of brain development, leading to maladaptive behaviors and abnormal development. Routine caregiving procedures (e.g. manipulation, diaper changes, endotracheal tube suctioning and repositioning, etc.) in critically ill preterm infants are associated with major circulatory fluctuations in cerebral hemodynamics that are underdetected by current bedside monitoring (3). However, introducing a developmentally appropriate NICU environment and stimulation has the potential to protect normal brain development (4, 5). Collectively, the effects of prematurity on neural development underscore the need for bedside brain monitoring tools (e.g. EEG, near infrared spectroscopy) to assess neurologic status and better understand the effects of stimulus modalities and caregiver manipulations on brain activity (6).
Defining normality of electrocortical activity represents a significant challenge in the NICU (7). Reduced-montage 2-channel EEG is becoming a common technique to monitor and map brain maturation and assess neurological status in preterm infants. Amplitude-integrated electroencephalography (aEEG) reflects integrated brain activity and allows for time-compressed, continuous electrocortical monitoring at bedside. Two pairs of surface electrodes (C3-P3 and C4-P4) are placed on the scalp over the centroparietal cortices, proximal to forelimb and orofacial sensorimotor cortical representations for aEEG recording. The aEEG provides many electrocortical features, including type of background activity (discontinuous/continuous), interburst intervals or burst rate, cyclic variation in the background activity corresponding to sleep-wake cycling, upper and lower amplitude margins, amplitude bandwidth, spectral composition, and the presence of seizure patterns (8).
The spectral signature of brain activity, including exogenous and autogenic frequency modulation (e.g. neonate state changes, external stimulation) and rhythmic electrocortical activity (resting state network dynamics), provides the clinician and neuroscientist with a window into the integrity and maturation of the human nervous system in the frequency domain. Spectral edge frequency fc=90% (SEF-90), defined as the frequency below which 90% of the power in a spectrum resides, is thought to reflect cerebral maturation. Application of the SEF measure in healthy newborns revealed a significant positive correlation with gestational age (GA) and varied with behavioral state and brain loci (9). Between approximately 30 to 40 weeks GA, whole brain SEF values increased from 5.4 to 12 Hz. Automated spectral power analyses showed that brain maturation is indicated by a significant shift from the lower to the higher EEG frequencies with advancing PMA and that gyral development in the parietotemporal cortex precedes that of other areas (10, 11). The evolution of preterm brain maturity as seen by EEG correlates with progression of gyration (12, 13).
To date, most studies using processed forms of EEG to examine preterm brain cortical activity have been aimed at mapping developmental features of maturation and/or pathologic brain activity. An additional consideration in critical care of the preterm infant is the local environment in the NICU, including the role of uni- or multimodal stimulation in brain development and neurodevelopmental outcome. The aim of this study is to determine the effects of highly controlled (pneumatic servo) pulsed orocutaneous stimulation presented during gavage feedings begun at around 32 weeks PMA on the modulation of cerebral activity in the frequency domain among medically stable preterm infants.
RESULTS
SEF
The presence of the pulsed pneumatic orocutaneous stimulation produced a visible reorganization of aEEG signal margins as shown in the time domain for the two examples in Figure 1. During somatosensory stimulation, a shift in lower margins is apparent for each infant sampled at 32 weeks PMA. Fourier analysis revealed that the pulsed orocutaneous stimulation produced a significant reorganization of SEF-90 when compared to the blind pacifier condition in the left (p=0.005) and right (p<0.0001) hemispheres (Tables 1, 2).
Figure 1.

Examples of EEG/aEEG using the C3-P3 and C4-P4 electrode montage on two preterm infants recorded at 32 weeks PMA. 3-minute orocutaneous stimulation epochs are indexed by vertical cursors in the aEEG record (upper panel:13,15 and 17; lower panel:7, 8 and 9). Note the presence of aEEG modulation during pulsed orocutaneous stimulation. Wake-sleep cycles preceding orocutaneous stimulation periods are present.
Table 1.
