Abstract
Objectives
The purpose of this study was to determine the relationship between carbohydrate antigen (CA) 19-9 levels and outcome in patients with borderline resectable pancreatic cancer treated with neoadjuvant therapy (NT).
Methods
This study included all patients with borderline resectable pancreatic cancer, a serum CA 19-9 level of ≥40 U/ml and bilirubin of ≤2 mg/dl, in whom NT was initiated at one institution between 2001 and 2010. The study evaluated the associations between pre- and post-NT CA 19-9, resection and overall survival.
Results
Among 141 eligible patients, CA 19-9 declined during NT in 116. Following NT, 84 of 141 (60%) patients underwent resection. For post-NT resection, the positive predictive value of a decline and the negative predictive value of an increase in CA 19-9 were 70% and 88%, respectively. The normalization of CA 19-9 (post-NT <40 U/ml) was associated with longer median overall survival among both non-resected (15 months versus 11 months; P = 0.022) and resected (38 months versus 26 months; P = 0.020) patients. Factors independently associated with shorter overall survival were no resection [hazard ratio (HR) 3.86, P < 0.001] and failure to normalize CA 19-9 (HR 2.13, P = 0.001).
Conclusions
The serum CA 19-9 level represents a dynamic preoperative marker of tumour biology and response to NT, and provides prognostic information in both non-resected and resected patients with borderline resectable pancreatic cancer.
Introduction
Serum carbohydrate antigen (CA) 19-9 is the most common tumour marker used in the clinical management of patients with pancreatic ductal adenocarcinoma (PDAC).1–5 Among patients with radiographically resectable PDAC, CA 19-9 has been used as an indicator of clinically occult metastatic disease,5 as a criterion for the use of diagnostic laparoscopy prior to surgery,3,4 as a prognostic marker following resection with or without adjuvant therapy6,7 and as a director of therapy following postoperative recurrence.8 Among patients with unresectable, locally advanced or metastatic disease, CA 19-9 has been used as a prognostic marker for survival, as well as an indicator of response to palliative therapies.1,7,9
Borderline resectable PDAC, irrespective of the radiographic criteria used to define it, represents a challenging subset of cancer manifestations with local tumour anatomy that lies between the clearly resectable and unresectable.10–12 Because patients with borderline resectable cancers are at high risk for margin-positive resection and early disease progression following surgery, patients with this stage of disease are routinely administered neoadjuvant therapy (NT) prior to planned resection.10,12–15 The goals of this treatment strategy include the achievement of local control of the primary tumour, sterilization of lymphadenopathy and surgical margins, early treatment of presumed micro-metastatic disease, and the evaluation of tumour biology prior to a potentially morbid abdominal operation.12,14
Although there is consensus that the use of chemotherapy and/or chemoradiation prior to surgical resection is rational in patients with borderline resectable PDAC, few data exist to guide the details of such management.10,14 The relative importance of chemotherapy and chemoradiation, the ideal sequence and duration of their administration, and the precise cytotoxic agents to be used in the individual patient are all unclear. In this regard, one of the primary obstacles to the assessment and optimization of multimodal treatment strategies is the absence of a well-defined clinical indicator of response to preoperative therapy. An inexpensive, reproducible and easily measured response metric would be clinically useful to evaluate the efficacy of different preoperative regimens, to determine the optimal duration of each component of multimodal therapy and to estimate prognosis following treatment.
The current authors previously demonstrated that the response of PDAC to NT can be estimated by histopathologic examination of the resected primary tumour. Fibrosis and a marked reduction in the residual viable cancer cells following the administration of NT were associated with a favourable prognosis and may reflect clinically significant cytotoxic activity.16 However, because a histologic evaluation of tumour response requires an examination of tumour cells and tissue architecture, this dynamic measure of tumour response is unavailable in the preoperative setting. Furthermore, although the change in tumour size as determined radiographically and as classified using RECIST (response evaluation criteria in solid tumours) parameters can be used to estimate response to therapy in patients with many types of cancer, the current authors have previously demonstrated that radiographic ‘response’ is rare among patients with borderline resectable PDAC treated with NT and, further, when observed, it is not associated with a favourable outcome among patients who undergo subsequent resection.17
Using data from two clinical trials of neoadjuvant chemoradiation, the current authors previously demonstrated that serum CA 19-9 levels can provide some clinically relevant prognostic information in patients with potentially resectable PDAC treated with NT.2,18–20 On this basis, the present study was conducted to verify the hypothesis that a change in CA 19-9 during NT can be used as a surrogate marker of tumour biology and response to therapy in patients with borderline resectable PDAC. The first objective of this analysis was to evaluate the change in serum CA 19-9 from baseline following NT in patients treated for borderline resectable PDAC. The primary aim was to evaluate the association between CA 19-9 levels and survival in this important group of patients.
Materials and methods
Patient selection and definitions
This study was approved by the institutional review board of the University of Texas MD Anderson Cancer Center (MDACC). The institution's prospectively maintained pancreatic translational database21 was queried for all patients with PDAC who were treated with neoadjuvant chemotherapy and/or chemoradiation and subsequently underwent restaging and consideration for surgery during the period 2001–2010. Strict anatomic staging criteria were assessed by multi-detector, contrast-enhanced computed tomography (CT).12,17 Included in this study were all patients who presented with primary tumour anatomy that met either MDACC criteria11,12 or those of the Americas Hepato-Pancreato-Biliary Association (AHPBA), the Society of Surgical Oncology (SSO) and the Society for Surgery of the Alimentary Tract (SSAT)10 for borderline resectable PDAC (Table 1), in whom serum CA 19-9 was assayed both prior and subsequent to the delivery of NT, and in whom an abnormal CA 19-9 level (defined as ≥40 U/ml) prior to treatment was established. Patients with baseline CA 19-9 levels obtained in association with a total bilirubin level of >2 mg/dl were excluded as biliary obstruction can lead to an artificial increase in serum CA 19-9 (Fig. 1).22
Table 1.
