Abstract
Background
The surgical portosystemic shunts (PSS) are a time-proven modality for treating portal hypertension. Recently, in the era of liver transplantation and the transjugular intrahepatic portosystemic shunts (TIPS), use of the PSS has declined.
Objectives
This study was conducted to evaluate changes in practice, referral patterns, and short-and longterm outcomes of the use of the surgical PSS before and after the introduction of the Model for End-stage Liver Disease (MELD).
Methods
A retrospective analysis of 47 patients undergoing PSS between 1996 and 2011 in a single university hospital was conducted.
Results
Subgroups of patients with cirrhosis (53%), Budd–Chiari syndrome (13%), portal vein thrombosis (PVT) (26%), and other pathologies (9%) differed significantly with respect to shunt type, Child–Pugh class, MELD score and perioperative mortality. Perioperative mortality at 60 days was 15%. Five-year survival was 68% (median: 70 months); 5-year shunt patency was 97%. Survival was best in patients with PVT and worst in those with Budd–Chiari syndrome compared to other subgroups. Patency was better in the subgroups of patients with cirrhosis and other pathologies compared with the PVT subgroup. Substantial changes in referral patterns coincided with the adoption of the MELD in 2002, with decreases in the incidence of cirrhosis and variceal bleeding, and increases in non-cirrhotics and hypercoagulopathy.
Conclusions
Although the spectrum of diseases benefiting from surgical PSS has changed, surgical shunts continue to constitute an important addition to the surgical armamentarium. Selected subgroups with variceal bleeding in well-compensated cirrhosis and PVT benefit from the excellent longterm patency offered by the surgical PSS.
Introduction
Since its introduction into clinical practice in the mid-20th century, surgical portosystemic shunts (PSS) have established themselves as a time-proven method of treating gastro-oesophageal variceal bleeding associated with end-stage liver disease or extrahepatic portal hypertension. In the absence of alternatives, the PSS represented the only viable treatment prior to the introduction and widespread adoption of endoscopic interventions such as endoscopic sclerotherapy and banding, followed later by the perfection of orthotopic liver transplantation (OLT) along with the emergence of the transjugular intrahepatic portosystemic shunts (TIPS).
During the 1990s, several prospective randomized trials demonstrated the superior longterm patency of surgical shunts and their better prevention of recurrent bleeding, along with acceptably low operative morbidity and encephalopathy.1–3 These reports, however, did not influence the rapid rise in the use of the TIPS, which has become the standard treatment of choice for portal decompression, despite the fact that longterm outcomes were inferior in both patency and survival.
Changing practice patterns, whereby cirrhotic patients were almost exclusively treated by our gastrointestinal medicine and radiology colleagues, along with the ‘minimally–invasive’ appeal of endoscopic interventions and TIPS, caused an abrupt decline in the use of surgical shunts. With the advent of OLT as the definitive treatment for end-stage liver disease, many non-surgeons have come to firmly believe that the role of shunts is currently limited to that of a ‘bridge to transplant’ and, because the TIPS is able to fulfil this role, there is minimal to no purpose for surgical shunts.4 By contrast, it is the present authors' belief that a large patient population could be served appropriately using the surgical PSS, including those with cirrhosis with compensated liver function and patients with pre-and post-hepatic portal hypertension who have minimal or no liver dysfunction. In this patient population and in some patients in whom liver transplantation is either premature or not indicated, the use of a surgical shunt may be lifesaving and may offer excellent control of symptoms and a positive longterm outcome. Removing shunt operations from the surgical armamentarium is premature.
The purpose of this report is therefore several-fold. It aims to: (i) reassess the safety of the surgical PSS, as well as their longterm survival and patency; (ii) report change in referral practice before and after the incorporation of the Model for End-stage Liver Disease (MELD) score into the United Network for Organ Sharing (UNOS) system; (iii) identify clinical subgroups of patients who continue to benefit from the surgical PSS during the current era of the TIPS and OLT, and (iv) review the existing literature in support of the continued use of the surgical PSS in current practice.
Materials and methods
After institutional review board approval had been obtained, a retrospective review of a prospectively collected database was performed. These data cover all patients who underwent the creation of a surgical PSS at a single university hospital performed by a single transplant and hepatobiliary surgeon during 1996–2011. Surgical shunts were of three varieties: the side-to-side portocaval shunt (SSPCS), the mesocaval with interposition H-or C-graft shunt, and the central splenorenal shunt. The appropriate techniques have been described elsewhere.5,6 Before and after the creation of the shunt, pressures in the portal vein and inferior vena cava (IVC) were measured and portosystemic gradients were calculated and recorded. All patients with known hypercoagulopathy received anticoagulation immediately after the creation of the shunt, and continued to receive warfarin indefinitely. Hypercoagulability was established by a history of thrombosis and protein C/S deficiency, Factor V Leiden mutation, lupus inhibitor, antiphospholipid syndrome or antithrombin III deficiency.
Baseline Doppler ultrasound was performed to assess shunt patency prior to discharge and during follow-up at 3-month intervals during the first year and yearly thereafter. If a shunt occlusion or stenosis was suspected based on recurrent bleeding or ascites, Doppler ultrasound was repeated and additional angiographic studies were performed as appropriate. During the follow-up period, patients with cirrhosis remained on a restricted sodium intake of 2 g/day and restricted dietary protein of 60 g/day. In patients with alcoholic cirrhosis, complete abstinence from alcohol was emphasized repeatedly. An episode of encephalopathy was defined on mental status examination, accompanied by an elevated serum ammonia, or presence of asterixis on physical examination. Encephalopathy was graded as mild if it was short-lived, corrected easily with dietary modification and an increase in lactulose, and did not require hospitalization, and as severe when it required hospitalization or prolonged management with lactulose, an oral antibiotic and significant dietary protein restriction.
Data points were collected on: age; gender; diagnosis and surgical indications; comorbidities; presence of hypercoagulopathy; type of portal hypertension (pre-, intra-or post-hepatic); preoperative Child–Pugh class and MELD score; liver chemistries; presence of gastro-oesophageal varices and number of episodes of variceal bleeding and endoscopic interventions; type of shunt performed; timing of procedure (emergent, urgent or elective); pre-and post-shunt gradient (in mm saline); estimated blood loss (in ml); results of liver biopsy; postoperative 30-and 60-day complications and mortality; length of hospital stay; shunt patency as determined by Doppler ultrasound; presence of post-shunt encephalopathy, ascites or re-bleeding; status of the patient at last follow-up, and date of death.
Associations between various variables and diagnostic subgroups were calculated using Pearson's chi-squared test and the Kruskal–Wallis test for categorical and continuous variables, respectively.
Patient survival and shunt patency were calculated using Kaplan–Meier survival analysis. Differences between groups were estimated by a log-rank test. Differences were considered significant if associated with a P-value of < 0.05.
A multivariate logistic regression model (for operative mortality) and Cox proportional hazard models (for overall survival and patency) were created to establish the associations of selected variables with these outcomes. Associations among various variables were calculated using Pearson's chi-squared test and the Wilcoxon signed-rank test for categorical and continuous variables, respectively.