Mixed model adjusted means
| Group | Exp-ON | Exp-OFF | Cont-ON | Cont-OFF | Group Effect | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Periods | P3 P5 P7 | P1 P2 P4 P6 P8 P9 | P3 P5 P7 | P1 P2 P4 P6 P8 P9 | |||||||
| mean | SE | mean | SE | mean | SE | mean | SE | F | df | p | |
| SEF (90) Left | 10.79 | 0.28 | 10.99 | 0.28 | 11.16 | 0.29 | 11.08 | 0.28 | 5.21 | 3,30 | 0.005 |
| SEF (90) Right | 11.38 | 0.20 | 11.27 | 0.19 | 10.87 | 0.20 | 10.77 | 0.19 | 21.52 | 3,30 | 0.000 |
SEF(90): Spectral Edge Frequency, fc=90%. Exp-ON: Experimental pacifier with pneumatic pulse stimulation. Exp-OFF: Experimental pacifier is removed from the infant's mouth. Cont-ON: Controls blind pacifier stimulation. Cont-OFF: Controls blind pacifier is removed from the infant's mouth. P1 through P9 represent nine sequential periods. SE: Standard Error of Mean.
Table 2.
Post-hoc pairwise comparison
| Variable | Bonferroni-adjusted p | |||||
|---|---|---|---|---|---|---|
| G1 - G2 | G1 - G3 | G1 - G4 | G2 - G3 | G2 - G4 | G3 - G4 | |
| SEF (90) Left | 0.062 | 0.007 | 0.014 | 0.388 | 1.000 | 1.000 |
| SEF (90) Right | 0.947 | 0.000 | 0.000 | 0.001 | 0.000 | 1.000 |
SEF (90): Spectral Edge Frequency, fc=90%. G1: Exp-ON (experimental pacifier with pneumatic pulse stimulation); G2: Exp-OFF (experimental pacifier is removed from the infant's mouth); G3: Cont-ON (Controls blind pacifier stimulation); G4: Cont-OFF (Controls blind pacifier is removed from the infant's mouth).
Asymmetry
Based on the mixed model for repeated measures, the adjusted means for SEF-90 showed a reversal in the polarity of frequency shift between the left and right hemispheres (Table 1). The adjusted means for the SEF-90 in the left hemisphere were 10.79 and 11.16 Hz for the patterned pneumatic orocutaneous and blind pacifier conditions, respectively. The adjusted means for the SEF-90 in the right hemisphere were 11.38 and 10.87 Hz for the patterned pneumatic orocutaneous and blind pacifier conditions, respectively. This demonstrates an intriguing hemispheric asymmetry in EEG frequency domain.
Cortical Adaptation
A significant pattern of cortical adaptation in the SEF measure was apparent in the present cohort for both the blind and pulsed orocutaneous pacifier stimulation conditions, and a significant difference was apparent between the two conditions as well (Figure 2). The response to stimulation on SEF in P5 and P7 were progressively attenuated compared to P3 in both groups. The 5.5 minute periods of ‘no-stimulation’ in the experimental infants showed a significant after-effect in the SEF-90, especially following the first and second stimulation epochs. The SEF-90 after-effect was not apparent to the same degree in the control infants. SEF was elevated overall by approximately 0.5 Hz in infants in the experimental group, compared to control infants, who did not receive the pulsatile stimulation. Comparing mean values of SEF-90 in the first stimulation session (Figure 3) showed that the SEF-90 baseline (periods 1 and 2) was not significantly different between sham and pulsed infant groups (p= 0.898). Moreover, the potent effects of the pulsatile orocutaneous stimulus are evident in the divergence of sham and pulsed pacifier stimulation functions during the infant's first recording session.
Figure 2.
Interval plot of SEF-90 for cross-head (C3-P3 and C4-P4) montage for blind ‘sham’ pacifier (circle symbols) versus the ‘pulsed’ pacifier (triangle symbols). Data are included from all stimulation sessions of all infants. Filled symbols represent stimulation conditions. Open symbols indicate periods when neither sham nor pulsed pacifier was in the baby's mouth. Contiguous EEG periods (data blocks) labeled 1 through 9 (periods). All periods are 3 minutes except for periods 4 and 6 which are 5.5 minutes in duration.
Figure 3.
Plot of the SEF-90 for cross-head (C3-P3 and C4-P4) montage for blind ‘sham’ pacifier (circle symbols) versus the ‘pulsed’ pacifier (triangle symbols) for the first stimulation session only. Filled symbols represent stimulation conditions. Open symbols indicate periods when neither sham nor pulsed pacifier was in the baby's mouth. Contiguous EEG periods (data blocks) labeled 1 through 9 (periods). All periods are 3 minutes except for periods 4 and 6 which are 5.5 minutes in duration.