Classifications of localized pancreatic adenocarcinoma as defined by the Americas Hepato-Pancreato-Biliary Association (AHPBA), the Society of Surgical Oncology (SSO) and the Society for Surgery of the Alimentary Tract (SSAT), and MD Anderson Cancer Center. Adapted from Katz et al. (2012)17
| AHPBA/SSO/SSAT10 |
MD Anderson Cancer Center11,12 |
|||||
|---|---|---|---|---|---|---|
| Potentially resectable | Borderline resectable | Locally advanced | Potentially resectable | Borderline resectable | Locally advanced | |
| SMV/PV | No abutmenta or encasementb | Abutment, encasement or occlusion | Not reconstructible | Abutment or encasement without occlusion | Short segment occlusion | Not reconstructible |
| SMA | No abutment or encasement | Abutment | Encasement | No abutment or encasement | Abutment | Encasement |
| CHA | No abutment or encasement | Abutment or short segment encasement | Long segment encasement | No abutment or encasement | Abutment or short segment encasement | Long segment encasement |
| CT | No abutment or encasement | No abutment or encasement | Abutment | No abutment or encasement | Abutment | Encasement |
≤180 degrees of vessel circumference.
>180 degrees of vessel circumference.
SMV, superior mesenteric vein; PV, portal vein; SMA, superior mesenteric artery; CHA, common hepatic artery; CT, coeliac trunk.
Figure 1.
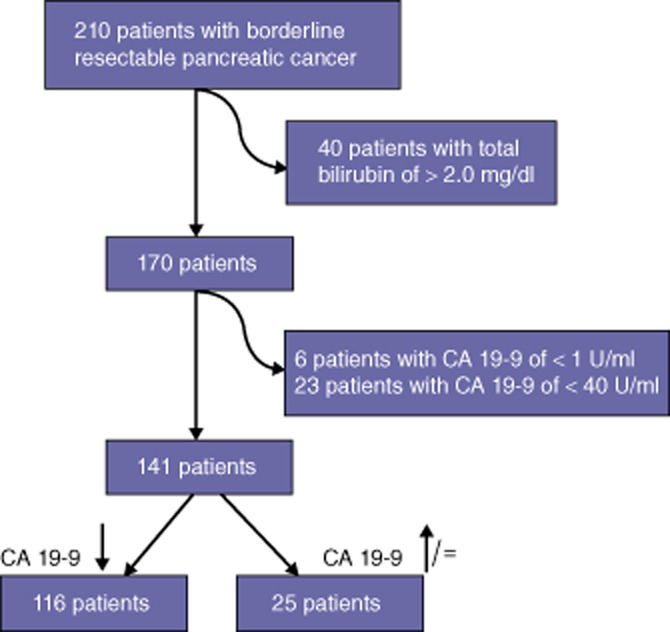
Flow diagram of patients selected for this study. CA 19-9, carbohydrate antigen 19-9 (serum)
Neoadjuvant therapy and treatment schema
The current authors routinely treated patients with potentially or borderline resectable PDAC with chemoradiation prior to surgical resection. Systemic chemotherapy was delivered selectively prior to chemoradiation, most commonly to patients meeting MDACC anatomic or clinical criteria for borderline resectable disease.11,12,18,20 Biliary decompression was generally accomplished with a metal endobiliary stent prior to the initiation of therapy.
The patients reported in this study received chemoradiation based on gemcitabine or 5-fluorouracil with or without induction gemcitabine-based systemic chemotherapy in the neoadjuvant setting.19,20,23 Prior to (‘pre-NT’) and following (‘post-NT’) the administration of NT, patients underwent a comprehensive staging evaluation that routinely included a measurement of serum CA 19-9. Serum samples acquired prior to 6 October 2004 were assayed for CA 19-9 by Quest Diagnostics, Inc. (Madison, NJ, USA). Subsequent samples were assayed using a similar method at MDACC.
Criteria for surgical resection following completion of NT included: (i) a performance status sufficient for major abdominal surgery; (ii) the adequate optimizing of comorbidities, and (iii) no radiographic or intraoperative detection of tumour progression.18 Serum CA 19-9 was not used as a primary criterion for surgery.
Follow-up
Following resection, patients were evaluated in an intensive surveillance programme every 3–4 months, with an institutional bias toward aggressive treatment of recurrence.24,25 Patients who did not undergo resection following the receipt of NT generally received palliative gemcitabine-based chemotherapy during this period.
Statistical analyses
Chi-squared and Fisher's exact tests were used to compare non-parametric categorical variables. The Mann–Whitney U-test was used to compare non-parametric continuous variables. A receiver operating characteristic (ROC) curve was constructed to evaluate the relationship between CA 19-9 level and the prediction of resection versus no resection after NT. The area under the curve (AUC) was calculated for pre-NT CA 19-9 and for post-NT CA 19-9 to compare the relative value of each test. Optimal cut-off values were determined graphically to maximize sensitivity and specificity simultaneously. Overall survival (OS) was defined as the interval between tissue diagnosis and death or last follow-up. Durations of OS associated with various CA 19-9 cut-off values were estimated using the Kaplan–Meier method and compared using the log-rank test. Clinically significant univariate factors were included in multivariate Cox regression models, as were statistically significant (P < 0.05) univariate factors associated with OS to determine independent associations with OS. Statistical analyses were performed using spss Statistics Version 19 (IBM Corp., Armonk, NY, USA). All tests were two-sided; differences of statistical significance were indicated by a P-value of <0.05.
