Abstract
Aim:
To test the hypothesis that different magnitude of resistance of denervated skeletal muscle to nondepolarizing muscle relaxants (NDMRs) is related to their varying potencies at ɛ-AChR and γ-AChR.
Methods:
Both innervated and denervated mouse muscle cells, and human embryonic kidney 293 (HEK293) cells expressing ɛ-AChR or γ-AChR were used. The effects of NDMRs on nAChR were explored using whole-cell patch clamp technique.
Results:
NDMRs vecuronium (VEC), atracurium (ATR) and rocuronium (ROC) produced reversible, dose-dependent inhibition on the currents induced by 30 μmol/L acetylcholine both in innervated and denervated skeletal muscle cells. Compared to those obtained in innervated skeletal muscle cells, denervation shifted the concentration-response curves rightward and significantly increased the 50% inhibitory concentration (IC50) values (VEC: from 11.2 to 39.2 nmol/L, P<0.01; ATR: from 24.4 to 129.0 nmol/L, P<0.01; ROC: from 37.9 to 101.4 nmol/L, P<0.01). In HEK293 cell expression system, ATR was less potent at γ-AChR than ɛ-AChR (IC50 values: 35.9 vs 22.3 nmol/L, P<0.01), VEC was equipotent at both receptor subtypes (IC50 values: 9.9 vs 10.2 nmol/L, P>0.05), while ROC was more potent at γ-AChR than ɛ-AChR (IC50 values: 22.3 vs 33.5 nmol/L, P<0.05).
Conclusion:
Magnitude differences of resistance to different NDMRs caused by denervation are associated with distinct potencies of NDMRs at nAChR subtypes.
Keywords: acetylcholine, atracurium, denervation, nicotinic acetylcholine receptor, rocuronium, nondepolarizing muscle relaxants, vecuronium
Introduction
Thermal injury and other forms of critical illness can cause denervation of skeletal muscle1. Denervation in turn causes pharmacological changes in skeletal muscle, with the main changes leading to potentially lethal hyperkalemia in response to the depolarizing muscle relaxant succinylcholine and resistance to nondepolarizing muscle relaxants (NDMRs), typified by d-tubocurarine (dTC)2, 3. Up-regulation (increase) of nicotinic acetylcholine receptors (nAChR) explains some of these pharmacological responses4, 5.
nAChRs include two subtypes: the adult form (ɛ-AChR) composed of α2βδɛ subunits and the fetal form (γ-AChR) containing α2βδγ subunits6, 7. γ-AChR is initially expressed in fetal muscle, and then replaced by ɛ-AChR when muscles are innervated. However, after denervation of adult muscles, γ-AChR is expressed de novo throughout the muscle membrane8. Clinically-used NDMRs competitively occupy the αδ and αɛ (or αγ) sites of the nAChR to achieve muscle relaxation during surgery and artificial ventilation. Denervation causes up-regulation of nAChRs and AChRγ mRNA5, 9 to increase the doses of NDMRs needed to competitively bind to nAChR. Also, expression of γ-AChR after denervation has clinical significance, because the two subtypes have different electrophysiological and drug-binding properties10.
Previous data have confirmed that denervated skeletal muscle displays resistance to NDMRs5, 11 and that potencies of different NDMRs are different at ɛ-AChR and γ-AChR12, 13, 14. Interestingly, Narimatsu et al15 observed that the magnitude of sepsis-induced attenuation of potencies of NDMRs depends on the kind of neuromuscular blocker.
The hypothesis of this study was that denervation would cause different magnitudes of resistance to different NDMRs due to their varying potencies at nAChR subtypes. The aim of this study was to test differences in the magnitude of resistance to vecuronium, atracurium, and rocuronium, which are commonly used NDMRs, in denervated skeletal muscle. Additionally, we examined the potencies of vecuronium, atracurium, and rocuronium at ɛ-AChR and γ-AChR expressed heterologously in human embryonic kidney 293 (HEK293) cells.