DISCUSSION
Cerebral and subcortical maturation and activity-dependent changes in the newborn are complex processes, and are reflected in EEG measures (14). The 24–32 week gestational period is characterized by the prominence of the transient cortical subplate, which expands to approximately five times the size and thickness of the cortical plate (15, 16). The subplate plays a critical role in the development of thalamocortical and corticocortical connections and contributes directly and indirectly to EEG activity. The subplate serves as a potential site for mechanisms underlying the spontaneous activity transients, as they are dominated by low frequency waves with superimposed high frequency components; a feature of early preterm EEG (17).
Diminished Thalamocortical Development in Prematurity
Subcortical inputs from the brainstem and thalamus provide an essential source of patterned activity to the developing neocortex (18). Diffusion tensor imaging has revealed significantly diminished preterm thalamocortical connectivity (19). These anatomical findings are corroborated by hemodynamic measures of connectivity in functional magnetic resonate imaging. Compared to full-term infants, very preterm infants, scanned at term equivalent age, exhibit reduced connectivity between the cerebral cortex and thalamus (20). Spectral EEG reflects complex processes related to the maturation of cerebral and subcortical pathways modulated by activity and experience. SEF has been shown to negatively correlate with the degree of white matter injury in premature infants (21).
Disruption of the thalamocortical system, correlated in EEG progression, represents a major component of preterm brain injury (22, 23) and negatively impacts long-term neurodevelopmental outcome. In normal infants, resting cortical activity is characterized by a distinct spectral peak in the alpha range (8-13 Hz). Slowing of this oscillatory peak toward the upper theta-band (6-8 Hz) contributes to altered thalamocortical dynamics, which are associated with neurologic and neuropsychiatric pathology (24). Thalamic input contributes to cortical oscillatory activity that is critical for perception and cognition (25). Magnetoencephalography was used in a controlled study of global spectral activity in 11 school-age children (~7.5 years of age) born very preterm (≤32 weeks gestation) without major intellectual or neurological impairment (24). The very preterm children exhibited a slowing of peak frequency toward the theta-band over their bilateral frontal cortex and a reduction in alpha-band power over their bilateral frontal and temporal cortices, suggesting that mildly dysrhythmic thalamocortical interactions may contribute to altered spontaneous cortical activity in children born very preterm.
SEF modulation
Modulation of EEG spectra during and after somatosensory stimulation as observed in this study reflects the dynamics of thalamocortical excitation. Pulsed pneumatic orocutaneous stimulation produces synchronous oral mechanoreceptive afferent activity in the maxillary (V2) and mandibular (V3) divisions of the trigeminal nerve. Based on the observed up-regulation of SEF-90 during and immediately after pulsed orocutaneous stimulation, we hypothesize that the frequency of electrocortical activity is influenced by bilateral trigeminal-thalamo-cortical input. The spectra modulation may reflect a form of plasticity, adaptation that facilitates increased rates cortical activity for several minutes after the stimulus is removed.
In preterm infants aEEG matures predominantly with PMA (26). We demonstrate here that pulsed orocutaneous stimulation to the lip and mouth produces changes in SEF-90. Such orocutaneous stimulus has been shown to improve sucking and feeding skills in preterm infants (5). Other early developmentally appropriate experiences, such as gentle care, dim lights, rest periods, flexion positioning, and holding improve the development of brain structure and function, as evidenced by EEG spectral coherence and neurobehavioral function (4). Massage of preterm infants increases aEEG amplitudes and delta, alpha, theta, and beta EEG frequency domains (27). Skin-to-skin contact facilitates brain maturation in preterm infants, resulting in a more mature sleep pattern, improved respiratory regularity, and less spectral beta activity (28).
SEF asymmetry
We demonstrate significant asymmetry in 32 week PMA infant cortical SEF during oral somatosensory stimulation. There is an abundance of evidence to support anatomic and functional cortical asymmetry in early life (29). However, obtaining a complete understanding of structural and functional hemispheric asymmetries in early life remains one of the main challenges in developmental neuroscience. One example of functional asymmetry is the right sided lateralization of pitch processing detected at 35 weeks PMA in infants born at greater than 30 weeks gestation (29). Macroscopic left-right differences are present from the fetal life and onward (30). The developmental time scale also differs between the cerebral hemispheres. The superior frontal gyrus, the superior temporal gyrus, and Heschl's gyrus appear 1 to 2 weeks sooner in the right hemisphere than in the left (31), whereas other evidence has shown that the planum temporale and Heschl's gyrus are larger in the left hemisphere in fetuses and infants (31). Dubois et al. (13) reported early fetal third trimester brain rightward morphological asymmetry, with the right temporal sulcus being larger than the left. Neonates, in contrast to in adults, tend to be larger gray and white matter volumes in the left hemisphere (32). In infants three to 11 months old, myelination asymmetries were observed (33); with slower myelenation in the right cerebellum and left cerebrum relative to the contralateral structures. Dubois et al. (30) used diffusion tensor imaging and spatial localization methods to demonstrate early leftward asymmetries in the arcuate fasciculus and corticospinal tract, suggesting that early macroscopic geometry, microscopic organization, and white matter bundle maturation may be related to later functional lateralization.