Results
Patients and treatment outcomes
During the study period, 210 patients presented with a primary tumour that met MDACC or AHPBA/SSO/SSAT anatomic criteria for borderline resectable PDAC and with serum CA 19-9 levels measured both before and after the delivery of NT. Of these, 40 patients were excluded because their pre-NT CA 19-9 levels were associated with jaundice. An additional 29 of the remaining 170 (17%) patients had normal or undetectable (n = 6) pre-NT CA 19-9 levels and were excluded.
Of the remaining 141 patients evaluated in this study, 84 (60%) underwent pancreatectomy after NT. The demographics and baseline clinical profiles of resected and non-resected patients were similar (Table 2). Median follow-up in resected and non-resected patients was 24.2 months (range: 5–113 months) and 12.0 months (range: 3–77 months), respectively. The median durations of OS in all patients, resected patients and non-resected patients were 19.1 months [95% confidence interval (CI) 13.9–24.3 months], 30.9 months (95% CI 26.8–35.0 months) and 12.0 months (95% CI 9.3–14.7 months), respectively (P < 0.001). Detection of metastases at restaging or at surgery was the most common reason for failure to undergo pancreatectomy following NT (37 of 57 patients, 65%) (Table 2).
Table 2.
Clinicopathologic profile of patients with borderline resectable pancreatic adenocarcinoma, non-resected and resected after neoadjuvant therapy (NT)
| Clinical characteristic | All patients | Patients not resected after NT | Patients resected after NT | P-value |
|---|---|---|---|---|
| Patients, n | 141 | 57 | 84 | |
| Baseline data (pre-NT) | ||||
| Age, years, median (range) | 63 (34–81) | 66 (38–81) | 63 (35–79) | 0.244 |
| Sex, female, n (%) | 74 (52.5%) | 25 (43.9%) | 49 (58.3%) | 0.091 |
| Race, White, n (%) | 115 (81.6%) | 48 (84.2%) | 67 (79.8%) | 0.611 |
| Head/uncinate (versus body/tail), n (%) | 126 (89.4%) | 52 (91.2%) | 74 (88.1%) | 0.554 |
| CA 19-9, U/ml, median (range) | 274 (42–8662) | 368 (46–5687) | 211 (42–8662) | 0.020 |
| CA 19-9 < 100, n (%) | 34 (24.1%) | 11 (19.3%) | 23 (27.4%) | 0.271 |
| CA 19-9 < 200, n (%) | 61 (43.3%) | 21 (36.8%) | 40 (47.6%) | 0.205 |
| CA 19-9 < 1000, n (%) | 113 (80.1%) | 36 (63.2%) | 77 (91.7%) | <0.001 |
| Post-NT serum CA 19-9 data | ||||
| CA 19-9, median (range) | 73 (1–17171) | 222 (9–17171) | 49 (1–2908) | <0.001 |
| CA 19-9 decrease (any %), n (%) | 116 (82.3%) | 35 (61.4%) | 81 (96.4%) | <0.001 |
| CA 19-9 of <40 U/ml (normalized), n (%) | 47 (33.3%) | 12 (21.1%) | 35 (41.7%) | 0.011 |
| CA 19-9 of <100 U/ml, n (%) | 81 (57.4%) | 17 (29.8%) | 64 (76.2%) | <0.001 |
| CA 19-9 of <200 U/ml, n (%) | 101 (71.6%) | 27 (47.4%) | 74 (88.1%) | <0.001 |
| CA 19-9 of <1000 U/ml, n (%) | 125 (88.7%) | 42 (73.7%) | 83 (98.8%) | <0.001 |
| Reason for non-resection, n (%) | ||||
| Metastases | 37 (64.9%) | |||
| Local anatomy | 13 (22.8%) | |||
| Performance status | 12 (12.3%) | |||
| Postoperative characteristics, n (%) | ||||
| Tumour size >2 cm | 40 (47.6%) | |||
| Lymph node negative | 43 (51.2%) | |||
| Lymph node ratio of <0.1 | 66 (78.6%) | |||
| Lymph node ratio of <0.15 | 74 (88.1%) | |||
| Margin negative | 77 (91.7%) | |||
| Complete pathologic response | 9 (10.7%) |
CA 19-9, carbohydrate antigen 19-9 (serum).
Serum CA 19-9 before and after NT
The median CA 19-9 level in non-resected patients was higher than that in resected patients both prior to (368 U/ml versus 211 U/ml; P = 0.020) and following (222 U/ml versus 49 U/ml; P < 0.001) the administration of NT (Table 2). Serum CA 19-9 in 116 of 141 (82%) patients declined over the course of NT by a median of 72% (range: 0.3–99%) and normalized in 47 (33%) patients. Serum CA 19-9 in 25 (18%) patients increased by a median of 109% (range: 0–564%) or did not change (n = 1) (Fig. 2).
Figure 2.
