Abstract
The objective of this study was to prepare and characterise nevirapine nanosuspensions so as to improve the dissolution rate of nevirapine. Nevirapine is a nonnucleoside reverse transcriptase inhibitor of immunodeficiency virus type-1 and it is poorly water-soluble antiretroviral drug. The low solubility of nevirapine can lead to decreased and variable oral bioavailability. Nanosuspension can overcome the oral bioavailability problem of nevirapine. Nevirapine nanosuspensions were prepared using nanoedge method. The suspensions were stabilised using surfactants Lutrol F 127 or Poloxamer 407 and hydroxypropyl methyl cellulose. The nanosuspension was characterised for particle size, polydispersibility index, crystalline state, particle morphology, in vitro drug release and pharmacokinetics in rats after oral administration. The results support the claim for the preparation of nanosuspensions with enhanced solubility and bioavailability.
Keywords: Nanoedge method, Polydispersibility index, AIDS, Nanosuspension, In vitro drug release, Pharmacokinetic study
Modification and improvement of solubility has been attempted with much success for the development of effective drug formulation[1]. This is especially true for Biopharmaceutical Classification System (BCS) class II drugs. Compounds exhibiting dissolution rate-limited bioavailability are considered class II according to the BCS classification and these compounds have low and variable bioavailability. The solubility of these drug candidates can be enhanced using suitable conventional formulation strategies, which include cosolvents, milling techniques, super critical processing, solid dispersions, including complexation and precipitation techniques[2]. There still remains an unmet need to equip the pharmaceutical industry with tools to effectively enhance the solubility of BCS class II compounds. With particle engineering technologies, the solubility can be enhanced. Nanosuspension technology is one such technique. With decrease in size, the surface area increases and thereby improved solubility can be achieved. In this regard, nanoscale formulations have attracted attention of various pharmaceutical scientists and these products especially, nanosuspensions, demonstrated clinical and market success[3]. The reduction of drug particles to nanosize leads to a significant increase in the dissolution rate, solubility and therefore enhances bioavailability. Nanosuspensions lead to a reduction in the particle size and/or can transform drugs from a crystalline to an amorphous state and thereby enhance the dissolution rate and solubility[4]. Variability of fed-fasted state bioavailability with nanosuspension formulation can be reduced. Increase in saturation solubility is achieved with nanosuspenions by an increase in dissolution rate of the compound. There is an increase in bioavailability due to increased dissolution rate, solubility as well as increased surface area with nanosuspensions[5]. There can be a greater increase in the bioavailability with nanosuspensions compared with micronisation, another technique commonly used in industry to enhance the solubility and dissolution rate by the way of reducing the surface area.
Acquired immunodeficiency syndrome (AIDS) is a serious disease afflicting several populations of the world. Several classes of the drugs are used in the treatment of AIDS. Of these, nonnucleoside reverse transcriptase inhibitors (NNRTIs) are a specific class of antiAIDS drugs[6]. Some of the NNRTIs use is limited due to low bioavailability resulting from dissolution rate-limited bioavailability. Their bioavailability can be improved by formulating as nanosuspensions[4]. In this study we chose nevirapine, a BCS class II NNRTI with undesirable solubility and dissolution kinetics from the dosage form. Its solubility in neutral pH is about 0.1 mg/ml. Although the drug appears to be well absorbed orally, at higher doses, its bioavailability is low and variable[7]. On occasions it is administered as 200 mg twice daily. Increasing the dose to 400 mg to provide once daily dosing resulted in variable bioavailability profiles[8]. This has been attributed to its limited solubility and dissolution rate-limited absorption. Developing novel nanosuspension formulations might help reduce this variability in bioavailability. Although once a day sustained release (SR) formulations have been developed by several groups to solve this problem[9,10,11], these formulations are not practical for paediatric use. Nevirapine is available as oral tablets as well as oral paediatric suspension. These conventional formulations of nevirapine were approved during 1996-98[6]. Later on, extended release formulations were also approved for clinical use. Clinical trials conducted during the beginning of this century proved that it is better than indinavir, nelfinavir and efavirenz. Because of its high potency, no food effect, low pill burden and low cost, combination of nevirapine with other antiAIDS drugs has been used as the first line therapy in developing countries[12]. The innovator of this product is Boehringer Ingelheim Pharmaceuticals, USA and patents with conventional dosage form have already been expired and currently several generic products are available in the USA and other regulated markets. Challenges still remain in the area of research for developing products of nevirapine to further enhance its solubility and reduce fluctuations in bioavailability. A nanosuspension can be one of such products, which can increase the bioavailability upon oral administration better than the marketed oral suspension, leading to increased in vivo efficacy. This type of approach has been previously reported for other drugs[13] as well as for nevirapine. In this study, nevirapine nanosuspensions were prepared and investigated for improved macrophage delivery after i.v. administration[14]. However, nevirapine nanosuspensions for oral delivery were not previously investigated. The nano formulations prepared in this study are mainly intended for paediatric purposes. The objective of the present study was to prepare and characterise nevirapine nanosuspensions intended for enhancement in oral bioavailability and compare the improvement with already existing marketed nevirapine oral suspension. Nanosuspensions of nevirapine were prepared using nanoedge technique and then they were subjected to in vitro and in vivo characterisation[4,15].