Materials and methods
Denervation
Balb/C mice (35 d old) were anesthetized with pentobarbital 40 mg/kg ip. A few millimeters of the right sciatic nerve was excised through a small (3–5 mm) incision over the hip. The incision was sutured with a single stitch. The left leg of each mouse served as the innervated control. The animals were killed 7 d after denervation by pentobarbital anesthesia and cervical dislocation. Animal welfare was monitored by the Animal Care and Use Committee of the Shanghai Jiaotong University School of Medicine and the research was performed in compliance with the WHO International Guiding Principles for Animal Research.
Isolation of muscle fibres
Single skeletal muscle cells from the flexor digitorum brevis (FDB) muscle were obtained from the hindfeet of each mouse. The muscles were incubated for 3 h with Dulbecco's modified Eagle's medium (DMEM) (InvitrogenTM, Grand Island, NY, USA) containing 10% fetal calf serum (InvitrogenTM, Grand Island, NY, USA), 100 μg/mL penicillin-streptomycin, and 0.2% collagenase 1A (Sigma Chemical Co, Saint Louis, USA) in a shaking bath at 37 °C. After 3 h of enzymatic treatment, the muscles were dissociated into single muscle cells using Pasteur pipettes with different tip sizes. Fibre length and width were measured using an ocular micrometer with 100 μm divisions. The fibres were viewed with a 40× objective, producing an optical resolution of 2.5 μm between the divisions. Half the distance between the divisions was easily resolved, giving a measurement error of ±1.25 μm.
Expression of nAChR in HEK293 cells
Expression plasmids Psp65α, Psp65β, Psp65δ, Psp64γ, and Pbssk(+)ɛ, encoding complementary DNA coding sequences for the mouse muscle nAChR subunits α, β, δ, γ, and ɛ, respectively, were provided by the Salk Institute in America. These plasmids were subcloned in pcDNA3.1+ (InvitrogenTM Life Technologies, Carlsbad, CA, USA). HEK293 cells were cultured in Dulbecco's modified Eagle's medium supplemented with 10% calf serum, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37 °C in a 5% CO2/95% O2 incubator. The HEK293 cells were transfected stably with LipofectamineTM 2000 according to the manufacturer's protocol (InvitrogenTM Life Technologies, Carlsbad, CA, USA). After transfection, the positive cell clones were selected with G418. The transfected cells were then incubated for 24 h before recording.
Electrophysiology
A conventional patch clamp technique with a whole cell configuration was used16. All experiments were performed at room temperature (20–24 °C). Patch pipettes were pulled from borosilicate glass using a Flaming Brown micropipette puller (P97; Sutter Instrument Co, Novato, CA, USA), ranging from 1 to 2 MΩ. The pipette electrode was filled with the following solution (mmol/L): 140 KCl, 10 HEPES, 10 EGTA, 1 CaCl2, and 1 MgCl2, and the pH was adjusted to 7.2 with KOH. The external solution contained (mmol/L): 5 KCl, 140 NaCl, 1 CaCl2, 1.25 MgCl2, 10 HEPES, and 10 glucose, with 0.5 μmol/L atropine sulfate, and pH was adjusted to 7.4 with NaOH. The cells were voltage-clamped at -80 mV in a whole-cell configuration after obtaining GΩ seals. 60%–80% of the series resistance was compensated for. Currents were recorded with an EPC10 amplifier (HEKA Elektronik, Germany) and PatchMaster software (HEKA Elektronik, Germany), sampled at 10 kHz or 20 kHz, and stored on a computer.
Acetylcholine (ACh) and atropine sulfate were purchased from Sigma (Saint Louis, USA). Muscle relaxants were obtained in preparations for clinical use: rocuronium, vecuronium (NV Organon, Netherlands), and atracurium (Jiangsu Hengrui Medicine Co, China). All drugs were dissolved in the external solution and applied by a gravity-driven perfusion system. Solutions and their dilutions to the experimental concentrations were prepared immediately before the experiments.