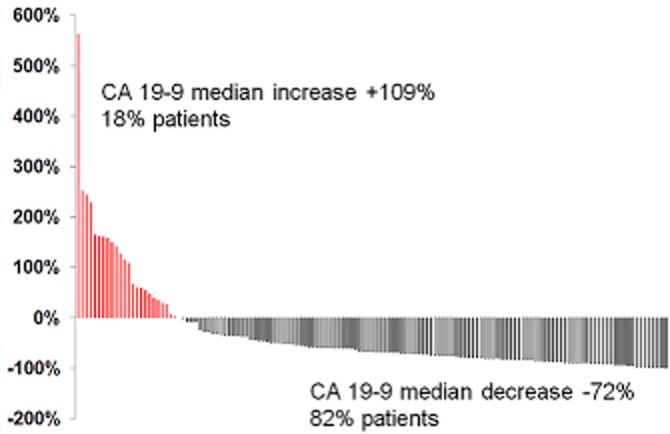
Pattern of carbohydrate antigen (CA) 19-9 change after neoadjuvant therapy showing that 82% of patients experienced a decrease in CA 19-9
Association between CA 19-9 change and resection status
The post-NT CA 19-9 level was more informative as a predictor of failure to undergo pancreatectomy than the pre-NT level based on ROC analysis (Fig. 3). For predicting resection after NT, the AUCs for pre-NT and post-NT CA 19-9 levels were 0.62 (95% CI 0.52–0.71; P < 0.020) and 0.78 (95% CI 0.70–0.86; P < 0.001) with optimum cut-off values of 989 U/ml (pre-NT) and 106 U/ml (post-NT), respectively.
Figure 3.
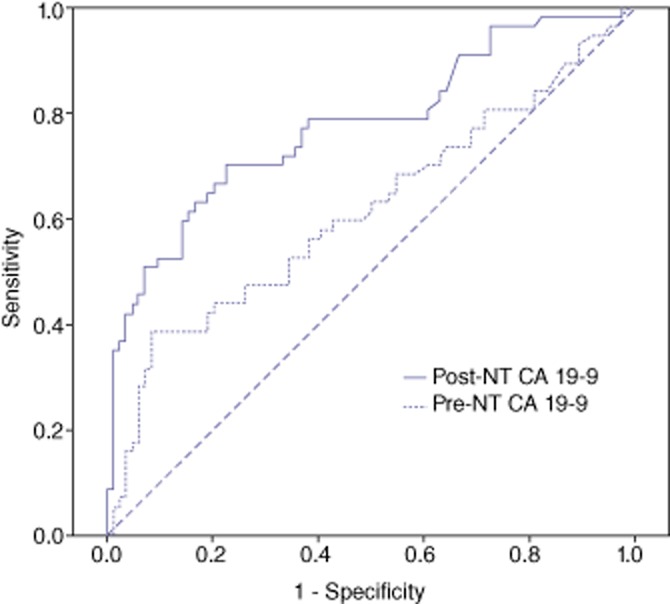
Receiver operating characteristic curves displaying associations of serum levels of carbohydrate antigen (CA) 19-9 before and after neoadjuvant therapy (NT) with resection after NT. The areas under the curve for pre-NT and post-NT CA 19-9 levels were 0.62 [95% confidence interval (CI) 0.52–0.71] and 0.78 (95% CI 0.70–0.86) with optimum cut-off values of 989 U/ml and 106 U/ml, respectively
In all, 81 of 116 patients in whom CA 19-9 decreased during NT underwent resection [positive predictive value (PPV): 70%] and 22 of 25 patients in whom CA 19-9 increased did not undergo resection [negative predictive value (NPV): 88%].
Change in CA 19-9 as a prognostic marker of OS
At post-NT CA 19-9 cut-offs of 1000 U/ml, 200 U/ml and 100 U/ml, low CA 19-9 was associated with a longer median OS in non-resected but not in resected patients (Table 3). Normalization of CA 19-9 was associated with a longer median OS among both resected [37.9 months (95% CI 30.5–45.3) versus 26.0 months (95% CI 19.6–32.4); P < 0.020] and non-resected patients [15.0 months (95% CI 7.6–22.4) versus 11.0 months (95% 9.4–12.6); P = 0.022] (Fig. 4).
Table 3.
Post-neoadjuvant therapy (NT) serum carbohydrate antigen (CA) 19-9 cut-offs and association with overall survival (OS) in patients with borderline resectable pancreatic adenocarcinoma
| Reached cut-off |
Did not reach cut-off |
||||
|---|---|---|---|---|---|
| Median OS, months | 95% CI | Median OS, months | 95% CI | P-value | |
| CA 19-9 level after NT, U/ml | |||||
| Any CA 19-9 decrease (resected patients) | 30.4 | 25.0–35.8 | 32.0 | 11.4–52.6 | 0.510 |
| Any CA 19-9 decrease (non-resected patients) | 14.2 | 12.6–15.8 | 8.3 | 5.54–11.1 | 0.003 |
| CA 19-9 of <1000 U/ml (resected patients) | 30.9 | 26.8–35.0 | 15.4 | a | 0.102 |
| CA 19-9 of <1000 U/ml (non-resected patients) | 13.6 | 10.6–16.6 | 7.9 | 5.5–10.3 | 0.011 |
| CA 19-9 of <200 U/ml (resected patients) | 30.9 | 27.1–34.8 | 25.7 | 0–58.5 | 0.813 |
| CA 19-9 of <200 U/ml (non-resected patients) | 15.0 | 12.9–17.1 | 8.3 | 5.9–10.7 | 0.001 |
| CA 19-9 of <100 U/ml (resected patients) | 30.4 | 24.3–36.5 | 32.0 | 25.7–38.3 | 0.733 |
| CA 19-9 of <100 U/ml (non-resected patients) | 15.0 | 12.7–17.3 | 10.8 | 9.4–12.2 | 0.030 |
| CA 19-9 normalizationb (resected patients) | 37.9 | 30.5–45.3 | 26.0 | 19.6–32.4 | 0.020 |
| CA 19-9 normalizationb (non-resected patients) | 15.0 | 7.6–22.4 | 11.0 | 9.4–12.6 | 0.022 |
Only one patient in this cohort.
