Abstract
Lead poisoning has been known to be associated with structural and functional abnormalities of multiple organ systems of human body. The aim of this investigation was to study the renal protective effects of ginger (Zingiber officinale) extract in lead induced toxicity rats. In this study renal glutathione (GSH) level, glutathione peroxidase (GPX), glutathione-s-transferase (GST), and catalase enzymes were measured in lead nitrate (300 mg/kg BW), and lead nitrate plus ginger extract (150 mg/kg BW) treated rat groups for 1 week and 3 weeks respectively. The glutathione level and GSH dependent antioxidant enzymes such as glutathione peroxidase, glutathione-s-transferase, and catalase significantly (P < 0.05) increased in ginger extract treated rat groups. In addition, histological studies showed lesser renal changes in lead plus ginger extract treated rat groups than that of lead alone treated rat groups. These results indicate that ginger extract alleviated lead toxic effects by enhancing the levels of glutathione, glutathione peroxidase, glutathione-s-transferase and catalase.
Keywords: Lead toxicity, Ginger (Zingiber officinale) extract, Rat kidney, Glutathione (GSH), Glutathione dependent antioxidant enzymes
Introduction
Rapid industrial growth has increased the lead toxicity substantially over the past four decades. Worldwide a large number of people are exposed to lead every year, especially those who are poor and live in developing countries. Lead is used in pottery, window making, shipbuilding, the arms industry, cosmetics, paint pigments, book printing, internal and topical medicinal preparations since recorded antiquity (Hernberg 2000; Meyer et al. 2008). This heavy use of lead in industries has caused the contamination of air, dust, and soil. Lead toxicity is associated with a variety of dysfunctions in experimental animals and humans (Hsu and Leon 2002) including haemopoietic system (De Silva 1981), liver (Sharama and Street 1980), kidneys (Humphreys 1991; Hernberg 2000), male and female reproductive systems (Lancranjan et al. 1975; Rom 1980). Several review reports on lead toxicity and its possible mechanisms of action are available in literature (Gurer and Ercal 2000; Hernberg 2000; Hsu and Leon 2002; Meyer et al. 2008). Although rising evidence of adverse health effects related to lead, it is still widely used in consumer products and released into the air through combustion of coals and oil, waste incineration, and fugitive emissions during mining and smelting (Meyer et al. 2008). Lead exposure causes oxidative stress by generation of Reactive Oxygen Species (ROS), reducing the antioxidant defense system of cells through depleting glutathione, inhibiting sulfhydryl-dependent enzymes, or increasing susceptibility of cells to oxidative attack by altering the membrane integrity (Gurer and Ercal 2000).
Ginger (Zingiber officinale) is commonly used as food spice in India and other Asian and African countries. In many traditional Chinese, Ayurvedic and Unani herbal medicines ginger has been recommended for the treatment of catarrh, rheumatism, nervous diseases, gingivitis, toothache, asthma, stroke, constipation and diabetes for centuries (Awang 1992; Wang and Wang 2005; Tapsell et al. 2006). Several reviews on medicinal values of ginger have been intensively reported in the literature (Ali et al. 2008; Grzanna et al. 2005; Ernst and Pittler 2000). Several recent studies reported the protective effects of ginger extracts against alcohol induced toxicity (Ali and Fahmy 2009), bromobenzene induced hepatotoxicity (El-sharaky et al. 2009), fenitrothion or lead induced developmental toxicity (Farag et al. 2010), fungicide induced liver toxicity (Sakr 2007), ethionine-induced toxicity (Habib et al. 2008), acetic acid-induced ulcerative colitis (El-abhar et al. 2008). Recent studies have also been reported the curing effects of ginger on diseases such as diabetes, cancer and heart attack and it was also found that ginger therapy is more effective in detoxification of cadmium and mercury toxicity (Egwurugwu et al. 2007; Vitalis et al. 2007).
Extensive research on lead reveals its toxic effects on animals. But sufficient work on detoxification could not be found in the literature. Hence, an attempt was made in this investigation to study the protective effects of ginger extract as a natural product on renal toxicity induced by lead in male rats. The aim of this study was to investigate potential role of ginger extract on oxidative stress-mediated renal toxicity on kidneys of male rats.
Materials and methods
Materials
The ginger was obtained from a local market, Anantapur, India. The ginger was cut into small pieces and air-dried for 5 to 6 days. The dried ginger was ground to fine powder using electronic grinder. Ginger powder (50 g) was extracted in 250 ml of ethanol for 18 h in a soxhlet apparatus following the method of Misra et al. (2005). The extract was dried at reduced pressure and stored at 0–4 °C until further experiments. The chemicals and solvents used in this study were of analytical grade.