MATERIALS AND METHODS
Nevirapine, poloxamer 407 were obtained from Hetero Labs (Hyderabad, India) hydroxypropyl methylcellulose (HPMC) was obtained from Merck India Ltd. (Mumbai). Sodium lauryl sulphate (SLS), Poloxamer were obtained from Hi Media Laboratories (Mumbai, India). Tween 80 and dichloromethane were obtained from S. D. Fine-Chem. Ltd. (Mumbai, India). PVPK30 and carboxymethylcellulose were obtained from Merck India.
Preparation of nanosuspensions:
Nanosuspensions were prepared using nanoedge method[4]. Initially nevirapine solubility studies in various solvents were performed to select a suitable solvent to be used in the formulation. The solvents used were ethanol, methanol, dichloromethane, isopropanol, acetone and ethyl acetate. To dissolve nevirapine, a cyclo mixture and a sonicator were used. Dichloromethane showed good solubility of 10 mg/ml. Hence dichloromethane was chosen as the solvent.
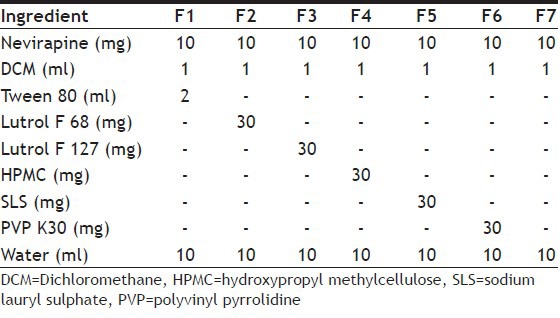
The method of preparation has two steps, first, precipitation using the solvent evaporation technique and second, high-pressure homogenisation. In the first step, the drug was dissolved in the solvent at room temperature. This solution was added drop wise using a syringe needle into different volumes of water containing different amounts of surfactant on a magnetic stirrer. Stirring was performed at room temperature and volatile solvents were allowed to evaporate. Stirring was kept for 3 h leading to precipitation of nanosuspension of the drug. A variety of nanosuspension formulations were prepared using different surfactants and other excipients (Table 1). The nanosuspension from step I was homoginised using Diax 900 homogeniser (Heidolph, Germany) for 5 min and the above prepared suspension was then subjected to sonication for 20 min using an ultrasonicator (Probe 12 T, Bandelin). This suspension was then passed through high-pressure homogenisation in for 10 cycles at 10 000 psi Using Emulsiflex-c5 HPH.
Table 1.
VARIOUS NEVIRAPINE NANOSUSPENSIONS AND THEIR COMPOSITIONS
Characterisation of nanosuspensions:
Particle size, surface morphology, crystallinity, saturation solubility and in vitro release profile of the nanosuspensions prepared were evaluated[15]. A photon correlation spectroscopy (PCS) on a submicron particle size analyser (N4 plus, Beckman-Coulter) at a scattering angle of 90° was used at 20° to determine the particle size. Surface morphology was determined using a scanning electron microscope. Briefly, a concentrated aqueous suspension was spread over a slab and dried under vacuum. The sample was shadowed with 20 nm thick gold layer in a cathodic evaporator. Photographs were taken using a JSM-5200 Scanning Electron Microscope (Tokyo, Japan) operated at 20 kV. Dried nevirapine nanosuspensions were analysed for crystallinity using the differential scanning calorimeter (DSC Q10, TA Instruments). Approximately 4-5 mg of each sample was sealed in a standard aluminium pan with lid. The temperature range of measurement was 200-250° and the heat flow rate was set to 10°/min. To determine saturation solubility of nanosuspensions in 0.1 N HCl, samples were subjected for shaking in screw-capped vials for 24 h. Samples were withdrawn and filtered through 0.22 micron membrane filters and analysed by ultraviolet (UV) for drug content. An in vitro drug release study was performed for all the formulations, marketed suspension and pure nevirapine powder using USP Type II dissolution apparatus (paddle) under the following conditions; dissolution medium 900 ml of 0.1 N HCl, rotation speed 50 rpm, temperature 37±0.5°. Sampling volume was 5 ml and sampling time chosen was 5, 10, 15, 30, 45 and 60 min. Samples were withdrawn using a 0.22 micron syringe filter. It was ensured that the particles are not taken up into the syringe. As the average particle size was more than the filter size, we assumed that only the dissolved drug was taken for analysis. Sink condition was maintained by replacing the volume equivalent to the quantity removed with fresh dissolution medium.
Pharmacokinetic study:
The studies were conducted with prior approval of the Institutional Animal Ethical Committee (IAEC/04/UCPSC/HETRO). Healthy male Wistar rats (weighing 190-220 g) were procured and divided into two groups containing six animals each, group 1 and group 2, which were orally administered with an optimised nanosuspension formulation prepared with Lutrol and a marketed nevirapine suspension (Veramune) at a dose of 50 mg/kg with the help of rat oral feeding tube, respectively.