Results
The study included 47 PSS patients treated over a 15-year period (1996–2011). Detailed patient demographic and clinical characteristics are presented in Table 1. The mean age of the patients was 44.6 ± 18.6 years. The youngest patient was a 7-month-old baby boy with Krabbe disease and a history of a bone marrow transplant with subsequent veno-occlusive disease (VOD) and massive ascites; the oldest was a 72-year-old man with non-alcoholic steatohepatitis (NASH) cirrhosis and variceal bleeding.
Table 1.
Patient demographics and clinical characteristics of patients submitted to portosystemic shunt surgery
| Study period | 1996–2011 |
| Total number of patients | 47 |
| Age, years, mean ± SD | 44.6 ± 18.6 |
| Male : female ratio | 0.53 : 0.47 |
| Hypercoagulopathy, n (%) | 14 (30%) |
| Evidence of gastro-oesophageal varices, n (%) | 37 (80%) |
| Clinically significant bleeding, n (%) | 34 (72%) |
| Oesophagogastroduodenoscopy, n (%) | 25 (55%) |
| Cirrhosis/no cirrhosis, n (%) | 25 (53%)/22 (47%) |
| Diagnostic subgroups, n (%) | |
| Cirrhosis | 25 (53%) |
| EtOH | 8 (17%) |
| Hepatitis C | 5 (11%) |
| Cryptogenic and other | 12 (26%) |
| Budd–Chiari syndrome | 6 (13%) |
| Portal vein thrombosis | 12 (26%) |
| Othera | 4 (9%) |
| Type of portal hypertension, n (%) | |
| Pre-hepatic | 13 (28%) |
| Intra-hepatic | 27 (57%) |
| Post-hepatic | 5 (11%) |
| Pre-and post-hepatic and pre-and intra-hepatic | 2 (4%) |
| Child–Pugh class, n (%) | |
| A | 24 (51%) |
| B | 19 (40%) |
| C | 4 (9%) |
| Era, n (%) | |
| Pre-MELD | 25 (53%) |
| Post-MELD | 22 (47%) |
| Timing of operation, n (%) | |
| Elective | 32 (70%) |
| Emergent (24–48 h) | 8 (17%) |
| Urgent (during index admission) | 7 (13%) |
| Type of shunt, n (%) | |
| SSPCS | 35 (74%) |
| Mesocaval | 5 (11%) |
| Central splenorenal | 7 (15%) |
| Portocaval gradient, mm saline, mean ± SD | |
| Before shunting | 258.3 ± 89.4 |
| After shunting | 10.4 ± 27.17 |
| Difference | 248.8 ± 87.9 |
| Mortality, n (%) | |
| 30-day | 3 (6%) |
| 60-day | 7 (15%) |
Congenital hepatoportal sclerosis, nodular regenerative hyperplasia, Krabbe disease, congenital hepatic fibrosis.
SD, standard deviation; MELD, Model for End-stage Liver Disease; SSPCS, side-to-side portocaval shunt.
Four definite subgroups were identified, comprising patients with: (i) cirrhosis (n = 25, 53%) (alcoholic, 17%; hepatitis C, 11%; cryptogenic and other cirrhosis, 26%); (ii) Budd–Chiari syndrome (n = 6, 13%); (iii) portal vein thrombosis (PVT) (n = 12, 26%), and (iv) other pathologies (n = 4, 9%), including congenital hepatoportal sclerosis, nodular regenerative hyperplasia, VOD and congenital hepatic fibrosis.
When grouped according to the presence or absence of cirrhosis, patients with cirrhosis comprised 53% and those without cirrhosis 47% (diagnostic subgroups ii–iv) of the study cohort. Of note, eight of 12 patients with cryptogenic cirrhosis were obese and had diabetes and would be reclassified as having NASH cirrhosis according to current diagnostic criteria.
Fourteen patients (30%) were either known to have hypercoagulopathy or were diagnosed as such during their hospital workup.
Thirty-eight patients (80%) showed evidence of gastro-oesophageal varices on oesophagogastroduodenoscopy or computed tomography. Of these, 34 (72%) had clinically significant bleeding (mean of 2.5 bleeding events per patient) and 26 (55%) underwent endoscopic intervention (mean of 1.96 interventions per patient).
Thirty-day operative morbidity was 43%. The majority of morbidity referred to ascites-related wound complications [ascites leak from the wound in the early postoperative period (n = 3), superficial wound infection (n = 7), and wound dehiscence with intra-abdominal abscess (n = 2)]. Other complications were: acute kidney injury, worsening ascites with IVC compression treated with IVC stenting and ascites resolution (n = 1); coecal perforation (n = 1); methicillin-resistant Staphylococcus aureus (MRSA) pneumonia requiring ventilator support (n = 1); new-onset atrial fibrillation (n = 1); recurrent pericardial effusion requiring a pericardial window (n = 1), and line infection (n = 1). Early postoperative shunt thrombosis with resultant recurrent variceal bleeding was identified in one patient who had a central splenorenal shunt in the setting of a hypercoagulable state not recognized prior to the first shunt. The recurrent bleeding was successfully treated by shunt revision on postoperative day 7.
Perioperative mortality rates at 30 days and 60 days were 6% (n = 3) and 15% (n = 7), respectively. Mortality details are presented in Table 2. In-hospital mortality in the emergent and urgent categories grouped together comprised of three of 15 patients (20%). The mean and median length of stay were 19.5 days and 10 days, respectively.
Table 2.