Among the most intriguing lateralized functions in humans are hand preference and speech-language function. Studies during late gestation and shortly after birth are needed to understand how these asymmetries arise during brain growth and development and to determine if left—right differences in brain structure are the source or the consequence of functional lateralization (30).
Cortical adaptation
This study showed cortical adaptation in the SEF measure. Sensory adaptation, defined as response attenuation to repeated stimulation, can be short term or long term and can occur in any anatomic location along the neurosensory pathway, from peripheral mechanosensors to the cerebral cortex. Cortical adaptation to sustained sensory input, as used in the present study, is regarded as one of the most ubiquitous forms of short term plasticity. This mechanism improves spatial and temporal resolution of sensory events, which in turn improves motor learning and development of categorical perception for higher cognitive and communicative function. Neural adaptation can be observed in either the amplitude or frequency domain. In terms of frequency-following capabilities, the rate of neural response adaptation tends to increase as one proceeds from the periphery to the S1/S2 cortex. A recent functional neuroimaging study using the same pulsed pneumatic orocutaneous stimulus with a Soothie silicone pacifier demonstrated adaptation in neuromagnetic response magnitude in S1 and S2 in young adults (34).
We observed two interesting phenomena in the cerebral SEF-90 measures that merit further discussion: an overall upward shift in the SEF-90 (Figure 2,3) and a persistent SEF after-effect once the pulsed orocutaneous stimulus was removed. These effects may reflect different underlying mechanisms of cortical adaptation in the preterm brain. We reasoned that the overall up shift in SEF-90 in the experimental group is likely due to long term neural adaptation, indicative of the collective effects of repeated stimulation over several sessions and days. To test this notion in the present data set, an additional analysis of the SEF modulation among sham and experimental infants was completed to rule out the possibility that their baseline SEF profiles were somehow different between groups at the beginning of the study. As shown in Figure 3, the baseline SEF profiles, indicated by periods 1 and 2 for sham and experimental infants, were not significantly different, indicating the observed overall difference in SEF-90 between the two groups developed after experience of the different stimulus conditions (sham versus pulsed orocutaneous).
A significant after-effect or persistence in SEF-90 adaptation also was observed in the preterm infants who received the pulsed orocutaneous stimulation. This represents a relatively short term form of cortical adaptation to our servo-controlled orocutaneous experience and may serve as a biomarker of brain maturation and/or map the effects of sensory intervention therapies. In the mature brain, sustained sensory stimulation leads to transformations in the thalamocortical encoding that impact the nature of information conveyed about the sensory stimuli. Changes in mid-layer cortical neuron spiking activity reflect a switch in their role with adaptation, from coincidence detectors (tuned for stimulus detection in a non-adapted state) to integrators (tuned for stimulus discrimination after adaptation) (35). With repetitive stimulation, a dynamic cortical inhibitory mechanism shapes the initial activity into a stimulus-specific spatial pattern of columnar assemblies (36). Lateral interactions between adjacent columns are the result of intrinsic inhibitory (GABA-ergic) and excitatory effects (glutamate) which lead to a differential modification (across columns) of the membrane potential that can last for as long as several seconds.
Conclusion
The present study describes the application of a new oral somatosensory stimulation regimen with bedside EEG monitoring, an emerging NICU technology for detecting the spectral signature of electrocortical activity. Features of this approach are highly effective for monitoring preterm infants, including (1) the precocial nature of trigeminal orofacial cutaneous sensitivity (37, 38), and (2) high cortical magnification factor for orofacial sensory representation in S1/S2 (39). Thus, the dual-channel aEEG/EEG montage, situated over the infant's lateral cerebral convexity, allows quantitative mapping of the effects of a highly controlled somatosensory input on SEF-90 electrocortical activity. This work lays a foundation for further elucidating mechanisms of short and long term neural adaptation and maturation of thalamocortical networks in preterm infants.