At appropriate predetermined time intervals after oral administration, blood samples were collected (0.33, 0.66, 1, 1.5, 2, 3, 4, 6, 9 and 12 h) by retro-orbital venous plexus puncture. The ethylenediamine tetracetic acid (EDTA) and blood samples were allowed to centrifuge for 10 min at 3000 rpm. The serum was separated and transferred into clean micro centrifuge tubes and stored at −20° until high-performance liquid chromatography (HPLC) analysis. The drug was then extracted. Briefly, to 100 μl of serum, 100 μl of internal standard was added and 100 μl of mobile phase and 200 μl of cold methanol was added and vortexed for 1 min and centrifuged at 3500 rpm for 15 min. Then supernatant was transferred to 2-ml Eppendorf tubes, 0.6 ml of dichloromethane was added and vortexed for 1 min followed by centrifugation at 10 000 rpm for 10 min. Exactly 20 μl of the supernatant sample was spiked onto the HPLC system for analysis. Nevirapine was quantified in rat plasma using a HPLC assay. The HPLC equipment consisted of a quaternary pump spectra system P4000, an auto sampler spectra system and a spectrophotometric detector. Hibar R 150.4,6 Purospher R STAR RP-18e (5 μm) column was used. The pharmacokinetic parameters of individual rats for optimised nevirapine nanosuspension and marketed suspension were calculated by noncompartmental estimations using Kinetica 2000 software. The statistical comparison of data was done by Student Unpaired t-test at significance level of P-value <0.05 using GraphPad Prism (version 5.0, 2007).
RESULTS AND DISCUSSION
Several formulations of nevirapine nanosuspensions (F1-F7) with different compositions were successfully prepared using nanoedge technique. The formulations are listed in Table 1. Formulation F5 and F6 used SLS and PVP K30 as the stabilisers of nanosuspensions. These formulations forms floccules immediately and after certain time they began to settle. The suspensions did not redisperse after shaking[16]. The other formulations, which were prepared by precipitation, followed by homogenisation were characterised for particle size after the step I (precipitation). The particle size was measured using an optical microscope. The particle sizes of F1, F2, F3, F4, F7 were 2.6, 3.1, 0.89, 0.98, 1.92 μm, respectively. The formulations, which used HPMC (F4), Lutrol F 127 (F3) and there combination (F7), showed good particle size, whereas the particles prepared using Tween 80 and Lutrol F 68 demonstrated higher size. The nanosuspensions F3, F4 and F7 were further taken to the homogenisation, sonication and high-pressure homogenisation steps. The particle size and polydispersity index (PDI) at the end of preparation of the suspensions is shown in Table 2. Comparison of mean size and PDI of nevirapine nanosuspension was done during various step of processing initially after homogenisation at 12 000 rpm for 5 min then sonication for 20 min followed by high pressure homogenisation 10 cycles at 10 000 psi. During various stages of the processing there was a decrease in particle size (Table 2). Formulation F3 stabilised using Lutrol F 127 has shown lesser particle size and PDI compared with other formulations and was further taken for characterisation and animal studies. Scanning electron microscope pictures of this optimised formulation indicated that the particles are spherical (fig. 1).
Table 2.
COMPARISON OF MEAN SIZE AND PDI OF NEVIRAPINE NANOSUSPENSIONS
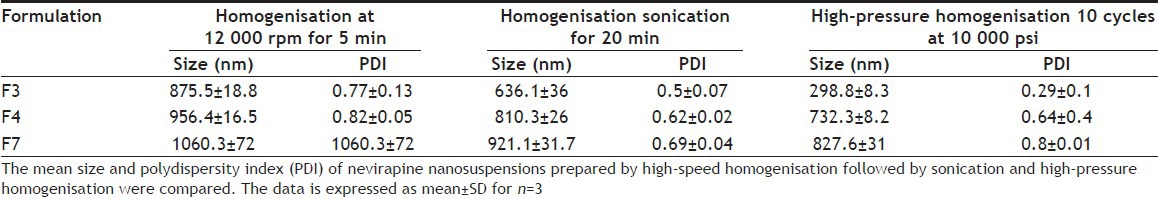
Fig. 1.

Scanning electron microscope pictures of nanosuspensions Magnification of a is ×10 and the magnification of b is ×50.
DSC studies were used to characterise the physical state of the drug in various formulations. Thermogram of pure nevirapine and optimised nevirapine nanosuspension using various excipients formulation has shown various peaks at different temperature points. The thermograms are shown in fig. 2. Pure drug has shown a peak at 229°. Nanosuspension prepared using HPMC has shown peak at 226.53° while formulation containing Lutrol F 127 has shown peak at 225.43°. The results suggested no change in melting endotherm, which was present in all the samples tested. There was a slight change in melting temperatures of the drug formulated into nanosuspensions with surfactant and stabiliser but melting endotherm is strong in both the cases and it is stronger than that of the pure drug. Thus, it can be inferred that the drug was not converted either fully or partly into amorphous state upon formulation into nanosuspension in the case of optimised formulation. As the concentration of Lutrol F127 in the formulation might be very low, we did not observe a melting endotherm for this excipient. Saturation solubility was determined for each of the optimised nanosuspensions, pure drug and the marketed formulation. The data is presented in Table 3. From the results we observe that the formulation using Lutrol F 127 has demonstrated higher saturation solubility value compared with marketed suspension. Drug release was studied with optimised formulation and compared with that of the pure drug as well as marketed suspension. The data indicates that the drug release from the optimised formulation was higher when compared with pure drug as well as the marketed suspension (fig. 3). Since there is an increase in the dissolution rate and saturation solubility without any change of physical state to amorphous form, it can be concluded that the enhancement in the dissolution as well as saturation solubility are due to nanonisation and not due to the conversion to a different physical state.