Animals and treatment
Male Wistar rats of body weight weighing 160 ± 10 g were procured from Sri Ragavendra Enterprises, Bangalore, India. The rats were maintained at 24 ± 2 °C and 12 h light/dark cycles. Rats were fed on standard rat diet and water was supplied ad libitum. Before experimentation rats were acclimatized for 2 weeks. Animals were handled according to the rules and regulations of Institutional Animal Ethics committee (IAEC), Sri Krishnadevaraya University, Anantapur, India. Animals were divided into five groups of six animals each. Group I treated with vehicle (distilled water) was kept as control. Group II treated with lead nitrate (300 mg/kg BW) daily for 1 week, group III was treated with lead nitrate (300 mg/kg BW) plus ginger extract (150 mg/kg BW) daily for 1 week. Group IV treated with lead nitrate (300 mg/kg BW) daily for 3 weeks, Group V was treated with lead nitrate (300 mg/kg BW) plus ginger extract (150 mg/kg BW) daily for 3 weeks. Lead nitrate was dissolved in distilled water and given through gastro intestinally with the help of a gavage. The ethanolic extract of ginger was dissolved in 5% tween-80 and administered through gastro intestinally with the help of a gavage after 6 h of lead nitrate incubation.
Animal sacrifice and organ collection
After one week of experimental period, three rats from group I, all six rats from group II and group III were sacrificed. From group I remaining three rats and from group IV and V all six rats were sacrificed after 3 weeks of exposure. Animals were sacrificed by cervical dislocation and immediately kidneys were removed and washed with ice-cold saline to remove blood and suspended in 0.1 M potassium chloride (KCl) in polypropylene container, sealed with parafilm, labeled and frozen at −80 °C until assays were carried out.
Preparation of tissue extraction for glutathione estimation
Ten percent tissue homogenate was prepared in 0.15 M KCl using a homogenizer at 0°C. The whole homogenate was used for estimation of glutathione.
Sample preparation for antioxidant enzyme assays
Ten percent kidney homogenate in 0.15 M KCl was prepared using a homogenizer at 0°C and centrifuged in cold (0–4°C) at 12000 rpm for 45 min. The supernatant thus obtained was distributed into eppendorf tubes, labeled and stored at −20 °C and all the antioxidant enzymes were assayed at the earliest.
Glutathione (GSH)
Total reduced glutathione content was measured by following the method of Ellman (1959). This method is based on the development of a yellow color, when 5, 5′-dithio-2-nitrobenzoic acid (DTNB) reacts with the compounds containing sulfhydryl groups with a maximum absorbance at 412 nm. Ten percent kidney homogenate (0.5 ml) was deproteinized with 3.5 ml of 5% Trichloroacetic acid (TCA) and centrifuged at 4000 rpm for 5 min. To 0.5 ml of supernatant, 3.0 ml 0.2 M phosphate buffer (pH 8.0) and 0.5 ml of Ellman’s reagent were added and the yellow color developed was measured at 412 nm. A series of standards (4–20 μg) were treated in a similar manner along with a blank and values were expressed as μg of GSH/mg protein.
Glutathione peroxidase (GPX)
A known amount of the enzyme preparation was allowed to react with H2O2 in the presence of GSH for specific time period according to the method of Rostruck et al. (1973) and remaining GSH was measured by following the method of Ellman (1959) as described for GSH estimation. To 0.5 ml 0.4 M phosphate buffer (pH 7.0), 0.2 ml of 10% kidney homogenate, 0.2 ml of GSH and 0.1 ml of H2O2 were added and incubated at room temperature (25 ± 2 °C) for 10 min along with a control tube containing all reagents except enzyme source. The reaction was arrested by adding 0.5 ml of TCA, centrifuged at 4000 rpm for 5 min and GSH content in 0.5 ml of supernatant was estimated. The activity was expressed as μg of GSH consumed/min/mg protein.
Glutathione-s-transferase (GST)
Glutathione-s-transferase activity was measured by monitoring the increase in the absorbance at 340 nm using 1-chloro-2,4-dinitrobenzene (CDNB) as a substrate according to the method of Habig et al. (1974). To 1.7 ml phosphate buffer (pH 6.5), 0.2 ml of GSH and 0.04 ml of kidney homogenate (40 μg protein) were added and the reaction was initiated by the addition of 0.06 ml CDNB. The change in absorbance was recorded at 1 min intervals at 340 nm for 5 min and the activity was calculated using extinction coefficient of CDNB-GSH conjugate as 9.6 mM -1 cm -1 and expressed as m moles of CDNB-GSH conjugate formed/min/mg protein.