METHODS
Participants
Santa Clara Valley Medical Center Human Subjects Internal Review Board approval was obtained and parental consent was obtained for all subjects before enrollment. Twenty two healthy preterm infants, (16M/6F) with a mean GA of 28.6 weeks (SD=2.1), birth weight of 1230 grams (SD=338), and PMA at test of 32.2 weeks (SD=1.09).
Inclusion Criteria
GA of 24-32 weeks and at least 28 weeks PMA at the time of enrollment.
Exclusion Criteria
Chromosomal abnormalities, multiple congenital anomalies or any major congenital anomalies. Infants with history of severe intraventricular hemorrhage, necrotizing enterocolitis (≥stage III), vocal cord paralysis, seizures, meningitis, or infants who were nippling all feeds at the time of enrollment.
Experimental Design
Study infants were randomly assigned to two groups, including those who received pulsed orocutaneous stimulation (Experimental), and those who did not (Control). The pulsed orocutaneous stimulation was delivered by a servo-controlled pneumatic amplifier (Ntrainer System, Innara Health, Shawnee, KS) specifically designed to transmit repeating pneumatic pulse trains to the soft tissues of the infant's lips-anterior tongue-intraoral mucosa-jaw through a regular (green) Soothie silicone pacifier (Philips AVENT Pacifier, Philips Healthcare, Andover, MA) (5). This 6-cycle orocutaneous stimulus burst was frequency modulated (interpulse cycle periods of 510, 526, 551, 580, and 626 ms) with an intertrain interval of 2 seconds and 50-ms pulse width (Figure 4). The pressure rise-fall time (10-90% intercepts) was 31 milliseconds, and resultant displacement at the pacifier-lip/tongue tissue interface was approximately 400 μm. Three-minute pneumatic orocutaneous stimulation periods were interleaved with 5.5 minute pause periods, where the pacifier was removed from the infant's mouth (Table 3). The control infants received a sham stimulation program in which infants were offered the same type of Soothie pacifier without patterned stimulation (blind pacifier). The staging of a single stimulation session was given concurrently with gavage. Infants had up to three daily sessions at routine feedings scheduled, every three hours. Infants were swaddled in a blanket with limbs positioned at midline, in a quiet-awake to drowsy state during stimulation.
Figure 4.
Frequency-modulated orocutaneous stimulus burst pattern consisting of 6 pulses followed by a 2-second pause period. A servo-controlled microprocessor generates the signal gating function (a) to dynamically ‘charge’ the intraluminal pressure of the silicone pacifier (b) resulting in rapid changes in pacifier geometry (c).
Table 3.
Stimulation schedule
| Periods | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 |
| Duration (minutes) | 3 | 3 | 3 | 5.5 | 3 | 5.5 | 3 | 3 | 3 |
| Baseline | Nasogastric Feeding | Post | |||||||
| Experimental | Exp-ON | Exp-OFF | Exp-ON | Exp-OFF | Exp-ON | ||||
| Control | Cont-ON | Cont-OFF | Cont-ON | Cont-OFF | Cont-ON | ||||
Nine sequential periods are indicated by P1 through P9. Exp-ON: Experimental pacifier with pneumatic pulse stimulation is presented to the infant. Exp-OFF: pacifier is removed from the infant's mouth. Cont-ON: Controls blind pacifier stimulation is presented to the infant. Cont-OFF: pacifier is removed from the infant's mouth. The pulsed or blind ‘sham’ pacifier is presented to the infant during P3, P5, and P7. The pacifier is not in the infant's mouth during P1, P2, P4, P6, P8, and P9.
EEG recording and signal processing
Four neonatal hydrogel sensors (Natus Medical Incorporated, San Carlos, CA) were placed in the C3, C4, P3, and P4 positions according to the international 10–20 system for EEG monitoring. EEG signals were recorded on a BRM3 monitor (Brainz, Natus Medical Incorporated, San Carlos, California) for up to 4-days beginning at around 32 weeks PMA (Figure 5). The right- and left-side EEG signals were amplified (Gain=5K), bandpass-filtered, first-order high-pass filter (fc −3 dB @ 1 Hz) and a fourth-order low-pass Butterworth (fc −3 dB @ 50 Hz), and digitized at a sampling rate of 256 Hz. Brainz Analyze Research (v1.5) software was used to derive the SEF-90 at 1-min intervals. These electroencephalographic measures were derived from nine sequential epochs (data blocks), spanning 32 minutes each, and centered over the pulsed orocutaneous or the blind pacifier conditions. A total of 1620 EEG blocks were analyzed among the 22 infants. The average number of orosensory sessions sampled per infant was 8.18 (SE=1.09). Portions of recordings were excluded from analysis if electrode impedance exceeded 10 kΩ, or if there was gross movement, electrical noise artifact, or asymmetry of baseline between the two channels (14).