Fig. 2.
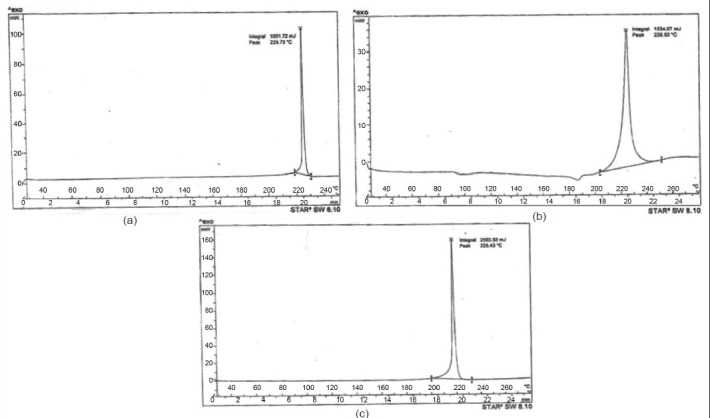
DSC thermograms.
DSC thermograms of a, pure nevirapine; b, nevirapine formulation with HPMC; and c, nevirapine formulation with Lutrol F127.
Table 3.
SATURATION SOLUBILITY OF PURE DRUG, MARKETED SUSPENSION AND NEVIRAPINE NANOSUSPENSION FORMULATIONS
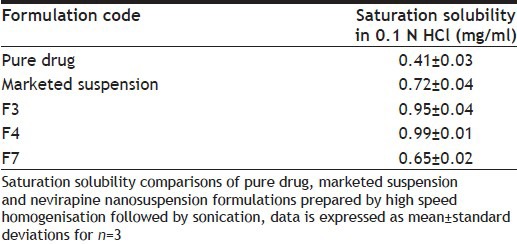
Fig. 3.
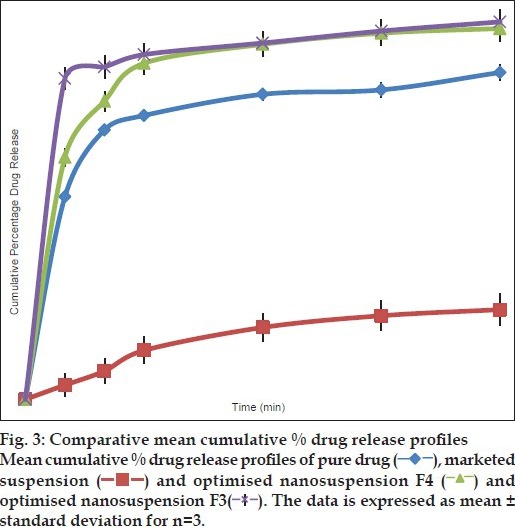
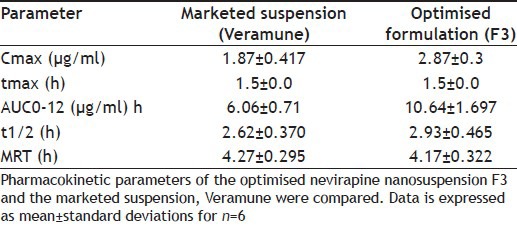
After demonstrated improved results with optimised formulations, these were further taken for animal studies to determine the comparative profiles in vivo. HPLC method to determine the drug levels in the plasma was developed. The following are the optimised parameters: mobile phase: 0.015 M phosphate buffer (pH 3.8 with OPA, 88%): acetonitrile (12%); flow rate: 1 ml/min; column: Hibar R 150-4, 6 Purospher STAR RP-18e (5 μm); column temperature: 30°; UV-detection at: 254 nm; detector sensitivity: 0.0005 AUFS; injection volume: 20 μl; retention time: 7.6 min. The chromatogram obtained is shown in fig. 4. Plasma profiles show that formulated nevirapine nanosuspension has good mean serum concentration than that of marketed suspension (fig. 5). Pharmacokinetic parameters were determined for individual groups and these are tabulated. It was found that optimised nanosuspension formulation showed high Cmax and area under the curve (AUC) values of 2.88±0.380 and 10.64±1.697 when compared with marketed suspension, which showed lower Cmax and AUC values of 1.86±0.417 and 6.07±0.71, respectively, and there is statistically significant difference between Cmax and AUC of the nanosuspension and marketed suspension was observed. Mean residence time (MRT) and t½ of optimised nanosuspension (4.17±0.322 and 2.92 h) showed no significant difference with that of marketed suspension (4.27±0.295 and 2.62 h). Tmax of both formulations was found to be the same and it was 1.5 h for each individual rat. The bioavailability of F3 nanosuspension was found to be increased by 1.752 times than that of Veramune and the relative bioavailability was 175.2% to that of marketed suspension (Table 4).
Fig. 4.