Catalase (CAT)
Catalase catalyses the breakdown of H2O2 to H2O and O2 and the rate of decomposition of H2O2 was measured spectrophotometrically at 240 nm following the method of Beers and Sizer (1952). To 1.9 ml of phosphate buffer (pH 7.0), 1.0 ml of H2O2.was added and then the reaction was initiated by the addition of 0.1 ml kidney homogenate (45 μg protein). The decrease in absorbance was monitored at 1 min intervals for 5 min at 240 nm and activity was calculated using a molar absorbance coefficient of H2O2 as 43.6 M-1 cm-1. The activity was expressed as m moles of H2O2 decomposed/min/mg protein.
Histology
The histology sections of the kidneys of male rats were taken by adopting the procedure as described by Humason (1972). The tissues were isolated and gently rinsed with saline solution to remove mucus and other debris adhering to them. They were fixed in Bouin’s fluid for 24 h and the fixative was removed by washing through running tap water for overnight. Then the tissues were processed for dehydration. The tissues were passed through successive series containing 30%, 50%, 70%, 80%, 90%, 95% and absolute alcohols. Then the tissues were cleaned in methyl benzoate and embedded in paraffin wax. Sections of 5 μ thickness were made using rotary microtome. The sections were stained with Harris hematoxylin (Harris 1900) and counter stained with eosin, dissolved in 95% alcohol. After dehydration and cleaning, the sections were mounted in Canada balsam. Photomicrographs of the section preparations were taken using Olympus photomicrography equipment (PM-6 model, Japan).
Statistical analysis
Statistical analysis was carried out using SPSS 11.0 for Windows (SPSS Inc., Chicago, IL). Each experiment repeated in triplicates and all data were represented as mean ± SD. Duncan’s multiple range test was used to determine the statistical significance between groups. The significance is presented at level of P < 0.05.
Results and discussion
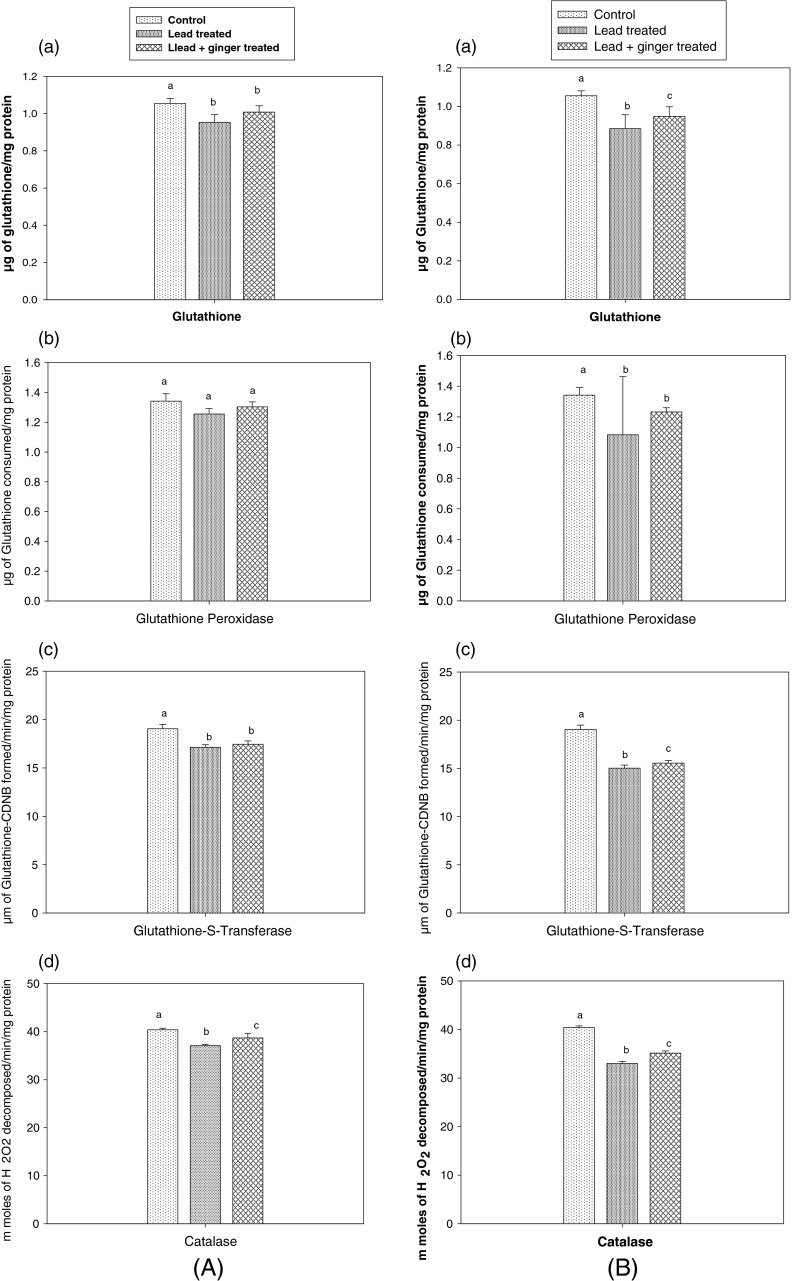
Figure 1 shows the renal glutathione (GSH) level and activities of glutathione peroxidase (GPX), glutathione-s-transferase (GST) and catalase (CAT) in control group, lead treated group and lead plus ginger extract treated group for one week and three weeks respectively. The results of this study has indicated that lead at a dose of 300 mg/kg BW had significantly (P < 0.05) decreased the renal glutathione, glutathione peroxidase, glutathione-s-transferase and catalase when compared to control group, whereas ginger extract at a dose of 150 mg/kg BW exposure has increased the levels of GSH, GPX, GST and CAT. This has demonstrated the protective role of ginger extract on renal toxicity caused by lead treatment. Lead is a non essential toxic heavy metal widely distributed in the environment and a chronic exposure to low levels of lead induces a broad range of physiological, biochemical and behavioural dysfunctions (Hsu and Leon 2002; Needleman 2004). Various mechanisms have been reported to explain lead toxicity, which include disturbances in the mineral metabolism, demyelization of nerve tissues and inactivation of several enzymes (Ercal et al. 2001). Recent studies have proposed that one possible mechanism of lead toxicity is the disturbance of prooxidant and antioxidant balance by generation of reactive oxygen species (ROS) (Gurer and Ercal 2000; Wang et al. 2001). Adonaylo and Oteiza (1999) reported that lead exposure has a dose response relationship with changes in antioxidant enzyme levels and their activities.