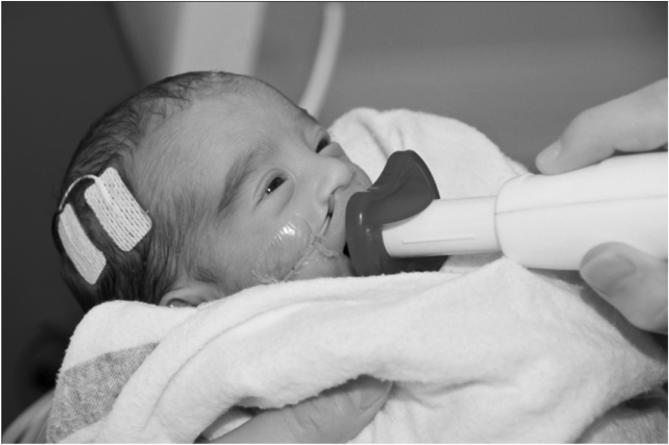
Figure 5.
Preterm infant with aEEG and pneumatically pulsed stimulation through a silicone pacifier coupled to the handpiece of the Ntrainer System (Innara Health, Inc., Shawnee, KS).
Statistical Analyses
Mixed models for repeated measures were used to compare the EEG SEF-90 measures among four stimulus conditions, including (1) Experimental pacifier with pneumatic pulse stimulation (Exp-ON), (2) Experimental pacifier without pneumatic pulse stimulation (Exp-Off), (3) Controls with blind (nonpulsitile) pacifier stimulation (Cont-On), and (4) Controls without blind (nonpulsitile) pacifier stimulation (Cont-Off). Adjusting for the infants’ GA and birth weight, mixed models estimated the stimulus effect on each outcome via the use of restricted maximum likelihood estimator and compound symmetric error covariance structure. When the stimulus effect was significant at 0.05 alpha level, adjusted means were pairwise compared using a Bonferroni-corrected p-value. All analyses were conducted using SAS 9.3 (SAS Institute, Cary, NC).
ACKNOWLEDGMENTS
Gratitude is expressed for the extreme dedication of Santa Clara Valley Medical Center NICU staff and the families of our preterm participants.
STATEMENT OF FINANCIAL SUPPORT: This study was supported in part by grants from Santa Clara County First Five (San Jose, CA), Valley Medical Center Foundation (San Jose, CA), National Institutes of Health (NIH) (Bethesda, MD; grant R01 DC003311 to SM Barlow, and grant P30 HD02528), and the Sutherland Family Foundation (Lawrence, KS).
Footnotes
DISCLOSURE: None of the authors have a direct financial relation with the manufacturers of the Soothie silicone pacifier, Brainz EEG monitor and Analyze software, or with the SAS statistical software. Dr. Barlow is the inventor of the Ntrainer System, which is licensed by the University of Kansas to Innara Health, Inc (Shawnee, KS).
REFERENCES
- 1.Penn AA, Shatz CJ. Brain waves and brain wiring: the role of endogenous and sensory-driven neural activity in development. Pediatr Res. 1999;45:447–58. doi: 10.1203/00006450-199904010-00001. [DOI] [PubMed] [Google Scholar]
- 2.Ball G, Boardman JP, Rueckert D, et al. The effect of preterm birth on thalamic and cortical development. Cereb Cortex. 2012;22:1016–24. doi: 10.1093/cercor/bhr176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Limperopoulos C, Gauvreau KK, O'Leary H, et al. Cerebral hemodynamic changes during intensive care of preterm infants. Pediatrics. 2008;122:e1006–13. doi: 10.1542/peds.2008-0768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Als H, Duffy FH, McAnulty G, et al. NIDCAP improves brain function and structure in preterm infants with severe intrauterine growth restriction. J Perinatol. 2012;32:797–803. doi: 10.1038/jp.2011.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barlow SM, Finan DS, Lee J, Chu S. Synthetic orocutaneous stimulation entrains preterm infants with feeding difficulties to suck. J Perinatol. 2008;28:541–8. doi: 10.1038/jp.2008.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Greisen G, Leung T, Wolf M. Has the time come to use near-infrared spectroscopy as a routine clinical tool in preterm infants undergoing intensive care? A Math Phys Eng Sci. 2011;369:4440–51. doi: 10.1098/rsta.2011.0261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Griesmaier E, Enot DP, Bachmann M, et al. Systematic characterization of amplitude-integrated EEG signals for monitoring the preterm brain. Pediatr Res. 2013;73:226–35. doi: 10.1038/pr.2012.171. [DOI] [PubMed] [Google Scholar]
- 8.Lena Hellstrom-Westas LSdV, Rosen Ingmar. Atlas of Amplitude-Integrated EEGs in the Newborn. 2nd Ed. Informa Healthcare; London, United Kingdom: 2008. [Google Scholar]