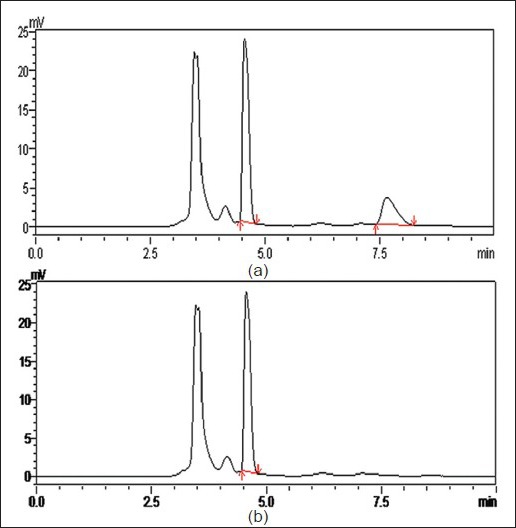
Chromatograms of nevirapine.
Chromatograms of nevirapine showing a. internal standard and nevirapine and b, blank with internal standard
Fig. 5.
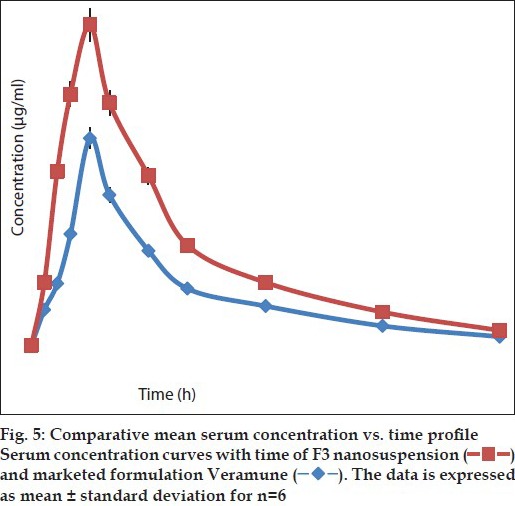
Table 4.
COMPARISON OF THE PHARMACOKINETIC PARAMETERS
Generic product development is a challenging task. Matching or excelling the innovator product in terms of various parameters is the criteria for the development of better and improved generic products. In this regard, dissolution and bioavailability are main selection parameters for product development of generics[17]. This is especially true for oral products whether tablets or suspensions. Formulation development studies were conducted in this work so as to aid in the development of newer proprietary formulations or better generic alternative for nevirapine, an antihuman immunodeficiency virus (antiHIV) drug mainly intended for paediatric use. The formulations developed in this study can also be further used in other population after transforming and formulating into a better tablet that can be given once a day with a 400-mg dose rather than use a complicated extended release dosage form. Nanosuspensions of nevirapine were investigated in this study so as to enhance the oral bioavailability. A nanosuspension is a submicron colloidal dispersion of drug particles stabilised by surfactants. The general particle size is in the range of <1 μm. The crystallinity of the drug is generally maintained while the dissolution rate is enhanced because of increased surface area. This enhancement in dissolution rate generally results in enhancement in bioavailability and reduces variability in bioavailability after oral administration[5]. It can also increase the saturation solubility. Further, if the drug is converted to amorphous form in the process of development of nanosuspension, the bioavailability is further enhanced. The issue of improving stability of an amorphous nanosuspension that can be incorporated either in to tablets or marketed as such has to be addressed at this stage in order to develop better formulations when compared with formulations developed using drug molecules as they are. However, in this study, crystalline nanosuspensions rather than amorphous nanosuspensions were formed with the process we used. This was confirmed using DSC thermograms. The pure drug gave a pure melting endotherm at its melting point. Further, the pure drug when processed to form a nanosuspension using same technique as used for the optimised nanosuspension in the presence of HPMC as stabiliser and DSC was performed, although there was a change in the melting point, a sharp endotherm resulted. This indicates that there is no formation of amorphous drug in the process of preparation of nanosuspension. When DSC was performed over optimised nanosuspension very similar results were obtained, suggesting that there is no formation of amorphous drug in the process. Such a conclusion is made because amorphous forms of drugs do not show a strong endotherm at the melting point. Further, we found that the heat of fusion is definitely higher for the nanosuspension compared with the pure drug corroborating the fact that there is no formation of amorphous nanosuspension in the process. Such an interpretation was previously made in the literature[18]. We can also conclude that although the process is affecting the melting point, the surfactant and stabiliser are not influencing the melting endotherm of the drug. Although a modulated DSC can be used to further elucidate the results, we thought that the data from simple DSC is strong enough to conclude the fact. Thus, this result clearly suggests that it was the reduction of the size of the particles in the nanorange that is leading to enhanced dissolution rate, solubility and bioavailability, and not because of conversion of drug into amorphous drug. Thus, increase in surface area for the drug is leading to enhanced bioavailability. Similar results were previously reported in the literature by taking solid dispersions of nevirapine[19,20]. Several studies clearly indicated that formation of solid dispersions increases the dissolution rate, solubility and bioavailability for this drug. In fact solid dispersions enhance the apparent surface area of the drug and leads to enhanced bioavailability. Thus, from our studies and earlier studies with solid dispersions, it can be clearly concluded that increase in surface area for nevirapine can result in enhanced bioavailability of the drug. In case of parenteral administration, the Cmax of the drug in the plasma can be enhanced with nanosuspensions while targeting into tumours by passive targeting can be achieved by enhanced penetration retention mechanism. The nanosuspensions can be administered using other routes of administration such as ocular, pulmonary, topical with added advantages. Currently several nanosuspension products are available in the market. This clearly indicates their success in patient treatment.