An agonist concentration of 30 μmol/L ACh was used for all experiments. Test solution containing either ACh 30 μmol/L alone or in combination with various concentrations of antagonist (NDMRs) were applied for 10 s to muscle cells and 5 s to HEK293 cells, and the peak current was determined. To determine the effect of the antagonist on the 30 μmol/L ACh-elicited current, the antagonist-containing solutions perfused the cells for 3 min prior to application of the solution of ACh 30 μmol/L plus the antagonist. The washout time between each drug application was at least 60 s in order to minimize the amount of desensitization throughout the course of an experiment. Currents were acquired from five muscle cells taken from five different mice or five HEK293 cells. The control current to ACh 30 μmol/L alone was repeated after washout of the antagonist. We took the mean value of these two ACh applications as the average control current, and calculated the antagonist response (percentage inhibition of average control current) using the equation:
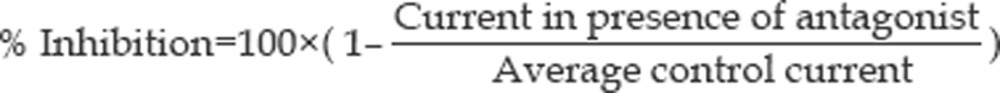 |
Statistical Analysis
Data analysis was performed off-line using Origin 8 (OriginLab, Northampton, MA, USA) and GraphPad Prism 4 (Graphpad software, Inc, San Diego, CA, USA). Concentration-response curves were fitted to the four-parameter logistic equation by nonlinear regression analysis, and the inhibition concentrations for the half-maximal responses (IC50) were determined. Results were represented as mean±SD. Statistical significance was assessed using one-way analysis of variance followed by Tukey's test and unpaired or paired two-tailed Student's t-tests. P<0.05 was considered significant.
Results
Effect of denervation on potency of different muscle relaxants
FDB muscle cells were chosen for this experiment due to their relatively small size for skeletal muscle cells, allowing for a better space clamp. The mean length and width of the muscle cells dissociated from innervated muscles were 394.2±8.5 and 17.1±0.8 μm (n=10), respectively. Denervation had minimal effect on muscle cell size (410.4±10.9 and 14.1±0.6 μm, respectively; n=10), with the small reduction in cells width being significant (P<0.05).