Fig. 1.
Comparison of (a) Glutathione level, (b) Glutathione peroxidase, (c) Glutathione-s-transferase, (d) Catalase enzymes in control group for (A) 1 week lead treated group and 1 week lead plus ginger treated group and (B) 3 weeks lead treated group and 3 weeks lead plus ginger treated group (n = 3). Different letters on the bars represent significantly different at P < 0.05. Same letters on the bars are not significantly different. The results are presented as mean ± SD of three determinations
Glutathione
Glutathione is an essential biofactor synthesized in all living cells. It forms an important substrate for GPX, GST and several other enzymes, which are involved in free radical scavenging. It can act as a nonenzymatic antioxidant by direct interaction of the SH groups with ROS or it can be involved in the enzymatic detoxificatin reactions for ROS as a cofactor (Ding et al. 2000). In the present study GSH levels were decreased in lead treated rat group when compared to control group. The GSH levels of control group was 1.05 μg of GSH/mg protein, and in 1 week lead exposed group was 0.95 μg of GSH/mg protein, and in 3 weeks lead exposed group was 0.88 μg of GSH/mg protein. Several studies reported that lead toxicity decreases GSH levels (Bechara 2004; Patra et al. 2001; Sivaprasad et al. 2003). In 3 weeks lead plus ginger treated group the GSH level was increased (0.94 μg of GSH/mg protein). As the GSH is substrate for GPX, GST and several other enzymes, it is believed that GSH level is also increased when these enzymes activity is increased. Kikuzaki and Nakatani (1993) and Lee and Ahn (1985) have also reported that GSH depended antioxidant activity is increased by ginger extracts.
Glutathione peroxidase (GPX)
Glutathione peroxidase (GPX) is an antioxidant enzyme that catalyzes the hydrogen peroxide-reduced glutathione reaction. GPX is an important component in the enzymatic defense system against the increase of free radicals (Crack et al. 2001). In this study, the activity of glutathione peroxidase was reduced in lead treated groups (1.25 and 1.08 μg of GSH consumed/min/mg protein, respectively for 1 week and 3 weeks lead treated groups) when compared to control group (1.34 μg of GSH consumed/min/mg protein). Farmand et al. (2005) reported that the decrease in GPX activity was due to lead toxicity. Lead plus ginger extract treated groups showed a significant increase of kidney GPX activity. The results obtained in present study further support that ginger can promote glutathione peroxidase as one of the antioxidant activity (Kikuzaki and Nakatani 1993; Lee and Ahn 1985).