Nanosuspensions are prepared using either bottom up technology or top down technology. In bottom up technology, the particles are prepared from solution form as the starting point. In top down technology, larger particles are broken down to smaller particles of nanosize such as done in any of the milling techniques[21]. In this study, nevirapine nanosuspensions were prepared using nanoedge technology. It is a combination of both bottom up and top down technologies[4]. In the initial step, particles were formed by precipitation technique. The size of these particles was further reduced with ultrasonication and homogenisation techniques. A variety of solvents can be used in the precipitation technique. Nevirapine nanosuspensions were successfully prepared using the techniques used in this study. A suitable solvent should be selected so that suspensions in nanosize can be obtained. In this study, different solvents have been investigated for their suitability in the preparation of nanosuspensions by precipitation technique. After a few trials, dichloromethane was considered suitable for nevirapine. The antisolvent was water. The next criterion in the formulation development is the use of stabilisers. Stabilisers play an important role in the formulation development of nanosuspensions. Without stabilisers, the high surface energy of nanoparticles can lead to agglomeration or aggregation. Stabilisers are also used for wetting the drug and prevent Ostwald ripening. This results in the preparation of stable nanosuspensions[22]. In this study, different stabilisers such as Tween 80, Lutrol F68, Lutrol F 127, HPMC, SLS and PVP K30 were investigated for suitability to prepare physically stable nanosuspensions. HPMC and Lutrol 127 were found to be suitable stabilisers for nevirapine nanosuspensions. The optimised formulation containing these stabilisers was further investigated. The solid state of the drug at the end of preparation of nanosuspensions was investigated using DSC. The results indicate no change in crystallinity as indicated by the presence of drug peaks in the thermograms of nanosuspensions. Saturation solubility of optimised nanosuspension was determined and was compared with that of the pure drug. The results suggest that the saturation solubility of nevirapine was enhanced with nanosuspension technology. Several reports previously demonstrated an enhancement of saturated solubility with nanosuspensions[23]. Subsequently, in vitro release of different formulation of nevirapine, namely, nanosuspensions, pure drug and marketed formulation were investigated. Nanosuspensions demonstrated improved in vitro release compared with that of marketed oral suspensions and pure drug. The dissolution of the drug from marketed formulation is significantly lower compared with that of pure drug. This could be attributed to the polymers used as stabilisers to form the suspensions. Literature clearly indicates that the use of sodium CMC and HPMC as suspending agent clearly reduces the dissolution of the drugs[24]. In the marketed formulation we used in this study, Carbomer 934P is used as the suspending agent. Although from literature review, we have not come across reports indicating reduced dissolution of the drug with this polymer, the reason for reduced dissolution of the drug in our study can be attributed to this polymer. Also, the release profiles of the nanoformulation prepared using different stabilisers have been compared. The results demonstrate better results with optimised formulation that was stabilised with Lutrol F127 when compared with those prepared using HPMC. Further, saturation solubility in both cases is similar. Thus, we considered the formulation with Lutrol F127 as optimised formulation and used it for conducting in vivo bioavailability. In vivo bioavailability studies with nanosuspensions were performed to understand the use of nanosuspensions as generic alternate for oral suspensions of nevirapine. Bioavailability studies indicated that the optimised nanosuspension formulation for nevirapine in this study is better when compared with marketed formulation Veramune in terms of drug absorption.
Nanosuspensions improved the oral bioavailability of nevirapine when compared with that of oral suspensions. This suggests that an alternate novel formulation with improvement in oral bioavailability has been developed for this drug. Alternatively, from the results of this study it could be concluded that this formulation can be developed into an improved generic formulation or a proprietary formulation for this drug. Previously, several studies developed a variety of novel formulations for improvement in the oral bioavailability of nevirapine. For instance, Sowjanya et al.[25] developed floating drug delivery systems for nevirapine so as to improve the oral bioavailability. Das et al.[26] enhanced solubility and dissolution rate of nevirapine using solid dispersion technique. The study suggested that the bioavailability of this drug could be enhanced with the use of solid dispersions. Although ample research has been conducted to enhance the bioavailability of drugs using solid dispersions, there are only seven solid dispersions that have moved forward into the market till 2009 without a great success[27]. The current research towards this approach is to develop improved amorphous solid dispersions. However, we preferably used nanosuspension technology in this study instead of solid dispersions since we believe that nanotechnology is a smarter technology[28] and also due to our experience in this technology. Although floating drug delivery systems could be used in the enhancement of bioavailability of nevirapine, their practical utilisation is a question mark. For instance, in the past, various gastro retentive drug delivery systems have been developed but market success of these formulations is very low[29]. Co-crystals of nevirapine were also investigated[30,31]. In these studies it was found that the solubility of nevirapine was increased several times with co-crystals. Although co-crystals are better than amorphous solid dispersions in terms of stability, co-crystals still have certain demerits. The preferred co-crystal not only should improve the physical properties but also contain a pharmaceutically acceptable conformer that is nontoxic, has high weight percentage of active pharmaceutical ingredient (API) compared with conformer, be chemically stable under different conditions, should be amenable to low cost manufacture using existing technologies and should remain stable over the shelf for desired periods. We consider nanosuspensions are ideal novel alternative formulations for nevirapine either to make better paediatric generics in the form of suspension or tablets that can be used as an alternative to once a day nevirapine SR formulation. As this study generated crystalline nanosuspensions, we believe that the technology can be taken further for production and marketing without compromising on the physical stability. Thus this study generated better generic alternate to available oral formulations in the market or proprietary products. Although the present study is aimed at developing generic alternative to nevirapine, it has the potential to provide impetus for the development of novel and improved formulations for antiretroviral drugs. For instance, Werling et al. reported, in their patent application, the use of nanosuspension of antiretroviral agents for increased central nervous system delivery[32]. Similarly, several groups are working on developing better formulations for antiretroviral drugs[33]. Thus, our study also provides technical and research leads to such studies.