ACh at different concentrations (1–500 μmol/L) was applied for 10 s to innervated mouse skeletal muscle cells. ACh elicited concentration-dependent inward currents. The data were fitted to a logistic equation. The ACh concentrations producing 50% of the maximal response (EC50) were 27.1 μmol/L for the nAChR in the innervated skeletal muscle (data not shown). In all following experiments, a concentration of 30 μmol/L was used to activate nAChR because this concentration was close to the EC50 concentration and smaller than the estimated transient peak concentrations within the neuromuscular junction17, but it elicited sufficient current responses without significant desensitization after repetitive application.
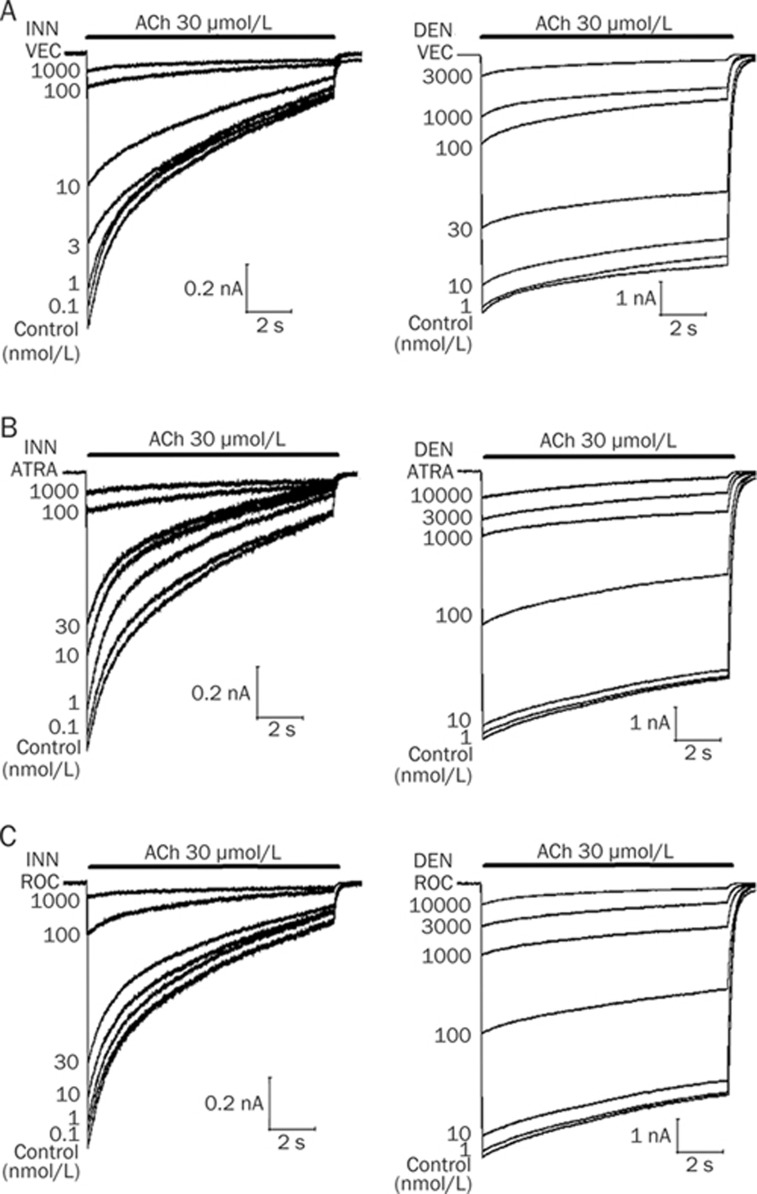
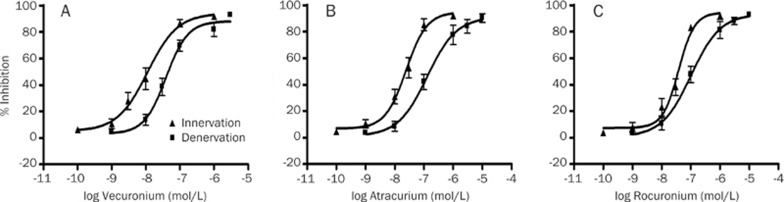
Next, we examined the effects of denervation on potency of the three clinically-used NDMRs by applying ACh to innervate or denervate skeletal muscle. We found that they reversibly inhibited ACh-elicited inward currents in a concentration-dependent manner (Figures 1 and 2). The rank order of potency using the IC50 concentrations for innervated skeletal muscle was vecuronium>atracurium>rocuronium and for denervated skeletal muscle was vecuronium>rocuronium∼atracurium. Compared with innervated skeletal muscle, in denervated skeletal muscle the IC50 values of vecuronium, atracurium, and rocuronium increased 3.7-, 5.5-, and 2.8-fold, respectively. Therefore, the rank order of the magnitude of resistance to the three NDMRs in denervated skeletal muscle, assessed by looking at the incremental fold in the IC50 values, was atracurium>vecuronium∼rocuronium (Table 1).
Figure 1.
Concentration-dependent effects of vecuronium (VEC) (A), atracurium (ATRA) (B), and rocuronium (ROC) (C) on the nicotinic acetylcholine receptors in innervated (INN) and denervated (DEN) skeletal muscle cells. Traces represent raw currents observed during the application of ACh 30 μmol/L for 10 s, either alone (control) or in combination with various concentrations of vecuronium, atracurium, or rocuronium, as indicated.
Figure 2.
Concentration-response effects of vecuronium (A), atracurium (B), and rocuronium (C) in inhibition of acetylcholine-induced currents in innervated and denervated skeletal muscle cells. Data points show the mean±SD (error bars) of five skeletal muscle cells. Error bars not visible are smaller than symbols.