Glutathione-s-transferase (GST)
Glutathione-s-transferase (GST) is an important enzymatic system of the cellular mechanism of detoxification that protects cells against reactive oxygen metabolites due to the conjugation of glutathione with electrophillic compounds. In this investigation the activity of renal GST was found to be 19.04, 17.13 and 15.02 μ moles of GSH-CDNB formed/min/mg protein, respectively for control, 1 week and 3 weeks lead treated groups. Significant improvement in the activity of GST was noticed in ginger supplemented groups. These findings further support the studies that reported the enhancement of GST activity by ginger extracts (Misra et al. 2005; Kikuzaki and Nakatani 1993).
Catalase (CAT)
Catalase (CAT) is a metalloprotein that protects the tissues from highly reactive hydroxyl radicals by reducing hydrogen peroxide. In this study the activity of catalase was found to be significantly (P < 0.05) increased in kidneys of lead plus ginger extract treated groups when compared to lead treated groups. These findings coincide with earlier studies that showed a significant increase in CAT activity due to ginger treatment (Misra et al. 2005; Kikuzaki and Nakatani 1993). The CAT activity was significantly (P < 0.05) decreased in lead treated groups when compared to control group. The decrease in CAT activity might be due to the inhibition of haembiosynthesis of catalase by lead (Patil et al. 2006). Mohammad et al. (2008) have also reported that decrease in CAT activity was due to lead toxicity.
Histopathology
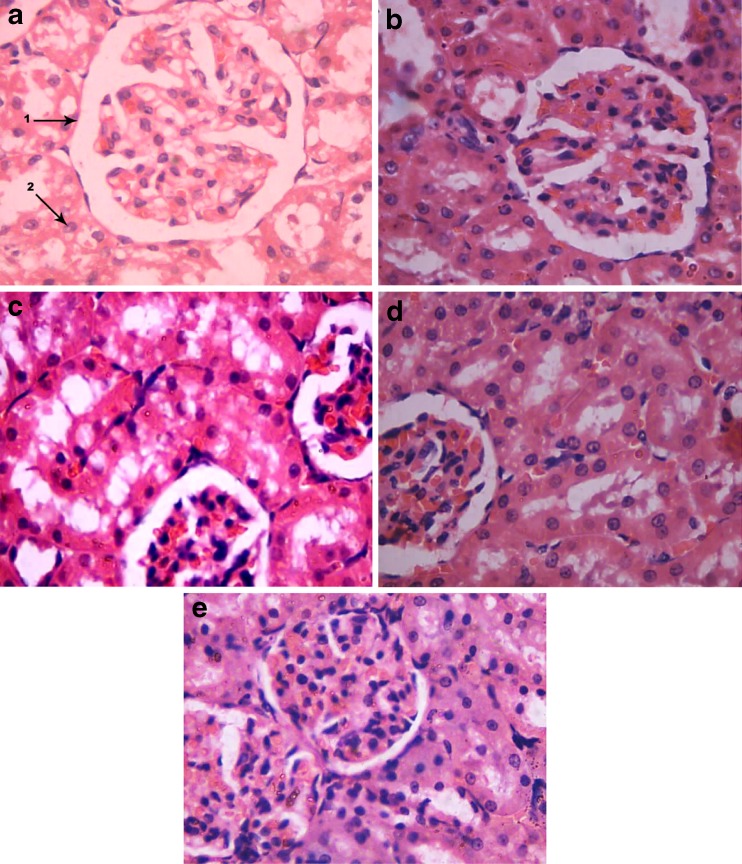
The results of histopathological examination of rat kidney tissue sections are presented in the Fig. 2. Kidney sections of 1 week lead exposed rat group showed architectural changes of glomerulus and tubular epithelial cells (Fig. 2b). The changes observed were dispersed in intratubular region. Whereas in kidney of lead exposed plus ginger extract treated rat group mild changes were observed when compared to the kidney of 1 week lead exposed rat group (Fig. 2c). Necrotic regions were observed and it seems to be fatty changes in between glomeruli and intratubular region. The kidney sections of 3 weeks lead exposed rat group showed severe changes than that of 1 week lead exposed group (Fig. 2d). Infiltration of inflammatory cells was seen in intratubular region. Tubular epithelial cells appear to be degenerative with mild necrotic changes. Whereas in 3 weeks lead plus ginger extract treated rat group kidney indicated lesser changes than 3 weeks lead exposed rat group (Fig. 2e).
Fig. 2.