Normal CA 19-9: <40 U/ml.
95% CI, 95% confidence interval.
Figure 4.
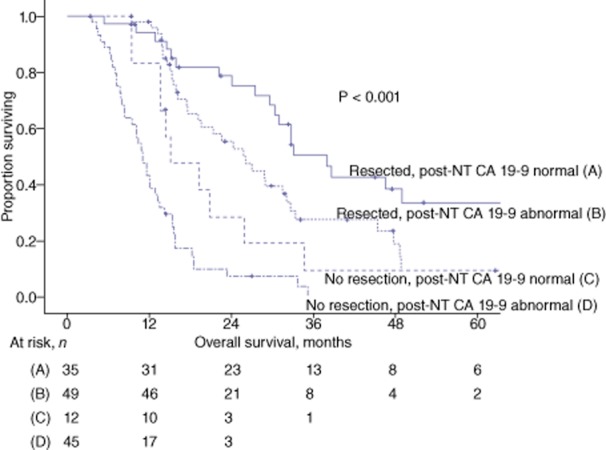
Kaplan–Meier estimates of survival in all patients stratified by resection status and carbohydrate antigen (CA) 19-9 normalization after neoadjuvant therapy (NT)
Multivariate survival model
For all patients, only resection and CA 19-9 normalization were significant covariates associated with OS. By multivariate analysis, both resection [hazard ratio (HR) 3.86, 95% CI 2.58–5.81; P < 0.001] and normalization of CA 19-9 (HR 2.13, 95% CI 1.37–3.22; P = 0.001) were independent factors associated with prolonged OS. Among resected patients, after adjusting for clinically relevant covariates (tumour size, margin status, lymph node status or ratio) which were entered into the multivariate analysis, only normalization of CA 19-9 remained independently associated with OS (HR 1.92, 95% CI 1.08–3.42; P = 0.027).
Discussion
The purpose of this study was to characterize the relationships between pre- and post-treatment serum CA 19-9 levels, and oncologic outcome following multimodal therapy in patients with borderline resectable PDAC. The data presented here suggest that the change in serum CA 19-9 that occurs during the administration of NT can be used clinically as a surrogate of treatment response and as a marker of longterm prognosis. Among 141 patients with borderline resectable PDAC who presented to MDACC with abnormal levels of CA 19-9 and who were treated with chemotherapy and/or chemoradiation prior to planned resection, CA 19-9 declined from baseline in 82% (116 of 141) and normalized in 33% (47 of 141) of patients. Failure of the CA 19-9 level to decline was an important clinical signal in that 88% (22 of 25) of patients with a stable or elevated CA 19-9 following NT did not complete resection, most commonly as a result of the interval development of metastatic disease. By contrast, CA 19-9 normalization was a favourable clinical indicator associated with prolonged OS in both resected and non-resected patients. These findings support the measurement of serum CA 19-9 both prior to and following the administration of NT in patients with borderline resectable PDAC, and emphasize its role as an easily assayed marker that provides insight into the biology of each patient's disease and the patient's likely longterm outcome following the completion of multimodal therapy.
Serum CA 19-9 is the most commonly assayed tumour marker in the clinical management of PDAC. Measurement of CA 19-9 has been incorporated into the clinical staging and treatment algorithms of both patients with localized and those with metastatic disease.3–6,22,26 Among patients with ostensibly localized cancers, CA 19-9 has been used primarily to predict resectability,26 to restrict preoperative laparoscopy to patients with the highest risk for radiographically occult metastatic disease,13,14 to predict the utility of adjuvant therapy after resection,6 and to predict the timing of postoperative recurrence.5 Several small studies have also investigated CA 19-9 levels in patients treated with chemotherapy or chemoradiation prior to surgery in an attempt to characterize the role of this marker in NT sequencing algorithms.27–29 Many of these studies, however, included heterogeneous populations of patients with advanced disease who were treated in the absence of a defined plan for resection. In a previous analysis of 174 patients with potentially resectable PDAC treated in two clinical trials of neoadjuvant chemoradiation, the current authors found that patients with low serum CA 19-9 both prior to and following the administration of NT had more favourable outcomes compared with those with high levels of CA 19-9, and that patients in whom CA 19-9 declined over the course of therapy had more favourable outcomes than those in whom CA 19-9 rose.2 However, the PPVs with respect to resection of both post-NT CA 19-9 and its change from the pre-NT level were similar to that derived from a standard radiologic and clinical restaging evaluation. Furthermore, both measures had a low NPV for the completion of resection: that is, a high post-NT level or rise in CA 19-9 during NT did not reliably exclude resection.