Mortality details in patients submitted to portosystemic shunt surgery (n = 47)
| Subject | Sex, age, years | Diagnosis | MELD score | Child–Pugh class | Type of shunt | Mortality time, PoD | Mortality details |
|---|---|---|---|---|---|---|---|
| 1 | M, 52 | Acute massive GI bleed, EToH cirrhosis, need for urgent shunt | 15 | C | Emergency SSPCS | 2 | Significant bleeding and coagulopathy intra-and postoperatively despite massive transfusions and factors |
| 2 | F, 30 | Budd–Chiari syndrome, fulminant liver failure, massive ascites, brain oedema, unsuccessful angioplasty | 37.8 | C | Emergency SSPCS | 4 | Fulminant liver failure, brain oedema, emergent shunt, postoperative herniation, brain death |
| 3 | M, 35 | Budd–Chiari syndrome, hypersplenism, polycythemia vera | 16.67 | B | SSPCS | 10 | Intractable third spacing, ascites, hypotension, resistant to pressors and albumin, suspected to be caused by TRALI, hepatorenal syndrome, haemodialysis, ARDS |
| 4 | F, 46 | Alcoholic cirrhosis, oesophageal bleeding bleed, need for emergent shunt | 10 | B | Emergency SSPCS | 39 | Postoperative ascites, recurrent paracentesis, encephalopathy, EtOH withdrawal, re-exploration for possible shunt revision (shunt was patent), repeat upper GI bleed despite repeat banding and splenic artery embolization, massive transfusion |
| 5 | F, 52 | Budd–Chiari syndrome, thrombosis of right and main hepatic veins, stenosis of left hepatic vein, polycythemia vera | 9.5 | B | SSPCS | 51 | Tense ascites, IVC clotting despite anticoagulation IVC stenting, IVC and shunt thrombectomy, revision of shunt to H-graft, bleeding duodenal ulcer, ARF, intra-abdominal bleed × 2, erosion of H-graft |
| 6 | M, 62 | Myeloproliferative syndrome, polycythemia vera, Budd–Chiari syndrome, multifocal non-occlusive PVT, recurrent intractable ascites | 21.5 | C | H-graft portocaval shunt, portal vein thrombectomy | 53 | Shunt thrombosis caused by extrinsic compression (caudate lobe), TIPS placement, recurrent ascites leak, malnutrition, liver failure, resistant hypercoagulable state (Jak 2 mutation, ATIII deficiency, Factor 7,8,9), failure to improve, withdrawal of care |
| 7 | F, 29 | Budd–Chiari syndrome, factor 5 Leiden, polycythemia vera | 20 | C | SSPCS | 63 | Wound dehiscence, re-exploration, shunt thrombosis, despite anticoagulation, thrombectomy ×2 suprahepatic IVC stenting, shunt revision to H-graft, portal vein thrombectomy, hepatocellular atrophy with progressive hepatocellular collapse, liver failure, renal failure, OLT, hypotension, hypothermia, cardiac arrest |
M ELD, Model for End-stage Liver Disease; PoD, postoperative day; M, male; F, female; GI, gastrointestinal; SSPCS, side-to-side portocaval shunt; TRALI, transfusion-related acute lung injury; ARDS, adult respiratory distress syndrome; EtOH, alcohol; IVC, inferior vena cava; ARF, acute renal failure; ATIII, antithrombin III; PVT, portal vein thrombosis; OLT, orthotopic liver transplantation; TIPS, transjugular intrahepatic portosystemic shunts.
Overall survival was 79%, 72%, 68% and 48% at 1, 3, 5 and 7 years, respectively. A total of 18% of patients survived 10 years (Fig. 1). Median survival was 70 months with a mean follow-up of 41 months.
Figure 1.
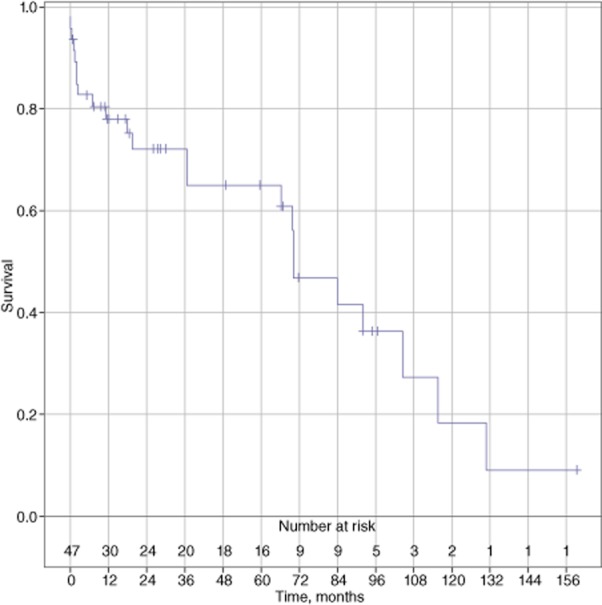
Overall survival (n = 47)
Overall primary shunt patency was 97% during the first 5-year interval and 58% during the second 5-year interval (Fig. 2). Secondary-assisted patency was 70% at 10 years, as one hypercoagulable patient experienced occlusion of a central splenorenal shunt after 6 years and underwent the successful creation of a mesocaval shunt with an interposition H-graft that remained patent 3 years afterwards.
Figure 2.
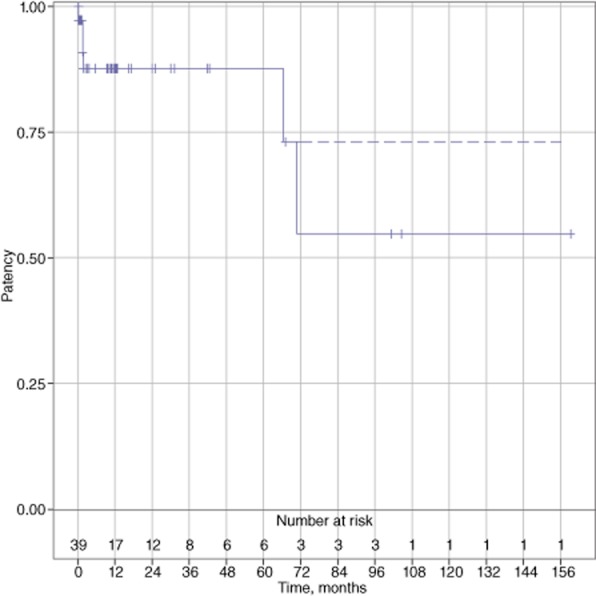
Overall shunt patency (n = 39)
Eleven patients (23%) had a single episode of mild post-shunt encephalopathy that was short-lived, was corrected easily with diet modification and an increase in lactulose, and did not require hospitalization; one patient had an episode of moderate encephalopathy that required brief hospitalization in addition to these measures. There were no episodes of severe or prolonged encephalopathy.
There were three episodes of re-bleeding during the follow-up period. One case signified shunt occlusion that was corrected with revision. The second patient bled from a duodenal ulcer, and the third patient had an emergency surgical shunt for bleeding varices and bled in the immediate postoperative period, with spontaneous resolution.
Associations among selected variables and diagnostic subgroups
When patients were stratified by diagnostic subgroups, various associations emerged (Table 3). There was a significant difference in age distribution, as patients with causes other than cirrhosis were on average 15–37 years younger than those with cirrhosis (P = 0.016). Hypercoagulopathy was found to be strongly associated with the Budd–Chiari syndrome and PVT subgroups (P < 0.001). Budd–Chiari syndrome was associated with a significantly higher MELD score and Child–Pugh classes B and C (P < 0.017 and P < 0.001, respectively). These patients also suffered from significantly higher 60-day mortality (P < 0.001). There was no difference in postoperative complications among the subgroups. Although patients with cirrhosis and PVT presented with gastro-oesophageal variceal bleeding, patients with Budd–Chiari syndrome and those in the subgroup with other causes of portal hypertension did so less frequently (P < 0.001), with ascites as the major manifestation of portal hypertension. An SSPCS was performed in 100% of patients with cirrhosis, Budd–Chiari syndrome and ‘other’ pathologies, whereas patients with PVT underwent a mesocaval shunt (42%) or central splenorenal shunt (58%) procedure (P < 0.001). No association was found between the urgency of the procedure and rates of perioperative morbidity or mortality (P = NS). During the pre-MELD era, patients with cirrhosis had constituted the majority of shunt patients (68%); after the adoption of the UNOS allocation of liver allografts by MELD score, the ratio reversed and non-cirrhotic patients came to represent the largest subgroup (64%) (P = 0.03). Portal hypertension associated with hypercoagulopathy became a more frequent indication for PSS after the introduction of MELD criteria (P = 0.028), whereas during this era variceal bleeding became a rare indication (P = 0.004).