ACKNOWLEDGEMENTS
The authors Anju Raju and A. Jagadeesh Reddy thank M/s. Hetero Labs for providing necessary facilities utilised in carrying out parts of the work. The authors, A. V. Jithan, Anju Raju and J. Satheesh would like to acknowledge the Management of Mother Teresa College of Pharmacy for providing necessary facilities for carrying out the work and for writing of the manuscript. A. V. Jithan and J. Satheesh would like to acknowledge Nanomission, Department of Science and Technology, Government of India for partially funding this work. J. Satheesh is a Junior Research Fellow supported under this project by DST.
Footnotes
Raju, et al.: Nevirapine Oral Nanosuspensions
REFERENCES
- 1.Beg S, Swain S, Rizwan M, Irfnuddin M, Malini DS. Bioavailability enhancement strategies: Basics, formulation approaches and regulatory considerations. Curr Drug Deliv. 2011;8:691–702. doi: 10.2174/156720111797635504. [DOI] [PubMed] [Google Scholar]
- 2.Buckley ST, Frank KJ, Fricker G, Brandl M. Biopharmaceutical classification of poorly soluble drugs with respect to “enabling formulations”. Eur J Pharm Sci. 2013;50:8–16. doi: 10.1016/j.ejps.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 3.Chavhan SS, Petkar KC, Sawant KK. Nanosuspensions in drug delivery: Recent advances, patent scenarios, and commercialization aspects. Crit Rev Ther Drug. 2011;28:447–88. doi: 10.1615/critrevtherdrugcarriersyst.v28.i5.20. [DOI] [PubMed] [Google Scholar]
- 4.Raval AJ, Patel MJ. Preparation and characterization for solubility and dissolution rate enhancement of meloxicam. Int Res J Pharm Sci. 2011;1:42–9. [Google Scholar]
- 5.Liu Y, Xie P, Zhang D, Zhang Q. A minireview of nanosuspensions development. J Drug Target. 2012;20:209–23. doi: 10.3109/1061186X.2011.645161. [DOI] [PubMed] [Google Scholar]
- 6.Ena J, Amador C, Benito C, Pasquau F. Pharmacological and clinical evidence of nevirapine immediate- and extended-release formulations. HIV AIDS (Auck) 2012;4:169–79. doi: 10.2147/HIV.S35564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sarkar M, Perumal OP, Panchagnula R. Solid-state characterization of nevirapine. Indian J Pharm Sci. 2008;70:619–30. doi: 10.4103/0250-474X.45401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kappelhoff BS, Huitema AD, van Leth F, Robinson PA, MacGregor TR, Lange JM, et al. Pharmacokinetics of nevirapine: Once-daily versus twice-daily dosing in the 2NN study. HIV Clin Trials. 2005;6:254–61. doi: 10.1310/B5VU-FU5F-QNWC-UDCK. [DOI] [PubMed] [Google Scholar]
- 9.Bogner JR. Nevirapine extended-release for the treatment of HIV-1 infection. Expert Rev Antiinfect Ther. 2012;10:21–30. doi: 10.1586/eri.11.145. [DOI] [PubMed] [Google Scholar]
- 10.Heil EL, Corbett AH. Guidelines for the use of extended-release nevirapine in HIV-injected patients. Exp Opin Pharmacother. 2011;12:2713–8. doi: 10.1517/14656566.2011.630391. [DOI] [PubMed] [Google Scholar]
- 11.Arasteh K, Ward D, Plettenberg A, Livrozet JM, Orkin C, Cordes C, et al. Twenty-four-week efficacy and safety of switching virologically suppressed HIV-1-injected patients from nevirapine immediate release 200 mg twice daily to nevirapine extended release 400 mg once daily (TRANxITION) HIV Med. 2012;13:236–44. doi: 10.1111/j.1468-1293.2011.00969.x. [DOI] [PubMed] [Google Scholar]
- 12.Grob PM, Wu JC, Cohen KA, Rngraham RH, Shih CK, Hargrave KD, et al. Nonnucleoside inhibitors of HIV-1 reverse transcriptase: Nevirapine as a prototype drug. AIDS Res Hum Retroviruses. 1992;8:145–52. doi: 10.1089/aid.1992.8.145. [DOI] [PubMed] [Google Scholar]
- 13.Kassem MA, Abdel Rahman AA, Ghorab MM, Ahmed MB, Khalil RM. Nanosuspension as an ophthalmic delivery system for certain glucocorticoid drugs. Int J Pharm. 2007;340:126–33. doi: 10.1016/j.ijpharm.2007.03.011. [DOI] [PubMed] [Google Scholar]
- 14.Shegokar R, Singh KK. Nevirapine nanosuspensions for HIV reservoir targeting. Pharmazie. 2011;66:408–15. [PubMed] [Google Scholar]
- 15.Tian X, Li H, Zhang D, Liu G, Jia L, Zheng D, et al. Nanosuspension for parenteral delivery of a p-terphenyl derivative: Preparation, characteristics and pharmacokinetic studies. Colloids Surf B Biointerfaces. 2013;108:29–33. doi: 10.1016/j.colsurfb.2013.02.038. [DOI] [PubMed] [Google Scholar]