Table 1. Potencies of nondepolarizing muscle relaxants at muscle nicotinic acetylcholine receptors in innervated and denervated skeletal muscles.
| Antagonist | IC50 nmol/L (95% CI) | P value | IC50 ratios | |
|---|---|---|---|---|
| Innervation | Denervation | |||
| Vecuronium | 11.2 (4.5–27.7) | 39.2 (26.3–52.6) | <0.01 | 3.66±0.95 |
| Atracurium | 24.4 (10.5–32.7) | 129.0 (81.0–152.7) | <0.01 | 5.52±1.48 |
| Rocuronium | 37.9 (23.3–61.3) | 101.4 (82.2–123.6) | <0.01 | 2.82±0.46 |
Potencies of nondepolarizing muscle relaxants at nicotinic acetylcholine receptors are expressed as the value achieving 50% of maximal inhibition (IC50) of the inward currents induced by coapplication of 30 μmol/L acetylcholine (95% confidence interval [CI]). Values are expressed as mean±SD in IC50 ratios [IC50 (denervation)/IC50 (innervation)]. For the innervation group, denervation group, and IC50 ratios, drug IC50 values or ratios that do not share a superscript were statistically different from each other using one-way analysis of variance and Tukey's multiple comparison method. Potency differences between the denervation and innervation groups were compared for each drug using paired two-tailed Student's t-tests (P values).
Potency of muscle relaxants on nAChR subtypes
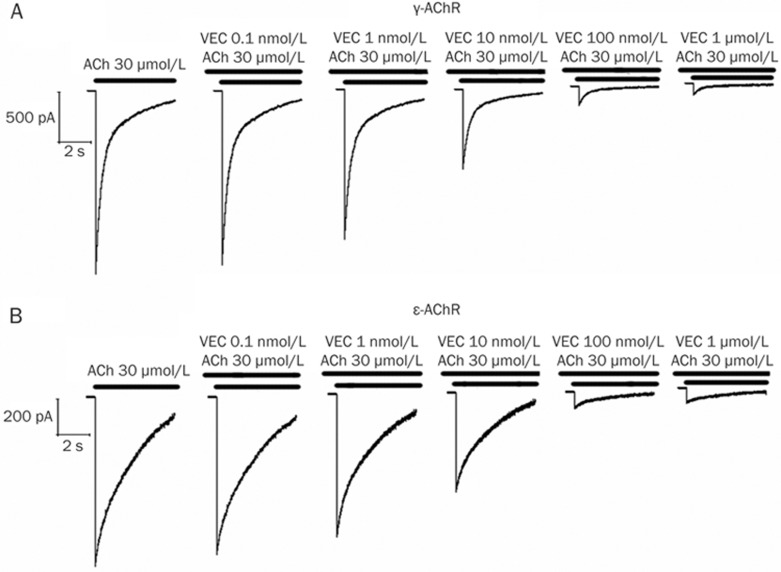
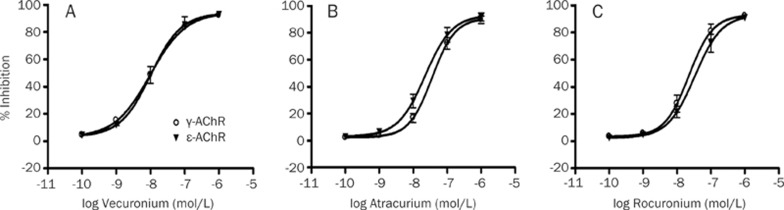
To further determine potency of the three NDMRs on nAChR subtypes, HEK293 cells were expressed with either ɛ- or γ-AChR, and then coapplied with ACh and each NDMR. We observed that all NDMRs reversibly inhibited the ACh-elicited currents in a concentration-dependent way (Figures 3 and 4). In a comparison of IC50 values, rocuronium was more potent at γ-AChR, but atracurium was more potent at ɛ-AChR; no significant difference was found for vecuronium. The rank order of potencies according to the estimated IC50 values for ɛ-AChR was vecuronium>atracurium>rocuronium and for γ-AChR was vecuronium>rocuronium>atracurium (Table 2). Together, these results suggest that differences of resistance to the NDMRs caused by denervation may be related to their potencies on nAChR subtypes.