Table 3.
Associations among selected variables and diagnostic subgroups in patients submitted to portosystemic shunt surgery (n = 47)
| Cirrhosis (combined) (n = 24) | Budd–Chiari syndrome (n = 6) | Portal vein thrombosis (n = 12) | Other causes of portal hypertension (n = 4) | P-value | |
|---|---|---|---|---|---|
| Age, years, median (IQR) | 53 (47–61) | 38 (31–61) | 37 (16–53) | 14 (6–32) | 0.016 |
| Sex, n (%) | 0.6 | ||||
| Female | 11 (44%) | 4 (67%) | 6 (50%) | 1 (25%) | |
| Male | 14 (56%) | 2 (33%) | 6 (50%) | 3 (75%) | |
| Hypercoagulopathy, n (%) | <0.001 | ||||
| No | 25 (100%) | 1 (17%) | 4 (33%) | 4 (100%) | |
| Yes | 0 | 5 (83%) | 8 (67%) | 0 | |
| Child–Pugh class, n (%) | 0.006 | ||||
| A | 15 (60%) | 0 | 7 (58%) | 2 (50%) | |
| B | 9 (36%) | 3 (50%) | 5 (42%) | 2 (50%) | |
| C | 1 (4%) | 3 (50%) | 0 | 0 | |
| MELD score, median (IQR) | 10.9 (8.8–13.7) | 19.4 (17.2–22.6) | 9.2 (7.8–13.55) | 10.6 (10.0–11.3) | 0.023 |
| GO variceal bleeding episodes, n, median (IQR)xs* | 2 (1–3) | 0 | 1.5 (1–3) | 0 | <0.001 |
| Type of shunt, n (%) | <0.001 | ||||
| Side-to-side portocaval | 25 (100%) | 6 (100%) | 0 | 4 (100%) | |
| Mesocaval | 0 | 0 | 5 (42%) | 0 | |
| Central splenorenal | 0 | 0 | 7 (58%) | 0 | |
| 60-day complications, n (%) | 0.8 | ||||
| No | 14 (56%) | 4 (67%) | 6 (50%) | 3 (75%) | |
| Yes | 11 (44%) | 2 (33%) | 6 (50%) | 1 (25%) | |
| 60-day mortality, n (%) | <0.001 | ||||
| No | 23 (92%) | 1 (17%) | 12 (100%) | 4 (100%) | |
| Yes | 2 (8%) | 5 (83%) | 0 | 0 | |
| Period, n (%) | 0.03 | ||||
| Pre-introduction of MELD | 17 (68%) | (Combined) 8 (36%) | |||
| Post-introduction of MELD | 8 (32%) | (Combined) 14 (64%) |
IQR, interquartile range; Model for End-stage Liver Disease; GO, gastro-oesophageal.
Survival stratified by diagnostic subgroups
When overall survival was stratified by diagnostic subgroups, patients with PVT were found to fare best (P = 0.006) and patients with Budd–Chiari syndrome to fare worst (P = 0.02) compared with the rest of the subgroups (Fig. 3). The poor survival of patients with Budd–Chiari syndrome corresponded to the significant postoperative mortality in this subgroup. Five of six patients with Budd–Chiari syndrome died in the hospital. Three of them had Child–Pugh class C cirrhosis and high MELD scores signifying advanced disease. One patient had fulminant hepatic failure and brain oedema and had been referred very late; another patient had shunt thrombosis caused by extrinsic compression by a hypertrophied caudate lobe and recalcitrant hypercoagulable and premalignant state, and the last patient developed recurrent shunt thrombosis requiring revision with a cryopreserved femoral vein H-graft and portal vein thrombectomy and later salvage OLT secondary to progressive liver failure, which led to death. An additional patient died despite aggressive measures including full anticoagulation, IVC stenting, IVC and shunt thrombectomy, and revision of the shunt with an internal jugular vein H-graft. This patient developed a bleeding duodenal ulcer, acute renal failure, and died of intra-abdominal bleeding secondary to the erosion of his duodenal ulcer into the H-graft. One remaining patient with Budd–Chiari syndrome remained alive at >13 years after her shunt operation. Overall, the survival of patients without cirrhosis did not differ statistically from that of their counterparts with cirrhosis (P = 0.15), nor was there any difference in overall survival between patients operated before and after the introduction of the MELD criteria. Of note, only one patient (noted above) in the entire series underwent OLT for subsequent treatment.
Figure 3.
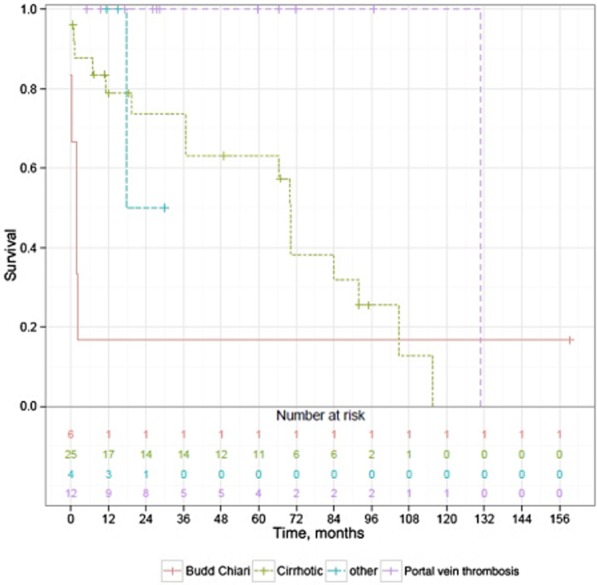
Survival stratified by diagnostic subgroups (n = 47)Notes: Difference between groups was estimated by a log-rank test. P = 0.006 PV thrombosis versus cirrhosis, and P = 0.02 PV thrombosis versus Budd-Chiari and cirrhosis versus Budd-Chiari. ‘Other’ subgroup is not large enough for statistical significance.
Longterm patency of the PSS
The patency of shunts stratified by diagnostic subgroups is illustrated in Fig. 4. In terms of longterm shunt patency, patients in the cirrhosis and ‘other’ pathologies subgroups fared better than those in the PVT subgroup (P = 0.04).
Figure 4.
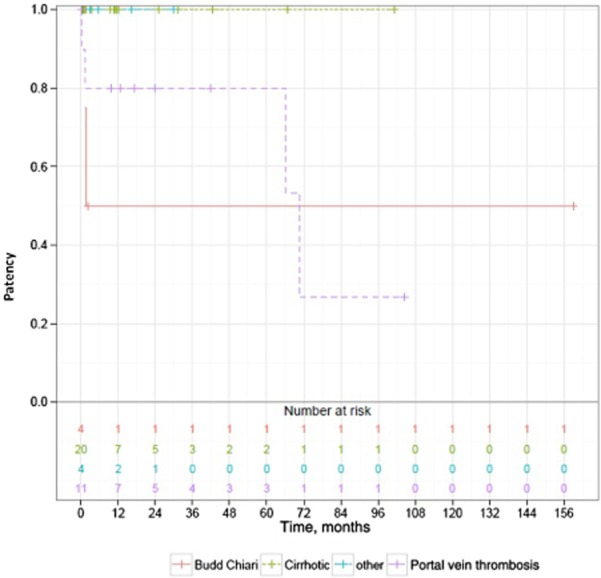
Shunt patency stratified by diagnostic subgroups (color-coded) (n = 39)Notes: Difference between groups was estimated by a log-rank test. P = 0.005 cirrhosis versus Budd-Chiari and P = 0.04 cirrhosis versus PV thrombosis. ‘Other’ subgroup is not large enough for statistical significance.