- 16.Abdelbary AA, Li X, El-Nabarawi M, Elassasy A, Jasti B. Effect of fixed aqueous layer thickness of polymeric stabilizers on zeta potential and stability of aripiprazole nanosuspensions. Pharm Dev Technol. 2013;18:730–5. doi: 10.3109/10837450.2012.727001. [DOI] [PubMed] [Google Scholar]
- 17.Pathak SM, Aggarwal D, Venkateswarlu V. Establishment of in vitro-in vivo equivalence of highly variable drugs - a generic product development perspective. Pharm Dev Technol. 2014;19:401–10. doi: 10.3109/10837450.2013.788513. [DOI] [PubMed] [Google Scholar]
- 18.Singh AK. Evaluation of orlistat solid dispersion using poloxomer 188 as hydrophilic carrier. Sch Res J. 2011;1:48–51. [Google Scholar]
- 19.Datta A, Ghosh NS, Ghosh S, Tapobana S, Samantha T, Das RC. Enhancement of solubility and dissolution profile of nevirapine by solid dispersion technique. Int J Chem Res. 2011;2:53–8. [Google Scholar]
- 20.Lokamatha KM, Shanta Kumar SM, Rao NR. Enhancement of solubility and dissolution rate of nevirapine by solid dispersion technique using dextran: Preparation and in vitro evaluation. Dig J Nanomater Biostruct. 2010;6:135–42. [Google Scholar]
- 21.Verma S, Gokhale R, Burgess DJ. A comparative study of top-down and bottom-up approaches for the preparation of micro/nanosuspensions. Int J Pharm. 2009;380:216–22. doi: 10.1016/j.ijpharm.2009.07.005. [DOI] [PubMed] [Google Scholar]
- 22.Patravale VB, Date AA, Kulkarni RM. Nanosuspensions: A promising drug delivery strategy. J Pharm Pharmacol. 2004;56:827–40. doi: 10.1211/0022357023691. [DOI] [PubMed] [Google Scholar]
- 23.Thakkar HP, Patel BV, Thakkar SP. Development and characterization of nanosuspensions of olmesartan medoxomil for bioavailability enhancement. J Pharm Bioallied Sci. 2011;3:426–34. doi: 10.4103/0975-7406.84459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Howard SA, Mauger J, Hsieh JW, Amin K. Suspending agent effects on steroid suspension dissolution profiles. J Pharm Sci. 1979;68:1475–9. doi: 10.1002/jps.2600681203. [DOI] [PubMed] [Google Scholar]
- 25.Sowjanya K, Shoba Deepthi K, Bharathi A. Formulation and evaluation of gastro-retentive floating matrix tablets of nevirapine. Int J Pharm Pharm Sci. 2012;4:368–75. [Google Scholar]
- 26.Das SK, Roy S, Kalimuthu Y, Khannam J, Nanda A. Solid dispersions: An approach to enhance the bioavailability of poorly water-soluble drugs. Int J Pharmacol Pharm Tech. 2012;1:37–46. [Google Scholar]
- 27.Janssens S, Mooter GV. Review: physical chemistry of solid dispersions. J Pharm Pharmacol. 2009;61:1571–86. doi: 10.1211/jpp/61.12.0001. [DOI] [PubMed] [Google Scholar]
- 28.Jithan AV, Madhavi K, Madhavi M, Prabhakar K. Preparation and characterization of albumin nanoparticles encapsulating curcumin intended for the treatment of breast cancer. Int J Pharm Investig. 2011;1:119–25. doi: 10.4103/2230-973X.82432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pawar VK, Kansal S, Asthana S, Chourasia MK. Industrial perspective of gastroretentive drug delivery systems: Physicochemical, biopharmaceutical, technological and regulatory consideration. Expert Opin Drug Deliv. 2012;9:551–65. doi: 10.1517/17425247.2012.677431. [DOI] [PubMed] [Google Scholar]
- 30.Caira MR, Bourne SA, Samsodien H, Engel E, Liebenberg W, Stieger N, et al. Co-crystals of the antiretroviral nevirapine: Crystal structures, thermal analysis and dissolution behavior. CrystEngComm. 2012;14:2541–51. [Google Scholar]
- 31.Gajar PP, Kokil AA, Karekar PS, Gurav YA, Yadav AV. Improvement in physicochemical properties of nevirapine co-crystals. Int J Pharm Technol. 2013;4:4831–42. [Google Scholar]
- 32.Werling J, Chaubal M, Kipp J, Rabinow B. Nanosuspensions of anti-retroviral agents for increased central nervous system delivery. US Patent Application No. US200502002094. [Google Scholar]
- 33.Swindells S, Flexner C, Fletcher CV, Jacobson JM. The critical need for alternative antiretroviral formulations and obstacles and their development. J Infect Dis. 2011;204:669–74. doi: 10.1093/infdis/jir370. [DOI] [PMC free article] [PubMed] [Google Scholar]