Figure 3.
Concentration-dependent effects of vecuronium (VEC) on fetal-type (γ-AChR) (A) and adult-type (ɛ-AChR) (B) nicotinic acetylcholine receptors expressed in HEK293 cells. Traces represent raw currents observed during the application of ACh 30 μmol/L for 5 s, either alone or in combination with various concentrations of vecuronium, as indicated.
Figure 4.
Concentration-response effects of vecuronium (A), atracurium (B), and rocuronium (C) in inhibition of acetylcholine-induced currents in HEK293 cells expressing fetal-type (γ-AChR) and adult-type (ɛ-AChR) nicotinic acetylcholine receptors. Data points show the mean±SD (error bars) of five HEK293 cells. Error bars not visible are smaller than symbols.
Table 2. Potencies of nondepolarizing muscle relaxants at adult and fetal forms of the muscle nicotinic acetylcholine receptors (ɛ-AChR and γ-AChR) expressed in HEK293 cells.
| Antagonist | IC50 (nmol/L) | P value | |
|---|---|---|---|
| ɛ-nAChR | γ-nAChR | ||
| Vecuronium | 10.2 (8.7–11.9) | 9.9 (4.6–21.0) | >0.05 |
| Atracurium | 22.3 (14.8–33.7) | 35.9 (23.7–54.5) | <0.01 |
| Rocuronium | 33.5 (25.3–44.3) | 22.3 (17.5–28.5) | <0.05 |
Potencies of nondepolarizing muscle relaxants at nicotinic acetylcholine receptors are expressed as the value achieving 50% of maximal inhibition (IC50) of the inward currents induced by coapplication of 30 μmol/L acetylcholine. For each receptor subtype, drug IC50 values that do not share a superscript were statistically different from each other following one-way analysis of variance and Tukey's multiple comparison method. Potency differences between receptor subtypes were compared for each drug using unpaired two-tailed Student's t-tests (P values).
Discussion
In this study, we used a whole-cell patch clamp technique on acutely dissociated skeletal muscle cells and a HEK293 cell expression system to investigate the effect of denervation on the potency of vecuronium, atracurium, rocuronium, and the possible mechanisms. We used a holding voltage of -80 mV because the antagonistic effects of NDMRs are independent of holding voltages ranging from -100 to -40 mV12, 13.
We established our denervation model in mice18, 19. We obtained innervated and denervated skeletal muscle cells 7 d after denervation. Compared with the values obtained from innervated skeletal muscle cells, the IC50 values of vecuronium, atracurium, and rocuronium in the denervated skeletal muscle cells were significantly larger. This indicates that denervation leads to resistance to NDMRs in skeletal muscle, a finding that has been reported by others5, 11. A possible mechanism for this resistance to NDMRs is that skeletal muscle denervation causes up-regulation of mature and immature nAChRs on the muscle membrane9, 20, which would increase the amount of NDMR required to competitively block ACh10. Ibebunjo and Martyn2, 5 reported that total (mature and immature) AChR protein on the tibialis muscle increased 5.4-fold (P<0.05) compared to the contralateral control tibialis on d 7 after a burn. In this study, the IC50 values of vecuronium, rocuronium, and atracurium in denervated muscle cells increased 3.7-, 2.8-, and 5.5-fold, respectively, compared with innervated muscle cells.
We also observed that IC50 ratios in innervation and denervation were different among the tested NDMRs, thus indicating that denervation caused different magnitudes in potency of the tested NDMRs. Comparing IC50 ratios, we found that post-denervation resistance to NDMRs was greater to atracurium than to vecuronium or rocuronium. We presumed that this different magnitude of resistance to NDMRs is associated with the presence of γ-AChR because there are different potencies of NDMRs on γ-AChR and ɛ-AChR12, 13, 14. So, we studied the action of the tested NDMRs at γ- and ɛ-AChR expressed in HEK293 cells and observed that rocuronium had greater potency at γ-AChR than at ɛ-AChR; vecuronium had equipotent effects at both receptor subtypes; and atracurium was less potent at γ-AChR than at ɛ-AChR. Thus, we deemed that after denervation, resistance to atracurium was greater, which is consistent with the known lesser potency of atracurium at γ-AChR compared with the potency at ɛ-AChR, and resistance to rocuronium was less, which again is consistent with a greater potency at γ-AChR than at ɛ-AChR.