Univariate and multivariate analysis
60-day operative mortality
On univariate analysis, a higher MELD score (P = 0.024), intraoperative transfusion (P = 0.041), Child–Pugh class B or C (P < 0.001) and the presence of Budd–Chiari syndrome (P < 0.001) were predictive of increased 60-day mortality (Table 4). In a multivariate model, MELD score was found to be significant [odds ratio (OR) 1.36; P = 0.03]. Budd–Chiari syndrome was so associated with 60-day mortality that a model of convergence could not be created.
Table 4.
Multivariate analysis of 60-day mortality, overall survival and longterm patency in patients submitted to portosystemic shunt surgery (n = 47)
| 60-day mortality | |||
|---|---|---|---|
| Variable | OR | 95% CI | P-value |
| Age (per year) | 0.993 | 0.937–1.06 | 0.792 |
| MELD (per point) | 1.366 | 1.063–1.959 | 0.032 |
| EBL (per 100 ml) | 1.051 | 0.956–1.234 | 0.523 |
| Transfusion (per unit) | 1.052 | 0.555–1.825 | 0.853 |
| Elective (versus urgent) surgery | 1.137 | 0.077–25.70 | 0.924 |
| Longterm survival | |||
| Variable | HR | 95% CI | P-value |
| Age (per year) | 1.030 | 0.984–1.079 | 0.207 |
| Sex (male versus female) | 0.321 | 0.060–1.714 | 0.184 |
| Child–Pugh score (reference: A) | |||
| B | 1.106 | 0.252–4.860 | 0.894 |
| C | 59.364 | 2.183–1614.688 | 0.015 |
| EBL (per 100 ml) | 1.043 | 0.928–1.171 | 0.479 |
| MELD (per point) | 0.864 | 0.698–1.070 | 0.182 |
| Diagnosis (reference: EtOH cirrhosis) | |||
| Budd–Chiari syndrome | 0.268 | 0.014–4.998 | 0.378 |
| Cryptogenic and other cirrhosis | 0.113 | 0.009–1.489 | 0.097 |
| Hepatitis C cirrhosis | 0.470 | 0.055–4.050 | 0.492 |
| Other | 0.675 | 0.053–8.641 | 0.763 |
| Portal vein thrombosis | 0.040 | 0.002–0.821 | 0.037 |
| Post-MELD (versus pre-MELD) | 1.122 | 0.251–5.024 | 0.880 |
| Longterm patency | |||
| Variable | HR | 95% CI | P-value |
| Age (per year) | 1.016 | 0.948–1.088 | 0.655 |
| Hypercoagulopathy | 0.484 | 0.030–7.743 | 0.608 |
| MELD (per point) | 1.478 | 0.941–2.323 | 0.090 |
| Shunt type (reference: portocaval) | |||
| Mesocaval shunt | 116.680 | 0.744–18298.675 | 0.065 |
| Splenorenal shunt | 22.430 | 0.610–824.879 | 0.091 |
OR, odds ratio; 95% CI, confidence interval; HR, hazard ratio; MELD, Model for End-stage Liver Disease; EBL, estimated blood loss.
Longterm survival
When perioperative mortality was excluded, multivariate analysis showed PVT, compared with EtOH cirrhosis, to be associated with increased longterm survival [hazard ratio (HR) 0.040; P = 0.037], whereas Child–Pugh class C status, compared with that of Child–Pugh class A, was associated with decreased longterm survival. (HR = 59; P = 0.015).
Longterm patency
On multivariate analysis mesocaval (HR = 116) and central splenorenal (HR = 22) shunts tended to have decreased longterm patency compared with the SSPCS, although these differences did not reach statistical significance (P = 0.065 and P = 0.091, respectively). This is likely to reflect the high shunt thrombosis rate in patients with Budd–Chiari syndrome and SSPCS and the relatively smaller numbers of patients who did not receive an SSPCS.
Discussion
The current study summarizes the present authors' experience with surgical PSS during a time when multiple non-invasive strategies and liver transplantation have become de facto modalities for indications previously reserved for surgical shunts. Despite the fact that only a small fraction of cirrhosis patients with bleeding will ultimately undergo liver transplantation after the TIPS placement (3–10% in most series),5–8 an unsubstantiated belief that those patients will be candidates for possible liver transplant has resulted in the adoption of the TIPS as the decompressive shunt of choice and as a bridge to transplant by clinical decision makers. In a randomized controlled trial (RCT) reported by Orloff (MJ) et al., which compared endoscopic sclerotherapy with emergency PSS in 211 unselected consecutive patients with advanced cirrhosis and bleeding oesophageal varices, only 6% were referred for OLT and only 2% underwent OLT, with worse 1-and 5-year survival rates.9 In addition, this analysis was supplemented by 1300 non-randomized cirrhosis patients who previously underwent SSPCS with 100% follow-up, of whom only 3.8% were referred for and only 1.5% actually underwent OLT, again with a worse 5-year survival rate.9
As the number of TIPS performed has increased, the number of surgical shunts has reciprocally decreased and, as a result, current surgical trainees are neither exposed to these operations nor comfortable with treating this group of patients, either surgically or medically. A review of case logs maintained by the Accreditation Council for Graduate Medical Education (ACGME) over the past 10 years reveals that the average number of PSS performed by a graduating resident ranged between one and five and averaged 0.3 per resident during 2000–2005 and 0.1 during 2005–2012.10 In addition, since February 2002, when UNOS adopted a modified version of the original MELD (Proposed Amended UNOS Policy 3.6. 2002; http://optn.transplant.hrsa.gov/policiesAndBylaws/policies.asp) in order to allow the allocation of liver organs to sicker patients, the volume of patients with cirrhosis referred for surgical shunts has declined in parallel. As sicker cirrhosis patients have become eligible for transplantation and have meanwhile been temporized by other means, the need for surgical shunts has continued to decrease further. This shift has contributed to the perception of surgical shunts in general as irrelevant.
In the present authors' experience, although cirrhosis patients constituted the majority of shunt patients (68%) prior to the introduction of the MELD system, after the adoption of MELD by UNOS the ratio reversed and patients without cirrhosis came to represent the majority (64%) (P = 0.03). Conditions associated with hypercoagulopathy came to represent a more frequent indication for the SSPCS during the ‘post-MELD’ era, whereas variceal bleeding constituted a rare indication in this period.