Comparison of photomicrographs of kidney tissue sections of a Control group, bLead treated group for 1 week, c Lead plus ginger extract treated group for 1 week, d Lead treated group for 3 weeks, e Lead plus ginger extract treated group for 3 weeks
The histopathological results obtained in the present investigation indicated that lead induced many histopathological changes in the kidney of rats treated with lead for 1 week and 3 weeks. Lead potentially induces oxidative stress and causes pathological changes of tissues like kidney (Ercal et al. 2000; Farmand et al. 2005; Khalil-Manesh et al. 1992). Physiological studies of lead transport in the kidney have shown this metal is taken up in proximal tubule cells (Vander et al. 1977; Vander et al. 1979). Exposure to high lead levels can produce renal tubular damage with glycosuria and aminociduria (Loghman-Adham 1997; Ehrlich et al. 1998). The results obtained in the present study showed that treating rats with lead plus ginger extract improved the histopathological changes induced in the kidney by lead. This suggested the effectiveness of ginger extract in prevention of lead induced renal toxicity.
Conclusions
In this study, Ginger extract showed the protective effects on lead induced renal toxicity by enhancing the levels of glutathione, glutathione peroxidase, glutathione-s-transferase and catalase.
References
- Adonaylo VN, Oteiza PI. Lead intoxication: antioxidant defenses and oxidative damage in rat brain. Toxicol. 1999;13:77–85. doi: 10.1016/S0300-483X(99)00051-7. [DOI] [PubMed] [Google Scholar]
- Ali AS, Fahmy GE. Effects of water extracts of thyme (Thymus vulgaris) and ginger (Zingiber officinale Roscoe) on alcohol abuse. Food Chem Toxicol. 2009;47:1945–1949. doi: 10.1016/j.fct.2009.05.007. [DOI] [PubMed] [Google Scholar]
- Ali BH, Blunden G, Tanira MO, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe). A review of recent research. Food Chem Toxicol. 2008;46:409–420. doi: 10.1016/j.fct.2007.09.085. [DOI] [PubMed] [Google Scholar]
- Awang DVC. Ginger. Canadian Pharmocol J. 1992;125:309–311. [Google Scholar]
- Bechara EJH. Lead poisoning and oxidative stress. Free Rad Biol Med. 2004;36(suppl):S22. [Google Scholar]
- Beers RF, Sizer IW. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem. 1952;195:133–140. [PubMed] [Google Scholar]
- Crack PJ, Taylor JM, Flentjar NJ, De Haan J, Hertzog P, Iannello RC, Kola I. Increased infarct size and exacerbated apoptosis in the glutathione peroxidase-1 (GPX-1) knock out mouse brain in response to ischemia/reperfusion injury. Pharmacol Rev. 2001;53:135–159. doi: 10.1046/j.1471-4159.2001.00535.x. [DOI] [PubMed] [Google Scholar]
- De Silva PE. Determination of lead in plasma and studies on its relationship to lead in erythrocytes. Brit J Indust Med. 1981;38:209–217. doi: 10.1136/oem.38.3.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding Y, Gonick HC, Vaziri ND. Lead promotes hydroxyl radical generation and lipid peroxidation in cultured aortic endothelial cells. Americ J Hyperten. 2000;13:552–5. doi: 10.1016/S0895-7061(99)00226-5. [DOI] [PubMed] [Google Scholar]
- Egwurugwu JN, Ufearo CS, Abanobi OC, Nwokocha CR, Duruibe JO, Adeleye GS, Ebunlomo AO, Adetola AO, Onwufuji O. Effects of ginger (Zingiber officinale) on cadmium toxicity. African J Biotech. 2007;6:2078–2082. [Google Scholar]
- Ehrlich R, Robins T, Jordaan E, Miller S, Mbuli S, Selby P, Wynchank S, Cantrell A, De Broe M, D’Haese P, Todd A, Landrigan P. Lead absorption and renal dysfunction in a South African battery factory. Occupat Environ Med. 1998;55:453–460. doi: 10.1136/oem.55.7.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-abhar HS, Hammad LN, Abdel HS. Modulating effect of ginger extract on rats with ulcerative colitis. J Ethnopharmacol. 2008;118:367–372. doi: 10.1016/j.jep.2008.04.026. [DOI] [PubMed] [Google Scholar]
- Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophy. 1959;82:70–77. doi: 10.1016/0003-9861(59)90090-6. [DOI] [PubMed] [Google Scholar]