Knowing the different magnitudes of resistance to NDMRs in denervated skeletal muscle is important for clinicians. Denervation causes a larger dose requirement to muscle relaxants to achieve desired clinical effect, but this requirement to different muscle relaxants is variable; therefore, monitoring of neuromuscular function is clinically essential to specifically determine the dose requirement of muscle relaxants and help reduce potential adverse events.
In summary, this study demonstrates that denervation leads to different magnitudes of resistance to NDMRs in mouse skeletal muscle. We observe different potencies of vecuronium, atracurium, and rocuronium at ɛ-AChR and γ-AChR expressed in HEK293 cells. Our findings suggest that the phenomenon of different magnitudes of resistance to NDMRs in denervated mouse skeletal muscle is due to the different potencies of individual NDMRs at nAChR subunits.
Author contribution
Hong WANG and Shi-tong LI designed the research; Hong WANG and Bin YANG performed the research; Yong-fu XU and Tao YAN analyzed the data; Hong WANG and Shi-tong LI wrote the paper.
Acknowledgments
This work was supported by National Natural Science Foundation of China (No 30571796).
Glossary
- NDMRs
nondepolarizing muscle relaxants
- nAChR
nicotinic acetylcholine receptor
- γ-AChR
fetal-type nicotinic acetylcholine receptor
- ɛ-AChR
adult-type nicotinic acetylcholine receptor
- IC50
half-maximal response
- dTC
d-tubocurarine
- HEK293
human embryonic kidney 293
- FDB
flexor digitorum brevis
- DMEM
Dulbecco's modified Eagle's medium
- ACh
Acetylcholine
- INN
innervated
- DEN
denervated
- VEC
vecuronium
- ATRA
atracurium
- ROC
rocuronium.
References
- Kim C, Martyn J, Fuke N. Burn injury to trunk of rat causes denervation-like responses in the gastrocnemius muscle. J Appl Physiol. 1988;65:1745–51. doi: 10.1152/jappl.1988.65.4.1745. [DOI] [PubMed] [Google Scholar]
- Ibebunjo C, Martyn JA. Thermal injury induces greater resistance to d-tubocurarine in local rather than in distant muscles in the rat. Anesth Analg. 2000;91:1243–9. doi: 10.1097/00000539-200011000-00036. [DOI] [PubMed] [Google Scholar]
- Yanez P, Martyn JA. Prolonged d-tubocurarine infusion and/or immobilization causes upregulation of acetylcholine receptors and hyperkalemia to succinylcholine. Anesthesiology. 1996;84:384–91. doi: 10.1097/00000542-199602000-00017. [DOI] [PubMed] [Google Scholar]
- Nosek MT, Martyn JA. Na+ channel and acetylcholine receptor changes in muscle at sites distant from burns do not simulate denervation. J Appl Physiol. 1997;82:1333–9. doi: 10.1152/jappl.1997.82.4.1333. [DOI] [PubMed] [Google Scholar]
- Hogue CW, Jr., Itani MS, Martyn JA. Resistance to d-tubocurarine in lower motor neuron injury is related to increased acetylcholine receptors at the neuromuscular junction. Anesthesiology. 1990;73:703–9. doi: 10.1097/00000542-199010000-00016. [DOI] [PubMed] [Google Scholar]
- Mishina M, Takai T, Imoto K, Noda M, Takahashi T, Numa S, et al. Molecular distinction between fetal and adult forms of muscle acetylcholine receptor. Nature. 1986;321:406–11. doi: 10.1038/321406a0. [DOI] [PubMed] [Google Scholar]