As in this group's previous study of potentially resectable patients, baseline serum CA 19-9 in patients with borderline resectable cancers prior to NT has less clinical relevance than the CA 19-9 level obtained following therapy or the change in levels over time. Indeed, in this study, the percentage of patients with an elevated CA 19-9 level at any tested cut-off value of <1000 U/ml who did not undergo resection did not differ from the percentage of patients who did undergo resection. By contrast, among historical patients with localized disease who undergo surgery first, an elevated CA 19-9 level is an important indicator of the presence of radiographically occult disease that precludes curative surgery. The difference in the clinical value of the pretreatment CA 19-9 in these two groups is likely to reflect the response of the cancer to the cytotoxic activity of drugs and radiation treatments administered in the preoperative setting, and a consequent alteration in the relationship between the baseline tumour marker and tumour burden over the course of therapy. The relative importance of the post-NT CA 19-9 level or its change in response to chemotherapy and/or chemoradiation administered in the preoperative setting is analogous to the use of carcinoembryonic antigen (CEA) in patients with colorectal cancer, in whom the post-treatment marker level is an important indicator of prognosis.30–32
In patients with borderline resectable PDAC, the CA 19-9 changes observed over the course of NT appear to provide insight into tumour biology and treatment response, which, unlike in other solid tumours, cannot be evaluated radiographically. In a previous study of 122 patients with borderline resectable PDAC, only 15 (12%) showed a change in tumour size sufficient to meet RECIST parameters for a treatment response following the administration of NT, and only one (1%) patient showed radiographic evidence of a reduction in vascular involvement sufficient to improve anatomic stage. Furthermore, the median duration of OS in patients who underwent resection in the absence of a radiographic ‘response’ was similar to that in patients whose tumours decreased in size.17 However, tumour response to therapy can be estimated histopathologically. In a study of 223 patients who received NT prior to resection of PDAC, the resection specimens of 42 patients contained minimal or no residual cancer. These patients with minimal or no residual cancer had higher rates of negative margins, lower rates of positive lymph nodes, and longer overall and recurrence-free survival than patients with more residual disease.16 Of course, this information can only be gathered after resection and thus cannot contribute to preoperative counselling.
One of the most important findings reported here is that the normalization of CA 19-9 following NT is a marker for improved OS in both patients who do and those who do not undergo resection. Indeed, among these patients with borderline resectable primary tumours, patients in whom serum CA 19-9 normalized and who subsequently underwent resection following NT had a median OS of 37.9 months. Even among non-resected patients CA 19-9 normalization was associated with a prolonged median survival relative to that in patients in whom CA 19-9 did not normalize, reflecting a tumour response to the chemotherapy and chemoradiation regimens used. Future studies should explore how CA 19-9 data can be incorporated into individualized treatment algorithms. For example, a persistent elevation in serum CA 19-9 following an initial course of therapy might be considered as an indication of the need to switch cytotoxic agents prior to surgery. It could also serve as a trigger for a staging laparoscopy prior to chemoradiation. The present authors have typically performed laparoscopy prior to surgery and have chosen not to laparoscope patients with borderline PDAC prior to the initiation of systemic chemotherapy as the identification of occult metastatic disease would not change the initial chemotherapy regimen. However, laparoscopies performed earlier in the treatment algorithm according to CA 19-9 findings might save patients from the morbidities associated with ineffective local therapies in the presence of extrapancreatic disease. In addition, the prognosis estimated by CA 19-9 might also be used in the planning of personalized postoperative surveillance strategies designed to optimize cost-effectiveness.24
It should be emphasized that although CA 19-9 is routinely assayed in all patients at the study institution, clinical staging is accomplished using defined clinical classifications based upon local tumour anatomy and crude assessments of tumour biology and patient condition and comorbidities.18 The preoperative regimens formulated by the multidisciplinary group are then delivered until completion or tumour progression. Finally, surgeons use objective indications for resection following the administration of NT, which include: (i) a performance status sufficient for major abdominal surgery; (ii) the adequate optimizing of comorbidities, and (iii) the absence of any radiographic or intraoperative detection of tumour progression. Although CA 19-9 is not generally used as a primary factor in decision making on the use of surgical resection following the completion of non-operative therapy, the possibility that CA 19-9 levels influenced decision making in some of the patients reported here cannot be entirely discounted.
Other strengths and limitations of this study also warrant discussion. Firstly, this retrospective analysis of patients treated at a single institution is subject to the biases and limitations inherent in any retrospective study. However, this study evaluated one of the largest reported cohorts of patients with borderline resectable PDAC in whom tumours were staged using objective radiographic criteria and classified using consensus guidelines. Follow-up in these patients was generally unbiased. This institution's prospective pancreatic cancer patient database has an overall follow-up rate of 92% attributable to a systematic intensive surveillance programme.25 Another potential limitation of this analysis is that 40 of 210 (19%) patients were excluded for reasons of hyperbilirubinaemia and the fact that CA 19-9 is not routinely checked after endobiliary stent placement. This is important because hyperbilirubinaemia can cause an elevation in CA 19-9 irrespective of tumour burden and might introduce significant error into analyses in which pre-NT CA 19-9 levels associated with and without jaundice are considered. This may account for the wide spectrum of pretreatment CA 19-9 cut-offs proposed in the surgical literature. The extent to which any change in CA 19-9 levels reflects tumour response and not biliary decompression depends on all CA 19-9 levels being measured in the absence of obstructive jaundice.
In conclusion, CA 19-9 decreases in four of five patients undergoing NT for borderline resectable PDAC. Although conventional radiographic ‘response’ criteria are of little value in the management of patients with this stage of disease, CA 19-9 appears to represent a dynamic marker of tumour biology and response to NT. A normalization of CA 19-9 following NT is associated with a favourable prognosis in both non-resected and resected patients with borderline resectable PDAC.