The current study has identified four distinct diagnostic subgroups, each of which demonstrates unique patient characteristics and associated factors. These subgroups (comprising patients with, respectively, cirrhosis, Budd–Chiari syndrome, PVT and ‘other’ pathologies) differed in approach, complications and short-and longterm prognosis. Cirrhosis patients were on average 15–37 years older than patients without cirrhosis. As expected, hypercoagulopathy was found to be strongly associated with Budd–Chiari syndrome and PVT. Although patients with cirrhosis and PVT frequently presented with gastro-oesophageal varices and bleeding, patients with Budd–Chiari syndrome and other pathologies presented mostly with ascites. In the present series, Budd–Chiari syndrome was associated with significantly higher MELD score and Child–Pugh class B or C status and also with significantly higher 60-day mortality. One of the earliest series of Budd–Chiari syndrome patients treated with surgical shunts was reported by Cameron et al. and indicated modest results.11 Only eight of 12 patients survived to discharge; of those, three developed shunt thrombosis shortly thereafter, and the remaining five patients had a patent shunt. Importantly, however, liver architecture normalized in patients with patent shunts who had not yet developed cirrhosis. Later, with early intervention, perfection of the surgical technique, and a combination of an SSPCS with a cavoatrial shunt, Orloff (MJ) et al. achieved significantly improved results and demonstrated that effective early decompression of the liver is imperative to halt the progression of cirrhosis in Budd–Chiari syndrome.12,13
The present results of the surgical treatment of Budd–Chiari syndrome (five of six in-hospital deaths) are discouraging. The following explanations are proposed. These patients were referred late for surgical shunt procedures, with significant liver and systemic decompensation, and higher MELD scores than patients in the other subgroups. They might possibly have been salvaged by early referral. The common practice has been medical treatment with anticoagulation and thrombolysis: this is known as a ‘stepwise approach’.14–16 It should be noted that patient deaths that occur as a result of difficult-to-control hypercoagulability causing recurrent shunt thrombosis would most likely also result in TIPS thrombosis, should this route have been elected. As a result, in light of the high mortality risk affecting patients with Budd–Chiari syndrome and established liver decompensation, the current authors recommend neither surgical shunt nor TIPS surgery for this subgroup. These patients should be promptly referred for definitive therapy with OLT.
All patients in the cirrhosis, Budd–Chiari syndrome and ‘other’ pathology subgroups received SSPCS, whereas patients with PVT received mesocaval (42%) and central splenorenal (58%) shunts. Although the proponents of distal splenorenal shunts argue that they achieve effective portal decompression without a high incidence of encephalopathy,3,17,18 it is the present authors' experience that central shunts, in particular the SSPCS, are technically straightforward, achieve effective portal decompression and have high longterm patency. The ensuing encephalopathy is usually mild and easily managed. The further advantage of the SSPCS is its use in the treatment of intractable ascites, whereas the distal splenorenal shunt often makes the ascites worse. The goal of the present authors was to create an SSPCS when possible and to use other types of central shunts when an SSPCS was not an option, as in patients with complete PVT. However, there was no difference in postoperative complications between patients with different types of central shunts, as shown in previous reports.19–21 The frequency of encephalopathy in the present series was low (about 20% mild encephalopathy) and comparable with those reported in other series of surgical shunt patients (3.0–18.75%)22–24 and lower than that associated with the TIPS (30%),25 and, when diagnosed, was easily managed.
In the present series, rates of 30-day and 60-day operative mortality were 6% (n = 3) and 15% (n = 7), respectively, and were similar or lower than some of those reported for surgical shunts in the literature.20,21,23,24 The majority (70%) of the present patients were operated electively and the remainder underwent urgent or emergent surgery. Prohibitively high mortality rates after emergent surgical shunts, reaching 50%, have historically represented a reason to choose the TIPS in acute variceal haemorrhage. However, the RCT of emergency endoscopic sclerotherapy versus emergency PSS for acute bleeding of oesophageal varices reported by Orloff (MJ) et al. provided uniquely excellent results, with 1-, 5-, 10-and 15-year survival rates of 81%, 73%, 46% and 46%, respectively, in the shunt group and much inferior survival rates of 72%, 21%, 9% and 9%, respectively, in the sclerotherapy group.26,27 In this randomized trial, 85% of shunt patients survived to discharge. The present study found a 60-day mortality rate of 20% in the emergent/urgent subgroup, which compares favourably with those of other reports. No association between the urgency of the procedure and rates of perioperative morbidity or mortality emerged in the present data. In the present series, univariate analysis showed 60-day mortality to be associated with higher MELD score, intraoperative transfusion, Child–Pugh class B or C status and the presence of Budd–Chiari syndrome. However, in the multivariate model only the association with MELD score was found to be significant. The presence of Budd–Chiari syndrome was nearly universally associated with 60-day mortality; therefore, a model of convergence could not be created. The present results may suggest that it is not the urgency of the operation that puts the patient at higher risk, but, rather, the severity of disease along with liver decompensation.
In the present study, overall survival amounted to 79%, 72%, 68% and 48% at 1, 3, 5 and 7 years, respectively, and 18% of patients survived 10 years. Median survival was 70 months, which is comparable with or superior to survival in other surgical shunt series, but significantly longer than in patients submitted to TIPS procedures. In a study by Rosemurgy et al. of H-graft portocaval shunts, the survival of all patients grouped together (Child–Pugh classes A–C) was 68% at 2 years and 47% at 5 years, and median survival was 52 months.24,28 Patients with a more favourable risk profile (Child–Pugh class A or B cirrhosis) were the subject of a comparative study performed by Helton et al.,19 who reported a 2-year survival of 90% in a subgroup of patients with well-compensated liver disease undergoing surgical PSS. Orloff (MJ) et al. reported longterm survival of 78%, 71% and 57% at 5 years, 10 years and 15 years, respectively, in emergent portocaval shunt patients, demonstrating enviable results unmatched by anyone else.29 In addition, Orloff (MJ) et al. have shown that the surgical PSS is far superior to endoscopic sclerotherapy or the TIPS in two randomized prospective trials in the treatment of bleeding oesophageal varices.21,26
Pooled together, the survival of non-cirrhotic patients was not statistically different from their cirrhotic counterparts, nor was there any difference in the overall survival between ‘pre-MELD’ and ‘post-MELD’ era. However, when overall survival was stratified by diagnostic subgroups, patients with PVT fared better than other subgroups.
The current study demonstrated an overall primary shunt patency of 97% during the first 5-year interval and of 58% at 6–10 years. Secondary-assisted patency was 70% at 10 years. Rosemurgy et al. reported an H-graft median patency period of 42 months28 (about 70% at 2 years and 45% at 5 years), whereas Orloff (MJ) et al. reported SSPCS patency of >95% at more than 10 years of follow-up.29 Multivariate analysis of data for the present patients showed that mesocaval and central splenorenal shunts tended to have decreased longterm patency compared with portocaval shunts, but the difference was not significant. The longterm patency of shunts placed in the cirrhosis and ‘other’ pathology subgroups fared better than that in patients with PVT. Although it was not the purpose of the present study to compare survival or patency in similar patients after TIPS procedures, a review of the recent literature found no study with outcomes after TIPS matching the longterm patency of surgical shunts in the present study.