- 9.Bell AH, McClure BG, McCullagh PJ, McClelland RJ. Variation in power spectral analysis of the EEG with gestational age. J Clin Neurophysiol. 1991;8:312–9. doi: 10.1097/00004691-199107010-00007. [DOI] [PubMed] [Google Scholar]
- 10.Niemarkt HJ, Jennekens W, Pasman JW, et al. Maturational changes in automated EEG spectral power analysis in preterm infants. Pediatr Res. 2011;70:529–34. doi: 10.1203/PDR.0b013e31822d748b. [DOI] [PubMed] [Google Scholar]
- 11.Niemarkt HJ, Jennekens W, Maartens IA, et al. Multi-channel amplitude-integrated EEG characteristics in preterm infants with a normal neurodevelopment at two years of corrected age. Early Hum Dev. 2012;88:209–16. doi: 10.1016/j.earlhumdev.2011.08.008. [DOI] [PubMed] [Google Scholar]
- 12.Biagioni E, Frisone MF, Laroche S, et al. Maturation of cerebral electrical activity and development of cortical folding in young very preterm infants. Clin Neurophysiol. 2007;118:53–9. doi: 10.1016/j.clinph.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 13.Dubois J, Benders M, Cachia A, et al. Mapping the early cortical folding process in the preterm newborn brain. Cereb Cortex. 2008;18:1444–54. doi: 10.1093/cercor/bhm180. [DOI] [PubMed] [Google Scholar]
- 14.Mizrahi EM HR, Kellaway P, Stockard-Pope JE. Atlas of neonatal electroencephalography. 3rd ed. Lippincott Williams & Wilkins; Philadelphia, PA: 2004. [Google Scholar]
- 15.Kostović I, Jovanov-Milosević N. The development of cerebral connections during the first 20-45 weeks’ gestation. Semin Fetal Neonatal Med. 2006;11:415–22. doi: 10.1016/j.siny.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 16.Kostovic I, Rakic P. Developmental history of the transient subplate zone in the visual and somatosensory cortex of the macaque monkey and human brain. J Comp Neurol. 1990;297:441–70. doi: 10.1002/cne.902970309. [DOI] [PubMed] [Google Scholar]
- 17.Vanhatalo S, Kaila K. Development of neonatal EEG activity: from phenomenology to physiology. Semin Fetal Neonatal Med. 2006;11:471–8. doi: 10.1016/j.siny.2006.07.008. [DOI] [PubMed] [Google Scholar]
- 18.Grant E, Hoerder-Suabedissen A, Molnár Z. Development of the corticothalamic projections. Front Neurosci. 2012;6:53. doi: 10.3389/fnins.2012.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ball G, Boardman JP, Aljabar P, et al. The influence of preterm birth on the developing thalamocortical connectome. Cortex. 2013 Jun;49(6):1711–21. doi: 10.1016/j.cortex.2012.07.006. doi: 10.1016/j.cortex.2012.07.006. Epub 2012 Aug 9. [DOI] [PubMed] [Google Scholar]
- 20.Smyser CD, Inder TE, Shimony JS, et al. Longitudinal analysis of neural network development in preterm infants. Cereb Cortex. 2010;20:2852–62. doi: 10.1093/cercor/bhq035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Inder TE, Buckland L, Williams CE, et al. Lowered electroencephalographic spectral edge frequency predicts the presence of cerebral white matter injury in premature infants. Pediatrics. 2003;111:27–33. doi: 10.1542/peds.111.1.27. [DOI] [PubMed] [Google Scholar]
- 22.Kostović I, Judas M. The development of the subplate and thalamocortical connections in the human foetal brain. Acta Paediatr. 2010;99:1119–27. doi: 10.1111/j.1651-2227.2010.01811.x. [DOI] [PubMed] [Google Scholar]
- 23.Miller SP, Ferriero DM. From selective vulnerability to connectivity: insights from newborn brain imaging. Trends Neurosci. 2009;32:496–505. doi: 10.1016/j.tins.2009.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Doesburg SM, Ribary U, Herdman AT, et al. Magnetoencephalography reveals slowing of resting peak oscillatory frequency in children born very preterm. Pediatr Res. 2011;70:171–5. doi: 10.1203/PDR.0b013e3182225a9e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ribary U. Dynamics of thalamo-cortical network oscillations and human perception. Prog Brain Res. 2005;150:127–42. doi: 10.1016/S0079-6123(05)50010-4. [DOI] [PubMed] [Google Scholar]