- El-Sharaky AS, Newairy AA, Kamel MA, Eweda SM. Protective effect of ginger extract against bromobenzene-induced hepatotoxicity in male rats. Food Chem Toxicol. 2009;47:1584–1590. doi: 10.1016/j.fct.2009.04.005. [DOI] [PubMed] [Google Scholar]
- Ercal N, Neal R, Treeratphan P, Luttz PM, Hammond TC, Dennery PA, Spitz DR. A role for oxidative stress in suppressing serum immunoglobulin levels in lead exposed fisher 344 rats. Arch Environ Contam Toxicol. 2000;39:251–6. doi: 10.1007/s002440010102. [DOI] [PubMed] [Google Scholar]
- Ercal N, Gurer-Orhan H, Aykin-Burns N. Toxic metals and oxidative stress part I: mechanisms involved in metal induced oxidative damage. Curr Top Med Chem. 2001;1:529–539. doi: 10.2174/1568026013394831. [DOI] [PubMed] [Google Scholar]
- Ernst E, Pittler MH. Efficacy of ginger for nausea and vomiting: a systematic review of randomized clinical trials. Brit J Anaesth. 2000;84:367–71. doi: 10.1093/oxfordjournals.bja.a013442. [DOI] [PubMed] [Google Scholar]
- Farag AGA, Elhalwagy MEA, Farid HEA. Effect of ginger supplementation on developmental toxicity induced by fenitrothion insecticide and/or lead in albino rats. Pesticide Biochem Physiol. 2010;97:267–274. doi: 10.1016/j.pestbp.2010.03.007. [DOI] [Google Scholar]
- Farmand F, Ehdaie A, Roberts C, Sindhu RK. Lead induced dysregulation of superoxide dismutase, catalase, glutathione peroxidase and guanylate cyclase. Environ Res. 2005;98:33–39. doi: 10.1016/j.envres.2004.05.016. [DOI] [PubMed] [Google Scholar]
- Grzanna R, Lindmark L, Frondoza CG. Ginger– an herbal medicinal product with broad anti-inflammatory actions. J Med Food. 2005;8:125–132. doi: 10.1089/jmf.2005.8.125. [DOI] [PubMed] [Google Scholar]
- Gurer H, Ercal N. Can antioxidants be beneficial in the treatment of Lead poisoning? Free Rad Biol Med. 2000;29:927–945. doi: 10.1016/S0891-5849(00)00413-5. [DOI] [PubMed] [Google Scholar]
- Habib SHM, Makpol S, Hamid NAA, Das S, Ngah WZW, Yusof YAM. Ginger extract (zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics. 2008;63:807–813. doi: 10.1590/S1807-59322008000600017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habig WH, Pabst MJ, Jakoby WB. Glutathione–S-transferase. The first enzymatic step in mercapturic acid formation. J Biol Chem. 1974;249:7130–7139. [PubMed] [Google Scholar]
- Harris HF. On the rapid conversion of haemolymph into haematin in staining reaction. J Appl Microsc Lab Meth. 1900;3:777. [Google Scholar]
- Hernberg S. Lead poisoning in a historical perspective. Americ J Indust Med. 2000;254:244–254. doi: 10.1002/1097-0274(200009)38:3<244::AID-AJIM3>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- Hsu P, Leon Y. Antioxidant nutrients and lead toxicity. Toxicology. 2002;180:33–44. doi: 10.1016/S0300-483X(02)00380-3. [DOI] [PubMed] [Google Scholar]
- Humason GL. Animal tisssue techniques. III. Sanfransisco: W.H. Freeman and Co; 1972. [Google Scholar]
- Humphreys DJ. Effects of exposure to excessive quantities of lead on animals. Brit Vet J. 1991;147:18–30. doi: 10.1016/0007-1935(91)90063-S. [DOI] [PubMed] [Google Scholar]
- Khalil-Manesh F, Gonick HC, Cohen AH, Alinovi R, Bergamaschi E, Mutti A, Rosen VJ. Experimental model of lead nephropathy I. Continuous high dose lead administration. Kidney Inter. 1992;41:1192–1203. doi: 10.1038/ki.1992.181. [DOI] [PubMed] [Google Scholar]
- Kikuzaki H, Nakatani N. Antioxidant effects of some ginger constituents. J Food Sci. 1993;58:1407–1410. doi: 10.1111/j.1365-2621.1993.tb06194.x. [DOI] [Google Scholar]
- Lancranjan I, Popescu HI, Gavenescu O, Klepsch I, Serbanescu M. Reproductive ability of workmen occupationally exposed to lead. Arch Environ Health. 1975;30:396–401. doi: 10.1080/00039896.1975.10666733. [DOI] [PubMed] [Google Scholar]
- Lee IK, Ahn SY. The antioxidant activity of gingerol. Korean J Food Sci Technol. 1985;17:55–59. [Google Scholar]