In our institution, surgical PSS are performed alongside the liver transplant programme. During the past two decades, significant changes in referral pattern, patient demographics, and in the indications for and types of shunts performed have been seen. The shift in referral pattern, whereby the majority of referrals prior to 2002 concerned patients with cirrhosis, and the majority after 2002 concerned non-cirrhotic patients with hypercoagulopathy and PVT may reflect several issues, including the more frequent use of endoscopic management and/or TIPS to control variceal bleeding coupled with the new MELD allocation strategy, which results in the referral of cirrhosis patients with bleeding varices and refractory ascites to interventional radiologists and their placement on a transplant list, whereas subgroups of patients with preserved liver function and portal hypertension are referred for a surgical shunt procedure. In the present cohort, many patients in the latter group had hypercoagulopathy and PVT, a combination that precludes the placement of a TIPS as a result of technical inability and patency concerns. These considerations, together with the fact that many of these patients were not in need of a liver transplant, deemed the surgical shunt particularly appropriate.
Several studies have been performed to directly compare the TIPS and different surgical shunts19,23,24 and have been summarized in a meta-analysis carried out by Clark et al.30 The latter included three prospective randomized trials and one retrospective case–control study, comprising 314 patients, and included both selective and non-selective surgical shunts in patients with cirrhosis of Child–Pugh class A or B status. The study showed equal 30-day mortality and survival at 1 year; however, there was a clear survival advantage at 2 years for patients undergoing surgical shunts. Three of four trials found significantly higher rates of rehaemorrhage after TIPS, resulting in a higher rate of TIPS failure at 2 years, which translated into more hospital readmissions, greater consumption of resources, higher costs, and a need for better longterm surveillance and care of TIPS patients. A recent RCT comparing surgical shunts with TIPS in unselected patients with cirrhosis over a period of >10 years again found that patients with surgical shunts had significantly better shunt patency, fewer re-bleeding events and re-interventions, less post-shunt encephalopathy and better survival across all Child–Pugh classes, in addition to lower overall costs.21 Most of the patients in the present study, particularly post-MELD, had preserved liver function and thus favoured the patency advantage of surgical shunts.
The present study has numerous limitations. Firstly, it is a retrospective review and is subject to all the disadvantages inherent in such a study design. Secondly, the number of patients is not large. Accordingly, each diagnostic subgroup has limited reliability of survival and patency analysis, particularly at the tails of the Kaplan–Meyer curves as numbers decrease further. However, the evaluation of diagnostic subgroups was of particular interest because it elucidates significant differences in approach, treatment and outcomes. Thirdly, comparing the survival and patency of subgroups with different disease aetiologies and other characteristics is less powerful; instead, a more worthwhile comparison would have been based on Child–Pugh class, MELD score and similar disease aetiology. However, this comparison, although more relevant statistically, would be of limited clinical interest because not only are the numbers of the present patient groups insufficient to warrant this type of study, but such research has been previously conducted and reported in the published literature.
In conclusion, the present authors acknowledge the fact that despite certain shortcomings, the TIPS is probably here to stay and will continue to be considered as a first-line therapy in patients with advanced cirrhosis and variceal bleeding or refractory ascites failing endoscopic or medical therapies. The use of the TIPS as a bridge to OLT in the short term is acceptable. However, this approach should not be extrapolated to subgroups of patients with preserved liver function and a good life expectancy, such as those with non-cirrhotic portal hypertension and well-compensated cirrhosis, who would best be served by a surgical PSS. The present report showed that surgical shunts in this population can be performed with low operative morbidity and mortality and may offer unmatched longterm patency, the prevention of recurrent events and, in consequence, possibly improved survival. Surgical shunts in advanced Budd–Chiari syndrome are associated with high mortality and those patients should be considered for transplant. Liver biopsy prior to proposing a surgical shunt will help to exclude this high-risk group.
Whether this report will change current practice when surgeons are no longer trained to carry out this procedure is questionable and beyond the scope of this discussion. However, it is to be hoped that the present results will overcome inertia of thinking with respect to the TIPS and alert treating clinicians to refer the appropriate subgroup of patients for whom the longterm benefits of surgical shunts are clear to the appropriate specialist surgeons.
Conflicts of interest
None declared.
References
- 1.Rosemurgy AS, Goode SE, Zwiebel BR, Black TJ, Brady PG. A prospective trial of transjugular intrahepatic portasystemic stent shunts versus small-diameter prosthetic H-graft portacaval shunts in the treatment of bleeding varices. Ann Surg. 1996;224:378–384. doi: 10.1097/00000658-199609000-00014. discussion 384–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Orloff MJ, Bell RH, Jr, Orloff MS, Hardison WG, Greenburg AG. Prospective randomized trial of emergency portacaval shunt and emergency medical therapy in unselected cirrhotic patients with bleeding varices. Hepatology. 1994;20(4 Pt 1):863–872. doi: 10.1002/hep.1840200414. [DOI] [PubMed] [Google Scholar]
- 3.Rikkers LF, Jin G, Burnett DA, Buchi KN, Cormier RA. Shunt surgery versus endoscopic sclerotherapy for variceal haemorrhage: late results of a randomized trial. Am J Surg. 1993;165:27–32. doi: 10.1016/s0002-9610(05)80400-3. discussion 33. [DOI] [PubMed] [Google Scholar]
- 4.Boyer TD, Haskal ZJ. The role of transjugular intrahepatic portosystemic shunt (TIPS) in the management of portal hypertension: update 2009. Hepatology. 2010;51:306. doi: 10.1002/hep.23383. [DOI] [PubMed] [Google Scholar]
- 5.Tripathi D, Helmy A, Macbeth K, Balata S, Lui HF, Stanley AJ, et al. Ten years' follow-up of 472 patients following transjugular intrahepatic portosystemic stent-shunt insertion at a single centre. Eur J Gastroenterol Hepatol. 2004;16:9–18. doi: 10.1097/00042737-200401000-00003. [DOI] [PubMed] [Google Scholar]
- 6.Stanley AJ, Jalan R, Forrest EH, Redhead DN, Hayes PC. Longterm follow-up of transjugular intrahepatic portosystemic stent shunt (TIPSS) for the treatment of portal hypertension: results in 130 patients. Gut. 1996;39:479–485. doi: 10.1136/gut.39.3.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhuang ZW, Teng GJ, Jeffery RF, Gemery JM, Janne d'Othee B, Bettmann MA. Longterm results and quality of life in patients treated with transjugular intrahepatic portosystemic shunts. AJR Am J Roentgenol. 2002;179:1597–1603. doi: 10.2214/ajr.179.6.1791597. [DOI] [PubMed] [Google Scholar]
- 8.Rossle M, Siegerstetter V, Euringer W, Olschewski M, Kromeier J, Kurz K, et al. The use of a polytetrafluoroethylene-covered stent graft for transjugular intrahepatic portosystemic shunt (TIPS): longterm follow-up of 100 patients. Acta Radiol. 2006;47:660–666. doi: 10.1080/02841850600806324. [DOI] [PubMed] [Google Scholar]
- 9.Orloff MJ, Isenberg JI, Wheeler HO, Haynes KS, Jinich-Brook H, Rapier R, et al. Liver transplantation in a randomized controlled trial of emergency treatment of acutely bleeding oesophageal varices in cirrhosis. Transplant Proc. 2010;42:4101–4108. doi: 10.1016/j.transproceed.2010.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Accreditation Council for Graduate Medical Education. 2000. Surgery. Residency Review Committee. General Surgery: National Resident Report online. Available at http://www.acgme.org/acgmeweb/tabid/274/DataCollectionSystems/ResidentCaseLogSystem/CaseLogsStatisticalReports.aspx (last accessed 24 May 2013) 2012.