Acknowledgments
The authors thank Joel Cox for his management of the University of Texas MD Anderson Cancer Center departmental pancreatic surgery database.
Conflicts of interest
None declared.
Funding sources
Khalifa Bin Zayed Al Nahyan Foundation and the Various Donor Pancreatic Research Fund at MD Anderson Cancer Center. MHGK is supported in part by the 2012 Alliance for Clinical Trials in Oncology Clinical Scholar Award.
References
- Ballehaninna UK, Chamberlain RS. The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: an evidence-based appraisal. J Gastrointest Oncol. 2012;3:105–119. doi: 10.3978/j.issn.2078-6891.2011.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz MH, Varadhachary GR, Fleming JB, Wolff RA, Lee JE, Pisters PW, et al. Serum CA 19-9 as a marker of resectability and survival in patients with potentially resectable pancreatic cancer treated with neoadjuvant chemoradiation. Ann Surg Oncol. 2010;17:1794–1801. doi: 10.1245/s10434-010-0943-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maithel SK, Maloney S, Winston C, Gonen M, D'Angelica MI, Dematteo RP, et al. Preoperative CA 19-9 and the yield of staging laparoscopy in patients with radiographically resectable pancreatic adenocarcinoma. Ann Surg Oncol. 2008;15:3512–3520. doi: 10.1245/s10434-008-0134-5. [DOI] [PubMed] [Google Scholar]
- Satoi S, Yanagimoto H, Toyokawa H, Inoue K, Wada K, Yamamoto T, et al. Selective use of staging laparoscopy based on carbohydrate antigen 19-9 level and tumour size in patients with radiographically defined potentially or borderline resectable pancreatic cancer. Pancreas. 2011;40:426–432. doi: 10.1097/MPA.0b013e3182056b1c. [DOI] [PubMed] [Google Scholar]
- Sugiura T, Uesaka K, Kanemoto H, Mizuno T, Sasaki K, Furukawa H, et al. Serum CA 19-9 is a significant predictor among preoperative parameters for early recurrence after resection of pancreatic adenocarcinoma. J Gastrointest Surg. 2012;16:977–985. doi: 10.1007/s11605-012-1859-9. [DOI] [PubMed] [Google Scholar]
- Humphris JL, Chang DK, Johns AL, Scarlett CJ, Pajic M, Jones MD, et al. The prognostic and predictive value of serum CA 19.9 in pancreatic cancer. Ann Oncol. 2012;23:1713–1722. doi: 10.1093/annonc/mdr561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin LK, Wei L, Trolli E, Bekaii-Saab T. Elevated baseline CA 19-9 levels correlate with adverse prognosis in patients with early- or advanced-stage pancreas cancer. Med Oncol. 2012;29:3101–3107. doi: 10.1007/s12032-012-0278-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strobel O, Hartwig W, Hackert T, Hinz U, Berens V, Grenacher L, et al. Re-resection for isolated local recurrence of pancreatic cancer is feasible, safe, and associated with encouraging survival. Ann Surg Oncol. 2013;20:964–972. doi: 10.1245/s10434-012-2762-z. [DOI] [PubMed] [Google Scholar]
- Bernhard J, Dietrich D, Glimelius B, Hess V, Bodoky G, Scheithauer W, et al. Estimating prognosis and palliation based on tumour marker CA 19-9 and quality of life indicators in patients with advanced pancreatic cancer receiving chemotherapy. Br J Cancer. 2010;103:1318–1324. doi: 10.1038/sj.bjc.6605929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callery MP, Chang KJ, Fishman EK, Talamonti MS, William Traverso L, Linehan DC. Pretreatment assessment of resectable and borderline resectable pancreatic cancer: expert consensus statement. Ann Surg Oncol. 2009;16:1727–1733. doi: 10.1245/s10434-009-0408-6. [DOI] [PubMed] [Google Scholar]
- Varadhachary GR, Tamm EP, Abbruzzese JL, Xiong HQ, Crane CH, Wang H, et al. Borderline resectable pancreatic cancer: definitions, management, and role of preoperative therapy. Ann Surg Oncol. 2006;13:1035–1046. doi: 10.1245/ASO.2006.08.011. [DOI] [PubMed] [Google Scholar]
- Katz MH, Pisters PW, Evans DB, Sun CC, Lee JE, Fleming JB, et al. Borderline resectable pancreatic cancer: the importance of this emerging stage of disease. J Am Coll Surg. 2008;206:833–846. doi: 10.1016/j.jamcollsurg.2007.12.020. discussion 846–848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Comprehensive Cancer Network. Fort Washington, PA: NCCN; 2012. Pancreatic adenocarcinoma Version 2.2012. NCCN Clinical Practice Guidelines in Oncology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz MH, Pisters PW, Lee JE, Fleming JB. Borderline resectable pancreatic cancer: what have we learned and where do we go from here? Ann Surg Oncol. 2011;18:608–610. doi: 10.1245/s10434-010-1460-y. [DOI] [PubMed] [Google Scholar]