- 26.Sisman J, Campbell DE, Brion LP. Amplitude-integrated EEG in preterm infants: maturation of background pattern and amplitude voltage with postmenstrual age and gestational age. J Perinatol. 2005;25:391–6. doi: 10.1038/sj.jp.7211291. [DOI] [PubMed] [Google Scholar]
- 27.Rudnicki J, Boberski M, Butrymowicz E, et al. Recording of amplitude-integrated electroencephalography, oxygen saturation, pulse rate, and cerebral blood flow during massage of premature infants. Am J Perinatol. 2012;29:561–6. doi: 10.1055/s-0032-1310529. [DOI] [PubMed] [Google Scholar]
- 28.Scher MS, Ludington-Hoe S, Kaffashi F, Johnson MW, Holditch-Davis D, Loparo KA. Neurophysiologic assessment of brain maturation after an 8-week trial of skin-to-skin contact on preterm infants. Clin Neurophysiol. 2009;120:1812–8. doi: 10.1016/j.clinph.2009.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mento G, Suppiej A, Altoè G, Bisiacchi PS. Functional hemispheric asymmetries in humans: electrophysiological evidence from preterm infants. Eur J Neurosci. 2010;31:565–74. doi: 10.1111/j.1460-9568.2010.07076.x. [DOI] [PubMed] [Google Scholar]
- 30.Dubois J, Hertz-Pannier L, Cachia A, Mangin JF, Le Bihan D, Dehaene-Lambertz G. Structural asymmetries in the infant language and sensori-motor networks. Cereb Cortex. 2009;19:414–23. doi: 10.1093/cercor/bhn097. [DOI] [PubMed] [Google Scholar]
- 31.Chi JG, Dooling EC, Gilles FH. Left-right asymmetries of the temporal speech areas of the human fetus. Arch Neurol. 1977;34:346–8. doi: 10.1001/archneur.1977.00500180040008. [DOI] [PubMed] [Google Scholar]
- 32.Gilmore JH, Lin W, Prastawa MW, et al. Regional gray matter growth, sexual dimorphism, and cerebral asymmetry in the neonatal brain. J Neurosci. 2007;27:1255–60. doi: 10.1523/JNEUROSCI.3339-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Deoni SC, Mercure E, Blasi A, et al. Mapping infant brain myelination with magnetic resonance imaging. J Neurosci. 2011;31:784–91. doi: 10.1523/JNEUROSCI.2106-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Popescu M, Barlow S, Popescu EA, et al. Cutaneous stimulation of the digits and lips evokes responses with different adaptation patterns in primary somatosensory cortex. Neuroimage. 2010;52:1477–86. doi: 10.1016/j.neuroimage.2010.05.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang Q, Webber RM, Stanley GB. Thalamic synchrony and the adaptive gating of information flow to cortex. Nat Neurosci. 2010;13:1534–41. doi: 10.1038/nn.2670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Whitsel BL FO, Kelly DG, Tommerdahl MA. Information Processing in Somatosensory System. Macmillan Press; London, England: 1991. Mechanisms of dynamic peri- and intracolumnar interactions in somatosensory cortex: stimulus specific contrast enhancement by NMDA receptor activation. pp. 353–69. [Google Scholar]
- 37.Humphrey T. The development of mouth opening and related reflexes involving the oral area of human fetuses. Ala J of Med Sci. 1968;5:126–57. [PubMed] [Google Scholar]
- 38.Barlow SM, Finan DS, Bradford PT, Andreatta RD. Transitional properties of the mechanically evoked perioral reflex from infancy through adulthood. Brain Res. 1993;623:181–8. doi: 10.1016/0006-8993(93)91425-r. [DOI] [PubMed] [Google Scholar]
- 39.Sathian K, Zangaladze A. Tactile spatial acuity at the human fingertip and lip: bilateral symmetry and interdigit variability. Neurology. 1996;46:1464–6. doi: 10.1212/wnl.46.5.1464. [DOI] [PubMed] [Google Scholar]