- Gu Y, Hall ZW. Characterization of acetylcholine receptor subunits in developing and in denervated mammalian muscle. J Biol Chem. 1988;263:12878–85. [PubMed] [Google Scholar]
- Witzemann V, Brenner HR, Sakmann B. Neural factors regulate AChR subunit mRNAs at rat neuromuscular synapses. J Cell Biol. 1991;114:125–41. doi: 10.1083/jcb.114.1.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma J, Shen J, Garrett JP, Lee CA, Li Z, Elsaidi GA, et al. Gene expression of myogenic regulatory factors, nicotinic acetylcholine receptor subunits, and GAP-43 in skeletal muscle following denervation in a rat model. J Orthop Res. 2007;25:1498–505. doi: 10.1002/jor.20414. [DOI] [PubMed] [Google Scholar]
- Martyn JA, White DA, Gronert GA, Jaffe RS, Ward JM. Up-and-down regulation of skeletal muscle acetylcholine receptors. Effects on neuromuscular blockers. Anesthesiology. 1992;76:822–43. doi: 10.1097/00000542-199205000-00022. [DOI] [PubMed] [Google Scholar]
- Han T, Kim H, Bae J, Kim K, Martyn JA. Neuromuscular pharmacodynamics of rocuronium in patients with major burns. Anesth Analg. 2004;99:386–92. doi: 10.1213/01.ANE.0000129992.07527.4B. [DOI] [PubMed] [Google Scholar]
- Fletcher GH, Steinbach JH. Ability of nondepolarizing neuromuscular blocking drugs to act as partial agonists at fetal and adult mouse muscle nicotinic receptor. Mol Pharmacol. 1996;49:938–47. [PubMed] [Google Scholar]
- Garland CM, Foreman RC, Chad JE, Holden-Dye L, Walker RJ. The actions of muscle relaxants at nicotinic acetylcholine receptor isoforms. Eur J Pharmacol. 1988;357:83–92. doi: 10.1016/s0014-2999(98)00542-1. [DOI] [PubMed] [Google Scholar]
- Paul M, Kindler CH, Fokt RM, Dresser MJ, Dipp NC, Yost CS. The potency of new muscle relaxants on recombinant muscle-type acetylcholine receptors. Anesth Analg. 2002;94:597–603. doi: 10.1097/00000539-200203000-00022. [DOI] [PubMed] [Google Scholar]
- Narimatsu E, Niiya T, Kawamata M, Namiki A. Sepsis stage dependently and differentially attenuates the effects of nondepolarizing neuromuscular blockers on the rat diaphragm in vitro. Anesth Analg. 2005;100:823–9. doi: 10.1213/01.ANE.0000145010.60144.ED. [DOI] [PubMed] [Google Scholar]
- Hamill OP, Marty A, Neher E, Sakmann B, Siqworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Kuffler SW, Yoshikami D. The number of transmitter molecules in a quantum: an estimate from iontophoretic application of acetylcholine at the neuromuscular synapse. J Physiol. 1975;251:465–82. doi: 10.1113/jphysiol.1975.sp011103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waqatsuma A, Osawa T. Time course of changes in angiogenesis-related factors in denervated muscle. Acta Physiol (Oxf) 2006;187:503–9. doi: 10.1111/j.1748-1716.2006.01594.x. [DOI] [PubMed] [Google Scholar]
- Lauritzen HP, Ploug T, Ai H, Donsmark M, Prats C, Galbo H. Denervation and high-fat diet reduce insulin signaling in T-tubules in skeletal muscle of living mice. Diabetes. 2008;57:13–23. doi: 10.2337/db07-0516. [DOI] [PubMed] [Google Scholar]
- Adams L, Carlson BM, Henderson L, Goldman D. Adaptation of nicotinic acetylcholine receptor, myogenin, and MRF4 gene expression to long-term muscle denervation. J Cell Biol. 1995;131:1341–9. doi: 10.1083/jcb.131.5.1341. [DOI] [PMC free article] [PubMed] [Google Scholar]