- 11.Cameron JL, Herlong HF, Sanfey H, Boitnott J, Kaufman SL, Gott VL, et al. The Budd–Chiari syndrome. Treatment by mesenteric-systemic venous shunts. Ann Surg. 1983;198:335–346. doi: 10.1097/00000658-198309000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Orloff MJ, Daily PO, Orloff SL, Girard B, Orloff MS. A 27-year experience with surgical treatment of Budd–Chiari syndrome. Ann Surg. 2000;232:340–352. doi: 10.1097/00000658-200009000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Orloff MJ, Isenberg JI, Wheeler HO, Daily PO, Girard B. Budd–Chiari syndrome revisited: 38 years' experience with surgical portal decompression. J Gastrointest Surg. 16:286–300. doi: 10.1007/s11605-011-1738-9. [DOI] [PubMed] [Google Scholar]
- 14.Molmenti EP, Segev DL, Arepally A, Hong J, Thuluvath PJ, Rai R, et al. The utility of TIPS in the management of Budd–Chiari syndrome. Ann Surg. 2005;241:978–981. doi: 10.1097/01.sla.0000164180.77824.12. discussion 982–983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Plessier A, Sibert A, Consigny Y, Hakime A, Zappa M, Denninger MH, et al. Aiming at minimal invasiveness as a therapeutic strategy for Budd–Chiari syndrome. Hepatology. 2006;44:1308–1316. doi: 10.1002/hep.21354. [DOI] [PubMed] [Google Scholar]
- 16.Janssen HL, Garcia-Pagan JC, Elias E, Mentha G, Hadengue A, Valla DC. Budd–Chiari syndrome: a review by an expert panel. J Hepatol. 2003;38:364–371. doi: 10.1016/s0168-8278(02)00434-8. [DOI] [PubMed] [Google Scholar]
- 17.Warren WD, Henderson JM, Millikan WJ, Galambos JT, Brooks WS, Riepe SP, et al. Distal splenorenal shunt versus endoscopic sclerotherapy for longterm management of variceal bleeding. Preliminary report of a prospective, randomized trial. Ann Surg. 1986;203:454–462. doi: 10.1097/00000658-198605000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rikkers LF, Jin G, Langnas AN, Shaw BW., Jr Shunt surgery during the era of liver transplantation. Ann Surg. 1997;226:51–57. doi: 10.1097/00000658-199707000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Helton WS, Maves R, Wicks K, Johansen K. Transjugular intrahepatic portasystemic shunt vs. surgical shunt in good-risk cirrhotic patients: a case–control comparison. Arch Surg. 2001;136:17–20. doi: 10.1001/archsurg.136.1.17. [DOI] [PubMed] [Google Scholar]
- 20.Henderson JM, Boyer TD, Kutner MH, Galloway JR, Rikkers LF, Jeffers LJ, et al. Distal splenorenal shunt versus transjugular intrahepatic portal systematic shunt for variceal bleeding: a randomized trial. Gastroenterology. 2006;130:1643–1651. doi: 10.1053/j.gastro.2006.02.008. [DOI] [PubMed] [Google Scholar]
- 21.Orloff MJ, Vaida F, Haynes KS, Hye RJ, Isenberg JI, Jinich-Brook H. Randomized controlled trial of emergency transjugular intrahepatic portosystemic shunt versus emergency portacaval shunt treatment of acute bleeding oesophageal varices in cirrhosis. J Gastrointest Surg. 2012;16:2094–2111. doi: 10.1007/s11605-012-2003-6. [DOI] [PubMed] [Google Scholar]
- 22.Knechtle SJ, D'Alessandro AM, Armbrust MJ, Musat A, Kalayoglu M. Surgical portosystemic shunts for treatment of portal hypertensive bleeding: outcome and effect on liver function. Surgery. 1999;126:708–711. discussion 711–713. [PubMed] [Google Scholar]
- 23.Khaitiyar JS, Luthra SK, Prasad N, Ratnakar N, Daruwala DK. Transjugular intrahepatic portosystemic shunt versus distal splenorenal shunt – a comparative study. Hepatogastroenterology. 2000;47:492–497. [PubMed] [Google Scholar]
- 24.Rosemurgy AS, Bloomston M, Clark WC, Thometz DP, Zervos EE. H-graft portacaval shunts versus TIPS: ten-year follow-up of a randomized trial with comparison to predicted survivals. Ann Surg. 2005;241:238–246. doi: 10.1097/01.sla.0000151884.67600.b6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sanyal AJ, Freedman AM, Shiffman ML, Purdum PP, Luketic 3rd VA, Cheatham AK. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology. 1994;20(1 Pt 1):46–55. doi: 10.1016/0270-9139(94)90133-3. [DOI] [PubMed] [Google Scholar]
- 26.Orloff MJ, Isenberg JI, Wheeler HO, Haynes KS, Jinich-Brook H, Rapier R, et al. Randomized trial of emergency endoscopic sclerotherapy versus emergency portacaval shunt for acutely bleeding oesophageal varices in cirrhosis. J Am Coll Surg. 2009;209:25–40. doi: 10.1016/j.jamcollsurg.2009.02.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Orloff MJ, Isenberg JI, Wheeler HO, Haynes KS, Jinich-Brook H, Rapier R, et al. Alcoholic versus non-alcoholic cirrhosis in a randomized controlled trial of emergency therapy of bleeding varices. J Surg Res. 2012;174:98–105. doi: 10.1016/j.jss.2010.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rosemurgy AS, Frohman HA, Teta AF, Luberice K, Ross SB. Prosthetic H-graft portacaval shunts vs. transjugular intrahepatic portasystemic stent shunts: 18-year follow-up of a randomized trial. J Am Coll Surg. 2012;214:445–453. doi: 10.1016/j.jamcollsurg.2011.12.042. discussion 453–455. [DOI] [PubMed] [Google Scholar]
- 29.Orloff MJ, Orloff MS, Orloff SL, Rambotti M, Girard B. Three decades of experience with emergency portacaval shunt for acutely bleeding oesophageal varices in 400 unselected patients with cirrhosis of the liver. J Am Coll Surg. 1995;180:257–272. [PubMed] [Google Scholar]
- 30.Clark W, Hernandez J, McKeon B, Villadolid D, Al-Saadi S, Mullinax J, et al. Surgical shunting versus transjugular intrahepatic portasystemic shunting for bleeding varices resulting from portal hypertension and cirrhosis: a meta-analysis. Am Surg. 2010;76:857–864. [PubMed] [Google Scholar]