- Tempero MA, Arnoletti JP, Behrman SW, Ben-Josef E, Benson AB, Casper ES, et al. Pancreatic adenocarcinoma, Version 2.2012 featured updates to the NCCN Guidelines. J Natl Compr Canc Netw. 2012;10:703–713. doi: 10.6004/jnccn.2012.0073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee D, Katz MH, Rashid A, Varadhachary GR, Wolff RA, Wang H, et al. Histologic grading of the extent of residual carcinoma following neoadjuvant chemoradiation in pancreatic ductal adenocarcinoma: a predictor for patient outcome. Cancer. 2012;118:3182–3190. doi: 10.1002/cncr.26651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz MH, Fleming JB, Bhosale P, Varadhachary G, Lee JE, Wolff R, et al. Response of borderline resectable pancreatic cancer to neoadjuvant therapy is not reflected by radiographic indicators. Cancer. 2012;118:5749–5756. doi: 10.1002/cncr.27636. [DOI] [PubMed] [Google Scholar]
- Tzeng CW, Fleming JB, Lee JE, Xiao L, Pisters PW, Vauthey JN, et al. Defined clinical classifications are associated with outcome of patients with anatomically resectable pancreatic adenocarcinoma treated with neoadjuvant therapy. Ann Surg Oncol. 2012;19:2045–2053. doi: 10.1245/s10434-011-2211-4. [DOI] [PubMed] [Google Scholar]
- Evans DB, Varadhachary GR, Crane CH, Sun CC, Lee JE, Pisters PW, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–3502. doi: 10.1200/JCO.2007.15.8634. [DOI] [PubMed] [Google Scholar]
- Varadhachary GR, Wolff RA, Crane CH, Sun CC, Lee JE, Pisters PW, et al. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3487–3495. doi: 10.1200/JCO.2007.15.8642. [DOI] [PubMed] [Google Scholar]
- Hwang RF, Wang H, Lara A, Gomez H, Chang T, Sieffert N, et al. Development of an integrated biospecimen bank and multidisciplinary clinical database for pancreatic cancer. Ann Surg Oncol. 2008;15:1356–1366. doi: 10.1245/s10434-008-9833-1. [DOI] [PubMed] [Google Scholar]
- Kang CM, Kim JY, Choi GH, Kim KS, Choi JS, Lee WJ, et al. The use of adjusted preoperative CA 19-9 to predict the recurrence of resectable pancreatic cancer. J Surg Res. 2007;140:31–35. doi: 10.1016/j.jss.2006.10.007. [DOI] [PubMed] [Google Scholar]
- Katz MH, Wang H, Fleming JB, Sun CC, Hwang RF, Wolff RA, et al. Longterm survival after multidisciplinary management of resected pancreatic adenocarcinoma. Ann Surg Oncol. 2009;16:836–847. doi: 10.1245/s10434-008-0295-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzeng CW, Abbott DE, Cantor SB, Fleming JB, Lee JE, Pisters PW, et al. Frequency and intensity of postoperative surveillance after curative treatment of pancreatic cancer: a cost-effectiveness analysis. Ann Surg Oncol. 2013;20:2197–2203. doi: 10.1245/s10434-013-2889-6. [DOI] [PubMed] [Google Scholar]
- Tzeng CW, Fleming JB, Lee JE, Wang X, Pisters PW, Vauthey JN, et al. Yield of clinical and radiographic surveillance in patients with resected pancreatic adenocarcinoma following multimodal therapy. HPB. 2012;14:365–372. doi: 10.1111/j.1477-2574.2012.00445.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartwig W, Strobel O, Hinz U, Fritz S, Hackert T, Roth C, et al. CA 19-9 in potentially resectable pancreatic cancer: perspective to adjust surgical and perioperative therapy. Ann Surg Oncol. 2012;20:2188–2196. doi: 10.1245/s10434-012-2809-1. [DOI] [PubMed] [Google Scholar]
- Small W, Jr, Berlin J, Freedman GM, Lawrence T, Talamonti MS, Mulcahy MF, et al. Full-dose gemcitabine with concurrent radiation therapy in patients with non-metastatic pancreatic cancer: a multicentre phase II trial. J Clin Oncol. 2008;26:942–947. doi: 10.1200/JCO.2007.13.9014. [DOI] [PubMed] [Google Scholar]
- Ohara K, Tatsuzaki H, Molotkova NG, Oda T, Yuzawa K, Saida Y, et al. Utility of serum CA 19-9 monitoring in preoperative radiotherapy for pancreatic cancer. Hepatogastroenterology. 2001;48:859–863. [PubMed] [Google Scholar]
- Willett CG, Daly WJ, Warshaw AL. CA 19-9 is an index of response to neoadjunctive chemoradiation therapy in pancreatic cancer. Am J Surg. 1996;172:350–352. doi: 10.1016/S0002-9610(97)89547-5. [DOI] [PubMed] [Google Scholar]
- Park IJ, Choi GS, Lim KH, Kang BM, Jun SH. Serum carcinoembryonic antigen monitoring after curative resection for colorectal cancer: clinical significance of the preoperative level. Ann Surg Oncol. 2009;16:3087–3093. doi: 10.1245/s10434-009-0625-z. [DOI] [PubMed] [Google Scholar]
- Park JW, Lim SB, Kim DY, Jung KH, Hong YS, Chang HJ, et al. Carcinoembryonic antigen as a predictor of pathologic response and a prognostic factor in locally advanced rectal cancer patients treated with preoperative chemoradiotherapy and surgery. Int J Radiat Oncol Biol Phys. 2009;74:810–817. doi: 10.1016/j.ijrobp.2008.08.057. [DOI] [PubMed] [Google Scholar]
- Yakabe T, Nakafusa Y, Sumi K, Miyoshi A, Kitajima Y, Sato S, et al. Clinical significance of CEA and CA 19-9 in postoperative follow-up of colorectal cancer. Ann Surg Oncol. 2010;17:2349–2356. doi: 10.1245/s10434-010-1004-5. [DOI] [PubMed] [Google Scholar]