- Loghman-Adham M. Renal effects of environmental and occupational lead exposure. Environ Health Pers. 1997;105:928–938. doi: 10.1289/ehp.97105928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer PA, Brown MJ, Falk H. Global approach to reducing lead exposure and poisoning. Mut Res. 2008;659:166–175. doi: 10.1016/j.mrrev.2008.03.003. [DOI] [PubMed] [Google Scholar]
- Misra DS, Rajkumar M, Saradindu B, Koushik D, Ghosh D. Protective effects of composite extract of Withania somnifera, Ocimum sanctum and Zingiber officinale on swimming in male rat. Iranian J Pharma Thera. 2005;4:110–117. [Google Scholar]
- Mohammad IK, Mahdi AA, Raviraja A, Najmul I, Iqbal A, Thuppil V. Oxidative stress in painters exposed to low lead levels. Arh Hig Rada Toksikol. 2008;59:161–169. doi: 10.2478/10004-1254-59-2008-1883. [DOI] [PubMed] [Google Scholar]
- Needleman H. Lead Poisoning. Annual Rev Med. 2004;55:209–22. doi: 10.1146/annurev.med.55.091902.103653. [DOI] [PubMed] [Google Scholar]
- Patil AJ, Bhagwat VR, Patil JA, Dongre NN, Ambekar JG, Jailkhani R, Das KK. Effect of lead exposure on the activity of superoxide dismutase and catalase in battery manufacturing workers (BMW) of western Maharashtra (India) with reference to heme biosynthesis. Intern J Environ Res Pub Health. 2006;3:329–337. doi: 10.3390/ijerph2006030041. [DOI] [PubMed] [Google Scholar]
- Patra RC, Swarup D, Dwidedi SK. Antioxidant effects of alpha-tocopherol, ascorbic acid and L-methionine on lead-induced oxidative stress of the liver, kidney and brain in rats. Toxicol. 2001;16:81–88. doi: 10.1016/S0300-483X(01)00345-6. [DOI] [PubMed] [Google Scholar]
- Rom WN (1980) Effects of lead on reproduction. In: Infante PF, Legator MS (eds), Proceedings of the Workshop on Methodology for Assessing Reproductive Hazards in the Workplace. National Institute for Occupational Safety and Health Washington DC pp. 33–42
- Rostruck JJ, Pope AL, Ganther HE, Swanson AB, Hofeman DG, Hoekstra WG. Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973;179:588–590. doi: 10.1126/science.179.4073.588. [DOI] [PubMed] [Google Scholar]
- Sakr SA. Ameliorative effect of ginger (Zingiber officinale) on mancozeb fungicide induced liver injury in albino rats. Liver. 2007;1:650–656. [Google Scholar]
- Sharama RP, Street JC. Public health aspects of toxic heavy metals in animal feeds. J Americ Vet Med Assoc. 1980;177:149–153. [PubMed] [Google Scholar]
- Sivaprasad R, Nagaraj M, Varalakshmi P. Combined efficacies of lipoic acid and meso-2,3-dimercapto succinic acid on lead induced erythrocyte membrane lipid peroxidation and antioxidant status in rats. Hum Experi Toxicol. 2003;22:183–192. doi: 10.1191/0960327103ht335oa. [DOI] [PubMed] [Google Scholar]
- Tapsell LC, Hemphill I, Cobiac L, Patch CS, Sullivan DR, Fenech M, Roodenrys S, Keogh JB, Clifton PM, Williams PG, Fazio VA, Inge KE. Health benefits of herbs and spices: the past, the present, the future. Med J Aust. 2006;185:S4–S24. doi: 10.5694/j.1326-5377.2006.tb00548.x. [DOI] [PubMed] [Google Scholar]
- Vander AJ, Taylor DL, Kalitis K, Mouw DR, Victery W. Renal handling of lead in dogs: clearence studies. Americ J Physiol. 1977;2:F532–F538. doi: 10.1152/ajprenal.1977.233.6.F532. [DOI] [PubMed] [Google Scholar]
- Vander AJ, Mouw DR, Cox J, Johnson B. Lead transport by renal slices and its inhibition by tin. Americ J Physiol. 1979;23:F373–F378. doi: 10.1152/ajprenal.1979.236.4.F373. [DOI] [PubMed] [Google Scholar]
- Vitalis EC, Chukwuemeka N, Philippe ME, Chinonso NC. Effects of zingiber officinale on liver function of mercuric chloride-induced hepatotoxicity in adult wistar rats. Electr J Biomed. 2007;3:40–45. [Google Scholar]
- Wang WH, Wang ZM. Studies of commonly used traditional medicine-ginger. Zhongguo Zhong Yao Za Zhi. 2005;30:1569–1573. [PubMed] [Google Scholar]
- Wang HP, Qiaa SY, Schafer FQ, Domann FE, Oberley LW, Buettner GR. Phospholipids hydroperoxide glutathione peroxidase protects against singlet oxygen-induced cell damage of photodynamic therapy. Free Rad Biol Med. 2001;3:825–835. doi: 10.1016/S0891-5849(01)00469-5. [DOI] [PubMed] [Google Scholar]